Success and Limitation of Equine Influenza Vaccination: The First Incursion in a Decade of a Florida Clade 1 Equine Influenza Virus that Shakes Protection Despite High Vaccine Coverage
Abstract
1. Introduction
2. Materials and Methods
2.1. Serum Samples
2.2. Single Radial Haemolysis (SRH)
2.3. EIV NP ELISA (DIVA Test)
2.4. EIV Genes Sequencing and Phylogenetic Analysis
2.5. Statistical Analysis
3. Results
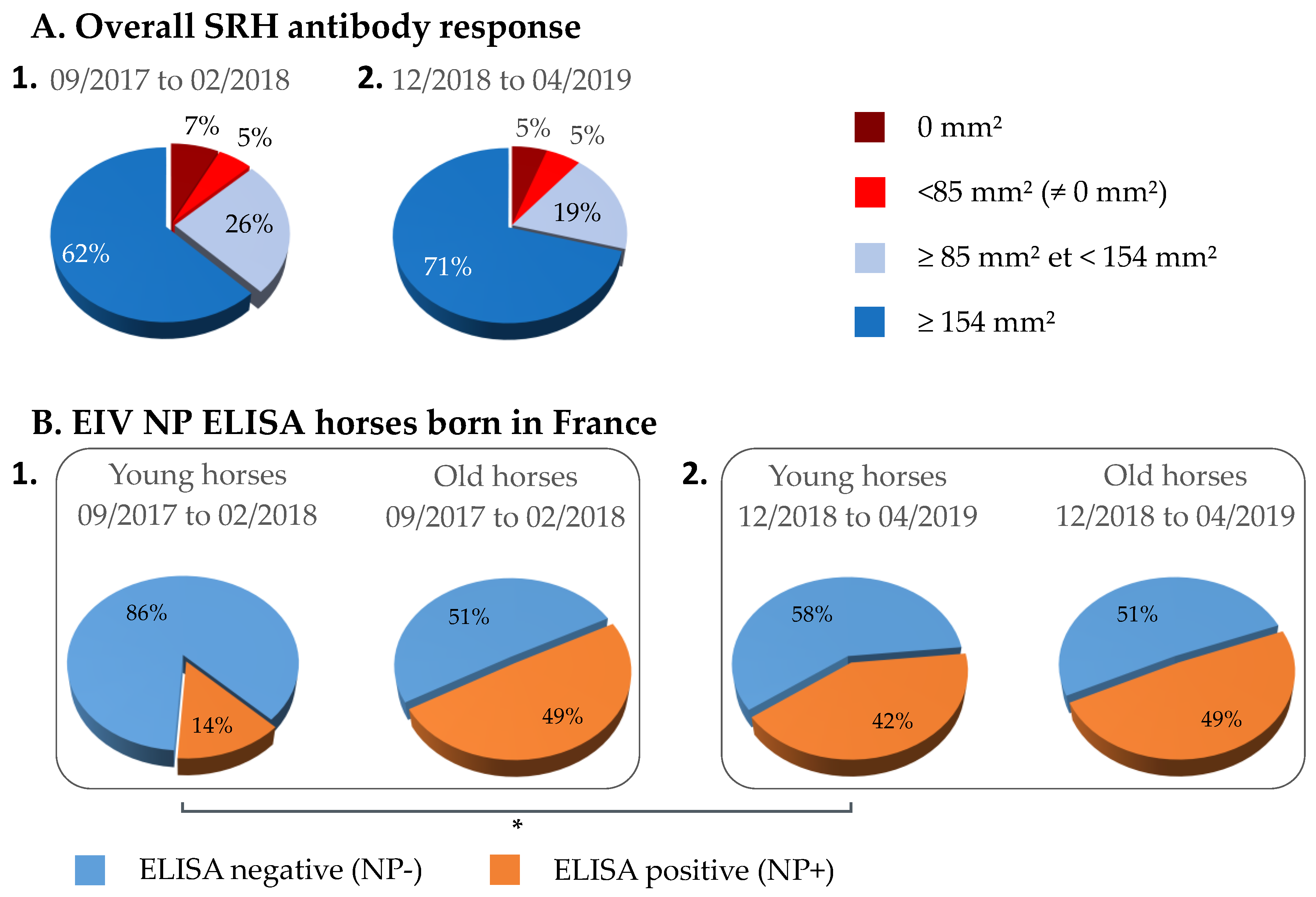
3.1. Overall SRH Antibody Response and Correlation with EI Immune Status
3.2. SRH Antibody Analysis by Geographic Region and Category
3.3. NP-ELISA Assays as DIVA (Differentiating Infected from Vaccinated Animals) Test
3.4. 2018–2019 Equine Influenza Outbreaks, Phylogenetic Characterization and Relationship with Immune Status
3.4.1. Equine Influenza Outbreak
3.4.2. Phylogenetic Characterization
3.4.3. 2018–2019 EI Outbreaks and EI Vaccination Status
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paillot, R.; El-Hage, C.M. The use of a recombinant canarypox-based equine influenza vaccine during the 2007 australian outbreak: A systematic review and summary. Pathogens 2016, 5, 42. [Google Scholar] [CrossRef]
- Heldens, J.G.; Pouwels, H.G.; Derks, C.G.; Van de Zande, S.M.; Hoeijmakers, M.J. Duration of immunity induced by an equine influenza and tetanus combination vaccine formulation adjuvanted with iscom-matrix. Vaccine 2010, 28, 6989–6996. [Google Scholar] [CrossRef]
- Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. A comparison of antibody responses to commercial equine influenza vaccines following primary vaccination of thoroughbred weanlings—A randomised blind study. Vaccine 2011, 29, 9214–9223. [Google Scholar] [CrossRef]
- Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. A comparison of antibody responses to commercial equine influenza vaccines following annual booster vaccination of national hunt horses—A randomised blind study. Vaccine 2011, 29, 3917–3922. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R. A systematic review of recent advances in equine influenza vaccination. Vaccines 2014, 2, 797–831. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Announcement: Final 2016-17 influenza vaccination coverage estimates available online. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1028. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chiu, A.P.Y.; Dushoff, J.; Yu, D.; He, D. Patterns of influenza vaccination coverage in the united states from 2009 to 2015. Int. J. Infect. Dis. 2017, 65, 122–127. [Google Scholar] [CrossRef]
- Marshall, E.; Salmon, D.; Bousfiha, N.; Togola, Y.; Ouedraogo, F.; Santantonio, M.; Dieng, C.K.; Tartiere, S.; Emmanuelli, X. Vaccination coverage among social and healthcare workers in ten countries of samu-social international sites. Vaccine 2017, 35, 5291–5296. [Google Scholar] [CrossRef]
- Seo, J.; Lim, J. Trends in influenza vaccination coverage rates in south korea from 2005 to 2014: Effect of public health policies on vaccination behavior. Vaccine 2018, 36, 3666–3673. [Google Scholar] [CrossRef]
- Uchida, M.; Kaneko, M.; Hidaka, Y.; Yamamoto, H.; Honda, T.; Takeuchi, S.; Saito, M.; Kawa, S. High vaccination coverage is associated with low epidemic level of seasonal influenza in elementary schools: An observational study in matsumoto city, Japan. BMC Infect. Dis. 2018, 18, 128. [Google Scholar] [CrossRef]
- Ireland, J.L.; Wylie, C.E.; Collins, S.N.; Verheyen, K.L.; Newton, J.R. Preventive health care and owner-reported disease prevalence of horses and ponies in great britain. Res. Vet. Sci. 2013, 95, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Satou, K.; Nishiura, H. Basic reproduction number for equine-2 influenza virus a (h3n8) epidemic in racehorse facilities in japan, 1971. J. Equine Vet. Sci. 2006, 26, 310–316. [Google Scholar] [CrossRef]
- Fougerolle, S.; Legrand, L.; Lecouturier, F.; Sailleau, C.; Paillot, R.; Hans, A.; Pronost, S. Genetic evolution of equine influenza virus strains (h3n8) isolated in france from 1967 to 2015 and the implications of several potential pathogenic factors. Virology 2017, 505, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Fitzpatrick, D.A.; Cullinane, A. Epidemiological and virological investigations of equine influenza outbreaks in ireland (2010–2012). Influenza Other Respir. Viruses 2013, 7 (Suppl. 4), 61–72. [Google Scholar] [CrossRef] [PubMed]
- Rash, A.; Morton, R.; Woodward, A.; Maes, O.; McCauley, J.; Bryant, N.; Elton, D. Evolution and divergence of h3n8 equine influenza viruses circulating in the united kingdom from 2013 to 2015. Pathogens 2017, 6, 6. [Google Scholar] [CrossRef]
- Favaro, P.F.; Fernandes, W.R.; Reischak, D.; Brandao, P.E.; Silva, S.O.S.; Richtzenhain, L.J. Evolution of equine influenza viruses (h3n8) during a brazilian outbreak, 2015. Braz. J. Microbiol. 2018, 49, 336–346. [Google Scholar] [CrossRef]
- Singh, R.K.; Dhama, K.; Karthik, K.; Khandia, R.; Munjal, A.; Khurana, S.K.; Chakraborty, S.; Malik, Y.S.; Virmani, N.; Singh, R.; et al. A comprehensive review on equine influenza virus: Etiology, epidemiology, pathobiology, advances in developing diagnostics, vaccines, and control strategies. Front. Microbiol. 2018, 9, 1941. [Google Scholar] [CrossRef]
- Woodward, A.L.; Rash, A.S.; Blinman, D.; Bowman, S.; Chambers, T.M.; Daly, J.M.; Damiani, A.; Joseph, S.; Lewis, N.; McCauley, J.W.; et al. Development of a surveillance scheme for equine influenza in the uk and characterisation of viruses isolated in europe, dubai and the USA from 2010–2012. Vet. Microbiol. 2014, 169, 113–127. [Google Scholar] [CrossRef]
- Paillot, R.; Pitel, P.H.; Pronost, S.; Legrand, L.; Fougerolle, S.; Jourdan, M.; Marcillaud-Pitel, C. Florida clade 1 equine influenza virus in france. Vet. Rec. 2019, 184, 101. [Google Scholar] [CrossRef]
- Mena, J.; Brito, B.; Moreira, R.; Tadich, T.; Gonzalez, I.; Cruces, J.; Ortega, R.; van Bakel, H.; Rathnasinghe, R.; Pizarro-Lucero, J.; et al. Reemergence of h3n8 equine influenza a virus in chile, 2018. Transbound. Emerg. Dis. 2018, 65, 1408–1415. [Google Scholar] [CrossRef]
- Daly, J.; Daas, A.; Behr-Gross, M.E. Collaborative study for the establishment of a candidate equine influenza subtype 2 american-like strain a/eq/south africa/4/03-horse antiserum biological reference preparation. Pharmeuropa Bio 2007, 2007, 7–14. [Google Scholar] [PubMed]
- Mumford, J.A.; Wood, J. Establishing an acceptability threshold for equine influenza vaccines. Dev. Biol. Stand. 1992, 79, 137–146. [Google Scholar] [PubMed]
- Mumford, J.A.; Jessett, D.M.; Dunleavy, U.; Wood, J.L.N.; Hannant, D.; Sundquist, B.; Cook, R.F. Antigenicity and immunogenicity of experimental equine influenza iscom vaccines. Vaccine 1994, 12, 857–863. [Google Scholar] [CrossRef]
- Rash, A.; Woodward, A.; Bryant, N.; McCauley, J.; Elton, D. An efficient genome sequencing method for equine influenza [h3n8] virus reveals a new polymorphism in the pa-x protein. Virol. J. 2014, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. Mega7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Gildea, S.; Arkins, S.; Cullinane, A. Management and environmental factors involved in equine influenza outbreaks in ireland 2007–2010. Equine Vet. J. 2011, 43, 608–617. [Google Scholar] [CrossRef]
- Fougerolle, S.; Legrand, L.; Garrett, D.; Birand, I.; Foursin, M.; D’Ablon, X.; Bayssat, P.; Newton, R.J.; Pronost, S.; Paillot, R. Influential factors inducing suboptimal humoral response to vector-based influenza immunisation in thoroughbred foals. Vaccine 2016, 34, 3787–3795. [Google Scholar] [CrossRef]
- Paillot, R.; Garrett, D.; Lopez-Alvarez, M.R.; Birand, I.; Montesso, F.; Horspool, L. The immunity gap challenge: Protection against a recent florida clade 2 equine influenza strain. Vaccines 2018, 6, 38. [Google Scholar] [CrossRef]
- Heldens, J.G.; Pouwels, H.G.; Derks, C.G.; Van de Zande, S.M.; Hoeijmakers, M.J. The first safe inactivated equine influenza vaccine formulation adjuvanted with iscom-matrix that closes the immunity gap. Vaccine 2009, 27, 5530–5537. [Google Scholar] [CrossRef]
- Heldens, J.G.; van Loon, A.A.; van de Zande, S. Is there a benefit from an early booster vaccination in the control of equine influenza? Vet. J. 2007, 174, 592–598. [Google Scholar] [CrossRef]
- Galvin, P.; Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. The evaluation of a nucleoprotein elisa for the detection of equine influenza antibodies and the differentiation of infected from vaccinated horses (diva). Influenza Other Respir. Viruses 2013, 7 (Suppl. 4), 73–80. [Google Scholar] [CrossRef]
- Perglione, C.O.; Gildea, S.; Rimondi, A.; Mino, S.; Vissani, A.; Carossino, M.; Cullinane, A.; Barrandeguy, M. Epidemiological and virological findings during multiple outbreaks of equine influenza in south america in 2012. Influenza Other Respir. Viruses 2016, 10, 37–46. [Google Scholar] [CrossRef]
- OIE Expert Surveillance Panel on Equine Influenza Vaccine Composition. OIE Bull. 2019, 2, 2–7. Available online: https://oiebulletin.com/index.php?officiel=08-4-1-2019-2-panel-en&edition=7913&pdf=officiel&article=11019 (accessed on 15 October 2019).
- Garrett, D.; Montesso, F.; Fougerolle, S.; Lopez-Alvarez, M.R.; Birand, I.; De Bock, M.; Huang, C.M.; Legrand, L.; Pronost, S.; Paillot, R. Refinement of the equine influenza model in the natural host: A meta-analysis to determine the benefits of individual nebulisation for experimental infection and vaccine evaluation in the face of decreased strain pathogenicity. Vet. Microbiol. 2017, 211, 150–159. [Google Scholar] [CrossRef]
- Paillot, R.; Rash, N.L.; Garrett, D.; Prowse-Davis, L.; Montesso, F.; Cullinane, A.; Lemaitre, L.; Thibault, J.C.; Wittreck, S.; Dancer, A. How to meet the last oie expert surveillance panel recommendations on equine influenza (ei) vaccine composition: A review of the process required for the recombinant canarypox-based ei vaccine. Pathogens 2016, 5, 64. [Google Scholar] [CrossRef]
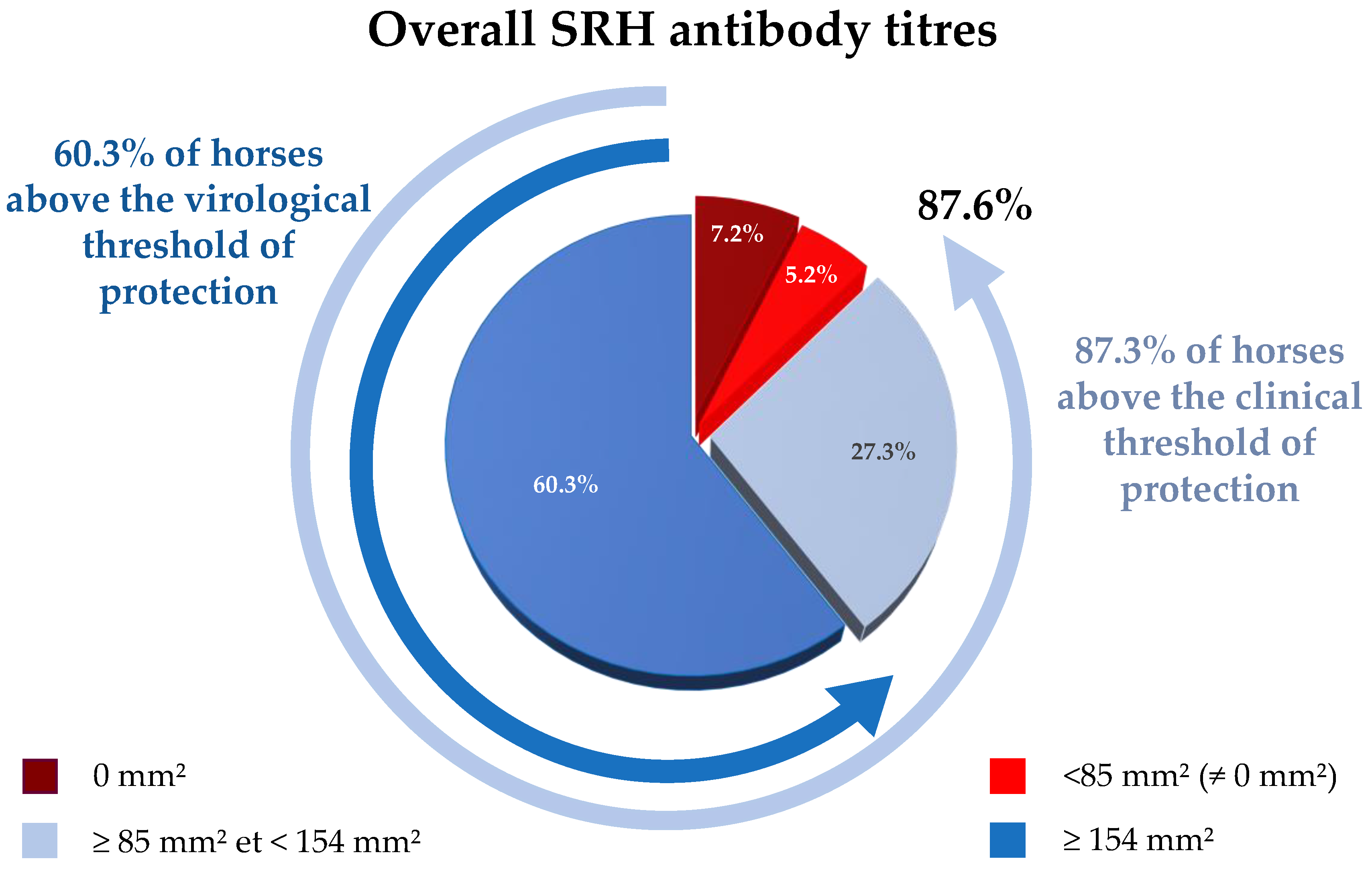

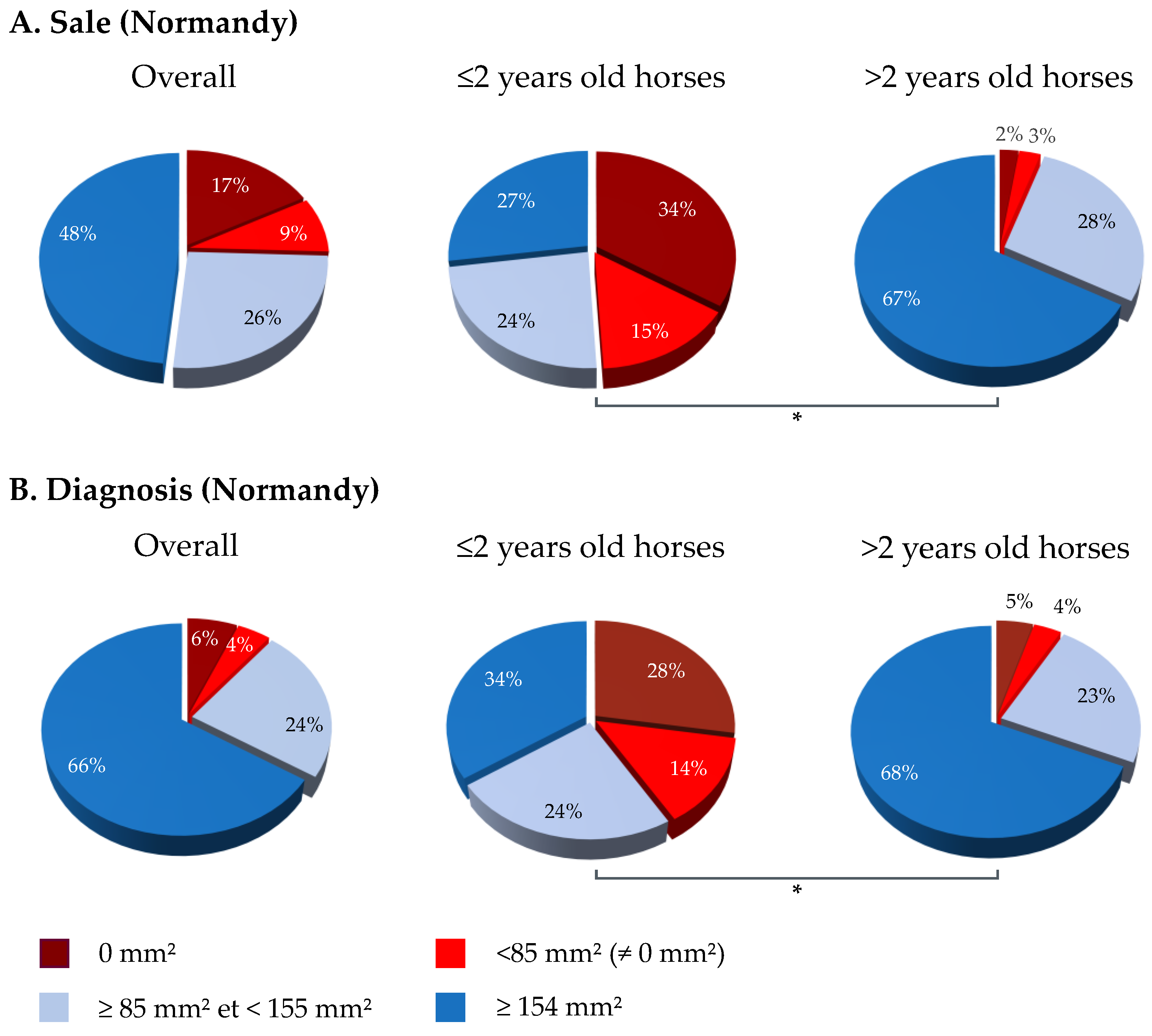

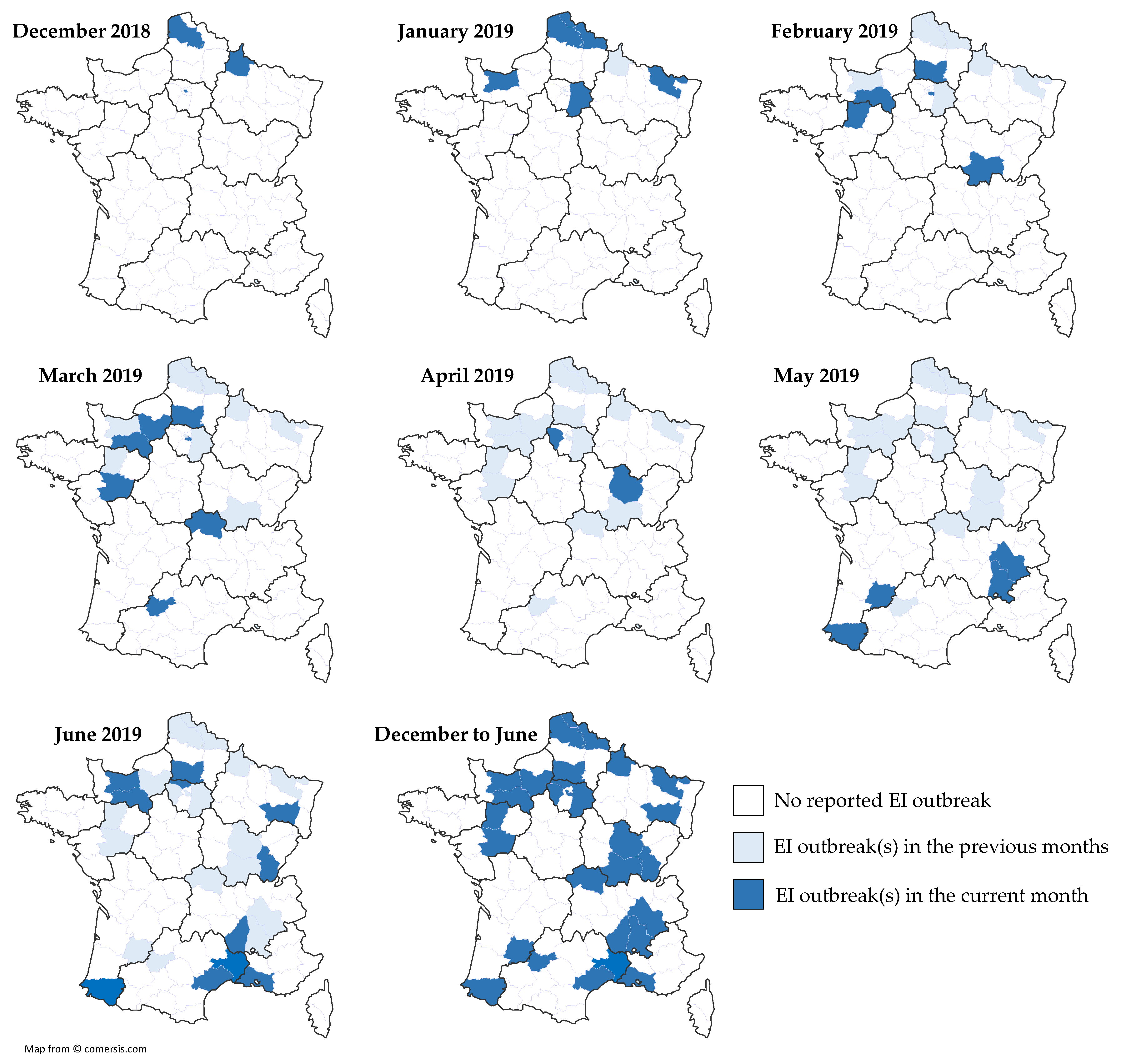
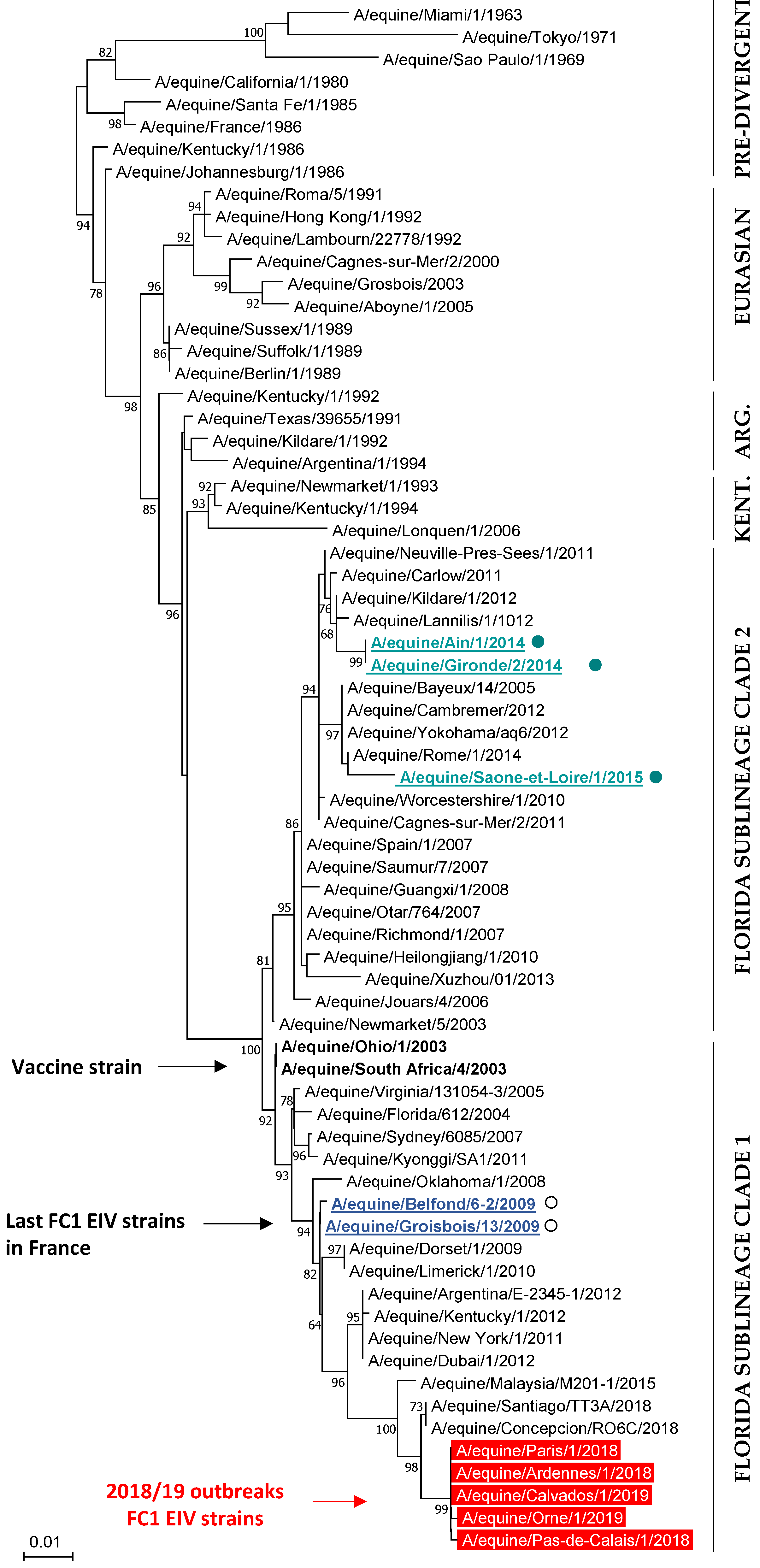
| N | Date | Location | Type | Nb. Horses/P | Nb. of Clinical Cases | Vaccination Status | ||
|---|---|---|---|---|---|---|---|---|
| #1 | 14DEC18 | Paris | Riding school | 100 | 9 | Yes | ||
| #2 | 21DEC18 | Pas de Calais | French Saddlebred | 50 | 14 | No | ||
| #3 | 28DEC18 | Ardennes | sport | unknown | 3 | No (young) | ||
| #4 | 02JAN19 | Nord | sport | 100 | 5 | Yes | ||
| #5 | 10JAN19 | Pas de Calais | Draft horse | 40 | 1 | No | ||
| #6 | 11JAN19 | Pas de Calais | Belgian Warmblood, Connemara, French Saddlebred | 300 | 2 | Yes | ||
| #7 | 11JAN19 | Pas de Calais | French Saddlebred | 20 | 3 | unknown | ||
| #8 | 11JAN19 | Moselle | sport | 130 | 3 | unknown | ||
| #9 | 16JAN19 | Calvados | unknown | unknown | 2 | unknown | ||
| #10 | 16JAN19 | Seine et Marne | Zangersheide | 40 | 1 | yes | ||
| #11 | 07FEB19 | Orne | French Trotters | 50 | 12 | yes | ||
| #12 | 13FEB19 | Val de Marne | TC French Trotters | 15 | 2 | yes | ||
| #13 | 18FEB19 | Saone et Loire | Paint horses, Arab. | 15 | 5 | yes | ||
| #14 | 20FEB19 | Orne | Stud farm TB | unknown | 2 | yes (lapsed) | ||
| #15 | 20FEB19 | Mayenne | TC Trotters | 150 | 10 | yes | ||
| #16 | 20FEB19 | Val-de-Marne | TC Trotters | 1 | 1 | yes | ||
| #17 | 26FEB19 | Oise | TC TB | 12 | 10 | unknown | ||
| #18 | 27FEB19 | Oise | TC TB | unknown | 2 | unknown | ||
| #19 | 28FEB19 | Val-de-Marne | TC Trotters | unknown | 3 | yes | ||
| #20 | 07MAR19 | Orne | Trotter | unknown | 1 | yes | ||
| #21 | 07MAR19 | Oise | TB | unknown | 2 (no clin.) | unknown | ||
| #22 | 08MAR19 | Val-de-Marne | Trotters | unknown | 4 | unknown | ||
| #23 | 09MAR19 | Orne | TB | unknown | 2 | yes | ||
| #24 | 15MAR19 | Oise | TB | unknown | 2 | unknown | ||
| #25 | 18MAR19 | Val-de-Marne | Gelding | 15 | 1 | yes | ||
| #26 | 20MAR19 | Orne | Trotters | 70 | 63 | unknown | ||
| #27 | 20MAR19 | Eure | unknown | unknown | 2 | unknown | ||
| #28 | 20MAR19 | Oise | TB | unknown | 1 | unknown | ||
| #29 | 20MAR19 | Allier | unknown | unknown | 1 | unknown | ||
| #30 | 26MAR19 | Maine et Loire | Trotters | unknown | 1 | unknown | ||
| #31 | 27MAR19 | Allier | unknown | unknown | 1 | unknown | ||
| #32 | 29MAR19 | Allier | TC TB | unknown | 2 | unknown | ||
| #33 | 29MAR19 | Tarn et Garonne | unknown | unknown | 1 | unknown | ||
| #34 | 08APR19 | Yvellines | unknown | unknown | 2 | unknown | ||
| #35 | 26APR19 | Côte d’Or | Sport | unknown | 9 | unknown | ||
| #36 | 06MAY19 | Isère | Leisure center | 4 | 1 | no | ||
| #37 | 17MAY19 | Lot et Garonne | Sport Arab. | 15 | 8 | unknown | ||
| #38 | 24MAY19 | Drôme | Breeding center, pony | 8 | 7 | no | ||
| #39 | 31MAY19 | Pyrénées Atlantiques | Donkey | unknown | 1 | unknown | ||
| #40 | 01JUN19 | Bouches du Rhône | unknown | unknown | 1 | unknown | ||
| #41 | 07JUN19 | Jura | Heavy horse | 15 | 15 | no | ||
| #42 | 11JUN19 | Hérault | unknown | unknown | 1 | unknown | ||
| #43 | 12JUN19 | Vosges | Heavy horse | 1 | 1 | no | ||
| #44 | 12JUN19 | Oise | unknown | unknown | unknown | unknown | ||
| #45 | 12JUN19 | Val d’Oise | unknown | unknown | unknown | unknown | ||
| #46 | 12JUN19 | Ardèche | Leisure center | 30 | 4 | yes | ||
| #47 | 12JUN19 | Orne | French Trotters | 28 | 10 | yes | ||
| #48 | 19JUN19 | Calvados | unknown | unknown | 3 | unknown | ||
| #49 | 22JUN19 | Orne | unknown | unknown | 1 | unknown | ||
| #50 | 26JUN19 | Orne | unknown | unknown | 6 | unknown | ||
| #51 | 27JUN19 | Gard | Camargue | 3 | 2 | no | ||
| #52 | 28JUN19 | Pyrénées Atlantiques | unknown | unknown | 1 † | unknown | ||
| #53 | 28JUN19 | Orne | unknown | unknown | 1 | unknown | ||
| Number of counties/total 1 | 29/96 | Total clinical cases | 246 | |||||
| Horse ID | Time Since Last Vaccination (V) | V-1/VT (Months between V and V-1) | V-2/VT (Months between V-1 and V-2) | SRH (mm2) 1 | EIV NP ELISA DIVA | Outbreak | |||
|---|---|---|---|---|---|---|---|---|---|
| Months | VT | Months | VT | Months | VT | ||||
| #1 | 1 | C | 9.5 | C | 7 | W1 | 115.9 1 | pos | #1 |
| #2 | 1 | W1 | 12 | C | 8.5 | C | nd | nd | #10 |
| #3 | 2 | C | 6 | C | 6 | C | 70.6 1 | neg | #1 |
| #4 | 2 | C | 6.5 | C | 1 | C | 74.05 1 | neg | #1 |
| #5 | 3 | C | 2 | C | 12 | C | 105.9 1 | neg | #1 |
| #6 | 3 | C | 11 | C | 6 | C | 177.2 2 | neg | #19 |
| #7 | 3.5 | C | 1 | C | na | na | 0 1 | borderline | #1 |
| #8 | 4 | C | 15 | C | 12 | C | 79.2 2 | neg | #19 |
| #9 | 6 | C | 12 | C | 11.5 | W2 | 66.9 1 | nd | #1 |
| #10 | 9 | C | 12 | C | 1.5 | C | 129.5 1 | neg | #1 |
| #11 | 9 | C | 11.5 | C | 6 | C | 93.9 2 | neg | #19 |
| #12 | 9.5 | C | 12 | C | 12 | C | nd | nd | #6 |
| #13 | 11 | C | 12 | C | 7 | C | 45.6 1 | neg | #1 |
| #14 | 19.5 | C | 12 | C | 12 | C | nd | nd | #6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fougerolle, S.; Fortier, C.; Legrand, L.; Jourdan, M.; Marcillaud-Pitel, C.; Pronost, S.; Paillot, R. Success and Limitation of Equine Influenza Vaccination: The First Incursion in a Decade of a Florida Clade 1 Equine Influenza Virus that Shakes Protection Despite High Vaccine Coverage. Vaccines 2019, 7, 174. https://doi.org/10.3390/vaccines7040174
Fougerolle S, Fortier C, Legrand L, Jourdan M, Marcillaud-Pitel C, Pronost S, Paillot R. Success and Limitation of Equine Influenza Vaccination: The First Incursion in a Decade of a Florida Clade 1 Equine Influenza Virus that Shakes Protection Despite High Vaccine Coverage. Vaccines. 2019; 7(4):174. https://doi.org/10.3390/vaccines7040174
Chicago/Turabian StyleFougerolle, Stéphanie, Christine Fortier, Loïc Legrand, Marion Jourdan, Christel Marcillaud-Pitel, Stéphane Pronost, and Romain Paillot. 2019. "Success and Limitation of Equine Influenza Vaccination: The First Incursion in a Decade of a Florida Clade 1 Equine Influenza Virus that Shakes Protection Despite High Vaccine Coverage" Vaccines 7, no. 4: 174. https://doi.org/10.3390/vaccines7040174
APA StyleFougerolle, S., Fortier, C., Legrand, L., Jourdan, M., Marcillaud-Pitel, C., Pronost, S., & Paillot, R. (2019). Success and Limitation of Equine Influenza Vaccination: The First Incursion in a Decade of a Florida Clade 1 Equine Influenza Virus that Shakes Protection Despite High Vaccine Coverage. Vaccines, 7(4), 174. https://doi.org/10.3390/vaccines7040174







