Remodeling of the Histoplasma Capsulatum Membrane Induced by Monoclonal Antibodies
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and Treatment
2.2. Sample Extraction
2.2.1. Metabolite, Protein, and Lipid Extraction (MPLEx)
2.2.2. Solid-Phase Extraction of Lipids
2.3. Proteomic Analysis
2.3.1. Protein Digestion
2.3.2. Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) Analysis
2.3.3. Data Analysis
2.4. Lipidomic Analysis
2.5. Metabolomic Analysis
2.5.1. Chemical Derivatization
2.5.2. GC-MS Analysis
2.5.3. GC-MS Data-Processing
2.6. Susceptibility to Amphotericin B
2.7. Enzyme-Linked Iimmunosorbent Assay (ELISA) of Histoplasma capsulatum Cells
3. Results
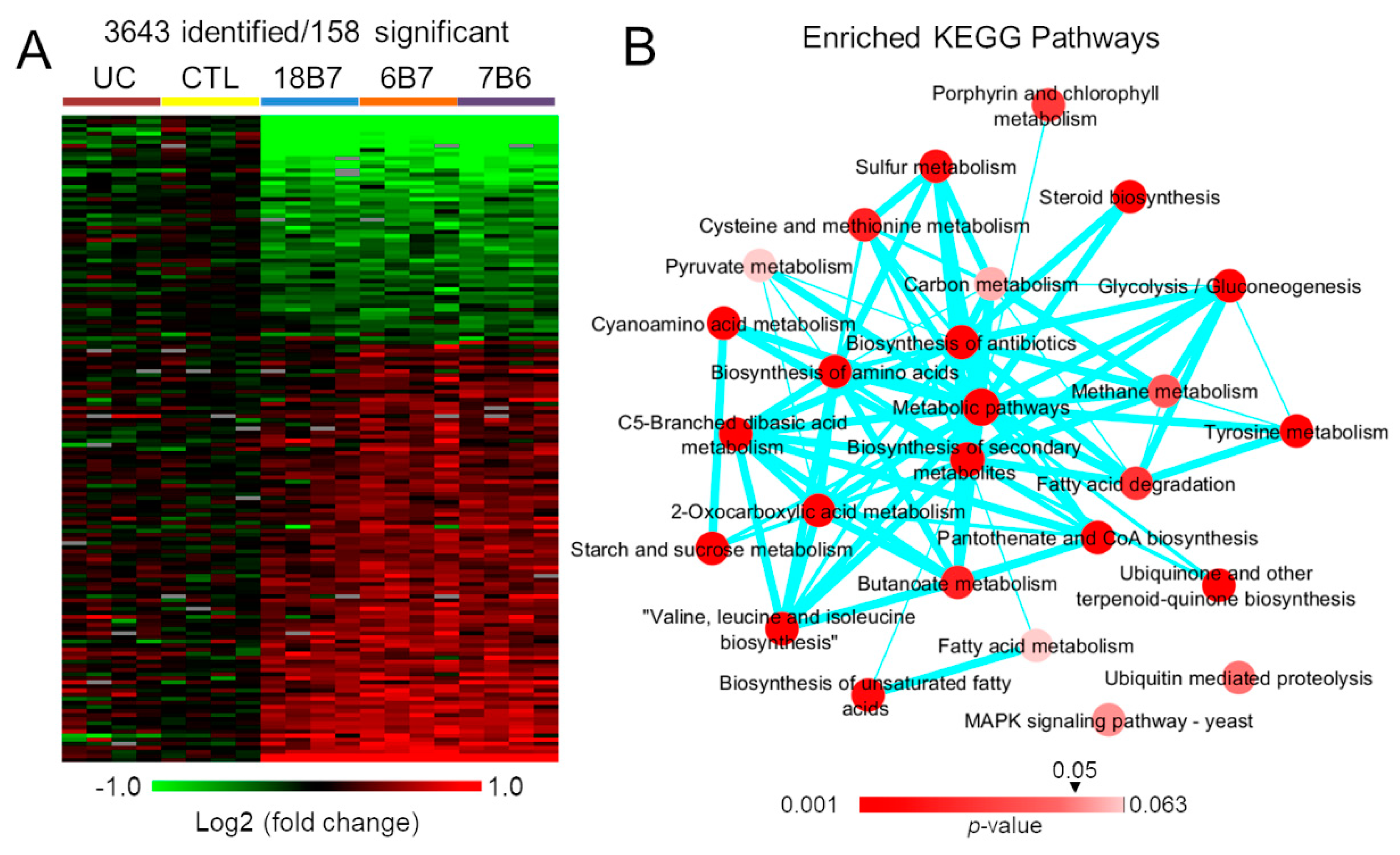
3.1. Proteomics Analysis of H. capsulatum Treated with Monoclonal Antibodies
3.2. Regulation of H. capsulatum Fatty Acid Desaturation Biosynthesis by Monoclonal Antibodies
3.3. The Impact of the Monoclonal Antibody Treatment on the Composition of Different Lipid Classes
3.4. Regulation of the H. capsulatum Sterol Metabolism by Monoclonal Antibodies
3.5. H. capsulatum Sensitivity to Amphotericin B in Cells Treated with Monoclonal Antibodies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Golay, J.; Introna, M. Mechanism of action of therapeutic monoclonal antibodies: Promises and pitfalls of in vitro and in vivo assays. Arch. Biochem. Biophys. 2012, 526, 146–153. [Google Scholar] [CrossRef]
- Martinez, L.R.; Moussai, D.; Casadevall, A. Antibody to Cryptococcus neoformans glucuronoxylomannan inhibits the release of capsular antigen. Infect. Immun. 2004, 72, 3674–3679. [Google Scholar] [CrossRef]
- Bowen, A.; Wear, M.P.; Cordero, R.J.; Oscarson, S.; Casadevall, A. A monoclonal antibody to Cryptococcus neoformans glucuronoxylomannan manifests hydrolytic activity for both peptides and polysaccharides. J. Biol. Chem. 2017, 292, 417–434. [Google Scholar] [CrossRef]
- Rodrigues, M.L.; Travassos, L.R.; Miranda, K.R.; Franzen, A.J.; Rozental, S.; de Souza, W.; Alviano, C.S.; Barreto-Bergter, E. Human antibodies against a purified glucosylceramide from Cryptococcus neoformans inhibit cell budding and fungal growth. Infect. Immun. 2000, 68, 7049–7060. [Google Scholar] [CrossRef]
- Liedke, S.C.; Miranda, D.Z.; Gomes, K.X.; Goncalves, J.L.S.; Frases, S.; Nosanchuk, J.D.; Rodrigues, M.L.; Nimrichter, L.; Peralta, J.M.; Guimaraes, A.J. Characterization of the antifungal functions of a wga-fc (igg2a) fusion protein binding to cell wall chitin oligomers. Sci. Rep. 2017, 7, 12187. [Google Scholar] [CrossRef]
- Nosanchuk, J.D.; Steenbergen, J.N.; Shi, L.; Deepe, G.S., Jr.; Casadevall, A. Antibodies to a cell surface histone-like protein protect against Histoplasma capsulatum. J. Clin. Invest. 2003, 112, 1164–1175. [Google Scholar] [CrossRef]
- Guimaraes, A.J.; Frases, S.; Gomez, F.J.; Zancope-Oliveira, R.M.; Nosanchuk, J.D. Monoclonal antibodies to heat shock protein 60 alter the pathogenesis of Histoplasma capsulatum. Infect. Immun. 2009, 77, 1357–1367. [Google Scholar] [CrossRef]
- McClelland, E.E.; Nicola, A.M.; Prados-Rosales, R.; Casadevall, A. Ab binding alters gene expression in Cryptococcus neoformans and directly modulates fungal metabolism. J. Clin. Invest. 2010, 120, 1355–1361. [Google Scholar] [CrossRef]
- Baltazar, L.M.; Zamith-Miranda, D.; Burnet, M.C.; Choi, H.; Nimrichter, L.; Nakayasu, E.S.; Nosanchuk, J.D. Concentration-dependent protein loading of extracellular vesicles released by Histoplasma capsulatum after antibody treatment and its modulatory action upon macrophages. Sci. Rep. 2018, 8, 8065. [Google Scholar] [CrossRef]
- Matos Baltazar, L.; Nakayasu, E.S.; Sobreira, T.J.; Choi, H.; Casadevall, A.; Nimrichter, L.; Nosanchuk, J.D. Antibody binding alters the characteristics and contents of extracellular vesicles released by Histoplasma capsulatum. mSphere 2016, 1. [Google Scholar] [CrossRef]
- Casadevall, A.; Cleare, W.; Feldmesser, M.; Glatman-Freedman, A.; Goldman, D.L.; Kozel, T.R.; Lendvai, N.; Mukherjee, J.; Pirofski, L.A.; Rivera, J.; et al. Characterization of a murine monoclonal antibody to Cryptococcus neoformans polysaccharide that is a candidate for human therapeutic studies. Antimicrob. Agents Chemother. 1998, 42, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Zamith-Miranda, D.; Heyman, H.M.; Burnet, M.C.; Couvillion, S.P.; Zheng, X.; Munoz, N.; Kyle, J.E.; Zink, E.M.; Weitz, K.K.; Bloodsworth, K.J.; et al. A Histoplasma capsulatum lipid metabolic map identifies antifungal targets and virulence factors. bioRxiv 2002, 2020.2003.2002.973412. [Google Scholar]
- Nakayasu, E.S.; Nicora, C.D.; Sims, A.C.; Burnum-Johnson, K.E.; Kim, Y.M.; Kyle, J.E.; Matzke, M.M.; Shukla, A.K.; Chu, R.K.; Schepmoes, A.A.; et al. Mplex: A robust and universal protocol for single-sample integrative proteomic, metabolomic, and lipidomic analyses. mSystems 2016, 1. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Cox, J. The maxquant computational platform for mass spectrometry-based shotgun proteomics. Nat. Protoc. 2016, 11, 2301–2319. [Google Scholar] [CrossRef]
- Cox, J.; Hein, M.Y.; Luber, C.A.; Paron, I.; Nagaraj, N.; Mann, M. Accurate proteome-wide label-free quantification by delayed normalization and maximal peptide ratio extraction, termed maxlfq. Mol. Cell Proteomics 2014, 13, 2513–2526. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Morishima, K. Blastkoala and ghostkoala: Kegg tools for functional characterization of genome and metagenome sequences. J. Mol. Biol. 2016, 428, 726–731. [Google Scholar] [CrossRef]
- Merico, D.; Isserlin, R.; Stueker, O.; Emili, A.; Bader, G.D. Enrichment map: A network-based method for gene-set enrichment visualization and interpretation. PLoS ONE 2010, 5, e13984. [Google Scholar] [CrossRef]
- Dautel, S.E.; Kyle, J.E.; Clair, G.; Sontag, R.L.; Weitz, K.K.; Shukla, A.K.; Nguyen, S.N.; Kim, Y.M.; Zink, E.M.; Luders, T.; et al. Lipidomics reveals dramatic lipid compositional changes in the maturing postnatal lung. Sci. Rep. 2017, 7, 40555. [Google Scholar] [CrossRef]
- Kyle, J.E.; Crowell, K.L.; Casey, C.P.; Fujimoto, G.M.; Kim, S.; Dautel, S.E.; Smith, R.D.; Payne, S.H.; Metz, T.O. Liquid: An-open source software for identifying lipids in lc-ms/ms-based lipidomics data. Bioinformatics 2017, 33, 1744–1746. [Google Scholar] [CrossRef]
- Pluskal, T.; Castillo, S.; Villar-Briones, A.; Oresic, M. Mzmine 2: Modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinform. 2010, 11, 395. [Google Scholar] [CrossRef]
- Kim, Y.M.; Schmidt, B.J.; Kidwai, A.S.; Jones, M.B.; Deatherage Kaiser, B.L.; Brewer, H.M.; Mitchell, H.D.; Palsson, B.O.; McDermott, J.E.; Heffron, F.; et al. Salmonella modulates metabolism during growth under conditions that induce expression of virulence genes. Mol. Biosyst. 2013, 9, 1522–1534. [Google Scholar] [CrossRef]
- Romine, M.F.; Rodionov, D.A.; Maezato, Y.; Anderson, L.N.; Nandhikonda, P.; Rodionova, I.A.; Carre, A.; Li, X.; Xu, C.; Clauss, T.R.; et al. Elucidation of roles for vitamin b12 in regulation of folate, ubiquinone, and methionine metabolism. Proc. Natl. Acad. Sci. USA 2017, 114, E1205–E1214. [Google Scholar] [CrossRef]
- Hiller, K.; Hangebrauk, J.; Jager, C.; Spura, J.; Schreiber, K.; Schomburg, D. Metabolitedetector: Comprehensive analysis tool for targeted and nontargeted gc/ms based metabolome analysis. Anal. Chem. 2009, 81, 3429–3439. [Google Scholar] [CrossRef]
- Kind, T.; Wohlgemuth, G.; Lee, D.Y.; Lu, Y.; Palazoglu, M.; Shahbaz, S.; Fiehn, O. Fiehnlib: Mass spectral and retention index libraries for metabolomics based on quadrupole and time-of-flight gas chromatography/mass spectrometry. Anal. Chem. 2009, 81, 10038–10048. [Google Scholar] [CrossRef]
- Xia, J.; Sinelnikov, I.V.; Han, B.; Wishart, D.S. Metaboanalyst 3.0--making metabolomics more meaningful. Nucleic Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef]
- Tang, G.Q.; Novitzky, W.P.; Carol Griffin, H.; Huber, S.C.; Dewey, R.E. Oleate desaturase enzymes of soybean: Evidence of regulation through differential stability and phosphorylation. Plant J. 2005, 44, 433–446. [Google Scholar] [CrossRef]
- Covino, R.; Ballweg, S.; Stordeur, C.; Michaelis, J.B.; Puth, K.; Wernig, F.; Bahrami, A.; Ernst, A.M.; Hummer, G.; Ernst, R. A eukaryotic sensor for membrane lipid saturation. Mol. Cell 2016, 63, 49–59. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Sakumoto, N.; Kaneko, Y.; Harashima, S. Mga2p is a putative sensor for low temperature and oxygen to induce ole1 transcription in Saccharomyces cerevisiae. Biochem. Biophys. Res. Commun. 2002, 291, 707–713. [Google Scholar] [CrossRef]
- Martin, C.E.; Oh, C.S.; Kandasamy, P.; Chellapa, R.; Vemula, M. Yeast desaturases. Biochem. Soc. Trans. 2002, 30, 1080–1082. [Google Scholar] [CrossRef]
- Zhang, S.; Skalsky, Y.; Garfinkel, D.J. Mga2 or spt23 is required for transcription of the delta9 fatty acid desaturase gene, ole1, and nuclear membrane integrity in Saccharomyces cerevisiae. Genetics 1999, 151, 473–483. [Google Scholar]
- O’Quin, J.B.; Bourassa, L.; Zhang, D.; Shockey, J.M.; Gidda, S.K.; Fosnot, S.; Chapman, K.D.; Mullen, R.T.; Dyer, J.M. Temperature-sensitive post-translational regulation of plant omega-3 fatty-acid desaturases is mediated by the endoplasmic reticulum-associated degradation pathway. J. Biol. Chem. 2010, 285, 21781–21796. [Google Scholar] [CrossRef]
- Carratu, L.; Franceschelli, S.; Pardini, C.L.; Kobayashi, G.S.; Horvath, I.; Vigh, L.; Maresca, B. Membrane lipid perturbation modifies the set point of the temperature of heat shock response in yeast. Proc. Natl. Acad. Sci. USA 1996, 93, 3870–3875. [Google Scholar] [CrossRef] [PubMed]
- Gargano, S.; Di Lallo, G.; Kobayashi, G.S.; Maresca, B. A temperature-sensitive strain of Histoplasma capsulatum has an altered delta 9-fatty acid desaturase gene. Lipids 1995, 30, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Cipak, A.; Jaganjac, M.; Tehlivets, O.; Kohlwein, S.D.; Zarkovic, N. Adaptation to oxidative stress induced by polyunsaturated fatty acids in yeast. Biochim. Biophys. Acta 2008, 1781, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Yazawa, H.; Iwahashi, H.; Kamisaka, Y.; Kimura, K.; Uemura, H. Production of polyunsaturated fatty acids in yeast Saccharomyces cerevisiae and its relation to alkaline ph tolerance. Yeast 2009, 26, 167–184. [Google Scholar] [CrossRef]
- Rodriguez-Vargas, S.; Sanchez-Garcia, A.; Martinez-Rivas, J.M.; Prieto, J.A.; Randez-Gil, F. Fluidization of membrane lipids enhances the tolerance of Saccharomyces cerevisiae to freezing and salt stress. Appl. Environ. Microbiol. 2007, 73, 110–116. [Google Scholar] [CrossRef]
- Nasution, O.; Lee, Y.M.; Kim, E.; Lee, Y.; Kim, W.; Choi, W. Overexpression of ole1 enhances stress tolerance and constitutively activates the mapk hog pathway in Saccharomyces cerevisiae. Biotechnol. Bioeng. 2017, 114, 620–631. [Google Scholar] [CrossRef]
- Casanovas, A.; Sprenger, R.R.; Tarasov, K.; Ruckerbauer, D.E.; Hannibal-Bach, H.K.; Zanghellini, J.; Jensen, O.N.; Ejsing, C.S. Quantitative analysis of proteome and lipidome dynamics reveals functional regulation of global lipid metabolism. Chem. Biol. 2015, 22, 412–425. [Google Scholar] [CrossRef]
- Tuller, G.; Hrastnik, C.; Achleitner, G.; Schiefthaler, U.; Klein, F.; Daum, G. Ydl142c encodes cardiolipin synthase (cls1p) and is non-essential for aerobic growth of Saccharomyces cerevisiae. FEBS Lett. 1998, 421, 15–18. [Google Scholar] [CrossRef]
- Li, Q.Q.; Tsai, H.F.; Mandal, A.; Walker, B.A.; Noble, J.A.; Fukuda, Y.; Bennett, J.E. Sterol uptake and sterol biosynthesis act coordinately to mediate antifungal resistance in Candida glabrata under azole and hypoxic stress. Mol. Med. Rep. 2018, 17, 6585–6597. [Google Scholar]
- Nagi, M.; Tanabe, K.; Nakayama, H.; Yamagoe, S.; Umeyama, T.; Oura, T.; Ohno, H.; Kajiwara, S.; Miyazaki, Y. Serum cholesterol promotes the growth of Candida glabrata in the presence of fluconazole. J. Infect Chemother. 2013, 19, 138–143. [Google Scholar] [CrossRef] [PubMed]
- DuBois, J.C.; Smulian, A.G. Sterol regulatory element binding protein (srb1) is required for hypoxic adaptation and virulence in the dimorphic fungus Histoplasma capsulatum. PLoS ONE 2016, 11, e0163849. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, A.J.; de Cerqueira, M.D.; Nosanchuk, J.D. Surface architecture of Histoplasma capsulatum. Front. Microbiol. 2011, 2, 225. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burnet, M.C.; Zamith-Miranda, D.; Heyman, H.M.; Weitz, K.K.; Bredeweg, E.L.; Nosanchuk, J.D.; Nakayasu, E.S. Remodeling of the Histoplasma Capsulatum Membrane Induced by Monoclonal Antibodies. Vaccines 2020, 8, 269. https://doi.org/10.3390/vaccines8020269
Burnet MC, Zamith-Miranda D, Heyman HM, Weitz KK, Bredeweg EL, Nosanchuk JD, Nakayasu ES. Remodeling of the Histoplasma Capsulatum Membrane Induced by Monoclonal Antibodies. Vaccines. 2020; 8(2):269. https://doi.org/10.3390/vaccines8020269
Chicago/Turabian StyleBurnet, Meagan C., Daniel Zamith-Miranda, Heino M. Heyman, Karl K. Weitz, Erin L. Bredeweg, Joshua D. Nosanchuk, and Ernesto S. Nakayasu. 2020. "Remodeling of the Histoplasma Capsulatum Membrane Induced by Monoclonal Antibodies" Vaccines 8, no. 2: 269. https://doi.org/10.3390/vaccines8020269
APA StyleBurnet, M. C., Zamith-Miranda, D., Heyman, H. M., Weitz, K. K., Bredeweg, E. L., Nosanchuk, J. D., & Nakayasu, E. S. (2020). Remodeling of the Histoplasma Capsulatum Membrane Induced by Monoclonal Antibodies. Vaccines, 8(2), 269. https://doi.org/10.3390/vaccines8020269





