Grippenet: A New Tool for the Monitoring, Risk-Factor and Vaccination Coverage Analysis of Influenza-Like Illness in Switzerland
Abstract
1. Introduction
2. Methods
2.1. Participant Recruitment
2.2. Data Collection
2.3. Definitions
2.3.1. Influenza-Like Illness
- Sudden onset of symptoms;
- AND (fever or fatigue or muscular/articular pain);
- AND (coughing or sore throat or shortness of breath).
2.3.2. Active User
2.4. National Data Sources
2.5. Statistical Analyses
2.6. Data Safety and Ethical Approval
3. Results
3.1. Participants’ Socio-Demographic and Medical Characteristics
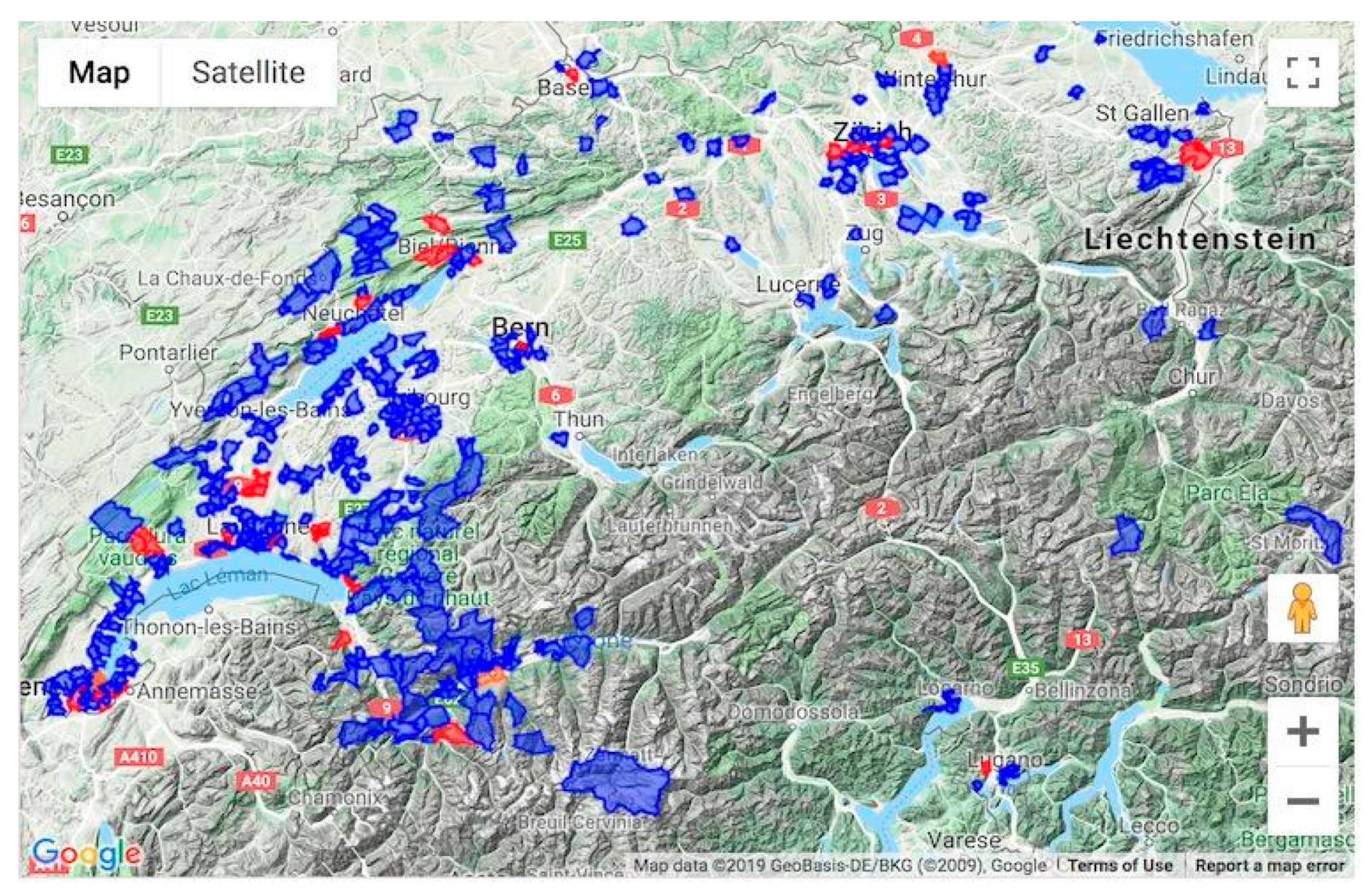
3.2. Participation
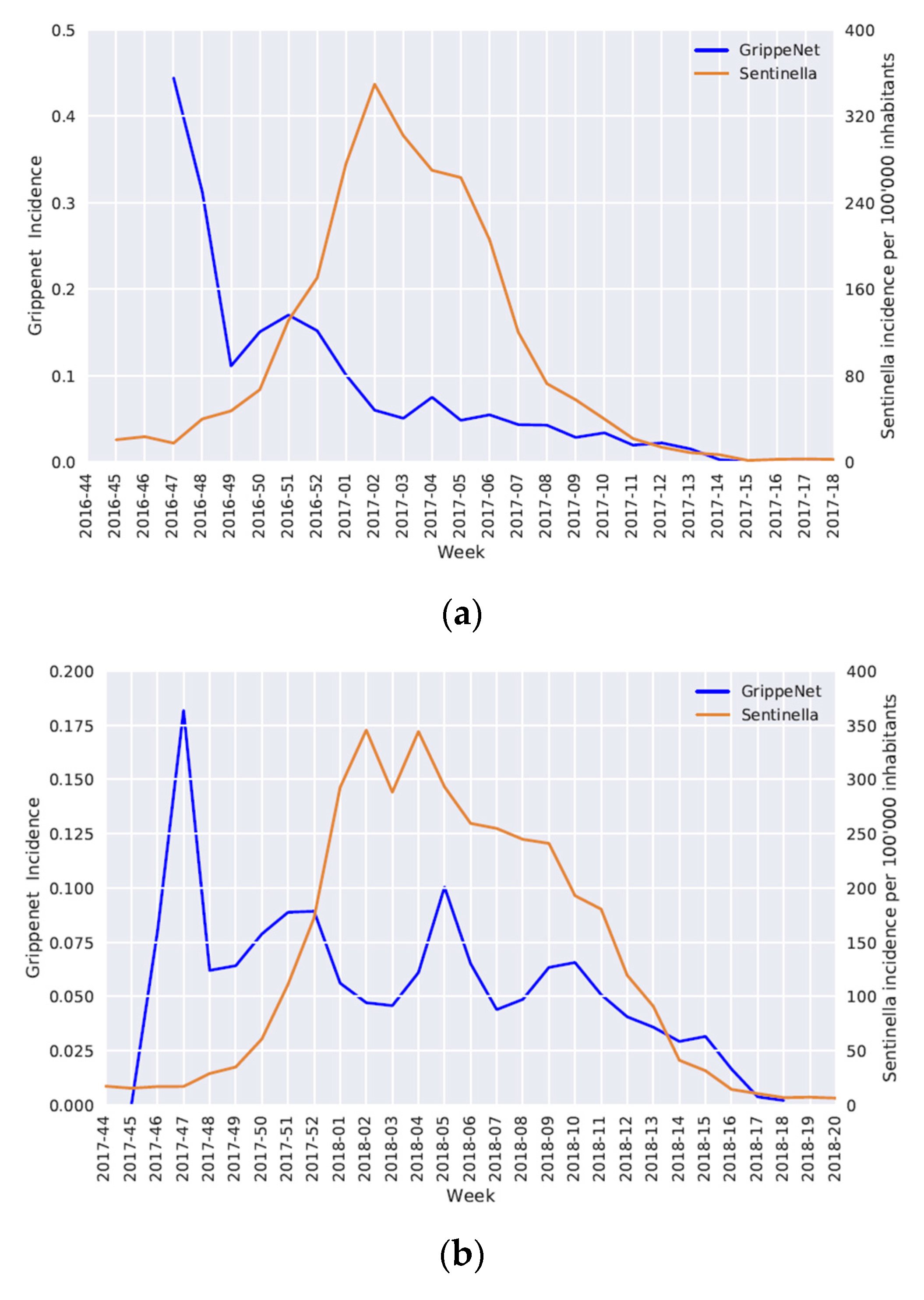
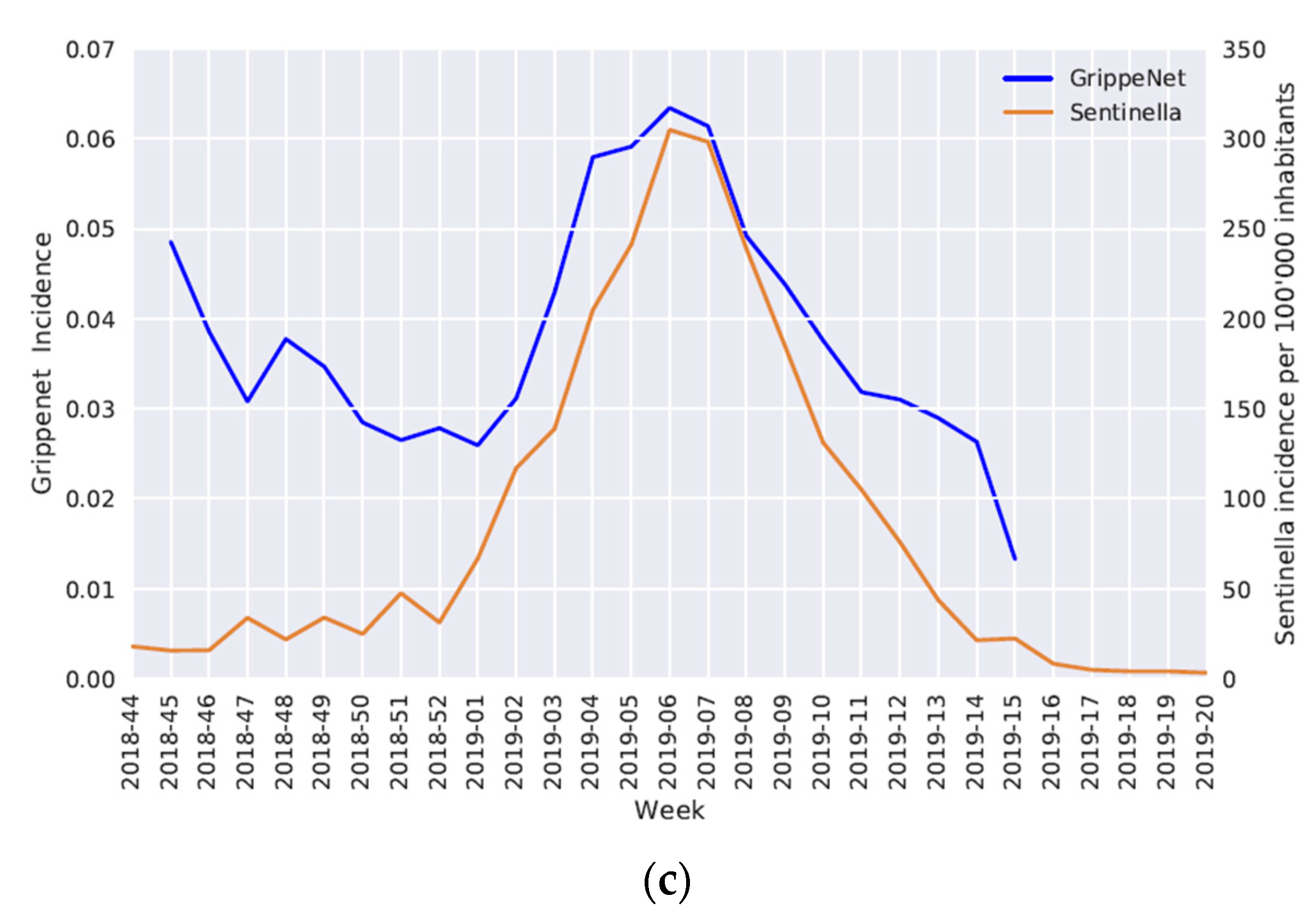
3.3. Epidemic Curves
3.4. Factors Associated with an Increased Risk of ILI
3.5. Vaccination Status
3.6. Medical Care Seeking Behavior
4. Discussion
4.1. Population
4.2. Participation
4.3. Epidemic Curves
4.4. Risk Factors Associated with the Occurrence of an ILI
4.5. Vaccination Status
4.6. Medical Care-Seeking Behavior
4.7. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO Global Influenza Strategy 2019–2030. Available online: https://apps.who.int/iris/handle/10665/311184 (accessed on 24 April 2020).
- Seasonal Influenza 2018/19. Available online: https://www.infovac.ch/docs/public/influenza/grippe-saisonnie--re-2018-19.pdf (accessed on 24 April 2020).
- Seasonal Influenza. Available online: https://www.who.int/fr/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 24 April 2020).
- Is Seasonal Influenza Dangerous? Available online: https://www.sevaccinercontrelagrippe.ch/fr-ch/la-grippe/la-grippe-saisonniere-est-elle-dangereuse.html (accessed on 24 April 2020).
- Welcome to Sentinella. Available online: https://www.sentinella.ch/fr/info (accessed on 22 April 2020).
- European Centre for Disease Prevention and Control European Influenza Surveillance Network (EISN). Available online: https://ecdc.europa.eu/en/about-us/partnerships-and-networks/disease-and-laboratory-networks/eisn (accessed on 24 April 2020).
- Federal Office of Public Health (FOPH). Available online: https://www.bag.admin.ch/bag/fr/home.html (accessed on 24 April 2020).
- Seasonal Influenza—Status Report. Available online: https://www.bag.admin.ch/bag/fr/home/krankheiten/ausbrueche-epidemien-pandemien/aktuelle-ausbrueche-epidemien/saisonale-grippe---lagebericht-schweiz.html (accessed on 24 April 2020).
- Pandemic Influenza Severity Assessment (PISA): A WHO Guide to Assess the Severity of Influenza Epidemics and Pandemics. Available online: https://apps.who.int/iris/bitstream/handle/10665/259392/WHO-WHE-IHM-GIP-2017.2-eng.pdf?sequence=1 (accessed on 24 April 2020).
- Laboratory Diagnosis for Infectious Diseases. Available online: https://www.bag.admin.ch/bag/fr/home/krankheiten/infektionskrankheiten-bekaempfen/labordiagnostik-infektionskrankheiten.html (accessed on 24 April 2020).
- World Health Organization. WHO Interim Global Epidemiological Surveillance Standards for Influenza; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Grippenet. Available online: https://fr.grippenet.ch/fr/propos-de-nous/ (accessed on 22 April 2020).
- Welcome to InfluenzaNet Analytics. Available online: http://influenzanet.info/#page/home (accessed on 24 April 2020).
- Smolinski, M.S.; Crawley, A.W.; Olsen, J.M.; Jayaraman, T.; Libel, M. Participatory Disease Surveillance: Engaging Communities Directly in Reporting, Monitoring, and Responding to Health Threats. JMIR Public Health Surveill. 2017, 3, e62. [Google Scholar] [CrossRef] [PubMed]
- Tilston, N.L.; Eames, K.T.; Paolotti, D.; Ealden, T.; Edmunds, W.J. Internet-based surveillance of Influenza-like-illness in the UK during the 2009 H1N1 influenza pandemic. BMC Public Health 2010, 10, 650. [Google Scholar] [CrossRef] [PubMed]
- Casalegno, J.-S.; Eibach, D.; Valette, M.; Enouf, V.; Daviaud, I.; Behillil, S.; Vabret, A.; Soulary, J.C.; Benchaib, M.; Cohen, J.M.; et al. Performance of influenza case definitions for influenza community surveillance: Based on the French influenza surveillance network GROG, 2009–2014. Euro Surveill. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- EU Case Definitions. Available online: https://www.ecdc.europa.eu/en/surveillance-and-disease-data/eu-case-definitions (accessed on 25 April 2020).
- Federal Statistical Office. Available online: https://www.bfs.admin.ch/bfs/en/home.html (accessed on 26 April 2020).
- Swissveg. Available online: https://www.swissveg.ch/about_us?language=en (accessed on 26 April 2020).
- Language—Facts and Figures. Available online: https://www.eda.admin.ch/aboutswitzerland/en/home/gesellschaft/sprachen/die-sprachen---fakten-und-zahlen.html (accessed on 15 June 2020).
- Utilisation d’Internet. Available online: https://www.bfs.admin.ch/bfs/fr/home/statistiques/culture-medias-societe-information-sport/societe-information/indicateurs/utilisation-internet.assetdetail.12307273.html (accessed on 28 April 2020).
- Bundorf, M.K.; Wagner, T.H.; Singer, S.J.; Baker, L.C. Who Searches the Internet for Health Information? Health Serv. Res. 2006, 41, 819–836. [Google Scholar] [CrossRef] [PubMed]
- Swiss Health Survey. Available online: https://www.bfs.admin.ch/bfs/fr/home/statistiques/sante/enquetes/sgb.html (accessed on 26 April 2020).
- Marquet, R.L.; Bartelds, A.I.; van Noort, S.P.; Koppeschaar, C.E.; Paget, J.; Schellevis, F.G.; van der Zee, J. Internet-based monitoring of influenza-like illness (ILI) in the general population of the Netherlands during the 2003–2004 influenza season. BMC Public Health 2006, 6, 242. [Google Scholar] [CrossRef] [PubMed]
- van Noort, S.P.; Muehlen, M.; de Andrade, R.H.; Koppeschaar, C.; Lourenço, L.J.M.; Gomes, M.G. Gripenet: An internet-based system to monitor influenza-like illness uniformly across Europe. Eurosurveillance 2007, 12, 5–6. [Google Scholar] [CrossRef] [PubMed]
- Friesema, I.H.M.; Koppeschaar, C.E.; Donker, G.A.; Dijkstra, F.; van Noort, S.P.; Smallenburg, R.; van der Hoek, W.; van der Sande, M.A.B. Internet-based monitoring of influenza-like illness in the general population: Experience of five influenza seasons in the Netherlands. Vaccine 2009, 27, 6353–6357. [Google Scholar] [CrossRef] [PubMed]
- Vandendijck, Y.; Faes, C.; Hens, N. Eight Years of the Great Influenza Survey to Monitor Influenza-Like Illness in Flanders. PLoS ONE 2013, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Adler, A.J.; Eames, K.T.; Funk, S.; Edmunds, W.J. Incidence and risk factors for influenza-like-illness in the UK: Online surveillance using Flusurvey. BMC Infect. Dis. 2014, 14, 232. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, D.; Tizzoni, M.; Paolotti, D. Using Participatory Web-based Surveillance Data to Improve Seasonal Influenza Forecasting in Italy. In Proceedings of the 26th International Conference on World Wide Web, Perth, Australia, 3 April–7 April 2017. [Google Scholar]
- de Lange, M.M.; Meijer, A.; Friesema, I.H.M.; Donker, G.A.; Koppeschaar, C.E.; Hooiveld, M.; Ruigrok, N.; van der Hoek, W. Comparison of five influenza surveillance systems during the 2009 pandemic and their association with media attention. BMC Public Health 2013, 13, 881. [Google Scholar] [CrossRef] [PubMed]
- van Noort, S.P.; Codeço, C.T.; Koppeschaar, C.E.; van Ranst, M.; Paolotti, D.; Gomes, M.G.M. Ten-year performance of Influenzanet: ILI time series, risks, vaccine effects, and care-seeking behaviour. Epidemics 2015, 13, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Kjelsø, C.; Galle, M.; Bang, H.; Ethelberg, S.; Krause, T.G. Influmeter—An online tool for self-reporting of influenza-like illness in Denmark. Infect. Dis. 2016, 48, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Koppeschaar, C.E.; Colizza, V.; Guerrisi, C.; Turbelin, C.; Duggan, J.; Edmunds, W.J.; Kjelsø, C.; Mexia, R.; Moreno, Y.; Meloni, S.; et al. Influenzanet: Citizens Among 10 Countries Collaborating to Monitor Influenza in Europe. JMIR Public Health Surveill. 2017, 3, e66. [Google Scholar] [CrossRef] [PubMed]
- Guerrisi, C.; Turbelin, C.; Blanchon, T.; Hanslik, T.; Bonmarin, I.; Levy-Bruhl, D.; Perrotta, D.; Paolotti, D.; Smallenburg, R.; Koppeschaar, C.; et al. Participatory Syndromic Surveillance of Influenza in Europe. J. Infect. Dis. 2016, 214, S386–S392. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.L.K.; Marques, D.F.P.; Cameron, R.L.; Potts, A.; Bishop, J.; von Wissmann, B.; William, N.; Reynolds, A.J.; Robertson, C.; McMenamin, J. Moving epidemic method (MEM) applied to virology data as a novel real time tool to predict peak in seasonal influenza healthcare utilisation. The Scottish experience of the 2017/18 season to date. Euro Surveill. 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Vega, T.; Lozano, J.E.; Meerhoff, T.; Snacken, R.; Mott, J.; de Lejarazu, O.R.; Nunes, B. Influenza surveillance in Europe: Establishing epidemic thresholds by the Moving Epidemic Method. Influenza Other Respir. Viruses 2013, 7, 546–558. [Google Scholar] [CrossRef] [PubMed]
- Vette, K.; Bareja, C.; Clark, R.; Lal, A. Establishing thresholds and parameters for pandemic influenza severity assessment, Australia. Bull World Health Organ. 2018, 96, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Grippe (Influenza). Available online: https://www.infovac.ch/fr/les-vaccins/par-maladie/grippe (accessed on 25 April 2020).
- World Health Assembly. Fifty-sixth World Health Assembly; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Covidnet.fr Become a Part of the Monitoring of COVID-19. Available online: https://www.covidnet.fr/fr/grippenet/ (accessed on 25 April 2020).
- Debin, M.; Colizza, V.; Blanchon, T.; Hanslik, T.; Turbelin, C.; Falchi, A. Effectiveness of 2012–2013 influenza vaccine against influenza-like illness in general population. Hum. Vaccin. Immunother. 2014, 10, 536–543. [Google Scholar] [CrossRef] [PubMed]

| Variable | Category | Ntot = 1247 | |
|---|---|---|---|
| N | % | ||
| Age (years) | 0–4 | 30 | 2.4 |
| 5–14 | 68 | 5.5 | |
| 15–29 | 207 | 16.6 | |
| 30–64 | 770 | 61.7 | |
| 65+ | 164 | 13.2 | |
| No answer | 8 | 0.6 | |
| Sex | Female | 714 | 57.3 |
| Male | 533 | 42.7 | |
| No answer | 0 | 0.0 | |
| Smoking | Yes | 165 | 13.2 |
| No | 971 | 77.9 | |
| No answer | 111 | 8.9 | |
| Respiratory allergies | Yes | 395 | 31.6 |
| No | 744 | 59.7 | |
| No answer | 108 | 8.7 | |
| Chronic medical condition(s) | Yes | 178 | 14.3 |
| No | 961 | 77.1 | |
| No answer | 108 | 8.7 | |
| Influenza vaccination 2018/19 | Yes | 364 | 29.2 |
| No | 549 | 44.0 | |
| No answer | 334 | 26.8 | |
| Pregnancy (women only) | Yes | 3 | 0.4 |
| No | 365 | 51.1 | |
| No answer | 346 | 48.5 | |
| Contact risk * | Yes | 728 | 58.3 |
| No | 411 | 33.0 | |
| No answer | 108 | 8.7 | |
| Completed Education | No former education | 60 | 4.8 |
| Secondary school | 354 | 28.4 | |
| Tertiary degree | 635 | 50.9 | |
| No answer | 198 | 15.9 | |
| Main activity | Paid employment, full time | 419 | 33.6 |
| Paid employment, part time | 275 | 22.1 | |
| Self-employed | 77 | 6.2 | |
| Attending daycare/school/college/university | 201 | 16.1 | |
| Home-maker, unemployed, long-term sick-leave or parental leave | 75 | 6.0 | |
| Retired | 162 | 13.0 | |
| No answer | 38 | 3.0 | |
| Household size ** | 1 | 241 | 19.3 |
| 2 | 294 | 23.6 | |
| 3 | 192 | 15.4 | |
| 4 | 246 | 19.7 | |
| 5 | 86 | 6.9 | |
| 6+ | 45 | 3.6 | |
| No answer | 143 | 11.5 | |
| Diet | No special diet | 986 | 79.1 |
| Vegetarian | 59 | 4.7 | |
| Vegan | 10 | 0.8 | |
| Low-calorie | 26 | 2.1 | |
| Other | 54 | 4.3 | |
| No answer | 112 | 9.0 | |
| Presence of a pet | Yes | 149 | 11.9 |
| No | 986 | 79.1 | |
| No answer | 112 | 9.0 | |
| ILI (N) | Non-ILI (N) | OR | 95% CI | AOR | 95% CI | ||
|---|---|---|---|---|---|---|---|
| Age group (years) | 0–4 | 20 | 10 | 1 | - | 1 | - |
| 5 to 14 | 30 | 38 | 0.4 | 0.17–0.97 | 0.6 | 0.19–0.99 | |
| 15–29 | 70 | 137 | 0.26 | 0.12–0.57 | 0.29 | 0.15–0.60 | |
| 30–64 | 375 | 395 | 0.48 | 0.22–1.03 | 0.49 | 0.23–1.05 | |
| 65+ | 75 | 89 | 0.42 | 0.19–0.96 | 0.38 | 0.16–0.93 | |
| Gender | F | 379 | 335 | 1 | - | 1 | - |
| M | 195 | 338 | 0.5 | 0.4–0.64 | 0.81 | 0.7–0.95 | |
| Respiratory allergy | Yes | 219 | 176 | 1.38 | 1.08–1.76 | 1.58 | 1.38–1.96 |
| No | 353 | 391 | 1 | - | 1 | - | |
| Chronic medical condition(s) | Yes | 99 | 79 | 1.29 | 0.94–1.8 | - | - |
| No | 473 | 488 | 1 | - | - | - | |
| Smoking | Yes | 84 | 81 | 1.04 | 0.79–1.44 | - | - |
| No | 486 | 485 | 1 | - | - | - | |
| Influenza vaccination 2018/2019 | Yes | 140 | 225 | 1 | - | 1 | - |
| No | 310 | 239 | 2.09 | 1.59–2.73 | 2.4 | 1.9–3.04 | |
| Contact risk | Yes | 378 | 350 | 1.21 | 0.95–1.54 | - | - |
| No | 194 | 217 | 1 | - | - | - | |
| Household size | 1 | 126 | 115 | 1 | - | 1 | - |
| 2 | 147 | 147 | 0.92 | 0.65–1.28 | - | - | |
| 3 | 109 | 83 | 1.04 | 0.74–1.44 | - | - | |
| 4 or more | 176 | 201 | 1.12 | 0.82–1.75 | - | - | |
| Education | No former education | 28 | 32 | 1 | - | 1 | - |
| Secondary school | 186 | 168 | 1.27 | 0.73–2.19 | - | - | |
| Tertiary degree | 311 | 324 | 1.09 | 0.65–1.86 | - | - | |
| Diet | No special diet | 492 | 494 | 1 | - | 1 | - |
| Vegan, vegetarian | 33 | 36 | 0.92 | 0.57–1.55 | - | - | |
| Low-calorie, other | 47 | 33 | 1.43 | 0.9–2.28 | - | - | |
| Pet(s) at home | Yes | 80 | 69 | 1 | - | 1 | - |
| No | 492 | 494 | 0.85 | 0.60–1.21 | - | - | |
| Main occupation | Paid employment, full time | 186 | 233 | 1 | - | 1 | - |
| Paid employment, part time | 141 | 134 | 1.32 | 0.98–1.79 | 1.21 | 0.87–1.68 | |
| Self-employed | 44 | 33 | 1.67 | 1.03–2.73 | 1.97 | 1.33–3.03 | |
| Attending daycare/school/college/university | 86 | 115 | 0.94 | 0.67–1.31 | 0.98 | 0.72–1.35 | |
| Retired, homemaker, unemployed, long-term sick leave or parental leave | 105 | 132 | 0.99 | 0.72–1.37 | 1.06 | 0.79–1.42 | |
| Main mean of transportation | Walking | 78 | 74 | 1 | - | 1 | - |
| Bike | 61 | 64 | 0.9 | 0.56–1.45 | - | - | |
| Car | 247 | 220 | 1.06 | 0.74–1.54 | - | - | |
| Public transportation (bus, train, metro, etc.) | 171 | 190 | 0.85 | 0.58–1.25 | - | - | |
| Daily commuting time | No time at all | 324 | 289 | 1 | - | 1 | - |
| 0–30 min | 117 | 139 | 0.75 | 0.56–1.01 | - | - | |
| 30 min–1.5 h | 99 | 114 | 0.77 | 0.57–1.06 | - | - | |
| 1.5 h and over | 32 | 25 | 1.18 | 0.68–2.03 | - | - |
| Indication to Vaccination | Vaccinated Users | |
|---|---|---|
| 2017–2018 | 2018–2019 | |
| Age > 65 | 54.2% (77/142) | 66.2% (47/71) |
| Contact Risk | 37.3% (135/362) | 44.5% (81/182) |
| Chronic Disease | 52.7% (58/110) | 53.8% (35/65) |
| Pregnancy | 0% (0/4) | 50% (1/2) |
| All users (with updated intake) | 37.4% (279/746) | 47.8% (165/345) |
| All at risk users | 43.7% (270/618) | 51.3% (164/320) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richard, A.; Müller, L.; Wisniak, A.; Thiabaud, A.; Merle, T.; Dietrich, D.; Paolotti, D.; Jeannot, E.; Flahault, A. Grippenet: A New Tool for the Monitoring, Risk-Factor and Vaccination Coverage Analysis of Influenza-Like Illness in Switzerland. Vaccines 2020, 8, 343. https://doi.org/10.3390/vaccines8030343
Richard A, Müller L, Wisniak A, Thiabaud A, Merle T, Dietrich D, Paolotti D, Jeannot E, Flahault A. Grippenet: A New Tool for the Monitoring, Risk-Factor and Vaccination Coverage Analysis of Influenza-Like Illness in Switzerland. Vaccines. 2020; 8(3):343. https://doi.org/10.3390/vaccines8030343
Chicago/Turabian StyleRichard, Aude, Laura Müller, Ania Wisniak, Amaury Thiabaud, Thibaut Merle, Damien Dietrich, Daniela Paolotti, Emilien Jeannot, and Antoine Flahault. 2020. "Grippenet: A New Tool for the Monitoring, Risk-Factor and Vaccination Coverage Analysis of Influenza-Like Illness in Switzerland" Vaccines 8, no. 3: 343. https://doi.org/10.3390/vaccines8030343
APA StyleRichard, A., Müller, L., Wisniak, A., Thiabaud, A., Merle, T., Dietrich, D., Paolotti, D., Jeannot, E., & Flahault, A. (2020). Grippenet: A New Tool for the Monitoring, Risk-Factor and Vaccination Coverage Analysis of Influenza-Like Illness in Switzerland. Vaccines, 8(3), 343. https://doi.org/10.3390/vaccines8030343







