Differences in the Evolution of Clinical, Biochemical, and Hematological Indicators in Hospitalized Patients with COVID-19 According to Their Vaccination Scheme: A Cohort Study in One of the World’s Highest Hospital Mortality Populations
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Measures and Follow-Up
2.3. Sample Size
2.4. Statistical Analysis
3. Results
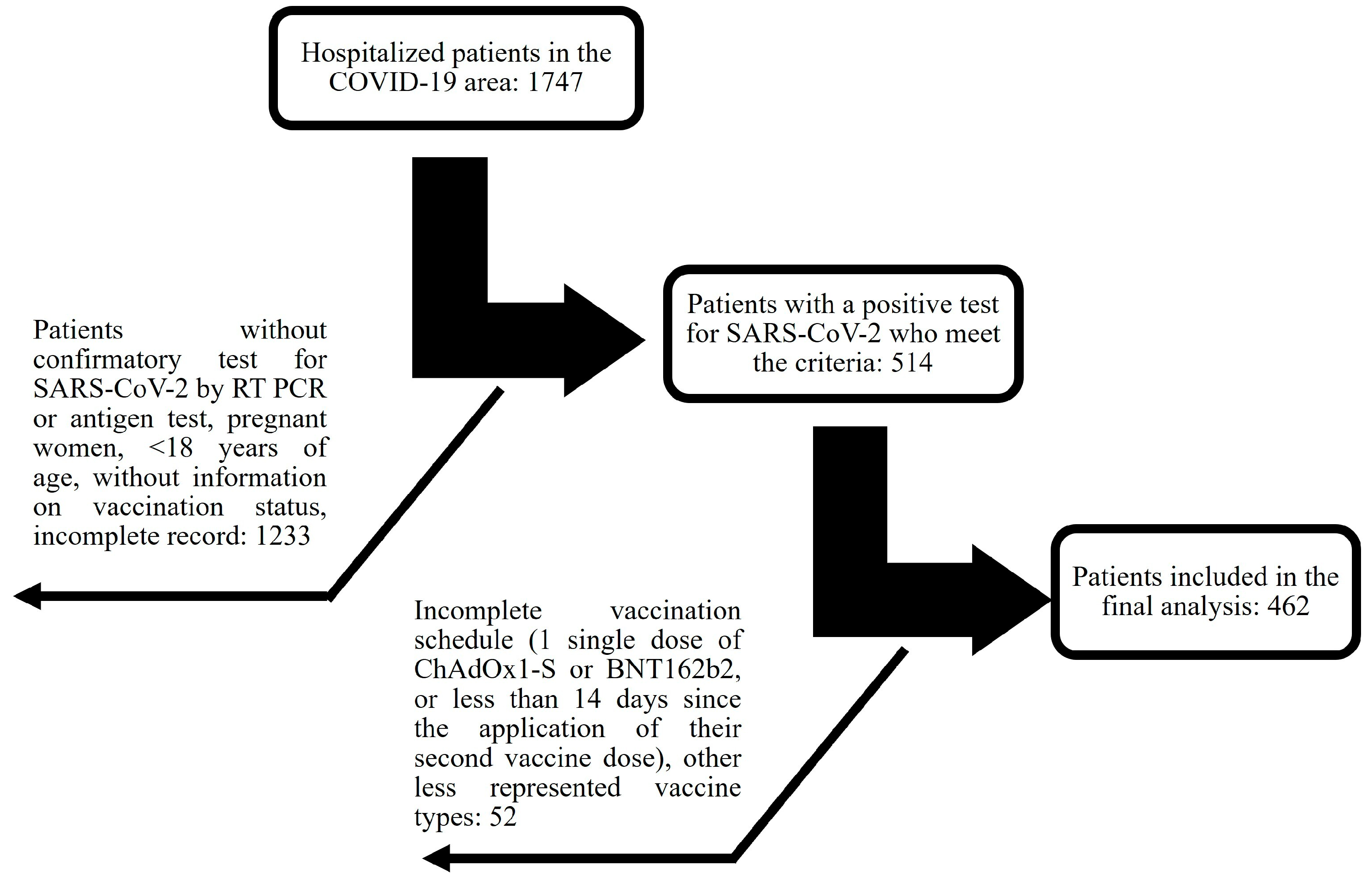
3.1. Patient Characteristics
3.2. Differences in Clinical Characteristics between Vaccinated and Unvaccinated Individuals
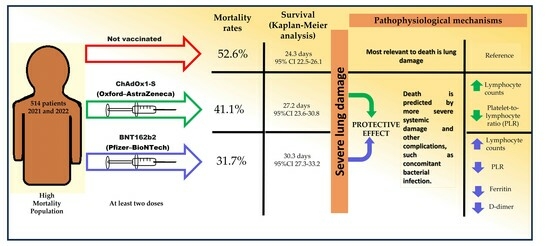
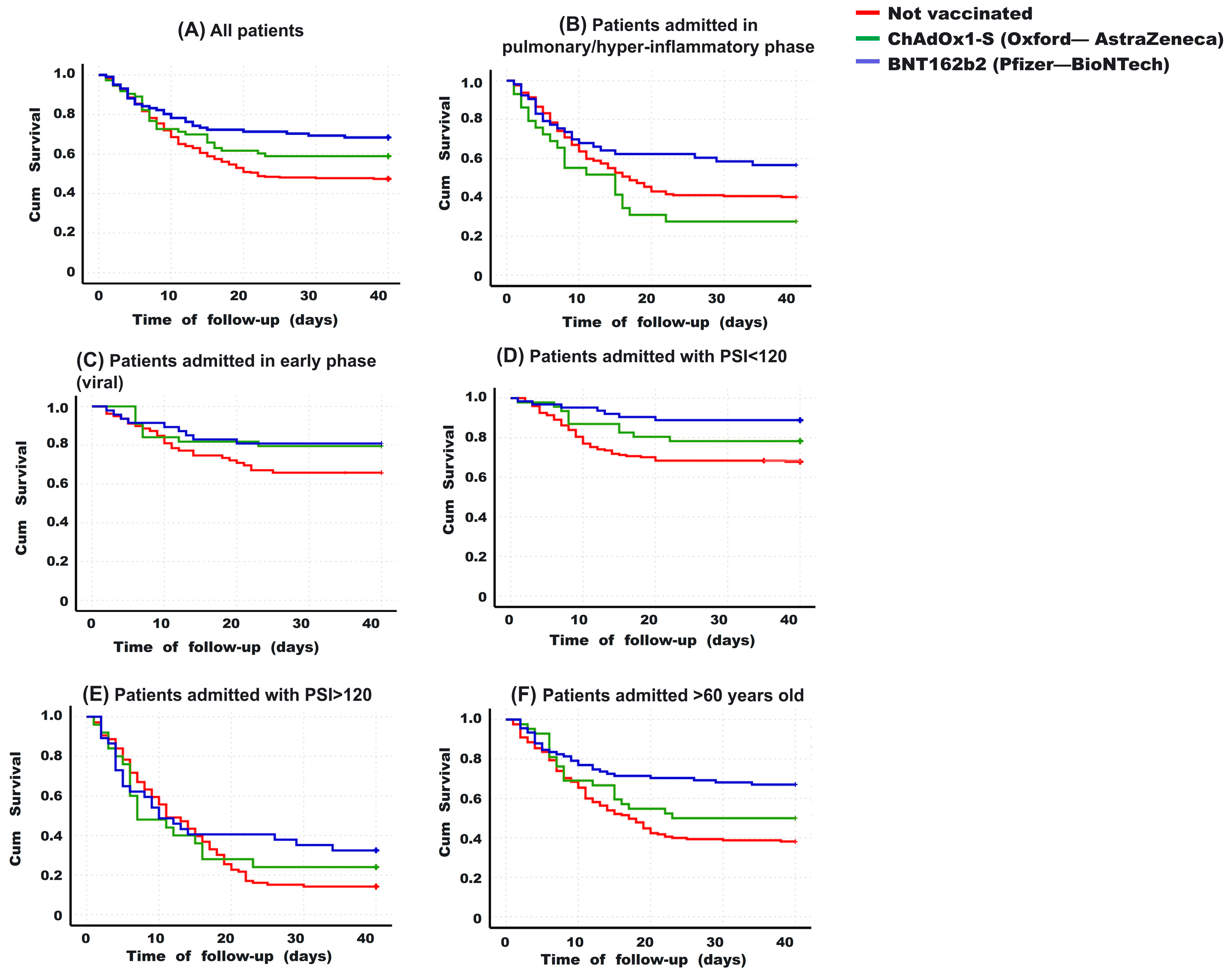
3.3. Hospitalization Days and Survival
3.4. Risk Factors for Death in Hospitalized Patients with COVID-19, including Vaccination Schedule
3.5. Clinical Differences between Those Vaccinated with BNT162b2 and ChAdOx1-S
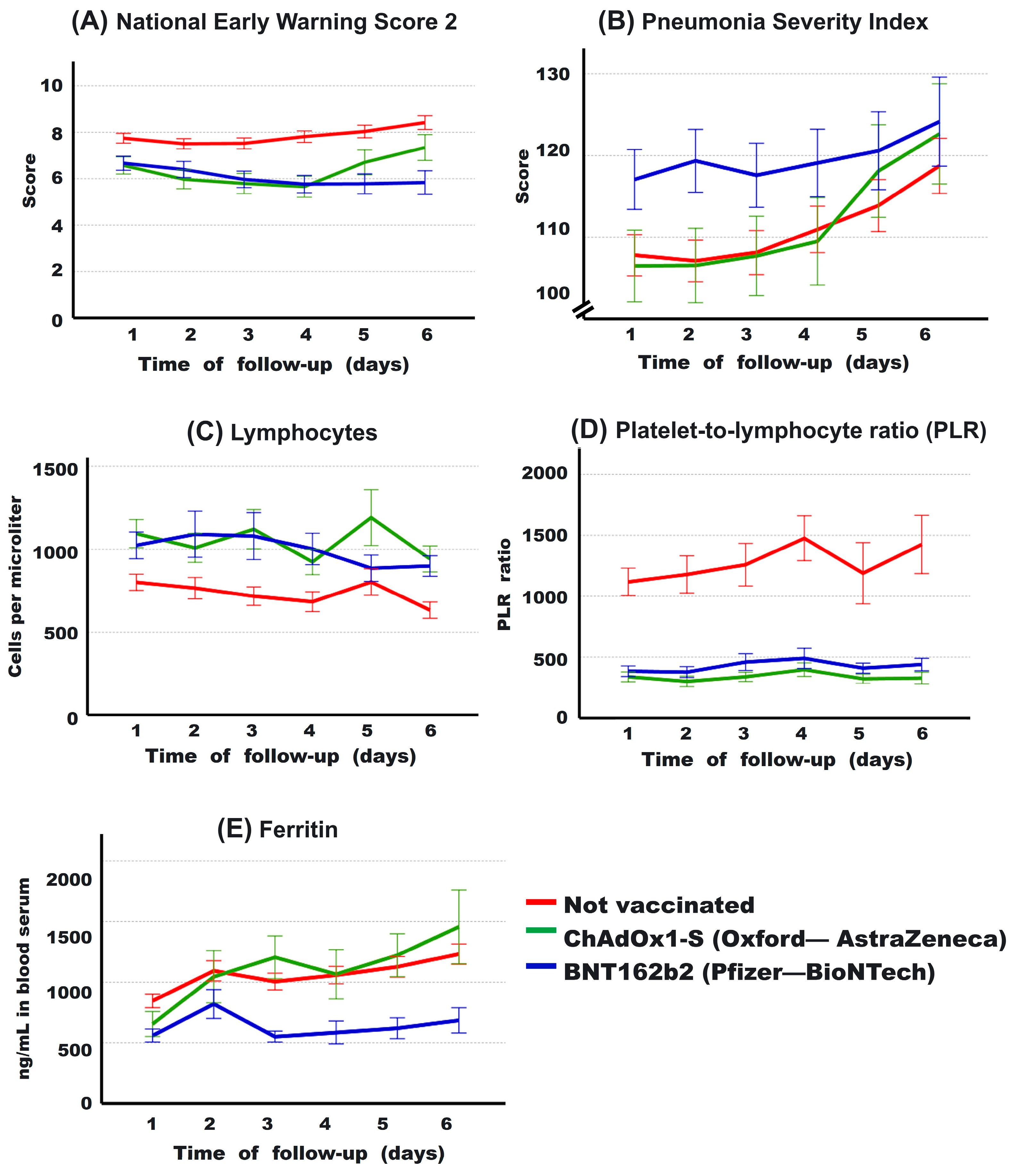
3.6. Evolution of Some Clinical Parameters during the First 6 Days
3.7. Predictors of Death Based on Vaccination Status
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rahmani, K.; Shavaleh, R.; Forouhi, M.; Disfani, H.F.; Kamandi, M.; Oskooi, R.K.; Foogerdi, M.; Soltani, M.; Rahchamani, M.; Mohaddespour, M.; et al. The Effectiveness of COVID-19 Vaccines in Reducing the Incidence, Hospitalization, and Mortality from COVID-19: A Systematic Review and Meta-Analysis. Front. Public Health 2022, 10, 873596. [Google Scholar] [PubMed]
- Guzman-Esquivel, J.; Mendoza-Hernandez, M.A.; Guzman-Solorzano, H.P.; Sarmiento-Hernandez, K.A.; Rodriguez-Sanchez, I.P.; Martinez-Fierro, M.L.; Paz-Michel, B.A.; Murillo-Zamora, E.; Rojas-Larios, F.; Lugo-Trampe, A.; et al. Clinical Characteristics in the Acute Phase of COVID-19 That Predict Long COVID: Tachycardia, Myalgias, Severity, and Use of Antibiotics as Main Risk Factors, While Education and Blood Group B Are Protective. Healthcare 2023, 11, 197. [Google Scholar] [CrossRef] [PubMed]
- Mascellino, M.T.; Di Timoteo, F.; De Angelis, M.; Oliva, A. Overview of the Main Anti-SARS-CoV-2 Vaccines: Mechanism of Action, Efficacy and Safety. Infect. Drug Resist. 2021, 14, 3459–3476. [Google Scholar] [CrossRef] [PubMed]
- Havers, F.P.; Pham, H.; Taylor, C.A.; Whitaker, M.; Patel, K.; Anglin, O.; Kambhampati, A.K.; Milucky, J.; Zell, E.; Moline, H.L.; et al. COVID-19-Associated Hospitalizations among Vaccinated and Unvaccinated Adults 18 Years or Older in 13 US States, January 2021 to April 2022. JAMA Intern. Med. 2022, 182, 1071. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Chang, X.; He, Y.; Tan, K.J.K. The Determinants of COVID-19 Morbidity and Mortality across Countries. Sci. Rep. 2022, 12, 5888. [Google Scholar] [CrossRef] [PubMed]
- Ascencio-Montiel, I.d.J.; Ovalle-Luna, O.D.; Rascón-Pacheco, R.A.; Borja-Aburto, V.H.; Chowell, G. Comparative Epidemiology of Five Waves of COVID-19 in Mexico, March 2020–August 2022. BMC Infect. Dis. 2022, 22, 813. [Google Scholar] [CrossRef]
- Huespe, I.A.; Ferraris, A.; Lalueza, A.; Valdez, P.R.; Peroni, M.L.; Cayetti, L.A.; Mirofsky, M.A.; Boietti, B.; Gómez-Huelgas, R.; Casas-Rojo, J.M.; et al. COVID-19 Vaccines Reduce Mortality in Hospitalized Patients with Oxygen Requirements: Differences between Vaccine Subtypes. A Multicontinental Cohort Study. J. Med. Virol. 2023, 95, e28786. [Google Scholar] [CrossRef]
- Busic, N.; Lucijanic, T.; Barsic, B.; Luksic, I.; Busic, I.; Kurdija, G.; Barbic, L.; Kunstek, S.; Jelic, T.; Lucijanic, M. Vaccination Provides Protection from Respiratory Deterioration and Death among Hospitalized COVID-19 Patients: Differences between Vector and MRNA Vaccines. J. Med. Virol. 2022, 94, 2849–2854. [Google Scholar] [CrossRef]
- Tandon, P.; Leibner, E.S.; Hackett, A.; Maguire, K.; Mashriqi, N.; Kohli-Seth, R. The Third Wave: Comparing Seasonal Trends in COVID-19 Patient Data at a Large Hospital System in New York City. Crit. Care Explor. 2022, 4, e0653. [Google Scholar] [CrossRef]
- Brosh-Nissimov, T.; Orenbuch-Harroch, E.; Chowers, M.; Elbaz, M.; Nesher, L.; Stein, M.; Maor, Y.; Cohen, R.; Hussein, K.; Weinberger, M.; et al. BNT162b2 Vaccine Breakthrough: Clinical Characteristics of 152 Fully Vaccinated Hospitalized COVID-19 Patients in Israel. Clin. Microbiol. Infect. 2021, 27, 1652–1657. [Google Scholar] [CrossRef]
- Vassallo, M.; Clement, N.; Lotte, L.; Manni, S.; Sindt, A.; Bertrand, P.M.; Durant, J. Prevalence and Main Clinical Characteristics of Fully Vaccinated Patients Admitted to Hospital for Delta Variant COVID-19. Front. Med. 2022, 9, 809154. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Pazgan-Simon, M.; Simon, K.; Łapiński, T.; Zarębska-Michaluk, D.; Szczepańska, B.; Chojnicki, M.; Mozer-Lisewska, I.; Flisiak, R. Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine. Vaccines 2021, 9, 781. [Google Scholar] [CrossRef] [PubMed]
- Abate, S.M.; Checkol, Y.A.; Mantefardo, B. Global Prevalence and Determinants of Mortality among Patients with COVID-19: A Systematic Review and Meta-Analysis. Ann. Med. Surg. 2021, 64, 102204. [Google Scholar] [CrossRef] [PubMed]
- Gray, W.K.; Navaratnam, A.V.; Day, J.; Wendon, J.; Briggs, T.W.R. COVID-19 Hospital Activity and in-Hospital Mortality during the First and Second Waves of the Pandemic in England: An Observational Study. Thorax 2022, 77, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- National Center for Health Statistics (NCHS); Centers for Disease Control and Prevention (CDC). Glossary, National Health Interview Survey. Available online: https://www.cdc.gov/nchs/nhis/tobacco/tobacco_glossary.htm (accessed on 1 November 2023).
- Romagnoli, S.; Peris, A.; De Gaudio, A.R.; Geppetti, P. SARS-CoV-2 and COVID-19: From the Bench to the Bedside. Physiol. Rev. 2020, 100, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Ku, E.; Amaral, S.; McCulloch, C.E.; Adey, D.B.; Li, L.; Johansen, K.L. Comparison of 2021 CKD-EPI Equations for Estimating Racial Differences in Preemptive Waitlisting for Kidney Transplantation. Clin. J. Am. Soc. Nephrol. 2022, 17, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Bahl, A.; Johnson, S.; Chen, N.-W. Timing of Corticosteroids Impacts Mortality in Hospitalized COVID-19 Patients. Intern. Emerg. Med. 2021, 16, 1593–1603. [Google Scholar] [CrossRef]
- Guzman-Esquivel, J.; Diaz-Martinez, J.; Ortega-Ortiz, G.J.; Murillo-Zamora, E.; Melnikov, V.; Tejeda-Luna, R.H.; Cosio-Medina, G.V.; Llerenas-Aguirre, I.K.; Guzman-Solorzano, A.J.; Hernandez-Fuentes, A.G.; et al. Interactions between the Principal Risk Factors for Reduction of the EGFR in Unvaccinated COVID-19 Survivors: Normal Pre-COVID-19 EGFR, Not Having Diabetes and Being Hospitalized. Exp. Ther. Med. 2023, 26, 580. [Google Scholar] [CrossRef]
- Baum, M.K.; Tamargo, J.A.; Diaz-Martinez, J.; Delgado-Enciso, I.; Meade, C.S.; Kirk, G.D.; Mehta, S.H.; Moore, R.; Kipke, M.D.; Shoptaw, S.J.; et al. HIV, Psychological Resilience, and Substance Misuse during the COVID-19 Pandemic: A Multi-Cohort Study. Drug Alcohol Depend. 2022, 231, 109230. [Google Scholar] [CrossRef]
- Jaffa, M.A.; Gebregziabher, M.; Luttrell, D.K.; Luttrell, L.M.; Jaffa, A.A. Multivariate Generalized Linear Mixed Models with Random Intercepts to Analyze Cardiovascular Risk Markers in Type-1 Diabetic Patients. J. Appl. Stat. 2016, 43, 1447–1464. [Google Scholar] [CrossRef]
- Naimi, A.I.; Whitcomb, B.W. Estimating Risk Ratios and Risk Differences Using Regression. Am. J. Epidemiol. 2020, 189, 508–510. [Google Scholar] [CrossRef] [PubMed]
- Gobierno de Mexico. Política Nacional de Vacunación Contra El Virus SARS-CoV-2, Para La Prevención de La COVID-19 En Mexico. Available online: https://coronavirus.gob.mx/wp-content/uploads/2021/04/28Abr2021_13h00_PNVx_COVID_19.pdf (accessed on 30 September 2023).
- Loza, A.; Wong-Chew, R.M.; Jiménez-Corona, M.-E.; Zárate, S.; López, S.; Ciria, R.; Palomares, D.; García-López, R.; Iša, P.; Taboada, B.; et al. Two-Year Follow-up of the COVID-19 Pandemic in Mexico. Front. Public Health 2023, 10, 1050673. [Google Scholar] [CrossRef] [PubMed]
- Zárate, S.; Taboada, B.; Rosales-Rivera, M.; García-López, R.; Muñoz-Medina, J.E.; Sanchez-Flores, A.; Herrera-Estrella, A.; Gómez-Gil, B.; Selem Mojica, N.; Salas-Lais, A.G.; et al. Omicron-BA.1 Dispersion Rates in Mexico Varied According to the Regional Epidemic Patterns and the Diversity of Local Delta Subvariants. Viruses 2023, 15, 243. [Google Scholar] [CrossRef] [PubMed]
- Ulises Pérez-Arenas, L.; Paola Plascencia-Roldán, J.; Irazú Morales-Reyes, G.; Flores-Vargas, G.; de Jesús Gallardo-Luna, M.; Navarro-Olivos, E.; Javier Magos-Vázquez, F.; Padilla-Raygoza, N. Variants of SARS-CoV-2 and COVID-19 in Mexico. In New COVID-19 Variants [Working Title]; IntechOpen: Rijeka, Croatia, 2023. [Google Scholar]
- Farrell, J.M.; Zhao, C.Y.; Tarquinio, K.M.; Brown, S.P. Causes and Consequences of COVID-19-Associated Bacterial Infections. Front. Microbiol. 2021, 12, 682571. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Lu, J.; Liu, D.; He, Y. Characteristics and Risk Factors of Secondary Bacterial Infections in COVID-19 Patients. Antimicrob. Steward. Healthc. Epidemiol. 2023, 3, e156. [Google Scholar] [CrossRef] [PubMed]
- Dietz, L.L.; Juhl, A.K.; Søgaard, O.S.; Reekie, J.; Nielsen, H.; Johansen, I.S.; Benfield, T.; Wiese, L.; Stærke, N.B.; Jensen, T.Ø.; et al. Impact of Age and Comorbidities on SARS-CoV-2 Vaccine-Induced T Cell Immunity. Commun. Med. 2023, 3, 58. [Google Scholar] [CrossRef] [PubMed]
- Montes-Galindo, D.A.; Espiritu-Mojarro, A.C.; Melnikov, V.; Moy-López, N.A.; Soriano-Hernandez, A.D.; Galvan-Salazar, H.R.; Guzman-Muñiz, J.; Guzman-Esquivel, J.; Martinez-Fierro, M.L.; Rodriguez-Sanchez, I.P.; et al. Adenovirus 5 Produces Obesity and Adverse Metabolic, Morphological, and Functional Changes in the Long Term in Animals Fed a Balanced Diet or a High-Fat Diet: A Study on Hamsters. Arch. Virol. 2019, 164, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Voss, J.D.; Atkinson, R.L.; Dhurandhar, N.V. Role of Adenoviruses in Obesity. Rev. Med. Virol. 2015, 25, 379–387. [Google Scholar] [CrossRef]
- Banoun, H. MRNA: Vaccine or Gene Therapy? The Safety Regulatory Issues. Int. J. Mol. Sci. 2023, 24, 10514. [Google Scholar] [CrossRef]
- Zsengellér, Z.K.; Wert, S.E.; Bachurski, C.J.; Kirwin, K.L.; Trapnell, B.C.; Whitsett, J.A. Recombinant Adenoviral Vector Disrupts Surfactant Homeostasis in Mouse Lung. Hum. Gene Ther. 1997, 8, 1331–1344. [Google Scholar] [CrossRef]
- Tsilingiris, D.; Vallianou, N.G.; Karampela, I.; Muscogiuri, G.; Dalamaga, M. Use of Adenovirus Type-5 Vector Vaccines in COVID-19: Potential Implications for Metabolic Health? Minerva Endocrinol. 2022, 47, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Flotte, T.R.; Gao, G. Immune Responses to Recombinant Adenoviruses As Gene Therapy Vectors and COVID-19 Vaccines: A Two-Edged Sword. Hum. Gene Ther. 2021, 32, 645–646. [Google Scholar] [CrossRef] [PubMed]
- Rosero-Bixby, L. The Effectiveness of Pfizer-BioNTech and Oxford-AstraZeneca Vaccines to Prevent Severe COVID-19 in Costa Rica: Nationwide, Ecological Study of Hospitalization Prevalence. JMIR Public Health Surveill. 2022, 8, e35054. [Google Scholar] [CrossRef] [PubMed]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca Vaccines on COVID-19 Related Symptoms, Hospital Admissions, and Mortality in Older Adults in England: Test Negative Case-Control Study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef]
| Clinical Characteristic | Vaccinated | |||
|---|---|---|---|---|
| All | No | Yes | p | |
| n = 514 | n = 288 | n = 226 | ||
| Medical personal history | ||||
| Age (years) | 63.3 ± 16.1 | 62.2 ± 16.3 | 64.6 ± 15.7 | 0.084 * |
| ≥60 years | 63.1% | 57.1% | 70.8% | 0.001 ** |
| Male (%) | 61.9% | 61.5% | 62.4% | 0.451 ** |
| Data on the COVID vaccination scheme | ||||
| Vaccinated | 56.0% | 44.0% | ||
| ChAdOx1-S a | 17.9% | |||
| BNT162b2 b | 21.7% | |||
| Ad5-nCoV | 2.7% | |||
| CorovaVac c | 1.7% | |||
| Complete primary series | 38.2% | |||
| Booster vaccinations d | 8.9% | |||
| Days since last application | 132.9 ± 97.0 | |||
| Clinical | Vaccinated | p * | |||||
|---|---|---|---|---|---|---|---|
| Characteristic | No | ChAdOx1-S | BNT162b2 | Inter- | No vs. | No vs. | ChAdOx1-S |
| n = 288 (100%) | n = 73 (100%) | n = 101 (100%) | Group | ChAdOx1-S | BNT162b2 | vs. BNT162b2 | |
| ≥60 years | 57.1% | 57.5% | 90.1% | <0.001 | 0.527 | <0.001 | <0.001 |
| Male (%) | 61.5% | 61.6% | 65.3% | 0.777 | |||
| Diabetes | 38.0% | 54.8% | 50.5% | 0.009 * | 0.007 | 0.019 | 0.342 |
| HBP | 34.3% | 56.2% | 54.5% | <0.001 * | 0.001 | <0.001 | 0.473 |
| BMI | 30.4 ± 7.2 | 29.3 ± 4.2 | 30.6 ± 7.5 | 0.264 | |||
| Smoking | 9.6% | 1.4% | 6.1% | 0.025 * | 0.013 | 0.197 | 0.135 |
| CKD | 16.3% | 40.3% | 29.7% | <0.001 | <0.001 | 0.003 | 0.100 |
| Charlson Index | 3.2 ± 2.1 | 4.2 ± 2.1 | 4.7 ± 1.7 | <0.001 * | 0.001 | <0.001 | 0.106 |
| Clinical data at hospital admission | |||||||
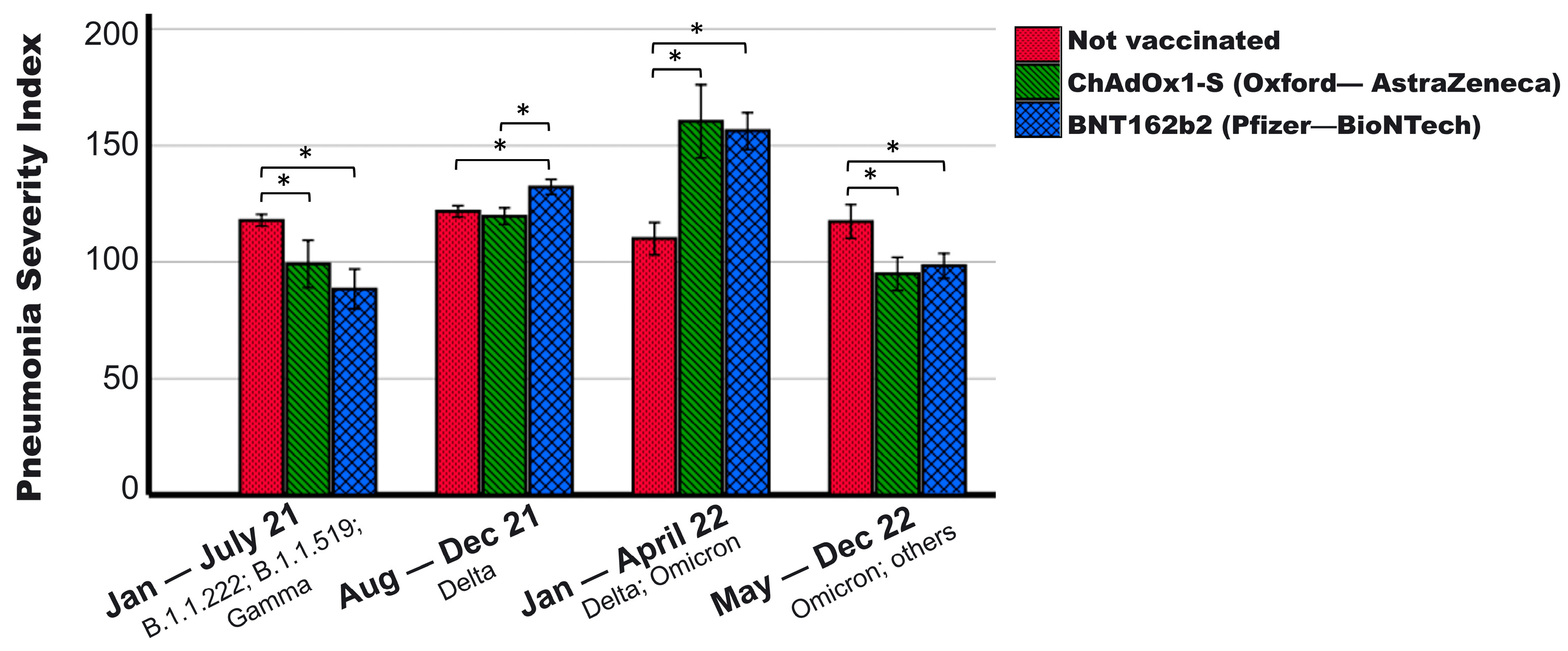
| PSI | 107 ± 42 | 106 ± 37 | 117 ± 36 | 0.110 | |||
| Disease phase | <0.001 | <0.001 | <0.001 | 0.058 | |||
| Viral | 27.7% | 60.3% | 45.0% | ||||
| Pulmonary | 57.1% | 28.8% | 30.0% | ||||
| Inflammation | 15.2% | 11.0% | 25.0% | ||||
| C-GRAM | 135 ± 36 | 131 ± 41 | 134 ± 32 | 0.692 | |||
| NEWS-2 | 7.7 ± 3.5 | 6.5 ± 3.1 | 6.6 ± 3.0 | 0.003 | 0.011 | 0.007 | 0.843 |
| Neutrophils | 7309 ± 5972 | 10,604 ± 7244 | 9606 ± 6101 | <0.001 | 0.001 | 0.001 | 0.336 |
| Lymphocytes | 801 ± 793 | 1094 ± 714 | 1023 ± 789 | 0.004 | 0.005 | 0.019 | 0.554 |
| PlateletsX1000 | 261 ± 122 | 267 ± 147 | 244 ± 103 | 0.390 | |||
| NLR | 13.0 ± 12.5 | 15.3 ± 21.6 | 13.6 ± 12.9 | 0.487 | |||
| D-Dimer | 2999 ± 2184 | 3142 ± 5579 | 2180 ± 3583 | 0.914 | |||
| ESR | 29.8 ± 11.6 | 34.5 ± 16.8 | 23.8 ± 11.7 | 0.050 | 0.226 | 0.047 | 0. 045 |
| CRP | 17.1 ± 18.6 | 15.5 ± 11.2 | 16.6 ± 28.8 | 0.971 * | |||
| Ferritin | 845.8 ± 638.5 | 655.2 ± 586.2 | 559.7 ± 336.1 | 0.017 | 0.122 | 0.009 | 0.394 |
| Creatinine | 2.2 ± 3.7 | 3.8 ± 4.4 | 2.4 ± 3.2 | 0.006 | 0.003 | 0.760 | 0.014 |
| eGFR | 73.9 ± 48.7 | 51.9 ± 58.3 | 54.1 ± 36.7 | <0.001 | 0.003 | 0.002 | 0.999 |
| AST | 50.1 ± 51.2 | 125.6 ± 397.6 | 67.5 ± 193.2 | 0.046 | 0.007 | 0.328 | 0.329 |
| ALT | 41.2 ± 38.8 | 83.2 ± 216.2 | 42.4 + 122.8 | 0.035 | 0.008 | 0.900 | 0.195 |
| ALP | 99.3 ± 65.6 | 139.8 ± 170.5 | 103.1 ± 70.8 | 0.068 | |||
| LDH | 384.2 ± 209.5 | 502.4 ± 954.6 | 345.8 ± 288.0 | 0.078 | |||
| Glucose | 191.5 ± 132.3 | 179.5 ± 129.2 | 214.9 ± 169.5 | 0.232 | |||
| INR | 1.14 + 0.25 | 1.13 + 0.31 | 1.19 + 0.59 | 0.690 | |||
| Main treatments during hospital stay | |||||||
| AKI | 16.7% | 35.6% | 18.7% | 0.002 | 0.001 | 0.545 | 0.004 |
| Paracetamol | 11.4% | 11.0% | 4.0% | 0.054 | |||
| Anticoagulants | 89.3% | 86.3% | 94.1% | 0.193 | |||
| Antibiotics | 48.7% | 42.3% | 49.0% | 0.746 | |||
| Amine support | 9.7% | 6.8% | 8.0% | 0.689 | |||
| Steroids | 90.6% | 93.2% | 93.1% | 0.642 | |||
| Diuretics | 14.6% | 29.5% | 22.1% | 0.150 | |||
| Mech. Vent. | 38.1% | 23.3% | 21.8% | 0.002 | 0.012 | 0.002 | 0.478 |
| Hemodialysis | 9.0% | 19.2% | 5.0% | 0.010 | 0.015 | 0.139 | 0.003 |
| Clinical | Vaccinated | p * | |||||
|---|---|---|---|---|---|---|---|
| Characteristic | No | ChAdOx1-S | BNT162b2 | Inter- | No vs. | No vs. | ChAdOx1-S |
| n = 288 | n = 73 | n = 101 | Group | ChAdOx1-S | BNT162b2 | vs. BNT162b2 | |
| Days hosp. | |||||||
| All | 8.9 ± 6.1 | 7.6 ± 5.1 | 8.4 ± 6.2 | 0.207 | |||
| D. alive | 7.6 ± 5.1 | 6.8 ± 4.2 | 7.9 ± 5.1 | 0.492 | |||
| D. dead | 10.2 ± 6.6 | 8.8 ± 6.0 | 9.2 ± 8.1 | 0.533 | |||
| Deaths | 52.6% | 41.1% | 31.7% | 0.001 | 0.052 | <0.001 | 0.132 |
| Death according to age | |||||||
| <60 years | 40.3% | 29.0% | 20.0% | 0.245 | |||
| ≥60 years | 61.8% | 50.0% | 33.0% | <0.001 | 0.112 | <0.001 | 0.047 |
| Deaths according to Disease phase at admission | |||||||
| Viral | 34.2% | 20.5% | 19.1% | 0.104 | |||
| Advanced ** | 59.8% | 72.4% | 43.4% | 0.024 | 0.134 | 0.023 | 0.010 |
| Deaths according to PSI at admission | |||||||
| PSI score < 120 | 32.2% | 21.7% | 11.1% | 0.002 | 0.115 | 0.001 | 0.107 |
| PSI score ≥ 120 | 85.8% | 76.0% | 67.6% | 0.046 | 0.180 | 0.016 | 0.336 |
| Death with booster vaccination | 38.1% | 30.4% | 0.414 | ||||
| 95% CI | ||||
|---|---|---|---|---|
| Covariate | Ad RR | Lower | Upper | p |
| Age ≥ 60 years | 1.46 | 0.91 | 2.37 | 0.119 |
| Male | 0.97 | 0.63 | 1.50 | 0.887 |
| Diabetes | 1.18 | 0.76 | 1.82 | 0.471 |
| HBP | 0.83 | 0.53 | 1.29 | 0.405 |
| Smoking | 4.50 | 2.00 | 10.14 | <0.001 |
| CKD | 1.42 | 0.72 | 2.83 | 0.314 |
| Charlson Index | 2.67 | 0.60 | 11.97 | 0.199 |
| Admission phase | 4.01 | 2.47 | 6.50 | <0.001 |
| NEWS-2 score ≥ 12 | 3.56 | 1.64 | 7.76 | 0.001 |
| PSI score > 120 | 8.14 | 5.06 | 13.09 | <0.001 |
| Mech. Vent. | 3.74 | 1.89 | 7.39 | <0.001 |
| Creatinine ≥ 4mg/dL | 0.89 | 0.39 | 2.04 | 0.777 |
| AKI | 1.10 | 0.63 | 1.92 | 0.726 |
| Hemodialysis | 0.70 | 0.16 | 3.09 | 0.633 |
| Neutrophils ≥ 8 × 10e3/uL | 3.76 | 2.34 | 6.02 | <0.001 |
| Lymphocytes ≥ 680/uL | 0.53 | 0.34 | 0.84 | 0.007 |
| AST ≥ 50UL | 1.40 | 0.81 | 2.42 | 0.229 |
| ALT ≥ 45UL | 0.45 | 0.26 | 0.79 | 0.005 |
| Ferritin ≥ 810 ng/mL | 0.91 | 0.58 | 1.42 | 0.668 |
| ESR ≥ 30 mm/h | 1.42 | 0.66 | 3.05 | 0.370 |
| Vaccine type | ||||
| BNT162b2/ChAdOx1-S 2 doses α | 0.54 | 0.30 | 0.97 | 0.041 |
| BNT162b2 2 doses α | 0.41 | 0.22 | 0.79 | 0.008 |
| ChAdOx1-S 2 doses α | 1.04 | 0.48 | 2.29 | 0.915 |
| Booster vaccination Ω | 1.74 | 0.68 | 4.45 | 0.247 |
| 95% CI | ||||
|---|---|---|---|---|
| Covariate | Ad RR | Lower | Upper | p |
| Age ≥ 60 years | 7.61 | 2.74 | 21.11 | <0.001 |
| Male | 1.42 | 0.62 | 3.24 | 0.407 |
| Smoking | 0.88 | 0.20 | 3.88 | 0.861 |
| CKD | 0.75 | 0.28 | 2.00 | 0.570 |
| Charlson Index | 1.07 | 0.82 | 1.39 | 0.633 |
| Admission phase | 2.30 | 1.14 | 4.63 | 0.020 |
| NEWS-2 score ≥ 12 | 1.84 | 0.55 | 6.20 | 0.322 |
| PSI score ≥ 120 | 3.91 | 1.50 | 10.22 | 0.006 |
| Mech. Vent. | 1.24 | 0.41 | 3.78 | 0.701 |
| Creatinine ≥ 4mg/dL | 0.58 | 0.17 | 2.00 | 0.389 |
| AKI | 0.88 | 0.37 | 2.06 | 0.762 |
| Neutrophils ≥ 8 × 10e3/uL | 0.87 | 0.37 | 2.05 | 0.753 |
| Lymphocytes ≥ 680/uL | 0.95 | 0.44 | 2.03 | 0.893 |
| AST ≥ 50UL | 1.42 | 0.61 | 3.30 | 0.410 |
| ALT ≥ 45UL | 0.46 | 0.17 | 1.24 | 0.123 |
| Ferritin ≥ 810 ng/mL | 0.21 | 0.09 | 0.46 | <0.001 |
| ESR ≥ 30 mm/h | 0.91 | 0.58 | 1.42 | 0.668 |
| Booster vaccination Ω | 0.32 | 0.13 | 0.75 | 0.009 |
| Death | 0.27 | 0.10 | 0.70 | 0.008 |
| 95% CI | ||||
|---|---|---|---|---|
| Covariate | Ad RR | Lower | Upper | p |
| Age ≥ 60 years | 3.25 | 1.70 | 6.22 | 0.000 |
| Male | 1.78 | 1.10 | 2.88 | 0.018 |
| Smoking | 0.38 | 0.15 | 0.94 | 0.037 |
| CKD | 0.69 | 0.35 | 1.37 | 0.297 |
| Charlson Index | 1.41 | 1.20 | 1.65 | 0.000 |
| Admission phase | 0.88 | 0.55 | 1.42 | 0.622 |
| NEWS-2 score ≥ 12 | 1.18 | 0.62 | 2.26 | 0.603 |
| PSI score ≥ 120 | 1.50 | 0.82 | 2.74 | 0.186 |
| Mech. Vent. | 1.13 | 0.60 | 2.12 | 0.686 |
| Creatinine ≥ 4mg/dL | 1.58 | 0.73 | 3.38 | 0.237 |
| AKI | 0.54 | 0.31 | 0.93 | 0.028 |
| Neutrophils ≥ 8 × 10e3/uL | 2.43 | 1.51 | 3.89 | 0.000 |
| Lymphocytes ≥ 680/uL | 3.63 | 2.34 | 5.62 | 0.000 |
| AST ≥ 50UL | 1.02 | 0.59 | 1.78 | 0.919 |
| ALT ≥ 45UL | 0.64 | 0.35 | 1.19 | 0.163 |
| Ferritin ≥ 810 ng/mL | 0.74 | 0.46 | 1.19 | 0.220 |
| ESR ≥ 30 mm/h | 1.03 | 0.47 | 2.22 | 0.939 |
| Death | 0.38 | 0.19 | 0.72 | 0.004 |
| Mean, First 6 Days | p-Value No vs. | ChAdOx1-S vs. | ||||
|---|---|---|---|---|---|---|
| No | ChAdOx1-S | BNT162b2 | ChAdOx1-S | BNT162b2 | BNT162b2 | |
| Ferritin | 1043 ± 758 | 1031 ± 813 | 632 ± 473 | 0.646 | <0.001 | <0.001 |
| Lymphocytes | 740.7 ± 776 | 1055 ± 699 | 1008 ± 836 | <0.001 | <0.001 | 0.148 |
| PLR | 1259 ± 2317 | 339 ± 301 | 423 ± 460 | <0.001 | <0.001 | 0.055 |
| NEWS-2 | 7.8 ± 3.8 | 6.3 ± 3.5 | 6.1 ± 3.4 | 0.004 | 0.028 | 0.450 |
| PSI score | 110.5 ± 44 | 110.6 ± 39 | 119.3 ± 38 | 0.302 | <0.001 | <0.001 |
| Variable | Group | AUC | S.E. | p | 95% CI | Cutoff | Sens | Spec | |
|---|---|---|---|---|---|---|---|---|---|
| PSI | Not vac | 0.92 | 0.01 | <0.001 | 0.91 | 0.93 | 118.0 | 0.80 | 0.10 |
| ChAdOx1-S | 0.89 | 0.01 | <0.001 | 0.86 | 0.92 | 123.5 | 0.78 | 0.17 | |
| BNT162b2 | 0.85 | 0.02 | <0.001 | 0.82 | 0.88 | 124.5 | 0.81 | 0.22 | |
| Oxemia | Not vac | 0.35 | 0.02 | <0.001 | 0.31 | 0.39 | 74.0 | 0.92 | 0.95 |
| ChAdOx1-S | 0.35 | 0.04 | 0.001 | 0.26 | 0.43 | 84.5 | 0.83 | 0.93 | |
| BNT162b2 | 0.41 | 0.03 | 0.007 | 0.34 | 0.47 | 82.5 | 0.83 | 0.86 | |
| Lymphocytes | Not vac | 0.39 | 0.02 | <0.001 | 0.36 | 0.42 | 535.0 | 0.44 | 0.59 |
| ChAdOx1-S | 0.32 | 0.03 | <0.001 | 0.27 | 0.37 | 831.4 | 0.37 | 0.67 | |
| BNT162b2 | 0.38 | 0.03 | <0.001 | 0.34 | 0.43 | 893.5 | 0.40 | 0.57 | |
| Neutrophils | Not vac | 0.69 | 0.01 | <0.001 | 0.67 | 0.72 | 7311.5 | 0.62 | 0.30 |
| ChAdOx1-S | 0.62 | 0.03 | <0.001 | 0.57 | 0.68 | 9710.9 | 0.65 | 0.43 | |
| BNT162b2 | 0.72 | 0.02 | <0.001 | 0.68 | 0.76 | 9430.1 | 0.70 | 0.37 | |
| LDH | Not vac | 0.75 | 0.02 | <0.001 | 0.72 | 0.78 | 367.5 | 0.62 | 0.25 |
| ChAdOx1-S | 0.87 | 0.03 | <0.001 | 0.82 | 0.92 | 332.7 | 0.79 | 0.17 | |
| BNT162b2 | 0.76 | 0.03 | <0.001 | 0.70 | 0.81 | 325.4 | 0.69 | 0.30 | |
| Ferritin | Not vac | 0.64 | 0.02 | <0.001 | 0.60 | 0.67 | 875.1 | 0.59 | 0.41 |
| ChAdOx1-S | 0.57 | 0.04 | 0.090 | 0.49 | 0.64 | 878.4 | 0.53 | 0.46 | |
| BNT162b2 | 0.67 | 0.03 | <0.001 | 0.61 | 0.73 | 648.3 | 0.58 | 0.41 | |
| CRP | Not vac | 0.74 | 0.02 | <0.001 | 0.71 | 0.78 | 12.2 | 0.60 | 0.25 |
| ChAdOx1-S | 0.57 | 0.05 | 0.162 | 0.47 | 0.67 | 12.3 | 0.56 | 0.49 | |
| BNT162b2 | 0.71 | 0.01 | <0.001 | 0.68 | 0.74 | 12.2 | 0.60 | 0.30 | |
| D dimer | Not vac | 0.74 | 0.02 | <0.001 | 0.70 | 0.77 | 1009.5 | 0.64 | 0.31 |
| ChAdOx1-S | 0.71 | 0.04 | <0.001 | 0.64 | 0.79 | 1231.7 | 0.62 | 0.48 | |
| BNT162b2 | 0.69 | 0.03 | <0.001 | 0.62 | 0.75 | 1083.0 | 0.63 | 0.40 | |
| eGFR | Not vac | 0.32 | 0.01 | <0.001 | 0.29 | 0.34 | 75.2 | 0.42 | 0.73 |
| ChAdOx1-S | 0.43 | 0.03 | 0.009 | 0.37 | 0.48 | 36.3 | 0.46 | 0.59 | |
| BNT162b2 | 0.431 | 0.024 | 0.005 | 0.384 | 0.479 | 60.00 | 0.436 | 0.598 | |
| Vaccine | p-Value No vs. | ChAdOx1-S vs. | ||||
|---|---|---|---|---|---|---|
| Variable | No | ChAdOx1-S | BNT162b2 | ChAdOx1-S | BNT162b2 | BNT162b2 |
| PSI | 119.1 ± 45.5 | 117.5 ± 41.1 | 128.1 ± 42.3 | 0.496 | <0.001 | <0.001 |
| Oxemia | 90.68 ± 10.18 | 90.91 ± 9.95 | 90.78 ± 10.43 | 0.826 | 0.925 | 0.807 |
| pH | 7.339 ± 0.16 | 7.333 ± 0.15 | 7.360 ± 0.14 | 0.314 | 0.034 | 0.014 |
| Lymphocytes | 691.9 ± 722.0 | 1040.4 ± 721.8 | 1091.1 ± 856.6 | <0.001 | <0.001 | 0.102 |
| Neutrophils | 8309.4 ± 6674.1 | 11,972.3 ± 7959.6 | 10,479.9 ± 5828.0 | <0.001 | <0.001 | 0.024 |
| LDH | 421.9 ± 400.2 | 416.3 ± 568.0 | 370.5 ± 258.2 | 0.814 | 0.046 | 0.166 |
| ALP | 92.4 ± 61.5 | 170.3 ± 162.8 | 106.1 ± 75.6 | <0.001 | 0.006 | 0.024 |
| AST | 56.4 ± 177.4 | 80.6 ± 224.9 | 62.0 ± 171.5 | 0.148 | 0.628 | 0.402 |
| ALT | 45.2 ± 58.6 | 63.6 ± 149.6 | 49.2 ± 121.7 | 0.002 | 0.389 | 0.520 |
| Ferritin | 1129.4 ± 769.2 | 1130.3 ± 790.9 | 807.8 ± 616.3 | 0.673 | <0.001 | <0.001 |
| Platelets | 257.5 ± 136.5 | 264.5 ± 155.6 | 270.1 ± 131.3 | 0.057 | 0.894 | 0.130 |
| MPV | 10.7 ± 1.1 | 10.5 ± 1.0 | 10.7 ± 1.2 | 0.006 | 0.706 | 0.042 |
| CRP | 14.8 ± 19.9 | 14.1 ± 9.1 | 13.0 ± 17.7 | 0.699 | 0.230 | 0.562 |
| D dimer | 2475.5 + 12,065.1 | 3312.1 ± 4438.8 | 2090.2 ± 2737.1 | 0.444 | 0.611 | 0.002 |
| eGFR | 84.8 ± 66.2 | 62.8 ± 65.4 | 70.9 ± 53.8 | 0.004 | < 0.001 | 0.826 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza-Hernandez, M.A.; Guzman-Esquivel, J.; Ramos-Rojas, M.A.; Santillan-Luna, V.V.; Sanchez-Ramirez, C.A.; Hernandez-Fuentes, G.A.; Diaz-Martinez, J.; Melnikov, V.; Rojas-Larios, F.; Martinez-Fierro, M.L.; et al. Differences in the Evolution of Clinical, Biochemical, and Hematological Indicators in Hospitalized Patients with COVID-19 According to Their Vaccination Scheme: A Cohort Study in One of the World’s Highest Hospital Mortality Populations. Vaccines 2024, 12, 72. https://doi.org/10.3390/vaccines12010072
Mendoza-Hernandez MA, Guzman-Esquivel J, Ramos-Rojas MA, Santillan-Luna VV, Sanchez-Ramirez CA, Hernandez-Fuentes GA, Diaz-Martinez J, Melnikov V, Rojas-Larios F, Martinez-Fierro ML, et al. Differences in the Evolution of Clinical, Biochemical, and Hematological Indicators in Hospitalized Patients with COVID-19 According to Their Vaccination Scheme: A Cohort Study in One of the World’s Highest Hospital Mortality Populations. Vaccines. 2024; 12(1):72. https://doi.org/10.3390/vaccines12010072
Chicago/Turabian StyleMendoza-Hernandez, Martha A., Jose Guzman-Esquivel, Marco A. Ramos-Rojas, Vanessa V. Santillan-Luna, Carmen A. Sanchez-Ramirez, Gustavo A. Hernandez-Fuentes, Janet Diaz-Martinez, Valery Melnikov, Fabian Rojas-Larios, Margarita L. Martinez-Fierro, and et al. 2024. "Differences in the Evolution of Clinical, Biochemical, and Hematological Indicators in Hospitalized Patients with COVID-19 According to Their Vaccination Scheme: A Cohort Study in One of the World’s Highest Hospital Mortality Populations" Vaccines 12, no. 1: 72. https://doi.org/10.3390/vaccines12010072
APA StyleMendoza-Hernandez, M. A., Guzman-Esquivel, J., Ramos-Rojas, M. A., Santillan-Luna, V. V., Sanchez-Ramirez, C. A., Hernandez-Fuentes, G. A., Diaz-Martinez, J., Melnikov, V., Rojas-Larios, F., Martinez-Fierro, M. L., Tiburcio-Jimenez, D., Rodriguez-Sanchez, I. P., Delgado-Enciso, O. G., Cabrera-Licona, A., & Delgado-Enciso, I. (2024). Differences in the Evolution of Clinical, Biochemical, and Hematological Indicators in Hospitalized Patients with COVID-19 According to Their Vaccination Scheme: A Cohort Study in One of the World’s Highest Hospital Mortality Populations. Vaccines, 12(1), 72. https://doi.org/10.3390/vaccines12010072