Cathelicidins: Immunomodulatory Antimicrobials
Abstract
1. Introduction
2. Structural Aspects
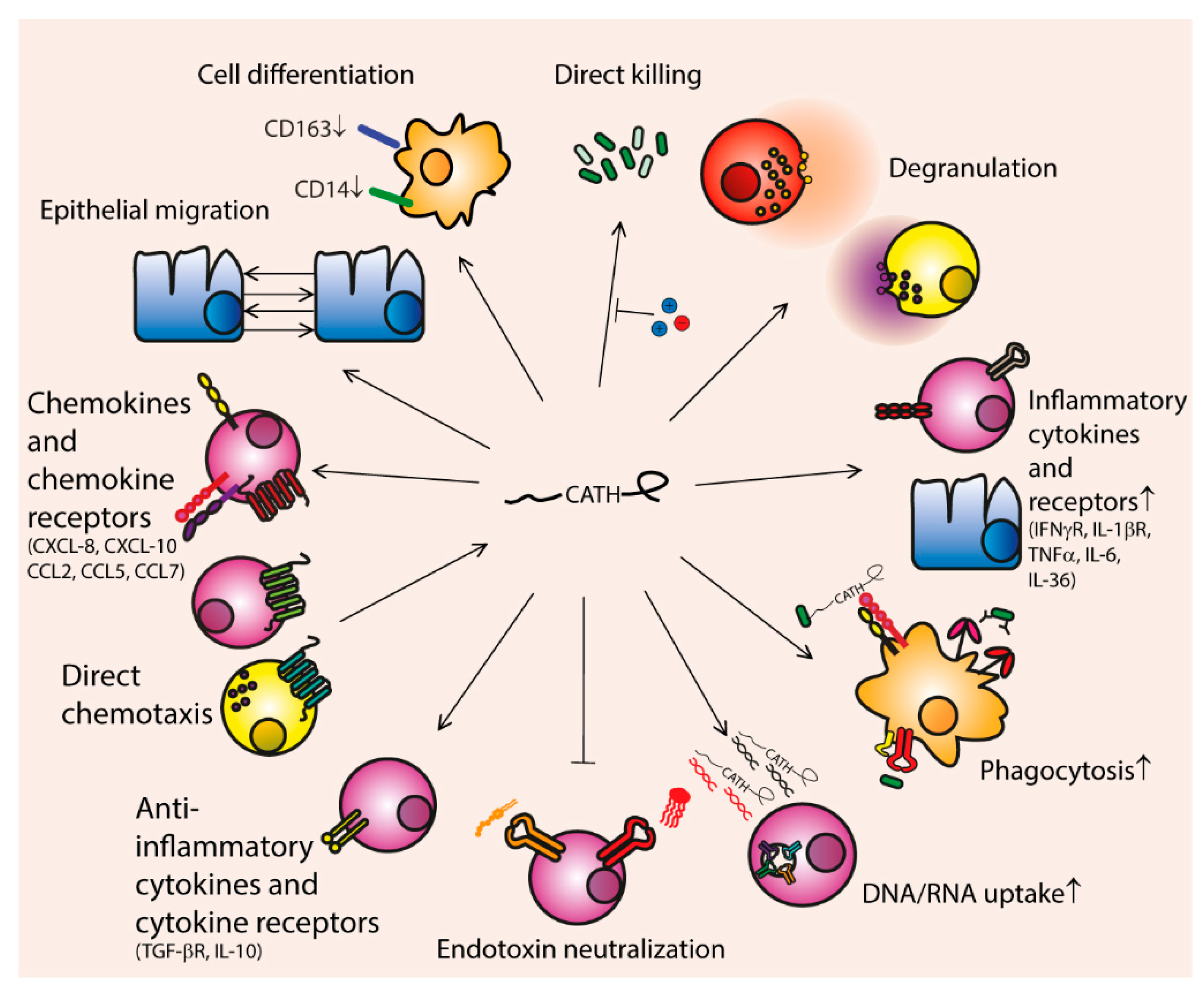
3. Biological Functions of Cathelicidins
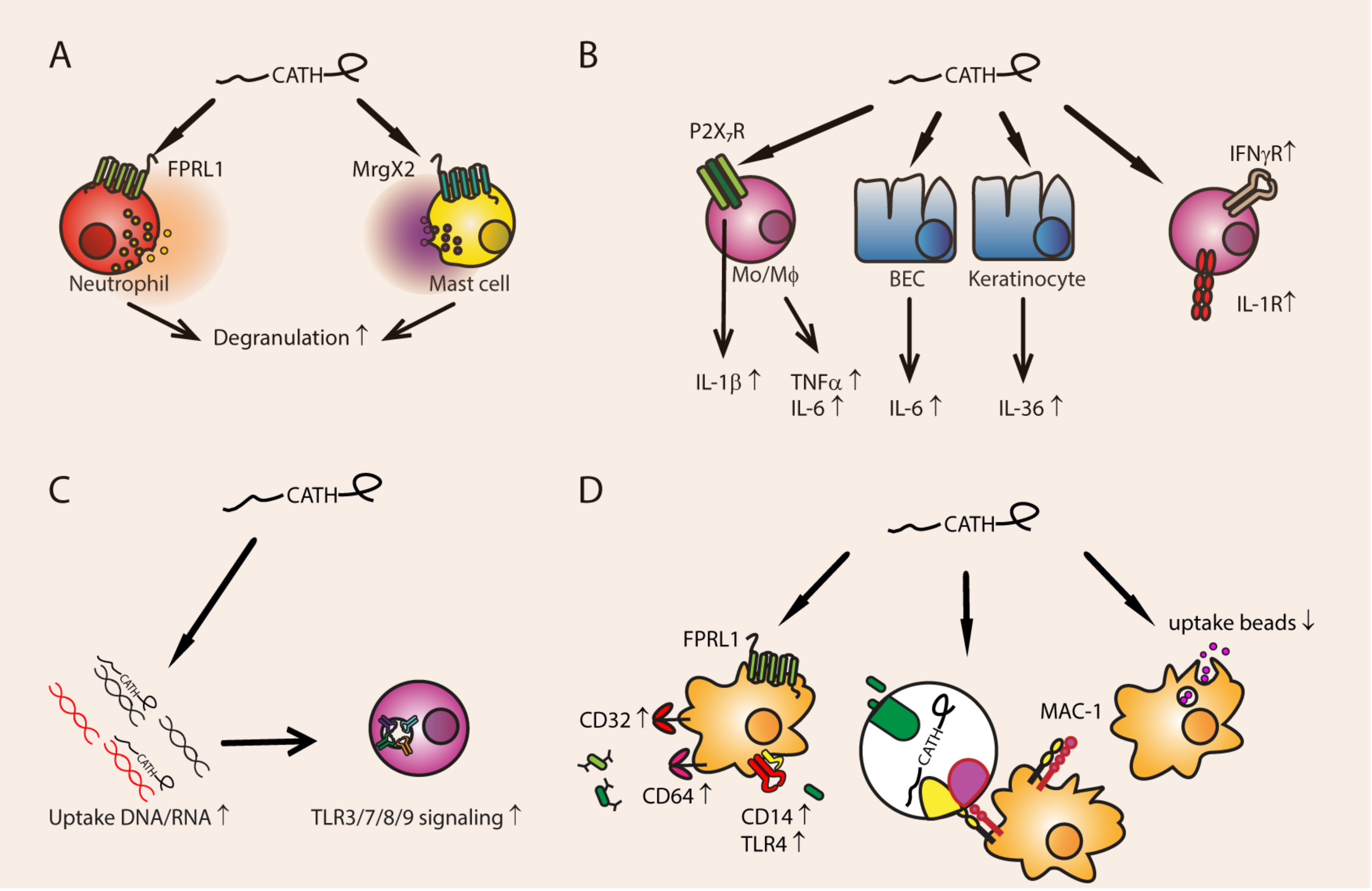
3.1. Degranulation
3.2. Pro-Inflammatory Immune Modulation
3.2.1. Cytokine and Cytokine Receptor Expression
3.2.2. Phagocytosis
3.2.3. DNA- and RNA-Mediated TLR Activation
3.3. Anti-Inflammatory Immune Modulation
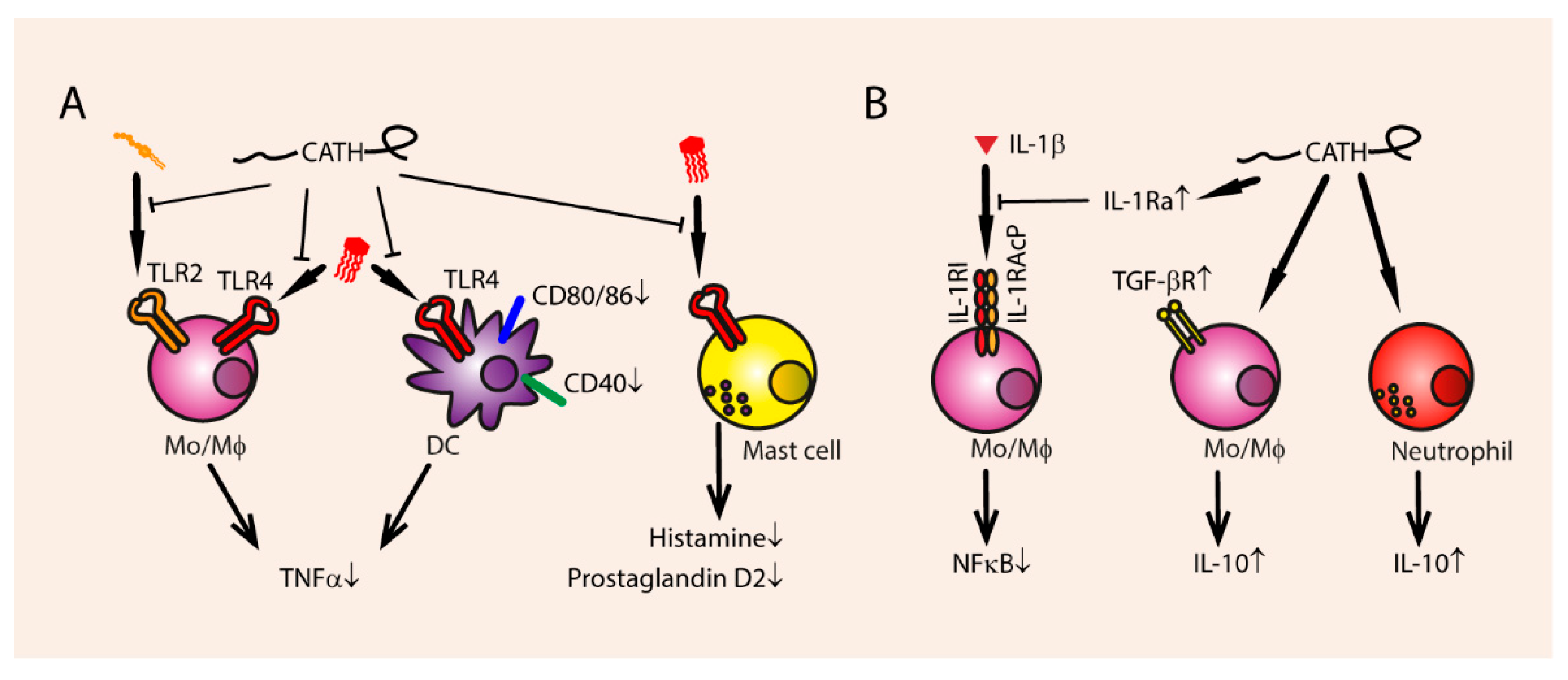
3.3.1. Inhibition of Endotoxin-Mediated TLR Activation
3.3.2. Secretion of Anti-Inflammatory Cytokines
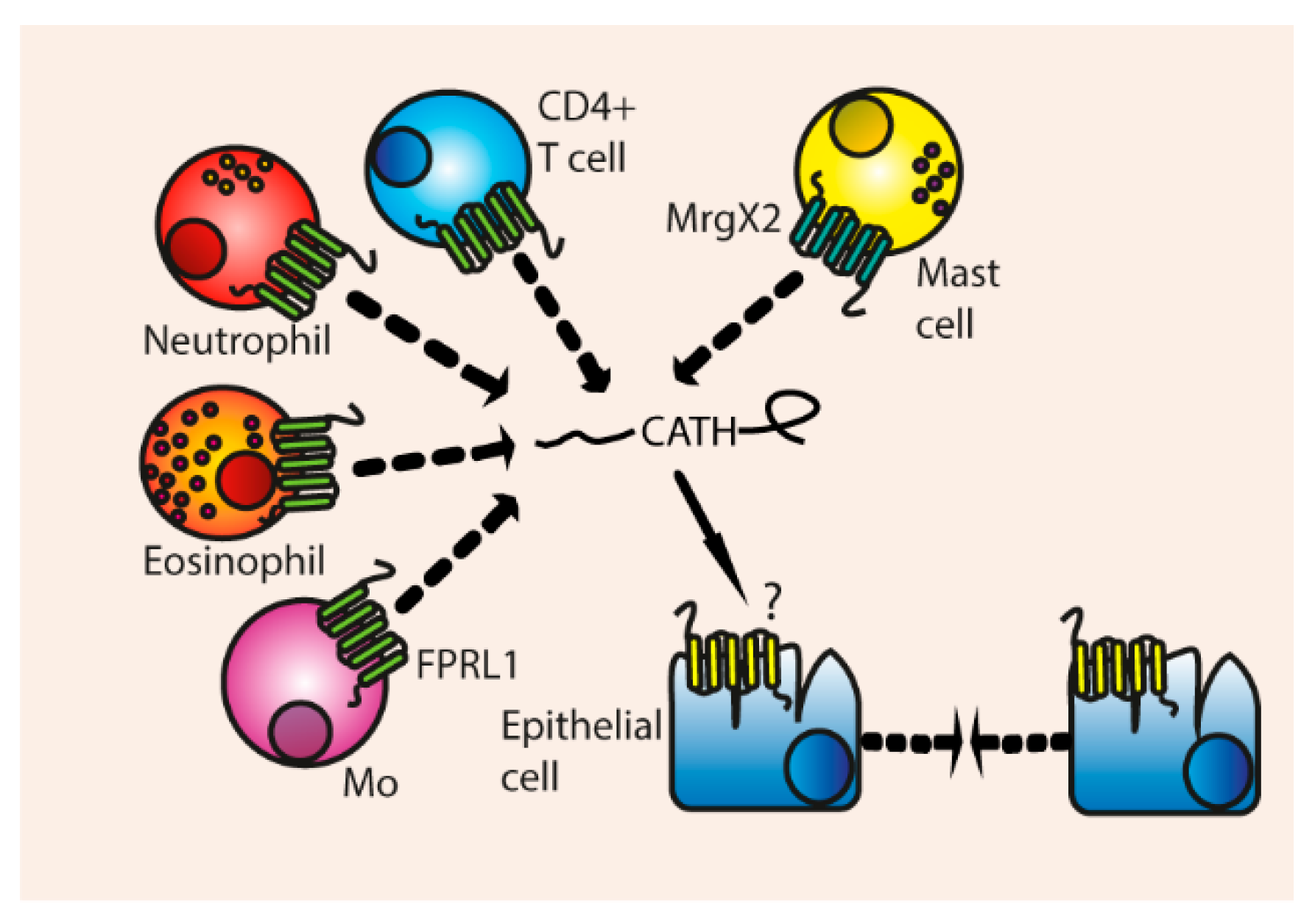
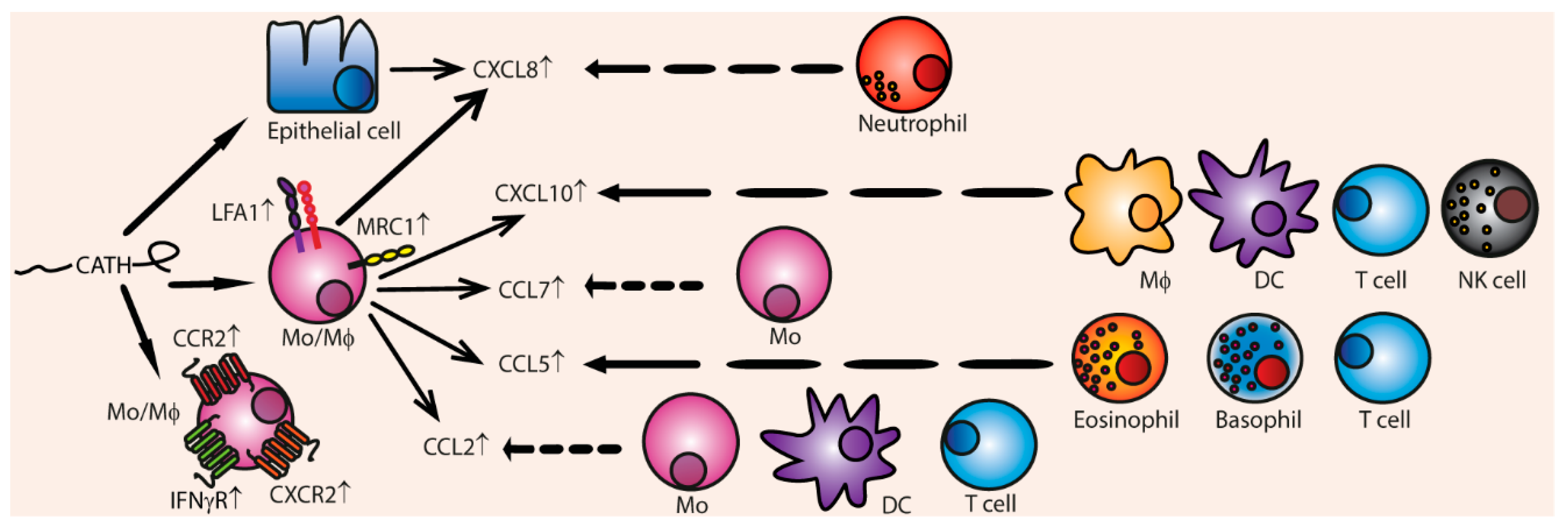
3.4. Chemotaxis
Indirect Chemotaxis
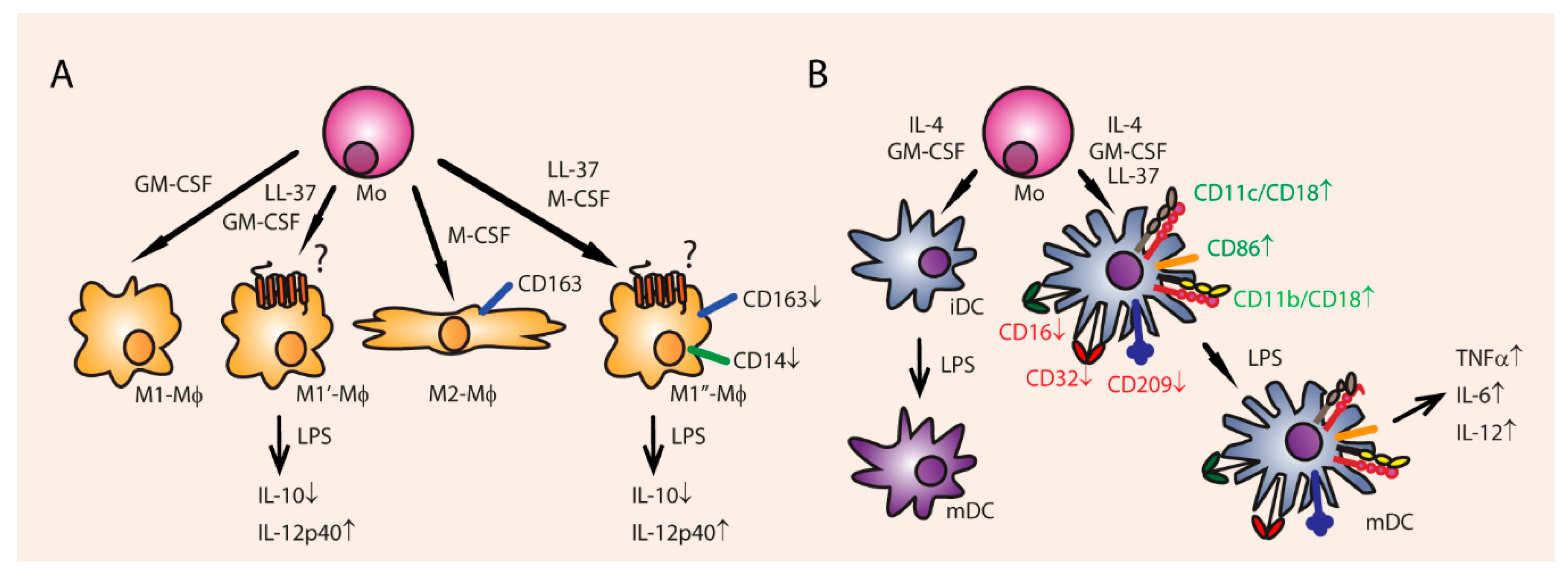
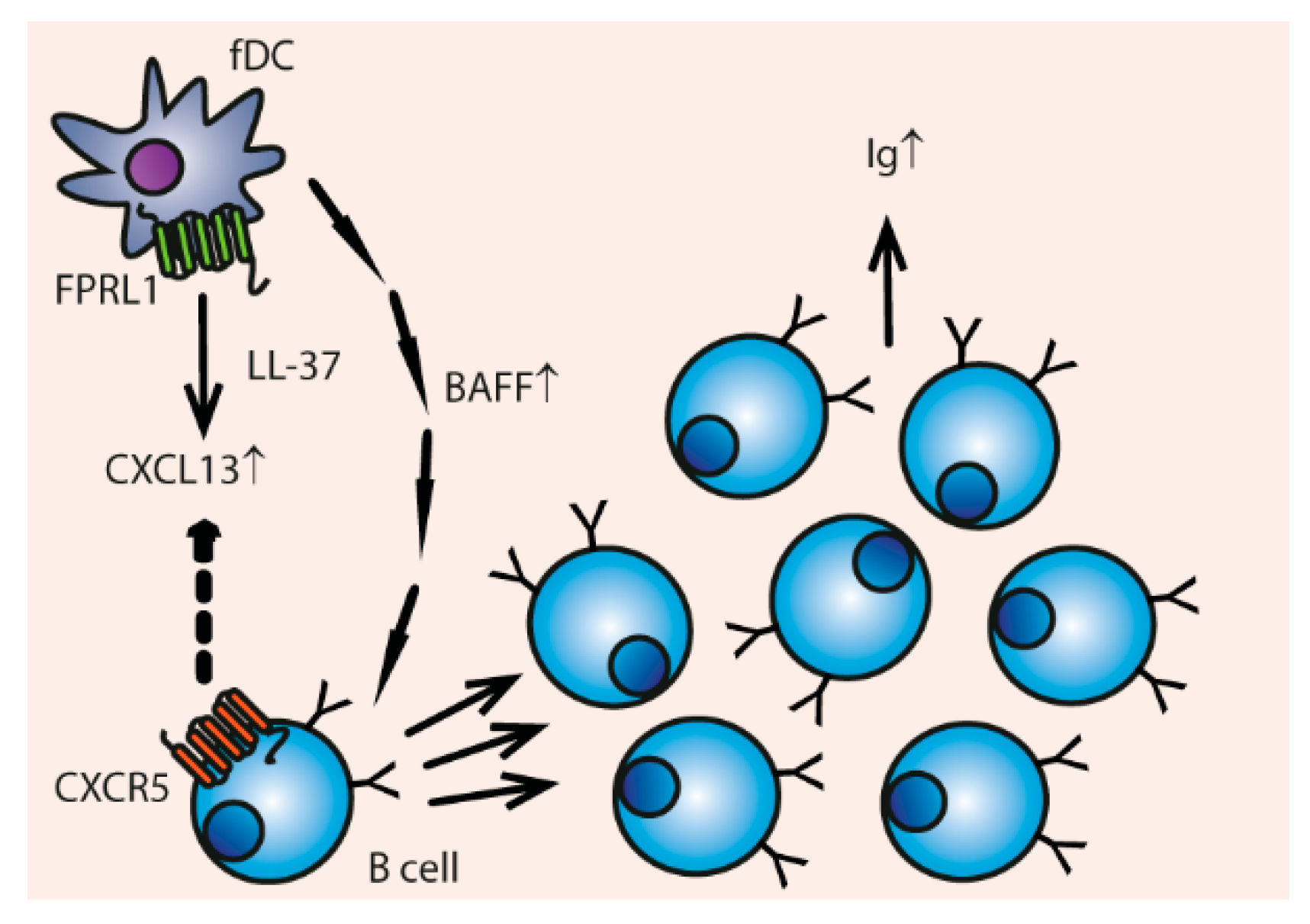
3.5. Cell Differentiation and Proliferation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.; Wertheim, H.F.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H. Antibiotic resistance—The need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Payne, D.J.; Gwynn, M.N.; Holmes, D.J.; Pompliano, D.L. Drugs for bad bugs: Confronting the challenges of antibacterial discovery. Nat. Rev. Drug Discov. 2007, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 2: Management strategies and new agents. Pharm. Ther. 2015, 40, 344–352. [Google Scholar]
- Baym, M.; Stone, L.K.; Kishony, R. Multidrug evolutionary strategies to reverse antibiotic resistance. Science 2016, 351. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Storek, K.M.; Auerbach, M.R.; Shi, H.; Garcia, N.K.; Sun, D.; Nickerson, N.N.; Vij, R.; Lin, Z.; Chiang, N.; Schneider, K.; et al. Monoclonal antibody targeting the beta-barrel assembly machine of escherichia coli is bactericidal. Proc. Natl. Acad. Sci. USA 2018, 115, 3692–3697. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, T.J.; Holthausen, D.J.; Hsieh, C.; Renken, C.; Mannella, C.A.; Benach, J.L. The bactericidal effect of a complement-independent antibody is osmolytic and specific to borrelia. Proc. Natl. Acad. Sci. USA 2009, 106, 10752–10757. [Google Scholar] [CrossRef] [PubMed]
- Szijártó, V.; Nagy, E.; Nagy, G. Directly bactericidal anti-escherichia coli antibody. Trends Microbiol. 2018, 26, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Ifrim, D.C.; Quintin, J.; Joosten, L.A.B.; Jacobs, C.; Jansen, T.; Jacobs, L.; Gow, N.A.R.; Williams, D.L.; van der Meer, J.W.M.; Netea, M.G. Trained immunity or tolerance: Opposing functional programs induced in human monocytes after engagement of various pattern recognition receptors. Clin. Vaccine Immunol. 2014, 21, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Joosten, L.A.B.; Latz, E.; Mills, K.H.G.; Natoli, G.; Stunnenberg, H.G.; O’Neill, L.A.J.; Xavier, R.J. Trained immunity: A program of innate immune memory in health and disease. Science 2016, 352. [Google Scholar] [CrossRef] [PubMed]
- Mitroulis, I.; Ruppova, K.; Wang, B.; Chen, L.-S.; Grzybek, M.; Grinenko, T.; Eugster, A.; Troullinaki, M.; Palladini, A.; Kourtzelis, I. Modulation of myelopoiesis progenitors is an integral component of trained immunity. Cell 2018, 172, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Arts, R.J.W.; Moorlag, S.J.C.F.M.; Novakovic, B.; Li, Y.; Wang, S.-Y.; Oosting, M.; Kumar, V.; Xavier, R.J.; Wijmenga, C.; Joosten, L.A.B.; et al. Bcg vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe 2018, 23, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Haney, E.F.; Gill, E.E. The immunology of host defence peptides: Beyond antimicrobial activity. Nat. Rev. Immunol. 2016, 16, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Hilchie, A.L.; Wuerth, K.; Hancock, R.E. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat. Chem. Biol. 2013, 9, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Mansour, S.C.; Pena, O.M.; Hancock, R.E.W. Host defense peptides: Front-line immunomodulators. Trends Immunol. 2014, 35, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Biragyn, A.; Kwak, L.W.; Oppenheim, J.J. Mammalian defensins in immunity: More than just microbicidal. Trends Immunol. 2002, 23, 291–296. [Google Scholar] [CrossRef]
- Ganz, T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003, 3, 710–720. [Google Scholar] [CrossRef] [PubMed]
- Hemshekhar, M.; Anaparti, V.; Mookherjee, N. Functions of cationic host defense peptides in immunity. Pharmaceuticals 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Ageitos, J.M.; Sánchez-Pérez, A.; Calo-Mata, P.; Villa, T.G. Antimicrobial peptides (amps): Ancient compounds that represent novel weapons in the fight against bacteria. Biochem. Pharmacol. 2017, 133, 117–138. [Google Scholar] [CrossRef] [PubMed]
- Kościuczuk, E.M.; Lisowski, P.; Jarczak, J.; Strzałkowska, N.; Jóźwik, A.; Horbańczuk, J.; Krzyzewski, J.; Zwierzchowski, L.; Bagnicka, E. Cathelicidins: Family of antimicrobial peptides. A review. Mol. Biol. Rep. 2012, 39, 10957–10970. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Blecha, F. Porcine host defense peptides: Expanding repertoire and functions. Dev. Comp. Immunol. 2009, 33, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Zanetti, M. The role of cathelicidins in the innate host defenses of mammals. Curr. Issues Mol. Biol. 2005, 7, 179–196. [Google Scholar] [PubMed]
- Agier, J.; Efenberger, M.; Brzezińska-Blaszczyk, E. Cathelicidin impact on inflammatory cells. Cent. Eur. J. Immunol. 2015, 40, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, O.E.; Gram, L.; Johnsen, A.H.; Andersson, E.; Bangsboll, S.; Tjabringa, G.S.; Hiemstra, P.S.; Malm, J.; Egesten, A.; Borregaard, N. Processing of seminal plasma hcap-18 to all-38 by gastricsin: A novel mechanism of generating antimicrobial peptides in vagina. J. Biol. Chem. 2003, 278, 28540–28546. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Lopez-Garcia, B.; Braff, M.; Dorschner, R.A.; Gallo, R.L. Postsecretory processing generates multiple cathelicidins for enhanced topical antimicrobial defense. J. Immunol. 2004, 172, 3070–3077. [Google Scholar] [CrossRef] [PubMed]
- Coorens, M.; Scheenstra, M.R.; Veldhuizen, E.J.; Haagsman, H.P. Interspecies cathelicidin comparison reveals divergence in antimicrobial activity, tlr modulation, chemokine induction and regulation of phagocytosis. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, M.; Jenssen, H.; Kindrachuk, J.; Scott, W.R.; Elliott, M.; Hilpert, K.; Cheng, J.T.; Hancock, R.E.; Straus, S.K. Structural studies of a peptide with immune modulating and direct antimicrobial activity. Chem. Biol. 2010, 17, 970–980. [Google Scholar] [CrossRef] [PubMed]
- Bommineni, Y.R.; Dai, H.; Gong, Y.X.; Soulages, J.L.; Fernando, S.C.; DeSilva, U.; Prakash, O.; Zhang, G. Fowlicidin-3 is an α-helical cationic host defense peptide with potent antibacterial and lipopolysaccharide-neutralizing activities. FEBS J. 2007, 274, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, A.; Molhoek, E.M.; Veldhuizen, E.J.A.; Bokhoven, J.L.M.T.v.; Wagendorp, E.; Bikker, F.; Haagsman, H.P. Identification of chicken cathelicidin-2 core elements involved in antibacterial and immunomodulatory activities. Mol. Immunol. 2009, 46, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- Tomasinsig, L.; Pizzirani, C.; Skerlavaj, B.; Pellegatti, P.; Gulinelli, S.; Tossi, A.; Virgilio, F.D.; Zanetti, M. The human cathelicidin ll-37 modulates the activities of the p2x7 receptor in a structure-dependent manner. J. Biol. Chem. 2008, 283, 30471–30481. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, O.; Cowland, J.B.; Askaa, J.; Borregaard, N. An elisa for hcap-18, the cathelicidin present in human neutrophils and plasma. J. Immunol. Methods 1997, 206, 53–59. [Google Scholar] [CrossRef]
- Bowdish, D.M.E.; Davidson, D.J.; Lau, Y.E.; Lee, K.; Scott, M.G.; Hancock, R.E.W. Impact of ll-37 on anti-infective immunity. J. Leukoc. Biol. 2005, 77, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.I.U.; Schaller-Bals, S.; Paul, K.P.; Wahn, U.; Bals, R. B-defensins and ll-37 in bronchoalveolar lavage fluid of patients with cystic fibrosis. J. Cyst. Fibros. 2004, 3, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Schaller-Bals, S.; Schulze, A.; Bals, R. Increased levels of antimicrobial peptides in tracheal apirates of newborn infants during infection. Am. J. Respir. Crit. Care Med. 2002, 165, 992–995. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C. Toll-like receptor triggering of a vitamin d-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Yim, S.; Dhawan, P.; Ragunath, C.; Christakos, S.; Diamond, G. Induction of cathelicidin in normal and cf bronchial epithelial cells by 1,25-dihydroxyvitamin d3. J. Cyst. Fibros. 2007, 6, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Rode, A.K.O.; Kongsbak, M.; Hansen, M.M.; Lopez, D.V.; Levring, T.B.; Woetmann, A.; Ødum, N.; Bonefeld, C.M.; Geisler, C. Vitamin d counteracts mycobacterium tuberculosis-induced cathelicidin downregulation in dendritic cells and allows th1 differentiation and ifnγ secretion. Front. Immunol. 2017, 8, 656. [Google Scholar] [CrossRef] [PubMed]
- Mansour, S.C.; Fuente-Núñez, C.; Hancock, R.E.W. Peptide idr-1018: Modulating the immune system and targeting bacterial biofilms to treat antibiotic-resistant bacterial infections. J. Pept. Sci. 2015, 21, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Pena, O.M.; Afacan, N.; Pistolic, J.; Chen, C.; Madera, L.; Falsafi, R.; Fjell, C.D.; Hancock, R.E. Synthetic cationic peptide idr-1018 modulates human macrophage differentiation. PLoS ONE 2013, 8, e52449. [Google Scholar] [CrossRef] [PubMed]
- Achtman, A.H.; Pilat, S.; Law, C.W.; Lynn, D.J.; Janot, L.; Mayer, M.L.; Ma, S.; Kindrachuk, J.; Finlay, B.B.; Brinkman, F.S.L.; et al. Effective adjunctive therapy by an innate defense regulatory peptide in a preclinical model of severe malaria. Sci. Transl. Med. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Pane, K.; Durante, L.; Crescenzi, O.; Cafaro, V.; Pizzo, E.; Varcamonti, M.; Zanfardino, A.; Izzo, V.; Di Donato, A.; Notomista, E. Antimicrobial potency of cationic antimicrobial peptides can be predicted from their amino acid composition: Application to the detection of “cryptic” antimicrobial peptides. J. Theor. Biol. 2017, 419, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Bosso, A.; Pirone, L.; Gaglione, R.; Pane, K.; Del Gatto, A.; Zaccaro, L.; Di Gaetano, S.; Diana, D.; Fattorusso, R.; Pedone, E.; et al. A new cryptic host defense peptide identified in human 11-hydroxysteroid dehydrogenase-1 β-like: From in silico identification to experimental evidence. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2342–2353. [Google Scholar] [CrossRef] [PubMed]
- Zanfardino, A.; Bosso, A.; Gallo, G.; Pistorio, V.; Di Napoli, M.; Gaglione, R.; Dell’Olmo, E.; Varcamonti, M.; Notomista, E.; Arciello, A.; et al. Human apolipoprotein e as a reservoir of cryptic bioactive peptides: The case of apoe 133-167. J. Pept. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Goitsuka, R.; Chen, C.-L.H.; Benyon, L.; Asano, Y.; Kitamura, D.; Cooper, M.D. Chicken cathelicidin-b1, an antimicrobial guardian at the mucosal m cell gateway. Proc. Natl. Acad. Sci. USA 2007, 104, 15063–15068. [Google Scholar] [CrossRef] [PubMed]
- Tossi, A.; Scocchi, M.; Zanetti, M.; Storici, P.; Gennaro, R. Pmap-37, a novel antibacterial peptide from pig myeloid cells. Cdna cloning, chemical synthesis and activity. Eur. J. Biochem. 1995, 228, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Veldhuizen, E.J.A.; Scheenstra, M.R.; Tjeerdsma-van Bokhoven, J.L.M.; Coorens, M.; Schneider, V.A.F.; Bikker, F.J.; van Dijk, A.; Haagsman, H.P. Antimicrobial and immunomodulatory activity of pmap-23 derived peptides. Protein Pept. Lett. 2017, 24, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Wessely-Szponder, J.; Majer-Dziedzic, B.; Smolira, A. Analysis of antimicrobial peptides from porcine neutrophils. J. Microbiol. Methods 2010, 83, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Cai, Y.; Bommineni, Y.R.; Fernando, S.C.; Prakash, O.; Gilliland, S.E.; Zhang, G. Identification and functional characterization of three chicken cathelicidins with potent antimicrobial activity. J. Biol. Chem. 2006, 281, 2858–2867. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.; Weldon, S.; Buchanan, P.J.; Schock, B.; Ernst, R.K.; McAuley, D.F.; Tunney, M.M.; Irwin, C.R.; Elborn, J.S.; Taggart, C.C. Evaluation of the ability of ll-37 to neutralise lps in vitro and ex vivo. PLoS ONE 2011, 6, e26525. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.J.; Travis, S.M.; Greenberg, E.P.; Welsh, M.J. Cystic fibrosis airway epithelia fail to kill bacteria because of abnormal airway surface fluid. Cell 1996, 85, 229–236. [Google Scholar] [CrossRef]
- De Buhr, N.; Reuner, F.; Neumann, A.; Stump-Guthier, C.; Tenenbaum, T.; Schroten, H.; Ishikawa, H.; Müller, K.; Beineke, A.; Hennig-Pauka, I.; et al. Neutrophil extracellular trap formation in the streptococcus suis-infected cerebrospinal fluid compartment. Cell. Microbiol. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, H.; Nakamura, K.; Hu, Z.; Tamura, H.; Reich, J.; Kuwahara-Arai, K.; Iba, T.; Tabe, Y.; Nagaoaka, I. Antimicrobial cathelicidin peptide ll37 induces net formation and suppresses the inflammatory response in a mouse septic model. Mol. Med. Rep. 2017, 16, 5618–5626. [Google Scholar] [CrossRef] [PubMed]
- Neumann, A.; Völlger, L.; Berends, E.T.M.; Molhoek, E.M.; Stapels, D.A.C.; Midon, M.; Friães, A.; Pingoud, A.; Rooijakkers, S.H.M.; Gallo, R.L.; et al. Novel role of the antimicrobial peptide ll-37 in the protection of neutrophil extracellular traps against degradation by bacterial nucleases. J. Innate Immunity 2014, 6, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Riool, M.; de Breij, A.; de Boer, L.; Kwakman, P.H.S.; Cordfunke, R.A.; Cohen, O.; Malanovic, N.; Emanuel, N.; Lohner, K.; Drijfhout, J.W.; et al. Controlled release of ll-37-derived synthetic antimicrobial and anti-biofilm peptides saap-145 and saap-276 prevents experimental biomaterial-associated staphylococcus aureus infection. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Onaizi, S.A.; Leong, S.S.J. Tethering antimicrobial peptides: Current status and potential challenges. Biotechnol. Adv. 2011, 29, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Bommineni, Y.R.; Pham, G.H.; Sunkara, L.T.; Achanta, M.; Zhang, G. Immune regulatory activities of fowlicidin-1, a cathelicidin host defense peptide. Mol. Immunol. 2014, 59, 55–63. [Google Scholar] [CrossRef] [PubMed]
- LaRock, C.N.; Döhrmann, S.; Todd, J.; Corriden, R.; Olson, J.; Johannssen, T.; Lepenies, B.; Gallo, R.L.; Ghosh, P.; Nizet, V. Group a streptococcal m1 protein sequesters cathelicidin to evade innate immune killing. Cell Host Microbe 2015, 18, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Nizet, V.; Ohtake, T.; Lauth, X.; Trowbridge, J.; Rudisill, J.; Dorschner, R.A.; Pestonjamasp, V.; Piraino, J.; Huttner, K.; Gallo, R.L. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature 2001, 414, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.H.A.; Ohtake, T.; Zaiou, M.; Murakami, M.; Rudisill, J.A.; Lin, K.H.; Gallo, R.L. Expression of an additional cathelicidin antimicrobial peptide protects against bacterial skin infection. Proc. Natl. Acad. Sci. USA 2005, 102, 3750–3755. [Google Scholar] [CrossRef] [PubMed]
- Cuperus, T.; van Dijk, A.; Matthijs, M.G.R.; Veldhuizen, E.J.A.; Haagsman, H.P. Protective effect of in ovo treatment with the chicken cathelicidin analog d-cath-2 against avian pathogenic E. coli. Sci. Rep. 2016, 6, 26622. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Subramanian, H.; Ali, H. Modulation of host defense peptide-mediated human mast cell activation by lps. Innate Immunity 2016, 22, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhang, Y.; Zhang, Y.; Lai, Y.; Chen, W.; Xiao, Z.; Zhang, W.; Jin, M.; Yu, B. Ll-37-induced human mast cell activation through g protein-coupled receptor mrgx2. Int. Immunopharmacol. 2017, 49, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, H.; Gupta, K.; Guo, Q.; Price, R.; Ali, H. Mas-related gene x2 (mrgx2) is a novel g protein-coupled receptor for the antimicrobial peptide ll-37 in human mast cells: Resistance to receptor phosphorylation, desensitization, and internalization. J. Biol. Chem. 2011, 286, 44739–44749. [Google Scholar] [CrossRef] [PubMed]
- Schiemann, F.; Brandt, E.; Gross, R.; Lindner, B.; Mittelstädt, J.; Sommerhoff, C.P.; Schulmistrat, J.; Petersen, F. The cathelicidin ll-37 activates human mast cells and is degraded by mast cell tryptase: Counter-regulation by cxcl4. J. Immunol. 2009, 183, 2223–2231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-Y.; Yu, Y.-Y.; Zhang, Y.-R.; Zhang, W.; Yu, B. The modulatory effect of tlr2 on ll-37-induced human mast cells activation. Biochem. Biophys. Res. Commun. 2016, 470, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Baumann, A.; Demoulins, T.; Python, S.; Summerfield, A. Porcine cathelicidins efficiently complex and deliver nucleic acids to plasmacytoid dendritic cells and can thereby mediate bacteria-induced ifn-alpha responses. J. Immunol. 2014, 193, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Niyonsaba, F.; Ushio, H.; Nagaoka, I.; Ikeda, S.; Okumura, K.; Ogawa, H. Cathelicidin ll-37 induces the generation of reactive oxygen species and release of human α-defensins from neutrophils. Br. J. Dermatol. 2007, 157, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Ross, C.R.; Leto, T.L.; Blecha, F.; Klebanoff, S.J. Pr-39, a proline-rich antibacterial peptide that inhibits phagocyte nadph oxidase activity by binding to src homology 3 domains of p47phox. Proc. Natl. Acad. Sci. USA 1996, 93, 6014–6018. [Google Scholar] [CrossRef] [PubMed]
- Neumann, A.; Berends, E.T.; Nerlich, A.; Molhoek, E.M.; Gallo, R.L.; Meerloo, T.; Nizet, V.; Naim, H.Y.; von Köckritz-Blickwede, M. The antimicrobial peptide ll-37 facilitates the formation of neutrophil extracellular traps. Biochem. J. 2014, 464, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Papayannopoulos, V.; Zychlinsky, A. Nets: A new strategy for using old weapons. Trends Immunol. 2009, 30, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Overhage, J.; Campisano, A.; Bains, M.; Torfs, E.C.W.; Rehm, B.H.A.; Hancock, R.E.W. Human host defense peptide ll-37 prevents bacterial biofilm formation. Infect. Immunity 2008, 76, 4176–4182. [Google Scholar] [CrossRef] [PubMed]
- Coorens, M.; van Dijk, A.; Bikker, F.; Veldhuizen, E.J.; Haagsman, H.P. Importance of endosomal cathelicidin degradation to enhance DNA-induced chicken macrophage activation. J. Immunol. 2015, 195, 3970–3977. [Google Scholar] [CrossRef] [PubMed]
- D’Este, F.; Tomasinsig, L.; Skerlavaj, B.; Zanetti, M. Modulation of cytokine gene expression by cathelicidin bmap-28 in lps-stimulated and -unstimulated macrophages. Immunobiology 2012, 217, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Veldhuizen, E.J.; Schneider, V.A.; Agustiandari, H.; Van Dijk, A.; Tjeerdsma-van Bokhoven, J.L.; Bikker, F.J.; Haagsman, H.P. Antimicrobial and immunomodulatory activities of pr-39 derived peptides. PLoS ONE 2014, 9, e95939. [Google Scholar] [CrossRef] [PubMed]
- Coorens, M.; Schneider, V.A.F.; de Groot, A.M.; van Dijk, A.; Meijerink, M.; Wells, J.M.; Scheenstra, M.R.; Veldhuizen, E.J.A.; Haagsman, H.P. Cathelicidins inhibit escherichia coli–induced tlr2 and tlr4 activation in a viability-dependent manner. J. Immunol. 2017, 199, 1418–1428. [Google Scholar] [CrossRef] [PubMed]
- Elssner, A.; Duncan, M.; Gavrilin, M.; Wewers, M.D. A novel p2x7 receptor activator, the human cathelicidin-derived peptide ll37, induces il-1 processing and release. J. Immunol. 2004, 172, 4987–4994. [Google Scholar] [CrossRef] [PubMed]
- Wiley, J.S.; Sluyter, R.; Gu, B.J.; Stokes, L.; Fuller, S.J. The human p2x7 receptor and its role in innate immunity. Tissue Antigens 2011, 78, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Yamasaki, K.; Saito, R.; Fukushi-Takahashi, S.; Shimada-Omori, R.; Asano, M.; Aiba, S. Alarmin function of cathelicidin antimicrobial peptide ll37 through il-36γ induction in human epidermal keratinocytes. J. Immunol. 2014, 193, 5140–5148. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.G.; Davidson, D.J.; Gold, M.R.; Bowdish, D.; Hancock, R.E. The human antimicrobial peptide ll-37 is a multifunctional modulator of innate immune responses. J. Immunol. 2002, 169, 3883–3891. [Google Scholar] [CrossRef] [PubMed]
- Pistolic, J.; Cosseau, C.; Li, Y.; Yu, J.; Filewod, N.C.J.; Gellatly, S.; Rehaume, L.M.; Bowdish, D.M.E.; Hancock, R.E.W. Host defence peptide ll-37 induces il-6 expression in human bronchial epithelial cells by activation of the nf-κb signaling pathway. J. Innate Immunity 2009, 1, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Medina Santos, C.E.; López Hurtado, C.N.; Rivas Santiago, B.; Gonzalez-Amaro, R.; Cataño Cañizales, Y.G.; Martínez Fierro, M.d.l.L.; Enciso-Moreno, J.A.; García Hernández, M.H. Ll-37, hnp-1, and hbd2/3 modulate the secretion of cytokines tnf-α, il-6, ifn-γ, il-10 and mmp1 in human primary cell cultures. Eur. Cytokine Netw. 2016, 27, 68–74. [Google Scholar] [PubMed]
- Mookherjee, N.; Lippert, D.N.D.; Hamill, P.; Falsafi, R.; Nijnik, A.; Kindrachuk, J.; Pistolic, J.; Gardy, J.; Miri, P.; Naseer, M.; et al. Intracellular receptor for human host defense peptide ll-37 in monocytes. J. Immunol. 2009, 183, 2688–2696. [Google Scholar] [CrossRef] [PubMed]
- Oren, Z.; Lerman, J.C.; Gudmundsson, G.H.; Agerberth, B.; Shai, Y. Structure and organization of the human antimicrobial peptide ll-37 in phospholipid membranes: Relevance to the molecular basis for its non-cell-selective activity. Biochem. J. 1999, 341, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.; Gudmundsson, G.H.; Rottenberg, M.E.; Berndt, K.D.; Agerberth, B. Conformation-dependent antibacterial activity of the naturally occurring human peptide ll-37. J. Biol. Chem. 1998, 273, 3718–3724. [Google Scholar] [CrossRef] [PubMed]
- Lishko, V.; Moreno, B.; Podolnikova, N.; Ugarova, T. Identification of human cathelicidin peptide ll-37 as a ligand for macrophage integrin αmβ2 (mac-1, cd11b/cd18) that promotes phagocytosis by opsonizing bacteria. Res. Rep. Biochem. 2016, 6, 39–55. [Google Scholar]
- Zhang, X.; Bajic, G.; Andersen, G.R.; Hill Christiansen, S.; Vorup-Jensen, T. The cationic peptide ll-37 binds mac-1 (cd11b/cd18) with a low dissociation rate and promotes phagocytosis. Biochim. Biophys. Acta Proteins Proteom. 2016, 1864, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Wan, M.; van der Does, A.M.; Tang, X.; Lindbom, L.; Agerberth, B.; Haeggstrom, J.Z. Antimicrobial peptide ll-37 promotes bacterial phagocytosis by human macrophages. J. Leukoc. Biol. 2014, 95, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Kress, E.; Merres, J.; Albrecht, L.-J.; Hammerschmidt, S.; Pufe, T.; Tauber, S.C.; Brandenburg, L.-O. Cramp deficiency leads to a pro-inflammatory phenotype and impaired phagocytosis after exposure to bacterial meningitis pathogens. Cell Commun. Signal. 2017, 15, 32. [Google Scholar] [CrossRef] [PubMed]
- Underhill, D.M.; Goodridge, H.S. Information processing during phagocytosis. Nat. Rev. Immunol. 2012, 12, 492–502. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, P.; Peh, C.A. Ll-37 promotes rapid sensing of cpg oligodeoxynucleotides by b lymphocytes and plasmacytoid dendritic cells. J. Immunol. 2010, 184, 1425–1435. [Google Scholar] [CrossRef] [PubMed]
- Lande, R.; Gregorio, J.; Facchinetti, V.; Chatterjee, B.; Wang, Y.-H.; Homey, B.; Cao, W.; Wang, Y.-H.; Su, B.; Nestle, F.O.; et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature 2007, 449, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Baumann, A.; Kiener, M.S.; Haigh, B.; Perreten, V.; Summerfield, A. Differential ability of bovine antimicrobial cathelicidins to mediate nucleic acid sensing by epithelial cells. Front. Immunol. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Qi, R.; Jordan, J.L.; San Mateo, L.; Kao, C.C. The human antimicrobial peptide ll-37, but not the mouse ortholog, mcramp, can stimulate signaling by poly(i:C) through a fprl1-dependent pathway. J. Biol. Chem. 2013, 288, 8258–8268. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, D.; Chamilos, G.; Lande, R.; Gregorio, J.; Meller, S.; Facchinetti, V.; Homey, B.; Barrat, F.J.; Zal, T.; Gilliet, M. Self-rna-antimicrobial peptide complexes activate human dendritic cells through tlr7 and tlr8. J. Exp. Med. 2009, 206, 1983–1994. [Google Scholar] [CrossRef] [PubMed]
- Uematsu, S.; Akira, A. Toll-Like Receptors and Innate Immunity. In Handbook of Experimental Pharmacology; Springer: Berlin, Germany, 2008; Volume 183. [Google Scholar]
- Ghiselli, R.; Giacometti, A.; Cirioni, O.; Circo, R.; Mocchegiani, F.; Skerlavaj, B.; D’Amato, G.; Scalise, G.; Zanetti, M.; Saba, V. Neutralization of endotoxin in vitro and in vivo by bac7(1-35), a proline-rich antibacterial peptide. Shock 2003, 19, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Giacometti, A.; Cirioni, O.; Ghiselli, R.; Bergnach, C.; Orlando, F.; D’Amato, G.; Mocchegiani, F.; Silvestri, C.; Del Prete, M.S.; Skerlavaj, B.; et al. The antimicrobial peptide bmap-28 reduces lethality in mouse models of staphylococcal sepsis. Crit. Care Med. 2004, 32, 2485–2490. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Shimomura, Y.; Yoshida, M.; Morgan, J.G.; Palings, I.; Wilson, D.; Yen, M.H.; Wright, S.C.; Larrick, J.W. Characterization of a rabbit cationic protein (cap18) with lipopolysaccharide-inhibitory activity. Infect. Immunity 1994, 62, 1421–1426. [Google Scholar]
- Larrick, F.W.; Hirata, M.; Balint, R.F.; Lee, J.; Zhong, J.; Wright, S.C. Human cap18: A novel antimicrobial lipopolysaccharide-binding protein. Infect. Immunity 1995, 63, 1291–1297. [Google Scholar]
- Bowdish, D.M.E.; Davidson, D.J.; Scott, M.G.; Hancock, R.E.W. Immunomodulatory activities of small host defense peptides. Antimicrob. Agents Chemother. 2005, 49, 1727–1732. [Google Scholar] [CrossRef] [PubMed]
- Mookherjee, N.; Brown, K.L.; Bowdish, D.M.E.; Doria, S.; Falsafi, R.; Hokamp, K.; Roche, F.M.; Mu, R.; Doho, G.H.; Pistolic, J.; et al. Modulation of the tlr-mediated inflammatory response by the endogenous human host defense peptide ll-37. J. Immunol. 2006, 176, 2455–2464. [Google Scholar] [CrossRef] [PubMed]
- Nijnik, A.; Pistolic, J.; Wyatt, A.; Tam, S.; Hancock, R.E.W. Human cathelicidin peptide ll-37 modulates the effects of ifn-ɣ on apcs. J. Immunol. 2009, 183, 5788–5798. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Y.; Shen, T.; Wang, Y.; Hou, M.; Li, J.; Sun, T. Antimicrobial peptide ll-37 attenuates lta induced inflammatory effect in macrophages. Int. Immunopharmacol. 2013, 15, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Mookherjee, N.; Wilson, H.L.; Doria, S.; Popowych, Y.; Falsafi, R.; Yu, J.J.; Li, Y.; Veatch, S.; Roche, F.M.; Brown, K.L.; et al. Bovine and human cathelicidin cationic host defense peptides similarly suppress transcriptional responses to bacterial lipopolysaccharide. J. Leukoc. Biol. 2006, 80, 1563–1574. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, A.; van Eldik, M.; Veldhuizen, E.J.; Tjeerdsma-van Bokhoven, H.L.; de Zoete, M.R.; Bikker, F.J.; Haagsman, H.P. Immunomodulatory and anti-inflammatory activities of chicken cathelicidin-2 derived peptides. PLoS ONE 2016, 11, e0147919. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, A.; Braff, M.H.; Taylor, K.R.; Na, C.; Granstein, R.D.; McInturff, J.E.; Krutzik, S.; Modlin, R.L.; Gallo, R.L. Cathelicidin antimicrobial peptides block dendritic cell tlr4 activation and allergic contact sensitization. J. Immunol. 2007, 178, 1829–1834. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.; Silva, J.P.; Rodrigues, A.C.; Costa, R.; Guardão, L.; Schmitt, F.; Soares, R.; Vilanova, M.; Domingues, L.; Gama, M. Wound healing activity of the human antimicrobial peptide ll37. Peptides 2011, 32, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Kandler, K.; Shaykhiev, R.; Kleemann, P.; Klescz, F.; Lohoff, M.; Vogelmeier, C.; Bals, R. The anti-microbial peptide ll-37 inhibits the activation of dendritic cells by tlr ligands. Int. Immunol. 2006, 18, 1729–1736. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, Y.; Papo, N.; Shai, Y. Endotoxin (lipopolysaccharide) neutralization by innate immunity host-defense peptides: Peptide properties and plausible modes of action. J. Biol. Chem. 2006, 281, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Kraaij, M.D.; van Dijk, A.; Haagsman, H.P. Cath-2 and ll-37 increase mannose receptor expression, antigen presentation and the endocytic capacity of chicken mononuclear phagocytes. Mol. Immunol. 2017, 90, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Cherryholmes, G.; Shively, J.E. Neutrophil secondary necrosis is induced by ll-37 derived from cathelicidin. J. Leukoc. Biol. 2008, 84, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.-Y.G.; Napper, S.; Mookherjee, N. Human cathelicidin ll-37 and its derivative ig-19 regulate interleukin-32-induced inflammation. Immunology 2014, 143, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Giacometti, A.; Cirioni, O.; Ghiselli, R.; Mocchegiani, F.; D’Amato, G.; Circo, R.; Orlando, F.; Skerlavaj, B.; Silvestri, C.; Saba, V.; et al. Cathelicidin peptide sheep myeloid antimicrobial peptide 29 prevents endotoxin-induced mortality in rat models of septic shock. Am. J. Respir. Crit. Care Med. 2004, 169, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Hing, T.C.; Ho, S.; Shih, D.Q.; Ichikawa, R.; Cheng, M.; Chen, J.; Chen, X.; Law, I.; Najarian, R.; Kelly, C.P.; et al. The antimicrobial peptide cathelicidin modulates clostridium difficile-associated colitis and toxin a-mediated enteritis in mice. Gut 2013, 62, 1295–1305. [Google Scholar] [CrossRef] [PubMed]
- Severino, P.; Ariga, S.K.; Barbeiro, H.V.; de Lima, T.M.; de Paula Silva, E.; Barbeiro, D.F.; Machado, M.C.C.; Nizet, V.; Pinheiro da Silva, F. Cathelicidin-deficient mice exhibit increased survival and upregulation of key inflammatory response genes following cecal ligation and puncture. J. Mol. Med. 2017, 95, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.Y.; Shamoon, M.; He, Y.; Bhatia, M.; Sun, J. Cathelicidin-related antimicrobial peptide modulates the severity of acute pancreatitis in mice. Mol. Med. Rep. 2016, 13, 3881–3885. [Google Scholar] [CrossRef] [PubMed]
- Tjabringa, G.S.; Ninaber, D.K.; Drijfhout, J.W.; Rabe, K.F.; Hiemstra, P.S. Human cathelicidin ll-37 is a chemoattractant for eosinophils and neutrophils that acts via formyl-peptide receptors. Int. Arch. Allergy Immunol. 2006, 140, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Chen, Q.; Schmidt, A.P.; Anderson, G.M.; Wang, J.M.; Wooters, J.; Oppenheim, J.J.; Chertov, O. Ll-37, the neutrophil granule–and epithelial cell–derived cathelicidin, utilizes formyl peptide receptor–like 1 (fprl1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and t cells. J. Exp. Med. 2000, 192, 1069–1074. [Google Scholar] [CrossRef] [PubMed]
- Niyonsaba, F.; Iwabuchi, K.; Someya, A.; Hirata, M.; Matsuda, H.; Ogawa, H.; Nagaoka, I. A cathelicidin family of human antibacterial peptide ll-37 induces mast cell chemotaxis. Immunology 2002, 106, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Chen, Q.; Le, Y.; Wang, J.M.; Oppenheim, J.J. Differential regulation of formyl peptide receptor-like 1 expression during the differentiation of monocytes to dendritic cells and macrophages. J. Immunol. 2001, 166, 4092–4098. [Google Scholar] [CrossRef] [PubMed]
- Shaykhiev, R. Human endogenous antibiotic ll-37 stimulates airway epithelial cell proliferation and wound closure. AJP Lung Cell. Mol. Physiol. 2005, 289. [Google Scholar] [CrossRef] [PubMed]
- Bowdish, D.M.E.; Davidson, D.J.; Speert, D.P.; Hancock, R.E.W. The human cationic peptide ll-37 induces activation of the extracellular signal-regulated kinase and p38 kinase pathways in primary human monocytes. J. Immunol. 2004, 172, 3758–3765. [Google Scholar] [CrossRef] [PubMed]
- Kurosaka, K.; Chen, Q.; Yarovinsky, F.; Oppenheim, J.J.; Yang, D. Mouse cathelin-related antimicrobial peptide chemoattracts leukocytes using formyl peptide receptor-like 1/mouse formyl peptide receptor-like 2 as the receptor and acts as an immune adjuvant. J. Immunol. 2005, 174, 6257–6265. [Google Scholar] [CrossRef] [PubMed]
- Kindrachuk, J.; Scruten, E.; Attah-Poku, S.; Bell, K.; Potter, A.; Babiuk, L.A.; Griebel, P.J.; Napper, S. Stability, toxicity, and biological activity of host defense peptide bmap28 and its inversed and retro-inversed isomers. Biopolymers 2011, 96, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Verbanac, D.; Zanetti, M.; Romeo, D. Chemotactic and protease-inhibiting activities of antibiotic peptide precursors. FEBS Lett. 1993, 317, 255–258. [Google Scholar] [CrossRef]
- Babolewska, E.; Pietrzak, A.; Brzezińska-Błaszczyk, E. Cathelicidin rcramp stimulates rat mast cells to generate cysteinyl leukotrienes, synthesize tnf and migrate: Involvement of plc/a2, pi3k and mapk signaling pathways. Int. Immunol. 2014, 26, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-J.; Ross, C.R.; Blecha, F. Chemoattractant properties of pr-39, a neutrophil antibacterial peptide. J. Leukoc. Biol. 1997, 61, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Tjabringa, G.S.; Aarbiou, J.; Ninaber, D.K.; Drijfhout, J.W.; Sørensen, O.E.; Borregaard, N.; Rabe, K.F.; Hiemstra, P.S. The antimicrobial peptide ll-37 activates innate immunity at the airway epithelial surface by transactivation of the epidermal growth factor receptor. J. Immunol. 2003, 171, 6690–6696. [Google Scholar] [CrossRef] [PubMed]
- Van der Does, A.M.; Beekhuizen, H.; Ravensbergen, B.; Vos, T.; Ottenhoff, T.H.M.; van Dissel, J.T.; Drijfhout, J.W.; Hiemstra, P.S.; Nibbering, P.H. Ll-37 directs macrophage differentiation toward macrophages with a proinflammatory signature. J. Immunol. 2010, 185, 1442–1449. [Google Scholar] [CrossRef] [PubMed]
- Davidson, D.J.; Currie, A.J.; Reid, G.S.D.; Bowdish, D.M.E.; MacDonald, K.L.; Ma, R.C.; Hancock, R.E.W.; Speert, D.P. The cationic antimicrobial peptide ll-37 modulates dendritic cell differentiation and dendritic cell-induced t cell polarization. J. Immunol. 2004, 172, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Xiang, Y.; Huang, J.; Gong, W.; Yoshimura, T.; Jiang, Q.; Tessarollo, L.; Le, Y.; Wang, J.M. The formylpeptide receptor 2 (fpr2) and its endogenous ligand cathelin-related antimicrobial peptide (cramp) promote dendritic cell maturation. J. Biol. Chem. 2014, 289, 17553–17563. [Google Scholar] [CrossRef] [PubMed]
- Bandholtz, L.; Ekman, G.J.; Vilhelmsson, M.; Buentke, E.; Agerberth, B.; Scheynius, A.; Gudmundsson, G.H. Antimicrobial peptide ll-37 internalized by immature human dendritic cells alters their phenotype. Scand. J. Immunol. 2006, 63, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Kin, N.W.; Chen, Y.; Stefanov, E.K.; Gallo, R.L.; Kearney, J.F. Cathelin-related antimicrobial peptide differentially regulates t- and b-cell function. Eur. J. Immunol. 2011, 41, 3006–3016. [Google Scholar] [CrossRef] [PubMed]
- Thomi, R.; Schlapbach, C.; Yawalkar, N.; Simon, D.; Yerly, D.; Hunger, R.E. Elevated levels of the antimicrobial peptide ll-37 in hidradenitis suppurativa are associated with a th1/th17 immune response. Exp. Dermatol. 2018, 27, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.J.; Jung, H.J.; Kim, M.J.; Roh, N.K.; Jung, J.W.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Serum levels of ll-37 and inflammatory cytokines in plaque and guttate psoriasis. Med. Inflamm. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Lande, R.; Botti, E.; Jandus, C.; Dojcinovic, D.; Fanelli, G.; Conrad, C.; Chamilos, G.; Feldmeyer, L.; Marinari, B.; Chon, S.; et al. The antimicrobial peptide ll37 is a t-cell autoantigen in psoriasis. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kim, Y.N.; Jang, Y.S. Cutting edge: Ll-37–mediated formyl peptide receptor-2 signaling in follicular dendritic cells contributes to b cell activation in peyer’s patch germinal centers. J. Immunol. 2017, 198, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Vissers, J.L.M.; Hartgers, F.C.; Lindhout, E.; Figdor, C.G.; Adema, G.J. Blc (cxcl13) is expressed by different dendritic cell subsets in vitro and in vivo. Eur. J. Immunol. 2001, 31, 1544–1549. [Google Scholar] [CrossRef]
- Piktel, E.; Niemirowicz, K.; Wnorowska, U.; Wątek, M.; Wollny, T.; Głuszek, K.; Góźdź, S.; Levental, I.; Bucki, R. The role of cathelicidin ll-37 in cancer development. Arch. Immunol. Ther. Exp. 2016, 64, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Luan, C.; Xie, Y.G.; Pu, Y.T.; Zhang, H.W.; Han, F.F.; Feng, J.; Wang, Y.Z. Recombinant expression of antimicrobial peptides using a novel self-cleaving aggregation tag in escherichia coli. Can. J. Microbiol. 2014, 60, 113–120. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Fu, A.Y.; Li, T.J. Expression and one-step purification of the antimicrobial peptide cathelicidin-bf using the intein system in bacillus subtilis. J. Ind. Microbiol. Biotechnol. 2015, 42, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.W.; Tian, S.X.; Gao, W.; Yang, N.; Qu, P.; Liu, D.; Jiao, J.; Wang, J.; Feng, X.J. Recombinant expression and biological characterization of the antimicrobial peptide fowlicidin-2 in pichia pastoris. Exp. Ther. Med. 2016, 12, 2324–2330. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.-J. Enhanced resistance to bacterial pathogen in transgenic tomato plants expressing cathelicidin antimicrobial peptide. Biotechnol. Bioprocess Eng. 2013, 18, 615–624. [Google Scholar] [CrossRef]
- Morassutti, C.; De Amicis, F.; Skerlavaj, B.; Zanetti, M.; Marchetti, S. Production of a recombinant antimicrobial peptide in transgenic plants using a modified vma intein expression system. FEBS Lett. 2002, 519, 141–146. [Google Scholar] [CrossRef]
- Shinnar, A.E.; Butler, K.L.; Park, H.J. Cathelicidin family of antimicrobial peptides: Proteolytic processing and protease resistance. Bioorg. Chem. 2003, 31, 425–436. [Google Scholar] [CrossRef]
- Sieprawska-Lupa, M.; Mydel, P.; Krawczyk, K.; Wójcik, K.; Puklo, M.; Lupa, B.; Suder, P.; Silberring, J.; Reed, M.; Pohl, J. Degradation of human antimicrobial peptide ll-37 by staphylococcus aureus-derived proteinases. Antimicrob. Agents Chemother. 2004, 48, 4673–4679. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.; Carvalho, I.F.; Montelaro, R.C.; Gomes, P.; Martins, M.C.L. Covalent immobilization of antimicrobial peptides (amps) onto biomaterial surfaces. Acta Biomater. 2011, 7, 1431–1440. [Google Scholar] [CrossRef] [PubMed]
- deGruyter, J.N.; Malins, L.R.; Baran, P.S. Residue-specific peptide modification: A chemist’s guide. Biochemistry 2017, 56, 3863–3873. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.L.; Suleman, L.; Vuotto, C.; Donelli, G. Healthcare-associated infections, medical devices and biofilms: Risk, tolerance and control. J. Med. Microbiol. 2015, 64, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Carretero, M.; Escámez, M.J.; García, M.; Duarte, B.; Holguín, A.; Retamosa, L.; Jorcano, J.L.; Río, M.D.; Larcher, F. In vitro and in vivo wound healing-promoting activities of human cathelicidin ll-37. J. Investig. Dermatol. 2008, 128, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.H.; Wu, W.K.K.; Tai, E.K.K.; Wong, H.P.S.; Lam, E.K.Y.; So, W.H.L.; Shin, V.Y.; Cho, C.H. The cationic host defense peptide rcramp promotes gastric ulcer healing in rats. J. Pharmacol. Exp. Ther. 2006, 318, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Koczulla, R.; Degenfeld, G.V.; Kupatt, C.; Krötz, F.; Zahler, S.; Gloe, T.; Issbrücker, K.; Unterberger, P.; Zaiou, M.; Lebherz, C.; et al. An angiogenic role for the human peptide antibiotic ll-37/hcap-18. J. Clin. Investig. 2003, 111, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Post, M.; Volk, R.; Gao, Y.; Li, M.; Metais, C.; Sato, K.; Tsai, J.; Aird, W.; Rosenberg, R.D.; et al. Pr39, a peptide regulator of angiogenesis. Nat. Med. 2000, 6, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Seth, A.; Wibowo, N.; Zhao, C.X.; Mitter, N.; Yu, C.; Middelberg, A.P.J. Nanoparticle vaccines. Vaccine 2014, 32, 327–337. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Harten, R.M.; Van Woudenbergh, E.; Van Dijk, A.; Haagsman, H.P. Cathelicidins: Immunomodulatory Antimicrobials. Vaccines 2018, 6, 63. https://doi.org/10.3390/vaccines6030063
Van Harten RM, Van Woudenbergh E, Van Dijk A, Haagsman HP. Cathelicidins: Immunomodulatory Antimicrobials. Vaccines. 2018; 6(3):63. https://doi.org/10.3390/vaccines6030063
Chicago/Turabian StyleVan Harten, Roel M., Esther Van Woudenbergh, Albert Van Dijk, and Henk P. Haagsman. 2018. "Cathelicidins: Immunomodulatory Antimicrobials" Vaccines 6, no. 3: 63. https://doi.org/10.3390/vaccines6030063
APA StyleVan Harten, R. M., Van Woudenbergh, E., Van Dijk, A., & Haagsman, H. P. (2018). Cathelicidins: Immunomodulatory Antimicrobials. Vaccines, 6(3), 63. https://doi.org/10.3390/vaccines6030063




