Changes in Invasive Pneumococcal Disease in the Paediatric Population in the Second COVID-19 Pandemic Year
Abstract
:1. Introduction
2. Material and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, Y.K.; Choi, Y.Y.; Lee, H.; Song, E.S.; Ahn, J.G.; Park, S.E.; Lee, T.; Cho, H.K.; Lee, J.; Kim, Y.J.; et al. Differential impact of nonpharmaceutical interventions on the epidemiology of invasive bacterial infections in children during the Coronavirus Disease 2019 Pandemic. Pediatr. Infect. Dis. J. 2022, 41, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Janapatla, R.P.; Chen, C.L.; Dudek, A.; Li, H.C.; Yang, H.P.; Su, L.H.; Chiu, C.H. Serotype transmission dynamics and reduced incidence of invasive pneumococcal disease caused by different serotypes after implementation of non-pharmaceutical interventions during COVID-19 pandemic. Eur. Respir. J. 2021, 58, 2100978. [Google Scholar] [CrossRef] [PubMed]
- Ciruela, P.; Broner, S.; Izquierdo, C.; Ayneto, X.; Coronas, L.; Muñoz Almagro, C.; Pallarès, R.; Ardanuy, C.; Martínez, M.; Cabezas, C.; et al. Epidemiology of Invasive Pneumococcal Disease in Catalonia, Report 2019–2020. 2022. Available online: https://salutpublica.gencat.cat/ca/detalls/Article/malaltia-pneumococcica-invasiva-00001 (accessed on 26 May 2023).
- Ciruela, P.; Soldevila, N.; García-Garcia, J.J.; González-Peris, S.; Díaz-Conradi, A.; Redin, A.; Viñado, B.; Izquierdo, C.; Muñoz-Almagro, C.; Domínguez, A.; et al. Effect of COVID-19 pandemic on invasive pneumococcal disease in children, Catalonia, Spain. Emerg. Infect. Dis. 2022, 28, 2321–2325. [Google Scholar] [CrossRef] [PubMed]
- Casanova, C.; Küffer, M.; Leib, S.L.; Hilty, M. Re-emergence of invasive pneumococcal disease (IPD) and increase of serotype 23B after easing of COVID-19 measures, Switzerland, 2021. Emerg. Microbes Infect. 2021, 10, 2202–2204. [Google Scholar] [CrossRef] [PubMed]
- Perniciaro, S.; van der Linden, M.; Weinberger, D.M. Reemergence of invasive pneumococcal disease in Germany during the spring and summer of 2021. Clin. Infect. Dis. 2022, 75, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Bertran, M.; Amin-Chowdhury, Z.; Sheppard, C.L.; Eletu, S.; Zamarreño, D.V.; Ramsay, M.E.; Litt, D.; Fry, N.K.; Ladhani, S.N. Increased incidence of invasive pneumococcal disease among children after COVID-19 pandemic, England. Emerg. Infect. Dis. 2022, 28, 1669–1672. [Google Scholar] [CrossRef] [PubMed]
- Rocafort, M.; Henares, D.; Brotons, P.; Launes, C.; Fernandez de Sevilla, M.; Fumado, V.; Barrabeig, I.; Arias, S.; Redin, A.; Ponomarenko, J.; et al. Impact of COVID-19 lockdown on the nasopharyngeal microbiota of children and adults self-confined at home. Viruses 2022, 14, 1521. [Google Scholar] [CrossRef] [PubMed]
- Real Decreto 463/2020, de 14 de Marzo, por el que se Declara el Estado de Alarma para la Gestión de la Situación de Crisis Sanitaria Ocasionada por el COVID-19. BOE núm. 67, de 14 de Marzo de 2020, Páginas 25390 a 25400. Available online: https://www.boe.es/eli/es/rd/2020/03/14/463 (accessed on 4 September 2023).
- Real Decreto-Ley 21/2020, de 9 de Junio, de Medidas Urgentes de Prevención, Contención y Coordinación para Hacer Frente a la Crisis Sanitaria Ocasionada por el COVID-19. BOE» núm. 163, de 10 de Junio de 2020, Páginas 38723 a 38752. Available online: https://www.boe.es/diario_boe/txt.php?id=BOE-A-2020-5895 (accessed on 4 September 2023).
- Crisis Sanitaria Covid 19. Evolución de la Gestión de la Crisis en España. Gobierno de España. Real Decreto 926/2020, de 25 de Octubre, por el que se Declara el Estado de Alarma Para Contener la Propagación de Infecciones Causadas por el SARS-CoV-2. Available online: https://www.boe.es/buscar/act.php?id=BOE-A-2020-12898 (accessed on 29 May 2023).
- Crisis Sanitaria Covid 19. Evolución de la Gestión de la Crisis en España. Real Decreto 956/2020, de 3 de Noviembre, por el que se Prorroga el Estado de Alarma Declarado por el Real Decreto 926/2020, de 25 de Octubre, por el que se Declara el Estado de Alarma para Contener la Propagación de Infecciones Causadas por el SARS-CoV-2. Available online: https://www.boe.es/buscar/act.php?id=BOE-A-2020-13494 (accessed on 29 May 2023).
- Ondacero, 07.05.2021. Available online: https://www.ondacero.es/emisoras/catalunya/noticies/levanta-confinamiento-elimina-toque-queda-partir-9-mayo-estas-son-restricciones-vigor_20210507609527c86c3eec000114c862.html (accessed on 29 May 2023).
- La Moncloa. Consejo de Ministros, 19.04.2022. Available online: https://www.lamoncloa.gob.es/consejodeministros/resumenes/Paginas/2022/190422-rp-cministros.aspx#:~:text=El%20Ejecutivo%20recomienda%20su%20uso%20responsable%20en%20la%20poblaci%C3%B3n%20vulnerable.&text=El%20Consejo%20de%20Ministros%20ha,partir%20del%2020%20de%20abril (accessed on 29 May 2023).
- Ministerio de Sanidad. Gobierno de España. Cobertura de Vacunación. 2020. Available online: https://www.sanidad.gob.es/areas/promocionPrevencion/vacunaciones/calendario-y-coberturas/coberturas/docs/Todas_las_tablas2020.pdf (accessed on 16 May 2023).
- Izquierdo, C.; Ciruela, P.; Hernández, S.; García-García, J.J.; Esteva, C.; Moraga-Llop, F.; Díaz-Conradi, A.; Martínez-Osorio, J.; Solé-Ribalta, A.; de Sevilla, M.F.; et al. Pneumococcal serotypes in children, clinical presentation and antimicrobial susceptibility in the PCV13 era. Epidemiol. Infect. 2020, 148, e279. [Google Scholar] [CrossRef] [PubMed]
- Silva-Costa, C.; Brito, M.J.; Pinho, M.D.; Friães, A.; Aguiar, S.I.; Ramirez, M.; Melo-Cristino, J.; Portuguese Group for the Study of Streptococcal Infections; Portuguese Study Group of Invasive Pneumococcal Disease of the Pediatric Infectious Disease Society. Pediatric complicated pneumonia caused by Streptococcus pneumoniae serotype 3 in 13-valent pneumococcal conjugate vaccinees, Portugal, 2010–2015. Emerg Infect Dis. 2018, 24, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Alfayate Miguélez, S.; Yague Guirao, G.; Menasalvas Ruíz, A.I.; Sanchez-Solís, M.; Domenech Lucas, M.; González Camacho, F.; Ortíz Romero, M.M.; Espejo García, P.; Guerrero Gómez, C.; Iofrío de Arce, A.; et al. Impact of pneumococcal vaccination in the nasopharyngeal carriage of Streptococcus pneumoniae in healthy children of the Murcia Region in Spain. Vaccines 2020, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Skosana, Z.; Von Gottberg, A.; Olorunju, S.; Mohale, T.; Du Plessis, M.; Adams, T.; Mbelle, N. Non-vaccine serotype pneumococcal carriage in healthy infants in South Africa following introduction of the 13-valent pneumococcal conjugate vaccine. S. Afr. Med. J. 2021, 111, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.M.; Shaik-Dasthagirisaheb, Y.B.; Congdon, M.; Young, R.R.; Patel, M.Z.; Mazhani, T.; Boiditswe, S.; Leburu, T.; Lechiile, K.; Arscott-Mills, T.; et al. Evolution of pneumococcal serotype epidemiology in Botswana following introduction of 13-valent pneumococcal conjugate vaccine. PLoS ONE 2022, 17, e0262225. [Google Scholar] [CrossRef] [PubMed]
- Kielbik Kielbik, K.; Pietras, A.; Jablonska, J.; Bakiera, A.; Borek, A.; Niedzielska, G.; Grzegorczyk, M.; Grywalska, E.; Korona-Glowniak, I. Impact of pneumococcal vaccination on nasopharyngeal carriage of Streptococcus pneumoniae and microbiota profiles in preschool children in South East Poland. Vaccines 2022, 10, 791. [Google Scholar] [CrossRef]
- Janoff, E.N.; Musher, D.M. Streptococcus pneumoniae. In Principles and Practice of Infectious Diseases, 9th ed.; Bennet, E.J., Dolin, R., Blaser, M.I., Eds.; Elsevier: Philadelphia, PA, USA, 2020; pp. 2473–2491. [Google Scholar]
- Heyman, D.L. Control of Communicable Diseases Manual, 2nd ed.; American Public Health Association: Washington, DC, USA, 2022. [Google Scholar]
- American Academy of Pediatrics. Streptococcus pneumoniae (Pneumococcal) infections. In Red Bood 2021 Report of the Committee on Infectious Diseases; Kimberlin, D.W., Barnett, E.D., Lynfield, R., Sawyer, M.H., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2021; pp. 717–727. [Google Scholar]
- Decret 203/2015, de 15 de Setembre, pel Qual es Crea la Xarxa de Vigilància Epidemiològica i es Regulen els Sistemes de Notificació de Malalties de Declaració Obligatòria i els Brots Epidèmics. DOGC, 17 September 2015. Available online: https://portaljuridic.gencat.cat/eli/es-ct/d/2015/09/15/203(accessed on 16 May 2023).


| Mean 2018–2019 | 2020 | 2021 | 2021 vs. 2018–2019 | p Value | Variation | 2021 vs. 2020 | p Value | Variation | |
|---|---|---|---|---|---|---|---|---|---|
| N (IR) | N (IR) | N (IR) | IRR (CI 95%) | % | IRR (CI 95%) | % | |||
| 0–17 years | |||||||||
| Emergency visits | 227,148 (43,661.3) | 148,637 (28,437.6) | 178,243 (34,570.5) | 0.79 (0.78–0.80) | <0.0001 | −21 | 1.22 (1.21–1.22) | <0.0001 | +22 |
| PCR samples (HSJD, HIVH) | 641 (123.2) | 497 (95.1) | 605 (117.3) | 0.95 (0.85–1.06) | 0.39 | NS− | 1.23 (1.10–1.39) | 0.0005 | +23 |
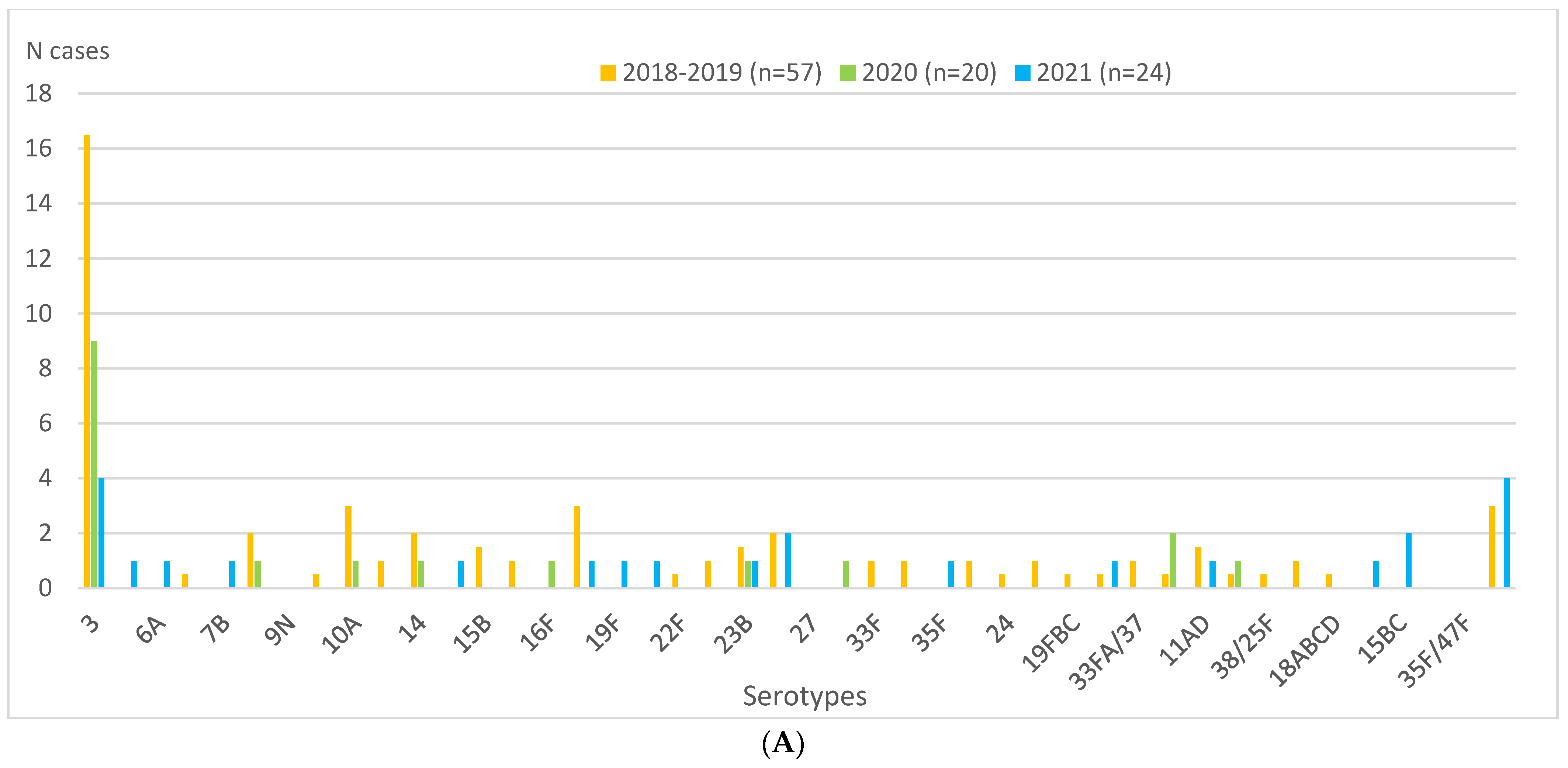
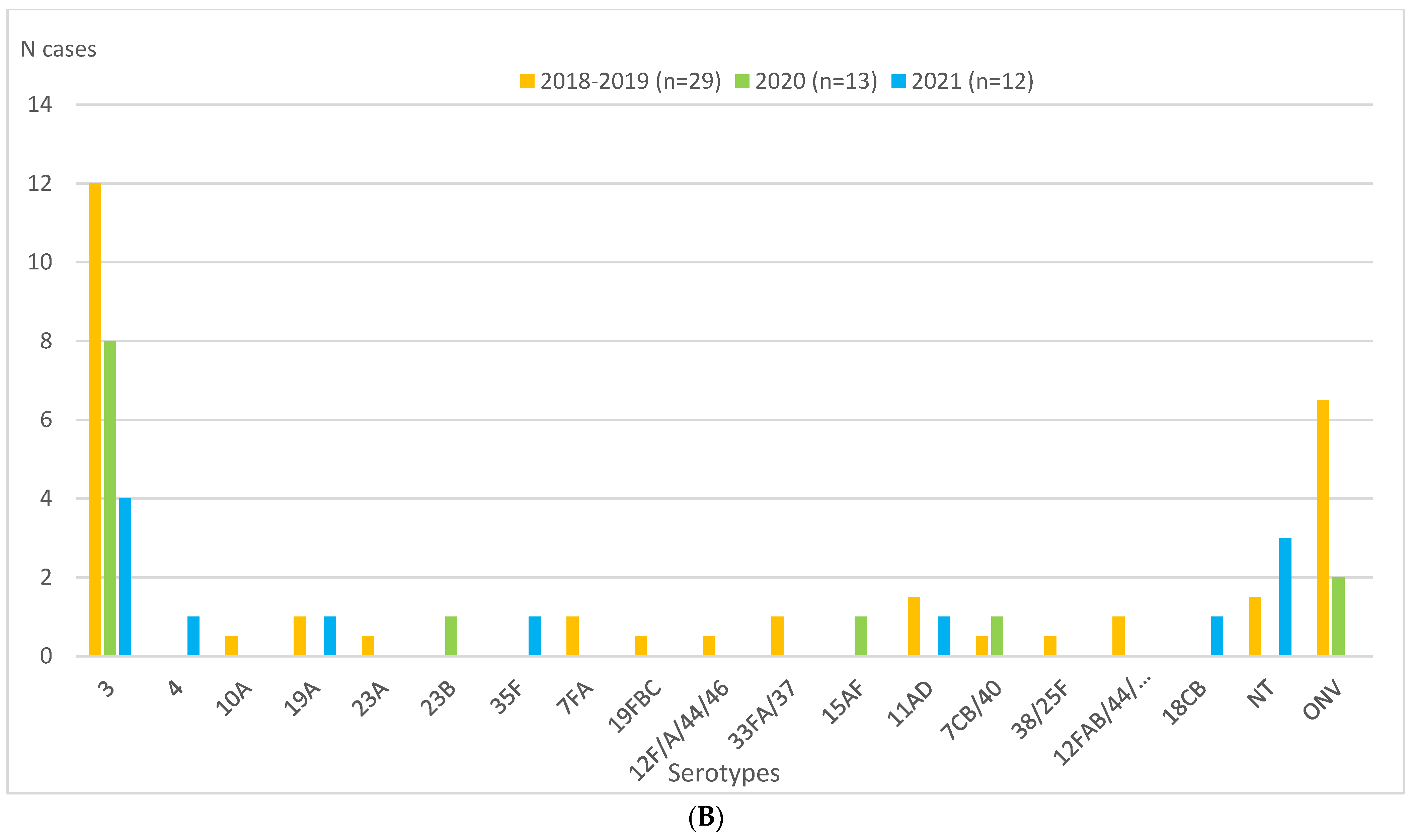
| IPD cases | 57 (11.0) | 20 (3.8) | 24 (4.6) | 0.42 (0.26–0.68) | <0.0001 | −58 | 1.22 (0.67–2.23) | 0.52 | NS+ |
| PCV13 serotypes | 25 (4.8) | 10 (1.9) | 8 (1.6) | 0.32 (0.15–0.72) | 0.003 | −68 | 0.81 (0.32–2.05) | 0.66 | NS− |
| Serotype 3 | 17 (3.3) | 9 (1.7) | 4 (0.8) | 0.24 (0.08–0.70) | 0.005 | −76 | 0.45 (0.14–1.46) | 0.17 | NS− |
| Non-PCV13 serotypes | 29 (5.6) | 10 (1.9) | 15 (2.9) | 0.52 (0.27–0.97) | 0.04 | −48 | 1.52 (0.68–3.51) | 0.31 | NS+ |
| 0–4 years | |||||||||
| Emergency visits | 108,757 (93,016.7) | 68,684 (60,617.9) | 104,023 (96,896.3) | 1.04 (1.03–1.05) | <0.0001 | +4 | 1.60 (1.58–1.61) | <0.0001 | +60 |
| PCR samples (HSJD, HIVH) | 459 (392.6) | 342 (301.8) | 449 (418.2) | 1.07 (0.96–1.21) | 0.34 | NS+ | 1.38 (1.20–1.59) | <0.0001 | +38 |
| IPD cases | 44 (37.6) | 15 (13.2) | 18 (16.8) | 0.44 (0.25–0.76) | 0.002 | −56 | 1.27 (0.63–2.56) | 0.50 | NS+ |
| PCV13 serotypes | 18 (15.4) | 8 (7.1) | 5 (4.6) | 0.30 (0.11–0.81) | 0.01 | −70 | 0.66 (0.22–2.02) | 0.46 | NS− |
| Serotype 3 | 12 (10.3) | 8 (7.1) | 2 (1.9) | 0.18 (0.04–0.81) | 0.01 | −82 | 0.26 (0.06–1.24) | 0.07 | NS− |
| Non-PCV13 serotypes | 25 (21.4) | 7 (6.2) | 12 (11.2) | 0.52 (0.25–1.03) | 0.06 | NS− | 1.80 (0.71–4.89) | 0.22 | NS+ |
| 5-17 years | |||||||||
| Emergency visits | 118,391 (29,353.5) | 79,953 (19,530.7) | 74,220 (18,180.6) | 0.62 (0.61–0.63) | <0.0001 | −38 | 0.93 (0.92–0.94) | <0.0001 | −7 |
| PCR samples (HSJD, HIVH) | 182 (45.1) | 155 (37.9) | 156 (38.2) | 0.85 (0.68–1.05) | 0.13 | NS− | 1.01 (0.81–1.26) | 0.93 | NS |
| IPD cases | 13 (3.2) | 5 (1.2) | 6 (1.5) | 0.46 (0.16–1.18) | 0.11 | NS− | 1.20 (0.35–4.28) | 0.77 | NS+ |
| PCV13 serotypes | 7 (1.7) | 2 (0.5) | 3 (0.7) | 0.42 (0.11–1.64) | 0.20 | NS− | 1.50 (0.25–9.00) | 0.65 | NS+ |
| Serotype 3 | 5 (1.2) | 1 (0.2) | 2 (0.5) | 0.39 (0.08–2.04) | 0.25 | NS− | 2.01 (0.18–22.12) | 0.56 | NS+ |
| Non-PCV13 serotypes | 4 (1.0) | 3 (0.7) | 3 (0.7) | 0.74 (0.14–3.59) | 0.72 | NS− | 1.00 (0.17–5.84) | 0.99 | NS |
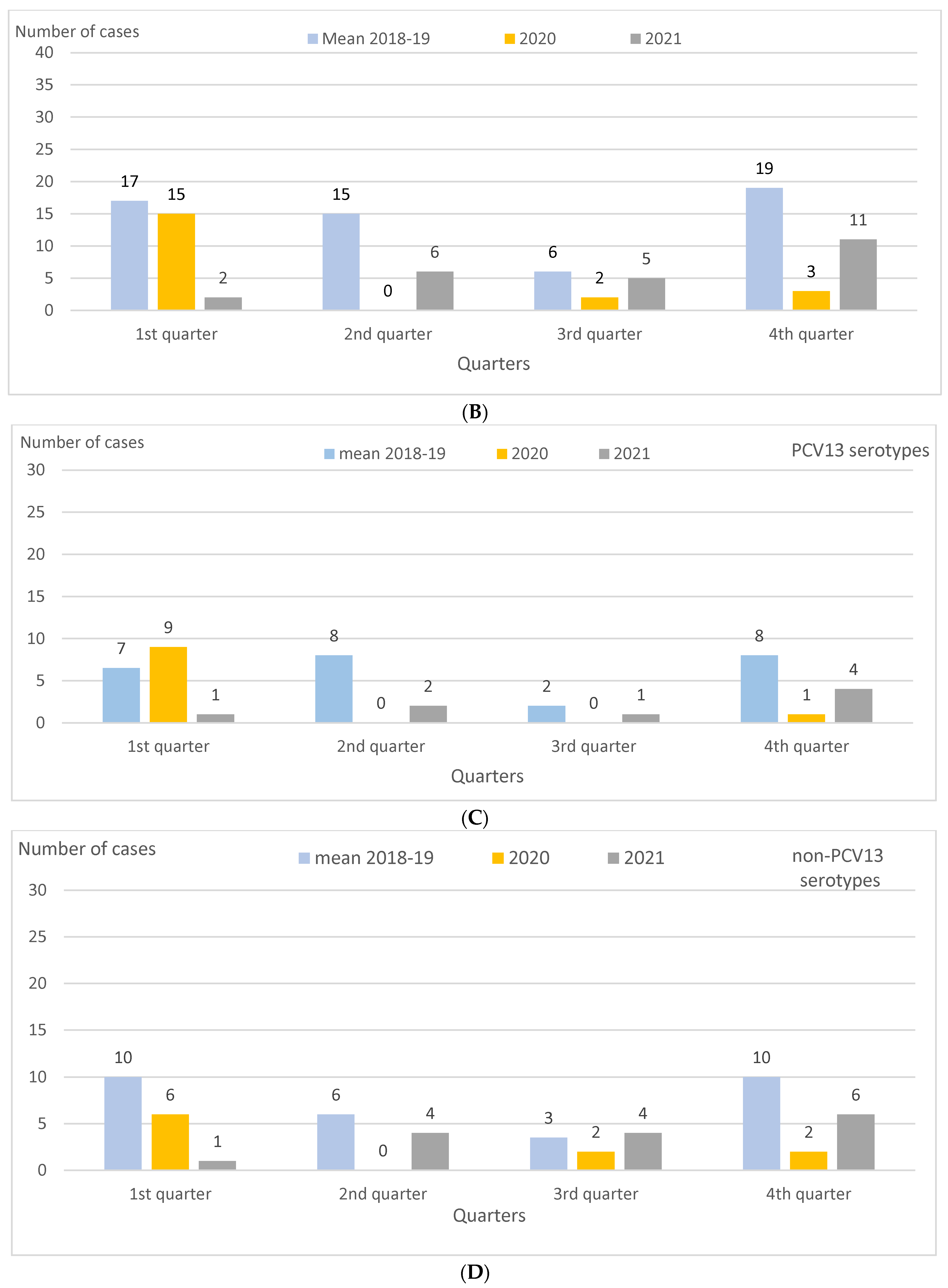
| Quarter | Mean 2018–2019 | 2020 | 2021 | 2021 vs. 2018–2019 | p Value | Variation | 2021 vs. 2020 | p Value | Variation |
|---|---|---|---|---|---|---|---|---|---|
| N (IR) | N (IR) | N (IR) | IRR (CI 95%) | % | IRR (CI 95%) | % | |||
| 1 st quarter | |||||||||
| Emergency visits | 61,590 (11,838.5) | 54,430 (10,413.7) | 37,324 (7239.0) | 0.61 (0.60–0.62) | <0.0001 | −39 | 0.70 (0.69–0.70) | <0.0001 | −30 |
| PCR samples (HSJD, HIVH) | 185 (35.6) | 182 (34.8) | 98 (19.0) | 0.53 (0.42–0.68) | <0.0001 | −47 | 0.54 (0.43–0.70) | <0.0001 | −46 |
| IPD cases | 17 (3.3) | 15 (2.9) | 2 (0.4) | 0.12 (0.02–0.45) | 0.0004 | −88 | 0.14 (0.02–0.52) | 0.0001 | −86 |
| PCV13 serotypes | 7 (1.3) | 9 (1.7) | 1 (0.2) | 0.14 (0.02–1.17) | 0.06 | NS− | 0.11 (0.01–0.89) | 0.01 | −89 |
| Serotype 3 | 5 (1.0) | 8 (1.5) | 0 (0) | - | 0.03 | (-Not calc) | - | 0.01 | (-Not calc) |
| Non-PCV13 serotypes | 10 (1.9) | 6 (1.1) | 1 (0.2) | 0.10 (0.01–0.60) | 0.001 | −90 | 0.17 (0.01–1.14) | 0.07 | NS- |
| 2 nd quarter | |||||||||
| Emergency visits | 55,519 (10,671.6) | 23,025 (4405.2) | 46,291 (8978.2) | 0.84 (0.83–0.85) | <0.0001 | −16 | 2.04 (2.01–2.07) | <0.0001 | +104 |
| PCR samples (HSJD, HIVH) | 141 (27.1) | 107 (20.5) | 198 (38.4) | 1.42 (1.14–1.76) | 0.001 | +42 | 1.88 (1.48–2.37) | <0.0001 | +88 |
| IPD cases | 15 (2.9) | 0 (0) | 6 (1.2) | 0.40 (0.14–1.02) | 0.05 | NS− | - | 0.02 | (+Not calc) |
| PCV13 serotypes | 8 (1.5) | 0 (0) | 2 (0.4) | 0.25 (0.05–1.19) | 0.06 | NS− | - | 0.15 | NS+ |
| Serotype 3 | 6 (1.2) | 0 (0) | 1 (0.2) | 0.17 (0.02–1.10) | 0.06 | NS− | - | 0.31 | NS+ |
| Non-PCV13 serotypes | 6 (1.2) | 0 (0) | 4 (0.8) | 0.67 (0.17–2.46) | 0.56 | NS− | - | 0.06 | NS+ |
| 3 rd quarter | |||||||||
| Emergency visits | 44,594 (8571.6) | 34,933 (6683.5) | 35,692 (6922.5) | 0.81 (0.80–0.82) | <0.0001 | −19 | 1.04 (1.02–1.05) | <0.0001 | +4 |
| PCR samples (HSJD, HIVH) | 112 (21.5) | 86 (16.5) | 158 (30.6) | 1.42 (1.12–1.81) | 0.004 | +42 | 1.86 (1.43–2.42) | <0.0001 | +86 |
| IPD cases | 6 (1.2) | 2 (0.4) | 5 (1.0) | 0.84 (0.24–2.88) | 0.78 | NS− | 2.53 (0.50–18.86) | 0.28 | NS+ |
| PCV13 serotypes | 2 (0.4) | 0 (0) | 1 (0.2) | 0.50 (0.05–5.56) | 0.57 | NS− | - | 0.31 | NS+ |
| Serotype 3 | 1 (0.2) | 0 (0) | 0 (0) | - | 0.32 | NS− | - | NS | |
| Non-PCV13 serotypes | 3 (0.6) | 2 (0.4) | 4 (0.8) | 1.34 (0.28–7.21) | 0.72 | NS+ | 2.03 (0.36–15.83) | 0.44 | NS+ |
| 4 th quarter | |||||||||
| Emergency visits | 65,445 (12,579.5) | 36,249 (6935.3) | 58,933 (11,430.1) | 0.91 (0.90–0.92) | <0.0001 | −9 | 1.65 (1.963–1.67) | <0.0001 | +65 |
| PCR samples (HSJD, HIVH) | 203 (39.0) | 122 (23.3) | 151 (29.3) | 0.75 (0.61–0.92) | 0.007 | −25 | 1.25 (0.99–1.59) | 0.06 | NS+ |
| IPD cases | 19 (3.7) | 3 (0.6) | 11 (2.1) | 0.58 (0.27–1.22) | 0.16 | NS− | 3.72 (1.04–13.32) | 0.03 | +272 |
| PCV13 serotypes | 8 (1.5) | 1 (0.2) | 4 (0.8) | 0.50 (0.15–1.67) | 0.25 | NS− | 4.05 (0.45–36.28) | 0.17 | NS+ |
| Serotype 3 | 5 (1.0) | 1 (0.2) | 3 (0.6) | 0.60 (0.14–2.53) | 0.49 | NS− | 3.04 (0.32–29.24) | 0.31 | NS+ |
| Non-PCV13 serotypes | 10 (1.9) | 2 (0.4) | 6 (1.2) | 0.60 (0.20–1.67) | 0.34 | NS− | 3.04 (0.64–21.89) | 0.17 | NS+ |
| Diagnostic Technique | Mean 2018–2019 | % | 2020 | % | 2021 | % | 2018–2021 | % |
|---|---|---|---|---|---|---|---|---|
| Culture only | 16 | 28.9 | 2 | 10 | 4 | 16.7 | 38 | 24.1 |
| PCR only | 29 | 50.0 | 13 | 65 | 12 | 50.0 | 83 | 52.5 |
| Culture + PCR | 12 | 21.1 | 5 | 25 | 8 | 33.3 | 37 | 23.4 |
| Total | 57 | 100.0 | 20 | 100 | 24 | 100 | 158 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izquierdo, C.; Ciruela, P.; Soldevila, N.; Garcia-Garcia, J.-J.; Gonzalez-Peris, S.; Díaz-Conradi, A.; Viñado, B.; F de Sevilla, M.; Moraga-Llop, F.; Muñoz-Almagro, C.; et al. Changes in Invasive Pneumococcal Disease in the Paediatric Population in the Second COVID-19 Pandemic Year. Vaccines 2023, 11, 1548. https://doi.org/10.3390/vaccines11101548
Izquierdo C, Ciruela P, Soldevila N, Garcia-Garcia J-J, Gonzalez-Peris S, Díaz-Conradi A, Viñado B, F de Sevilla M, Moraga-Llop F, Muñoz-Almagro C, et al. Changes in Invasive Pneumococcal Disease in the Paediatric Population in the Second COVID-19 Pandemic Year. Vaccines. 2023; 11(10):1548. https://doi.org/10.3390/vaccines11101548
Chicago/Turabian StyleIzquierdo, Conchita, Pilar Ciruela, Núria Soldevila, Juan-Jose Garcia-Garcia, Sebastia Gonzalez-Peris, Alvaro Díaz-Conradi, Belen Viñado, Mariona F de Sevilla, Fernando Moraga-Llop, Carmen Muñoz-Almagro, and et al. 2023. "Changes in Invasive Pneumococcal Disease in the Paediatric Population in the Second COVID-19 Pandemic Year" Vaccines 11, no. 10: 1548. https://doi.org/10.3390/vaccines11101548
APA StyleIzquierdo, C., Ciruela, P., Soldevila, N., Garcia-Garcia, J.-J., Gonzalez-Peris, S., Díaz-Conradi, A., Viñado, B., F de Sevilla, M., Moraga-Llop, F., Muñoz-Almagro, C., Domínguez, A., & Barcino Working Group. (2023). Changes in Invasive Pneumococcal Disease in the Paediatric Population in the Second COVID-19 Pandemic Year. Vaccines, 11(10), 1548. https://doi.org/10.3390/vaccines11101548









