Reduced Renal Colonization and Enhanced Protection by Leptospiral Factor H Binding Proteins as a Multisubunit Vaccine against Leptospirosis in Hamsters
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Bacterial Strains and Culture Conditions
2.3. LMQ Adjuvant Preparation
2.4. Production of Recombinant FHBPs
2.5. Western Blotting
2.6. Mouse Immunization
2.7. Hamster Immunization and Challenge with Leptospira
2.8. FHBP-Specific Antibodies Detection
2.9. Human FH Binding Assay
2.10. Histopathology
2.11. Leptospira Detection in Hamsters
2.12. Statistical Analysis
3. Results
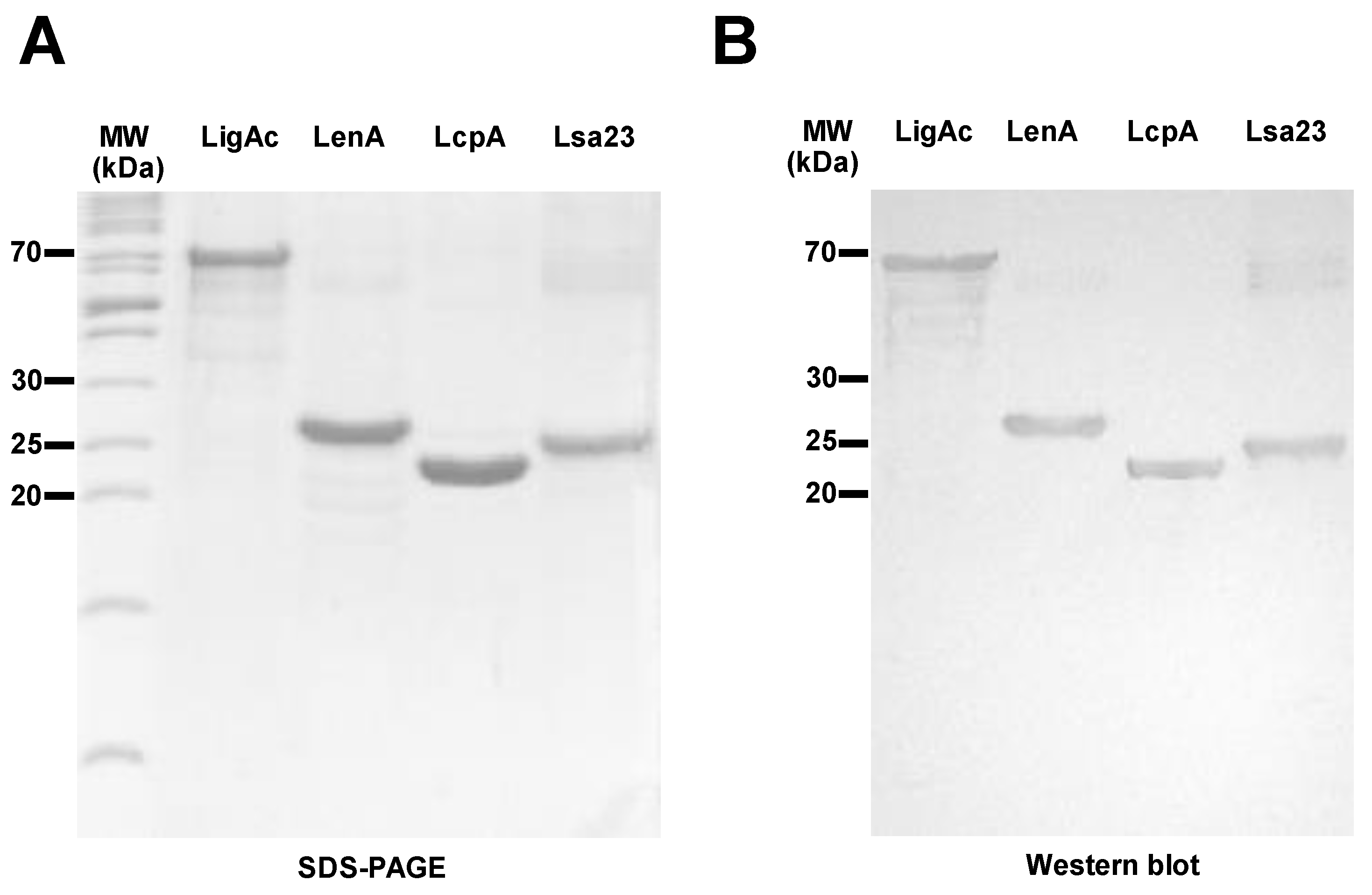
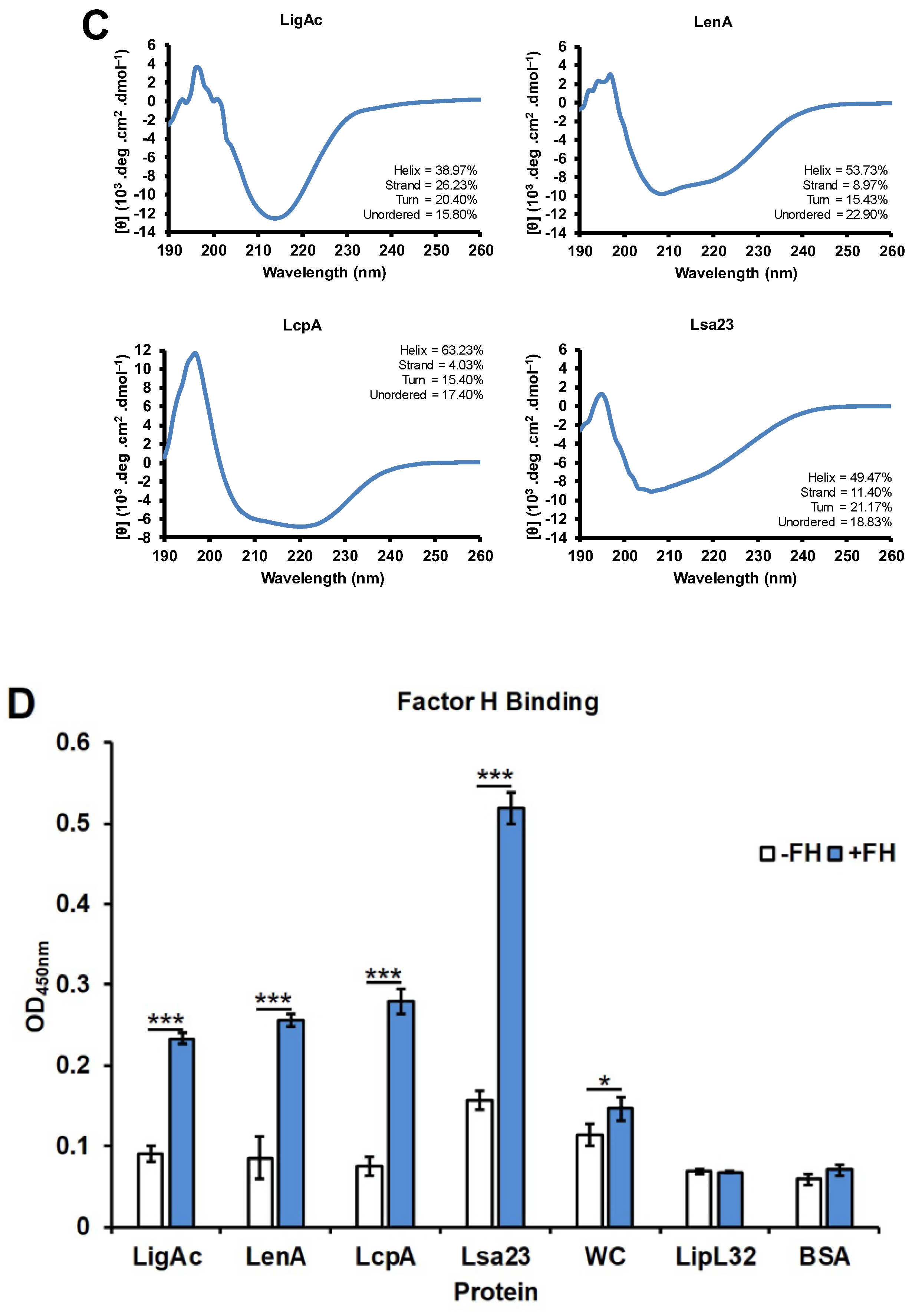
3.1. Preparation and Characterization of Recombinant FHBPs
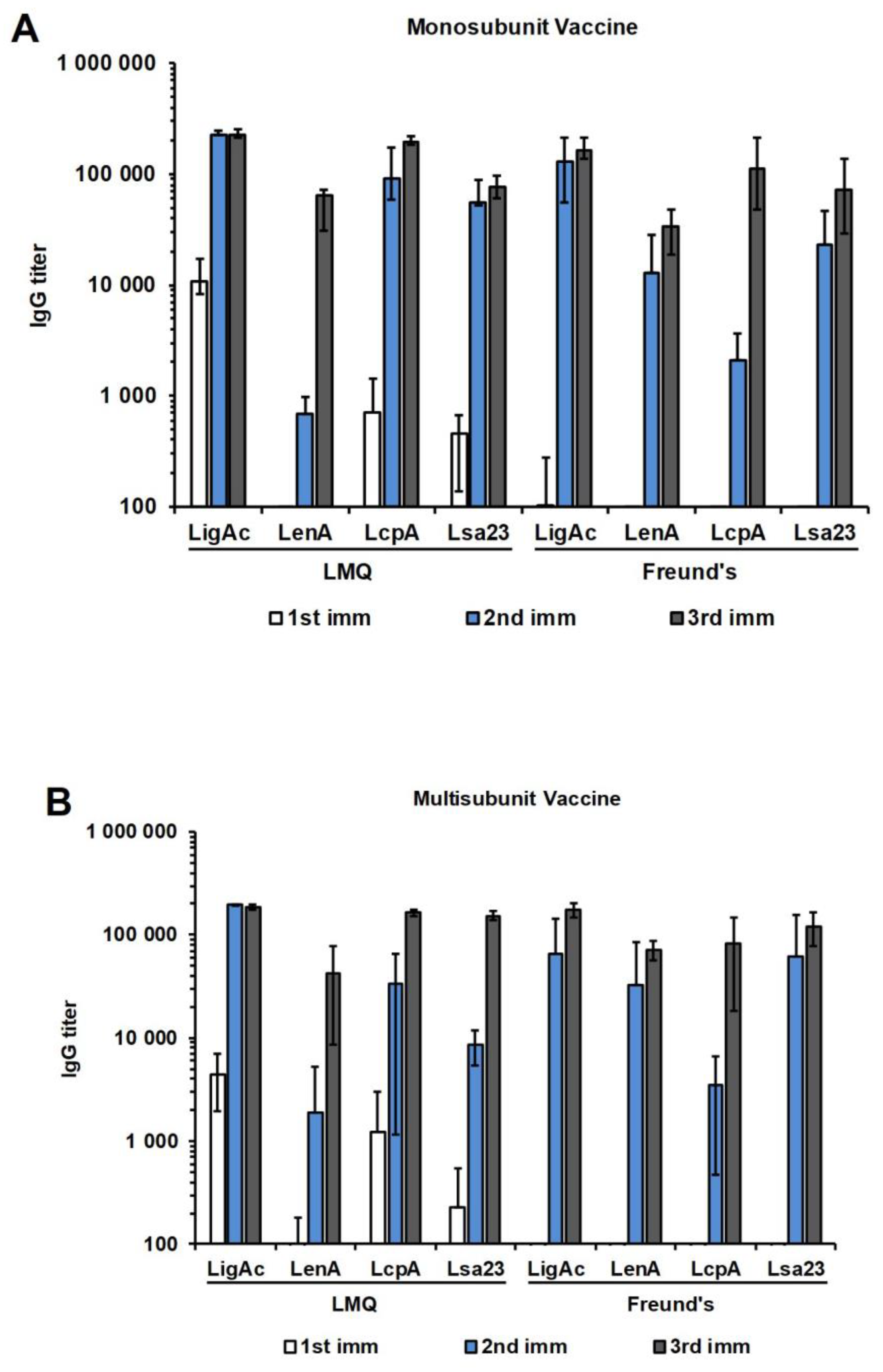
3.2. Immunogenicity of Mono or Multisubunit FHBP Vaccines in Mice
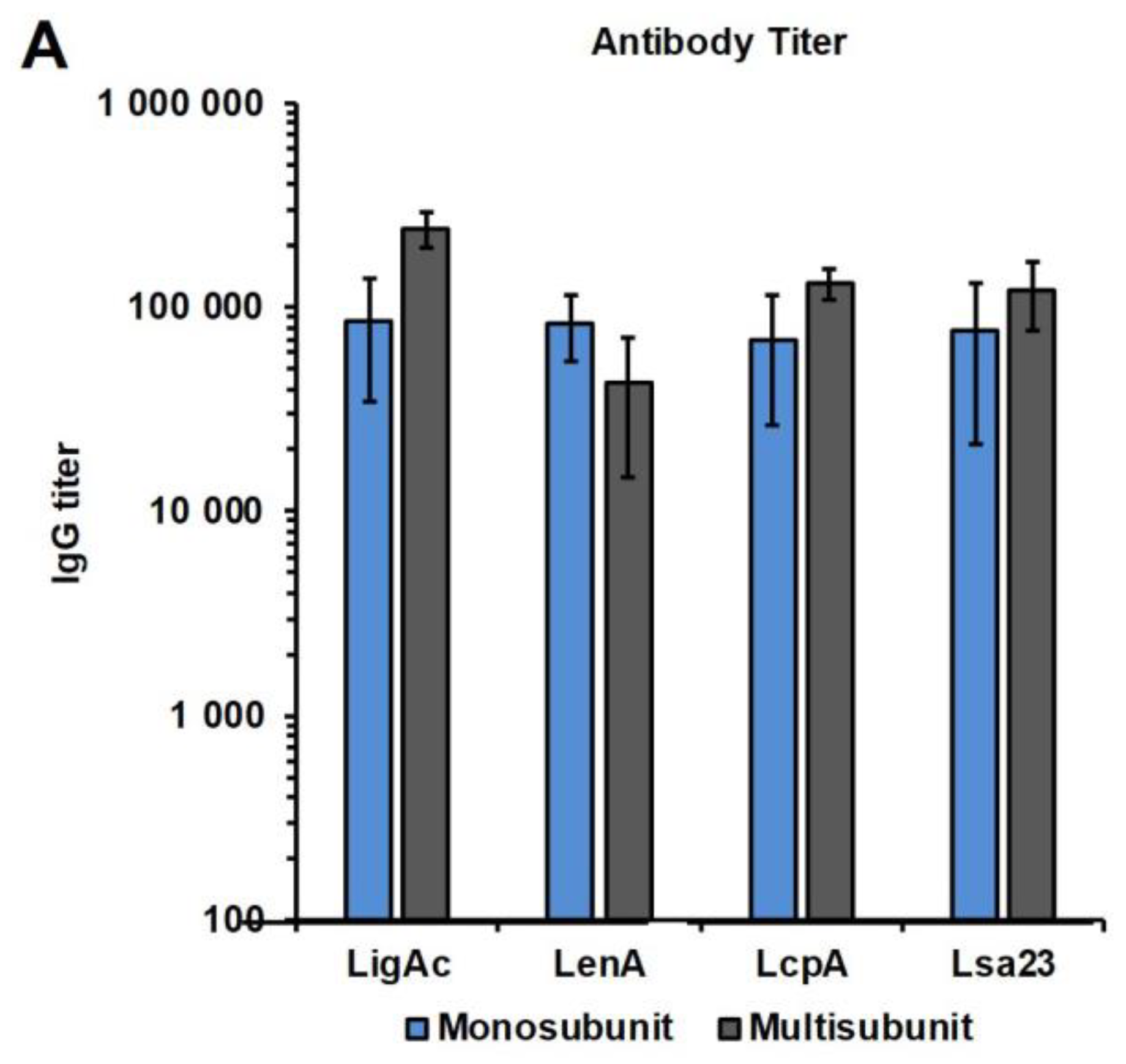
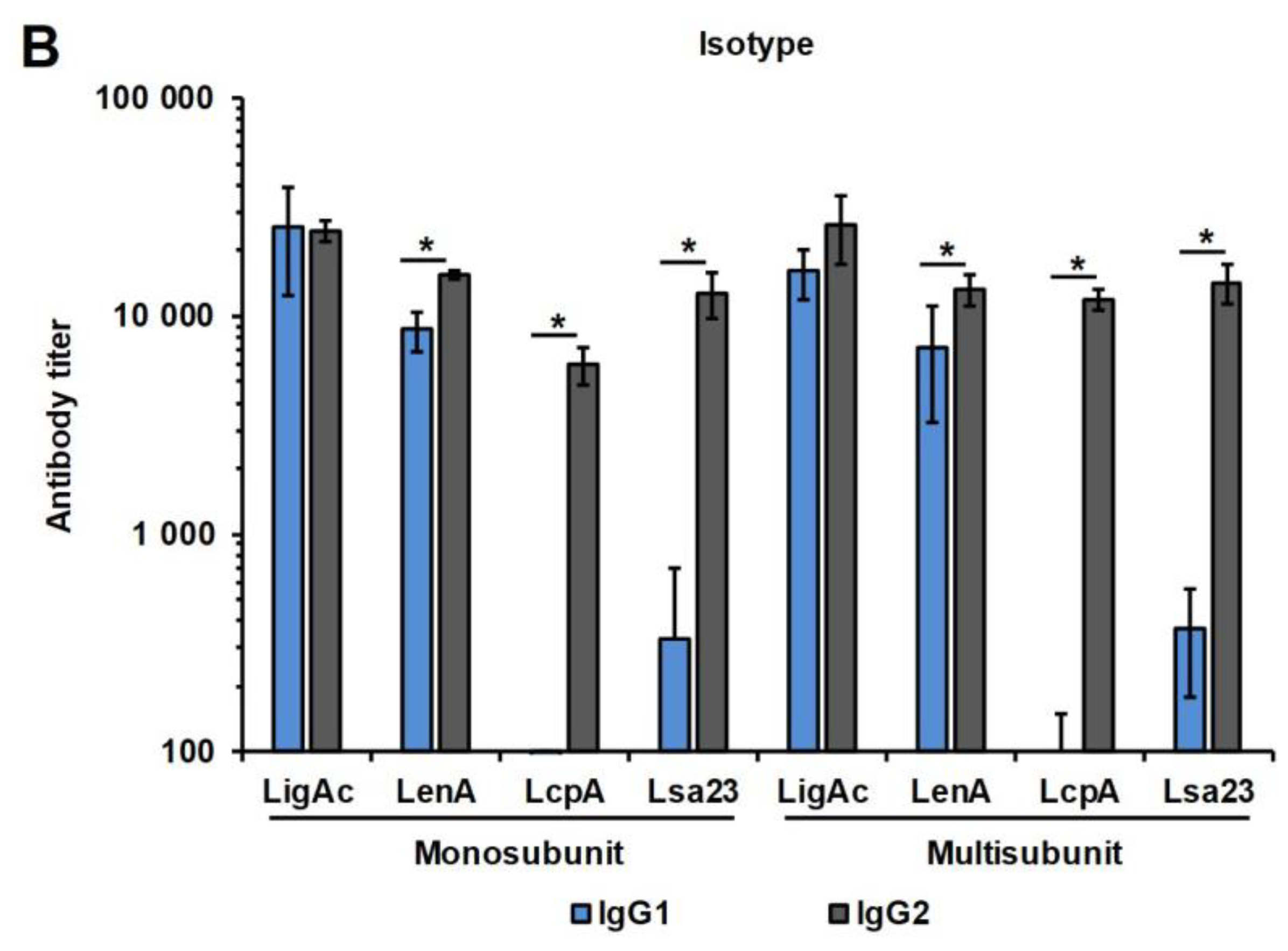
3.3. Immunogenicity of Mono or Multisubunit FHBPs in Hamsters
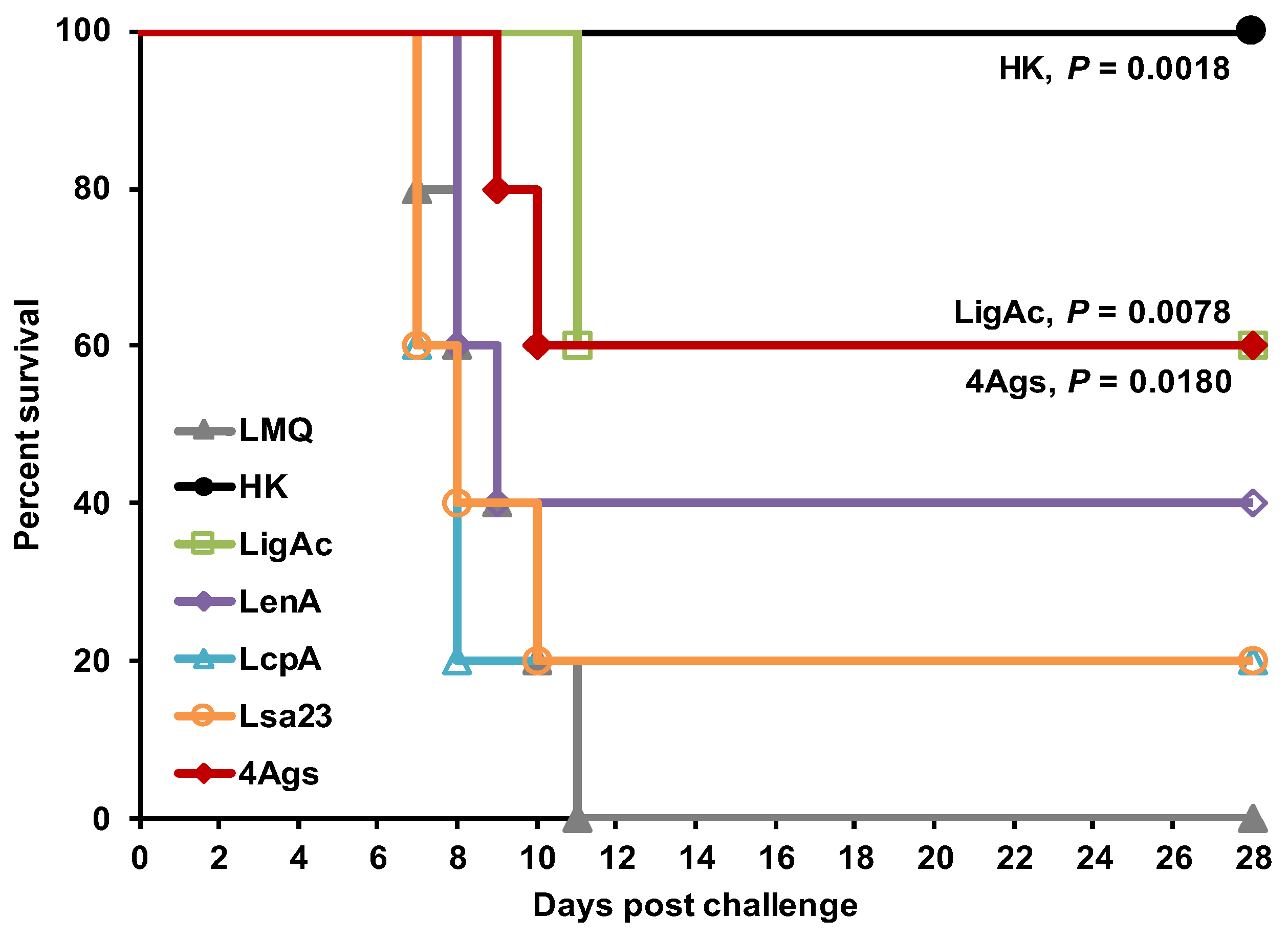
3.4. Protective Efficacy of Multisubunit FHBP Vaccine in Hamsters
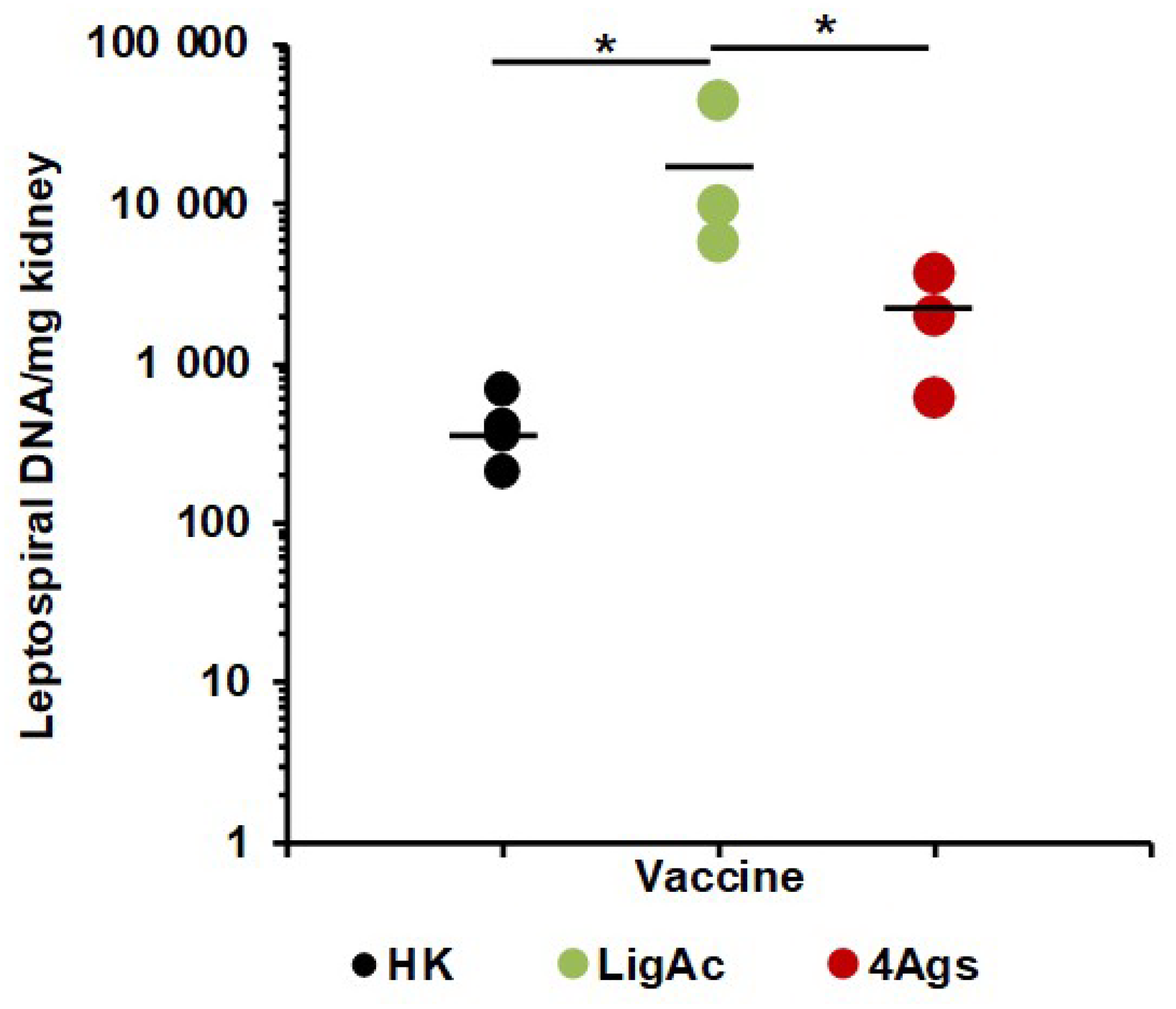
3.5. Effect of Multisubunit FHBP Vaccine on Target Organ Invasion by Leptospira
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Adler, B.; de la Pena Moctezuma, A. Leptospira and leptospirosis. Vet. Microbiol. 2010, 140, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Adler, B. Vaccines Against Leptospirosis. In Leptospira and Leptospirosis; Adler, B., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 251–272. [Google Scholar]
- Haake, D.A.; Mazel, M.K.; McCoy, A.M.; Milward, F.; Chao, G.; Matsunaga, J.; Wagar, E.A. Leptospiral outer membrane proteins OmpL1 and LipL41 exhibit synergistic immunoprotection. Infect. Immun. 1999, 67, 6572–6582. [Google Scholar] [PubMed]
- Palaniappan, R.U.; McDonough, S.P.; Divers, T.J.; Chen, C.S.; Pan, M.J.; Matsumoto, M.; Chang, Y.F. Immunoprotection of recombinant leptospiral immunoglobulin-like protein A against Leptospira interrogans serovar Pomona infection. Infect. Immun. 2006, 74, 1745–1750. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Faisal, S.M.; McDonough, S.P.; Chang, C.F.; Pan, M.J.; Akey, B.; Chang, Y.F. Identification and characterization of OmpA-like proteins as novel vaccine candidates for Leptospirosis. Vaccine 2010, 28, 2277–2283. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, D.D.; Forster, K.M.; Oliveira, T.L.; Amaral, M.; McBride, A.J.; Dellagostin, O.A. A prime-boost strategy using the novel vaccine candidate, LemA, protects hamsters against leptospirosis. Clin. Vaccine Immunol. 2013, 20, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Umthong, S.; Buaklin, A.; Jacquet, A.; Sangjun, N.; Kerdkaew, R.; Patarakul, K.; Palaga, T. Immunogenicity of a DNA and Recombinant Protein Vaccine Combining LipL32 and Loa22 for Leptospirosis Using Chitosan as a Delivery System. J. Microbiol. Biotechnol. 2015, 25, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.L.; Grassmann, A.A.; Schuch, R.A.; Seixas Neto, A.C.; Mendonca, M.; Hartwig, D.D.; McBride, A.J.; Dellagostin, O.A. Evaluation of the Leptospira interrogans Outer Membrane Protein OmpL37 as a Vaccine Candidate. PLoS ONE 2015, 10, e0142821. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, M.L.; Choy, H.A.; Kelley, M.M.; Matsunaga, J.; Babbitt, J.T.; Lewis, M.S.; Aleixo, J.A.; Haake, D.A. A LigA three-domain region protects hamsters from lethal infection by Leptospira interrogans. PLoS Negl. Trop. Dis. 2011, 5, e1422. [Google Scholar] [CrossRef]
- Silva, E.F.; Medeiros, M.A.; McBride, A.J.; Matsunaga, J.; Esteves, G.S.; Ramos, J.G.; Santos, C.S.; Croda, J.; Homma, A.; Dellagostin, O.A.; et al. The terminal portion of leptospiral immunoglobulin-like protein LigA confers protective immunity against lethal infection in the hamster model of leptospirosis. Vaccine 2007, 25, 6277–6286. [Google Scholar] [CrossRef]
- Monaris, D.; Sbrogio-Almeida, M.E.; Dib, C.C.; Canhamero, T.A.; Souza, G.O.; Vasconcellos, S.A.; Ferreira, L.C.; Abreu, P.A. Protective Immunity and Reduced Renal Colonization Induced by Vaccines Containing Recombinant Leptospira interrogans Outer Membrane Proteins and Flagellin Adjuvant. Clin. Vaccine Immunol. 2015, 22, 965–973. [Google Scholar] [CrossRef]
- Johnson, R.C.; Muschel, L.H. Antileptospiral Activity of Normal Serum. J. Bacteriol. 1965, 89, 1625–1626. [Google Scholar] [PubMed]
- Meri, T.; Murgia, R.; Stefanel, P.; Meri, S.; Cinco, M. Regulation of complement activation at the C3-level by serum resistant leptospires. Microb. Pathog. 2005, 39, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.S.; Abreu, P.A.; Vasconcellos, S.A.; Morais, Z.M.; Goncales, A.P.; Silva, A.S.; Daha, M.R.; Isaac, L. Immune evasion of Leptospira species by acquisition of human complement regulator C4BP. Infect. Immun. 2009, 77, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.B.; Miragaia Ldos, S.; Breda, L.C.; Abe, C.M.; Schmidt, M.C.; Moro, A.M.; Monaris, D.; Conde, J.N.; Jozsi, M.; Isaac, L.; et al. Pathogenic Leptospira species acquire factor H and vitronectin via the surface protein LcpA. Infect. Immun. 2015, 83, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Granoff, D.M. Commentary: European Medicines Agency recommends approval of a broadly protective vaccine against serogroup B meningococcal disease. Pediatr. Infect. Dis. J. 2013, 32, 372–373. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.L.; Absalon, J.; Beeslaar, J.; Balmer, P.; Jansen, K.U.; Jones, T.R.; Harris, S.; York, L.J.; Jiang, Q.; Radley, D.; et al. From research to licensure and beyond: Clinical development of MenB-FHbp, a broadly protective meningococcal B vaccine. Expert Rev. Vaccines 2018, 17, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Beernink, P.T.; Shaughnessy, J.; Braga, E.M.; Liu, Q.; Rice, P.A.; Ram, S.; Granoff, D.M. A meningococcal factor H binding protein mutant that eliminates factor H binding enhances protective antibody responses to vaccination. J. Immunol. 2011, 186, 3606–3614. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, G.H.; Atzingen, M.V.; de Souza, G.O.; Vasconcellos, S.A.; Nascimento, A.L. Leptospira interrogans Lsa23 protein recruits plasminogen, factor H and C4BP from normal human serum and mediates C3b and C4b degradation. Microbiology 2016, 162, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Choy, H.A. Multiple activities of LigB potentiate virulence of Leptospira interrogans: Inhibition of alternative and classical pathways of complement. PLoS ONE 2012, 7, e41566. [Google Scholar] [CrossRef]
- Siqueira, G.H.; Atzingen, M.V.; Alves, I.J.; de Morais, Z.M.; Vasconcellos, S.A.; Nascimento, A.L. Characterization of three novel adhesins of Leptospira interrogans. Am. J. Trop. Med. Hyg. 2013, 89, 1103–1116. [Google Scholar] [CrossRef]
- Barbosa, A.S.; Monaris, D.; Silva, L.B.; Morais, Z.M.; Vasconcellos, S.A.; Cianciarullo, A.M.; Isaac, L.; Abreu, P.A. Functional characterization of LcpA, a surface-exposed protein of Leptospira spp. that binds the human complement regulator C4BP. Infect. Immun. 2010, 78, 3207–3216. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, J.; Barocchi, M.A.; Croda, J.; Young, T.A.; Sanchez, Y.; Siqueira, I.; Bolin, C.A.; Reis, M.G.; Riley, L.W.; Haake, D.A.; et al. Pathogenic Leptospira species express surface-exposed proteins belonging to the bacterial immunoglobulin superfamily. Mol. Microbiol. 2003, 49, 929–945. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Hellwage, J.; Artiushin, S.; Zipfel, P.F.; Kraiczy, P.; Timoney, J.F.; Stevenson, B. LfhA, a novel factor H-binding protein of Leptospira interrogans. Infect. Immun. 2006, 74, 2659–2666. [Google Scholar] [CrossRef] [PubMed]
- Castiblanco-Valencia, M.M.; Fraga, T.R.; Silva, L.B.; Monaris, D.; Abreu, P.A.; Strobel, S.; Jozsi, M.; Isaac, L.; Barbosa, A.S. Leptospiral immunoglobulin-like proteins interact with human complement regulators factor H, FHL-1, FHR-1, and C4BP. J. Infect. Dis. 2012, 205, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Srikram, A.; Zhang, K.; Bartpho, T.; Lo, M.; Hoke, D.E.; Sermswan, R.W.; Adler, B.; Murray, G.L. Cross-protective immunity against leptospirosis elicited by a live, attenuated lipopolysaccharide mutant. J. Infect. Dis. 2011, 203, 870–879. [Google Scholar] [CrossRef]
- Zuerner, R.L. Laboratory maintenance of pathogenic Leptospira. Curr. Protoc. Microbiol. 2005, 1. [Google Scholar] [CrossRef]
- Li, Y.; Leneghan, D.B.; Miura, K.; Nikolaeva, D.; Brian, I.J.; Dicks, M.D.; Fyfe, A.J.; Zakutansky, S.E.; de Cassan, S.; Long, C.A.; et al. Enhancing immunogenicity and transmission-blocking activity of malaria vaccines by fusing Pfs25 to IMX313 multimerization technology. Sci. Rep. 2016, 6, 18848. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, B.; Choy, H.A.; Pinne, M.; Rotondi, M.L.; Miller, M.C.; Demoll, E.; Kraiczy, P.; Cooley, A.E.; Creamer, T.P.; Suchard, M.A.; et al. Leptospira interrogans endostatin-like outer membrane proteins bind host fibronectin, laminin and regulators of complement. PLoS ONE 2007, 2, e1188. [Google Scholar] [CrossRef]
- Tanyaratsrisakul, S.; Malainual, N.; Jirapongsananuruk, O.; Smith, W.A.; Thomas, W.R.; Piboonpocanun, S. Structural and IgE binding analyses of recombinant Der p 2 expressed from the hosts Escherichia coli and Pichia pastoris. Int. Arch. Allergy Immunol. 2010, 151, 190–198. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Atzingen, M.V.; Goncales, A.P.; de Morais, Z.M.; Araujo, E.R.; De Brito, T.; Vasconcellos, S.A.; Nascimento, A.L. Characterization of leptospiral proteins that afford partial protection in hamsters against lethal challenge with Leptospira interrogans. J. Med. Microbiol. 2010, 59, 1005–1015. [Google Scholar] [CrossRef]
- National Center for the Replacement Refinement and Reduction of Animals in Research. Cardiac Puncture with Recovery. Available online: https://www.nc3rs.org.uk/hamster-cardiac-puncture-recovery-surgical (accessed on 24 May 2019).
- Conrad, N.L.; Cruz McBride, F.W.; Souza, J.D.; Silveira, M.M.; Felix, S.; Mendonca, K.S.; Santos, C.S.; Athanazio, D.A.; Medeiros, M.A.; Reis, M.G.; et al. LigB subunit vaccine confers sterile immunity against challenge in the hamster model of leptospirosis. PLoS Negl. Trop. Dis. 2017, 11, e0005441. [Google Scholar] [CrossRef]
- Buaklin, A.; Palaga, T.; Hannaman, D.; Kerdkaew, R.; Patarakul, K.; Jacquet, A. Optimization of the immunogenicity of a DNA vaccine encoding a bacterial outer membrane lipoprotein. Mol. Biotechnol. 2014, 56, 903–910. [Google Scholar] [CrossRef]
- Faisal, S.M.; Yan, W.; McDonough, S.P.; Chang, Y.F. Leptospira immunoglobulin-like protein A variable region (LigAvar) incorporated in liposomes and PLGA microspheres produces a robust immune response correlating to protective immunity. Vaccine 2009, 27, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Stoddard, R.A.; Gee, J.E.; Wilkins, P.P.; McCaustland, K.; Hoffmaster, A.R. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn. Microbiol. Infect. Dis. 2009, 64, 247–255. [Google Scholar] [CrossRef]
- Evangelista, K.V.; Lourdault, K.; Matsunaga, J.; Haake, D.A. Immunoprotective properties of recombinant LigA and LigB in a hamster model of acute leptospirosis. PLoS ONE 2017, 12, e0180004. [Google Scholar] [CrossRef] [PubMed]
- Lucas, D.S.; Cullen, P.A.; Lo, M.; Srikram, A.; Sermswan, R.W.; Adler, B. Recombinant LipL32 and LigA from Leptospira are unable to stimulate protective immunity against leptospirosis in the hamster model. Vaccine 2011, 29, 3413–3418. [Google Scholar] [CrossRef]
- Garcon, N.; Chomez, P.; Van Mechelen, M. GlaxoSmithKline Adjuvant Systems in vaccines: Concepts, achievements and perspectives. Expert Rev. Vaccines 2007, 6, 723–739. [Google Scholar] [CrossRef] [PubMed]
- Seder, R.A.; Hill, A.V. Vaccines against intracellular infections requiring cellular immunity. Nature 2000, 406, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Berzofsky, J.A.; Ahlers, J.D.; Janik, J.; Morris, J.; Oh, S.; Terabe, M.; Belyakov, I.M. Progress on new vaccine strategies against chronic viral infections. J. Clin. Investig. 2004, 114, 450–462. [Google Scholar] [CrossRef]
- Chlibek, R.; Bayas, J.M.; Collins, H.; de la Pinta, M.L.; Ledent, E.; Mols, J.F.; Heineman, T.C. Safety and immunogenicity of an AS01-adjuvanted varicella-zoster virus subunit candidate vaccine against herpes zoster in adults >=50 years of age. J. Infect. Dis. 2013, 208, 1953–1961. [Google Scholar] [CrossRef]
- Chlibek, R.; Smetana, J.; Pauksens, K.; Rombo, L.; Van den Hoek, J.A.; Richardus, J.H.; Plassmann, G.; Schwarz, T.F.; Ledent, E.; Heineman, T.C. Safety and immunogenicity of three different formulations of an adjuvanted varicella-zoster virus subunit candidate vaccine in older adults: A phase II, randomized, controlled study. Vaccine 2014, 32, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Leroux-Roels, I.; Devaster, J.M.; Leroux-Roels, G.; Verlant, V.; Henckaerts, I.; Moris, P.; Hermand, P.; Van Belle, P.; Poolman, J.T.; Vandepapeliere, P.; et al. Adjuvant system AS02V enhances humoral and cellular immune responses to pneumococcal protein PhtD vaccine in healthy young and older adults: Randomised, controlled trials. Vaccine 2015, 33, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Baldridge, J.R.; McGowan, P.; Evans, J.T.; Cluff, C.; Mossman, S.; Johnson, D.; Persing, D. Taking a Toll on human disease: Toll-like receptor 4 agonists as vaccine adjuvants and monotherapeutic agents. Expert Opin. Biol. Ther. 2004, 4, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Kashala, O.; Amador, R.; Valero, M.V.; Moreno, A.; Barbosa, A.; Nickel, B.; Daubenberger, C.A.; Guzman, F.; Pluschke, G.; Patarroyo, M.E. Safety, tolerability and immunogenicity of new formulations of the Plasmodium falciparum malaria peptide vaccine SPf66 combined with the immunological adjuvant QS-21. Vaccine 2002, 20, 2263–2277. [Google Scholar] [CrossRef]
- Lourdault, K.; Wang, L.C.; Vieira, A.; Matsunaga, J.; Melo, R.; Lewis, M.S.; Haake, D.A.; Gomes-Solecki, M. Oral immunization with Escherichia coli expressing a lipidated form of LigA protects hamsters against challenge with Leptospira interrogans serovar Copenhageni. Infect. Immun. 2014, 82, 893–902. [Google Scholar] [CrossRef]
- Oliveira, T.L.; Rizzi, C.; da Cunha, C.E.P.; Dorneles, J.; Seixas Neto, A.C.P.; Amaral, M.G.; Hartwig, D.D.; Dellagostin, O.A. Recombinant BCG strains expressing chimeric proteins derived from Leptospira protect hamsters against leptospirosis. Vaccine 2019, 37, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Bacelo, K.L.; Hartwig, D.D.; Seixas, F.K.; Schuch, R.; Moreira Ada, S.; Amaral, M.; Collares, T.; Vendrusculo, C.T.; McBride, A.J.; Dellagostin, O.A. Xanthan gum as an adjuvant in a subunit vaccine preparation against leptospirosis. BioMed Res. Int. 2014, 2014, 636491. [Google Scholar] [CrossRef] [PubMed]
- Coe, J.E.; Schell, R.F.; Ross, M.J. Immune response in the hamster: Definition of a novel IgG not expressed in all hamster strains. Immunology 1995, 86, 141–148. [Google Scholar] [PubMed]
- Fernandes, L.G.; Teixeira, A.F.; Filho, A.F.; Souza, G.O.; Vasconcellos, S.A.; Heinemann, M.B.; Romero, E.C.; Nascimento, A.L. Immune response and protective profile elicited by a multi-epitope chimeric protein derived from Leptospira interrogans. Int. J. Infect. Dis. 2017, 57, 61–69. [Google Scholar] [CrossRef]
- Da Cunha, C.E.P.; Bettin, E.B.; Bakry, A.; Seixas Neto, A.C.P.; Amaral, M.G.; Dellagostin, O.A. Evaluation of different strategies to promote a protective immune response against leptospirosis using a recombinant LigA and LigB chimera. Vaccine 2019, 37, 1844–1852. [Google Scholar] [CrossRef]
- Wunder, E.A., Jr.; Figueira, C.P.; Santos, G.R.; Lourdault, K.; Matthias, M.A.; Vinetz, J.M.; Ramos, E.; Haake, D.A.; Picardeau, M.; Dos Reis, M.G.; et al. Real-Time PCR Reveals Rapid Dissemination of Leptospira interrogans after Intraperitoneal and Conjunctival Inoculation of Hamsters. Infect. Immun. 2016, 84, 2105–2115. [Google Scholar] [CrossRef] [PubMed]
- Breda, L.C.; Hsieh, C.L.; Castiblanco Valencia, M.M.; da Silva, L.B.; Barbosa, A.S.; Blom, A.M.; Chang, Y.F.; Isaac, L. Fine Mapping of the Interaction between C4b-Binding Protein and Outer Membrane Proteins LigA and LigB of Pathogenic Leptospira interrogans. PLoS Negl. Trop. Dis. 2015, 9, e0004192. [Google Scholar]
- Choy, H.A.; Kelley, M.M.; Chen, T.L.; Moller, A.K.; Matsunaga, J.; Haake, D.A. Physiological osmotic induction of Leptospira interrogans adhesion: LigA and LigB bind extracellular matrix proteins and fibrinogen. Infect. Immun. 2007, 75, 2441–2450. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Brissette, C.A.; Bowman, A.A.; Shah, S.T.; Zipfel, P.F.; Stevenson, B. Leptospiral endostatin-like protein A is a bacterial cell surface receptor for human plasminogen. Infect. Immun. 2010, 78, 2053–2059. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Castiblanco-Valencia, M.M.; Fraga, T.R.; Pagotto, A.H.; Serrano, S.M.; Abreu, P.A.; Barbosa, A.S.; Isaac, L. Plasmin cleaves fibrinogen and the human complement proteins C3b and C5 in the presence of Leptospira interrogans proteins: A new role of LigA and LigB in invasion and complement immune evasion. Immunobiology 2016, 221, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.F.; Chen, C.S.; Palaniappan, R.U.; He, H.; McDonough, S.P.; Barr, S.C.; Yan, W.; Faisal, S.M.; Pan, M.J.; Chang, C.F. Immunogenicity of the recombinant leptospiral putative outer membrane proteins as vaccine candidates. Vaccine 2007, 25, 8190–8197. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.L.; Schuch, R.A.; Inda, G.R.; Roloff, B.C.; Neto, A.; Amaral, M.; Dellagostin, O.A.; Hartwig, D.D. LemA and Erp Y-like recombinant proteins from Leptospira interrogans protect hamsters from challenge using AddaVax as adjuvant. Vaccine 2018, 36, 2574–2580. [Google Scholar] [CrossRef]
- Zuerner, R.L.; Alt, D.P.; Palmer, M.V. Development of chronic and acute golden Syrian hamster infection models with Leptospira borgpetersenii serovar Hardjo. Vet. Pathol. 2012, 49, 403–411. [Google Scholar] [CrossRef]
| Vaccine Formulation a | % Protection b | Endpoint Day | Kidney Positive Detection c | ||
|---|---|---|---|---|---|
| Antigen | Adjuvant | Culture | qPCR | ||
| PBS | LMQ | 0 | 7, 8, 9, 10, 11 | 0/5 | 5/5 |
| Heat-killed Leptospira | Freund’s | 100 ** | 28, 28, 28, 28, 28 | 0/5 | 5/5 |
| LigAc | LMQ | 60 ** | 11, 11, 28, 28, 28 | 0/5 | 5/5 |
| LenA | LMQ | 40 | 8, 8, 9, 28, 28 | 0/5 | 5/5 |
| LcpA | LMQ | 20 | 7, 7, 8, 8, 28 | 0/5 | 5/5 |
| Lsa23 | LMQ | 20 | 7, 7, 8, 10, 28 | 0/5 | 5/5 |
| LigAc + LenA + LcpA + Lsa23 | LMQ | 60 * | 9, 10, 28, 28, 28 | 0/5 | 5/5 |
| Vaccine Formulation a | Mean Pathological Score b | |||
|---|---|---|---|---|
| Antigen | Adjuvant | Lung | Liver | Kidney |
| PBS | LMQ | 0.8 ± 1.1 | 1.0 ± 1.4 | 0.8 ± 0.5 |
| Heat-killed Leptospira | Freund’s | 0.6 ± 0.6 | 1.0 ± 0.0 | 0.0 ± 0.0 * |
| LigAc | LMQ | 0.6 ± 0.9 | 0.4 ± 0.6 | 1.2 ± 0.8 |
| LenA | LMQ | 1.4 ± 1.3 | 1.0 ± 0.7 | 0.4 ± 0.9 |
| LcpA | LMQ | 2.0 ± 1.2 | 1.2 ± 0.4 | 0.4 ± 0.6 |
| Lsa23 | LMQ | 1.2 ± 0.8 | 0.8 ± 0.5 | 1.0 ± 0.7 |
| LigAc, LenA, LcpA, Lsa23 | LMQ | 1.2 ± 0.6 | 1.2 ± 0.6 | 1.0 ± 1.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Techawiwattanaboon, T.; Barnier-Quer, C.; Palaga, T.; Jacquet, A.; Collin, N.; Sangjun, N.; Komanee, P.; Piboonpocanun, S.; Patarakul, K. Reduced Renal Colonization and Enhanced Protection by Leptospiral Factor H Binding Proteins as a Multisubunit Vaccine against Leptospirosis in Hamsters. Vaccines 2019, 7, 95. https://doi.org/10.3390/vaccines7030095
Techawiwattanaboon T, Barnier-Quer C, Palaga T, Jacquet A, Collin N, Sangjun N, Komanee P, Piboonpocanun S, Patarakul K. Reduced Renal Colonization and Enhanced Protection by Leptospiral Factor H Binding Proteins as a Multisubunit Vaccine against Leptospirosis in Hamsters. Vaccines. 2019; 7(3):95. https://doi.org/10.3390/vaccines7030095
Chicago/Turabian StyleTechawiwattanaboon, Teerasit, Christophe Barnier-Quer, Tanapat Palaga, Alain Jacquet, Nicolas Collin, Noppadon Sangjun, Pat Komanee, Surapon Piboonpocanun, and Kanitha Patarakul. 2019. "Reduced Renal Colonization and Enhanced Protection by Leptospiral Factor H Binding Proteins as a Multisubunit Vaccine against Leptospirosis in Hamsters" Vaccines 7, no. 3: 95. https://doi.org/10.3390/vaccines7030095
APA StyleTechawiwattanaboon, T., Barnier-Quer, C., Palaga, T., Jacquet, A., Collin, N., Sangjun, N., Komanee, P., Piboonpocanun, S., & Patarakul, K. (2019). Reduced Renal Colonization and Enhanced Protection by Leptospiral Factor H Binding Proteins as a Multisubunit Vaccine against Leptospirosis in Hamsters. Vaccines, 7(3), 95. https://doi.org/10.3390/vaccines7030095





