A Pentavalent HIV-1 Subtype C Vaccine Containing Computationally Selected gp120 Strains Improves the Breadth of V1V2 Region Responses
Abstract
1. Introduction
2. Materials and Methods
2.1. Env Subunit Proteins for Vaccination
2.2. Immunization of Guinea Pigs and Cynomolgus Macaques
2.3. HIV-1 Specific Binding Antibody Assay
2.4. Peptide Microarray Linear Epitope Mapping
2.5. Monoclonal Antibody Competition ELISAs
2.6. Magnitude–Breadth Score Calculation
2.7. Statistical Analyses
3. Results
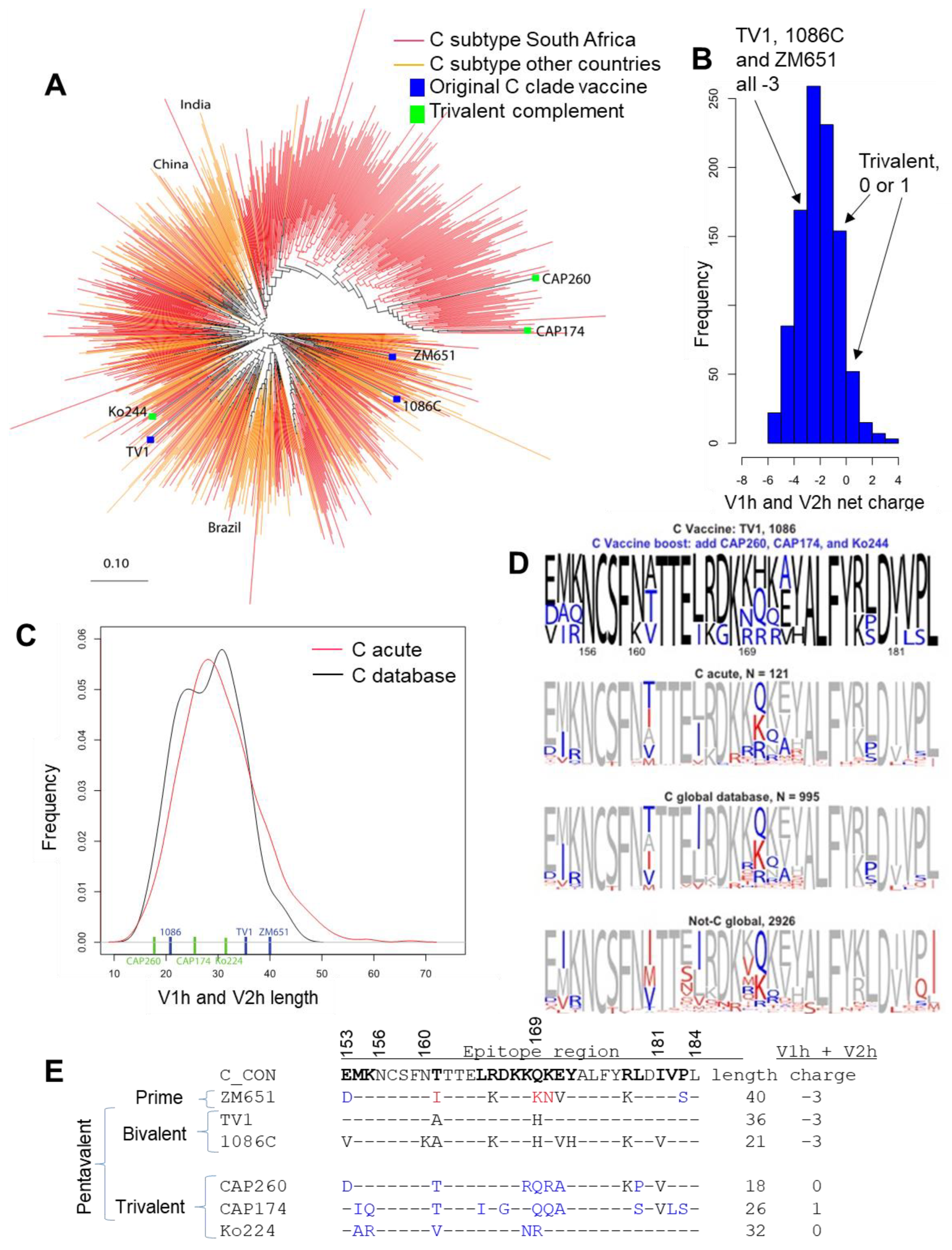
3.1. Selection of Novel Subtype C Env for Complimentary V1V2 Coverage
3.2. Pentavalent C Immunogenicity Study in Guinea Pigs
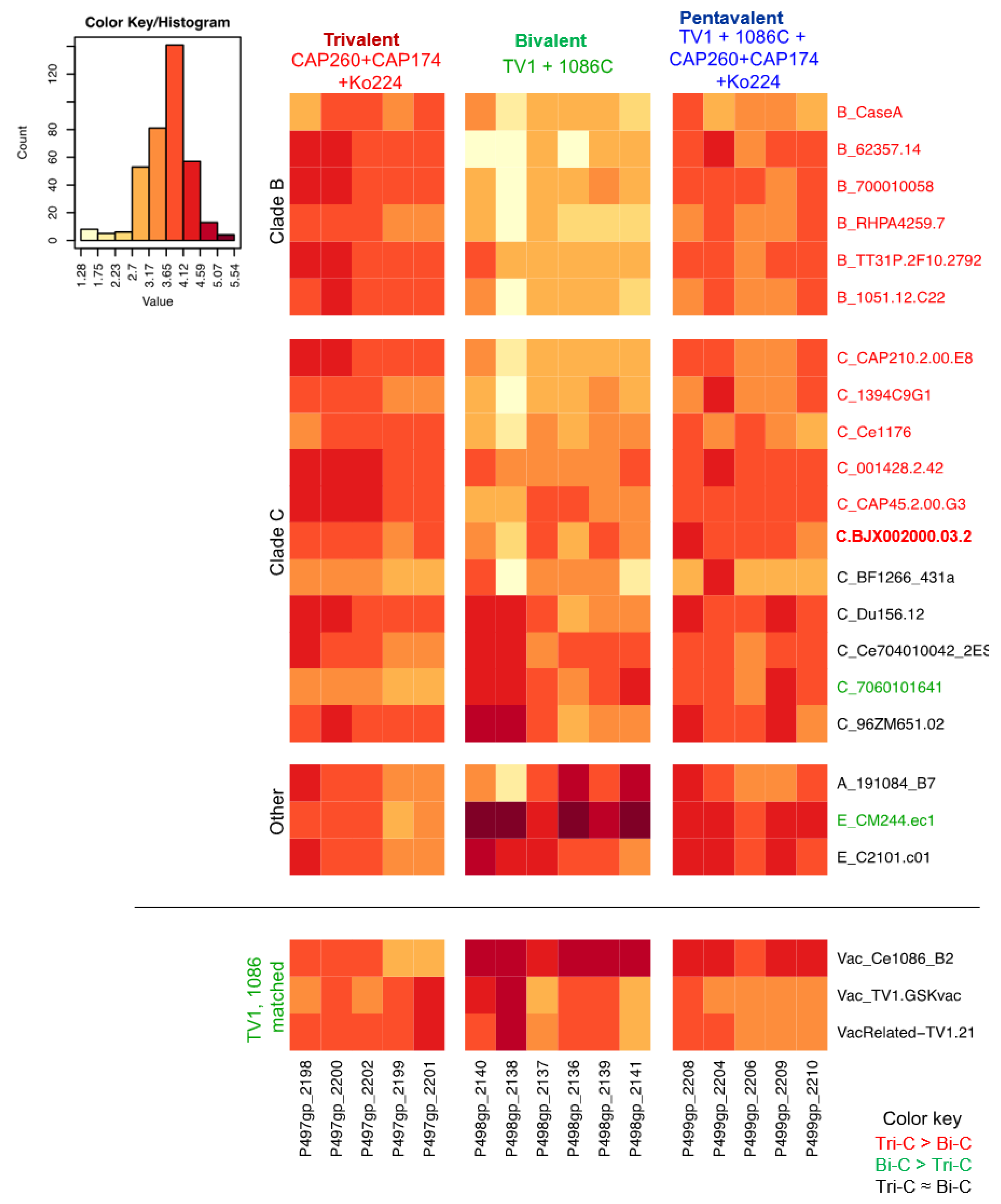
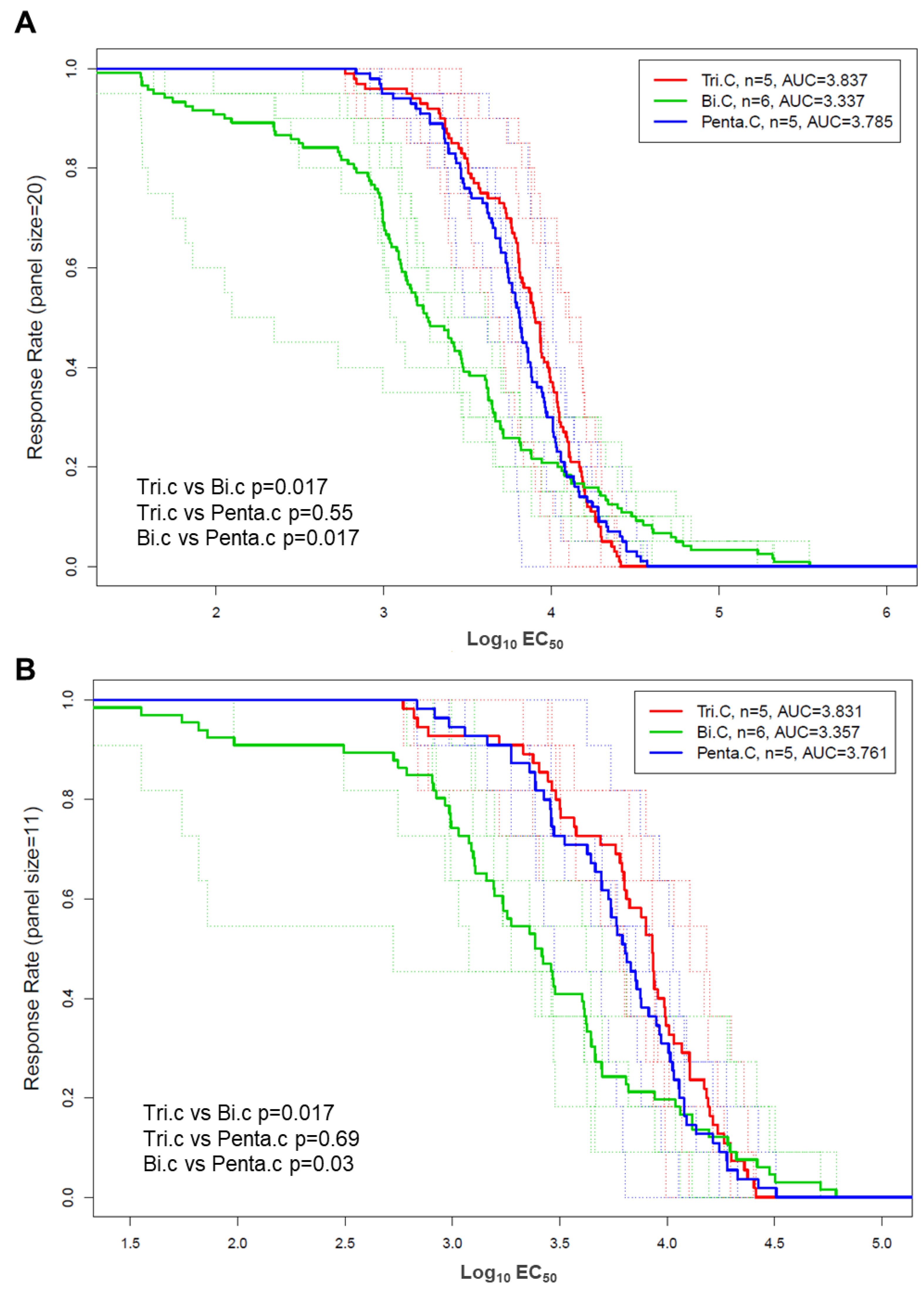
3.3. Differences Between Trivalent and Bivalent Groups for Magnitude of V1V2 Response
3.4. V1V2 Responses in Pentavalent Group
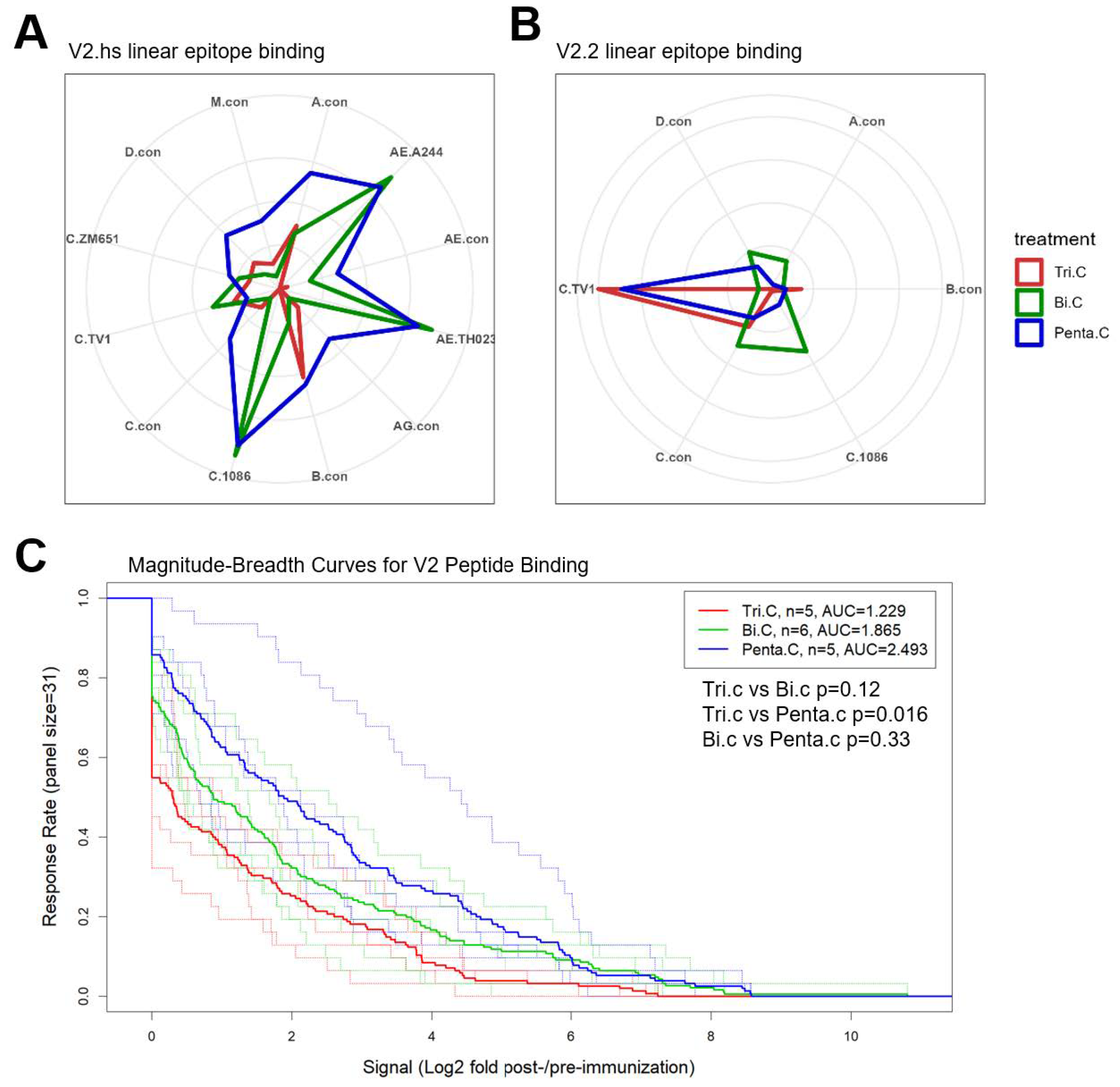
3.5. Improved Breadth of V1V2-IgG Binding Response in Trivalent and Pentavalent Groups Compared to Bivalent Group
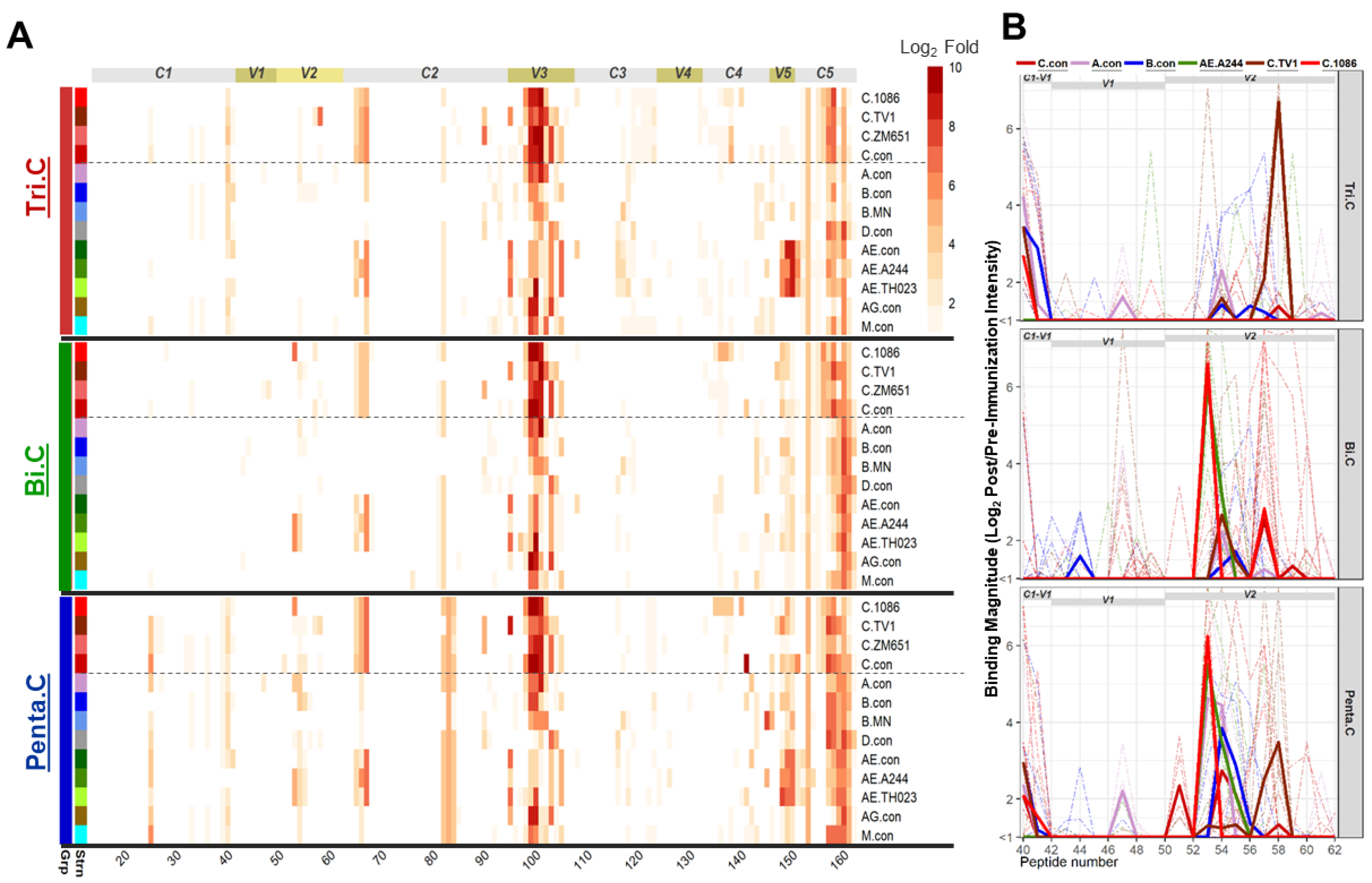
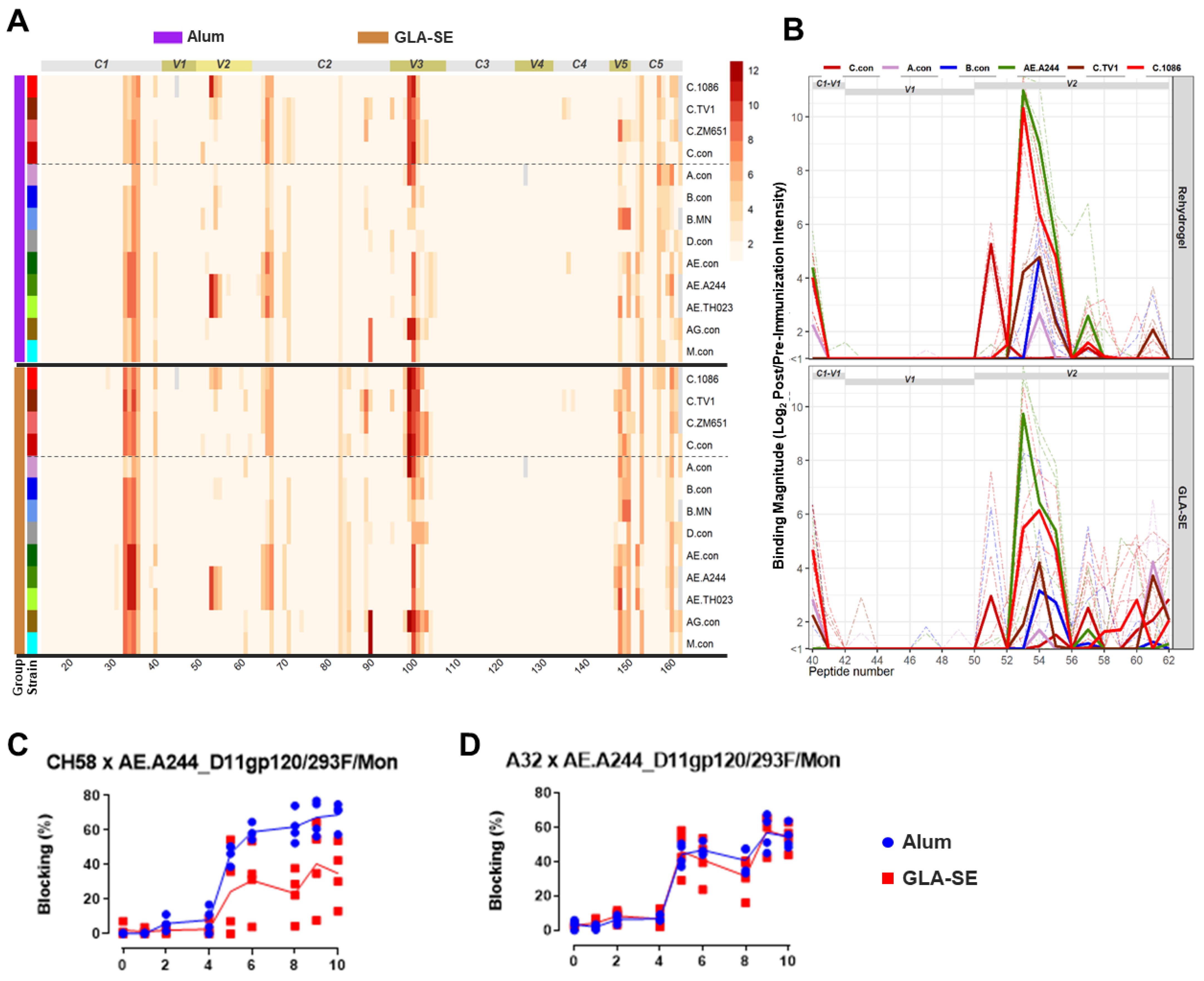
3.6. Binding Responses Against V1V2 and V2 Tags and Env Proteins
3.7. Pentavalent Group Targeted Both V2 Hotspot and V2.2 Linear Epitopes
3.8. Different Strain and Epitope Coverage by Bivalent and Trivalent Immunizations
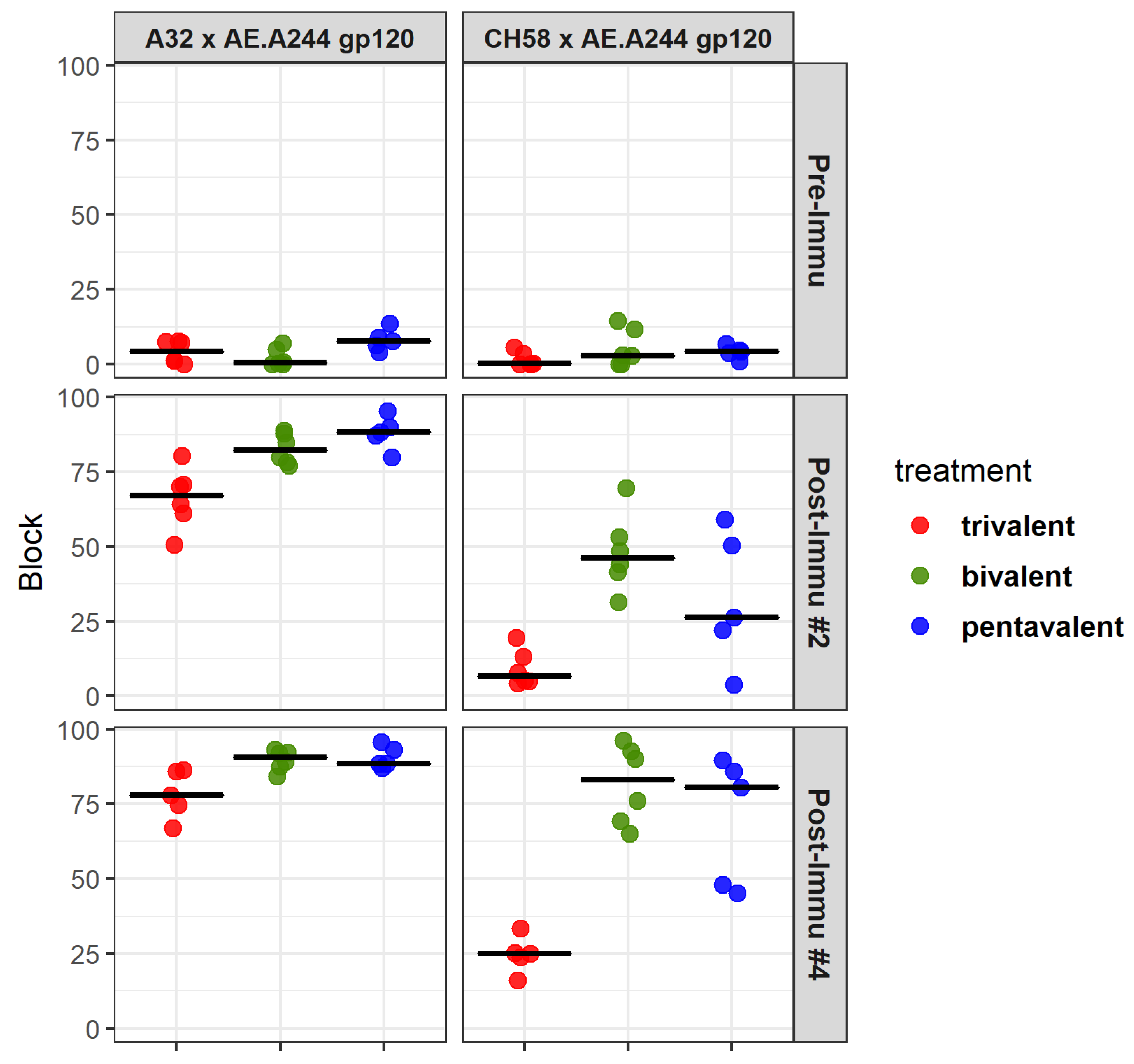
3.9. Development of Antibody Specificities That Target Binding Sites of Known Antibodies with Virus Inhibitory Functions
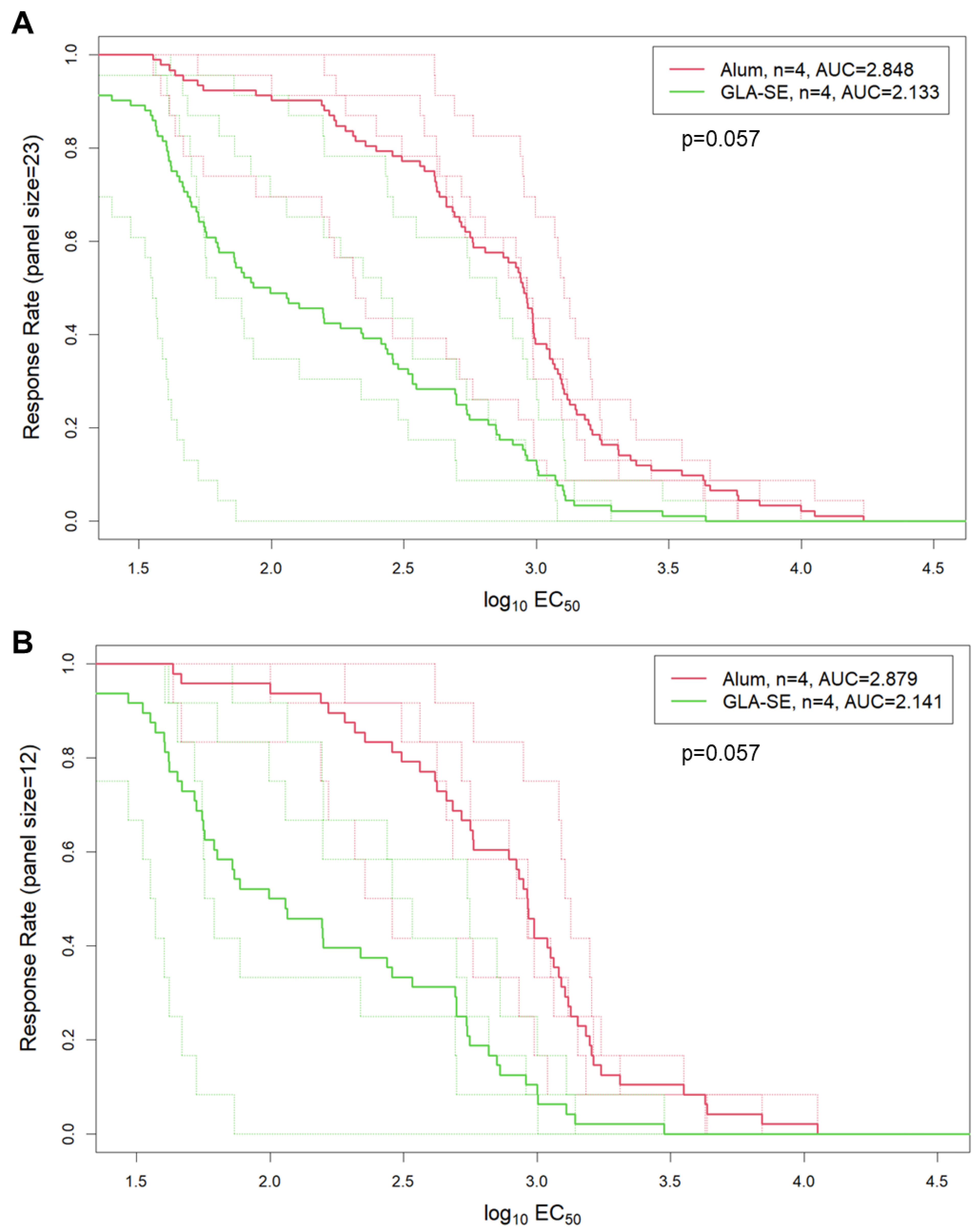
3.10. Pentavalent Subtype E/C Vaccine in Non-Human Primates
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zolla-Pazner, S.; deCamp, A.; Gilbert, P.B.; Williams, C.; Yates, N.L.; Williams, W.T.; Howington, R.; Fong, Y.; Morris, D.E.; Soderberg, K.A.; et al. Vaccine-induced IgG antibodies to V1V2 regions of multiple HIV-1 subtypes correlate with decreased risk of HIV-1 infection. PLoS ONE 2014, 9, e87572. [Google Scholar] [CrossRef] [PubMed]
- Haynes, B.F.; Gilbert, P.B.; McElrath, M.J.; Zolla-Pazner, S.; Tomaras, G.D.; Alam, S.M.; Evans, D.T.; Montefiori, D.C.; Karnasuta, C.; Sutthent, R.; et al. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N. Engl. J. Med. 2012, 366, 1275–1286. [Google Scholar] [CrossRef] [PubMed]
- Yates, N.L.; Liao, H.X.; Fong, Y.; deCamp, A.; Vandergrift, N.A.; Williams, W.T.; Alam, S.M.; Ferrari, G.; Yang, Z.Y.; Seaton, K.E.; et al. Vaccine-induced Env V1-V2 IgG3 correlates with lower HIV-1 infection risk and declines soon after vaccination. Sci. Transl. Med. 2014, 6, 228ra39. [Google Scholar] [CrossRef] [PubMed]
- Rolland, M.; Edlefsen, P.T.; Larsen, B.B.; Tovanabutra, S.; Sanders-Buell, E.; Hertz, T.; deCamp, A.C.; Carrico, C.; Menis, S.; Magaret, C.A.; et al. Increased HIV-1 vaccine efficacy against viruses with genetic signatures in Env V2. Nature 2012, 490, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Hessell, A.J.; Powell, R.; Jiang, X.; Luo, C.; Weiss, S.; Dussupt, V.; Itri, V.; Fox, A.; Shapiro, M.B.; Pandey, S.; et al. Multimeric Epitope-Scaffold HIV Vaccines Target V1V2 and Differentially Tune Polyfunctional Antibody Responses. Cell Rep. 2019, 28, 877–895.e6. [Google Scholar] [CrossRef] [PubMed]
- Roederer, M.; Keele, B.F.; Schmidt, S.D.; Mason, R.D.; Welles, H.C.; Fischer, W.; Labranche, C.; Foulds, K.E.; Louder, M.K.; Yang, Z.Y.; et al. Immunological and virological mechanisms of vaccine-mediated protection against SIV and HIV. Nature 2014, 505, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Zolla-Pazner, S.; Alvarez, R.; Kong, X.P.; Weiss, S. Vaccine-induced V1V2-specific antibodies control and or protect against infection with HIV, SIV and SHIV. Curr. Opin. HIV AIDS 2019, 14, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Karasavvas, N.; Billings, E.; Rao, M.; Williams, C.; Zolla-Pazner, S.; Bailer, R.T.; Koup, R.A.; Madnote, S.; Arworn, D.; Shen, X.; et al. The Thai Phase III HIV Type 1 Vaccine trial (RV144) regimen induces antibodies that target conserved regions within the V2 loop of gp120. AIDS Res. Hum. Retroviruses 2012, 28, 1444–1457. [Google Scholar] [CrossRef] [PubMed]
- Gottardo, R.; Bailer, R.T.; Korber, B.T.; Gnanakaran, S.; Phillips, J.; Shen, X.; Tomaras, G.D.; Turk, E.; Imholte, G.; Eckler, L.; et al. Plasma IgG to linear epitopes in the V2 and V3 regions of HIV-1 gp120 correlate with a reduced risk of infection in the RV144 vaccine efficacy trial. PLoS ONE 2013, 8, e75665. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.X.; Bonsignori, M.; Alam, S.M.; McLellan, J.S.; Tomaras, G.D.; Moody, M.A.; Kozink, D.M.; Hwang, K.K.; Chen, X.; Tsao, C.Y.; et al. Vaccine induction of antibodies against a structurally heterogeneous site of immune pressure within HIV-1 envelope protein variable regions 1 and 2. Immunity 2013, 38, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Tay, M.Z.; Liu, P.; Williams, L.D.; McRaven, M.D.; Sawant, S.; Gurley, T.C.; Xu, T.T.; Dennison, S.M.; Liao, H.X.; Chenine, A.L.; et al. Antibody-Mediated Internalization of Infectious HIV-1 Virions Differs among Antibody Isotypes and Subclasses. PLoS Pathog. 2016, 12, e1005817. [Google Scholar] [CrossRef] [PubMed]
- Musich, T.; Li, L.; Liu, L.; Zolla-Pazner, S.; Robert-Guroff, M.; Gorny, M.K. Monoclonal Antibodies Specific for the V2, V3, CD4-Binding Site, and gp41 of HIV-1 Mediate Phagocytosis in a Dose-Dependent Manner. J. Virol. 2017, 91, e02325-16. [Google Scholar] [CrossRef] [PubMed]
- UNAIDS. 2023 Data. Available online: https://www.unaids.org (accessed on 12 October 2024).
- Russell, N.D.; Marovich, M.A. Pox-Protein Public Private Partnership program and upcoming HIV vaccine efficacy trials. Curr. Opin. HIV AIDS 2016, 11, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Korber, B.; Muldoon, M.; Theiler, J.; Gao, F.; Gupta, R.; Lapedes, A.; Hahn, B.H.; Wolinsky, S.; Bhattacharya, T. Timing the ancestor of the HIV-1 pandemic strains. Science 2000, 288, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Travers, S.A.; Clewley, J.P.; Glynn, J.R.; Fine, P.E.; Crampin, A.C.; Sibande, F.; Mulawa, D.; McInerney, J.O.; McCormack, G.P. Timing and reconstruction of the most recent common ancestor of the subtype C clade of human immunodeficiency virus type 1. J. Virol. 2004, 78, 10501–10506. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hraber, P.; Korber, B.T.; Lapedes, A.S.; Bailer, R.T.; Seaman, M.S.; Gao, H.; Greene, K.M.; McCutchan, F.; Williamson, C.; Kim, J.H.; et al. Impact of clade, geography, and age of the epidemic on HIV-1 neutralization by antibodies. J. Virol. 2014, 88, 12623–12643. [Google Scholar] [CrossRef] [PubMed]
- Mkhize, N.N.; Yssel, A.E.J.; Kaldine, H.; van Dorsten, R.T.; Woodward Davis, A.S.; Beaume, N.; Matten, D.; Lambson, B.; Modise, T.; Kgagudi, P.; et al. Neutralization profiles of HIV-1 viruses from the VRC01 Antibody Mediated Prevention (AMP) trials. PLoS Pathog. 2023, 19, e1011469. [Google Scholar] [CrossRef] [PubMed]
- Bekker, L.-G.; Moodie, Z.; Grunenberg, N.; Laher, F.; Tomaras, G.D.; Cohen, K.W.; Allen, M.; Malahleha, M.; Mngadi, K.; Daniels, B.; et al. Subtype C ALVAC-HIV and bivalent subtype C gp120/MF59 HIV-1 vaccine in low-risk, HIV-uninfected, South African adults: A phase 1/2 trial. Lancet HIV 2018, 5, e366–e378. [Google Scholar] [CrossRef]
- Gray, G.E.; Huang, Y.; Grunenberg, N.; Laher, F.; Roux, S.; Andersen-Nissen, E.; De Rosa, S.C.; Flach, B.; Randhawa, A.K.; Jensen, R.; et al. Immune correlates of the Thai RV144 HIV vaccine regimen in South Africa. Sci. Transl. Med. 2019, 11, eaax1880. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Laher, F.; Moodie, Z.; McMillan, A.S.; Spreng, R.L.; Gilbert, P.B.; Huang, Y.; Yates, N.L.; Grunenberg, N.; Juliana McElrath, M.; et al. HIV-1 Vaccine Sequences Impact V1V2 Antibody Responses: A Comparison of Two Poxvirus Prime gp120 Boost Vaccine Regimens. Sci. Rep. 2020, 10, 2093. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.E.; Bekker, L.-G.; Laher, F.; Malahleha, M.; Allen, M.; Moodie, Z.; Grunenberg, N.; Huang, Y.; Grove, D.; Prigmore, B.; et al. Vaccine Efficacy of ALVAC-HIV and Bivalent Subtype C gp120–MF59 in Adults. New Engl. J. Med. 2021, 384, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- HVTN. Experimental Phase 2B HIV Vaccine Regimen Provides Insufficient Protection in Preventing HIV 2021. Available online: https://www.hvtn.org/news/news-releases/2021/08/experimental-phase-2b-hiv-vaccine-regimen-provides-insufficient-.html (accessed on 31 August 2021).
- HVTN. Phase 3 Mosaic-Based Investigational HIV Vaccine Study Discontinued Following Disappointing Results of Planned Data Review 2023. Available online: https://www.hvtn.org/news/news-releases/2023/01/phase-3-mosaic-based-investigational-hiv-vaccine-study-discontinued-following-disappointing-results-planned-data-review.html (accessed on 14 October 2024).
- Zolla-Pazner, S.; deCamp, A.C.; Cardozo, T.; Karasavvas, N.; Gottardo, R.; Williams, C.; Morris, D.E.; Tomaras, G.; Rao, M.; Billings, E.; et al. Analysis of V2 antibody responses induced in vaccinees in the ALVAC/AIDSVAX HIV-1 vaccine efficacy trial. PLoS ONE 2013, 8, e53629. [Google Scholar] [CrossRef] [PubMed]
- Israel, Z.R.; Gorny, M.K.; Palmer, C.; McKeating, J.A.; Zolla-Pazner, S. Prevalence of a V2 epitope in clade B primary isolates and its recognition by sera from HIV-1-infected individuals. Aids 1997, 11, 128–130. [Google Scholar] [PubMed]
- Gorny, M.K.; Pan, R.; Williams, C.; Wang, X.H.; Volsky, B.; O'Neal, T.; Spurrier, B.; Sampson, J.M.; Li, L.; Seaman, M.S.; et al. Functional and immunochemical cross-reactivity of V2-specific monoclonal antibodies from HIV-1-infected individuals. Virology 2012, 427, 198–207. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, L.; Li, L.; Nanfack, A.; Mayr, L.M.; Soni, S.; Kohutnicki, A.; Agyingi, L.; Wang, X.H.; Tuen, M.; Shao, Y.; et al. Anti-V2 antibody deficiency in individuals infected with HIV-1 in Cameroon. Virology 2019, 529, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Bradley, T.; Pollara, J.; Santra, S.; Vandergrift, N.; Pittala, S.; Bailey-Kellogg, C.; Shen, X.; Parks, R.; Goodman, D.; Eaton, A.; et al. Pentavalent HIV-1 vaccine protects against simian-human immunodeficiency virus challenge. Nat. Commun. 2017, 8, 15711. [Google Scholar] [CrossRef] [PubMed]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree: Computing large minimum evolution trees with profiles instead of a distance matrix. Mol. Biol. Evol. 2009, 26, 1641–1650. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.X.; Sutherland, L.L.; Xia, S.M.; Brock, M.E.; Scearce, R.M.; Vanleeuwen, S.; Alam, S.M.; McAdams, M.; Weaver, E.A.; Camacho, Z.; et al. A group M consensus envelope glycoprotein induces antibodies that neutralize subsets of subtype B and C HIV-1 primary viruses. Virology 2006, 353, 268–282. [Google Scholar] [CrossRef] [PubMed]
- Gautam, R.; Nishimura, Y.; Pegu, A.; Nason, M.C.; Klein, F.; Gazumyan, A.; Golijanin, J.; Buckler-White, A.; Sadjadpour, R.; Wang, K.; et al. A single injection of anti-HIV-1 antibodies protects against repeated SHIV challenges. Nature 2016, 533, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Moody, M.A.; Santra, S.; Vandergrift, N.A.; Sutherland, L.L.; Gurley, T.C.; Drinker, M.S.; Allen, A.A.; Xia, S.M.; Meyerhoff, R.R.; Parks, R.; et al. Toll-like receptor 7/8 (TLR7/8) and TLR9 agonists cooperate to enhance HIV-1 envelope antibody responses in rhesus macaques. J. Virol. 2014, 88, 3329–3339. [Google Scholar] [CrossRef] [PubMed]
- Tomaras, G.D.; Yates, N.L.; Liu, P.; Qin, L.; Fouda, G.G.; Chavez, L.L.; Decamp, A.C.; Parks, R.J.; Ashley, V.C.; Lucas, J.T.; et al. Initial B-cell responses to transmitted human immunodeficiency virus type 1: Virion-binding immunoglobulin M (IgM) and IgG antibodies followed by plasma anti-gp41 antibodies with ineffective control of initial viremia. J. Virol. 2008, 82, 12449–12463. [Google Scholar] [CrossRef] [PubMed]
- Ritz, C.; Baty, F.; Streibig, J.C.; Gerhard, D. Dose-Response Analysis Using R. PLoS ONE 2016, 10, e0146021. [Google Scholar]
- Shen, X.; Duffy, R.; Howington, R.; Cope, A.; Sadagopal, S.; Park, H.; Pal, R.; Kwa, S.; Ding, S.; Yang, O.O.; et al. Vaccine-Induced Linear Epitope-Specific Antibodies to Simian Immunodeficiency Virus SIVmac239 Envelope Are Distinct from Those Induced to the Human Immunodeficiency Virus Type 1 Envelope in Nonhuman Primates. J. Virol. 2015, 89, 8643–8650. [Google Scholar] [CrossRef] [PubMed]
- Saunders, K.O.; Nicely, N.I.; Wiehe, K.; Bonsignori, M.; Meyerhoff, R.R.; Parks, R.; Walkowicz, W.E.; Aussedat, B.; Wu, N.R.; Cai, F.; et al. Vaccine Elicitation of High Mannose-Dependent Neutralizing Antibodies against the V3-Glycan Broadly Neutralizing Epitope in Nonhuman Primates. Cell Rep. 2017, 18, 2175–2188. [Google Scholar] [CrossRef] [PubMed]
- Bricault, C.A.; Yusim, K.; Seaman, M.S.; Yoon, H.; Theiler, J.; Giorgi, E.E.; Wagh, K.; Theiler, M.; Hraber, P.; Macke, J.P.; et al. HIV-1 Neutralizing Antibody Signatures and Application to Epitope-Targeted Vaccine Design. Cell Host Microbe 2019, 25, 59–72.e8. [Google Scholar] [CrossRef] [PubMed]
- Ringe, R.; Phogat, S.; Bhattacharya, J. Subtle alteration of residues including N-linked glycans in V2 loop modulate HIV-1 neutralization by PG9 and PG16 monoclonal antibodies. Virology 2012, 426, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Doores, K.J.; Burton, D.R. Variable loop glycan dependency of the broad and potent HIV-1-neutralizing antibodies PG9 and PG16. J. Virol. 2010, 84, 10510–10521. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Lopez, C.A.; Derdeyn, C.A.; Jones, M.S.; Pinter, A.; Korber, B.; Gnanakaran, S. Effect of Glycosylation on an Immunodominant Region in the V1V2 Variable Domain of the HIV-1 Envelope gp120 Protein. PLoS Comput. Biol. 2016, 12, e1005094. [Google Scholar] [CrossRef] [PubMed]
- Yates, N.L.; deCamp, A.C.; Korber, B.T.; Liao, H.X.; Irene, C.; Pinter, A.; Peacock, J.; Harris, L.J.; Sawant, S.; Hraber, P.; et al. HIV-1 Envelope Glycoproteins from Diverse Clades Differentiate Antibody Responses and Durability among Vaccinees. J. Virol. 2018, 92, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Arthos, J.; Cicala, C.; Martinelli, E.; Macleod, K.; Van Ryk, D.; Wei, D.; Xiao, Z.; Veenstra, T.D.; Conrad, T.P.; Lempicki, R.A.; et al. HIV-1 envelope protein binds to and signals through integrin alpha4beta7, the gut mucosal homing receptor for peripheral T cells. Nat. Immunol. 2008, 9, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Devasundaram, S.; Rosati, M.; Valentin, A.; Weiss, S.; Itri, V.; Trinh, H.V.; Bear, J.; Chowdhury, B.; LaBranche, C.C.; Montefiori, D.; et al. Priming with DNA Expressing Trimeric HIV V1V2 Alters the Immune Hierarchy Favoring the Development of V2-Specific Antibodies in Rhesus Macaques. J. Virol. 2020, 95, e01193-20. [Google Scholar] [CrossRef] [PubMed]
- Zolla-Pazner, S.; Edlefsen, P.T.; Rolland, M.; Kong, X.P.; deCamp, A.; Gottardo, R.; Williams, C.; Tovanabutra, S.; Sharpe-Cohen, S.; Mullins, J.I.; et al. Vaccine-induced Human Antibodies Specific for the Third Variable Region of HIV-1 gp120 Impose Immune Pressure on Infecting Viruses. eBioMedicine 2014, 1, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Schifanella, L.; Barnett, S.W.; Bissa, M.; Galli, V.; Doster, M.N.; Vaccari, M.; Tomaras, G.D.; Shen, X.; Phogat, S.; Pal, R.; et al. ALVAC-HIV B/C candidate HIV vaccine efficacy dependent on neutralization profile of challenge virus and adjuvant dose and type. PLoS Pathog. 2019, 15, e1008121. [Google Scholar] [CrossRef] [PubMed]
- Peachman, K.K.; Karasavvas, N.; Chenine, A.L.; McLinden, R.; Rerks-Ngarm, S.; Jaranit, K.; Nitayaphan, S.; Pitisuttithum, P.; Tovanabutra, S.; Zolla-Pazner, S.; et al. Identification of New Regions in HIV-1 gp120 Variable 2 and 3 Loops that Bind to alpha4beta7 Integrin Receptor. PLoS ONE 2015, 10, e0143895. [Google Scholar] [CrossRef] [PubMed]
- Easternhoff, D.; Peachman, K.K.; Rao, M.; Haynes, B.F. HIV vaccine delayed boosting increases Env variable region 2–specific antibody effector functions. JCI Insight. 2020, 5, e131437. [Google Scholar] [CrossRef]
- Kenny, A.; van Duijn, J.; Dintwe, O.; Heptinstall, J.; Burnham, R.; Sawant, S.; Zhang, L.; Mielke, D.; Khuzwayo, S.; Omar, F.L.; et al. Immune correlates analysis of the Imbokodo (HVTN 705/HPX2008) efficacy trial of a mosaic HIV-1 vaccine regimen evaluated in Southern African people assigned female sex at birth: A two-phase case-control study. eBioMedicine 2024, 108, 105320. [Google Scholar] [CrossRef] [PubMed]
- Rademeyer, C.; Korber, B.; Seaman, M.S.; Giorgi, E.E.; Thebus, R.; Robles, A.; Sheward, D.J.; Wagh, K.; Garrity, J.; Carey, B.R.; et al. Features of recently transmitted HIV-1 clade C viruses that impact antibody recognition: Implications for active and passive immunization. PLoS Pathog. 2016, 12, e1005742. [Google Scholar] [CrossRef]

| Trivalent (Tri-V) | Bivalent (Bi-V) | Pentavalent (Penta-V) | |
|---|---|---|---|
| Immunogen | CAP260 gp120 Δ11 env CAP174 gp120 Δ11 env Ko224 gp120 Δ11 env | TV1c8 gp120 Δ11 env 1086C gp120 Δ7 env | CAP260 gp120 Δ11 env CAP174 gp120 Δ11 env Ko224 gp120 Δ11 env TV1c8 gp120 Δ11 env 1086C gp120 Δ7 env |
| Dose | 100 μg per animal per protein | 100 μg per animal per protein | 100 μg per animal per protein |
| Adjuvant | STR8S-C | STR8S-C | STR8S-C |
| Immunization time | Days 0, 21, 42, 63 | Days 0, 21, 42, 63 | Days 0, 21, 42, 63 |
| Group size | N = 5 * | N = 6 | N = 5 * |
| Group Median | p Value *, Wilcoxon Rank Sum Test | ||||||
|---|---|---|---|---|---|---|---|
| Category | Analyte | Tri-C | Bi-C | Penta-C | Tri- vs. Bi-C | Tri- vs. Penta-C | Bi- vs. Penta-C |
| Heterologous | gp70.001428.2.42_V1V2 | 19,671 | 2921 | 8903 | 0.004 | 0.310 | 0.004 |
| V1V2 | gp70.1051.12.C22_V1V2 | 9041 | 1046 | 2970 | 0.004 | 0.095 | 0.004 |
| scaffold | gp70.1394C9G1_V1V2 | 7560 | 896 | 2897 | 0.009 | 0.841 | 0.004 |
| gp70.62357.14_V1V2 | 13,049 | 298 | 4633 | 0.004 | 0.151 | 0.004 | |
| gp70.700010058_V1V2 | 10,312 | 989 | 6103 | 0.004 | 0.095 | 0.004 | |
| gp70.B.CaseA_V1V2 | 5385 | 1228 | 1647 | 0.009 | 0.151 | 0.429 | |
| gp70.BJOX002000.03.2_V1V2 | 9518 | 2542 | 5591 | 0.030 | 0.548 | 0.052 | |
| gp70.CAP210.2.00.E8_V1V2 | 10,188 | 1105 | 5455 | 0.004 | 0.056 | 0.004 | |
| gp70.CAP45.2.00.G3_V1V2 | 16,340 | 1998 | 10,624 | 0.004 | 0.222 | 0.009 | |
| gp70.Ce1176_V1V2 | 7958 | 1702 | 4248 | 0.017 | 0.095 | 0.082 | |
| gp70.RHPA4259.7_V1V2 | 6351 | 277 | 2301 | 0.004 | 0.310 | 0.004 | |
| gp70.TT31P.2F10.2792_V1V2 | 12,452 | 1015 | 7609 | 0.004 | 0.421 | 0.009 | |
| gp70.7060101641_V1V2 | 2790 | 14,347 | 11,982 | 0.017 | 0.056 | 0.329 | |
| gp70.CM244.ec1_V1V2 | 6399 | 189,146 | 27,970 | 0.004 | 0.032 | 0.009 | |
| gp70.191084_B7_V1V2 | 10,988 | 9728 | 10,177 | 0.662 | 1.000 | 0.662 | |
| gp70.96ZM651.02_V1V2 | 9707 | 8667 | 6374 | 0.931 | 0.841 | 1.000 | |
| gp70.BF1266_431a_V1V2 | 2141 | 3363 | 965 | 0.662 | 0.841 | 0.931 | |
| gp70.C2101.c01_V1V2 | 11,113 | 14,661 | 14,489 | 0.429 | 0.310 | 0.931 | |
| gp70.Ce704010042_2ES_V1V2 | 8530 | 10,108 | 9396 | 0.429 | 1.000 | 0.537 | |
| gp70.Du156.12_V1V2 | 8725 | 3956 | 9215 | 0.429 | 1.000 | 0.537 | |
| Vaccine- | gp70.Ce1086_B2_V1V2 | 6338 | 63,496 | 29,731 | 0.004 | 0.008 | 0.004 |
| matched | gp70.TV1.21_V1V2 | 8546 | 6296 | 4345 | 0.329 | 0.016 | 0.792 |
| V1V2 | gp70.TV1.GSKvacV1V2 | 5122 | 7338 | 2401 | 0.792 | 0.151 | 0.537 |
| V1V2 and | AE.A244.V1V2_Tags | 1530 | 6554 | 5594 | 0.009 | 0.056 | 0.429 |
| V2 Tags | C.1086C.V1V2_Tags | 1703 | 9758 | 5762 | 0.004 | 0.056 | 0.004 |
| AE.A244.V2_Tags | 2488 | 5627 | 5137 | 0.126 | 0.222 | 0.537 | |
| C.1086.V2_Tags | 6135 | 4107 | 5065 | 0.537 | 0.690 | 0.329 | |
| Env | CH505.TF.gp120 | 44,770 | 33,091 | 40,088 | 0.247 | 0.548 | 0.662 |
| Con.6.gp120_B | 17,953 | 9163 | 23,578 | 0.052 | 0.310 | 0.009 | |
| Con.S.gp140.CFI | 64,043 | 268,439 | 57,605 | 0.537 | 1.000 | 0.429 | |
| TV1c8_D11gp120.avi | 40,029 | 335,328 | 53,661 | 0.082 | 0.222 | 0.082 | |
| Group Median | p Value *, Wilcoxon Rank Sum Test | ||||||
|---|---|---|---|---|---|---|---|
| Epitope | Strain | Tri-C | Bi-C | Penta-C | Tri- vs. Bi-C | Tri- vs. Penta-C | Bi- vs. Penta-C |
| V2.hotspot | C.1086 | 0.0 | 6.6 | 6.2 | 0.004 | 0.008 | 0.628 |
| V2.hotspot | C.TV1 | 1.8 | 2.7 | 1.3 | 0.429 | 0.802 | 0.502 |
| V2.hotspot | AnyStrain # | 4.4 | 7.3 | 7.6 | 0.017 | 0.032 | 0.500 |
| V2.2 | C.1086 | 0.1 | 2.8 | 0.7 | 0.050 | 0.206 | 0.459 |
| V2.2 | C.TV1 | 6.7 | 0.5 | 5.8 | 0.022 | 0.841 | 0.082 |
| V2.2 | AnyStrain | 6.7 | 6.6 | 6.0 | 1.000 | 0.841 | 0.931 |
| Group Median | p Value *, Wilcoxon Rank Sum Test | ||||||
|---|---|---|---|---|---|---|---|
| Time Point | Blocking Pair | Tri-C | Bi-C | Penta-C | Tri- vs. Bi-C | Tri- vs. Penta-C | Bi- vs. Penta-C |
| Post Immu.#2 | A32 x AE.A244 gp120 | 67.1 | 82.3 | 88.3 | 0.016 | 0.011 | 0.1 |
| Post Immu.#2 | CH58 x AE.A244 gp120 | 6.6 | 46.3 | 26.4 | 0.004 | 0.100 | 0.27 |
| Post Immu.#4 | A32 x AE.A244 gp120 | 78.0 | 90.6 | 88.5 | 0.018 | 0.009 | 0.86 |
| Post Immu.#4 | CH58 x AE.A244 gp120 | 25.0 | 83.0 | 80.5 | 0.006 | 0.009 | 0.27 |
| Group | Alum Pentavalent E/C | GLA-SE Pentavalent E/C |
|---|---|---|
| Immunogen | AE.A244_D11gp120/293F/Mon | AE.A244_D11gp120/293F/Mon |
| CAP260 gp120 Δ11 env | CAP260 gp120 Δ11 env | |
| CAP174 gp120 Δ11 env | CAP174 gp120 Δ11 env | |
| Ko224 gp120 Δ11 env | Ko224 gp120 Δ11 env | |
| 1086C gp120 Δ7 env | 1086C gp120 Δ7 env | |
| Dose | 100 mg per animal per protein | 100 mg per animal per protein |
| Adjuvant | Alum rehydrogel | GLA-SE |
| Immunization time | Week 0, 4, 8 | Week 0, 4, 8 |
| Group size | N = 4 | N = 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, X.; Korber, B.; Spreng, R.L.; Sawant, S.S.; deCamp, A.; McMillan, A.S.; Mathura, R.; Zolla-Pazner, S.; Pinter, A.; Parks, R.; et al. A Pentavalent HIV-1 Subtype C Vaccine Containing Computationally Selected gp120 Strains Improves the Breadth of V1V2 Region Responses. Vaccines 2025, 13, 133. https://doi.org/10.3390/vaccines13020133
Shen X, Korber B, Spreng RL, Sawant SS, deCamp A, McMillan AS, Mathura R, Zolla-Pazner S, Pinter A, Parks R, et al. A Pentavalent HIV-1 Subtype C Vaccine Containing Computationally Selected gp120 Strains Improves the Breadth of V1V2 Region Responses. Vaccines. 2025; 13(2):133. https://doi.org/10.3390/vaccines13020133
Chicago/Turabian StyleShen, Xiaoying, Bette Korber, Rachel L. Spreng, Sheetal S. Sawant, Allan deCamp, Arthur S. McMillan, Ryan Mathura, Susan Zolla-Pazner, Abraham Pinter, Robert Parks, and et al. 2025. "A Pentavalent HIV-1 Subtype C Vaccine Containing Computationally Selected gp120 Strains Improves the Breadth of V1V2 Region Responses" Vaccines 13, no. 2: 133. https://doi.org/10.3390/vaccines13020133
APA StyleShen, X., Korber, B., Spreng, R. L., Sawant, S. S., deCamp, A., McMillan, A. S., Mathura, R., Zolla-Pazner, S., Pinter, A., Parks, R., Bowman, C., Sutherland, L., Scearce, R., Yates, N. L., Montefiori, D. C., Haynes, B. F., & Tomaras, G. D. (2025). A Pentavalent HIV-1 Subtype C Vaccine Containing Computationally Selected gp120 Strains Improves the Breadth of V1V2 Region Responses. Vaccines, 13(2), 133. https://doi.org/10.3390/vaccines13020133







