Relative Clinical and Cost Burden of Community-Acquired Pneumonia Hospitalizations in Older Adults in the United States—A Cross-Sectional Analysis
Abstract
1. Introduction
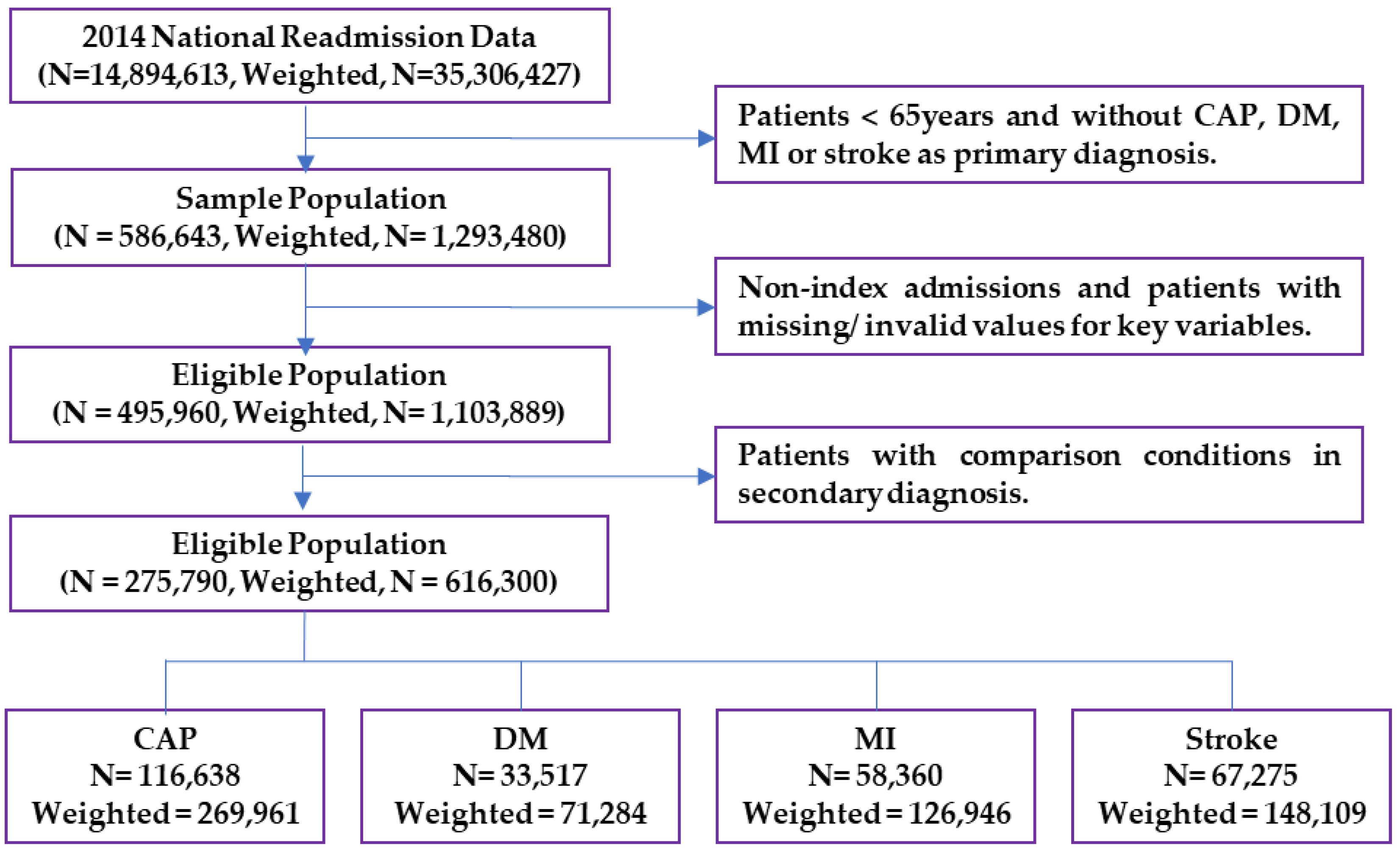
2. Materials and Methods
3. Results
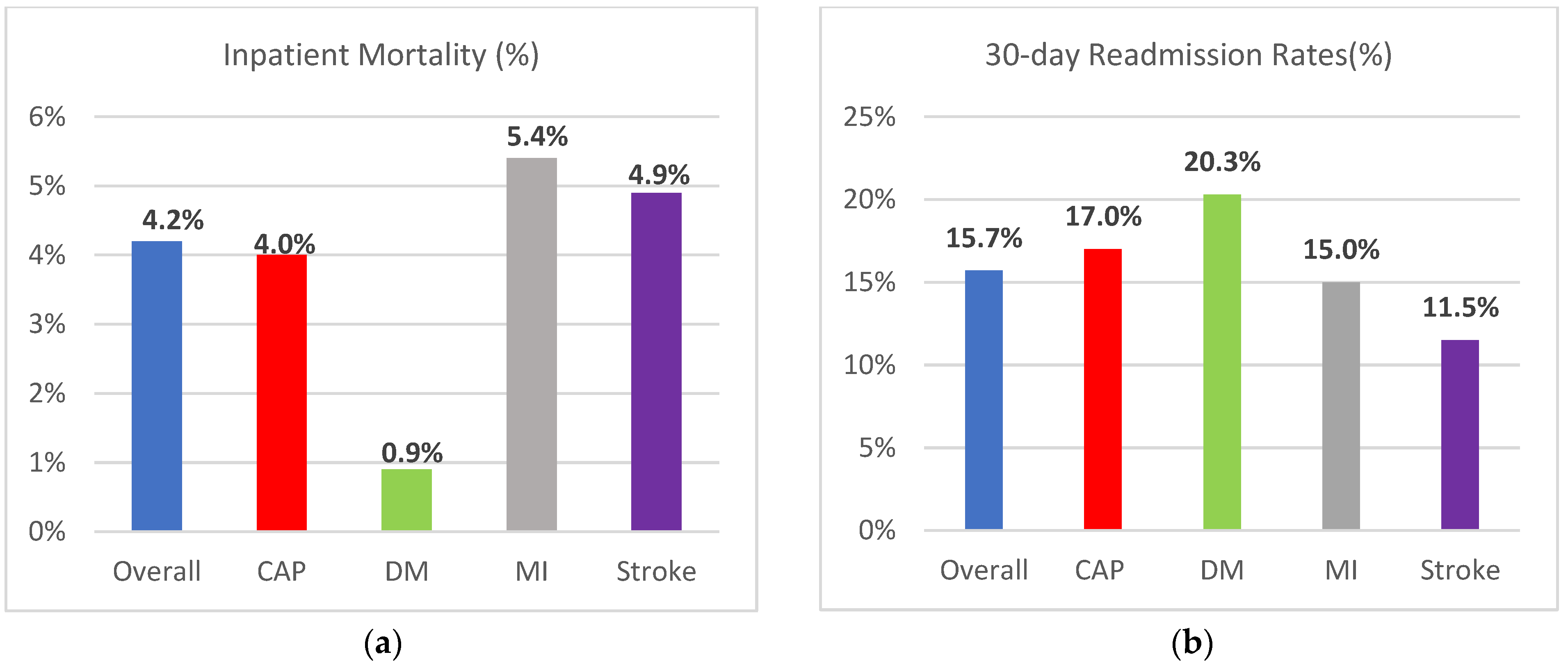
3.1. Inpatient Mortality
3.2. Length of Hospital Stay, 30-Day Readmission Rates, and Costs
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Disease State | ICD-9 Codes |
|---|---|
| Community-Acquired Pneumonia (CAP) | (1) 480.x–486.x, 487.0 Pneumonia as a principal diagnosis; OR (2) 518.8x Respiratory Failure or 038.xx Septicemia as a principal diagnosis with Pneumonia as a secondary diagnosis on the same claim line |
| Myocardial Infarction | 410.xx, 412.xx |
| Stroke | 433.x1, 434.x1, 436.xx |
| Diabetes Mellitus (with or without complications) | 250.0x–250.9x |
References
- 10 Leading Causes of Death by Age Group, United States—2015. Available online: https://www.cdc.gov/injury/images/lc-charts/leading_causes_of_death_age_group_2015_1050w740h.gif (accessed on 22 August 2017).
- Weycker, D.; Strutton, D.; Edelsberg, J.; Sato, R.; Jackson, A. Clinical and economic burden of pneumococcal disease in older US adults. Vaccine 2010, 28, 4955–4960. [Google Scholar] [CrossRef] [PubMed]
- Stupka, J.E.; Mortensen, E.M.; Anzueto, A.; Restrepo, M.I. Community-acquired pneumonia in elderly patients. Aging Halth 2009, 5, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Janssens, J.P.; Krause, K.H. Pneumonia in the very old. Lancet Infect. Dis. 2004, 4, 112–124. [Google Scholar] [CrossRef]
- Kaplan, V.; Angus, D.C.; Griffin, M.F.; Clermont, G.; Scott, W.R.; Linde-Zwirble, W.T. Hospitalized community-acquired pneumonia in the elderly: Age-and sex-related patterns of care and outcome in the United States. Am. J. Respir. Crit. Care Med. 2002, 165, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Musher, D.M.; Thorner, A.R. Community-acquired pneumonia. N. Engl. J. Med. 2014, 371, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Steel, H.C.; Cockeran, R.; Anderson, R.; Feldman, C. Overview of community-acquired pneumonia and the role of inflammatory mechanisms in the immunopathogenesis of severe pneumococcal disease. Mediators Inflamm. 2013. [Google Scholar] [CrossRef] [PubMed]
- Fine, M.J.; Smith, M.A.; Carson, C.A.; Mutha, S.S.; Sankey, S.S.; Weissfeld, L.A.; Kapoor, W.N. Prognosis and outcomes of patients with community-acquired pneumonia: A meta-analysis. JAMA 1996, 275, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.L.; Neuzil, K.M.; Thompson, W.W.; Shay, D.K.; Yu, O.; Hanson, C.A.; Jackson, L.A. The burden of community-acquired pneumonia in seniors: Results of a population-based study. Clin Infect Dis. 2004, 39, 1642–1650. [Google Scholar] [CrossRef] [PubMed]
- National Heart Lung and Blood Institute. Pneumonia: Causes, Symptoms, Risk Factors. Available online: www.nhlbi.nih.gov/health/health-topics/topics/pnu (accessed on 24 August 2017).
- Torres, A.; Blasi, F.; Dartois, N.; Akova, M. Which individuals are at increased risk of pneumococcal disease and why? impact of COPD, asthma, smoking, diabetes, and/or chronic heart disease on community-acquired pneumonia and invasive pneumococcal disease. Thorax 2015, 70, 984–989. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.; Exuzides, A.; Spangler, L.; O’Malley, C.; Colby, C.; Johnston, K.; Agodoa, I.; Baker, J.; Kagan, R. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin. Proc. 2015, 90, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, P.B.; Farooq, M.U.; Min, J. Population-based approaches for reducing stroke risk. Expert Rev. Cardiovasc. Ther. 2015, 13, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.D.; Harnett, J.; Chambers, R.; Sato, R. The relative burden of community-acquired pneumonia hospitalizations in older adults: A retrospective observational study in the United States. BMC Geriatr. 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- Number of Discharges from Short-Stay Hospitals, by First-Listed Diagnosis and Age: United States 2010. Available online: https://www.cdc.gov/nchs/data/nhds/3firstlisted/2010first3_numberage.pdf (accessed on 3 May 2018).
- File, T.M.; Marrie, T.J. Burden of community-acquired pneumonia in north american adults. Postgrad Med. J. 2010. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Rubin, J.; Dunning, S.; Li, S.; Sato, R. Clinical and economic burden of community-acquired pneumonia in the Medicare fee-for-service population. J. Am. Geriatr. Soc. 2012, 60, 2137–2143. [Google Scholar] [CrossRef] [PubMed]
- Welte, T.; Torres, A.; Nathwani, D. Clinical and economic burden of community-acquired pneumonia among adults in Europe. Thorax 2012, 67, 71–79. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.M.; Johnson, M.H.; Kagan, S.A.; Baer, S.L. Clinical and economic burden of community-acquired pneumonia in the Veterans Health Administration, 2011: A retrospective cohort study. Infection 2015, 43, 671–680. [Google Scholar] [CrossRef] [PubMed]
- HCUP National Readmissions Database. Available online: https://hcup-us.ahrq.gov/nrdoverview.jsp (accessed on 15 July 2018).
- Barber, C.; Lacaille, D.; Fortin, P.R. Systematic review of validation studies of the use of administrative data to identify serious infections. Arthritis Care Res. 2013, 65, 1343–1357. [Google Scholar] [CrossRef] [PubMed]
- Whittle, J.; Fine, M.J.; Joyce, D.Z.; Lave, J.R.; Young, W.W.; Hough, L.J.; Kapoor, W.N. Community-acquired pneumonia: Can it be defined with claims data? Am. J. Med. Qual. 1997, 12, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Whitney, C.; Pilishvili, T.; Farley, M.; Schaffner, W.; Craig, A.S.; Lynfield, R.; Nyquist, A.C.; Gershman, K.A.; Vazquez, M.; Bennett, N.M.; et al. Effectiveness of seven-valent pneumococcal conjugate vaccine against invasive pneumococcal disease: A matched case-control study. Lancet 2006, 368, 1495–1502. [Google Scholar] [CrossRef]
- Fine, M.; Smith, M.; Carson, C.; Meffe, F.; Sankey, S.S.; Weissfeld, L.A.; Detsky, A.S.; Kapoor, W.N. Efficacy of pneumococcal vaccination in adults. A meta-analysis of randomized controlled trials. Arch. Intern. Med. 1994, 154, 2666–2677. [Google Scholar] [CrossRef] [PubMed]
- Immunization and Infectious Diseases. Available online: https://www.healthypeople.gov/2020/topics-objectives/topic/immunization-and-infectious-diseases (accessed on 15 February 2017).
- Tomczyk, S.; Bennett, N.; Stoecker, C.; Gierke, R.; Moore, M.R.; Whitney, C.G.; Hadler, S.; Pilishvili, T. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among adults aged ≥65 years: Recommendations of the advisory committee on immunization practices (ACIP). MMWR Morb. Mortal. Wkly. Rep. 2014, 63, 822–825. [Google Scholar] [PubMed]
- Pneumococcal Vaccination: Information for Healthcare Professionals. Available online: https://www.cdc.gov/vaccines/vpd/pneumo/hcp/index.html (accessed on 18 February 2017).
- Kim, D.K.; Riley, L.E.; Harriman, K.H.; Hunter, P.; Bridges, C.B. Recommended immunization schedule for adults aged 19 years or older, United States, 2017. Ann. Intern. Med. 2017, 166, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Grijalva, C.G.; Zhu, Y.; Williams, D.J.; Self, W.H.; Ampofo, K.; Pavia, A.T.; Stockman, C.R.; McCullers, J.; Arnold, S.R.; Wunderink, R.G.; et al. Association between hospitalization with community-acquired laboratory-confirmed influenza pneumonia and prior receipt of influenza vaccination. JAMA 2015, 314, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Mandell, L.; Wunderink, R.; Anzueto, A.; Bartlett, J.; Campbell, G.; Dean, N.; Dowell, S.; File, T.; Musher, D.; Niederman, M.; et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin. Infect. Dis. 2007, 44, S27–S72. [Google Scholar] [CrossRef] [PubMed]

| Individual Characteristics | Weighted | CAP | DM | MI | Stroke |
|---|---|---|---|---|---|
| N = 616,300 | N = 269,961 | N = 71,284 | N = 126,946 | N = 148,109 | |
| 100% | 3.80% | 11.60% | 20.60% | 24.00% | |
| Demographics | N (%) | N (%) | N (%) | N (%) | N (%) |
| Age in years (Mean ± SD) | 78.8 ± 8.1 | 79.7 ± 8.0 | 75.4 ± 7.5 | 77.6 ± 8.2 | 78.5 ± 7.9 |
| Female | 334,253 (54.2) | 151,117 (56.0) | 37, 047 (52.0) | 58, 831 (46.3) | 87, 258 (58.9) |
| Patient Location: NCHS Urban-Rural Code (PL_NCHS) 1 | |||||
| Central counties ≥1 million | 125,386 (20.4) | 47,555 (17.6) | 21,198 (29.7) | 25,049 (19.7) | 31,584 (21.3) |
| Fringe counties of ≥1 million | 154,046 (25.0) | 66,274 (24.5) | 17,626 (24.7) | 31,199 (24.6) | 38,948 (26.3) |
| Metro areas of 250,000–999,999 | 132,376 (21.5) | 56,686 (21.0) | 14,027 (19.7) | 28,527 (22.5) | 33,136 (22.4) |
| Metro areas of 50,000–249,999 | 65,328 (10.6) | 29,151 (10.8) | 6067 (8.5) | 14,117 (11.1) | 15,993 (10.8) |
| Micropolitan counties | 70,712 (11.5) | 34,748 (12.9) | 6511 (9.1) | 14,663 (11.6) | 14,789 (10.0) |
| Not metropolitan/Micropolitan | 67,119 (10.9) | 35,033 (13.0) | 5708 (8.0) | 13,057 (10.3) | 13,320 (9.0) |
| Median Household Income National Quartile for Patient ZIP Code | |||||
| 0–25th percentile | 163,896 (27.0) | 71,832 (26.6) | 24,673 (34.6) | 31,360 (24.7) | 36,030 (24.3) |
| 26–50th percentile (median) | 173,612 (28.6) | 78,077 (28.9) | 18,734 (26.3) | 36,538 (28.8) | 40,263 (27.2) |
| 51–75th percentile | 143,008 (23.6) | 62,303 (23.1) | 14,480 (20.3) | 30,627 (24.1) | 35,598 (24.0) |
| 76th–100th percentile | 126,375 (20.8) | 53,566 (19.8) | 12,329 (17.3) | 26,408 (20.8) | 34,072 (23.0) |
| Primary Expected Payer | |||||
| Medicare | 564,041 (91.6) | 249,869 (92.6) | 64,028 (89.8) | 114,597 (90.3) | 135,547 (91.5) |
| Medicaid | 7393 (1.2) | 2838 (1.1) | 1741 (2.4) | 1208 (1.0) | 1605 (1.1) |
| Private insurance | 34,370 (5.6) | 13,112 (4.9) | 4142 (5.8) | 8789 (6.9) | 8327 (5.6) |
| Self-pay | 2309 (0.4) | 794 (0.3) | 370 (0.5) | 493 (0.4) | 652 (0.4) |
| No charge | 134 (0.0) | 43 (0.0) | 22 (0.0) | 37 (0.0) | 32 (0.0) |
| Other | 7491 (1.2) | 3034 (1.1) | 927 (1.3) | 1741 (1.4) | 1790 (1.2) |
| Admission Day: Weekday | 460,414 (74.7) | 201,862 (74.8) | 55,676 (78.1) | 93,639 (73.8) | 109,237 (73.8) |
| Admission Type: Non-elective | 581,468 (94.5) | 252,374 (93.5) | 64,903 (91.0) | 121,077 (95.4) | 143,114 (96.6) |
| Comorbidities | |||||
| AIDS | 134 (0) | <11 (0) | 46 (0) | 38 (0) | 48 (0) |
| Alcohol abuse | 13,700 (2.2) | 4741 (1.8) | 1560(2.2) | 2883 (2.3) | 4516 (3.0) |
| Deficiency anemias | 128,379 (20.8) | 70,587 (26.1) | 18,515 (26.0) | 20,833 (16.4) | 18,443 (12.5) |
| Congestive heart failure | 105,396 (17.1) | 71,244 (26.4) | 13,556 (19.0) | 622 (0.5) | 19,974 (13.5) |
| Chronic pulmonary disease | 194,822 (31.6) | 132,651 (49.1) | 12,670 (017.8) | 26,654 (21.0) | 22,847 (15.4) |
| Depression | 70,617 (11.5) | 36,623 (13.6) | 7903 (11.1) | 9872 (7.8) | 16,219 (11.0) |
| Hypertension | 452,538 (73.4) | 180,662 (66.9) | 56,654 (79.5) | 92,833 (73.1) | 122,389 (82.6) |
| Hypothyroidism | 111,022 (18.0) | 52,655 (19.5) | 11,557 (16.2) | 19,672 (15.5) | 27,139 (18.3) |
| Liver disease | 8409 (1.4) | 3932 (1.5) | 2041 (2.9) | 1342 (1.1) | 1094 (0.1) |
| Fluid and electrolyte disorders | 199,800 (32.4) | 105,284 (39.0) | 30,861 (43.3) | 29,617 (2.3) | 34,039 (23.0) |
| Other neurological disorders | 57,590 (9.3) | 38,231 (14.2) | 8914 (12.5) | 9989 (7.9) | 456 (0.3) |
| Obesity | 42,866 (7.0) | 14,900 (5.5) | 10,135 (14.2) | 10,061 (7.9) | 7770 (5.2) |
| Peripheral vascular disorders | 59,707 (9.7) | 20,041 (7.4) | 11,567 (16.2) | 15,551 (12.3) | 12,548 (8.5) |
| Pulmonary circulation disorders | 28,529 (4.6) | 19,370 (7.2) | 2165 (3.0) | 142 (0.1) | 19,370 (13.1) |
| Renal failure | 116,945 (19.0) | 50,083 (18.6) | 22,256 (31.2) | 25,040 (19.7) | 19,566 (13.2) |
| Solid tumor without metastasis | 20,093 (3.3) | 12,722 (4.7) | 1775 (2.5) | 2444 (1.2) | 3151 (2.1) |
| Metastatic cancer | 15,903 (2.6) | 10,568 (3.9) | 1331 (1.9) | 1522 (1.2) | 2483 (1.7) |
| No. of chronic conditions (Mean ± SD) | 6.6 ± 2.9 | 6.0 ± 2.9 | 6.9 ± 2.9 | 7.1 ± 2.8 | 7.0 ± 2.7 |
| Median | 6 | 6 | 7 | 7 | 7 |
| Hospital Characteristics | |||||
| Bed size of hospital: Large | 319,657 (51.9) | 125,093 (43.6) | 37,579 (52.7) | 74,603 (58.8) | 82,382 (55.6) |
| Control/Ownership of Hospital: | |||||
| Government, nonfederal | 73,390 (11.9) | 35,895 (13.3) | 8533 (12.0) | 11,978 (9.4) | 16,985 (11.5) |
| Private, not-profit | 448,313 (72.7) | 192,915 (71.5) | 49,736 (69.8) | 94,960 (74.8) | 110,702 (74.7) |
| Private, invest-own | 94,597 (15.3) | 41,152 (15.2) | 13,015 (18.3) | 20,009 (15.8) | 20,421 (13.8) |
| Hospitalization | Total Sample | Died during Hospitalization | Unadjusted RR | Adjusted RR |
|---|---|---|---|---|
| N (%) | N (%) | |||
| CAP | 269,961 (43.8) | 10,807 (4.0) | Ref | Ref |
| DM | 71,284 (11.6) | 606 (0.9) | 0.20 (0.18–0.23) | 0.37 (0.29–0.46) |
| MI | 126,946 (20.6) | 6841 (5.4) | 1.33 (1.27–1.40) | 1.67 (1.50–1.85) |
| Stroke | 148,109 (24.0) | 7309 (4.9) | 1.19 (1.14–1.26) | 1.67 (1.51–1.83) |
| Total | 616,300 (100%) | 25,563 (4.2) | - | - |
| Factors | Risk Ratio | 95% CI | p-Value | |
|---|---|---|---|---|
| Age in Years at Admission | ||||
| 75–84 vs. 65–74 | 1.35 | 1.23 | 1.49 | <0.0001 |
| 85–89 vs. 65–74 | 2.07 | 1.86 | 2.30 | <0.0001 |
| 90+ vs. 65–74 | 2.49 | 2.24 | 2.78 | <0.0001 |
| Sex: Male vs. Female | 0.95 | 0.89 | 1.02 | 0.17 |
| Patient Location: NCHS Urban–Rural Code | ||||
| Central counties of metro areas of ≥1 million population (Ref) | - | - | - | - |
| Fringe counties of metro areas of ≥1 million population | 0.98 | 0.88 | 1.09 | 0.69 |
| Counties in metro areas of 250,000–999,999 population | 1.13 | 1.02 | 1.25 | 0.02 |
| Counties in metro areas of 50,000–249,999 population | 1.39 | 1.23 | 1.58 | <0.0001 |
| Micropolitan counties | 1.05 | 0.88 | 1.24 | 0.62 |
| Not metropolitan or micropolitan counties | 1.44 | 1.22 | 1.68 | <0.0001 |
| Median Household Income National Quartile for Patient’s ZIP Code | ||||
| 0–25th percentile (Ref) | - | - | - | - |
| 26th to 50th percentile (median) | 0.96 | 0.88 | 1.06 | 0.45 |
| 51st to 75th percentile | 1.02 | 0.92 | 1.13 | 0.69 |
| 76th to 100th percentile | 1.09 | 0.98 | 1.21 | 0.13 |
| Primary Expected Payer | ||||
| Medicaid vs. Medicare | 1.08 | 0.76 | 1.52 | 0.67 |
| Private insurance vs. Medicare | 1.51 | 1.32 | 1.74 | <0.0001 |
| Self-pay vs. Medicare | 1.29 | 0.81 | 2.05 | 0.29 |
| No charge vs. Medicare | 0.00 | 0.00 | N/A | 1.00 |
| Others vs. Medicare | 3.12 | 2.60 | 3.76 | <0.0001 |
| Admission Day: Admitted Weekday vs. Weekend | 1.03 | 0.96 | 1.12 | 0.39 |
| Admission Type: Non-elective vs. Elective admission | 1.11 | 0.90 | 1.36 | 0.33 |
| All Patient Refined DRG: Severity of Illness Subclass | ||||
| Minor loss of function (Ref) | - | - | - | - |
| Moderate loss of function | 2.02 | 1.49 | 2.73 | <0.0001 |
| Major loss of function | 9.57 | 7.14 | 12.84 | <0.0001 |
| Extreme loss of function | 48.57 | 36.15 | 65.26 | <0.0001 |
| Comorbidities | ||||
| AIDS | 0.00 | 0.00 | N/A | 1.00 |
| Alcohol abuse | 1.24 | 0.99 | 1.55 | 0.06 |
| Deficiency anemias | 0.87 | 0.80 | 0.95 | 0.00 |
| Rheumatoid arthritis/collagen vascular diseases | 1.07 | 0.89 | 1.29 | 0.45 |
| Chronic blood loss anemia | 0.73 | 0.49 | 1.10 | 0.14 |
| Congestive heart failure | 1.20 | 1.10 | 1.31 | <0.0001 |
| Chronic pulmonary disease | 0.93 | 0.86 | 1.01 | 0.08 |
| Coagulopathy | 1.18 | 1.05 | 1.32 | 0.01 |
| Depression | 0.83 | 0.73 | 0.94 | 0.00 |
| Drug abuse | 0.57 | 0.33 | 0.96 | 0.04 |
| Hypertension (combine uncomplicated and complicated) | 0.88 | 0.81 | 0.95 | 0.00 |
| Hypothyroidism | 0.89 | 0.81 | 0.98 | 0.02 |
| Liver disease | 1.21 | 0.94 | 1.56 | 0.14 |
| Lymphoma | 0.77 | 0.58 | 1.02 | 0.07 |
| Fluid and electrolyte disorders | 1.11 | 1.03 | 1.19 | 0.00 |
| Other neurological disorders | 1.16 | 1.04 | 1.30 | 0.01 |
| Obesity | 0.77 | 0.66 | 0.91 | 0.00 |
| Paralysis | 1.21 | 1.04 | 1.41 | 0.01 |
| Peripheral vascular disorders | 0.97 | 0.87 | 1.09 | 0.65 |
| Psychoses | 1.26 | 1.01 | 1.58 | 0.04 |
| Pulmonary circulation disorders | 0.94 | 0.82 | 1.08 | 0.41 |
| Renal failure | 0.96 | 0.88 | 1.05 | 0.34 |
| Solid tumor without metastasis | 1.58 | 1.35 | 1.85 | <0.0001 |
| Metastatic cancer | 2.30 | 2.00 | 2.64 | <0.0001 |
| Valvular disease | 0.77 | 0.69 | 0.86 | <0.0001 |
| Weight loss | 0.74 | 0.68 | 0.80 | <0.0001 |
| Number of Chronic Conditions | ||||
| 1–4 (Ref) | - | - | - | - |
| 5–6 | 0.90 | 0.81 | 1.00 | 0.06 |
| 7–8 | 0.84 | 0.75 | 0.95 | 0.00 |
| >8 | 0.80 | 0.70 | 0.92 | 0.00 |
| Hospital Characteristics | ||||
| Bed size of hospital: Medium vs. Small | 1.06 | 0.94 | 1.18 | 0.35 |
| Large vs. Small | 1.00 | 0.90 | 1.11 | 0.99 |
| Hospital urban–rural designation | ||||
| Large metropolitan areas with at least 1 million residents (Ref) | - | - | - | - |
| Small metropolitan areas with less than 1 million residents | 1.32 | 1.10 | 1.58 | 0.0025 |
| Micropolitan areas | 0.67 | 0.51 | 0.88 | 0.0044 |
| Not metropolitan or micropolitan (non-urban residual) | 1.00 | 1.00 | 1.00 | N/A |
| Teaching status of urban hospitals: | ||||
| Metropolitan teaching vs. metropolitan non-teaching | 0.91 | 0.84 | 0.99 | 0.02 |
| Control/ownership of hospital: | ||||
| Private, not-profit vs. government, nonfederal | 1.01 | 0.92 | 1.12 | 0.80 |
| Private, invest-own vs. government, nonfederal | 1.14 | 1.00 | 1.30 | 0.04 |
| Hospitalization Metric | Overall Sample | CAP | DM | MI | Stroke | p-Value |
|---|---|---|---|---|---|---|
| LOS (days) | 5.5 ± 4.7 | 5.2 ± 3.8 | 4.6 ± 3.8 | 4.3 ± 3.6 | 5.6 ± 5.5 | - |
| LOS (Median) | 4 | 4 | 3 | 3 | 4 | <0.0001 |
| Total charges | $51,612 ± 52,478 | $35,480 ± 36,149 | $35,341 ± 38,580 | $72,077 ± 60,105 | $46,785 ± 44,219 | - |
| Total charges (Median) | $33,637 | $24,309 | $23,222 | $56,016 | $32,458 | <0.0001 |
| Total Cost | $13,425 ± 12,366 | $9686 ± 8340 | $9057 ± 9246 | $18,148 ± 13,088 | $12,188 ± 10,283 | - |
| Total Cost (Median) | $9317 | $7282 | $6217 | $14,802 | $8772 | <0.0001 |
| Total summed costs (Millions) | $3312 | $1130 | $304 | $1059 | $819 | <0.0001 |
| 30-day readmission rates | 15.7% | 17.0% | 20.3% | 15.0% | 11.5% | <0.0001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olasupo, O.; Xiao, H.; Brown, J.D. Relative Clinical and Cost Burden of Community-Acquired Pneumonia Hospitalizations in Older Adults in the United States—A Cross-Sectional Analysis. Vaccines 2018, 6, 59. https://doi.org/10.3390/vaccines6030059
Olasupo O, Xiao H, Brown JD. Relative Clinical and Cost Burden of Community-Acquired Pneumonia Hospitalizations in Older Adults in the United States—A Cross-Sectional Analysis. Vaccines. 2018; 6(3):59. https://doi.org/10.3390/vaccines6030059
Chicago/Turabian StyleOlasupo, Omotola, Hong Xiao, and Joshua D. Brown. 2018. "Relative Clinical and Cost Burden of Community-Acquired Pneumonia Hospitalizations in Older Adults in the United States—A Cross-Sectional Analysis" Vaccines 6, no. 3: 59. https://doi.org/10.3390/vaccines6030059
APA StyleOlasupo, O., Xiao, H., & Brown, J. D. (2018). Relative Clinical and Cost Burden of Community-Acquired Pneumonia Hospitalizations in Older Adults in the United States—A Cross-Sectional Analysis. Vaccines, 6(3), 59. https://doi.org/10.3390/vaccines6030059






