Vaccine Hesitancy and Associated Factors Amongst Health Professionals: A Scoping Review of the Published Literature
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Search
2.3. Data Extraction and Analysis
3. Results
3.1. Screening and Procedure
3.2. Overview of Characteristics of the Included Studies
3.3. Measures
3.4. Vaccination History and Hesitancy
3.5. Socio-Demographic Characteristics and COVID-19-Related Variables Associated with Vaccination Hesitancy
3.6. Vaccination Knowledge and Attitudes
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ashmore, P.; Sherwood, E. An overview of COVID-19 global epidemiology and discussion of potential drivers of variable global pandemic impacts. J. Antimicrob. Chemother. 2023, 78, ii2–ii11. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, S.H.; Mansatta, K.; Mallett, G.; Harris, V.; Emary, K.R.W.; Pollard, A.J. What defines an efficacious COVID-19 vaccine? A review of the challenges assessing the clinical efficacy of vaccines against SARS-CoV-2. Lancet Infect. Dis. 2021, 21, e26–e35. [Google Scholar] [CrossRef] [PubMed]
- Adebisi, Y.A.; Alaran, A.J.; Bolarinwa, O.A.; Akande-Sholabi, W.; Lucero-Prisno, D.E. When it is available, will we take it? Social media users’ perception of hypothetical COVID-19 vaccine in Nigeria. Pan Afr. Med. J. 2021, 38, 230. [Google Scholar] [CrossRef] [PubMed]
- De Gier, B.; van Asten, L.; Boere, T.M.; van Roon, A.; van Roekel, C.; Pijpers, J.; van Werkhoven, C.H.H.; van den Ende, C.; Hahné, S.J.M.; de Melker, H.E.; et al. Effect of COVID-19 vaccination on mortality by COVID-19 and on mortality by other causes, the Netherlands, January 2021–January 2022. Vaccine 2023, 41, 4488–4496. [Google Scholar] [CrossRef]
- Wouters, O.J.; Shadlen, K.C.; Salcher-Konrad, M.; Pollard, A.J.; Larson, H.J.; Teerawattananon, Y.; Jit, M. Challenges in ensuring global access to COVID-19 vaccines: Production, affordability, allocation, and deployment. Lancet 2021, 397, 1023–1034. [Google Scholar] [CrossRef]
- Harder, T.; Koch, J.; Vygen-Bonnet, S.; Külper-Schiek, W.; Pilic, A.; Reda, S.; Scholz, S.; Wichmann, O. Efficacy and effectiveness of COVID-19 vaccines against SARS-CoV-2 infection: Interim results of a living systematic review, 1 January to 14 May 2021. Eurosurveillance 2021, 26, 2100563. [Google Scholar] [CrossRef]
- Chakraborty, C.; Bhattacharya, M.; Dhama, K. SARS-CoV-2 vaccines, vaccine development technologies, and significant efforts in vaccine development during the pandemic: The lessons learned might help to fight against the next pandemic. Vaccines 2023, 11, 682. [Google Scholar] [CrossRef]
- Wu, N.; Joyal-Desmarais, K.; Ribeiro, P.A.B.; Vieira, A.M.; Stojanovic, J.; Sanuade, C.; Yip, D.; Bacon, S.L. Long-term effectiveness of COVID-19 vaccines against infections, hospitalisations, and mortality in adults: Findings from a rapid living systematic evidence synthesis and meta-analysis up to December, 2022. Lancet Respir. Med. 2023, 11, 439–452. [Google Scholar] [CrossRef]
- Naaber, P.; Tserel, L.; Kangro, K.; Sepp, E.; Jürjenson, V.; Adamson, A.; Haljasmägi, L.; Rumm, A.P.; Maruste, R.; Kärner, J.; et al. Dynamics of antibody response to BNT162b2 vaccine after six months: A longitudinal prospective study. Lancet Reg. Health Eur. 2021, 10, 100208. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Vaccine Efficacy, Effectiveness and Protection. Available online: https://www.who.int/news-room/feature-stories/detail/vaccine-efficacy-effectiveness-and-protection (accessed on 20 June 2024).
- World Health Organization (WHO). WHO SAGE Roadmap on Uses of COVID-19 Vaccines in the Context of OMICRON and Substantial Population Immunity. Available online: https://iris.who.int/handle/10665/366671 (accessed on 14 April 2024).
- Peterson, C.J.; Lee, B.; Nugent, K. COVID-19 Vaccination Hesitancy among Healthcare Workers—A Review. Vaccines 2022, 10, 948. [Google Scholar] [CrossRef]
- Razai, M.S.; Oakeshott, P.; Esmail, A.; Wiysonge, C.S.; Viswanath, K.; Mills, M.C. COVID-19 vaccine hesitancy: The five Cs to tackle behavioural and sociodemographic factors. J. R. Soc. Med. 2021, 114, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Y.; Cheng, X.; Li, X.; Yang, Y.; Li, J. Acceptance of coronavirus disease 2019 (COVID-19) vaccines among healthcare workers: A meta-analysis. Front. Public Health 2022, 10, 881903. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Mullen, J.; Smith, D.; Kotarba, M.; Kaplan, S.J.; Tu, P. Healthcare Providers’ Vaccine Perceptions, Hesitancy, and Recommendation to Patients: A Systematic Review. Vaccines 2021, 9, 713. [Google Scholar] [CrossRef] [PubMed]
- Ramonfaur, D.; Limaye, R.J.; Hinojosa-González, D.E.; Barrera, F.J.; Rodríguez-Gómez, G.P.; Castillo-Salgado, C. COVID-19 vaccine hesitancy prevalence in Mexico: A systematic review and metanalysis. Vaccine X 2024, 18, 100488. [Google Scholar] [CrossRef]
- Rahbeni, T.A.; Satapathy, P.; Itumalla, R.; Marzo, R.R.; Mugheed, K.A.L.; Khatib, M.N.; Gaidhane, S.; Zahiruddin, Q.S.; Rabaan, A.A.; Alrasheed, H.A.; et al. COVID-19 Vaccine Hesitancy: Umbrella Review of Systematic Reviews and Meta-Analysis. JMIR Public Health Surveill. 2024, 10, e54769. [Google Scholar] [CrossRef]
- Weinstein, N.; Schwarz, K.; Chan, I.; Kobau, R.; Alexander, R.; Kollar, L.; Rodriguez, L.; Mansergh, G.; Repetski, T.; Gandhi, P.; et al. COVID-19 Vaccine Hesitancy Among US Adults: Safety and Effectiveness Perceptions and Messaging to Increase Vaccine Confidence and Intent to Vaccinate. Public Health Rep. 2024, 139, 102–111. [Google Scholar] [CrossRef]
- Bhattacharya, O.; Siddiquea, B.N.; Shetty, A.; Afroz, A.; Billah, B. COVID-19 vaccine hesitancy among pregnant women: A systematic review and meta-analysis. BMJ Open 2022, 12, e061477. [Google Scholar] [CrossRef]
- Ma, Y.; Ren, J.; Zheng, Y.; Cai, D.; Li, S.; Li, Y. Chinese parents’ willingness to vaccinate their children against COVID-19: A systematic review and meta-analysis. Front. Public Health 2022, 10, 1087295. [Google Scholar] [CrossRef]
- Burrowes, S.A.B.; Casey, S.M.; Dobbins, S.; Hall, T.; Ma, M.; Bano, R.; Drainoni, M.L.; Schechter-Perkins, E.M.; Garofalo, C.; Perkins, R.B.; et al. Healthcare workers’ perspectives on the COVID-19 vaccine and boosters for themselves, their patients, and their communities: A mixed methods study. J. Public Health 2024, 32, 123–136. [Google Scholar] [CrossRef]
- Maraqa, B.; Nazzal, Z.; Baroud, H.; Douden, M.; El Hamshary, Y.; Jalamneh, T. Healthcare workers’ attitudes toward and factors influencing their acceptance of an annual COVID-19 booster vaccine: A cross-sectional study in Palestine. BMC Health Serv. Res. 2024, 24, 624. [Google Scholar] [CrossRef]
- Aldakhlan, H.A.; Khan, A.S.; Alabdulbaqi, D. Hesitancy Over the COVID-19 Vaccine Among Various Healthcare Workers: An International Narrative Review. Cureus 2024, 16, e53059. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control (ECDC). Vaccine Hesitancy. Available online: https://www.ecdc.europa.eu/en/immunisation-vaccines/vaccine-hesitancy (accessed on 10 July 2024).
- Oliver, K.; Raut, A.; Pierre, S.; Silvera, L.; Boulos, A.; Gale, A.; Baum, A.; Chory, A.; Davis, N.J.; D’Souza, D.; et al. Factors associated with COVID-19 vaccine receipt at two integrated healthcare systems in New York City: A cross-sectional study of healthcare workers. BMJ Open 2022, 12, e053641. [Google Scholar] [CrossRef] [PubMed]
- Toth-Manikowski, S.M.; Swirsky, E.S.; Gandhi, R.; Piscitello, G. COVID-19 vaccination hesitancy among health care workers, communication, and policy-making. Am. J. Infect. Control. 2022, 50, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, L.; Jin, H.; Lin, L. Vaccination against COVID-19: A systematic review and meta-analysis of acceptability and its predictors. Prev. Med. 2021, 150, 106694. [Google Scholar] [CrossRef] [PubMed]
- Painter, E.M.; Ussery, E.N.; Patel, A.; Hughes, M.M.; Zell, E.R.; Moulia, D.L.; Scharf, L.G.; Lynch, M.; Ritchey, M.D.; Toblin, R.L.; et al. Demographic Characteristics of Persons Vaccinated During the First Month of the COVID-19 Vaccination Program—United States, December 14, 2020–January 14, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 174–177. [Google Scholar] [CrossRef]
- Farah, W.; Breeher, L.; Shah, V.; Hainy, C.; Tommaso, C.P.; Swift, M.D. Disparities in COVID-19 vaccine uptake among health care workers. Vaccine 2022, 40, 2749–2754. [Google Scholar] [CrossRef]
- Lee, J.T. Disparities in COVID-19 vaccination coverage among health care personnel working in long-term care facilities, by job category, National Healthcare Safety Network—United States, March 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 1036–1039. [Google Scholar] [CrossRef]
- Wilpstra, C.D.; Morrell, S.; Mirza, N.A.; Ralph, J.L. Consequences of COVID-19 Vaccine Hesitancy Among Healthcare Providers During the First 10 Months of Vaccine Availability: Scoping Review. Can. J. Nurs. Res. 2024, 56, 204–224. [Google Scholar] [CrossRef]
- Wróblewski, M.; Stankowska, J.; Kawiak-Jawor, E. ‘We’re at war.’ Healthcare workers’ experience with organisational change, uncertainty and vaccine hesitancy in 2021 and 2022 during the COVID-19 vaccination programe in Poland. Int. J. Health Plan. Manag. 2024, 39, 1298–1312. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, Y.; Jing, S.; Liu, X.; Ren, T.; Liu, X.; Dai, Z.; Fu, J.; Chen, X.; Xiao, W.; et al. The second dose of COVID-19 vaccine booster hesitancy among health care workers in China: A multicenter cross-sectional study. Am. J. Infect. Control. 2024, 52, 525–532. [Google Scholar] [CrossRef]
- Thampy, P.; Sharma, S.; Joshi, P.; Raj, M.S.; Rupani, A.; Tyagi, S.; Joshi, A. COVID-19 Vaccine Hesitancy Among Healthcare Workers: A Phenomenological Study of Skepticism. Cureus 2024, 16, e58445. [Google Scholar] [CrossRef] [PubMed]
- Thaivalappil, A.; Young, I.; MacKay, M.; Pearl, D.L.; Papadopoulos, A. A qualitative study exploring healthcare providers’ and trainees’ barriers to COVID-19 and influenza vaccine uptake. Health Psychol. Behav. Med. 2022, 10, 695–712. [Google Scholar] [CrossRef] [PubMed]
- Bedston, S.; Lowthian, E.; Jarvis, C.I.; Akbari, A.; Beggs, J.; Bradley, D.; de Lusignan, S.; Griffiths, R.; Herbert, L.; Hobbs, R.; et al. COVID-19 booster vaccination uptake and infection breakthrough amongst health care workers in Wales: A national prospective cohort study. Vaccine 2023, 41, 1378–1389. [Google Scholar] [CrossRef]
- Peters, M.D.; Godfrey, C.; McInerney, P.; Munn, Z.; Tricco, A.C.; Khalil, H. Chapter 11: Scoping reviews. JBI Man. Evid. Synth. 2020, 169, 467–473. [Google Scholar]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Mak, S.; Thomas, A. Steps for Conducting a Scoping Review. J. Grad. Med. Educ. 2022, 14, 565–567. [Google Scholar] [CrossRef]
- Arshad, M.S.; Masood, I.; Imran, I.; Saeed, H.; Ahmad, I.; Ishaq, I.; Yaseen, H.; Akbar, M.; Chaudhry, M.O.; Rasool, M.F. COVID-19 Vaccine Booster Hesitancy (VBH) among Healthcare Professionals of Pakistan, a Nationwide Survey. Vaccines 2022, 10, 1736. [Google Scholar] [CrossRef]
- Dale, C.; Seage, C.H.; Phillips, R.; James, D. The Role of Medication Beliefs in COVID-19 Vaccine and Booster Uptake in Healthcare Workers: An Exploratory Study. Healthcare 2023, 11, 1967. [Google Scholar] [CrossRef]
- Della Polla, G.; Miraglia Del Giudice, G.; Folcarelli, L.; Napoli, A.; Angelillo, I.F. Willingness to accept a second COVID-19 vaccination booster dose among healthcare workers in Italy. Front. Public Health 2022, 10, 1051035. [Google Scholar] [CrossRef]
- Dudley, M.Z.; Schuh, H.B.; Forr, A.; Shaw, J.; Salmon, D.A. Changes in vaccine attitudes and recommendations among US Healthcare Personnel during the COVID-19 pandemic. NPJ Vaccines 2024, 9, 49. [Google Scholar] [CrossRef]
- Galanis, P.; Vraka, I.; Katsiroumpa, A.; Siskou, O.; Konstantakopoulou, O.; Katsoulas, T.; Mariolis-Sapsakos, T.; Kaitelidou, D. Predictors of second COVID-19 booster dose or new COVID-19 vaccine hesitancy among nurses: A cross-sectional study. J. Clin. Nurs. 2023, 32, 3943–3953. [Google Scholar] [CrossRef] [PubMed]
- George, G.; Nota, P.; Strauss, M.; Lansdell, E.; Peters, R.P.H.; Brysiewicz, P.; Nadesan-Reddy, N.; Wassenaar, D. Examining the uptake of COVID-19 vaccine booster doses among healthcare workers in South Africa: A mixed-methods study. PLoS Glob. Public Health 2023, 3, e0002639. [Google Scholar] [CrossRef]
- Gu, F.; Lin, H.; Chen, Z.; Ambler, G.; Chen, X.; Chen, X.; Xia, P.; Liu, N.; Du, H. Future COVID-19 Booster Vaccine Refusal in Healthcare Workers after a Massive Breakthrough Infection Wave, a Nationwide Survey-Based Study. Vaccines 2023, 11, 987. [Google Scholar] [CrossRef]
- Guarducci, G.; Mereu, G.; Golinelli, D.; Galletti, G.; Gemmi, F.; Cartocci, A.; Holczer, N.; Bacci, L.; Sergi, A.; Messina, G.; et al. Factors Influencing the Healthcare Workers’ Willingness to Receive the COVID-19 Booster Dose in Tuscany (Italy). Vaccines 2023, 11, 1751. [Google Scholar] [CrossRef]
- Krishna, E.; Karthikeyan, V.; Ahmad, S.; Ranjan, A.; Hasan Km, A.; Pandey, S.; Kumar, P.; Singh, C.M. Acceptance of Annual Booster Doses of COVID-19 Vaccines Among Indian Healthcare Professionals: A Pan-India Cross-Sectional Survey. Cureus 2023, 15, e49363. [Google Scholar] [CrossRef]
- Lubad, M.A.; Abu-Helalah, M.A.; Alahmad, I.F.; Al-Tamimi, M.M.; QawaQzeh, M.S.; Al-Kharabsheh, A.M.; Alzoubi, H.; Alnawafleh, A.H.; Kheirallah, K.A. Willingness of Healthcare Workers to Recommend or Receive a Third COVID-19 Vaccine Dose: A Cross-Sectional Study from Jordan. Infect Dis. Rep. 2023, 15, 210–221. [Google Scholar] [CrossRef]
- Paudel, K.; Shah, S.; Bhusal, S.; Dahal, K.; Bhatta, N.; Pokhrel, S.; Dahal, S.; Gaihre, M.; Mudvari, A.; Gyanwali, P. Knowledge and attitude toward COVID-19 booster dose among health care professionals in Nepal: A cross-sectional study. Ann. Med. Surg. 2023, 85, 772–777. [Google Scholar] [CrossRef]
- Ramot, S.; Tal, O. Attitudes of Healthcare Workers in Israel towards the Fourth Dose of COVID-19 Vaccine. Vaccines 2023, 11, 385. [Google Scholar] [CrossRef]
- Rathinakumar, N.K.; Nishanthi, A.; Manickam, S. Perception and practices on COVID-19 vaccination and booster dose acceptability among health-care workers—A questionnaire-based study. Perspect Clin. Res. 2024, 15, 10–17. [Google Scholar] [CrossRef]
- Salah, H.; Sinan, I.; Alsamani, O.; Abdelghani, L.S.; ElLithy, M.H.; Bukamal, N.; Jawad, H.; Hussein, R.R.S.; Elgendy, M.O.; Rabie, A.S.I.; et al. COVID-19 Booster Doses: A Multi-Center Study Reflecting Healthcare Providers’ Perceptions. Vaccines 2023, 11, 1061. [Google Scholar] [CrossRef]
- Sansone, V.; Miraglia Del Giudice, G.; Della Polla, G.; Angelillo, I.F. Impact of the COVID-19 pandemic on behavioral changes in healthcare workers in Italy. Front. Public Health 2024, 12, 1335953. [Google Scholar] [CrossRef] [PubMed]
- Viskupič, F.; Wiltse, D.L. Drivers of COVID-19 booster uptake among nurses. Am. J. Infect Control. 2023, 51, 895–899. [Google Scholar] [CrossRef] [PubMed]
- Zoumpoulis, G.; Deligiorgi, P.; Lamprinos, D.; Georgakopoulos, P.; Oikonomou, E.; Siasos, G.; Rachiotis, G.; Damaskos, C.; Papagiannis, D.; Papavassiliou, K.A.; et al. Attitudes and Practices Related to COVID-19 Vaccination with the Second Booster Dose among Members of Athens Medical Association: Results from a Cross-Sectional Study. Vaccines 2023, 11, 1480. [Google Scholar] [CrossRef]
- Digregorio, M.; Van Ngoc, P.; Domen, J.; Bognar, Z.; Duysburgh, E.; Hendrickx, G.; Van Damme, P.; Coenen, S.; Scholtes, B. Primary Healthcare Providers’ Views on Periodic COVID-19 Booster Vaccination for Themselves and Their Patients: A 2023 Nationwide Survey in Belgium. Vaccines 2024, 12, 740. [Google Scholar] [CrossRef]
- Kolomba, B.M.; Kalenga Luhembwe, F.; Ndala, D.B.B.; Kanku Wa Ilunga, P.; Ciamala Mukendi, P.; Ngongo Kitenge, A.; Ngoy Lumbule, J.; Kilolo Ngoy, E.; Umba Ilunga, A.; Mbidi Miema, J.; et al. Healthcare workers’ willingness to receive COVID-19 booster dose and associated factors in the Democratic Republic of the Congo. Hum. Vaccin. Immunother. 2024, 20, 2357214. [Google Scholar] [CrossRef]
- Pandarathodiyil, A.K.; Veerabhadrappa, S.K.; Nabillah Ghani, W.M.; Termizi Bin Zamzuri, A. COVID-19 Booster Vaccination Adverse Effects and Willingness to Receive a Yearly Booster Dose among Members of Health Sciences Faculties: A Descriptive Cross-Sectional Study. J. Pharm. Bioallied. Sci. 2024, 16, S1776–S1783. [Google Scholar] [CrossRef]
- Pristov, Z.; Lobe, B.; Sočan, M. Factors Influencing COVID-19 Vaccination among Primary Healthcare Nurses in the Pandemic and Post-Pandemic Period: Cross-Sectional Study. Vaccines 2024, 12, 602. [Google Scholar] [CrossRef]
- Roberts, S.C.; Willebrand, K.; Fredrick, J.; Pischel, L.; Patel, K.; Murray, T.S.; Martinello, R.A. Characterizing healthcare personnel attitudes toward receipt of a voluntary bivalent COVID-19 booster vaccine during a COVID-19 outbreak at a behavioral health hospital in Connecticut. Antimicrob. Steward. Healthc. Epidemiol. 2024, 4, e87. [Google Scholar] [CrossRef]
- Russ, S.; Myers, C.; Licherdell, E.; Bowden, A.; Chinchilli, E.; Dahhan, R.; Van Wijngaarden, E.; Plumb, I.D.; Dumyati, G. Sociodemographic and Occupational Characteristics Associated with Early and Continued COVID-19 Vaccine Uptake Among Healthcare Personnel: Monroe County, NY. Vaccine 2024, 42, 2585–2591. [Google Scholar] [CrossRef]
- Baghani, M.; Fathalizade, F.; Loghman, A.H.; Samieefar, N.; Ghobadinezhad, F.; Rashedi, R.; Baghsheikhi, H.; Sodeifian, F.; Rahimzadegan, M.; Akhlaghdoust, M. COVID-19 vaccine hesitancy worldwide and its associated factors: A systematic review and meta-analysis. Sci. One Health 2023, 2, 100048. [Google Scholar] [CrossRef]
- Dror, A.A.; Eisenbach, N.; Taiber, S.; Morozov, N.G.; Mizrachi, M.; Zigron, A.; Srouji, S.; Sela, E. Vaccine hesitancy: The next challenge in the fight against COVID-19. Eur. J. Epidemiol. 2020, 35, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Barello, S.; Nania, T.; Dellafiore, F.; Graffigna, G.; Caruso, R. ‘Vaccine hesitancy’ among university students in Italy during the COVID-19 pandemic. Eur. J. Epidemiol. 2020, 35, 781–783. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Coppeta, L.; Olesen, O.F. Vaccine Hesitancy among Healthcare Workers in Europe: A Systematic Review. Vaccines 2023, 11, 1657. [Google Scholar] [CrossRef]
- Dudley, M.Z.; Halsey, N.A.; Omer, S.B.; Orenstein, W.A.; T O’Leary, S.; Limaye, R.J.; Salmon, D.A. The state of vaccine safety science: Systematic reviews of the evidence. Lancet Infect. Dis. 2020, 20, e80–e89. [Google Scholar] [CrossRef]
- Lip, A.; Pateman, M.; Fullerton, M.M.; Chen, H.M.; Bailey, L.; Houle, S.; Davidson, S.; Constantinescu, C. Vaccine hesitancy educational tools for healthcare providers and trainees: A scoping review. Vaccine 2023, 41, 23–35. [Google Scholar] [CrossRef]
- Suliman, D.M.; Nawaz, F.A.; Mohanan, P.; Modber, M.; Musa, M.K.; Musa, M.B.; El Chbib, D.; Elhadi, Y.A.M.; Essar, M.Y.; Isa, M.A.; et al. UAE efforts in promoting COVID-19 vaccination and building vaccine confidence. Vaccine 2021, 39, 6341–6345. [Google Scholar] [CrossRef]
- Bianchi, F.P.; Stefanizzi, P.; Brescia, N.; Lattanzio, S.; Martinelli, A.; Tafuri, S. COVID-19 vaccination hesitancy in Italian healthcare workers: A systematic review and meta-analysis. Expert Rev. Vaccines 2022, 21, 1289–1300. [Google Scholar] [CrossRef]
- Hall, C.M.; Northam, H.; Webster, A.; Strickland, K. Determinants of seasonal influenza vaccination hesitancy among healthcare personnel: An integrative review. J. Clin. Nurs. 2022, 31, 2112–2124. [Google Scholar] [CrossRef]
- Alalawi, M.; Alsalloum, M.A.; Garwan, Y.M.; Abuzeid, M.; Alalawi, H.; Eljaaly, K.; Thabit, A.K.; Jose, J. COVID-19 vaccine hesitancy among healthcare workers in Arab Countries: A systematic review and meta-analysis. PLoS ONE 2024, 19, e0296432. [Google Scholar] [CrossRef]
- Mollalo, A.; Tatar, M. Spatial Modeling of COVID-19 Vaccine Hesitancy in the United States. Int. J. Environ. Res. Public Health 2021, 18, 9488. [Google Scholar] [CrossRef] [PubMed]
- Moradpour, J.; Shajarizadeh, A.; Carter, J.; Chit, A.; Grootendorst, P. The impact of national income and vaccine hesitancy on country-level COVID-19 vaccine uptake. PLoS ONE 2023, 18, e0293184. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, G.O.; Leland, R.; Emanuel, E.J. Making vaccines available to other countries before offering domestic booster vaccinations. JAMA 2021, 326, 903–904. [Google Scholar] [CrossRef]
- Diseases, T.L.I. COVID-19 vaccine equity and booster doses. Lancet. Infect. Dis. 2021, 21, 1193. [Google Scholar] [CrossRef]
- Tselebis, A.; Sikaras, C.; Milionis, C.; Sideri, E.P.; Fytsilis, K.; Papageorgiou, S.M.; Ilias, I.; Pachi, A. A Moderated Mediation Model of the Influence of Cynical Distrust, Medical Mistrust, and Anger on Vaccination Hesitancy in Nursing Staff. Eur. J. Investig. Health Psychol. Educ. 2023, 13, 2373–2387. [Google Scholar] [CrossRef]
- Galanis, P.; Moisoglou, I.; Vraka, I.; Siskou, O.; Konstantakopoulou, O.; Katsiroumpa, A.; Kaitelidou, D. Predictors of COVID-19 vaccine uptake in healthcare workers: A cross-sectional study in Greece. J. Occup. Environ. Med. 2022, 64, e191–e196. [Google Scholar] [CrossRef]
- Bell, S.; Clarke, R.M.; Ismail, S.A.; Ojo-Aromokudu, O.; Naqvi, H.; Coghill, Y.; Donovan, H.; Letley, L.; Paterson, P.; Mounier-Jack, S. COVID-19 vaccination beliefs, attitudes, and behaviours among health and social care workers in the UK: A mixed-methods study. PLoS ONE 2022, 17, e0260949. [Google Scholar] [CrossRef]
- Kigongo, E.; Kabunga, A.; Tumwesigye, R.; Musinguzi, M.; Izaruku, R.; Acup, W. Prevalence and predictors of COVID-19 vaccination hesitancy among healthcare workers in Sub-Saharan Africa: A systematic review and meta-analysis. PLoS ONE 2023, 18, e0289295. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y. Multilevel determinants of COVID-19 vaccination hesitancy in the United States: A rapid systematic review. Prev. Med. Rep. 2022, 25, 101673. [Google Scholar] [CrossRef]
- Bianchi, F.P.; Stefanizzi, P.; Cuscianna, E.; Riformato, G.; Di Lorenzo, A.; Giordano, P.; Germinario, C.A.; Tafuri, S. COVID-19 vaccination hesitancy among Italian parents: A systematic review and meta-analysis. Hum. Vaccin. Immunother. 2023, 19, 2171185. [Google Scholar] [CrossRef]
- Issaris, V.; Kalogerakos, G.; Milas, G.P. Vaccination Hesitancy Among Greek Orthodox Christians: Is There a Conflict Between Religion and Science? J. Relig. Health 2023, 62, 1373–1378. [Google Scholar] [CrossRef]
- Irrgang, P.; Gerling, J.; Kocher, K.; Lapuente, D.; Steininger, P.; Habenicht, K.; Wytopil, M.; Beileke, S.; Schäfer, S.; Zhong, J.; et al. Class switch toward noninflammatory, spike-specific IgG4 antibodies after repeated SARS-CoV-2 mRNA vaccination. Sci. Immunol. 2023, 8, eade2798. [Google Scholar] [CrossRef]
- Uversky, V.N.; Redwan, E.M.; Makis, W.; Rubio-Casillas, A. IgG4 Antibodies Induced by Repeated Vaccination May Generate Immune Tolerance to the SARS-CoV-2 Spike Protein. Vaccines 2023, 11, 991. [Google Scholar] [CrossRef]
- Stowe, J.; Miller, E.; Andrews, N.; Whitaker, H.J. Risk of myocarditis and pericarditis after a COVID-19 mRNA vaccine booster and after COVID-19 in those with and without prior SARS-CoV-2 infection: A self-controlled case series analysis in England. PLoS Med. 2023, 20, e1004245. [Google Scholar] [CrossRef]
- Abraham, N.; Spruin, S.; Rossi, T.; Fireman, B.; Zafack, J.; Blaser, C.; Shaw, A.; Hutchings, K.; Ogunnaike-Cooke, S. Myocarditis and/or pericarditis risk after mRNA COVID-19 vaccination: A Canadian head to head comparison of BNT162b2 and mRNA-1273 vaccines. Vaccine 2022, 40, 4663–4671. [Google Scholar] [CrossRef]
- Jain, N.; Chaudhary, P.; Shrivastava, A.; Kaur, T.; Kaur, S.; Brar, H.S.; Jindal, R. Thrombosis with Thrombocytopenia Syndrome (TTS) After ChAdOx1 nCoV-19 Immunization: An Investigative Case Report. Am. J. Case Rep. 2023, 24, e938878. [Google Scholar] [CrossRef]
- Islam, A.; Bashir, M.S.; Joyce, K.; Rashid, H.; Laher, I.; Elshazly, S. An Update on COVID-19 Vaccine Induced Thrombotic Thrombocytopenia Syndrome and Some Management Recommendations. Molecules 2021, 26, 5004. [Google Scholar] [CrossRef]
- Tran, H.A.; Deng, L.; Wood, N.; Choi, P.; Singleton, S.; Clarke, L.; Khanlari, S.; Maitland-Scott, I.; Bird, R.; Brown, S.; et al. The clinicopathological features of thrombosis with thrombocytopenia syndrome following ChAdOx1-S (AZD1222) vaccination and case outcomes in Australia: A population-based study. Lancet Reg. Health West. Pac. 2023, 40, 100894. [Google Scholar] [CrossRef]
- Lang, A.L.; Hohmuth, N.; Višković, V.; Konigorski, S.; Scholz, S.; Balzer, F.; Remschmidt, C.; Leistner, R. COVID-19 Vaccine Effectiveness and Digital Pandemic Surveillance in Germany (eCOV Study): Web Application-Based Prospective Observational Cohort Study. J. Med. Internet. Res. 2024, 26, e47070. [Google Scholar] [CrossRef]
- Gram, M.A.; Emborg, H.D.; Schelde, A.B.; Friis, N.U.; Nielsen, K.F.; Moustsen-Helms, I.R.; Legarth, R.; Lam, J.U.H.; Chaine, M.; Malik, A.Z.; et al. Vaccine effectiveness against SARS-CoV-2 infection or COVID-19 hospitalization with the Alpha, Delta, or Omicron SARS-CoV-2 variant: A nationwide Danish cohort study. PLoS Med. 2022, 19, e1003992. [Google Scholar] [CrossRef] [PubMed]
- Garrett, M.E.; Galloway, J.G.; Wolf, C.; Logue, J.K.; Franko, N.; Chu, H.Y.; Matsen, F.A.t.; Overbaugh, J.M. Comprehensive characterization of the antibody responses to SARS-CoV-2 Spike protein finds additional vaccine-induced epitopes beyond those for mild infection. Elife 2022, 11, e73490. [Google Scholar] [CrossRef] [PubMed]
- Katz, M.A.; Rojas Castro, M.Y.; Chakhunashvili, G.; Chitadze, N.; Ward, C.L.; McKnight, C.J.; Lucaccioni, H.; Finci, I.; Zardiashvili, T.; Pebody, R.; et al. Primary series COVID-19 vaccine effectiveness among health care workers in the country of Georgia, March–December 2021. PLoS ONE 2024, 19, e0307805. [Google Scholar] [CrossRef]
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K.; et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Haque, A.; Pant, A.B. Mitigating Covid-19 in the face of emerging virus variants, breakthrough infections and vaccine hesitancy. J. Autoimmun. 2022, 127, 102792. [Google Scholar] [CrossRef]
- Graña, C.; Ghosn, L.; Evrenoglou, T.; Jarde, A.; Minozzi, S.; Bergman, H.; Buckley, B.S.; Probyn, K.; Villanueva, G.; Henschke, N.; et al. Efficacy and safety of COVID-19 vaccines. Cochrane Database Syst. Rev. 2022, 12, Cd015477. [Google Scholar] [CrossRef]
- De Waele, A.; Hendrickx, G.; Valckx, S.; Domínguez, À.; Toledo, D.; Castilla, J.; Tuells, J.; Van Damme, P. The Vaccine Training Barometer: Assessing healthcare providers’ confidence to answer vaccine-related questions and their training needs. Vaccine 2024, 42, 2421–2428. [Google Scholar] [CrossRef]
- Paterson, P.; Meurice, F.; Stanberry, L.R.; Glismann, S.; Rosenthal, S.L.; Larson, H.J. Vaccine hesitancy and healthcare providers. Vaccine 2016, 34, 6700–6706. [Google Scholar] [CrossRef]
- Moghnieh, R.; Haddad, W.; Jbeily, N.; El-Hassan, S.; Eid, S.; Baba, H.; Sily, M.; Saber, Y.; Abdallah, D.; Bizri, A.R.; et al. Immunogenicity and real-world effectiveness of COVID-19 vaccines in Lebanon: Insights from primary and booster schemes, variants, infections, and hospitalization. PLoS ONE 2024, 19, e0306457. [Google Scholar] [CrossRef]
- Barosa, M.; Ioannidis, J.P.A.; Prasad, V. Evidence base for yearly respiratory virus vaccines: Current status and proposed improved strategies. Eur. J. Clin. Investig. 2024, 54, e14286. [Google Scholar] [CrossRef] [PubMed]
- Nusbaum, N.J. The COVID Vaccination Hesitancy Epidemic. J. Community Health 2024, 49, 377–378. [Google Scholar] [CrossRef]
- Diekema, D.S. Responding to parental refusals of immunization of children. Pediatrics 2005, 115, 1428–1431. [Google Scholar] [CrossRef]
- Alya, W.A.; Maraqa, B.; Nazzal, Z.; Odeh, M.; Makhalfa, R.; Nassif, A.; Aabed, M. COVID-19 vaccine uptake and its associated factors among Palestinian healthcare workers: Expectations beaten by reality. Vaccine 2022, 40, 3713–3719. [Google Scholar] [CrossRef]
- Zintel, S.; Flock, C.; Arbogast, A.L.; Forster, A.; von Wagner, C.; Sieverding, M. Gender differences in the intention to get vaccinated against COVID-19: A systematic review and meta-analysis. J. Public Health 2023, 31, 1303–1327. [Google Scholar] [CrossRef]
- Nachtigall, I.; Bonsignore, M.; Hohenstein, S.; Bollmann, A.; Günther, R.; Kodde, C.; Englisch, M.; Ahmad-Nejad, P.; Schröder, A.; Glenz, C.; et al. Effect of gender, age and vaccine on reactogenicity and incapacity to work after COVID-19 vaccination: A survey among health care workers. BMC Infect. Dis. 2022, 22, 291. [Google Scholar] [CrossRef]
- Di Resta, C.; Ferrari, D.; Viganò, M.; Moro, M.; Sabetta, E.; Minerva, M.; Ambrosio, A.; Locatelli, M.; Tomaiuolo, R. The Gender Impact Assessment among Healthcare Workers in the SARS-CoV-2 Vaccination—An Analysis of Serological Response and Side Effects. Vaccines 2021, 9, 522. [Google Scholar] [CrossRef] [PubMed]
- Sallam, M.; Dababseh, D.; Eid, H.; Al-Mahzoum, K.; Al-Haidar, A.; Taim, D.; Yaseen, A.; Ababneh, N.A.; Bakri, F.G.; Mahafzah, A. High rates of COVID-19 vaccine hesitancy and its association with conspiracy beliefs: A study in Jordan and Kuwait among other Arab countries. Vaccines 2021, 9, 42. [Google Scholar] [CrossRef]
- She, J.; Hou, D.; Chen, C.; Bi, J.; Song, Y. Challenges of vaccination and herd immunity in COVID-19 and management strategies. Clin. Respir. J. 2022, 16, 708–716. [Google Scholar] [CrossRef]
- Dassarma, B.; Tripathy, S.; Chabalala, M.; Matsabisa, M.G. Challenges in Establishing Vaccine Induced Herd Immunity through Age Specific Community Vaccinations. Aging Dis. 2022, 13, 29–36. [Google Scholar] [CrossRef]
- Goldberg, A.R.; Langwig, K.E.; Brown, K.L.; Marano, J.M.; Rai, P.; King, K.M.; Sharp, A.K.; Ceci, A.; Kailing, C.D.; Kailing, M.J.; et al. Widespread exposure to SARS-CoV-2 in wildlife communities. Nat. Commun. 2024, 15, 6210. [Google Scholar] [CrossRef]
- Delahay, R.J.; de la Fuente, J.; Smith, G.C.; Sharun, K.; Snary, E.L.; Flores Girón, L.; Nziza, J.; Fooks, A.R.; Brookes, S.M.; Lean, F.Z.X.; et al. Assessing the risks of SARS-CoV-2 in wildlife. One Health Outlook 2021, 3, 7. [Google Scholar] [CrossRef]
- Valencak, T.G.; Csiszar, A.; Szalai, G.; Podlutsky, A.; Tarantini, S.; Fazekas-Pongor, V.; Papp, M.; Ungvari, Z. Animal reservoirs of SARS-CoV-2: Calculable COVID-19 risk for older adults from animal to human transmission. Geroscience 2021, 43, 2305–2320. [Google Scholar] [CrossRef]
- Shaheen, M.N.F. The concept of one health applied to the problem of zoonotic diseases. Rev. Med. Virol. 2022, 32, e2326. [Google Scholar] [CrossRef]
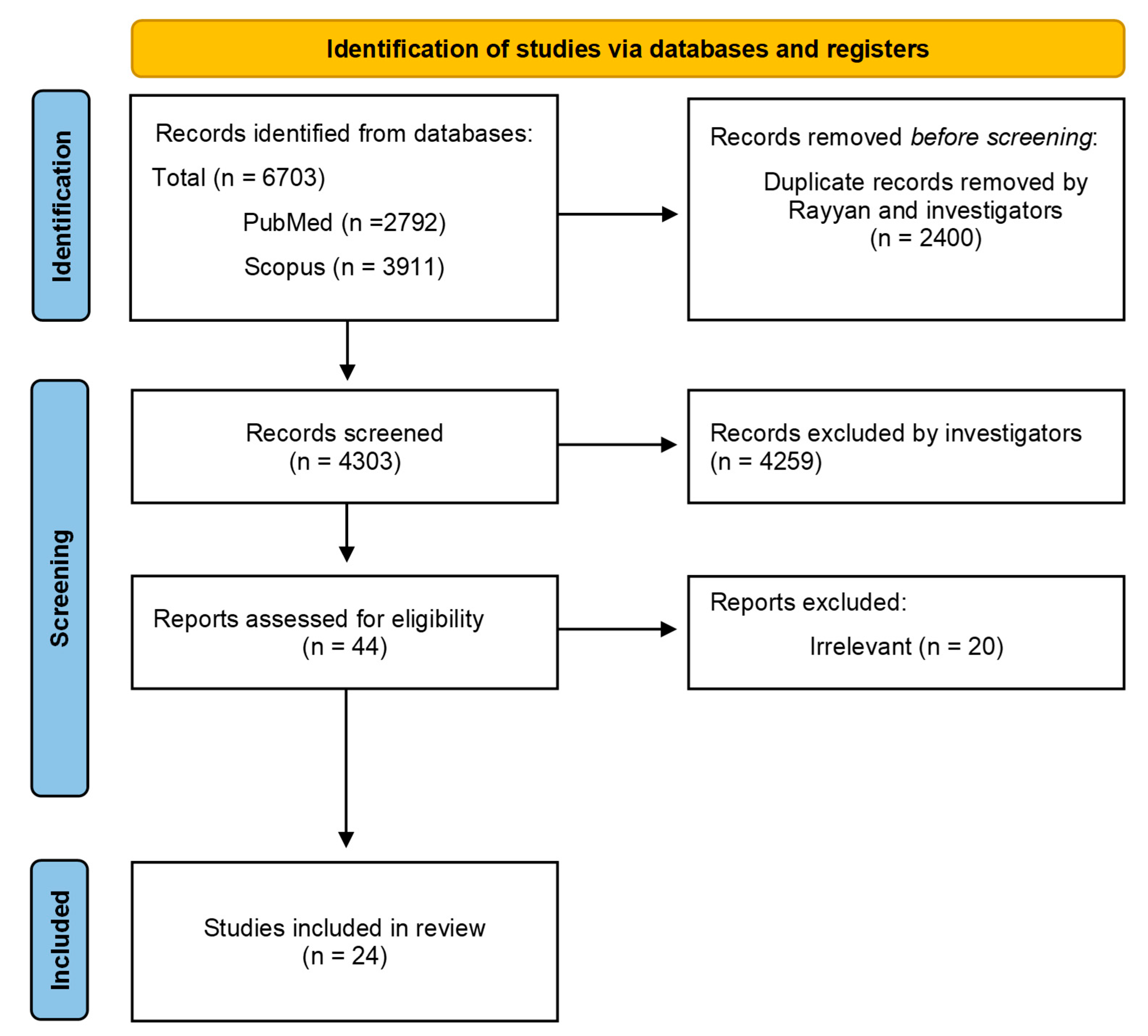
| Author/Year (Ref.) | Study Type | Country | No of Participants | Vaccination History (%) Number of BDs Received | Vaccine Hesitancy (%) | Hesitancy Among HCPs by Specialty |
|---|---|---|---|---|---|---|
| Arshad et al., 2022 [40] | Quantitative | Pakistan | n = 1164 | 9.9% at least one BD | 47.9% | 24.4% Medical professionals 23.7% Allied Health Professionals |
| Dale et al., 2023 [41] | Quantitative | England | n = 91 | 77.1% at least one BD 4.9% 2 BDs 1.2% 3 BDs | Oxford Vaccine hesitancy scale score: 13.56/35.00 (17.1% being above the midpoint). | NR |
| Della Polla et al., 2022 [42] | Quantitative | Italy | n = 496 | 94.9% at least one BD 48.1% at least 2 BDs | 47.4% | NR |
| Digregorio et al., 2024 [57] | Quantitative | Belgium | n = 1814 | 66.8% 2 BDs 6.3% 3 BDs | 14% | NR |
| Dudley et al., 2023 [43] | Quantitative | USA | n = 1207 | 82% HCPs at least one BD | NR | 6.3% Pediatrician 13% Family medicine 26.7% Physician assistant, Nurse Practitioners, and Nurse 26% Pharmacist |
| Galanis et al., 2023 [44] | Quantitative | Greece | n = 795 | NR | 30.9% | NR |
| George et al., 2023 [45] | Mixed-Methods | South Africa | n = 6235 | 56% at least one BD | 27% | 27.9% Nurse 17.5% Doctor |
| Gu et al., 2023 [46] | Quantitative | China | n = 1618 | 78.4% at least one BD | 41.8% | 43.7% Physician 37% Nurse |
| Guarducci et al., 2023 [47] | Quantitative | Italy | n = 1309 | 96.5% one BD | NR | NR |
| Kolomba et al., 2024 [58] | Quantitative | Congo | n = 514 | 24.3% one BD | 31.1% | 23.1% Doctor |
| Krishna et al., 2023 [48] | Quantitative | India | n = 535 | 62.2% one BD | 40% | NR |
| Lubad et al., 2023 [49] | Quantitative | Jordan | n = 300 | NR | 31.6% | NR |
| Maraga et al., 2024 [22] | Quantitative | Palestine | n = 919 | NR | 66.5% | NR |
| Pandarathodiyil et al., 2024 [59] | Quantitative | Malaysa | n = 392 | 100% at least one BD | 22% | NR |
| Paudel et al., 2023 [50] | Quantitative | Nepal | n = 300 | 29% one BD | 12% | NR |
| Pristov al., 2024 [60] | Quantitative | Slovenia | n = 560 | 50.9% at least one BD | NR | NR |
| Ramot et al., 2023 [51] | Quantitative | Israel | n = 124 | 88.7% one BD 38.7% 2 BDs | 61.3% | NR |
| Rathinakumar et al., 2024 [52] | Quantitative | South India | n = 572 | 12.6% one BD | 19.7% | 23.2% Paramedical workers 12.8% Doctor |
| Roberts et al. 2024 [61] | Quantitative | USA | n = 182 | 55% one BD | NR | NR |
| Russ et al., 2024 [62] | Quantitative | USA | n = 3375 | 85% one BD | NR | NR |
| Salah et al., 2023 [53] | Quantitative | Kingdom of Bahrain and Egypt | n = 389 | NR | 46.1% | 46.1% Physicians 26% Nurses 34.6% Pharmacists |
| Sansone et al., 2024 [54] | Quantitative | Italy | n = 521 | 5.2% at least one BD | 60.2% | NR |
| Viskupič et al., 2023 [55] | Quantitative | USA | n = 1084 | 63.2% one BD | NR | NR |
| Zoumpoulis et al., 2023 [56] | Quantitative | Greece | n = 1224 | 52.4% one BD 47.5% 2 BDs | 27.4% | NR |
| Factors | Associated with Hesitancy | Number of Studies |
|---|---|---|
| Age | Younger age [39,41,49,50,51,54,56]. | 7 |
| Gender | Being male [34,39]. Being female [43,45,47,48,51,54]. | 8 |
| Race | Black African [39]. Black [55]. Non-Hispanic Black [56]. Hispanic [56]. | 3 |
| Education | Lower education [38,44,56]. | 3 |
| Occupation | Non-prescribers [51]. Other than physicians [37,38,39,43,44,46,48,52]. Physicians [41,45,47]. Pharmacists [47]. Νot in direct contact with patients [39]. Less job experience [39]. Working experience more than 5 years [52]. Wards of activity with lower risk of infection (Medical vs. Emergency/Critical/Infectious Disease wards) [48]. | 12 |
| Political Leanings | Republican self-identification [49]. | 1 |
| Marital Status | Single/not married [34,46]. Married [38]. | 3 |
| Friends/family | Influence of friends/family [35,38,42,52]. | 4 |
| Area of Residence | Rural [34,37]. Highly socially vulnerable census tract [56]. | 3 |
| Low income | Low annual household income (<USD 50,000) [56]. | 1 |
| Comorbidity/chronic illness | Absence of chronic conditions [38,39,52,54]. Permanent or temporary medical conditions [37]. History of allergy [40]. Obesity [47]. | 7 |
| Health status | Good/very good self-perceived physical health [38]. Unhealthy dietary habits [54]. | 2 |
| Time constraints | Lack of time [35] | 1 |
| Flu Vaccination | Lack of flu vaccination [17,38,39,49,50]. | 5 |
| Hygiene measures | Increased compliance [38,41,52]. | 3 |
| Relating to COVID-19 | Previous COVID-19 infection [34,35,49]. No previous infection [38]. | 4 |
| Relating to COVID-19 vaccination | No previous vaccination [34] Type of vaccine (non-mRNA COVID-19 vaccines) [34,53] Less previous vaccine doses [44,48,51,52] Uptake of the first booster dose [42] Previous side effects [34] | 10 |
| Knowledge/Attitudes | Associated with Hesitancy | Number of Studies |
|---|---|---|
| Trust-related issues | Vaccine safety [34,35,38,39,40,41,42,43,45,47,52]. | 11 |
| Pregnancy safety [35]. | ||
| Vaccine effectiveness [17,34,36,38,39,40,41,42,43,45,46,47,51,52,53,55]. | 16 | |
| Vaccine necessity [35,36,38,39,40,45,46,47,52,55]. | 10 | |
| Vaccine Side effects [17,36,37,46,50,52,55]. | 7 | |
| Mistrust in government/scientists [37,42,43,50]. | 4 | |
| Rapid development of the vaccines [37,42,50]. | 3 | |
| Distrust due to racism and previous unethical treatment of minorities [37]. | 1 | |
| Reliability of clinical trials (not including HCPs) [37]. | 1 | |
| Low trust and satisfaction in COVID-19 vaccination [34,38,51]. | 3 | |
| Wanting to wait more [37,42,43,45]. | 4 | |
| Information | Lack of information/misinformation [42,47,48]. | 3 |
| Beliefs and attitudes about health and prevention | Low risk of COVID-19 infection [36,37,40,50,51]. | 5 |
| Immune system capable of fighting COVID-19 [43,51]. | 1 | |
| Lower perception of the severity of COVID-19 [36,41,51,52]. | 4 | |
| Against vaccines in general [50,52]. | 2 | |
| Belief in greater efficacy of complementary alternative medicine [50]. | 1 | |
| Ethics | Mandatory Vaccination [39,41,48]. | 3 |
| Other | Not being very likely to suggest the vaccine to patients [51]. | 1 |
| Tiredness due to the vaccination procedure [38]. | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christodoulakis, A.; Bouloukaki, I.; Aravantinou-Karlatou, A.; Zografakis-Sfakianakis, M.; Tsiligianni, I. Vaccine Hesitancy and Associated Factors Amongst Health Professionals: A Scoping Review of the Published Literature. Vaccines 2024, 12, 1411. https://doi.org/10.3390/vaccines12121411
Christodoulakis A, Bouloukaki I, Aravantinou-Karlatou A, Zografakis-Sfakianakis M, Tsiligianni I. Vaccine Hesitancy and Associated Factors Amongst Health Professionals: A Scoping Review of the Published Literature. Vaccines. 2024; 12(12):1411. https://doi.org/10.3390/vaccines12121411
Chicago/Turabian StyleChristodoulakis, Antonios, Izolde Bouloukaki, Antonia Aravantinou-Karlatou, Michail Zografakis-Sfakianakis, and Ioanna Tsiligianni. 2024. "Vaccine Hesitancy and Associated Factors Amongst Health Professionals: A Scoping Review of the Published Literature" Vaccines 12, no. 12: 1411. https://doi.org/10.3390/vaccines12121411
APA StyleChristodoulakis, A., Bouloukaki, I., Aravantinou-Karlatou, A., Zografakis-Sfakianakis, M., & Tsiligianni, I. (2024). Vaccine Hesitancy and Associated Factors Amongst Health Professionals: A Scoping Review of the Published Literature. Vaccines, 12(12), 1411. https://doi.org/10.3390/vaccines12121411










