Overview of the Development, Impacts, and Challenges of Live-Attenuated Oral Rotavirus Vaccines
Abstract
1. Introduction—Overview of Rotavirus Particles
2. Rotavirus Vaccines—Implementation, Efficiency, Cost, and Challenge
3. Initial Concepts to Produce Live-Attenuated Oral Rotavirus Vaccines
4. The Renaissance of Live-Attenuated Oral Rotavirus Vaccines Today–The Journal so Far
5. Comparative Analysis of Vaccine Coverage, Effectiveness, and Efficacy
6. Factors Affecting Rotavirus Vaccine Efficiency
6.1. Breastfeeding
6.2. Maternal Antibodies Acquired Transplacentally
6.3. Microbiota/Probiotic Diversities
6.4. Malnutrition
6.5. Co-Infection
6.6. Overage
6.7. Underage
6.8. Juvenile Immune System
6.9. Tropical Enteropathy
6.10. Histo-Blood Group Antigens
6.11. Vaccine Interference from Co-Administration with Other Childhood Routine Vaccines
6.12. Sequential Vaccine Doses
7. Problems Associated with the Live-Attenuated Rotavirus Vaccines
7.1. Live-Attenuated Rotavirus Vaccines Cause Intussusception
7.2. Incidence of Adventitious Contaminants in the Live-Attenuated Oral Rotavirus Vaccines
7.3. Live-Attenuated Oral Rotavirus Vaccines Reassort
7.4. Live-Attenuated Oral Rotavirus Vaccines Require Cold Storage Facility and Transportation
7.5. Formulation, Processing, and Packaging of the Live-Attenuated Oral Rotavirus Vaccines
7.6. Rate of Viral Passaging May Induce Vaccine Adverse Events/Reactogenicity
8. Impact of Rotavirus Vaccines on the Genotype Distribution Pattern
9. Vaccine- and Natural-Acquired Protections against Rotavirus Infections
10. Conclusions, Recommendation, and Future Direction
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 116E–Rotavac® |
| ACIP = US Advisory Committee on Immunization Practices |
| AD = any dose |
| AGE = acute gastroenteritis |
| AGMKC = African green monkey kidney cells |
| ARD = acute rotavirus diarrhoea |
| BCG = Bacille Calmette-Guérin |
| BRV = bovine rotavirus |
| BRV-PV = bovine rotavirus pentavalent vaccine |
| CDC = Centers for Disease Control and Prevention |
| CEMR = cryo-electron microscopy reconstruction |
| CMPKEC = Cynomolgus monkey primary kidney epithelial cells |
| COVID-19 = coronavirus disease (discovered in 2019) |
| D = dose |
| DALY = disability-adjusted life year |
| DLP = double-layered particle |
| DNA = deoxyribonucleic acid |
| DPT = Diptheria, Pertussis, and Tetanus same as DTaP or DTP |
| dsRNA = double-stranded ribonucleic acid |
| EE = environmental enteropathy |
| EED = environmental enteric dysfunction |
| EPI = Expanded Programme on Immunisation |
| ESPGHAN = European Society for Paediatric, Gastroenterology, Hepatology and Nutrition |
| ESPID = European Society for Paediatric Infectious Diseases |
| EV = emergency visit |
| FBKC = foetal bovine kidney cell |
| FD = full doses |
| FFU = focus-forming unit |
| FUT = fucosyltransferase gene |
| GalNAc = N-acetylgalactosamine |
| GAVI = Global Alliance for Vaccines and Immunisation |
| GE = gastroenteritis |
| GMC = geometric mean titre |
| GRSN = Global Rotavirus Surveillance Network |
| GSK = Glaxo SmithKline (formerly Glaxo Wellcome) |
| HBGAs = histo-blood group antigens |
| HICs = high income countries |
| HMICs = high-middle income countries |
| IgA = immunoglobulin A |
| IPV = inactivated polio vaccine |
| IS = intussusception |
| ITT = intention-to-treat |
| ITTP = intention-to-treat population |
| LAORoVs = live-attenuated oral rotavirus vaccines |
| Le = Lewis factor |
| LICs = low income countries |
| LMICs = low-to-middle income countries |
| LRTI = lower respiratory tract infection |
| MenAV = Meningococcal A conjugate vaccine |
| MICs = middle income countries |
| mRNA = messenger ribonucleic acid |
| MRV = Measles-Rubella Vaccine |
| MV = Measles vaccine |
| NCDV = Nebraska Calf Diarrhoea Virus |
| NGO = non-governmental organisation |
| NIH = National Institute of Health |
| NIP = National Immunisation Program |
| NSP = non-structural protein |
| NSP1 = interferon antagonist |
| NSP2 = NTPase |
| NSP3 = translational enhancer |
| NSP4 = enterotoxin |
| NSP5/6 = phosphoprotein |
| OPV = oral polio vaccine |
| ORF = open reading frame |
| ORS = oral rehydrating solution |
| PAHO = Pan American Health Organisation |
| PATH = Program for Appropriate Technology in Health |
| PCVs = porcine circoviruses |
| PKC = porcine kidney cell |
| PPP = per-protocol population |
| REST = rotavirus efficacy and safety trial |
| RIX4414 = Rotarix® |
| RNA = ribonucleic acid |
| RoV = rotavirus vaccine |
| RoVE = rotavirus vaccine effectiveness |
| RoVs = rotavirus vaccines |
| RRV-PV rhesus rotavirus pentavalent vaccine |
| RRV-TV = rhesus rotavirus tetravalent (RotaShield®) |
| RTI = respiratory tract infection |
| RV1 = Rotarix® |
| RV4 = RotaShield® |
| RV5 = RotaTeq® |
| RVD = rotavirus diarrhoea |
| RVGE = rotavirus gastroenteritis |
| RVI = rotavirus infection |
| RVIs = rotavirus infection |
| RV-VLPs = rotavirus virus-like particles |
| SAGE = Strategic Advisory Group of Experts |
| SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2 |
| SGE = severe gastroenteritis |
| SLP = single-layered particle |
| SRVD = severe rotavirus diarrhoea |
| SRVGE = severe rotavirus gastroenteritis |
| TLP = triple-layered particleUN = United Nations |
| UNICEF = United Nations Children’s Fund (United Nations International Children’s Emergency Fund) |
| VE = vaccine effectiveness |
| VP = viral protein |
| VP1 = RNA-dependent RNA polymerase |
| VP2 = core protein |
| VP3 = methyltransferase-guanylyltransferase |
| VP4 = spike-like protease-sensitive neutralising antigen |
| VP6 = inner capsid |
| VP7 = glycosylated neutralising antigen |
| VP8* = sialic-binding protein |
| VSRVGE = very severe rotavirus gastroenteritis |
| WHO = World Health Organisation |
| WUENIC = WHO/UNICEF Estimates of National Immunization Coverage |
| YFV = Yellow fever vaccine |
References
- Kapahnke, R.; Rappold, W.; Desselberger, U.; Riesner, D. The stiffness of dsRNA: Hydrodynamic studies on fluorescence-labelled RNA segments of bovine rotavirus. Nucleic Acids Res. 1986, 14, 3215–3228. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Labbe, M.; Baudoux, P.; Charpilienne, A.; Poncet, D.; Cohen, J. Identification of the nucleic acid binding domain of the rotavirus VP2 protein. J. Gen. Virol. 1994, 75(Pt. 12), 3423–3430. [Google Scholar] [CrossRef]
- Kozak, M. Adherence to the First-Aug Rule When a Second Aug Codon Follows Closely Upon the First. Proc. Natl. Acad. Sci. USA 1995, 92, 2662–2666. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, A.R.; Both, G.W. Molecular biology of rotaviruses. In Advances in Virus Research; Elsevier: Amsterdam, The Netherlands, 1990; Volume 38, pp. 1–43. [Google Scholar]
- Estes, M.K.; Cohen, J. Rota gene structure and function. Microbiol. Rev. 1989, 410–449. [Google Scholar] [CrossRef]
- Mattion, N.M.; Mitchell, D.B.; Both, G.W.; Estes, M.K. Expression of rotavirus proteins encoded by alternative open reading frames of genome segment 11. Virology 1991, 181, 295–304. [Google Scholar] [CrossRef]
- Nuttall, S.D.; Hum, C.P.; Holmes, I.H.; Dyall-Smith, M.L. Sequences of VP9 genes from short and supershort rotavirus strains. Virology 1989, 171, 453–457. [Google Scholar] [CrossRef]
- Prasad, B.V.; Rothnagel, R.; Zeng, C.Q.; Jakana, J.; Lawton, J.A.; Chiu, W.; Estes, M.K. Visualization of ordered genomic RNA and localization of transcriptional complexes in rotavirus. Nature 1996, 382, 471–473. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Ciarlet, M.; Rahman, M.; Attoui, H.; Banyai, K.; Estes, M.K.; Gentsch, J.R.; Iturriza-Gomara, M.; Kirkwood, C.D.; Martella, V.; et al. Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Arch. Virol. 2008, 153, 1621–1629. [Google Scholar] [CrossRef]
- Mihalov-Kovacs, E.; Gellert, A.; Marton, S.; Farkas, S.L.; Feher, E.; Oldal, M.; Jakab, F.; Martella, V.; Banyai, K. Candidate New Rotavirus Species in Sheltered Dogs, Hungary. Emerg. Infect. Dis. 2015, 21, 660–663. [Google Scholar] [CrossRef]
- Banyai, K.; Kemenesi, G.; Budinski, I.; Foldes, F.; Zana, B.; Marton, S.; Varga-Kugler, R.; Oldal, M.; Kurucz, K.; Jakab, F. Candidate new rotavirus species in Schreiber’s bats, Serbia. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2017, 48, 19–26. [Google Scholar] [CrossRef]
- Estes, M.K.; Greenberg, H.B. Rotavirus. In Fields Virology; Knipe, P.M., Howley, D.M., Eds.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 1347–1401. [Google Scholar]
- Jayaram, H.; Estes, M.K.; Prasad, B.V. Emerging themes in rotavirus cell entry, genome organization, transcription and replication. Virus Res. 2004, 101, 67–81. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO). Immunization, Vaccines and Biologicals-Rotavirus; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Collaborators, G.B.D.C.o.D. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Steele, A.D.; Duque, J.; Parashar, U.D.; S, W.C.G.R. 2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: A systematic review and meta-analysis. Lancet Infect. Dis. 2012, 12, 136–141. [Google Scholar] [CrossRef]
- Troeger, C.; Khalil, I.A.; Rao, P.C.; Cao, S.; Blacker, B.F.; Ahmed, T.; Armah, G.; Bines, J.E.; Brewer, T.G.; Colombara, D.V.; et al. Rotavirus Vaccination and the Global Burden of Rotavirus Diarrhea Among Children Younger Than 5 Years. JAMA Pediatr. 2018, 172, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, Z.; Xie, G.; Wang, H.; Zhang, Q.; Sun, X.; Guo, N.; Pang, L.; Duan, Z. Genotype of Rotavirus Vaccine Strain LLR in China is G10P[15]. Bing Du Xue Bao Chin. J. Virol. 2015, 31, 170–173. [Google Scholar]
- Le, T.L.; Nguyen, T.H.; Dang, D.A.; Nguyen, V.T.; Tran, B.H.; Tran, T.G.H.; Tran, T.O.; Pham, G.K.; Jiang, B.; Glass, R. Advances in research on Rotavin-M1. Vietnam J. Sci. Technol. Eng. 2017, 59, 43–48. [Google Scholar] [CrossRef]
- Msimang, V.M.Y.; Page, N.; Groome, M.J.; Moyes, J.; Cortese, M.M.; Seheri, M.; Kahn, K.; Chagan, M.; Madhi, S.A.; Cohen, C. Impact of Rotavirus Vaccine on Childhood Diarrheal Hospitalization After Introduction Into the South African Public Immunization Program. Pediatr. Infect. Dis. J. 2013, 32, 1359–1364. [Google Scholar] [CrossRef]
- Richardson, V.; Parashar, U.; Patel, M. Childhood diarrhea deaths after rotavirus vaccination in Mexico. N. Engl. J. Med. 2011, 365, 772–773. [Google Scholar] [CrossRef]
- Mwenda, J.M.; Mandomando, I.; Jere, K.C.; Cunliffe, N.A.; Duncan Steele, A. Evidence of reduction of rotavirus diarrheal disease after rotavirus vaccine introduction in national immunization programs in the African countries: Report of the 11(th) African rotavirus symposium held in Lilongwe, Malawi. Vaccine 2019, 37, 2975–2981. [Google Scholar] [CrossRef]
- Jere, K.C.; Chaguza, C.; Bar-Zeev, N.; Lowe, J.; Peno, C.; Kumwenda, B.; Nakagomi, O.; Tate, J.E.; Parashar, U.D.; Heyderman, R.S.; et al. Emergence of Double- and Triple-Gene Reassortant G1P[8] Rotaviruses Possessing a DS-1-Like Backbone after Rotavirus Vaccine Introduction in Malawi. J. Virol. 2018, 92, e01246-17. [Google Scholar] [CrossRef]
- Velasquez, D.E.; Parashar, U.; Jiang, B. Decreased performance of live attenuated, oral rotavirus vaccines in low-income settings: Causes and contributing factors. Expert Rev. Vaccines 2018, 17, 145–161. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.S.; Groome, M.J.; Velasquez, D.E.; Parashar, U.D.; Jones, S.; Koen, A.; van Niekerk, N.; Jiang, B.; Madhi, S.A. Prevaccination Rotavirus Serum IgG and IgA Are Associated With Lower Immunogenicity of Live, Oral Human Rotavirus Vaccine in South African Infants. Clin. Infect. Dis. 2016, 62, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Becker-Dreps, S.; Vilchez, S.; Velasquez, D.; Moon, S.-S.; Hudgens, M.G.; Zambrana, L.E.; Jiang, B. Rotavirus-specific IgG antibodies from mothers’ serum may inhibit infant immune responses to the pentavalent rotavirus vaccine. Pediatr. Infect. Dis. J. 2015, 34, 115. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Moon, S.; Wang, Y.; Jiang, B. Multiple virus infection alters rotavirus replication and expression of cytokines and Toll-like receptors in intestinal epithelial cells. Virus Res. 2012, 167, 48–55. [Google Scholar] [CrossRef]
- Harris, V.C.; Armah, G.; Fuentes, S.; Korpela, K.E.; Parashar, U.; Victor, J.C.; Tate, J.; de Weerth, C.; Giaquinto, C.; Wiersinga, W.J.; et al. Significant Correlation Between the Infant Gut Microbiome and Rotavirus Vaccine Response in Rural Ghana. J. Infect. Dis. 2017, 215, 34–41. [Google Scholar] [CrossRef]
- Harris, V.; Ali, A.; Fuentes, S.; Korpela, K.; Kazi, M.; Tate, J.; Parashar, U.; Wiersinga, W.J.; Giaquinto, C.; de Weerth, C.; et al. Rotavirus vaccine response correlates with the infant gut microbiota composition in Pakistan. Gut Microbes 2018, 9, 93–101. [Google Scholar] [CrossRef]
- Valdez, Y.; Brown, E.M.; Finlay, B.B. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014, 35, 526–537. [Google Scholar] [CrossRef]
- Greenberg, H.B.; Estes, M.K. Rotaviruses: From pathogenesis to vaccination. Gastroenterology 2009, 136, 1939–1951. [Google Scholar] [CrossRef]
- Chandran, A.; Fitzwater, S.; Zhen, A.; Santosham, M. Prevention of rotavirus gastroenteritis in infants and children: Rotavirus vaccine safety, efficacy, and potential impact of vaccines. Biologics 2010, 4, 213–229. [Google Scholar] [CrossRef]
- World Health Organisation. Immunization Coverage; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Parashar, U.D. Global, regional, and national estimates of rotavirus mortality in children <5 years of age, 2000–2013. Clin. Infect. Dis. 2016, 62, S96–S105. [Google Scholar] [CrossRef]
- RotaCouncil. Global Introduction Status-Rotavirus vaccine Introduction and Coverage; Johns Hopkins Bloomberg School of Public Health and International Vacccine Access Center (IVAC): Baltimore, MD, USA, 2020. [Google Scholar]
- Sabin Vaccine Institute. Expanding Rotavirus Vaccine Impact: Improving Coverage and Equity. In Proceedings of the 13th International Rotavirus Symposium, Minsk, Belarus, 29–30 August 2018; Sabin Vaccine Institute: Washinton, DC, USA, 2018; p. 34. [Google Scholar]
- United Nations Children’s Fund (UNICEF). Vaccine Resourse Library; PATH: Washington, DC, USA, 2018. [Google Scholar]
- Burnett, E.; Parashar, U.; Tate, J. Rotavirus Vaccines: Effectiveness, Safety, and Future Directions. Paediatr. Drugs 2018, 20, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Burnett, E.; Tate, J.E.; Kirkwood, C.D.; Nelson, E.A.S.; Santosham, M.; Steele, A.D.; Parashar, U.D. Estimated impact of rotavirus vaccine on hospitalizations and deaths from rotavirus diarrhea among children < 5 in Asia. Expert Rev. Vaccines 2018, 17, 453–460. [Google Scholar] [PubMed]
- World Health Organisation (WHO). State of the World’s Vaccines and Immunizations; World Health Organization; United Nations Children’s Fund & World Bank; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Global Alliance for Vaccines and Immunizations (GAVI). Gavi Supports Rotavirus Vaccine Introduction in Uganda; GAVI: Geneva, Switzerland, 2018. [Google Scholar]
- United Nation Children’s Fund (UNICEF). Supply of Children’s Five-in-One Vaccine Secured at Lowest-Ever Price; UNICEF: Copenhagen, Denmark, 2016. [Google Scholar]
- GlaxoSmithKline (GSK). Millions of Children in the World’s Poorest Countries could Receive Vaccination Against Rotavirus Diarrhoeal Disease under New Offer made by GSK to the GAVI Alliance; GlaxoSmithKline: Brentford, UK, 2011. [Google Scholar]
- Merck & Co. Inc. Merck Commends GAVI Alliance on Continued Efforts to IMPROVE access; Merck & Co. Inc.: Kenilworth, NJ, USA, 2011. [Google Scholar]
- Madhi, S.A.; Bamford, L.; Ngcobo, N. Effectiveness of pneumococcal conjugate vaccine and rotavirus vaccine introduction into the South African public immunisation programme. Samj S. Afr. Med. J. 2014, 104, 228–234. [Google Scholar] [CrossRef] [PubMed]
- United Nations Children’s Fund (UNICEF). Supplies and Logistics. In Vaccine Price Data; UNICEF: Copenhagen, Denmark, 2019. [Google Scholar]
- Centers for Disease Control Prevention (CDC). Vaccines for Children Program (VFC); National Center for Immunization and Respiratory Diseases: Atlanta, GO, USA, 2020. [Google Scholar]
- Ngabo, F.; Mvundura, M.; Gazley, L.; Gatera, M.; Rugambwa, C.; Kayonga, E.; Tuyishime, Y.; Niyibaho, J.; Mwenda, J.M.; Donnen, P. The economic burden attributable to a child’s inpatient admission for diarrheal disease in Rwanda. PLoS ONE 2016, 11, e0149805. [Google Scholar] [CrossRef]
- Nonvignon, J.; Atherly, D.; Pecenka, C.; Aikins, M.; Gazley, L.; Groman, D.; Narh, C.T.; Armah, G. Cost-effectiveness of rotavirus vaccination in Ghana: Examining impacts from 2012 to 2031. Vaccine 2018, 36, 7215–7221. [Google Scholar] [CrossRef]
- Anwari, P.; Debellut, F.; Pecenka, C.; Parwiz, S.M.; Clark, A.; Groman, D.; Safi, N. Potential impact and cost-effectiveness of rotavirus vaccination in Afghanistan. Vaccine 2018, 36, 7769–7774. [Google Scholar] [CrossRef]
- Rheingans, R.D.; Constenla, D.; Antil, L.; Innis, B.L.; Breuer, T. Potential cost-effectiveness of vaccination for rotavirus gastroenteritis in eight Latin American and Caribbean countries. Revista Panamericana de Salud Pública 2007, 21, 205–216. [Google Scholar] [CrossRef]
- De la Hoz, F.; Alvis, N.; Narvaez, J.; Cediel, N.; Gamboa, O.; Velandia, M. Potential epidemiological and economical impact of two rotavirus vaccines in Colombia. Vaccine 2010, 28, 3856–3864. [Google Scholar] [CrossRef]
- Atherly, D.E.; Lewis, K.D.; Tate, J.; Parashar, U.D.; Rheingans, R.D. Projected health and economic impact of rotavirus vaccination in GAVI-eligible countries: 2011–2030. Vaccine 2012, 30 (Suppl. 1), A7–A14. [Google Scholar] [CrossRef]
- Atherly, D.; Dreibelbis, R.; Parashar, U.D.; Levin, C.; Wecker, J.; Rheingans, R.D. Rotavirus Vaccination: Cost-Effectiveness and Impact on Child Mortality in Developing Countries. J. Infect. Dis. 2009, 200, S28–S38. [Google Scholar] [CrossRef]
- Pecenka, C.; Debellut, F.; Bar-Zeev, N.; Anwari, P.; Nonvignon, J.; Shamsuzzaman, M.; Clark, A. Re-evaluating the cost and cost-effectiveness of rotavirus vaccination in Bangladesh, Ghana, and Malawi: A comparison of three rotavirus vaccines. Vaccine 2018, 36, 7472–7478. [Google Scholar] [CrossRef] [PubMed]
- Ogden, K.M.; Tan, Y.; Akopov, A.; Stewart, L.S.; McHenry, R.; Fonnesbeck, C.J.; Piya, B.; Carter, M.H.; Fedorova, N.B.; Halpin, R.A.; et al. Multiple introductions and antigenic mismatch with vaccines may contribute to increased predominance of G12P[8] rotaviruses in the United States. J. Virol. 2018, 93, e01476-18. [Google Scholar] [CrossRef] [PubMed]
- Zaman, K.; Dang, D.A.; Victor, J.C.; Shin, S.; Yunus, M.; Dallas, M.J.; Podder, G.; Vu, D.T.; Le, T.P.; Luby, S.P.; et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in Asia: A randomised, double-blind, placebo-controlled trial. Lancet 2010, 376, 615–623. [Google Scholar] [CrossRef]
- Kassim, P.; Eslick, G.D. Risk of intussusception following rotavirus vaccination: An evidence based meta-analysis of cohort and case-control studies. Vaccine 2017, 35, 4276–4286. [Google Scholar] [CrossRef] [PubMed]
- Murphy, T.V.; Gargiullo, P.M.; Massoudi, M.S.; Nelson, D.B.; Jumaan, A.O.; Okoro, C.A.; Zanardi, L.R.; Setia, S.; Fair, E.; LeBaron, C.W.; et al. Intussusception among infants given an oral rotavirus vaccine. N. Engl. J. Med. 2001, 344, 564–572. [Google Scholar] [CrossRef]
- Bhandari, N.; Rongsen-Chandola, T.; Bavdekar, A.; John, J.; Antony, K.; Taneja, S.; Goyal, N.; Kawade, A.; Kang, G.; Rathore, S.S.; et al. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: A randomised, double-blind, placebo-controlled trial. Lancet 2014, 383, 2136–2143. [Google Scholar] [CrossRef]
- Kulkarni, P.S.; Desai, S.; Tewari, T.; Kawade, A.; Goyal, N.; Garg, B.S.; Kumar, D.; Kanungo, S.; Kamat, V.; Kang, G.; et al. A randomized Phase III clinical trial to assess the efficacy of a bovine-human reassortant pentavalent rotavirus vaccine in Indian infants. Vaccine 2017, 35, 6228–6237. [Google Scholar] [CrossRef]
- Parashar, U.D.; Cortese, M.M.; Payne, D.C.; Lopman, B.; Yen, C.; Tate, J.E. Value of post-licensure data on benefits and risks of vaccination to inform vaccine policy: The example of rotavirus vaccines. Vaccine 2015, 33, D55–D59. [Google Scholar] [CrossRef]
- Simonsen, L.; Viboud, C.; Elixhauser, A.; Taylor, R.J.; Kapikian, A.Z. More on RotaShield and intussusception: The role of age at the time of vaccination. J. Infect. Dis. 2005, 192, S36–S43. [Google Scholar] [CrossRef]
- Global Alliance for Vaccines and Immunizations (GAVI). The Vaccine Alliance Risk and Assurance Report 2018. In Risk & Assurance Report 2018; GAVI: Geneva, Switzerland, 2018; pp. 1–37. [Google Scholar]
- World Health Organisation (WHO). Introduction of rotavirus vaccines. In Information for Policy Makers, Programme Managers, and Health Workers; Expanded Programme on Immunization of the Department of Immunization, Vaccines, and Biologicals: Geneva, Switzerland, 2013; pp. 1–76. [Google Scholar]
- Bishop, R.F. Development of candidate rotavirus vaccines. Vaccine 1993, 11, 247–254. [Google Scholar] [CrossRef]
- Kapikian, A.Z.; Flores, J.; Midthun, K.; Hoshino, Y.; Green, K.Y.; Gorziglia, M.; Nishikawa, K.; Chanock, R.M.; Potash, L.; Perez-Schael, I. Strategies for the development of a rotavirus vaccine against infantile diarrhea with an update on clinical trials of rotavirus vaccines. In The Immune Response to Viral Infections; Springer: Cham, Switzerland, 1989; pp. 67–89. [Google Scholar]
- Lobmann, M.; Charlier, P.; Delem, A.; Zygraich, N.; Lambert, J.P.; Zissis, G. Challenge experiments in colostrum deprived piglets previously immunized with human type 2 and bovine, RIT4237, rotavirus strains; evidence of homologous and heterologous protection. In Proceedings of the Fifth International Congress of Virology, Strasbourg, France, 2–7 August 1981; p. 195. [Google Scholar]
- Lobmann, M.; Charlier, P.; Delem, A.; Zygraich, N.; Lambert, J.P.; Zissis, G. Cross-protection studies in piglets artificially infected with the bovine rotavirus strain RIT 4237, and challenged with human rotavirus type 2 and type 3. In Proceedings of the Recent Advances in Enteric Infections, Brugge, Belgium, 8–11 September 1981; p. 37. [Google Scholar]
- Zissis, G.; Lambert, J.P.; Marbebant, P.; Marissens, D.; Lobmann, M.; Charlier, P.; Delem, A.; Zygraich, N. Protection studies in colostrum-deprived piglets of a bovine rotavirus vaccine candidate using human rotavirus strains for challenge. J. Infect. Dis. 1983, 148, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Mebus, C.A.; Kono, M.; Underdahl, N.R.; Twiehaus, M.J. Cell culture propagation of neonatal calf diarrhea (scours) virus. Can. Vet. J. 1971, 12, 69. [Google Scholar] [PubMed]
- Delem, A.; Lobmann, M.; Zygraich, N. A bovine rotavirus developed as a candidate vaccine for use in humans. J. Biol. Stand. 1984, 12, 443–445. [Google Scholar] [CrossRef]
- Vesikari, T.; Isolauri, E.; Delem, A.; D’Hondt, E.; André, F.; Zissis, G. Immunogenicity and safety of live oral attenuated bovine rotavirus vaccine strain RIT 4237 in adults and young children. Lancet 1983, 322, 807–811. [Google Scholar] [CrossRef]
- Vesikari, T.; Isolauri, E.; D’Hondt, E.; Delem, A.; Andre, F.E.; Zissis, G. Protection of infants against rotavirus diarrhoea by RIT 4237 attenuated bovine rotavirus strain vaccine. Lancet 1984, 1, 977–981. [Google Scholar] [CrossRef]
- Maldonado, Y.; Hestvik, L.; Wilson, M.; Townsend, T.; O’Hare, J.; Wee, S.; Yolken, R. Safety and immunogenicity of bovine rotavirus vaccine RIT 4237 in 3-month-old infants. J. Pediatr. 1986, 109, 931–935. [Google Scholar] [CrossRef]
- Hanlon, P.; Hanlon, L.; Marsh, V.; Byass, P.; Shenton, F.; Hassan-King, M.; Jobe, O.; Sillah, H.; Hayes, R.; M’Boge, B.H.; et al. Trial of an attenuated bovine rotavirus vaccine (RIT 4237) in Gambian infants. Lancet 1987, 1, 1342–1345. [Google Scholar] [CrossRef]
- De Mol, P.; Zissis, G.; Butzler, J.P.; Mutwewingabo, A.; Andre, F.E. Failure of live, attenuated oral rotavirus vaccine. Lancet 1986, 2, 108. [Google Scholar] [CrossRef]
- Bernstein, D.I.; Glass, R.I.; Rodgers, G.; Davidson, B.L.; Sack, D.A. Evaluation of rhesus rotavirus monovalent and tetravalent reassortant vaccines in US children. US Rotavirus Vaccine Efficacy Group. JAMA 1995, 273, 1191–1196. [Google Scholar] [CrossRef]
- Rennels, M.B.; Glass, R.I.; Dennehy, P.H.; Bernstein, D.I.; Pichichero, M.E.; Zito, E.T.; Mack, M.E.; Davidson, B.L.; Kapikian, A.Z. Safety and efficacy of high-dose rhesus-human reassortant rotavirus vaccines—report of the National Multicenter Trial. Pediatrics 1996, 97, 7–13. [Google Scholar]
- Losonsky, G.A.; Rennels, M.B.; Kapikian, A.Z.; Midthun, K.; Ferra, P.J.; Fortier, D.N.; Hoffman, K.M.; Baig, A.; Levine, M.M. Safety, infectivity, transmissibility and immunogenicity of rhesus rotavirus vaccine (MMU 18006) in infants. Pediatr. Infect. Dis. 1986, 5, 25–29. [Google Scholar] [CrossRef]
- Gothefors, L.; Wadell, G.; Juto, P.; Taniguchi, K.; Kapikian, A.Z.; Glass, R.I. Prolonged efficacy of rhesus rotavirus vaccine in Swedish children. J. Infect. Dis. 1989, 159, 753–757. [Google Scholar] [CrossRef]
- Vesikari, T.; Rautanen, T.; Varis, T.; Beards, G.M.; Kapikian, A.Z. Rhesus rotavirus candidate vaccine: Clinical trial in children vaccinated between 2 and 5 months of age. Am. J. Dis. Child. 1990, 144, 285–289. [Google Scholar] [CrossRef]
- Christy, C.; Madore, H.P.; Pichichero, M.E.; Gala, C.; Pincus, P.; Vosefski, D.; Hoshino, Y.; Kapikian, A.; Dolin, R. Field trial of rhesus rotavirus vaccine in infants. Pediatr. Infect. Dis. J. 1988, 7, 645–650. [Google Scholar] [CrossRef]
- Wallace, R.E.; Vasington, P.J.; Petricciani, J.C.; Hopps, H.E.; Lorenz, D.E. Diploid cell lines from subhuman primates as substrates for virus vaccine production. Prog. Immunobiol. Stand. 1971, 5, 181. [Google Scholar]
- Kapikian, A.Z.; Midthun, K.; Hoshino, Y.; Flores, J.; Wyatt, R.G.; Glass, R.I.; Askaa, J.; Nakagomi, O.; Nakagomi, T.; Chanock, R.M.; et al. Rhesus rotavirus: A candidate vaccine for prevention of human rotavirus disease. In Vaccines 85: Molecular and Chemical Basis of Resistance to Parasitic, Bacterial and Viral Diseases; Lerner, R.A., Chanock, R.M., Brown, F., Eds.; Cold Spring Harbor Laboratory: Cold Spring Harbor, NJ, USA, 1985; pp. 357–367. [Google Scholar]
- Vesikari, T.; Kapikian, A.Z.; Delem, A.; Zissis, G. A comparative trial of rhesus monkey (RRV-1) and bovine (RIT 4237) oral rotavirus vaccines in young children. J. Infect. Dis. 1986, 153, 832–839. [Google Scholar] [CrossRef]
- Vesikari, T.; Ruuska, T.; Delem, A.; AndrÉ, F.E. Oral rotavirus vaccination in breast-and bottle-fed infants aged 6 to 12 months. Acta Pædiatrica 1986, 75, 573–578. [Google Scholar] [CrossRef]
- Wyatt, R.G.; Mebus, C.A.; Yolken, R.H.; Kalica, A.R.; James, H.D.; Kapikian, A.Z.; Chanock, R.M. Rotaviral immunity in gnotobiotic calves: Heterologous resistance to human virus induced by bovine virus. Science 1979, 203, 548–550. [Google Scholar] [CrossRef]
- Woode, G.N.; Bridger, J.C.; Jones, J.M.; Flewett, T.H.; Bryden, A.S.; Davies, H.A.; White, G.B.B. Morphological and Antigenic Relationships between Viruses (Rotaviruses) from Acute Gastroenteritis of Children, Calves, Piglets, Mice, and Foals. Infect. Immun. 1976, 14, 804–810. [Google Scholar] [CrossRef]
- Kapikian, A.Z.; Wyatt, R.G.; Greenberg, H.B.; Kalica, A.R.; Kim, H.W.; Brandt, C.D.; Rodriguez, W.J.; Parrott, R.H.; Chanock, R.M. Approaches to immunization of infants and young children against gastroenteritis due to rotaviruses. Rev. Infect. Dis. 1980, 2, 459–469. [Google Scholar] [CrossRef]
- Bernstein, D.I.; Smith, V.E.; Sander, D.S.; Pax, K.A.; Schiff, G.M.; Ward, R.L. Evaluation of WC3 rotavirus vaccine and correlates of protection in healthy infants. J. Infect. Dis. 1990, 162, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Clark, H.F.; Borian, F.E.; Bell, L.M.; Modesto, K.; Gouvea, V.; Plotkin, S.A. Protective effect of WC3 vaccine against rotavirus diarrhea in infants during a predominantly serotype 1 rotavirus season. J. Infect. Dis. 1988, 158, 570–587. [Google Scholar] [CrossRef]
- Ward, R.L.; Sander, D.S.; Schiff, G.M.; Bernstein, D.I. Effect of vaccination on serotype-specific antibody responses in infants administered WC3 bovine rotavirus before or after a natural rotavirus infection. J. Infect. Dis. 1990, 162, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Kapikian, A.Z.; Chanock, R.M. Rotaviruses. In Virology; Fields, B.N., Knipe, D.M., Eds.; Raven Press Ltd.: London, UK, 1990; pp. 1353–1404. [Google Scholar]
- Vesikari, T.; Ruuska, T.; Koivu, H.-P.; Green, K.Y.; Flores, J.; Kapikian, A.Z. Evaluation of the M37 human rotavirus vaccine in 2-to 6-month-old infants. Pediatr. Infect. Dis. J. 1991, 10, 912–917. [Google Scholar] [CrossRef]
- Midthun, K.; Halsey, N.A.; Jett-Goheen, M.; Clements, M.L.; Steinhoff, M.; King, J.C.; Karron, R.; Wilson, M.; Burns, B.; Perkis, V. Safety and immunogenicity of human rotavirus vaccine strain M37 in adults, children, and infants. J. Infect. Dis. 1991, 164, 792–796. [Google Scholar] [CrossRef]
- Flores, J.; Perez-Schael, I.; Blanco, M.; White, L.; Garcia, D.; Vilar, M.; Cunto, W.; Gonzalez, R.; Urbina, C.; Boher, J.; et al. Comparison of reactogenicity and antigenicity of M37 rotavirus vaccine and rhesus-rotavirus-based quadrivalent vaccine. Lancet 1990, 336, 330–334. [Google Scholar] [CrossRef]
- Barnes, G.L.; Lund, J.S.; Adams, L.; Mora, A.; Mitchell, S.V.; Caples, A.; Bishop, R.F. Phase 1 trial of a candidate rotavirus vaccine (RV3) derived from a human neonate. J. Paediatr. Child. Health 1997, 33, 300–304. [Google Scholar] [CrossRef]
- Bishop, R.F.; Barnes, G.L.; Cipriani, E.; Lund, J.S. Clinical immunity after neonatal rotavirus infection. A prospective longitudinal study in young children. N. Engl. J. Med. 1983, 309, 72–76. [Google Scholar] [CrossRef]
- Barnes, G.L.; Lund, J.S.; Mitchell, S.V.; De Bruyn, L.; Piggford, L.; Smith, A.L.; Furmedge, J.; Masendycz, P.J.; Bugg, H.C.; Bogdanovic-Sakran, N.; et al. Early phase II trial of human rotavirus vaccine candidate RV3. Vaccine 2002, 20, 2950–2956. [Google Scholar] [CrossRef]
- Hoshino, Y.; Kapikian, A.Z. Rotavirus vaccine development for the prevention of severe diarrhea in infants and young children. Trends Microbiol. 1994, 2, 242–249. [Google Scholar] [CrossRef]
- Hoshino, Y.; Kapikian, A.Z.; Chanock, R.M. Attenuated human rotavirus vaccine. U.S. Patent US7150984B2, 19 December 2006. [Google Scholar]
- United States—Department of State—Bureau of Oceans—International Environmental and Scientific Affairs. The United States-Japan Cooperative Medical Science Program.: Fifth Five-year Report, 1986–1990; Bureau of Oceans and International Environmental and Scientific Affairs, University of Minnesota: Minneapolis, MN, USA, 1990; Volume 9761, p. 329. [Google Scholar]
- Shigeo, M.; Shigeki, M.; Mitsuo, T.; Masami, H.; Sakae, I.; Ayako, H.; Konosuke, F. Cold-adaptation of human rotavirus. Virus Res. 1987, 7, 273–280. [Google Scholar] [CrossRef]
- Edelman, R. Perspective on the development and deployment of rotavirus vaccines. Pediatr. Infect. Dis. J. 1987, 6, 704–710. [Google Scholar] [CrossRef]
- Midthun, K.; Greenberg, H.B.; Hoshino, Y.; Kapikian, A.Z.; Wyatt, R.G.; Chanock, R.M. Reassortant rotaviruses as potential live rotavirus vaccine candidates. J. Virol. 1985, 53, 949–954. [Google Scholar] [CrossRef]
- Kapikian, A.Z.; Hoshino, Y.; Flores, J.; Midthun, K.; Glass, R.I.; Nakagomi, O.; Nakogomi, T.; Chanock, R.M.; Potash, L.; Levine, M.M.; et al. Alternative approaches to the development of a rotavirus vaccine. In Development of Vaccines and Drugs Against Diarrhoea: 11th Nobel Conference, Stockholm, 1985; Holmgren, J., Lindberg, A., Mollby, R., Eds.; Student Litteratur: Lund, Sweden, 1986; pp. 192–214. [Google Scholar]
- Vesikari, T.; Ruuska, T.; Green, K.Y.; Flores, J.; Kapikian, A.Z. Protective efficacy against serotype 1 rotavirus diarrhea by live oral rhesus-human reassortant rotavirus vaccines with human rotavirus VP7 serotype 1 or 2 specificity. Pediatr. Infect. Dis. J. 1992, 11, 535–542. [Google Scholar] [CrossRef]
- Flores, J.; Perez-Schael, I.; Blanco, M.; Vilar, M.; Garcia, D.; Perez, M.; Daoud, N.; Midthun, K.; Kapikian, A.Z. Reactions to and antigenicity of two human-rhesus rotavirus reassortant vaccine candidates of serotypes 1 and 2 in Venezuelan infants. J. Clin. Microbiol. 1989, 27, 512–518. [Google Scholar] [CrossRef]
- Perez-Schael, I.; Blanco, M.; Vilar, M.; Garcia, D.; White, L.; Gonzalez, R.; Kapikian, A.Z.; Flores, J. Clinical studies of a quadrivalent rotavirus vaccine in Venezuelan infants. J. Clin. Microbiol. 1990, 28, 553. [Google Scholar] [CrossRef]
- Uhnoo, I.; Riepenhoff-Talty, M.; Dharakul, T.; Chegas, P.; Fisher, J.E.; Greenberg, H.B.; Ogra, P.L. Extramucosal spread and development of hepatitis in immunodeficient and normal mice infected with rhesus rotavirus. J. Virol. 1990, 64, 361–368. [Google Scholar] [CrossRef]
- Clark, H.F.; Borian, F.E.; Modesto, K.; Plotkin, S.A. Serotype 1 reassortant of bovine rotavirus WC3, strain WI79-9, induces a polytypic antibody response in infants. Vaccine 1990, 8, 327–332. [Google Scholar] [CrossRef]
- Midthun, K.; Kapikian, A.Z. Rotavirus vaccines: An overview. Clin. Microbiol. Rev. 1996, 9, 423–434. [Google Scholar] [CrossRef]
- Santosham, M.; Letson, G.W.; Wolff, M.; Reid, R.; Gahagan, S.; Adams, R.; Callahan, C.; Sack, R.B.; Kapikian, A.Z. A field study of the safety and efficacy of two candidate rotavirus vaccines in a Native American population. J. Infect. Dis. 1991, 163, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Bishop, R. The present status of rotavirus vaccine development. Southeast. Asian J. Trop. Med. Public Health 1988, 19, 429–435. [Google Scholar] [PubMed]
- Ward, R.L.; Knowlton, D.R.; Zito, E.T.; Davidson, B.L.; Rappaport, R.; Mack, M.E. Serologic correlates of immunity in a tetravalent reassortant rotavirus vaccine trial. US Rotavirus Vaccine Efficacy Group. J. Infect. Dis. 1997, 176, 570–577. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murphy, B.R.; Morens, D.M.; Simonsen, L.; Chanock, R.M.; La Montagne, J.R.; Kapikian, A.Z. Reappraisal of the association of intussusception with the licensed live rotavirus vaccine challenges initial conclusions. J. Infect. Dis. 2003, 187, 1301–1308. [Google Scholar] [CrossRef]
- Clark, H.F.; Offit, P.A.; Ellis, R.W.; Eiden, J.J.; Krah, D.; Shaw, A.R.; Pichichero, M.; Treanor, J.J.; Borian, F.E.; Bell, L.M. The Development of Multivalent Bovine Rotavirus (Strain WC3) Reassortant. J. Infect. Dis. 1996, 174, S73–S80. [Google Scholar] [CrossRef]
- Payne, D.C.; Selvarangan, R.; Azimi, P.H.; Boom, J.A.; Englund, J.A.; Staat, M.A.; Halasa, N.B.; Weinberg, G.A.; Szilagyi, P.G.; Chappell, J. Long-term consistency in rotavirus vaccine protection: RV5 and RV1 vaccine effectiveness in US children, 2012–2013. Clin. Infect. Dis. 2015, 61, 1792–1799. [Google Scholar] [CrossRef]
- Ciarlet, M.; Schodel, F. Development of a rotavirus vaccine: Clinical safety, immunogenicity, and efficacy of the pentavalent rotavirus vaccine, RotaTeq®. Vaccine 2009, 27, G72–G81. [Google Scholar] [CrossRef]
- Vesikari, T.; Itzler, R.; Karvonen, A.; Korhonen, T.; Van Damme, P.; Behre, U.; Bona, G.; Gothefors, L.; Heaton, P.M.; Dallas, M. RotaTeq®, a pentavalent rotavirus vaccine: Efficacy and safety among infants in Europe. Vaccine 2009, 28, 345–351. [Google Scholar] [CrossRef]
- Velazquez, F.R.; Matson, D.O.; Calva, J.J.; Guerrero, L.; Morrow, A.L.; Carter-Campbell, S.; Glass, R.I.; Estes, M.K.; Pickering, L.K.; Ruiz-Palacios, G.M. Rotavirus infection in infants as protection against subsequent infections. N. Engl. J. Med. 1996, 335, 1022–1028. [Google Scholar] [CrossRef]
- Vesikari, T.; Matson, D.O.; Dennehy, P.; Van Damme, P.; Santosham, M.; Rodriguez, Z.; Dallas, M.J.; Heyse, J.F.; Goveia, M.G.; Black, S.B. Safety and efficacy of a pentavalent human–bovine (WC3) reassortant rotavirus vaccine. N. Engl. J. Med. 2006, 354, 23–33. [Google Scholar] [CrossRef]
- Vesikari, T.; Karvonen, A.; Prymula, R.; Schuster, V.; Tejedor, J.C.; Cohen, R.; Damaso, S.; Han, H.H.; De Vos, B.; Meurice, F. Human rotavirus vaccine RotarixTM (RIX4414) is highly efficacious in Europe. In Proceedings of the 24th Annual Meeting of the European Society for Paediatric Infectious Diseases (ESPID), Basel, Switzerland, 3–5 May 2006; p. 75. [Google Scholar]
- Kirkwood, C.D.; Ma, L.-F.; Carey, M.E.; Steele, A.D. The rotavirus vaccine development pipeline. Vaccine 2019, 37, 7328–7335. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; van Zandvoort, K.; Flasche, S.; Sanderson, C.; Bines, J.; Tate, J.; Parashar, U.; Jit, M. Efficacy of live oral rotavirus vaccines by duration of follow-up: A meta-regression of randomised controlled trials. Lancet Infect. Dis. 2019, 19, 717–727. [Google Scholar] [CrossRef]
- Ruiz-Palacios, G.M.; Perez-Schael, I.; Velazquez, F.R.; Abate, H.; Breuer, T.; Clemens, S.C.; Cheuvart, B.; Espinoza, F.; Gillard, P.; Innis, B.L.; et al. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N. Engl. J. Med. 2006, 354, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Vesikari, T.; Van Damme, P.; Giaquinto, C.; Dagan, R.; Guarino, A.; Szajewska, H.; Usonis, V. European Society for Paediatric Infectious Diseases consensus recommendations for rotavirus vaccination in Europe: Update 2014. Pediatr. Infect. Dis. J. 2015, 34, 635–643. [Google Scholar] [CrossRef]
- Payne, D.C.; Boom, J.A.; Staat, M.A.; Edwards, K.M.; Szilagyi, P.G.; Klein, E.J.; Selvarangan, R.; Azimi, P.H.; Harrison, C.; Moffatt, M. Effectiveness of pentavalent and monovalent rotavirus vaccines in concurrent use among US children <5 years of age, 2009–2011. Clin. Infect. Dis. 2013, 57, 13–20. [Google Scholar]
- Madhi, S.A.; Cunliffe, N.A.; Steele, D.; Witte, D.; Kirsten, M.; Louw, C.; Ngwira, B.; Victor, J.C.; Gillard, P.H.; Cheuvart, B.B. Research Article (New England Journal of Medicine) Effect of human rotavirus vaccine on severe diarrhea in African infants. Malawi Med. J. 2016, 28, 108–114. [Google Scholar]
- Zaman, K.; Sack, D.A.; Neuzil, K.M.; Yunus, M.; Moulton, L.H.; Sugimoto, J.D.; Fleming, J.A.; Hossain, I.; Arifeen, S.E.; Azim, T.; et al. Effectiveness of a live oral human rotavirus vaccine after programmatic introduction in Bangladesh: A cluster-randomized trial. PLoS Med. 2017, 14, e1002282. [Google Scholar] [CrossRef]
- Buttery, J.P.; Danchin, M.H.; Lee, K.J.; Carlin, J.B.; McIntyre, P.B.; Elliott, E.J.; Booy, R.; Bines, J.E.; Group, P.A.S. Intussusception following rotavirus vaccine administration: Post-marketing surveillance in the National Immunization Program in Australia. Vaccine 2011, 29, 3061–3066. [Google Scholar] [CrossRef]
- Patel, M.M.; López-Collada, V.R.; Bulhões, M.M.; De Oliveira, L.H.; Márquez, A.B.; Flannery, B.; Esparza-Aguilar, M.; Montenegro Renoiner, E.I.; Luna-Cruz, M.E.; Sato, H.K.; et al. Intussusception risk and health benefits of rotavirus vaccination in Mexico and Brazil. N. Engl. J. Med. 2019, 364, 2283–2292. [Google Scholar] [CrossRef]
- Vesikari, T.; Karvonen, A.; Ferrante, S.A.; Ciarlet, M. Efficacy of the pentavalent rotavirus vaccine, RotaTeq®, in Finnish infants up to 3 years of age: The Finnish Extension Study. Eur. J. Pediatr. 2010, 169, 1379–1386. [Google Scholar] [CrossRef]
- Vesikari, T.; Karvonen, A.; Prymula, R.; Schuster, V.; Tejedor, J.C.; Cohen, R.; Meurice, F.; Han, H.H.; Damaso, S.; Bouckenooghe, A. Efficacy of human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in European infants: Randomised, double-blind controlled study. Lancet 2007, 370, 1757–1763. [Google Scholar] [CrossRef]
- Armah, G.E.; Sow, S.O.; Breiman, R.F.; Dallas, M.J.; Tapia, M.D.; Feikin, D.R.; Binka, F.N.; Steele, A.D.; Laserson, K.F.; Ansah, N.A.; et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-Saharan Africa: A randomised, double-blind, placebo-controlled trial. Lancet 2010, 376, 606–614. [Google Scholar] [CrossRef]
- Bar-Zeev, N.; Jere, K.C.; Bennett, A.; Pollock, L.; Tate, J.E.; Nakagomi, O.; Iturriza-Gomara, M.; Costello, A.; Mwansambo, C.; Parashar, U.D.; et al. Population Impact and Effectiveness of Monovalent Rotavirus Vaccination in Urban Malawian Children 3 Years After Vaccine Introduction: Ecological and Case-Control Analyses. Clin. Infect. Dis. 2016, 62 (Suppl. 2), S213–S219. [Google Scholar] [CrossRef]
- Weldegebriel, G.; Mwenda, J.M.; Chakauya, J.; Daniel, F.; Masresha, B.; Parashar, U.D.; Tate, J.E. Impact of rotavirus vaccine on rotavirus diarrhoea in countries of East and Southern Africa. Vaccine 2018, 36, 7124–7130. [Google Scholar] [CrossRef]
- Mwenda, J.M.; Parashar, U.D.; Cohen, A.L.; Tate, J.E. Impact of rotavirus vaccines in Sub-Saharan African countries. Vaccine 2018, 36, 7119–7123. [Google Scholar] [CrossRef]
- Karafillakis, E.; Hassounah, S.; Atchison, C. Effectiveness and impact of rotavirus vaccines in Europe, 2006-2014. Vaccine 2015, 33, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- International Vaccine Access Center (IVAC). Map Gallery-Rotavirus; Johns Hopkins Bloomberg School of Public Health, Johns Hopkins University: Baltimore, MD, USA, 2019. [Google Scholar]
- Aliabadi, N.; Antoni, S.; Mwenda, J.M.; Weldegebriel, G.; Biey, J.N.M.; Cheikh, D.; Fahmy, K.; Teleb, N.; Ashmony, H.A.; Ahmed, H.; et al. Global impact of rotavirus vaccine introduction on rotavirus hospitalisations among children under 5 years of age, 2008–2016: Findings from the Global Rotavirus Surveillance Network. Lancet Glob. Health 2019, 7, E893–E903. [Google Scholar] [CrossRef]
- McQuade, E.T.R.; Platts-Mills, J.A. Monitoring the impact of rotavirus vaccines on a global scale. Lancet Glob. Health 2019, 7, e817–e818. [Google Scholar] [CrossRef]
- Pietsch, C.; Liebert, U.G. Rotavirus vaccine effectiveness in preventing hospitalizations due to gastroenteritis: A descriptive epidemiological study from Germany. Clin. Microbiol. Infect. 2019, 25, 102–106. [Google Scholar] [CrossRef]
- Lopman, B.A.; Curns, A.T.; Yen, C.; Parashar, U.D. Infant Rotavirus Vaccination May Provide Indirect Protection to Older Children and Adults in the United States. J. Infect. Dis. 2011, 204, 980–986. [Google Scholar] [CrossRef]
- Anderson, E.J.; Shippee, D.B.; Weinrobe, M.H.; Davila, M.D.; Katz, B.Z.; Reddy, S.; Cuyugan, M.G.; Lee, S.Y.; Simons, Y.M.; Yogev, R.; et al. Indirect protection of adults from rotavirus by pediatric rotavirus vaccination. Clin. Infect. Dis. 2013, 56, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Paulke-Korinek, M.; Kundi, M.; Rendi-Wagner, P.; de Martin, A.; Eder, G.; Schmidle-Loss, B.; Vecsei, A.; Kollaritsch, H. Herd immunity after two years of the universal mass vaccination program against rotavirus gastroenteritis in Austria. Vaccine 2011, 29, 2791–2796. [Google Scholar] [CrossRef] [PubMed]
- Sahakyan, G.; Grigoryan, S.; Wasley, A.; Mosina, L.; Sargsyan, S.; Asoyan, A.; Gevorgyan, Z.; Kocharyan, K.; Avagyan, T.; Lopman, B.; et al. Impact and Effectiveness of Monovalent Rotavirus Vaccine in Armenian Children. Clin. Infect. Dis. 2016, 62, S147–S154. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). The Global Vaccine Action Plan. (GVAP) 2011–2020; WHO: Geneva, Switzerland, 2013; pp. 1–77. [Google Scholar]
- World Health Organization (WHO); United Nation Children’s Fund (UNICEF). Progress Towards Global Immunisation Goals, 2018: Summary Presentation of Key Indicators; WHO: Geneva, Switzerland, 2019; pp. 1–29. [Google Scholar]
- World Health Organization (WHO); United Nation Children’s Fund (UNICEF). Change in vaccine vial monitor (VVM) assignment for GlaxoSmithKline Rotarix® Product; WHO: Geneva, Switzerland, 2017; pp. 1–3. [Google Scholar]
- Merck & Co. Inc. Merck Reaffirms Commitment to Increase Vaccine Production and Clarifies Rotavirus Vaccine Transition Plan with Gavi Countries; Merck & Co. Inc.: Keniwolrth, NJ, USA, 2018. [Google Scholar]
- Global Alliance for Vaccines and Immunizations (GAVI). The Vacine Alliance Risks and Assurance Report 2019; GAVI: Geneva, Switzerland, 2019. [Google Scholar]
- Ruuska, T.; Vesikari, T. Rotavirus disease in Finnish children: Use of numerical scores for clinical severity of diarrhoeal episodes. Scand. J. Infect. Dis. 1990, 22, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Flores, J.; Perez-Schael, I.; Gonzalez, M.; Garcia, D.; Perez, M.; Daoud, N.; Cunto, W.; Chanock, R.M.; Kapikian, A.Z. Protection against severe rotavirus diarrhoea by rhesus rotavirus vaccine in Venezuelan infants. Lancet 1987, 1, 882–884. [Google Scholar] [CrossRef]
- Clark, H.F.; Furukawa, T.; Bell, L.M.; Offit, P.A.; Perrella, P.A.; Plotkin, S.A. Immune response of infants and children to low-passage bovine rotavirus (strain WC3). Am. J. Dis. Child. 1986, 140, 350–356. [Google Scholar] [CrossRef]
- Li, J.-S.; Cao, B.; Gao, H.-C.; Lin, L.; Li, L.I.L.; Liu, N.; Duan, Z.-J. Faecal shedding of rotavirus vaccine in Chinese children after vaccination with Lanzhou lamb rotavirus vaccine. Sci. Rep. 2018, 8, 1–7. [Google Scholar] [CrossRef]
- Fu, C.; He, Q.; Xu, J.; Xie, H.; Ding, P.; Hu, W.; Dong, Z.; Liu, X.; Wang, M. Effectiveness of the Lanzhou lamb rotavirus vaccine against gastroenteritis among children. Vaccine 2012, 31, 154–158. [Google Scholar] [CrossRef]
- Armah, G.E.; Kapikian, A.Z.; Vesikari, T.; Cunliffe, N.; Jacobson, R.M.; Burlington, D.B.; Ruiz, L.P., Jr. Efficacy, immunogenicity, and safety of two doses of a tetravalent rotavirus vaccine RRV-TV in Ghana with the first dose administered during the neonatal period. J. Infect. Dis. 2013, 208, 423–431. [Google Scholar] [CrossRef]
- Vesikari, T.; Karvonen, A.; Forrest, B.D.; Hoshino, Y.; Chanock, R.M.; Kapikian, A.Z. Neonatal administration of rhesus rotavirus tetravalent vaccine. Pediatr. Infect. Dis. J. 2006, 25, 118–122. [Google Scholar] [CrossRef]
- Keating, G.M. Rotavirus vaccine (RotaTeq). Paediatr. Drugs 2006, 8, 197–202, discussion 203–194. [Google Scholar] [CrossRef] [PubMed]
- Hemming, M.; Vesikari, T. Vaccine-derived human-bovine double reassortant rotavirus in infants with acute gastroenteritis. Pediatr. Infect. Dis. J. 2012, 31, 992–994. [Google Scholar] [CrossRef] [PubMed]
- Markkula, J.; Hemming, M.; Vesikari, T. Detection of vaccine-derived rotavirus strains in nonimmunocompromised children up to 3–6 months after RotaTeq® vaccination. Pediatr. Infect. Dis. J. 2015, 34, 296–298. [Google Scholar] [CrossRef] [PubMed]
- Breiman, R.F.; Zaman, K.; Armah, G.; Sow, S.O.; Anh, D.D.; Victor, J.C.; Hille, D.; Ciarlet, M.; Neuzil, K.M. Analyses of health outcomes from the 5 sites participating in the Africa and Asia clinical efficacy trials of the oral pentavalent rotavirus vaccine. Vaccine 2012, 30 (Suppl. 1), A24–A29. [Google Scholar] [CrossRef] [PubMed]
- Vesikari, T.; Karvonen, A.V.; Majuri, J.; Zeng, S.-Q.; Pang, X.-L.; Kohberger, R.; Forrest, B.D.; Hoshino, Y.; Chanock, R.M.; Kapikian, A.Z. Safety, efficacy, and immunogenicity of 2 doses of bovine-human (UK) and Rhesus–Rhesus-Human rotavirus reassortant tetravalent vaccines in Finnish children. J. Infect. Dis. 2006, 194, 370–376. [Google Scholar] [CrossRef]
- Naik, S.P.; Zade, J.K.; Sabale, R.N.; Pisal, S.S.; Menon, R.; Bankar, S.G.; Gairola, S.; Dhere, R.M. Stability of heat stable, live attenuated Rotavirus vaccine (ROTASIIL®). Vaccine 2017, 35, 2962–2969. [Google Scholar] [CrossRef]
- Dennehy, P.H. Rotavirus vaccines: An overview. Clin. Microbiol. Rev. 2008, 21, 198–208. [Google Scholar] [CrossRef]
- Dennehy, P.H.; Brady, R.C.; Halperin, S.A.; Ward, R.L.; Alvey, J.C.; Fischer, F.H.; Innis, B.L.; Rathfon, H.; Schuind, A.; De Vos, B.; et al. Comparative evaluation of safety and immunogenicity of two dosages of an oral live attenuated human rotavirus vaccine. Pediatr. Infect. Dis. J. 2005, 24, 481–488. [Google Scholar] [CrossRef]
- O’Ryan, M. Rotarix™(RIX4414): An oral human rotavirus vaccine. Expert Rev. Vaccines 2007, 6, 11–19. [Google Scholar] [CrossRef]
- Tate, J.E.; Arora, R.; Kang, G.; Parashar, U.D. Rotavirus vaccines at the threshold of implementation in India. Natl. Med. J. India 2014, 27, 245–248. [Google Scholar]
- Danchin, M.; Kirkwood, C.D.; Lee, K.J.; Bishop, R.F.; Watts, E.; Justice, F.A.; Clifford, V.; Cowley, D.; Buttery, J.P.; Bines, J.E. Phase I trial of RV3-BB rotavirus vaccine: A human neonatal rotavirus vaccine. Vaccine 2013, 31, 2610–2616. [Google Scholar] [CrossRef] [PubMed]
- Bines, J.E.; Danchin, M.; Jackson, P.; Handley, A.; Watts, E.; Lee, K.J.; West, A.; Cowley, D.; Chen, M.Y.; Barnes, G.L.; et al. Safety and immunogenicity of RV3-BB human neonatal rotavirus vaccine administered at birth or in infancy: A randomised, double-blind, placebo-controlled trial. Lancet Infect. Dis. 2015, 15, 1389–1397. [Google Scholar] [CrossRef]
- Bines, J.E.; At Thobari, J.; Satria, C.D.; Handley, A.; Watts, E.; Cowley, D.; Nirwati, H.; Ackland, J.; Standish, J.; Justice, F.; et al. Human Neonatal Rotavirus Vaccine (RV3-BB) to Target Rotavirus from Birth. N. Engl. J. Med. 2018, 378, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Dennehy, P.H.; North American Human Rotavirus Vaccine Study Group. A short report on the highlights of world-wide development of RIX4414: A North American experience comparative evaluation of safety and immunogenicity of two dosages of an oral live attenuated human rotavirus vaccine (RIX4414) in infants in the United States and Canada. Vaccine 2006, 24, 3780–3781. [Google Scholar] [CrossRef] [PubMed]
- Paternina-Caicedo, A.; Parashar, U.D.; Alvis-Guzmán, N.; De Oliveira, L.H.; Castaño-Zuluaga, A.; Cotes-Cantillo, K.; Gamboa-Garay, O.; Coronell-Rodríguez, W.; De la Hoz-Restrepo, F. Effect of rotavirus vaccine on childhood diarrhea mortality in five Latin American countries. Vaccine 2015, 33, 3923–3928. [Google Scholar] [CrossRef]
- Patel, M.M.; Patzi, M.; Pastor, D.; Nina, A.; Roca, Y.; Alvarez, L.; Iniguez, V.; Rivera, R.; Tam, K.I.; Quaye, O. Effectiveness of monovalent rotavirus vaccine in Bolivia: Case-control study. BMJ 2013, 346, f3726. [Google Scholar] [CrossRef]
- De Oliveira, L.H.; Giglio, N.; Ciapponi, A.; Marti, S.G.; Kuperman, M.; Sanwogou, N.J.; Ruiz-Matus, C.; de Sousa, M.F.M. Temporal trends in diarrhea-related hospitalizations and deaths in children under age 5 before and after the introduction of the rotavirus vaccine in four Latin American countries. Vaccine 2013, 31, C99–C108. [Google Scholar] [CrossRef]
- Patel, M.; Pedreira, C.; De Oliveira, L.H.; Umana, J.; Tate, J.; Lopman, B.; Sanchez, E.; Reyes, M.; Mercado, J.; Gonzalez, A.; et al. Duration of protection of pentavalent rotavirus vaccination in Nicaragua. Pediatrics 2012, 130, e365–e372. [Google Scholar] [CrossRef]
- Cunliffe, N.A.; Witte, D.; Ngwira, B.M.; Todd, S.; Bostock, N.J.; Turner, A.M.; Chimpeni, P.; Victor, J.C.; Steele, A.D.; Bouckenooghe, A.; et al. Efficacy of human rotavirus vaccine against severe gastroenteritis in Malawian children in the first two years of life: A randomized, double-blind, placebo controlled trial. Vaccine 2012, 30 (Suppl. 1), A36–A43. [Google Scholar] [CrossRef]
- Steele, A.D.; Neuzil, K.M.; Cunliffe, N.A.; Madhi, S.A.; Bos, P.; Ngwira, B.; Witte, D.; Todd, S.; Louw, C.; Kirsten, M. Human rotavirus vaccine Rotarix™ provides protection against diverse circulating rotavirus strains in African infants: A randomized controlled trial. BMC Infect. Dis. 2012, 12, 213. [Google Scholar] [CrossRef]
- Burke, R.M.; Tate, J.E.; Kirkwood, C.D.; Steele, A.D.; Parashar, U.D. Current and new rotavirus vaccines. Curr. Opin. Infect. Dis. 2019, 32, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Isanaka, S.; Guindo, O.; Langendorf, C.; Matar Seck, A.; Plikaytis, B.D.; Sayinzoga-Makombe, N.; McNeal, M.M.; Meyer, N.; Adehossi, E.; Djibo, A.; et al. Efficacy of a Low-Cost, Heat-Stable Oral Rotavirus Vaccine in Niger. N. Engl. J. Med. 2017, 376, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, N.; Rongsen-Chandola, T.; Bavdekar, A.; John, J.; Antony, K.; Taneja, S.; Goyal, N.; Kawade, A.; Kang, G.; Rathore, S.S.; et al. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian children in the second year of life. Vaccine 2014, 32 (Suppl. 1), A110–A116. [Google Scholar] [CrossRef] [PubMed]
- Dadonaite, B.; Ritchie, H. Rotavirus vaccine – An effective tool that prevents children dying from diarrhea. In Our World in Data; Global Change Data Lab (GCDL): England, UK, 2019. [Google Scholar]
- Vesikari, T.; Karvonen, A.; Ferrante, S.A.; Kuter, B.J.; Ciarlet, M. Sustained Efficacy of the Pentavalent Rotavirus Vaccine, RV5, up to 3.1 Years Following the Last Dose of Vaccine. Pediatr. Infect. Dis. J. 2010, 29, 957–963. [Google Scholar] [CrossRef]
- Staat, M.A.; Payne, D.C.; Donauer, S.; Weinberg, G.A.; Edwards, K.M.; Szilagyi, P.G.; Griffin, M.R.; Hall, C.B.; Curns, A.T.; Gentsch, J.R. Effectiveness of pentavalent rotavirus vaccine against severe disease. Pediatrics 2011, 128, e267–e275. [Google Scholar] [CrossRef]
- Mast, T.C.; Khawaja, S.; Espinoza, F.; Paniagua, M.; Del Carmen, L.P.; Cardellino, A.; Sánchez, E. Case-control study of the effectiveness of vaccination with pentavalent rotavirus vaccine in Nicaragua. Pediatr. Infect. Dis. J. 2011, 30, e209–e215. [Google Scholar] [CrossRef]
- Ichihara, M.Y.; Rodrigues, L.C.; Teles Santos, C.A.; Teixeira Mda, G.; De Jesus, S.R.; Alvim De Matos, S.M.; Gagliardi Leite, J.P.; Barreto, M.L. Effectiveness of rotavirus vaccine against hospitalized rotavirus diarrhea: A case-control study. Vaccine 2014, 32, 2740–2747. [Google Scholar] [CrossRef]
- Burnett, E.; Lopman, B.; Parashar, U.D. Potential for a booster dose of rotavirus vaccine to further reduce diarrhea mortality. Vaccine 2017, 35, 7198–7203. [Google Scholar] [CrossRef]
- Hodgson, K.; Phuong, L.K.; Manley, B. Rotavirus vaccine for neonates. Acta Paediatr. 2019, 108, 774. [Google Scholar] [CrossRef]
- Steele, A.D.; Peenze, I.; De Beer, M.C.; Pager, C.T.; Yeats, J.; Potgieter, N.; Ramsaroop, U.; Page, N.A.; Mitchell, J.O.; Geyer, A. Anticipating rotavirus vaccines: Epidemiology and surveillance of rotavirus in South Africa. Vaccine 2003, 21, 354–360. [Google Scholar] [CrossRef]
- Madhi, S.A.; Kirsten, M.; Louw, C.; Bos, P.; Aspinall, S.; Bouckenooghe, A.; Neuzil, K.M.; Steele, A.D. Efficacy and immunogenicity of two or three dose rotavirus-vaccine regimen in South African children over two consecutive rotavirus-seasons: A randomized, double-blind, placebo-controlled trial. Vaccine 2012, 30 (Suppl. 1), A44–A51. [Google Scholar] [CrossRef]
- Clarke, E.; Desselberger, U. Correlates of protection against human rotavirus disease and the factors influencing protection in low-income settings. Mucosal. Immunol. 2015, 8, 1–17. [Google Scholar] [CrossRef]
- Lopman, B.A.; Pitzer, V.E.; Sarkar, R.; Gladstone, B.; Patel, M.; Glasser, J.; Gambhir, M.; Atchison, C.; Grenfell, B.T.; Edmunds, W.J.; et al. Understanding reduced rotavirus vaccine efficacy in low socio-economic settings. PLoS ONE 2012, 7, e41720. [Google Scholar] [CrossRef] [PubMed]
- Superti, F.; Ammendolia, M.G.; Valenti, P.; Seganti, L. Antirotaviral activity of milk proteins: Lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29. Med. Microbiol. Immunol. 1997, 186, 83–91. [Google Scholar] [CrossRef]
- Newburg, D.S.; Peterson, J.A.; Ruiz-Palacios, G.M.; Matson, D.O.; Morrow, A.L.; Shults, J.; de Lourdes Guerrero, M.; Chaturvedi, P.; Newburg, S.O.; Scallan, C.D. Role of human-milk lactadherin in protectoin against symptomatic rotavirus infection. Lancet 1998, 351, 1160–1164. [Google Scholar] [CrossRef]
- Yolken, R.H.; Peterson, J.A.; Vonderfecht, S.L.; Fouts, E.T.; Midthun, K.; Newburg, D.S. Human milk mucin inhibits rotavirus replication and prevents experimental gastroenteritis. J. Clin. Investig. 1992, 90, 1984–1991. [Google Scholar] [CrossRef]
- Moon, S.S.; Wang, Y.; Shane, A.L.; Nguyen, T.; Ray, P.; Dennehy, P.; Baek, L.J.; Parashar, U.; Glass, R.I.; Jiang, B. Inhibitory effect of breast milk on infectivity of live oral rotavirus vaccines. Pediatr. Infect. Dis. J. 2010, 29, 919–923. [Google Scholar] [CrossRef]
- Moon, S.S.; Tate, J.E.; Ray, P.; Dennehy, P.H.; Archary, D.; Coutsoudis, A.; Bland, R.; Newell, M.L.; Glass, R.I.; Parashar, U.; et al. Differential profiles and inhibitory effect on rotavirus vaccines of nonantibody components in breast milk from mothers in developing and developed countries. Pediatr. Infect. Dis. J. 2013, 32, 863–870. [Google Scholar] [CrossRef]
- Groome, M.J.; Moon, S.S.; Velasquez, D.; Jones, S.; Koen, A.; van Niekerk, N.; Jiang, B.; Parashar, U.D.; Madhi, S.A. Effect of breastfeeding on immunogenicity of oral live-attenuated human rotavirus vaccine: A randomized trial in HIV-uninfected infants in Soweto, South Africa. Bull. World Health Organ. 2014, 92, 238–245. [Google Scholar] [CrossRef]
- Ali, A.; Kazi, A.M.; Cortese, M.M.; Fleming, J.A.; Moon, S.; Parashar, U.D.; Jiang, B.; McNeal, M.M.; Steele, D.; Bhutta, Z. Impact of withholding breastfeeding at the time of vaccination on the immunogenicity of oral rotavirus vaccine—A randomized trial. PLoS ONE 2015, 10, e0127622. [Google Scholar]
- Rongsen-Chandola, T.; Strand, T.A.; Goyal, N.; Flem, E.; Rathore, S.S.; Arya, A.; Winje, B.A.; Lazarus, R.; Shanmugasundaram, E.; Babji, S.; et al. Effect of withholding breastfeeding on the immune response to a live oral rotavirus vaccine in North Indian infants. Vaccine 2014, 32 (Suppl. 1), A134–A139. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.S.; Kazi, M.; Cortese, M.; Fleming, J.; Parahar, U.; Jiang, B. Impact of with-holding breastfeeding around the time of vaccination on the immunogenicity of the human rotavirus vaccine in Pakistan—A randomized controlled trial. Vaccines Enteric Dis. 2013, 11, 6–8. [Google Scholar]
- Gastañaduy, P.A.; Steenhoff, A.P.; Mokomane, M.; Esona, M.D.; Bowen, M.D.; Jibril, H.; Pernica, J.M.; Mazhani, L.; Smieja, M.; Tate, J.E. Effectiveness of monovalent rotavirus vaccine after programmatic implementation in Botswana: A multisite prospective case-control study. Clin. Infect. Dis. 2016, 62, S161–S167. [Google Scholar] [CrossRef] [PubMed]
- Goveia, M.G.; DiNubile, M.J.; Dallas, M.J.; Heaton, P.M.; Kuter, B.J.; Team, R.S. Efficacy of pentavalent human-bovine (WC3) reassortant rotavirus vaccine based on breastfeeding frequency. Pediatr. Infect. Dis. J. 2008, 27, 656–658. [Google Scholar] [CrossRef] [PubMed]
- Rennels, M.B.; Wasserman, S.S.; Glass, R.I.; Keane, V.A. Comparison of immunogenicity and efficacy of rhesus rotavirus reassortant vaccines in breastfed and nonbreastfed children. US Rotavirus Vaccine Efficacy Group. Pediatrics 1995, 96, 1132–1136. [Google Scholar] [PubMed]
- Pichichero, M.E. Effect of breast-feeding on oral rhesus rotavirus vaccine seroconversion: A metaanalysis. J. Infect. Dis. 1990, 162, 753–755. [Google Scholar] [CrossRef] [PubMed]
- Appaiahgari, M.B.; Glass, R.; Singh, S.; Taneja, S.; Rongsen-Chandola, T.; Bhandari, N.; Mishra, S.; Vrati, S. Transplacental rotavirus IgG interferes with immune response to live oral rotavirus vaccine ORV-116E in Indian infants. Vaccine 2014, 32, 651–656. [Google Scholar] [CrossRef]
- Brussow, H.; Sidoti, J.; Lerner, L.; Rahim, H.; Eckstein, W.; Werchau, H.; Mietens, C. Antibodies to seven rotavirus serotypes in cord sera, maternal sera, and colostrum of German women. J. Clin. Microbiol. 1991, 29, 2856–2859. [Google Scholar] [CrossRef]
- McLean, B.; Holmes, I.H. Transfer of antirotaviral antibodies from mothers to their infants. J. Clin. Microbiol. 1980, 12, 320. [Google Scholar] [CrossRef]
- Thouless, M.E.; Bryden, A.S.; Flewett, T.H. Rotavirus neutralisation by human milk. Br. Med. J. 1977, 2, 1390. [Google Scholar] [CrossRef]
- Totterdell, B.M.; Banatvala, J.E.; Chrystie, I.L. Studies on human lacteal rotavirus antibodies by immune electron microscopy. J. Med. Virol. 1983, 11, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Ringenbergs, M.; Albert, M.J.; Davidson, G.P.; Goldsworthy, W.; Haslam, R. Serotype-specific antibodies to rotavirus in human colostrum and breast milk and in maternal and cord blood. J. Infect. Dis. 1988, 158, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.G.; Kelkar, S.D. Prevalence of Neutralizing Antibodies against Different Rotavirus Serotypes in Children with Severe Rotavirus-Induced Diarrhea and Their Mothers. Clin. Diagn. Lab. Immunol. 2004, 11, 186. [Google Scholar] [CrossRef][Green Version]
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 2007, 449, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Bik, E.M.; Costello, E.K.; Dethlefsen, L.; Haque, R.; Relman, D.A.; Singh, U. Distinct distal gut microbiome diversity and composition in healthy children from Bangladesh and the United States. PLoS ONE 2013, 8, e53838. [Google Scholar] [CrossRef]
- Praharaj, I.; John, S.M.; Bandyopadhyay, R.; Kang, G. Probiotics, antibiotics and the immune responses to vaccines. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140144. [Google Scholar] [CrossRef]
- Kamada, N.; Seo, S.U.; Chen, G.Y.; Nunez, G. Role of the gut microbiota in immunity and inflammatory disease. Nat. Rev. Immunol. 2013, 13, 321–335. [Google Scholar] [CrossRef]
- Twitchell, E.L.; Tin, C.; Wen, K.; Zhang, H.; Becker-Dreps, S.; Azcarate-Peril, M.A.; Vilchez, S.; Li, G.; Ramesh, A.; Weiss, M.; et al. Modeling human enteric dysbiosis and rotavirus immunity in gnotobiotic pigs. Gut Pathog. 2016, 8, 51. [Google Scholar] [CrossRef]
- Vlasova, A.N.; Chattha, K.S.; Kandasamy, S.; Liu, Z.; Esseili, M.; Shao, L.; Rajashekara, G.; Saif, L.J. Lactobacilli and bifidobacteria promote immune homeostasis by modulating innate immune responses to human rotavirus in neonatal gnotobiotic pigs. PLoS ONE 2013, 8, e76962. [Google Scholar] [CrossRef]
- Liu, F.; Li, G.; Wen, K.; Wu, S.; Zhang, Y.; Bui, T.; Yang, X.; Kocher, J.; Sun, J.; Jortner, B.; et al. Lactobacillus rhamnosus GG on rotavirus-induced injury of ileal epithelium in gnotobiotic pigs. J. Pediatr. Gastroenterol. Nutr. 2013, 57, 750–758. [Google Scholar] [CrossRef]
- Kandasamy, S.; Chattha, K.S.; Vlasova, A.N.; Rajashekara, G.; Saif, L.J. Lactobacilli and Bifidobacteria enhance mucosal B cell responses and differentially modulate systemic antibody responses to an oral human rotavirus vaccine in a neonatal gnotobiotic pig disease model. Gut Microbes 2014, 5, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Gu, C.; Hu, H.; Tang, J.; Chen, D.; Yu, B.; He, J.; Yu, J.; Luo, J.; Tian, G. Dietary Lactobacillus rhamnosus GG supplementation improves the mucosal barrier function in the intestine of weaned piglets challenged by porcine rotavirus. PLoS ONE 2016, 11, e0146312. [Google Scholar] [CrossRef] [PubMed]
- Parker, E.P.K.; Praharaj, I.; Zekavati, A.; Lazarus, R.P.; Giri, S.; Operario, D.J.; Liu, J.; Houpt, E.; Iturriza-Gómara, M.; Kampmann, B.; et al. Influence of the intestinal microbiota on the immunogenicity of oral rotavirus vaccine given to infants in south India. Vaccine 2018, 36, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, R.P.; John, J.; Shanmugasundaram, E.; Rajan, A.K.; Thiagarajan, S.; Giri, S.; Babji, S.; Sarkar, R.; Kaliappan, P.S.; Venugopal, S.; et al. The effect of probiotics and zinc supplementation on the immune response to oral rotavirus vaccine: A randomized, factorial design, placebo-controlled study among Indian infants. Vaccine 2018, 36, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Narang, A.; Bose, A.; Pandit, A.N.; Dutta, P.; Kang, G.; Bhattacharya, S.K.; Datta, S.; Delem, A.; Han, H.H.; Bock, H.L. Immunogenicity, reactogenicity and safety of human rotavirus vaccine (RIX4414) in Indian infants. Hum. Vaccines 2009, 5, 414–419. [Google Scholar] [CrossRef]
- Hoest, C.; Seidman, J.C.; Pan, W.; Ambikapathi, R.; Kang, G.; Kosek, M.; Knobler, S.; Mason, C.J.; Miller, M.; Investigators, M.-E.N. Evaluating associations between vaccine response and malnutrition, gut function, and enteric infections in the MAL-ED cohort study: Methods and challenges. Clin. Infect. Dis 2014, 59 (Suppl. 4), S273–S279. [Google Scholar] [CrossRef]
- Prendergast, A.J. Malnutrition and vaccination in developing countries. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140141. [Google Scholar] [CrossRef]
- Khagayi, S.; Omore, R.; Otieno, G.; Ogwel, B.; Ochieng, J.; Juma, J.; Apondi, E.; Bigogo, G.; Onyango, C.; Ngama, M.; et al. Effectiveness of monovalent rotavirus vaccine against hospitalization with acute rotavirus gastroenteritis in Kenyan children. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2019. [Google Scholar] [CrossRef]
- Mujuru, H.A.; Burnett, E.; Nathoo, K.J.; Ticklay, I.; Gonah, N.A.; Mukaratirwa, A.; Berejena, C.; Manangazira, P.; Rupfutse, M.; Weldegebriel, G.G. Monovalent rotavirus vaccine effectiveness against rotavirus hospitalizations among children in Zimbabwe. Clin. Infect. Dis. 2018, 69, 1339–1344. [Google Scholar] [CrossRef]
- Linhares, A.C.; Carmo, K.B.d.; Oliveira, K.K.; Oliveira, C.S.; Freitas, R.B.d.; Bellesi, N.; Monteiro, T.A.F.; Gabbay, Y.B.; Mascarenhas, J.D. Nutritional status in relation to the efficacy of the rhesus-human reassortant, tetravalent rotavirus vaccine (RRV-TV) in infants from Belem, para state, Brazil. Revista do Instituto de Medicina Tropical de São Paulo 2002, 44, 13–16. [Google Scholar] [CrossRef]
- Perez-Schael, I.; Salinas, B.; Tomat, M.; Linhares, A.C.; Guerrero, M.L.; Ruiz-Palacios, G.M.; Bouckenooghe, A.; Yarzábal, J.P. Efficacy of the human rotavirus vaccine RIX4414 in malnourished children. J. Infect. Dis. 2007, 196, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.D.; Groome, M.J. Measuring Rotavirus Vaccine Impact in Sub-Saharan Africa. Clin. Infect. Dis. 2019, 10.1093/cid/ciz918. [Google Scholar] [CrossRef] [PubMed]
- Taniuchi, M.; Platts-Mills, J.A.; Begum, S.; Uddin, M.J.; Sobuz, S.U.; Liu, J.; Kirkpatrick, B.D.; Colgate, E.R.; Carmolli, M.P.; Dickson, D.M.; et al. Impact of enterovirus and other enteric pathogens on oral polio and rotavirus vaccine performance in Bangladeshi infants. Vaccine 2016, 34, 3068–3075. [Google Scholar] [CrossRef] [PubMed]
- Bhavnani, D.; Goldstick, J.E.; Cevallos, W.; Trueba, G.; Eisenberg, J.N.S. Synergistic effects between rotavirus and coinfecting pathogens on diarrheal disease: Evidence from a community-based study in northwestern Ecuador. Am. J. Epidemiol. 2012, 176, 387–395. [Google Scholar] [CrossRef]
- Hung, T.Y.; Liu, M.C.; Hsu, C.F.; Lin, Y.C. Rotavirus infection increases the risk of bacteremia in children with nontyphoid Salmonella gastroenteritis. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 425–428. [Google Scholar] [CrossRef]
- Lee, W.T.; Lin, P.C.; Lin, L.C.; Chen, H.L.; Yang, R.C. Salmonella/rotavirus coinfection in hospitalized children. Kaohsiung J. Med. Sci. 2012, 28, 595–600. [Google Scholar] [CrossRef]
- Valentini, D.; Vittucci, A.C.; Grandin, A.; Tozzi, A.E.; Russo, C.; Onori, M.; Menichella, D.; Bartuli, A.; Villani, A. Coinfection in acute gastroenteritis predicts a more severe clinical course in children. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 909–915. [Google Scholar] [CrossRef]
- Patel, M.; Pedreira, C.; De Oliveira, L.H.; Tate, J.; Orozco, M.; Mercado, J.; Gonzalez, A.; Malespin, O.; Amador, J.J.; Umana, J.; et al. Association Between Pentavalent Rotavirus Vaccine and Severe Rotavirus Diarrhea Among Children in Nicaragua. JAMA 2009, 301, 2243–2251. [Google Scholar] [CrossRef]
- Pringle, K.D.; Patzi, M.; Tate, J.E.; Iniguez Rojas, V.; Patel, M.; Inchauste Jordan, L.; Montesano, R.; Zarate, A.; De Oliveira, L.; Parashar, U. Sustained effectiveness of rotavirus vaccine against very severe rotavirus disease through the second year of life, Bolivia 2013–2014. Clin. Infect. Dis. 2016, 62, S115–S120. [Google Scholar] [CrossRef]
- Boom, J.A.; Tate, J.E.; Sahni, L.C.; Rench, M.A.; Quaye, O.; Mijatovic-Rustempasic, S.; Patel, M.M.; Baker, C.J.; Parashar, U.D. Sustained Protection from Pentavalent Rotavirus Vaccination during the Second Year of Life at a Large, Urban United States Pediatric Hospital. Pediatr. Infect. Dis. J. 2010, 29, 1133–1135. [Google Scholar] [CrossRef]
- Correia, J.B.; Patel, M.M.; Nakagomi, O.; Montenegro, F.M.; Germano, E.M.; Correia, N.B.; Cuevas, L.E.; Parashar, U.D.; Cunliffe, N.A.; Nakagomi, T. Effectiveness of monovalent rotavirus vaccine (Rotarix) against severe diarrhea caused by serotypically unrelated G2P[4] strains in Brazil. J. Infect. Dis. 2010, 201, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Gheorghita, S.; Birca, L.; Donos, A.; Wasley, A.; Birca, I.; Cojocaru, R.; Melnick, A.; Ciobanu, S.; Mosina, L.; Cortese, M.M.; et al. Impact of Rotavirus Vaccine Introduction and Vaccine Effectiveness in the Republic of Moldova. Clin. Infect. Dis. 2016, 62 (Suppl. 2), S140–S146. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; Beristain, X.; Martinez-Artola, V.; Navascues, A.; Cenoz, M.G.; Alvarez, N.; Polo, I.; Mazon, A.; Gil-Setas, A.; Barricarte, A. Effectiveness of rotavirus vaccines in preventing cases and hospitalizations due to rotavirus gastroenteritis in Navarre, Spain. Vaccine 2012, 30, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.M.; Immergluck, L.C.; Held, M.; Jain, S.; Chan, T.; Grizas, A.P.; Khizer, S.; Barrett, C.; Quaye, O.; Mijatovic-Rustempasic, S.; et al. Effectiveness of monovalent and pentavalent rotavirus vaccine. Pediatrics 2013, 132, e25–e33. [Google Scholar] [CrossRef]
- de Palma, O.; Cruz, L.; Ramos, H.; de Baires, A.; Villatoro, N.; Pastor, D.; de Oliveira, L.H.; Kerin, T.; Bowen, M.; Gentsch, J.; et al. Effectiveness of rotavirus vaccination against childhood diarrhoea in El Salvador: Case-control study. BMJ 2010, 340, c2825. [Google Scholar] [CrossRef]
- Cotes-Cantillo, K.; Paternina-Caicedo, A.; Coronell-Rodriguez, W.; Alvis-Guzman, N.; Parashar, U.D.; Patel, M.; De la Hoz-Restrepo, F. Effectiveness of the monovalent rotavirus vaccine in Colombia: A case-control study. Vaccine 2014, 32, 3035–3040. [Google Scholar] [CrossRef]
- Leshem, E.; Givon-Lavi, N.; Tate, J.E.; Greenberg, D.; Parashar, U.D.; Dagan, R. Real-world effectiveness of pentavalent rotavirus vaccine among Bedouin and Jewish children in southern Israel. Clin. Infect. Dis. 2016, 62, S155–S160. [Google Scholar] [CrossRef]
- Bonkoungou, I.J.O.; Aliabadi, N.; Leshem, E.; Kam, M.; Nezien, D.; Drabo, M.K.; Nikiema, M.; Ouedraogo, B.; Medah, I.; Konate, S.; et al. Impact and effectiveness of pentavalent rotavirus vaccine in children <5 years of age in Burkina Faso. Vaccine 2018, 36, 7170–7178. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Rotavirus vaccines: An update. Weekly Epidemiological Record Relevé Épidémiologique Hebdomadaire 2009, 84, 533–537. [Google Scholar]
- Patel, M.M.; Clark, A.D.; Sanderson, C.F.B.; Tate, J.; Parashar, U.D. Removing the age restrictions for rotavirus vaccination: A benefit-risk modeling analysis. PLoS Med. 2012, 9, e1001330. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Meeting of the Strategic Advisory Group of Experts on Immunization, April 2012—conclusions and recommendations. Weekly Epidemiological Record Relevé Épidémiologique Hebdomadaire 2012, 87, 201–216. [Google Scholar]
- Paulke-Korinek, M.; Kollaritsch, H.; Aberle, S.W.; Zwazl, I.; Schmidle-Loss, B.; Vécsei, A.; Kundi, M. Sustained low hospitalization rates after four years of rotavirus mass vaccination in Austria. Vaccine 2013, 31, 2686–2691. [Google Scholar] [CrossRef] [PubMed]
- Zlamy, M.; Kofler, S.; Orth, D.; Würzner, R.; Heinz-Erian, P.; Streng, A.; Prelog, M. The impact of Rotavirus mass vaccination on hospitalization rates, nosocomial Rotavirus gastroenteritis and secondary blood stream infections. BMC Infect. Dis. 2013, 13, 112. [Google Scholar] [CrossRef] [PubMed]
- Vesikari, T.; Karvonen, A.; Korhonen, T.; Espo, M.; Lebacq, E.; Forster, J.; Zepp, F.; Delem, A.; De Vos, B. Safety and immunogenicity of RIX4414 live attenuated human rotavirus vaccine in adults, toddlers and previously uninfected infants. Vaccine 2004, 22, 2836–2842. [Google Scholar] [CrossRef]
- Vesikari, T. Neonatal rotavirus vaccination making headway. Lancet Infect. Dis. 2015, 15, 1362–1363. [Google Scholar] [CrossRef]
- Ruuska, T.; Vesikari, T.; Delem, A.; André, F.E.; Beards, G.M.; Flewett, T.H. Evaluation of RIT 4237 bovine rotavirus vaccine in newborn infants: Correlation of vaccine efficacy to season of birth in relation to rotavirus epidemic period. Scand. J. Infect. Dis. 1990, 22, 269–278. [Google Scholar] [CrossRef]
- Vesikari, T.; Isolauri, E.; Delem, A.; Dhondt, E.; Andre, F.E.; Beards, G.M.; Flewett, T.H. Clinical Efficacy of the Rit-4237 Live Attenuated Bovine Rotavirus Vaccine in Infants Vaccinated before a Rotavirus Epidemic. J. Pediatr. 1985, 107, 189–194. [Google Scholar] [CrossRef]
- Delem, A.; Vesikari, T. Detection of serum antibody responses to RIT 4237 rotavirus vaccine by ELISA and neutralization assays. J. Med. Virol. 1987, 21, 231–238. [Google Scholar] [CrossRef]
- Simon, A.K.; Hollander, G.A.; McMichael, A. Evolution of the immune system in humans from infancy to old age. Proc. Biol. Sci. 2015, 282, 20143085. [Google Scholar] [CrossRef]
- Lagos, R.; Fasano, A.; Wasserman, S.S.; Prado, V.; Martin, O.S.; Abrego, P.; Losonsky, G.A.; Alegria, S.; Levine, M.M. Effect of Small Bowel Bacterial Overgrowth on the Immunogenicity of Single-Dose Live Oral Cholera Vaccine CVD 103-HgR. J. Infect. Dis. 1999, 180, 1709–1712. [Google Scholar] [CrossRef][Green Version]
- Simon, G.L.; Gorbach, S.L. Intestinal flora in health and disease. Gastroenterology 1984, 86, 174–193. [Google Scholar] [CrossRef]
- Fagundes-Neto, U.; Viaro, T.; Wehba, J.; Patricio, F.R.; Machado, N.L. Tropical enteropathy (environmental enteropathy) in early childhood: A syndrome caused by contaminated environment. J. Trop. Pediatr. 1984, 30, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Neto, U.F.; Martins, M.C.; Lima, F.L.; Patricio, F.R.; Toledo, M.R. Asymptomatic environmental enteropathy among slum-dwelling infants. J. Am. Coll. Nutr. 1994, 13, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Korpe, P.S.; Petri, W.A., Jr. Environmental enteropathy: Critical implications of a poorly understood condition. Trends Mol. Med. 2012, 18, 328–336. [Google Scholar] [CrossRef]
- Naylor, C.; Lu, M.; Haque, R.; Mondal, D.; Buonomo, E.; Nayak, U.; Mychaleckyj, J.C.; Kirkpatrick, B.; Colgate, R.; Carmolli, M.; et al. Environmental Enteropathy, Oral Vaccine Failure and Growth Faltering in Infants in Bangladesh. EBioMedicine 2015, 2, 1759–1766. [Google Scholar] [CrossRef]
- D’Haens, G.; Ferrante, M.; Vermeire, S.; Baert, F.; Noman, M.; Moortgat, L.; Geens, P.; Iwens, D.; Aerden, I.; Van Assche, G.; et al. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflamm. Bowel Dis. 2012, 18, 2218–2224. [Google Scholar] [CrossRef]
- Kosek, M.; Haque, R.; Lima, A.; Babji, S.; Shrestha, S.; Qureshi, S.; Amidou, S.; Mduma, E.; Lee, G.; Yori, P.P. Fecal markers of intestinal inflammation and permeability associated with the subsequent acquisition of linear growth deficits in infants. Am. J. Trop. Med. Hygiene 2013, 88, 390–396. [Google Scholar] [CrossRef]
- Campbell, D.I.; Murch, S.H.; Elia, M.; Sullivan, P.B.; Sanyang, M.S.; Jobarteh, B.; Lunn, P.G. Chronic T cell-mediated enteropathy in rural west African children: Relationship with nutritional status and small bowel function. Pediatr. Res. 2003, 54, 306–311. [Google Scholar] [CrossRef]
- Campbell, D.I.; Elia, M.; Lunn, P.G. Growth faltering in rural Gambian infants is associated with impaired small intestinal barrier function, leading to endotoxemia and systemic inflammation. J. Nutr. 2003, 133, 1332–1338. [Google Scholar] [CrossRef]
- Becker-Dreps, S.; Paniagua, M.; Dominik, R.; Cao, H.; Shah, N.K.; Morgan, D.R.; Moreno, G.; Espinoza, F. Changes in childhood diarrhea incidence in nicaragua following 3 years of universal infant rotavirus immunization. Pediatr. Infect. Dis. J. 2011, 30, 243–247. [Google Scholar] [CrossRef]
- Becker, D.J.; Lowe, J.B. Fucose: Biosynthesis and biological function in mammals. Glycobiology 2003, 13, 41R–53R. [Google Scholar] [CrossRef]
- Sun, X.; Guo, N.; Li, D.; Jin, M.; Zhou, Y.; Xie, G.; Pang, L.; Zhang, Q.; Cao, Y.; Duan, Z.-J. Binding specificity of P[8] VP8* proteins of rotavirus vaccine strains with histo-blood group antigens. Virology 2016, 495, 129–135. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, Y.; Tan, M. Histo-blood group antigens as receptors for rotavirus, new understanding on rotavirus epidemiology and vaccine strategy. Emerg. Microbes Infect. 2017, 6, e22. [Google Scholar] [CrossRef]
- Hu, L.; Crawford, S.E.; Czako, R.; Cortes-Penfield, N.W.; Smith, D.F.; Le Pendu, J.; Estes, M.K.; Prasad, B.V. Cell attachment protein VP8* of a human rotavirus specifically interacts with A-type histo-blood group antigen. Nature 2012, 485, 256–259. [Google Scholar] [CrossRef]
- Huang, P.; Xia, M.; Tan, M.; Zhong, W.; Wei, C.; Wang, L.; Morrow, A.; Jiang, X. Spike protein VP8* of human rotavirus recognizes histo-blood group antigens in a type-specific manner. J. Virol. 2012, 86, 4833–4843. [Google Scholar] [CrossRef]
- Liu, Y.; Ramelot, T.A.; Huang, P.; Liu, Y.; Li, Z.; Feizi, T.; Zhong, W.; Wu, F.-T.; Tan, M.; Kennedy, M.A.; et al. Glycan Specificity of P[19] Rotavirus and Comparison with Those of Related P Genotypes. J. Virol. 2016, 90, 9983. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, P.; Tan, M.; Liu, Y.; Biesiada, J.; Meller, J.; Castello, A.A.; Jiang, B.; Jiang, X. Rotavirus VP8*: Phylogeny, Host Range, and Interaction with Histo-Blood Group Antigens. J. Virol. 2012, 86, 9899. [Google Scholar] [CrossRef]
- Nordgren, J.; Sharma, S.; Bucardo, F.; Nasir, W.; Gunaydin, G.; Ouermi, D.; Nitiema, L.W.; Becker-Dreps, S.; Simpore, J.; Hammarstrom, L.; et al. Both Lewis and secretor status mediate susceptibility to rotavirus infections in a rotavirus genotype-dependent manner. Clin. Infect. Dis. 2014, 59, 1567–1573. [Google Scholar] [CrossRef]
- Kambhampati, A.; Payne, D.C.; Costantini, V.; Lopman, B.A. Host Genetic Susceptibility to Enteric Viruses: A Systematic Review and Metaanalysis. Clin. Infect. Dis. 2015, 62, 11–18. [Google Scholar] [CrossRef]
- Lee, B.; Dickson, D.M.; deCamp, A.C.; Ross Colgate, E.; Diehl, S.A.; Uddin, M.I.; Sharmin, S.; Islam, S.; Bhuiyan, T.R.; Alam, M.; et al. Histo–Blood Group Antigen Phenotype Determines Susceptibility to Genotype-Specific Rotavirus Infections and Impacts Measures of Rotavirus Vaccine Efficacy. J. Infect. Dis. 2018, 217, 1399–1407. [Google Scholar] [CrossRef]
- Kazi, A.M.; Cortese, M.M.; Yu, Y.; Lopman, B.; Morrow, A.L.; Fleming, J.A.; McNeal, M.M.; Steele, A.D.; Parashar, U.D.; Zaidi, A.K.M.; et al. Secretor and Salivary ABO Blood Group Antigen Status Predict Rotavirus Vaccine Take in Infants. J. Infect. Dis. 2017, 215, 786–789. [Google Scholar] [CrossRef]
- Patel, M.; Steele, A.D.; Parashar, U.D. Influence of oral polio vaccines on performance of the monovalent and pentavalent rotavirus vaccines. Vaccine 2012, 30 (Suppl. 1), A30–A35. [Google Scholar] [CrossRef]
- Emperador, D.M.; Velasquez, D.E.; Estivariz, C.F.; Lopman, B.; Jiang, B.; Parashar, U.; Anand, A.; Zaman, K. Interference of Monovalent, Bivalent, and Trivalent Oral Poliovirus Vaccines on Monovalent Rotavirus Vaccine Immunogenicity in Rural Bangladesh. Clin. Infect. Dis. 2016, 62, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Ciarlet, M.; Sani-Grosso, R.; Yuan, G.; Liu, G.F.; Heaton, P.M.; Gottesdiener, K.M.; Arredondo, J.L.; Schodel, F. Concomitant use of the oral pentavalent human-bovine reassortant rotavirus vaccine and oral poliovirus vaccine. Pediatr. Infect. Dis. J. 2008, 27, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Ciarlet, M.; Sani-Grosso, R.; Yuan, G.; Liu, G.; Liu, P.M.; Gottesdiener, K.M. Safety and immunogenicity of concomitant use of the pentavalent rotavirus vaccine (RotaTeq) and oral poliovirus vaccine (OPV). In Proceedings of the 25th Annual Meeting of the European Society for Paediatric Infectious Diseases, Porto, Portugal, 2–4 May 2007. [Google Scholar]
- Cortese, M.M.; Parashar, U.D. Prevention of rotavirus gastroenteritis among infants and children: Recommendations of the Advisory Committee on Immunization Practices (ACIP). Morb. Mortal. Weekly Rep. Recomm. Rep. 2009, 58, 1–25. [Google Scholar]
- Zaman, K.; Sack, D.A.; Yunus, M.; Arifeen, S.E.; Podder, G.; Azim, T.; Luby, S.; Breiman, R.F.; Neuzil, K.; Datta, S.K. Successful co-administration of a human rotavirus and oral poliovirus vaccines in Bangladeshi infants in a 2-dose schedule at 12 and 16 weeks of age. Vaccine 2009, 27, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Zaman, K.; Fleming, J.A.; Victor, J.C.; Yunus, M.; Bari, T.I.A.; Azim, T.; Rahman, M.; Mowla, S.M.N.; Bellini, W.J.; McNeal, M. Noninterference of rotavirus vaccine with measles-rubella vaccine at 9 months of age and improvements in antirotavirus immunity: A randomized trial. J. Infect. Dis. 2016, 213, 1686–1693. [Google Scholar] [CrossRef]
- Haidara, F.C.; Tapia, M.D.; Sow, S.O.; Doumbia, M.; Coulibaly, F.; Diallo, F.; Traore, A.; Kodio, M.; Kelly, C.L.; Fitzpatrick, M.; et al. Evaluation of a Booster Dose of Pentavalent Rotavirus Vaccine Coadministered With Measles, Yellow Fever, and Meningitis A Vaccines in 9-Month-Old Malian Infants. J. Infect. Dis. 2018, 218, 606–613. [Google Scholar] [CrossRef]
- Chandola, T.R.; Taneja, S.; Goyal, N.; Antony, K.; Bhatia, K.; More, D.; Bhandari, N.; Cho, I.; Mohan, K.; Prasad, S.; et al. ROTAVAC® does not interfere with the immune response to childhood vaccines in Indian infants: A randomized placebo controlled trial. Heliyon 2017, 3, e00302. [Google Scholar] [CrossRef]
- Desai, S.; Rathi, N.; Kawade, A.; Venkatramanan, P.; Kundu, R.; Lalwani, S.K.; Dubey, A.P.; Venkateswara Rao, J.; Narayanappa, D.; Ghildiyal, R.; et al. Non-interference of Bovine-Human reassortant pentavalent rotavirus vaccine ROTASIIL(R) with the immunogenicity of infant vaccines in comparison with a licensed rotavirus vaccine. Vaccine 2018, 36, 5519–5523. [Google Scholar] [CrossRef]
- Rathi, N.; Desai, S.; Kawade, A.; Venkatramanan, P.; Kundu, R.; Lalwani, S.K.; Dubey, A.P.; Venkateswara Rao, J.; Narayanappa, D.; Ghildiyal, R.; et al. A Phase III open-label, randomized, active controlled clinical study to assess safety, immunogenicity and lot-to-lot consistency of a bovine-human reassortant pentavalent rotavirus vaccine in Indian infants. Vaccine 2018, 36, 7943–7949. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.D.; De Vos, B.; Tumbo, J.; Reynders, J.; Scholtz, F.; Bos, P.; de Beer, M.C.; Van der Merwe, C.F.; Delem, A. Co-administration study in South African infants of a live-attenuated oral human rotavirus vaccine (RIX4414) and poliovirus vaccines. Vaccine 2010, 28, 6542–6548. [Google Scholar] [CrossRef] [PubMed]
- Vesikari, T.; Karvonen, A.; Prymula, R.; Schuster, V.; Tejedor, J.C.; Thollot, F.; Garcia-Corbeira, P.; Damaso, S.; Han, H.-H.; Bouckenooghe, A. Immunogenicity and safety of the human rotavirus vaccine Rotarix™ co-administered with routine infant vaccines following the vaccination schedules in Europe. Vaccine 2010, 28, 5272–5279. [Google Scholar] [CrossRef] [PubMed]
- Dennehy, P.H.; Bertrand, H.R.; Silas, P.E.; Damaso, S.; Friedland, L.R.; Abu-Elyazeed, R. Coadministration of RIX4414 oral human rotavirus vaccine does not impact the immune response to antigens contained in routine infant vaccines in the United States. Pediatrics 2008, 122, e1062–e1066. [Google Scholar] [CrossRef]
- Rodriguez, Z.M.; Goveia, M.G.; Stek, J.E.; Dallas, M.J.; Boslego, J.W.; DiNubile, M.J.; Heaton, P.M. Concomitant Use of an Oral Live Pentavalent Human-Bovine Reassortant Rotavirus Vaccine With Licensed Parenteral Pediatric Vaccines in the United States. Pediatr. Infect. Dis. J. 2007, 26, 221–227. [Google Scholar] [CrossRef]
- Ing, D.J.; Glass, R.I.; Woods, P.A.; Simonetti, M.; Pallansch, M.A.; Wilcox, W.D.; Davidson, B.L.; Sievert, A.J. Immunogenicity of tetravalent rhesus rotavirus vaccine administered with buffer and oral polio vaccine. Am. J. Dis. Child. 1991, 145, 892–897. [Google Scholar] [CrossRef]
- Rennels, M.B.; Ward, R.L.; Mack, M.E.; Zito, E.T.; Group, U.S.R.V.E. Concurrent oral poliovirus and rhesus-human reassortant rotavirus vaccination: Effects on immune responses to both vaccines and on efficacy of rotavirus vaccines. J. Infect. Dis. 1996, 173, 306–313. [Google Scholar] [CrossRef]
- Migasena, S.; Simasathien, S.; Samakoses, R.; Pitisuttitham, P.; Sangaroon, P.; van Steenis, G.; Coen Beuvery, E.; Bugg, H.; Bishop, R.; Davidson, B.L.; et al. Simultaneous administration of oral rhesus-human reassortant tetravalent (RRV-TV) rotavirus vaccine and oral poliovirus vaccine (OPV) in Thai infants. Vaccine 1995, 13, 168–174. [Google Scholar] [CrossRef]
- Ho, M.S.; Floyd, R.L.; Glass, R.I.; Pallansch, M.A.; Jones, B.; Hamby, B.; Woods, P.; Penaranda, M.E.; Kapikian, A.Z.; Bohan, G.; et al. Simultaneous administration of rhesus rotavirus vaccine and oral poliovirus vaccine: Immunogenicity and reactogenicity. Pediatr. Infect. Dis. J. 1989, 8, 692–696. [Google Scholar] [CrossRef]
- Jalil, F.; Zaman, S.; Carlsson, B.; Glass, R.I.; Kapikian, A.Z.; Mellander, L.; Hanson, L.A. Immunogenicity and reactogenicity of rhesus rotavirus vaccine given in combination with oral or inactivated poliovirus vaccines and diphtheria-tetanus-pertussis vaccine. Trans. R. Soc. Trop. Med. Hyg. 1991, 85, 292–296. [Google Scholar] [CrossRef]
- Vodopija, I.; Baklaic, Z.; Vlatkovic, R.; Bogaerts, H.; Delem, A.; Andre, F.E. Combined vaccination with live oral polio vaccine and the bovine rotavirus RIT 4237 strain. Vaccine 1986, 4, 233–236. [Google Scholar] [CrossRef]
- Giammanco, G.; De Grandi, V.; Lupo, L.; Mistretta, A.; Pignato, S.; Teuween, D.; Bogaerts, H.; Andre, F.E. Interference of oral poliovirus vaccine on RIT 4237 oral rotavirus vaccine. Eur. J. Epidemiol. 1988, 4, 121–123. [Google Scholar] [CrossRef]
- Boom, J.A.; Tate, J.E.; Sahni, L.C.; Rench, M.A.; Hull, J.J.; Gentsch, J.R.; Patel, M.M.; Baker, C.J.; Parashar, U.D. Effectiveness of pentavalent rotavirus vaccine in a large urban population in the United States. Pediatrics 2010, 125, e199–e207. [Google Scholar] [CrossRef]
- Donauer, S.; Payne, D.C.; Edwards, K.M.; Szilagyi, P.G.; Hornung, R.W.; Weinberg, G.A.; Chappell, J.; Hall, C.B.; Parashar, U.D.; Staat, M.A. Determining the effectiveness of the pentavalent rotavirus vaccine against rotavirus hospitalizations and emergency department visits using two study designs. Vaccine 2013, 31, 2692–2697. [Google Scholar] [CrossRef][Green Version]
- Sonia, P.D.; Federico, M.F.; José María, M.S.; Lorenzo, R.C.; Marta, B.A. Effectiveness of rotavirus vaccination in Spain. Hum. Vaccines 2011, 7, 757–761. [Google Scholar] [CrossRef]
- Beres, L.K.; Tate, J.E.; Njobvu, L.; Chibwe, B.; Rudd, C.; Guffey, M.B.; Stringer, J.S.; Parashar, U.D.; Chilengi, R. A Preliminary Assessment of Rotavirus Vaccine Effectiveness in Zambia. Clin. Infect. Dis. 2016, 62 (Suppl. 2), S175–S182. [Google Scholar] [CrossRef]
- Armah, G.; Pringle, K.; Enweronu-Laryea, C.C.; Ansong, D.; Mwenda, J.M.; Diamenu, S.K.; Narh, C.; Lartey, B.; Binka, F.; Grytdal, S.; et al. Impact and Effectiveness of Monovalent Rotavirus Vaccine Against Severe Rotavirus Diarrhea in Ghana. Clin. Infect. Dis. 2016, 62, S200–S207. [Google Scholar] [CrossRef]
- Armah, G.; Lewis, K.D.; Cortese, M.M.; Parashar, U.D.; Ansah, A.; Gazley, L.; Victor, J.C.; McNeal, M.M.; Binka, F.; Steele, A.D. A Randomized, Controlled Trial of the Impact of Alternative Dosing Schedules on the Immune Response to Human Rotavirus Vaccine in Rural Ghanaian Infants. J. Infect. Dis. 2016, 213, 1678–1685. [Google Scholar] [CrossRef]
- Kompithra, R.Z.; Paul, A.; Manoharan, D.; Babji, S.; Sarkar, R.; Mathew, L.G.; Kang, G. Immunogenicity of a three dose and five dose oral human rotavirus vaccine (RIX4414) schedule in south Indian infants. Vaccine 2014, 32 (Suppl. 1), A129–A133. [Google Scholar] [CrossRef]
- Ramani, S.; Sowmyanarayanan, T.V.; Gladstone, B.P.; Bhowmick, K.; Asirvatham, J.R.; Jana, A.K.; Kuruvilla, K.A.; Kumar, M.; Gibikote, S.; Kang, G. Rotavirus infection in the neonatal nurseries of a tertiary care hospital in India. Pediatr. Infect. Dis. J. 2008, 27, 719–723. [Google Scholar] [CrossRef]
- Lokeshwar, M.R.; Bhave, S.; Gupta, A.; Goyal, V.K.; Walia, A. Immunogenicity and safety of the pentavalent human-bovine (WC3) reassortant rotavirus vaccine (PRV) in Indian infants. Hum. Vaccines Immunother. 2013, 9, 172–176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ali, S.A.; Kazi, A.M.; Cortese, M.M.; Fleming, J.A.; Parashar, U.D.; Jiang, B.; McNeal, M.M.; Steele, D.; Bhutta, Z.; Zaidi, A. Impact of different dosing schedules on the immunogenicity of the human rotavirus vaccine in infants in Pakistan: A randomized trial. J. Infect. Dis. 2014, 210, 1772–1779. [Google Scholar] [CrossRef] [PubMed]
- Rattan, K.N.; Khurana, P.; Malik, V.; Maggu, S. Intestinal intussusception in children: A review of 70 cases. Indian J. Gastroenterol. Off. J.Indian Soc. Gastroenterol. 2000, 19, 92. [Google Scholar]
- Dawod, S.T.; Osundwa, V.M. Intussusception in children under 2 years of age in the State of Qatar: Analysis of 67 cases. Ann. Trop. Paediatr. 1992, 12, 121–126. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Acute Intussusception in Infants and Children: Incidence, Clinical Representation and Management: A Global Perspective; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Bines, J.E.; Liem, N.T.; Justice, F.A.; Son, T.N.; Kirkwood, C.D.; De Campo, M.; Barnett, P.; Bishop, R.F.; Robins-Browne, R.; Carlin, J.B. Risk factors for intussusception in infants in Vietnam and Australia: Adenovirus implicated, but not rotavirus. J. Pediatr. 2006, 149, 452–460. [Google Scholar] [CrossRef]
- Jiang, J.; Jiang, B.; Parashar, U.; Nguyen, T.; Bines, J.; Patel, M.M. Childhood intussusception: A literature review. PLoS ONE 2013, 8, e68482. [Google Scholar] [CrossRef]
- Carlin, J.B.; Macartney, K.K.; Lee, K.J.; Quinn, H.E.; Buttery, J.; Lopert, R.; Bines, J.; McIntyre, P.B. Intussusception risk and disease prevention associated with rotavirus vaccines in Australia’s National Immunization Program. Clin. Infect. Dis. 2013, 57, 1427–1434. [Google Scholar] [CrossRef]
- Stowe, J.; Andrews, N.; Ladhani, S.; Miller, E. The risk of intussusception following monovalent rotavirus vaccination in England: A self-controlled case-series evaluation. Vaccine 2016, 34, 3684–3689. [Google Scholar] [CrossRef]
- Rennels, M.B.; Parashar, U.D.; Holman, R.C.; Le, C.T.; Chang, H.-G.; Glass, R.I. Lack of an apparent association between intussusception and wild or vaccine rotavirus infection. Pediatr. Infect. Dis. J. 1998, 17, 924–925. [Google Scholar] [CrossRef]
- Mangete, E.D.; Allison, A.B. Intussusception in infancy and childhood: Analysis of 69 cases. East. African Med. J. 1993, 70, 734–736. [Google Scholar]
- Sutthiwan, P.; Darnwiriyagul, L.; Sritanyaratana, S. Intussusception. J. Med. Assoc. Thailand Chotmaihet Thangphaet 1982, 65, 403. [Google Scholar]
- Bruce, J.; Huh, Y.S.; Cooney, D.R.; Karp, M.P.; Allen, J.E.; Jewett, T.C., Jr. Intussusception: Evolution of current management. J. Pediatr. Gastroenterol. Nutr. 1987, 6, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.Y.; Kao, C.L.; Huang, L.M.; Ni, Y.H.; Lai, H.S.; Lin, F.Y.; Chang, M.H. Viral etiology of intussusception in Taiwanese childhood. Pediatr. Infect. Dis. J. 1998, 17, 893–898. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.; Gargiullo, P.; Murphy, T.V.; Parashar, U.D.; Patel, M.M. Sociodemographic and Dietary Risk Factors for Natural Infant Intussusception in the United States. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Intussusception among recipients of rotavirus vaccine—United States, 1998–1999. MMWR. Morb. Mortal. Wkly. Rep. 1999, 48, 577. [Google Scholar]
- Chang, H.G.H.; Smith, P.F.; Ackelsberg, J.; Morse, D.L.; Glass, R.I. Intussusception, rotavirus diarrhea, and rotavirus vaccine use among children in New York State. Pediatrics 2001, 108, 54–60. [Google Scholar] [CrossRef]
- Simonsen, L.; Morens, D.M.; Elixhauser, A.; Gerber, M.; Van Raden, M.; Blackwelder, W.C. Effect of rotavirus vaccination programme on trends in admission of infants to hospital for intussusception. Lancet 2001, 358, 1224–1229. [Google Scholar] [CrossRef]
- Haber, P.; Patel, M.; Pan, Y.; Baggs, J.; Haber, M.; Museru, O.; Yue, X.; Lewis, P.; Destefano, F.; Parashar, U.D. Intussusception after rotavirus vaccines reported to US VAERS, 2006–2012. Pediatrics 2013, 131, 1042–1049. [Google Scholar] [CrossRef]
- Yen, C.; Healy, K.; Tate, J.E.; Parashar, U.D.; Bines, J.; Neuzil, K.; Santosham, M.; Steele, A.D. Rotavirus vaccination and intussusception—Science, surveillance, and safety: A review of evidence and recommendations for future research priorities in low and middle income countries. Hum. Vaccine Immunother. 2016, 12, 2580–2589. [Google Scholar] [CrossRef]
- Verstraeten, T.; Baughman, A.L.; Cadwell, B.; Zanardi, L.; Haber, P.; Chen, R.T.; Vaccine Adverse Event Reporting System Team. Enhancing Vaccine Safety Surveillance: A Capture-Recapture Analysis of Intussusception after Rotavirus Vaccination. Am. J. Epidemiol. 2001, 154, 1006–1012. [Google Scholar] [CrossRef]
- Ugwu, B.T.; Legbo, J.N.; Dakum, N.K.; Yiltok, S.J.; Mbah, N.; Uba, F.A. Childhood intussusception: A 9-year review. Ann. Trop. Paediatr. 2000, 20, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Yung, C.-F.; Chan, S.P.; Soh, S.; Tan, A.; Thoon, K.C. Intussusception and Monovalent Rotavirus Vaccination in Singapore: Self-Controlled Case Series and Risk-Benefit Study. J. Pediatr. 2015, 167, 163–168.e1. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.; Tate, J.E.; Steiner, C.A.; Cortese, M.M.; Patel, M.M.; Parashar, U.D. Trends in Intussusception Hospitalizations Among US Infants Before and After Implementation of the Rotavirus Vaccination Program, 2000–2009. J. Infect. Dis. 2012, 206, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Allan, G.M.; McNeilly, F.; Cassidy, J.P.; Reilly, G.A.; Adair, B.; Ellis, W.A.; McNulty, M.S. Pathogenesis of porcine circovirus; experimental infections of colostrum deprived piglets and examination of pig foetal material. Vet. Microbiol. 1995, 44, 49–64. [Google Scholar] [CrossRef]
- Tischer, I.; Mields, W.; Wolff, D.; Vagt, M.; Griem, W. Studies on epidemiology and pathogenicity of porcine circovirus. Arch. Virol. 1986, 91, 271–276. [Google Scholar] [CrossRef]
- Victoria, J.G.; Wang, C.; Jones, M.S.; Jaing, C.; McLoughlin, K.; Gardner, S.; Delwart, E.L. Viral nucleic acids in live-attenuated vaccines: Detection of minority variants and an adventitious virus. J. Virol. 2010, 84, 6033–6040. [Google Scholar] [CrossRef]
- McClenahan, S.D.; Krause, P.R.; Uhlenhaut, C. Molecular and infectivity studies of porcine circovirus in vaccines. Vaccine 2011, 29, 4745–4753. [Google Scholar] [CrossRef]
- Martinón-Torres, F.; Vesikari, T.; Giaquinto, C.; Huppertz, H.-I. How France reversed its position on rotavirus vaccination - Presented by Pierre Pothier. In Proceedings of the 4th European Expert Meeting on Rotavirus Vaccination (EEROVAC), Santiago de Compostela, Spain, 23–25 March 2015; pp. 1–36. [Google Scholar]
- Dulac, G.C.; Afshar, A. Porcine circovirus antigens in PK-15 cell line (ATCC CCL-33) and evidence of antibodies to circovirus in Canadian pigs. Can. J. Vet. Res. 1989, 53, 431–433. [Google Scholar]
- Raye, W.; Muhling, J.; Warfe, L.; Buddle, J.R.; Palmer, C.; Wilcox, G.E. The detection of porcine circovirus in the Australian pig herd. Aust. Vet. J. 2005, 83, 300–304. [Google Scholar] [CrossRef]
- Allan, G.M.; McNeilly, F.; Kennedy, S.; Daft, B.; Clarke, E.G.; Ellis, J.A.; Haines, D.M.; Meehan, B.M.; Adair, B.M. Isolation of porcine circovirus-like viruses from pigs with a wasting disease in the USA and Europe. J. Vet. Diagn. Invest. 1998, 10, 3–10. [Google Scholar] [CrossRef]
- Hanley, K.A. The double-edged sword: How evolution can make or break a live-attenuated virus vaccine. Evolution (N Y) 2011, 4, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Donato, C.M.; Ch’ng, L.S.; Boniface, K.F.; Crawford, N.W.; Buttery, J.P.; Lyon, M.; Bishop, R.F.; Kirkwood, C.D. Identification of strains of RotaTeq rotavirus vaccine in infants with gastroenteritis following routine vaccination. J. Infect. Dis. 2012, 206, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Hemming, M.; Vesikari, T. Detection of rotateq vaccine-derived, double-reassortant rotavirus in a 7-year-old child with acute gastroenteritis. Pediatr. Infect. Dis. J. 2014, 33, 655–656. [Google Scholar] [CrossRef]
- Rose, T.L.; da Silva, M.F.M.; Goméz, M.M.; Resque, H.R.; Ichihara, M.Y.T.; de Mello Volotão, E.; Leite, J.P.G. Evidence of vaccine-related reassortment of rotavirus, Brazil, 2008–2010. Emerg. Infect. Dis. 2013, 19, 1843. [Google Scholar] [CrossRef] [PubMed]
- Boom, J.A.; Sahni, L.C.; Payne, D.C.; Gautam, R.; Lyde, F.; Mijatovic-Rustempasic, S.; Bowen, M.D.; Tate, J.E.; Rench, M.A.; Gentsch, J.R.; et al. Symptomatic infection and detection of vaccine and vaccine-reassortant rotavirus strains in 5 children: A case series. J. Infect. Dis. 2012, 206, 1275–1279. [Google Scholar] [CrossRef][Green Version]
- Donato, C.M.; Zhang, Z.A.; Donker, N.C.; Kirkwood, C.D. Characterization of G2P[4] rotavirus strains associated with increased detection in Australian states using the RotaTeq (R) vaccine during the 2010–2011 surveillance period. Infect. Genet. Evol. 2014, 28, 398–412. [Google Scholar] [CrossRef] [PubMed]
- Dóró, R.; László, B.; Martella, V.; Leshem, E.; Gentsch, J.; Parashar, U.; Bányai, K. Review of global rotavirus strain prevalence data from six years post vaccine licensure surveillance: Is there evidence of strain selection from vaccine pressure? Infect. Genet. Evol. 2014, 28, 446–461. [Google Scholar] [CrossRef]
- Matthias, D.M.; Robertson, J.; Garrison, M.M.; Newland, S.; Nelson, C. Freezing temperatures in the vaccine cold chain: A systematic literature review. Vaccine 2007, 25, 3980–3986. [Google Scholar] [CrossRef]
- Levin, A.; Levin, C.; Kristensen, D.; Matthias, D. An economic evaluation of thermostable vaccines in Cambodia, Ghana and Bangladesh. Vaccine 2007, 25, 6945–6957. [Google Scholar] [CrossRef]
- Chen, D.X.; Kristensen, D. Opportunities and challenges of developing thermostable vaccines. Expert Rev. Vaccines 2009, 8, 547–557. [Google Scholar] [CrossRef]
- Hansen, L.J.J.; Daoussi, R.; Vervaet, C.; Remon, J.P.; De Beer, T.R.M. Freeze-drying of live virus vaccines: A review. Vaccine 2015, 33, 5507–5519. [Google Scholar] [CrossRef] [PubMed]
- Kawade, A.; Babji, S.; Kamath, V.; Raut, A.; Kumar, C.M.; Kundu, R.; Venkatramanan, P.; Lalwani, S.K.; Bavdekar, A.; Juvekar, S.; et al. Immunogenicity and lot-to-lot consistency of a ready to use liquid bovine-human reassortant pentavalent rotavirus vaccine (ROTASIIL—Liquid) in Indian infants. Vaccine 2019, 37, 2554–2560. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.I.; Sack, D.A.; Rothstein, E.; Reisinger, K.; Smith, V.E.; O’Sullivan, D.; Spriggs, D.R.; Ward, R.L. Efficacy of live, attenuated, human rotavirus vaccine 89-12 in infants: A randomised placebo-controlled trial. Lancet 1999, 354, 287–290. [Google Scholar] [CrossRef]
- Joensuu, J.; Koskenniemi, E.; Vesikari, T. Symptoms associated with rhesus-human reassortant rotavirus vaccine in infants. Pediatr. Infect. Dis. J. 1998, 17, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Roczo-Farkas, S.; Kirkwood, C.D.; Cowley, D.; Barnes, G.L.; Bishop, R.F.; Bogdanovic-Sakran, N.; Boniface, K.; Donato, C.M.; Bines, J.E. The Impact of Rotavirus Vaccines on Genotype Diversity: A Comprehensive Analysis of 2 Decades of Australian Surveillance Data. J. Infect. Dis. 2018, 218, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Markkula, J.; Hemming-Harlo, M.; Salminen, M.T.; Savolainen-Kopra, C.; Pirhonen, J.; al-Hello, H.; Vesikari, T. Rotavirus epidemiology 5–6 years after universal rotavirus vaccination: Persistent rotavirus activity in older children and elderly. Infect. Dis. 2017, 49, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Cowley, D.; Donato, C.M.; Roczo-Farkas, S.; Kirkwood, C.D. Emergence of a novel equine-like G3P[8] inter-genogroup reassortant rotavirus strain associated with gastroenteritis in Australian children. J. Gen. Virol. 2016, 97, 403–410. [Google Scholar] [CrossRef]
- Luchs, A.; Timenetsky Mdo, C. Group A rotavirus gastroenteritis: Post-vaccine era, genotypes and zoonotic transmission. Einstein (Sao Paulo) 2016, 14, 278–287. [Google Scholar] [CrossRef]
- Pitzer, V.E.; Bilcke, J.; Heylen, E.; Crawford, F.W.; Callens, M.; De Smet, F.; Van Ranst, M.; Zeller, M.; Matthijnssens, J. Did large-scale vaccination drive changes in the circulating rotavirus population in Belgium? Sci. Rep. 2015, 5, 18585. [Google Scholar] [CrossRef]
- Yen, C.; Figueroa, J.R.; Uribe, E.S.; Carmen-Hernández, L.D.; Tate, J.E.; Parashar, U.D.; Patel, M.M.; Richardson López-Collado, V. Monovalent rotavirus vaccine provides protection against an emerging fully heterotypic G9P[4] rotavirus strain in Mexico. J. Infect. Dis. 2011, 204, 783–786. [Google Scholar] [CrossRef]
- Groome, M.J.; Page, N.; Cortese, M.M.; Moyes, J.; Zar, H.J.; Kapongo, C.N.; Mulligan, C.; Diedericks, R.; Cohen, C.; Fleming, J.A.; et al. Effectiveness of monovalent human rotavirus vaccine against admission to hospital for acute rotavirus diarrhoea in South African children: A case-control study. Lancet Infect. Dis. 2014, 14, 1096–1104. [Google Scholar] [CrossRef]
- Braeckman, T.; Van Herck, K.; Meyer, N.; Pircon, J.Y.; Soriano-Gabarro, M.; Heylen, E.; Zeller, M.; Azou, M.; Capiau, H.; De Koster, J.; et al. Effectiveness of rotavirus vaccination in prevention of hospital admissions for rotavirus gastroenteritis among young children in Belgium: Case-control study. BMJ 2012, 345, e4752. [Google Scholar] [CrossRef] [PubMed]
- Simwaka, J.C.; Mpabalwani, E.M.; Seheri, M.; Peenze, I.; Monze, M.; Matapo, B.; Parashar, U.D.; Mufunda, J.; Mphahlele, J.M.; Tate, J.E.; et al. Diversity of rotavirus strains circulating in children under five years of age who presented with acute gastroenteritis before and after rotavirus vaccine introduction, University Teaching Hospital, Lusaka, Zambia, 2008–2015. Vaccine 2018, 36, 7243–7247. [Google Scholar] [CrossRef] [PubMed]
- Seheri, L.M.; Magagula, N.B.; Peenze, I.; Rakau, K.; Ndadza, A.; Mwenda, J.M.; Weldegebriel, G.; Steele, A.D.; Mphahlele, M.J. Rotavirus strain diversity in Eastern and Southern African countries before and after vaccine introduction. Vaccine 2018, 36, 7222–7230. [Google Scholar] [CrossRef] [PubMed]
- Zaki, A.M.; DuPont, H.L.; el Alamy, M.A.; Arafat, R.R.; Amin, K.; Awad, M.M.; Bassiouni, L.; Imam, I.Z.; el Malih, G.S.; el Marsafie, A.; et al. The Detection of Enteropathogens in Acute Diarrhea in a Family Cohort Population in Rural Egypt. Am. J. Trop. Med. Hyg. 1986, 35, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Leshem, E.; Lopman, B.; Glass, R.; Gentsch, J.; Bányai, K.; Parashar, U.; Patel, M. Distribution of rotavirus strains and strain-specific effectiveness of the rotavirus vaccine after its introduction: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 847–856. [Google Scholar] [CrossRef]
- Hull, J.J.; Teel, E.N.; Kerin, T.K.; Freeman, M.M.; Esona, M.D.; Gentsch, J.R.; Cortese, M.M.; Parashar, U.D.; Glass, R.I.; Bowen, M.D.; et al. United States rotavirus strain surveillance from 2005 to 2008: Genotype prevalence before and after vaccine introduction. Pediatr. Infect. Dis. J. 2011, 30, S42–S47. [Google Scholar] [CrossRef]
- Kirkwood, C.D.; Boniface, K.; Barnes, G.L.; Bishop, R.F. Distribution of Rotavirus Genotypes After Introduction of Rotavirus Vaccines, Rotarix® and RotaTeq®, into the National Immunization Program of Australia. Pediatr. Infect. Dis. J. 2011, 30, 48–53. [Google Scholar] [CrossRef]
- Zeller, M.; Rahman, M.; Heylen, E.; De Coster, S.; De Vos, S.; Arijs, I.; Novo, L.; Verstappen, N.; Van Ransta, M.; Matthijnssens, J. Rotavirus incidence and genotype distribution before and after national rotavirus vaccine introduction in Belgium. Vaccine 2010, 28, 7507–7513. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Bilcke, J.; Ciarlet, M.; Martella, V.; Banyai, K.; Rahman, M.; Zeller, M.; Beutels, P.; Van Damme, P.; Van Ranst, M. Rotavirus disease and vaccination: Impact on genotype diversity. Future Microbiol. 2009, 4, 1303–1316. [Google Scholar] [CrossRef]
- Zeller, M.; Donato, C.; Trovao, N.S.; Cowley, D.; Heylen, E.; Donker, N.C.; McAllen, J.K.; Akopov, A.; Kirkness, E.F.; Lemey, P.; et al. Genome-Wide Evolutionary Analyses of G1P[8] Strains Isolated Before and After Rotavirus Vaccine Introduction. Genome Biol. Evol. 2015, 7, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.I.; Sander, D.S.; Smith, V.E.; Schiff, G.M.; Ward, R.L. Protection from rotavirus reinfection: 2-year prospective study. J. Infect. Dis. 1991, 164, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.D.; Madhi, S.A.; Cunliffe, N.A.; Vesikari, T.; Phua, K.B.; Lim, F.S.; Nelson, E.A.; Lau, Y.L.; Huang, L.M.; Karkada, N.; et al. Incidence of rotavirus gastroenteritis by age in African, Asian and European children: Relevance for timing of rotavirus vaccination. Hum. Vaccine Immunother. 2016, 12, 2406–2412. [Google Scholar] [CrossRef] [PubMed]
- Cunliffe, N.; Zaman, K.; Rodrigo, C.; Debrus, S.; Benninghoff, B.; Pemmaraju Venkata, S.; Han, H.H. Early exposure of infants to natural rotavirus infection: A review of studies with human rotavirus vaccine RIX4414. BMC Pediatr. 2014, 14, 295. [Google Scholar] [CrossRef] [PubMed]
- Moulton, L.H.; Staat, M.A.; Santosham, M.; Ward, R.L. The Protective Effectiveness of Natural Rotavirus Infection in an American Indian Population. J. Infect. Dis. 1998, 178, 1562–1566. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ward, R.L.; Bernstein, D.I. Protection against Rotavirus Disease after Natural Rotavirus Infection. J. Infect. Dis. 1994, 169, 900–904. [Google Scholar] [CrossRef]
- Fischer, T.K.; Valentiner-Branth, P.; Steinsland, H.; Perch, M.; Santos, G.; Aaby, P.; Molbak, K.; Sommerfelt, H. Protective immunity after natural rotavirus infection: A community cohort study of newborn children in Guinea-Bissau, west Africa. J. Infect. Dis. 2002, 186, 593–597. [Google Scholar] [CrossRef]
- Mata, L.; Simhon, A.; Urrutia, J.J.; Kronmal, R.A.; Fernández, R.; Garcia, B. Epidemiology of Rotaviruses in a Cohort of 45 Guatamalan Mayan Indian Children Observed from Birth to the Age of Three Years. J. Infect. Dis. 1983, 148, 452–461. [Google Scholar] [CrossRef]
- Zheng, B.J.; Lo, S.K.; Tam, J.S.; Lo, M.; Yeung, C.Y.; Ng, M.H. Prospective study of community-acquired rotavirus infection. J. Clin. Microbiol. 1989, 27, 2083. [Google Scholar] [CrossRef]
- Espinoza, F.; Paniagua, M.; Hallander, H.; Svensson, L.; Strannegard, O. Rotavirus infections in young Nicaraguan children. Pediatr. Infect. Dis. J. 1997, 16, 564–571. [Google Scholar] [CrossRef]
- Naficy, A.B.; Abu-Elyazeed, R.; Holmes, J.L.; Rao, M.R.; Savarino, S.J.; Kim, Y.; Wierzba, T.F.; Peruski, L.; Lee, Y.J.; Gentsch, J.R.; et al. Epidemiology of Rotavirus Diarrhea in Egyptian Children and Implications for Disease Control. Am. J. Epidemiol. 1999, 150, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Reves, R.R.; Hossain, M.M.; Midthun, K.; Kapikian, A.Z.; Naguib, T.; Zaki, A.M.; DuPont, H.L. An observational study of naturally acquired immunity to rotaviral diarrhea in a cohort of 363 Egyptian children. Calculation of risk for second episodes using age-specific person-years of observation. Am. J. Epidemiol. 1989, 130, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, B.P.; Ramani, S.; Mukhopadhya, I.; Muliyil, J.; Sarkar, R.; Rehman, A.M.; Jaffar, S.; Gomara, M.I.; Gray, J.J.; Brown, D.W.; et al. Protective effect of natural rotavirus infection in an Indian birth cohort. N. Engl. J. Med. 2011, 365, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Bhan, M.K.; Lew, J.F.; Sazawal, S.; Das, B.K.; Gentsch, J.R.; Glass, R.I. Protection conferred by neonatal rotavirus infection against subsequent rotavirus diarrhea. J. Infect. Dis. 1993, 168, 282–287. [Google Scholar] [CrossRef]
- Gentsch, J.R.; Woods, P.A.; Ramachandran, M.; Das, B.K.; Leite, J.P.; Alfieri, A.; Kumar, R.; Bhan, M.K.; Glass, R.I. Review of G and P typing results from a global collection of rotavirus strains: Implications for vaccine development. J. Infect. Dis. 1996, 174 (Suppl. 1), S30–S36. [Google Scholar] [CrossRef]
- Woods, P.A.; Gentsch, J.; Gouvea, V.; Mata, L.; Santosham, M.; Bai, Z.S.; Urasawa, S.; Glass, R.I. Distribution of serotypes of human rotavirus in different populations. J. Clin. Microbiol. 1992, 30, 781–785. [Google Scholar] [CrossRef]
- Steele, A.D.; Van Niekerk, M.C.; Mphahlele, M.J. Geographic distribution of human rotavirus VP4 genotypes and VP7 serotypes in five South African regions. J. Clin. Microbiol. 1995, 33, 1516–1519. [Google Scholar] [CrossRef]
- Fischer, T.K.; Steinsland, H.; Molbak, K.; Ca, R.; Gentsch, J.R.; Valentiner-Branth, P.; Aaby, P.; Sommerfelt, H. Genotype profiles of rotavirus strains from children in a suburban community in Guinea-Bissau, Western Africa. J. Clin. Microbiol. 2000, 38, 264–267. [Google Scholar]
- Ramachandran, M.; Das, B.K.; Vij, A.; Kumar, R.; Bhambal, S.S.; Kesari, N.; Rawat, H.; Bahl, L.; Thakur, S.; Woods, P.A. Unusual diversity of human rotavirus G and P genotypes in India. J. Clin. Microbiol. 1996, 34, 436–439. [Google Scholar] [CrossRef]
- Chatterjee, B.; Husain, M.; Kavita; Seth, P.; Broor, S. Diversity of rotavirus strains infecting pediatric patients in New Delhi, India. J. Trop. Pediatr. 1996, 42, 207–210. [Google Scholar] [CrossRef][Green Version]
- Gouvea, V.; de Castro, L.; Timenetsky, M.C.; Greenberg, H.; Santos, N. Rotavirus serotype G5 associated with diarrhea in Brazilian children. J. Clin. Microbiol. 1994, 32, 1408–1409. [Google Scholar] [CrossRef] [PubMed]
- Leite, J.P.G.; Alfieri, A.A.; Woods, P.A.; Glass, R.I.; Gentsch, J.R. Rotavirus G and P types circulating in Brazil: Characterization by RT-PCR, probe hybridization, and sequence analysis. Arch. Virol. 1996, 141, 2365–2374. [Google Scholar] [CrossRef]
- Mphahlele, M.J.; Steele, A.D. Relative frequency of human rotavirus VP4 (P) genotypes recovered over a ten-year period from South African children with diarrhea. J. Med. Virol. 1995, 47, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Global Alliance for Vaccines and Immunizations (GAVI). GAVI Alliance Secures Lower Price for Rotavirus Vaccine; GAVI: Geneva, Switzeralnd, 2016. [Google Scholar]
- Global Alliance for Vaccines and Immunizations (GAVI). Vaccine Pricing: Gavi Transitioning Countries; WHO: Geneva, Switzeralnd, 2017; pp. 2–7. [Google Scholar]
- Maggie, F. Childhood Vaccination Plunge Since Covid-19 Pandemic Started, CDC Says. Available online: https://edition.cnn.com/2020/05/08/health/childhood-vaccines-cdc--covid-wellness/index.html (accessed on 8 May 2020).
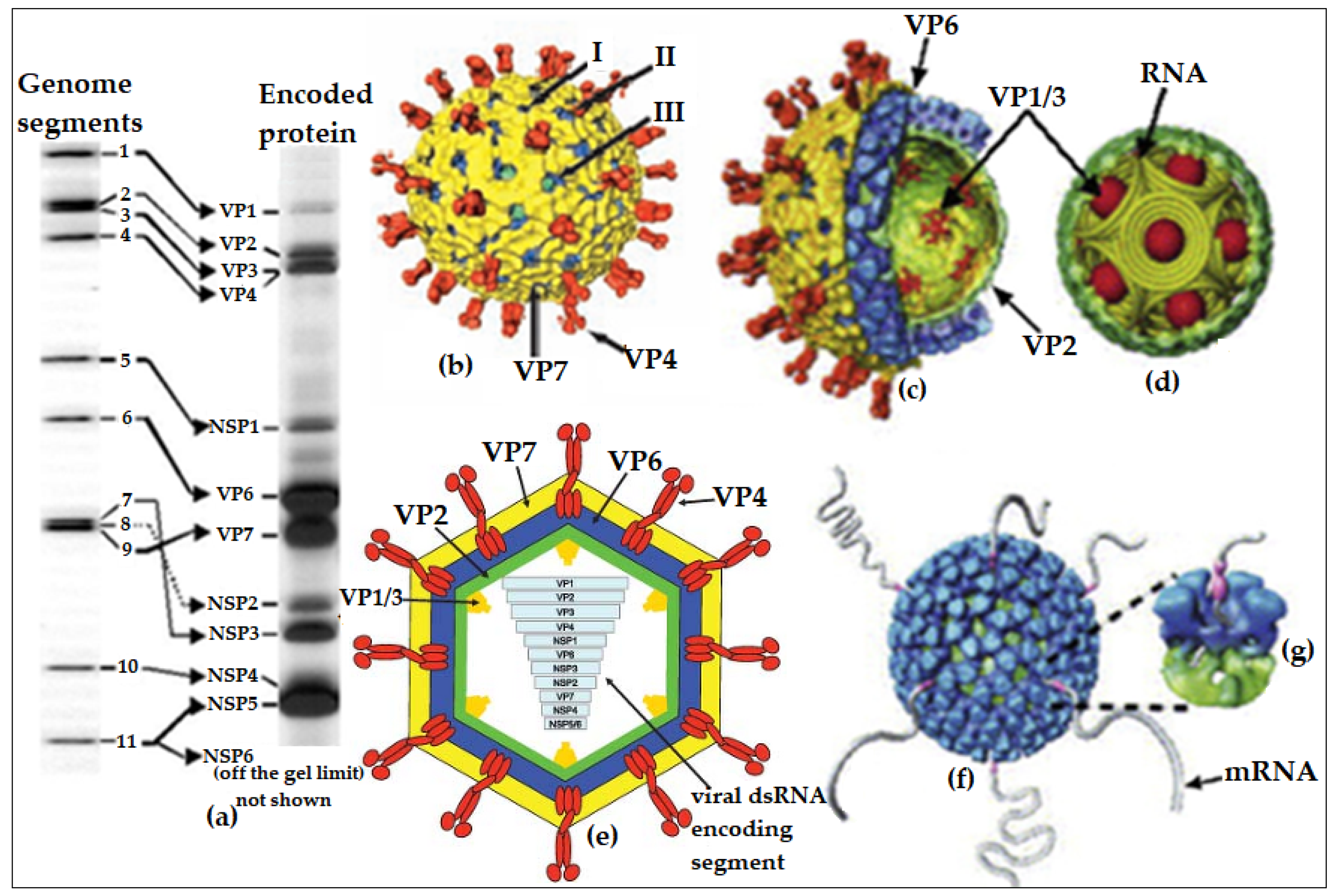
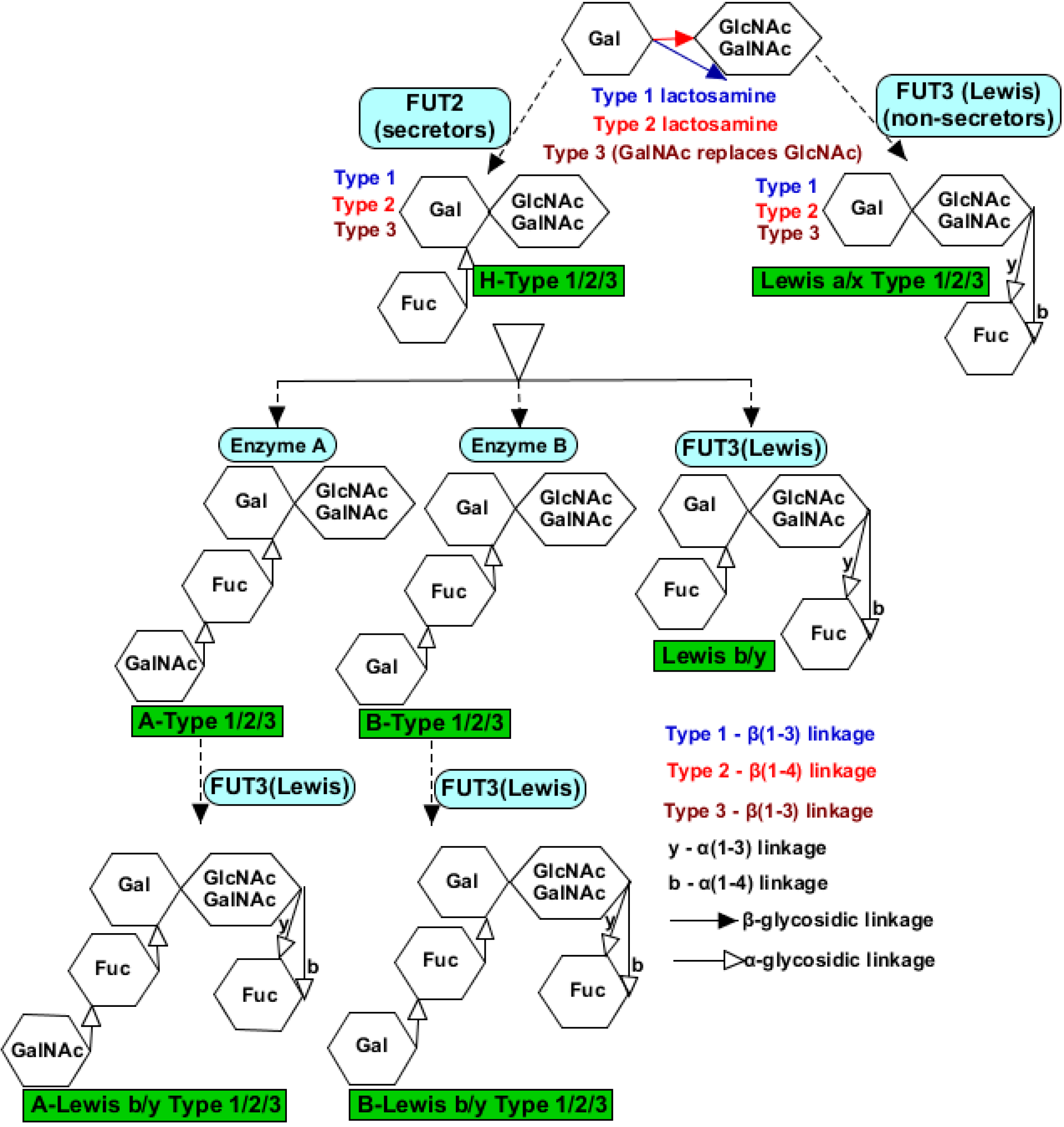
| Vaccine | Strain | Developer/Manufacturer | Type of Vaccine/Status | Vaccine Development and Assessment |
|---|---|---|---|---|
| From animal rotavirus strains (first generation of rotavirus vaccines) | ||||
| RIT4237 (monovalent) | G6P6[1], BRV NCDV strain | SmithKline-RIT (Rixensart, Belgium) | Jennerian, but withdrawn | About 154 cell culture passages with high titre value of over 108 TCID50/mL, first to be tested in human, higher efficacy in the developed countries, negative gastric effect, breast milk did not affect but suppressed by OPV [154]. |
| RRV-MMU (monovalent) | G3P[5] RRV strain | National Institute of Health (NIH) | Modified Jennerian-based vaccine, but withdrawn | Passaged 9 and 7 times in monkey kidney and foetal rhesus lung cells, respectively, with 105 PFU/dose titre level, reactogenic for fever, very virulent and later used as a backbone for RotaShield® [81,155]. |
| WC3 (monovalent) | Wistar calf strain G6P[7] | Institut Merieux/Wistar Institute | Jennerian, but withdrawn | Passaged 20 times with 107 PFU/mL titre value, less efficacious compared with RRV-MMU but later used as a backbone for RotaTeq® [91,156]. |
| LLR-37 (monovalent) | Lanzhou lamb-derived rotavirus G10P[15] | Lanzhou Institute of Biological Products (China) | Jennerian, licensed for use in China only since the year 2000 | Ovine attenuated vaccine, efficacious in 2 doses at 2 months and 2 years, replicate with faecal shedding within 15 days post-vaccination. Over 60 million doses rolled out as at the end of 2014 [157,158]. |
| Animal–human reassortant strains (second generation of rotavirus vaccines)—Jennerian-based vaccines | ||||
| RotaShield® (RV4) | Tetravalent human–rhesus reassortant (G1-G4)P7 [5] | Wyeth (Madison, NJ, USA) | Jennerian and first licensed to be used in the USA by 1998 but withdrawn in 1999 due to IS, but now in phase II trials for neonatal administration in Ghana | First reassortant prototype, stable at 25 °C, formulated with G1, G2, and G4 VP7 antigens on rhesus G3P[5] backbone, reactogenic for fever, formulated with 4 × 104 PFU/mL with two doses and caused IS but tested to be safe in neonates in Ghana [159], which might favour its re-introduction [63,160]. |
| RotaTeq® (RV5) | Pentavalent human–bovine reassortant G1-G4+P7 [5]; G6+P1A [8] | Merck & Co., Inc. (Burlington, MA, USA). Presented as a 2.5 mL liquid vaccine suspended in a buffer and administered via a squeeze tube. Scheduled as 3Ds | Jennerian, pre-qualified for global use since 2008 but first licensed in 2006. Usually co-administered with DPT 1, 2, and 3 vaccine doses | It is formulated with 2–2.8 × 106 PFU/mL with immunogenicity tested in 70,000 infants [161]. The overall efficacy was 95% against (G1–G4, G9)P[8] and displayed heterotypic protection against G2P[4]. Shedding and diarrhoea were observed in <1% infants [162,163]. Similarly, high efficacy was reported in 34,035 vaccinated infants from 11 countries across Latin America, America, and Europe [123]. VE increased with severity but decreased over 2 years of follow-up in Africa and Asia with 6674 infants from Ghana, Kenya, Mali, Bangladesh, and Vietnam (4705 African and 1969 Asian) [164]. The vaccine, however, requires cold-chain (2–8 °C) storage and transportation. |
| BRV-TV or BRV-PV | Bovine–human tetravalent reassortant G1-G4+P7 [5]; G1-G4+G9-P7 [5] | Biotecnics Instituto Butantan Shantha Biotecnics Limited Serum Institute of India (Pune, India) Wuhan (China) | Phase I trial in Brazil Phase II/III trials in India Phase III trial in China - | Started by Wyeth but discontinued at the same time RotaShield® was withdrawn; however, the prototype has been licensed by NIH to Brazil, India, and China [165]. |
| RotaSIIL® (BRV-PV) | Pentavalent human–bovine reassortant serotypes G(1–4) and G9 | Serum Institute of India Pvt Ltd. (SIIPL). Presented in two forms–2.5 mL reconstituted lyophilised vial and 2.5 mL prepared liquid vial. Each scheduled as 3Ds | Pre-qualified for global use in 2018 but licensed for use in India since 2016. Co-administered with DPT 1, 2, and 3 vaccine doses | World’s first cost-effective, thermostable vaccine which can be stored without refrigeration or below 25 °C. It is a bovine UK rotavirus strain from NIH (USA), which was reassorted with human VP7 genome encoding segments [166]. The vaccine has a lifespan of 20 months at 37 °C or 7 months at 45 °C. |
| Human rotavirus strains (second generation of rotavirus vaccines)—modified Jennerian-based vaccines | ||||
| Rotarix® (RIX-4414) | Monovalent human strain G1P1A [8] | GlaxoSmithKline, GSK (Belgium). Presented in two forms. First, as 1 mL liquid vaccine with oral applicator or as a squeeze tube. Secondly, as 1.5 mL freeze-dried reconstituted with buffer and used with an oral applicator. Scheduled as 2Ds | Pre-qualified for global use since 2009 but first licensed in 2006. Co-administered with DPT 1, 2, and 3 vaccine doses | The most extensively used vaccine that was initially developed in Cincinnati, USA. The parent strain was first isolated from a child with natural RVI during the serotype 1 outbreak in 1988–1989 [91] and passaged 45 times in MRC-5 cells to remove the infectivity. Initially developed by Avant Immunotherapeutics Inc. Massachusetts, USA but licensed to GSK for further modification [167]. With 60,000 infants, Rotarix® induced >90% efficacy with significant homotypic and heterotypic protection against HRV strains, reaching 100% efficacy against a more severe case [127]. Summarily, Phases I–III trials with over 70,000 children proved the vaccine was non-reactogenic, well-tolerated, effective at first dose, but increased seropositivity rate after the second dose, not associated with IS but provided >85% protection against moderate to SRVGE, and reduced GE-related hospitalisation by >40% in Latin America and >75% in Europe [168,169]. This vaccine also requires cold-chain facilities (2–8 °C). |
| Rotavac® | Human neonatal strain 116E G9P[11] | Bharat Biotech International Limited (India). Presented as a 0.5 mL liquid vaccine with a separate dropper | Pre-qualified for global use in 2018 but licensed for use in India since 2014 [170]. Co-administered with DPT 1, 2, and 3 vaccine doses | It is a naturally occurring reassortant strain G9P[11], containing one BRV gene P[11] and ten HRV genes [60] at 105 FFU/dose formulation, taking in 3 single doses at 4 weeks interval starting from 6 weeks of age. This vaccine was licensed and introduced into the NIP (National Immunisation Program) of India by 2014 [60]. This vaccine must be stored at −20 °C. |
| Rotavin-M1® | Human strain G1P1A [8] | Centre for Research and Production of Vaccines and Biologicals - Polyvac (Vietnam) | Licensed to be used in Vietnam since 2007 | It was derived from an attenuated G1P[8] strain (KH0118-2003) isolated from a Vietnamese infant, passaged in Vero cell at 106.3 FFU/dose, given in 2 doses (2 months apart) or 3 doses (1 month apart), safety, and immunogenicity tested in 160 infants with rotavirus-specific IgA seroconversion rate of 73% vs. 58% in Rotarix® and rotavirus shedding between 44% and 48% vs. 65% for Rotarix®. Over 500,000 doses already rolled out. The vaccine must be stored at −20 °C [19]. |
| RV3-BB | Human neonatal strain G3P2A [6] | Murdoch Children Research Institute, Australia Biofarma, Indonesia | Phase III trial in Indonesia | RV3 is based on a neonatal G3P[6] strain from Melbourne. A Vero cell-adapted derivative of the strain RV3-BB grew to a higher titre 8.3 × 106 FFU/mL and was more immunogenic than the parent strain RV3 [171]. The Phase IIa trial with 3Ds in New Zealand showed 63% neonate and 74% infant with IgA seroconversion and shedding was 70% in the neonate and 78% in the infant. The vaccine was well-tolerated and immunogenic [172]. Analysis in the Phase IIb in Indonesia showed the efficacy of 75%, 51%, and 63% in the neonate, infant, and combined groups against SRVGE, respectively [173]. Vaccine response/“take” of RV3-BB was 94% and 99% in the neonatal- and infant-scheduled groups, respectively [173]. |
| Vaccine | Seasonal Efficacy/Follow-up/Doses | Remark | |
|---|---|---|---|
| First-Year Follow-up | Second-Year Follow-up | ||
| North America and Europe | |||
| Rotarix® was tested against RVGE in infants from six different European countries | 87.1% efficacy against any SRVGE and 75–96% efficacy against the corresponding genotypes G1–G4 and G9 | 71.9% efficacy against any SRVGE and 69–84% efficacy against the corresponding G1–G4 and G9. About 85.6% efficacy against SRVGE and 77–97% efficacy against the corresponding G1–G4 and G9 | Observed combined efficacy against any SRVGE and SRVGE was 78.9% and 90.4% respectively. The combined efficacy against all-cause SRVGE and hospital admission was 49.6% and 71.5%, respectively [135]. Though similar reactogenic was observed in the tested groups, serious and nonserious adverse effects were reported, but no IS case in the USA and Canada [168]. |
| Rotarix® was evaluated for safety and immunogenicity | 95.8% efficacy against SRVGE and 74–100% against the G1–G4 and G9 | 2Ds of RV1 are well tolerated with a high vaccine “take”, highly immunogenic to induce IgA without side effect or interference with routine childhood vaccines [174]. | |
| RotaTeq® evaluated for efficacy and safety in >30,000 infants out of 70,000 infants’ enrolment for REST analysis in 5 European countries | 100% efficacy against SRVGE and 72% efficacy against any RVGE | 94.3% efficacy against SRVGE and 58.5% efficacy against any RVGE | RotaTeq® showed the efficacy of 98.3% and 68% respectively against SRVGE and all-cause of RVGE due to any serotype for the two rotavirus seasons in Europe. Further, 94.5% efficacy against RVGE hospitalisation and EV due to any serotype over 2 years of vaccination without any significant safety concern [121]. |
| Latin America | |||
| Rotarix® trial in 63,225 infants in 1-year Phase III trial from 11 Latin American and Finland | FDs produced 85% efficacy against SRVGE and hospitalisation, reaching 100% against VSRVGE with 45% efficacy in reducing any-cause of rotavirus diarrhoea hospitalisation | Two oral doses of the live attenuated G1P[8] HRV vaccine protected infants without associating with an increased risk of IS [127]. | |
| Rotarix® effectiveness in Bolivia | RV1 offered sustainable protection through two years of the life of Bolivian children, with similar VE against hospital admission <1 year and >1 year of age | RV1 offered significant protection against hospital admission for diarrhoea caused by diverse serotypes: G9P[8], G3P[8], G2P[4], and G9P[6], which are either partially and fully heterotypic to the G1P[8] vaccine [175]. This protection was sustained over two years of life against diverse serotypes different from the vaccine strain. | |
| Rotarix® introduction vs. reduction of all-cause diarrhoea death in Brazil, El Salvador, Mexico, Nicaragua, Panama, Venezuela, Bolivia, and Honduras | There was a significant reduction in all-cause diarrhoea mortality in the post-vaccination period among children <5 years old between 2006 and 2009 compared with the pre-vaccination period [176]. This reduction, 30–50%, was significant in all the vaccine adopter countries except Nicaragua. However, non-vaccine adopter countries (Argentina, Chile, Costa Rica, and Paraguay) showed a greater mortality reduction in the pre-vaccination period, 2002–2005 [176] | Significant reduction after post-vaccination in these countries was observed except in Panama, where this reduction was not observed. Concomitantly, countries such as Argentina, Chile, Costa Rica, and Paraguay were not using the vaccine at the time of this assessment [176]. Vaccine impact on the reduction of deaths was more than the hospitalisation [177]. | |
| Duration of protection offered by RotaTeq® in Nicaragua | Risk of rotavirus hospitalisation was two-fold lower in vaccinated children <1 year compared with ≥1 year | There was no significant reduction in rotavirus hospitalisation in the second year after the last vaccine dose | Waning immunity was suspected, and a booster dose was suggested in Nicaragua [178]. |
| RotaTeq® introduction vs. reduction of all-cause diarrhoea death rates in Mexico and Nicaragua | Significant reduction in all-cause diarrhoea mortality in <5 years between 2006 and 2009 when comparing postvaccination and pre-vaccination together | A significant reduction was observed in Mexico, but not in Nicaragua [176]. | |
| Africa | |||
| Rotarix® efficacy against severe diarrhoea | After the first year of follow-up, 61.2%, 58.7%, and 63.7% overall efficacy against SRVGE were observed in South Africa and Malawi combined. In South Africa, the efficacies of 2 and 3Ds were 72.2% and 81.5% and a pooled of 76.9%, while in Malawi, 49.2% and 49.7% with a pooled of 49.4% were respectively observed. Efficacy against all-cause SRVGE was 30.2%, and when pooled, South Africa was 44.1%, while Malawi was 25.1% [130] | There was a marginal reduction in vaccine efficacy in the second year of follow-up in Malawi. The 2Ds, 3Ds, and pooled efficacies against SRVGE were 2.6%, 33.1%, and 17.6% while against severe any-cause RVGE were 13.2%, 5.2%, and 9.3% respectively. There was 9.3% efficacy against all SRVGE, while the entire follow-up yielded 15.9% vaccine efficacy [179] | RV1 significantly reduced the incidence of RVGE among African infants during the first year of life. No difference in vaccine efficacy against G1 and non-G1 (G2, G8, G12) [180,181]. Madhi and co-workers observed a higher preventable episode of SRVGE/100 infants vaccinated/year in Malawi (6.7) than South Africa (4.2), though vaccine efficacy was lower in the former than the latter [130]. From Malawi, RV1 significantly reduced the incidence of SRVGE caused by diverse rotavirus strains and more episodes of SRVGE, and any SRVGE seemed to be prevented at 2Ds more than 3Ds, even after the entire follow-up [179]. Vaccine-acquired immunity seemed not to endure beyond the first year of vaccination. Three doses boost vs. 2Ds of RV1 in Malawi at the first year of follow-up produced 27.2% vs. 22.9% efficacies against all-cause SRVGE, while after the second year of follow-up, 5.2% vs. 13.2% efficacies were observed and the entire follow-up yielded 15.7% vs. 16% efficacies [179]. |
| RotaTeq® efficacy between 2007 and 2009 | Prevention of SRVGE in Ghana was 65%, Kenya 83.4%, Mali 1%, and overall was 64.2% | Prevention of SRVGE in Ghana was 29.4%, Kenya 54.7%, Mali 19.2% and overall was 19.6% | Overall efficacies of 30.5%, 39.3%, 40.7%, and 100% were observed against any severity, moderate-to-severe, severe, VSRVGE, respectively. GE was the most common adverse event associated with this vaccine, and this occurred within 14 days of any dose. There was no significant difference in this adverse events rate (0.6% each) between the vaccinated and the placebo. High IgA sero-response rates of 78.9%, 73.8%, and 82.5% were observed in Ghana, Kenya, and Mali, respectively, but very low serotype-response rates to G1–G4 and P1A[8]. Overall efficacy against moderate-to-severe RVGE was 55.5%, 63.9%, and 17.6% in Ghana, Kenya, and Mali, respectively. Generally, the efficacy of RV5 in sub-Sahara Africa was higher with higher severe cases of RVD prevented/100 vaccinated/year in the first year of follow-up than the second year, however, because of the intense mortality rates in Africa, the magnitude of prevented cases is very high than any other region [136]. Note: Infants with HIV infection were included in this study |
| RotaSIIL® efficacy assessed in 3508 infants from Niger | In a 2-stratum treatment (ITTP vs. PPP), the efficacy of 3Ds of BRV-PV against all RVGE (33% vs. 34.5%), against SRVGE (69.1% vs. 66.7%), against VSRVGE (77.4% vs. 78.8%), against all GE (−12.4% vs. −0.6%) against SGE (16.2% vs. 10.7%), and against VSGE (67.7% vs. 68%). Cases of SRVGE/100 infant vaccinated/year in the vaccine vs. placebo groups were 2.14 vs. 6.44 | At 66.7% efficacy, BRV-PV prevented 4.3 episodes of SRVGE/100 infants vaccinated/year. No significant difference in the adverse events and mortality rate. No IS cases. Efficacy increases with clinical severity. This vaccine offered protection against SRVGE resulting in mortality and morbidity of infants, and there is no need for cold storage facility (stable for 2 years at 37 °C or 6 months at 40 °C) [182]. | |
| Southeast Asia | |||
| RotaTeq® efficacy in 2036 recruited infants from Bangladesh and Vietnam | Overall efficacy against SRVGE in Bangladesh was 42.7% with the first-year follow-up of 45.7% while Vietnam was 63.9% with the first-year follow-up of 72.3% | The second-year follow-up for Bangladesh was 39.3%, and Vietnam was 64.6% | Overall efficacy of 42.5%, 48.3%, and 70% against any severity, SRVGE, and VSRVGE, respectively, for nearly 2 years of follow-up was reported. Vaccine efficacy increases with clinical severity. Higher efficacy was consistently found in Vietnam than in Bangladesh. However, efficacy against the VSRVGE (Vesikari score ≥ 15) was higher in Bangladesh because, before the vaccination, the infant mortality rate in Bangladesh (29.7/1000 live births) was twice Vietnam’s (14.7/1000 live births), and again the SRVGE cases were substantially higher in Bangladesh than Vietnam [57]. Pneumonia was one of the serious adverse events, which occurred within 14 days of any dose, and there was no significant difference in this adverse event between the vaccinated and the placebo [57]. |
| India | |||
| Rotavac® assessed for efficacy in India with 4532 infants ranging from 6 to 8-weeks-old | Efficacy of 56.4% against SRVGE with an incidence rate of 1.5 in vaccine vs. 3.2 in placebo groups. Six intussusception and 25 deaths in vaccine vs. 2 intussusception and 17 deaths in placebo groups were observed after the 3rd dose | Efficacy of 55.1% against SRVGE with an incidence rate of 1.3 in vaccine vs. 2.9 in placebo groups. Eight intussusception and 30 deaths in vaccine vs. 3 intussusception and 18 deaths in placebo groups were observed in the second year of follow-up | Whether from ITTP or PPP analysis, Rotavac® (116E) was effective and well-tolerated in Indian infants and maintained this efficacy in the second year after the 3Ds vaccination program. The reported adverse events were the same in the two groups—vaccine vs. placebo. IS occurred after 112 vs. 36 days at 1st year and 112–587 vs. 36–60 days at 2nd year, respectively. Increased seroconversion 4 weeks after the 3rd dose was observed, which suggested wildtype RVI in infants before the vaccination period [183] |
| RotaSIIL® assessed for efficacy in Phase III clinical trial in India with 3749 infants ranging from 6 to 8-weeks-old | Efficacy against VSRVGE was 60.5% with PPP analysis or 61.3% with ITTP analysis | Efficacy against VSRVGE was 54.7% with PPP analysis or 52.9% with ITTP analysis | Reactogenic response compared to placebo after 1st dose showed mild irritability (42.6% vs. 36.1%), severe fever ≥39.5–<40.5 °C (4 vs. 1 case), and LRTI (60 vs. 38 cases). All other adverse events were the same in the two arms. Overall reduction of SRVGE incidence was 1.7 cases/100 person/year in the ITTP. Generally low serum anti-rotavirus IgA seroresponses (33.6% vs. 9.7%), however, BRV-PV was well tolerated and a safe vaccine in Indian infants, but the efficacy is very low especially against SRVGE [61]. There was no vaccine interference with OPV because similar seroconversion to OPV was observed in the two arms. |
| Dose | RV3-BB | RIX4414 ≈ Rotarix® | ||||||
|---|---|---|---|---|---|---|---|---|
| IgA Response (Neonates) | Neonatal Stool Shedding | IgA Response (Infants) | Infant Stool Shedding | Vaccine Concentration (FFU/mL) | IgA Response (Infants) | Infant Stool Shedding | ||
| 1D | 2Ds | |||||||
| 1 | 23% | 4% | Placebo | - | 104.7 (with antacid) | 100% | 42% | 8% |
| 2 | 54% | 42% | 44% | 41% | 104.1 (with buffer) | 74% | 38% | 13% |
| 3 | 94% | 70% | 86% | 62% | 104.7 (with buffer) | 93% | 60% | 8% |
| 4 | Placebo | - | 99% | 76% | 105.8 (with buffer) | 96% | 55% | 0 |
| P Genogroup | P Genotype | Animals | Human | HBGAs Preference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| P[I] | P[1] | Sialic acid-dependent | ||||||||
| P[2] | ||||||||||
| P[3] | ||||||||||
| P[5] | ||||||||||
| P[7] | ||||||||||
| P[10] | ||||||||||
| P[12]–[13] | ||||||||||
| P[15]–[16] | ||||||||||
| P[18] | ||||||||||
| P[20]–[24] | ||||||||||
| P[26]–[29] | ||||||||||
| P[32]–[34] | ||||||||||
| P[II] | P[4] | Type 1 precursors, H-type 1 and Lewis b P[6] binds only H-type 1 | ||||||||
| P[6] | ||||||||||
| P[8] | ||||||||||
| P[19] | ||||||||||
| P[III] | P[9] | A-type antigen | ||||||||
| P[14] | ||||||||||
| P[25] | ||||||||||
| P[IV] | P[11] | Type 2 precursors | ||||||||
| P[V] | P[17] | - | ||||||||
| P[30]–[31] | ||||||||||
| P[35] | ||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Folorunso, O.S.; Sebolai, O.M. Overview of the Development, Impacts, and Challenges of Live-Attenuated Oral Rotavirus Vaccines. Vaccines 2020, 8, 341. https://doi.org/10.3390/vaccines8030341
Folorunso OS, Sebolai OM. Overview of the Development, Impacts, and Challenges of Live-Attenuated Oral Rotavirus Vaccines. Vaccines. 2020; 8(3):341. https://doi.org/10.3390/vaccines8030341
Chicago/Turabian StyleFolorunso, Olufemi Samuel, and Olihile M. Sebolai. 2020. "Overview of the Development, Impacts, and Challenges of Live-Attenuated Oral Rotavirus Vaccines" Vaccines 8, no. 3: 341. https://doi.org/10.3390/vaccines8030341
APA StyleFolorunso, O. S., & Sebolai, O. M. (2020). Overview of the Development, Impacts, and Challenges of Live-Attenuated Oral Rotavirus Vaccines. Vaccines, 8(3), 341. https://doi.org/10.3390/vaccines8030341





