Cost Effectiveness of Quadrivalent Versus Trivalent Inactivated Influenza Vaccines for the Portuguese Elderly Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Key Features of the Economic Evaluation
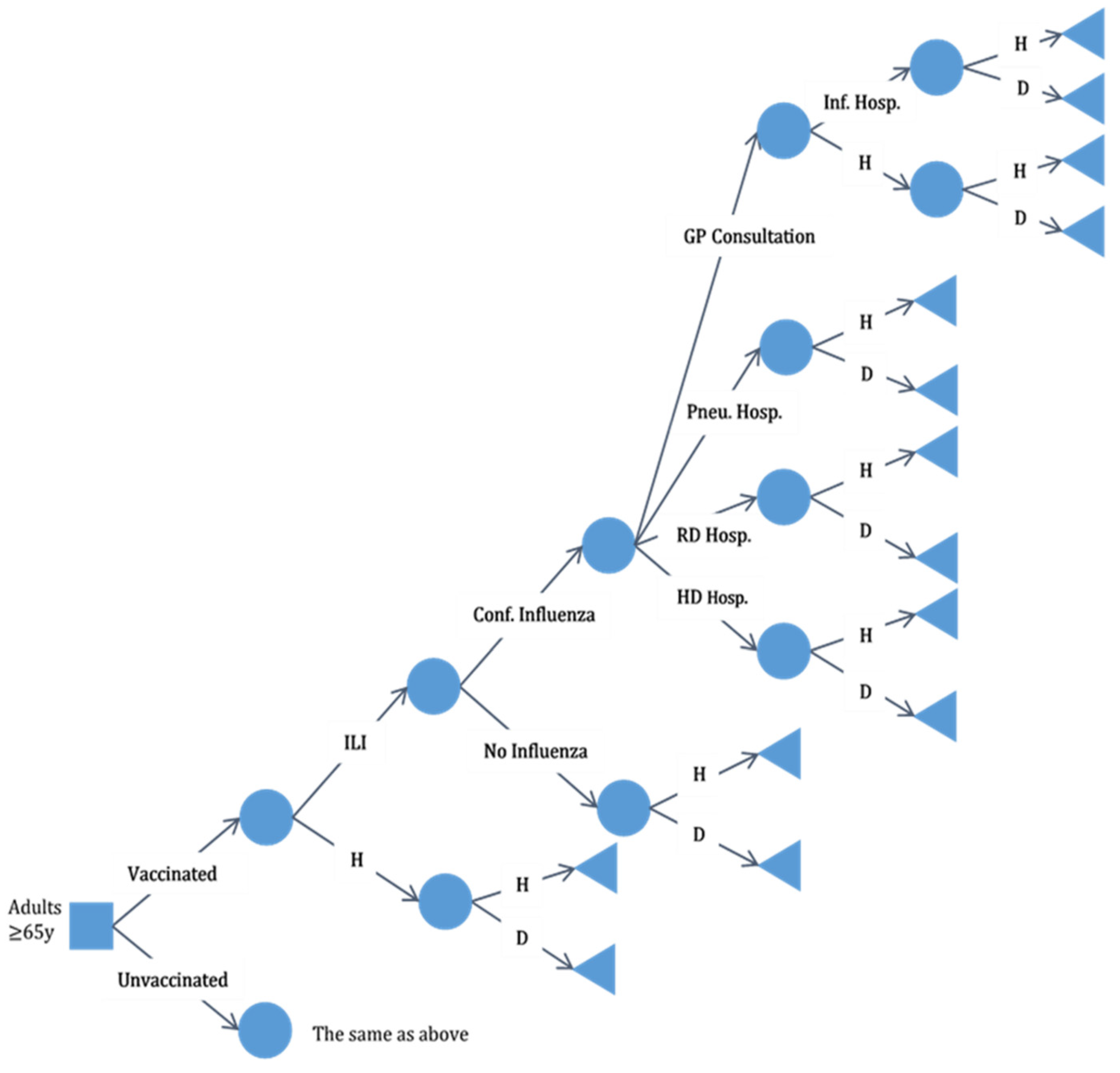
2.2. Decision Tree
2.3. Input Parameters and Computation of the Probabilities
2.4. Scenario Analysis
3. Results
3.1. Base Case Analysis
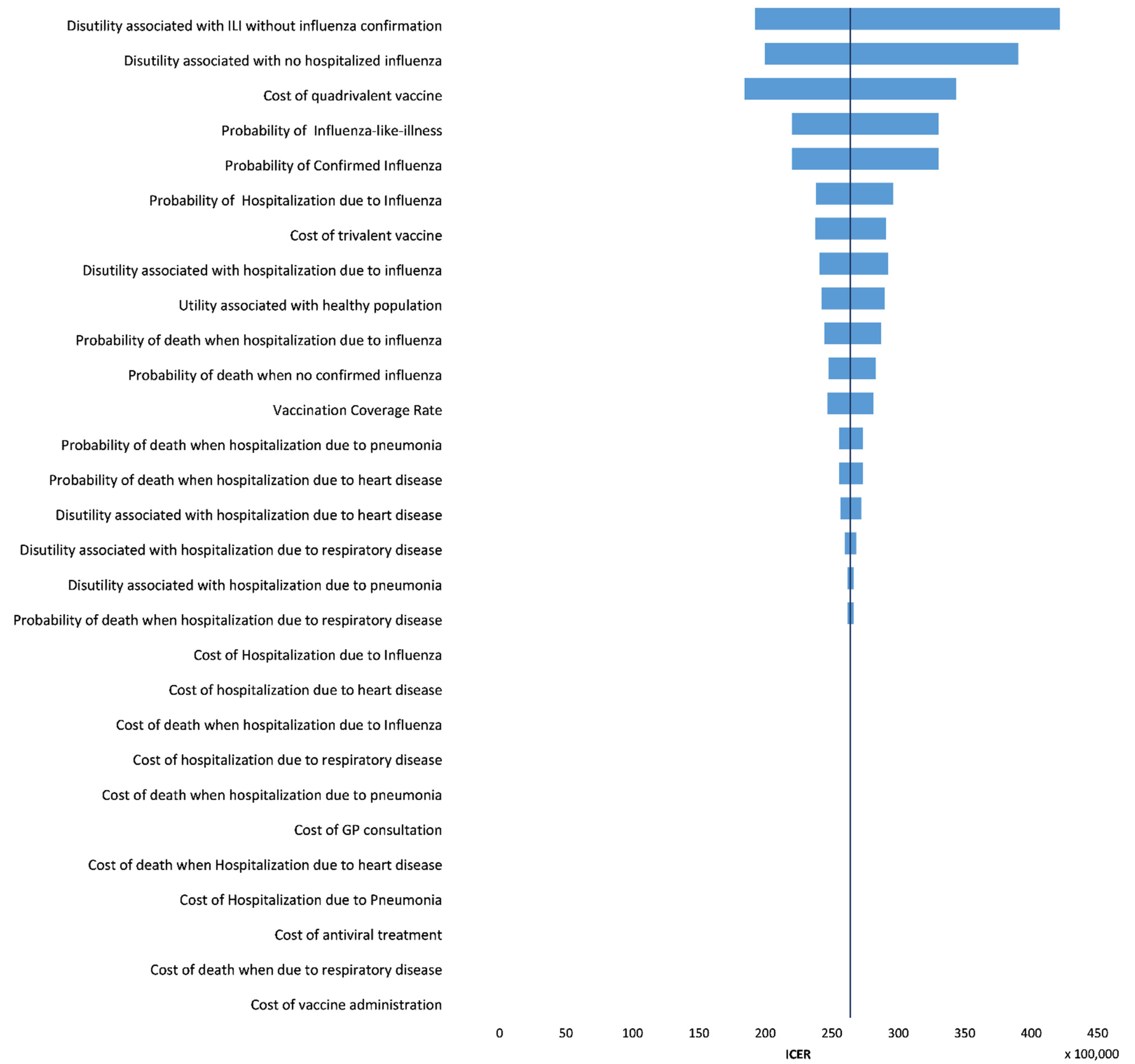
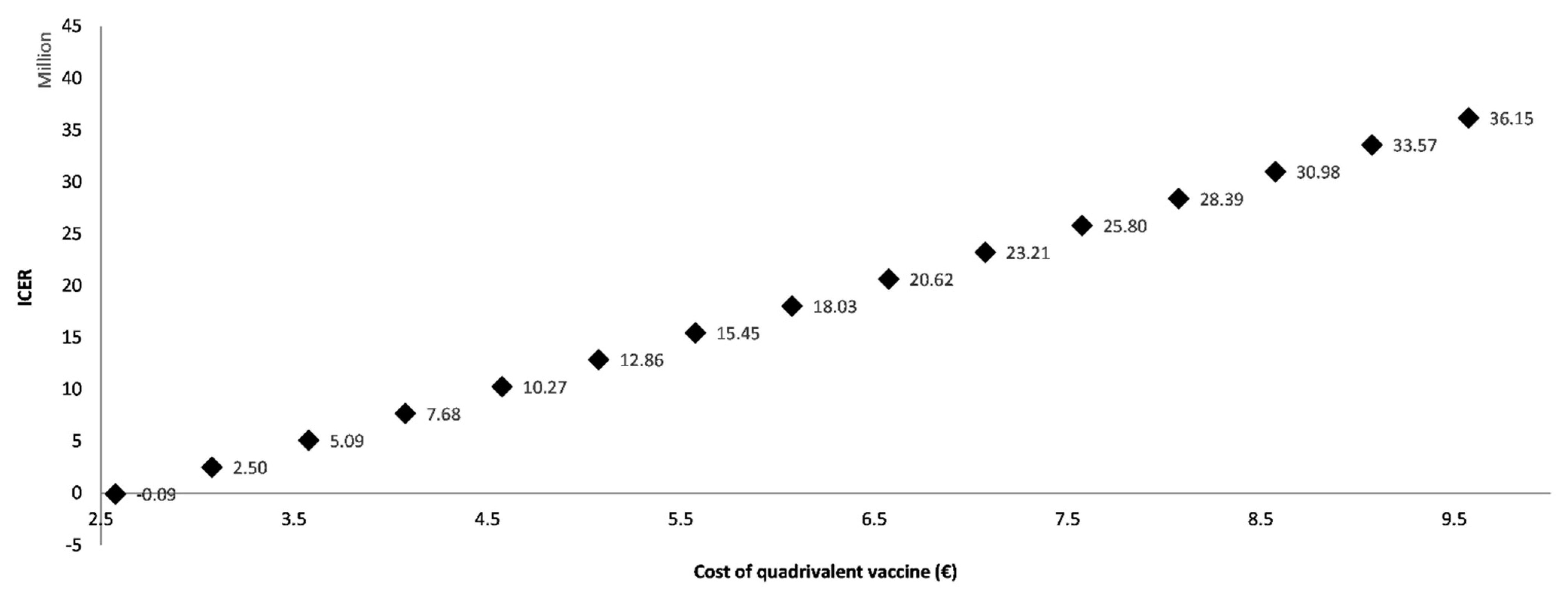
3.2. One-Way Sensitivity Analysis
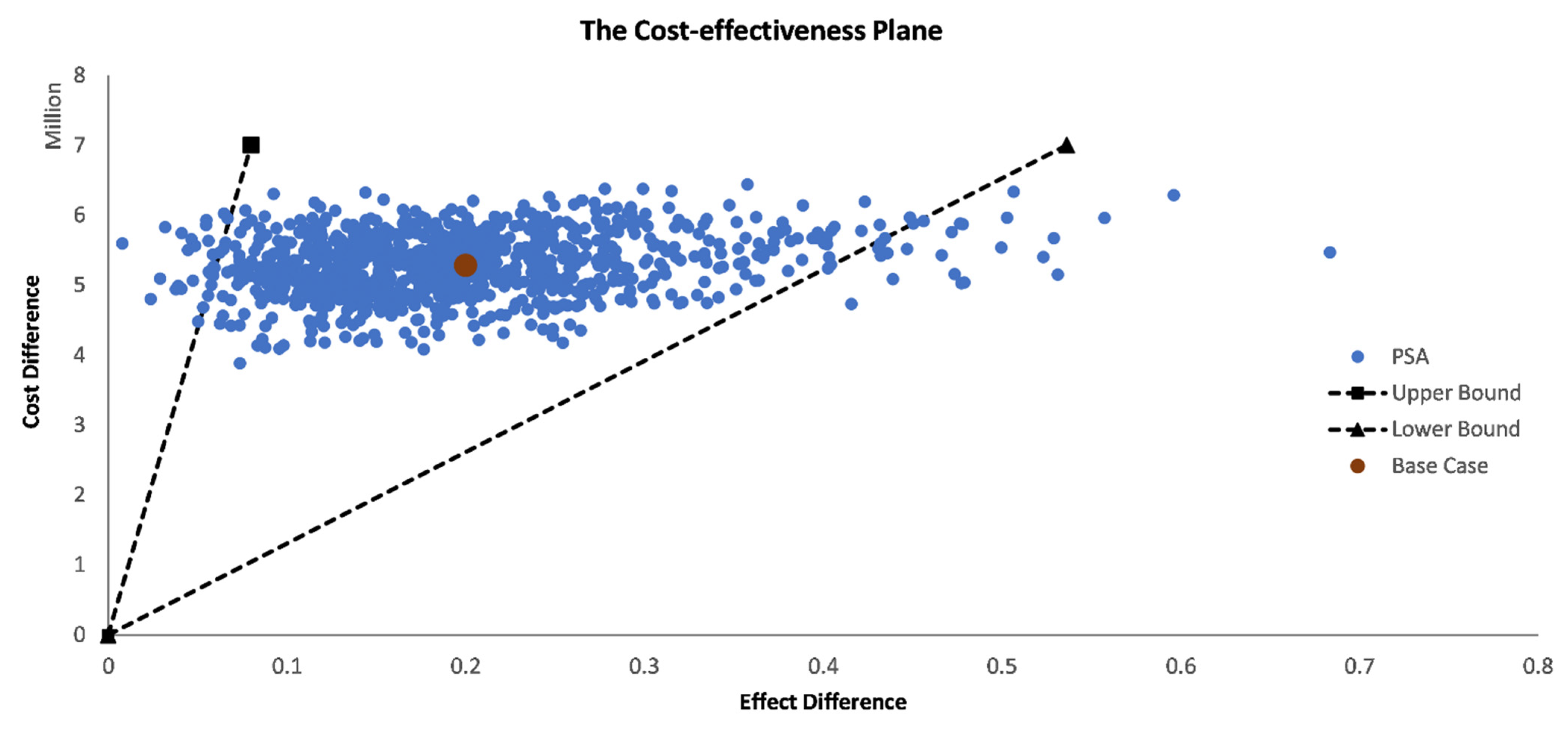
3.3. Probabilistic Sensitivity Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
Appendix B
Appendix B.1. Model Input Parameters
| Season | Proportion of Influenza B | Proportion of B/Victoria | Proportion of B/Yamagata | B—lineage in TIV | Proportion of B Lineage Mismatch | SE | Source |
|---|---|---|---|---|---|---|---|
| 2010/11 | 43.20% | 42.70% | 0.50% | Victoria | 0.50% | 0.00022 | [28] |
| 2011/12 | 2.30% | 0.00% | 2.30% | Victoria | 2.30% | 0.00058 | [29] |
| 2012/13 | 51.30% | 1.80% | 49.50% | Yamagata | 1.80% | 0.00038 | [30] |
| 2013/14 | 0.80% | 0.10% | 0.70% | Yamagata | 0.10% | 0.00011 | [31] |
| 2014/15 | 0.36% | 0.00% | 0.36% | Yamagata | 0.00% | 0.00000 | [32] |
| 2015/16 | 8.30% | 7.80% | 0.50% | Yamagata | 7.80% | 0.00084 | [33] |
| 2016/17 | 0.20% | 0.20% | 0.00% | Victoria | 0.00% | 0.00000 | [34] |
| 2017/18 | 66.00% | 9.00% | 57.00% | Victoria | 57.00% | 0.00354 | [35] |
| 2010/11–2017/18 | 21.56% | 8.69% | 0.00213 |
Appendix B.2. Computation of the Probabilities
Appendix B.3. Costs
References
- WHO. Influenza (Seasonal). 2018. Available online: http://www.who.int/mediacentre/factsheets/fs211/en/ (accessed on 2 February 2020).
- Shi, A.; Zeng, D.; Lin, S.-S.; Caldwell, L. Harrison’s—Principles of Internal Medicine, 16th ed.; McGraw-Hill: New York, NY, USA, 2011; p. 515. [Google Scholar]
- Villacampa, T. Manual Curso Intensivo Mir Asturias—Enfermedades Infecciosas; Curso Intensivo MIR Asturias: Oviedo, Spain, 2009. [Google Scholar]
- George, F. Orientação DGS: Vacinação contra a gripe. Época 2017/2018; Direção-Geral da Saúde: Lisbon, Portugal, 2017; pp. 1–6. [Google Scholar]
- Types of Influenza Viruses|Seasonal Influenza (Flu)|CDC. Available online: https://www.cdc.gov/flu/about/viruses/types.htm (accessed on 6 March 2018).
- George, F. História da Gripe; Direção-Geral da Saúde: Lisbon, Portugal, 2014; pp. 1–28. [Google Scholar]
- Sousa-Uva, M.; Kislaya, I.; Roquette, R.; Rodrigues, A.P.; Machado, A. Vacinação Antigripal da População Portuguesa na época 2015/2016: Estudo na Amostra ECOS; Instituto Nacional de Saúde Dr. Ricardo Jorge: Lisbon, Portugal, 2016. [Google Scholar]
- Goméz, V.; Guiomar, R.; Rodrigues, A.P.; Pechirra, P.; Conde, P.; Cristóvão, P.; Costa, I.; Nunes, B.; Machado, A. Influenza Vaccine Effectiveness in Portugal: Season 2015–16 Report; Instituto Nacional de Saúde Dr. Ricardo Jorge: Lisbon, Portugal, 2017. [Google Scholar]
- Key Facts About Influenza (Flu)|Seasonal Influenza (Flu)|CDC. Available online: https://www.cdc.gov/flu/keyfacts.htm (accessed on 9 March 2018).
- Jorgensen, P.; Mereckiene, J.; Cotter, S.; Johansen, K.; Tsolova, S.; Brown, C. How Close are Countries of the WHO European Region to Achieving the Goal of Vaccinating 75% of Key Risk Groups against Influenza? Results from National Surveys on Seasonal Influenza Vaccination Programmes, 2008/2009 to 2014/2015. Vaccine 2017, 36, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.; Torres, A.R.; Kislaya, I.; Neto, M. Vacinação Antigripal da População Portuguesa nas épocas 2016/2017 e 2017/2018. Cobertura e Características do Ato Vacinal; Instituto Nacional de Saúde Dr. Ricardo Jorge: Lisbon, Portugal, 2019. [Google Scholar]
- Demicheli, V.; Jefferson, T.; Pietrantonj, C.D.; Ferroni, E.; Thorning, S.; Thomas, R.E.; Rivetti, A. Vaccines for preventing influenza in the elderly (Review). Cochrane Database Syst. Rev. 2018, 2, CD004876. [Google Scholar] [CrossRef] [PubMed]
- Influenza—European Commission. Available online: https://ec.europa.eu/health/vaccination/influenza_en (accessed on 15 March 2018).
- WHO. Types of Seasonal Influenza Vaccine. 22 March 2018. Available online: http://www.euro.who.int/en/health-topics/communicable-diseases/influenza/vaccination/types-of-seasonal-influenza-vaccine (accessed on 22 March 2018).
- Sanders, G.D.; Neumann, P.J.; Basu, A.; Brock, D.W.; Feeny, D.; Krahn, M.; Kuntz, K.M.; Meltzer, D.O.; Owens, D.K.; Prosser, L.A.; et al. Recommendations for Conduct, Methodological Practices, and Reporting of Cost-effectiveness Analyses: Second Panel on Cost-Effectiveness in Health and Medicine. JAMA 2016, 316. [Google Scholar] [CrossRef]
- Ferreira, L.N.; Ferreira, P.L.; Pereira, L.N.; Oppe, M. EQ-5D Portuguese population norms. Qual Life Res. 2014, 23, 425–430. [Google Scholar] [CrossRef]
- Hollmann, M.; Garin, O.; Galante, M.; Ferrer, M.; Dominguez, A.; Alonso, J. Impact of Influenza on Health-Related Quality of Life among Confirmed (H1N1)2009 Patients. PLoS ONE 2013, 8, e60477. [Google Scholar] [CrossRef]
- Greenland, S. Sensitivity analysis, Monte Carlo risk analysis, and Bayesian uncertainty assessment. Risk Anal. 2001, 21, 579–583. [Google Scholar] [CrossRef]
- Drummond, M.F.; Sculpher, M.J.; Claxton, K.; Stoddart, G.L.; Torrance, G.W. Methods for the Economic Evaluation of Health Care Programmes, 4th ed.; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Laires, P.A.; Ejzykowicz, F.; Hsu, T.-Y.; Ambegaonkar, B.; Davies, G. Cost–effectiveness of adding ezetimibe to atorvastatin vs switching to rosuvastatin therapy in Portugal. J. Med. Econ. 2015, 18, 565–572. [Google Scholar] [CrossRef]
- Cameron, D.; Ubels, J.; Norström, F. On what basis are medical cost-effectiveness thresholds set? Clashing opinions and an absence of data: A systematic review. Glob. Health Action 2018, 11, 1447828. [Google Scholar] [CrossRef] [PubMed]
- Marques, A.; Lourenço, Ó.; Ortsäter, G.; Borgström, F.; Kanis, J.A.; da Silva, J.A.P. Cost-Effectiveness of Intervention Thresholds for the Treatment of Osteoporosis Based on FRAX® in Portugal. Calcif Tissue Int. 2016, 99, 131–141. [Google Scholar] [CrossRef]
- Areia, M.; Spaander, M.C.W.; Kuipers, E.J.; Dinis-Ribeiro, M. Endoscopic screening for gastric cancer: A cost-utility analysis for countries with an intermediate gastric cancer risk. United Eur. Gastroenterol. J. 2018, 6, 192–202. [Google Scholar] [CrossRef]
- World Development Indicators|DataBank. Available online: http://databank.worldbank.org/data/reports.aspx?source=2&country=PRT (accessed on 15 September 2018).
- Briggs, A.; Sculpher, M.; Claxton, K. Decision Modelling for Health Economic Evaluation; Oxford University Press: Oxford, UK, 2006; p. 445. [Google Scholar]
- Instituto Nacional de Estatística. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_indicadores&indOcorrCod=0000256&contexto=bd&selTab=tab2&xlang=en (accessed on 4 May 2018).
- Guiomar, R.; Conde, P.; Cristóvão, P.; Pechirra, P.; Nunes, B. Programa Nacional de Vigilância da Gripe—Relatório da Época 2012/2013; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2013. [Google Scholar]
- Guiomar, R.; Conde, P.; Cristóvão, P.; Pechirra, P.; Nunes, B. Programa Nacional de Vigilância da Gripe—Relatório da Época 2013/2014; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2015. [Google Scholar]
- Guiomar, R.; Costa, I.; Cristóvão, P.; Pechirra, P.; Rodrigues, A.P.; Nunes, B. Programa Nacional de Vigilância da Gripe—Relatório da época 2014/2015; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2015. [Google Scholar]
- Guiomar, R.; Pechirra, P.; Cristóvão, P.; Costa, I.; Conde, P.; Rodrigues, A.P.; Silva, S.P.; Kislaya, I.; Nunes, B. Programa Nacional de Vigilância da Gripe-Relatório da época 2016/2017; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2017. [Google Scholar]
- Guiomar, R.; Pechirra, P.; Rodrigues, A.P.; Silva, S.P.; Machado, A.; Nunes, B. Programa Nacional de Vigilância da Gripe—Relatório da época 2015/2016; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2016. [Google Scholar]
- Gonçalves, P.; Pechirra, P.; Conde, P.; Guiomar, R.; Nunes, B. Programa Nacional de Vigilância da Gripe—Relatório da Época 2010/2011; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2013. [Google Scholar]
- Gonçalves, P.; Pechirra, P.; Conde, P.; João, I.; Guiomar, R.; Nunes, B. Programa Nacional de Vigilância da Gripe–Relatório da Época 2011/2012; Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisbon, Portugal, 2013. [Google Scholar]
- Petri, E.; Ruiz-aragón, J.A. Comparison of Cost Effectiveness of Seasonal Influenza vaccines in Spain. In Proceedings of the IVW Conference, Albufeira, Portugal, 6–9 October 2015; p. 7015. [Google Scholar]
- Instituto Nacional de Saúde Doutor Ricardo Jorge. Boletim de Vigilância Epidemiológica da Gripe. Época 2017/2018-Semana 20; INSA: Lisbon, Portugal, 2018. [Google Scholar]
- Inverno e Saúde—Evolução Semanal. Available online: http://saudesazonal.min-saude.pt/#consultaSindromeGripal/149/757 (accessed on 9 May 2018).
- Administração Central do Sistema de Saúde, I.P. Base de dados Central de Morbilidade Hospitalar.
- Instituto Nacional de Estatística. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_indicadores&indOcorrCod=0008180&contexto=bd&selTab=tab2 (accessed on 4 May 2018).
- Coelho, P.P.; Morgado, C.; Albuquerque, D.D.; Rodrigues, M. Diário da República 1.a série, No153—Portaria 234/2015 de 7 de agosto—Anexo-II.; Ministério da Saúde: Lisbon, Portugal, 2015. [Google Scholar]
- BASE:Contratos Públicos Online. Available online: http://www.base.gov.pt/Base/pt/Homepage (accessed on 4 May 2018).
- George, F. Orientação 007/2015 Terapêutica e Quimioprofilaxia da Gripe Sazonal, época Gripal 2014/2015; Direção Geral da Saúde: Lisbon, Portugal, 2015; pp. 1–7. [Google Scholar]
- Serviços Partilhados do Ministério da Saúde. Contrato Fornecimento de V13—Vacina Contra a Gripe Inativada [UDOSE] à Administração Regional de Saúde do Alentejo; Ministério da Saúde: Lisbon, Portugal, 2016. [Google Scholar]
- BASE: Contratos Públicos Online. Available online: http://www.base.gov.pt/Base/pt/Pesquisa/Contrato?a=5864096 (accessed on 27 January 2020).
- Portal do INE—Índice de Preços no Consumidor (Taxa de Variação Média dos Últimos 12 Meses—Base 2012-%) por Localização Geográfica e Agregados Especiais; Mensal. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_indicadores&indOcorrCod=0002390&contexto=pi&selTab=tab0 (accessed on 13 January 2020).
- Boer, P.T.; Crépey, P.; Pitman, R.J.; Macabeo, B.; Chit, A.; Postma, M.J. Cost-Effectiveness of Quadrivalent versus Trivalent Influenza Vaccine in the United States. Value Health 2016, 19, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Rondy, M.; Kissling, E.; Emborg, H.-D.; Gherasim, A.; Pebody, R.; Trebbien, R.; Pozo, F.; Larrauri, A.; McMenamin, J.; Valenciano, M. I-MOVE/I-MOVE+ group Interim 2017/18 influenza seasonal vaccine effectiveness: Combined results from five European studies. Eurosurveillance 2018, 23, 18-00086. [Google Scholar] [CrossRef]
- Rondy, M.; Gherasim, A.; Casado, I.; Launay, O.; Rizzo, C.; Pitigoi, D.; Mickiene, A.; Marbus, S.D.; Machado, A.; Syrjänen, R.K.; et al. Low 2016/17 season vaccine effectiveness against hospitalised influenza A(H3N2) among elderly: Awareness warranted for 2017/18 season. Eurosurveillance 2017, 22, 17-00645. [Google Scholar] [CrossRef] [PubMed]
- Rondy, M.; Larrauri, A.; Casado, I.; Alfonsi, V.; Pitigoi, D.; Launay, O.; Syrjänen, R.K.; Gefenaite, G.; Machado, A.; Vučina, V.V.; et al. 2015/16 seasonal vaccine effectiveness against hospitalisation with influenza A (H1N1) pdm09 and B among elderly people in Europe: Results from the I-MOVE + project. Eurosurveillance 2015, 22, 30580. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B.; Machado, A.; Pechirra, P.; Falcão, I.; Gonçalves, P.; Conde, P.; Guiomar, R.; Batista, I.; Falcão, J.M. Efetividade da vacina antigripal na época 2010-2011 em Portugal: Resultados do projeto EuroEVA. Rev. Port. De Med. Geral E Fam. 2012, 28, 271–284. [Google Scholar] [CrossRef]
- Kissling, E.; Valenciano, M.; Pozo, F.; Vilcu, A.-M.; Reuss, A.; Rizzo, C.; Larrauri, A.; Horváth, J.K.; Brytting, M.; Domegan, L.; et al. 2015/16 I-MOVE/I-MOVE+ multicentre case control study in Europe: Moderate vaccine effectiveness estimates against influenza A(H1N1)pdm09 and low estimates against lineage mismatched influenza B among children. Influenza Other Respir Viruses 2018, 12, 423–437. [Google Scholar] [CrossRef]
- You, J.H.S.; Ming, W.K.; Chan, P.K.S. Cost-effectiveness analysis of quadrivalent influenza vaccine versus trivalent influenza vaccine for elderly in Hong Kong. BMC Infect. Dis. 2014, 14, 618. [Google Scholar] [CrossRef]
- You, J.H.S.; Ming, W.K.; Chan, P.K.S. Cost-effectiveness of quadrivalent influenza vaccine in Hong Kong—A decision analysis. Hum. Vaccines Immunother. 2015, 11, 564–571. [Google Scholar] [CrossRef]
- Chit, A.; Roiz, J.; Aballea, S. An assessment of the expected cost-effectiveness of quadrivalent influenza vaccines in Ontario, Canada using a static model. PLoS ONE 2015, 10, e0133606. [Google Scholar] [CrossRef]
- Chit, A.; Roiz, J.; Briquet, B.; Greenberg, D.P. Expected cost effectiveness of high-dose trivalent influenza vaccine in US seniors. Vaccine 2015, 33, 734–741. [Google Scholar] [CrossRef]
- Van Bellinghen, L.A.; Meier, G.; Van Vlaenderen, I. The potential cost-effectiveness of quadrivalent versus trivalent influenza vaccine in elderly people and clinical risk groups in the UK: A lifetime multi-cohort model. PLoS ONE 2014, 9, e98437. [Google Scholar] [CrossRef] [PubMed]
- Dolk, C.; Eichner, M.; Welte, R.; Anastassopoulou, A.; Van Bellinghen, L.A.; Poulsen Nautrup, B.; Vlaenderen, I.V.; Schmidt-Ott, R.; Schwehm, M.; Postma, M. Cost-Utility of Quadrivalent Versus Trivalent Influenza Vaccine in Germany, Using an Individual-Based Dynamic Transmission Model. Pharmacoeconomics 2016, 34, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Ortiz de Lejarazu, R.; Reina, J.; Callejo, D.; Cuervo, J.; Morano Larragueta, R. Cost–effectiveness analysis of quadrivalent influenza vaccine in Spain. Hum. Vaccines Immunother. 2016, 12, 2269–2277. [Google Scholar] [CrossRef] [PubMed]
- Meier, G.; Gregg, M.; Poulsen Nautrup, B. Cost-effectiveness analysis of quadrivalent influenza vaccination in at-risk adults and the elderly: An updated analysis in the UK. J. Med. Econ. 2015, 18, 746–761. [Google Scholar] [CrossRef] [PubMed]
- Mullikin, M.; Tan, L.; Jansen, J.P.; Van Ranst, M.; Farkas, N.; Petri, E. A Novel Dynamic Model for Health Economic Analysis of Influenza Vaccination in the Elderly. Infect. Dis. Ther. 2015, 4, 459–487. [Google Scholar] [CrossRef][Green Version]
- Capri, S.; Barbieri, M.; de Waure, C.; Boccalini, S.; Panatto, D. Cost-effectiveness analysis of different seasonal influenza vaccines in the elderly Italian population. Hum. Vaccines Immunother. 2018, 14, 1331–1341. [Google Scholar] [CrossRef]
- Aguiar, M.; Dosi, G.; Knopoff, D.A.; Virgillito, M.E. A multiscale network-based model of contagion dynamics: Heterogeneity, spatial distancing and vaccination. Math. Models Methods Appl. Sci. 2021, 31, 2425–2454. [Google Scholar] [CrossRef]
- Weinstein, M.C.; Torrance, G.; McGuire, A. QALYs: The Basics. Value Health 2009, 12, S5–S9. [Google Scholar] [CrossRef]
| Input Parameters | Base Case | Source |
|---|---|---|
| Population | 2,067,654 | [26] |
| Vaccination coverage rate | 0.501 | [7] |
| Trivalent vaccine effectiveness | 0.580 | [12] |
| Quadrivalent vaccine effectiveness | 0.599 | [27,28,29,30,31,32,33,34,35] |
| Probability of influenza-like illness | 0.007 | [7,12,26,31,36] |
| Probability of confirmed influenza | 0.278 | [31] |
| Probability of GP consultation | 0.952 | [31,36] |
| Probability of hospitalization due to influenza | 0.091 | [37] |
| Probability of death when hosp. influenza | 0.079 | [37] |
| Probability of hospitalization due to pneumonia | 0.008 | [37] |
| Probability of death due to hosp. pneumonia | 0.381 | [37] |
| Probability of hospitalization due to respiratory disease | 0.014 | [37] |
| Probability of death when hosp. RD | 0.050 | [37] |
| Probability of hospitalization due to heart disease | 0.026 | [37] |
| Probability of death when hosp. HD | 0.110 | [37] |
| Probability of death when no confirmed influenza | 0.043 | [26,38] |
| Cost of GP consultation | 31.000 | [39] |
| Cost of hospitalization due to influenza | 2644.011 | [37,39] |
| Cost of death when hospitalization due to influenza | 6364.597 | [37,39] |
| Cost of hospitalization due to pneumonia | 2906.749 | [37,39] |
| Cost of death when hospitalization due to pneumonia | 11,298.219 | [37,39] |
| Cost of hospitalization due to respiratory disease | 2534.841 | [37,39] |
| Cost of death when hospitalization due to RD | 1363.460 | [37,39] |
| Cost of hospitalization due to heart disease | 2978.842 | [37,39] |
| Cost of death when hospitalization due to HD | 7785.951 | [37,39] |
| Cost of antiviral treatment | 18.300 | [40,41] |
| Cost of trivalent vaccine | 2.576 | [42] |
| Cost of quadrivalent vaccine | 7.692 | [43,44] |
| Cost of vaccine administration | 3.700 | [39] |
| Disutility associated with ILI without influenza confirmation | 0.009 | [17] |
| Disutility associated with no hospitalized influenza | 0.009 | [17] |
| Disutility associated with hospitalization due to influenza | 0.031 | [17] |
| Disutility associated with hospitalization due to pneumonia | 0.031 | [17] |
| Disutility associated with hospitalization due to respiratory disease | 0.031 | [17] |
| Disutility associated with hospitalization due to heart disease | 0.031 | [17] |
| Utility associated with healthy population | 0.625 | [16,26] |
| Parameter | TIV | QIV | Difference (QIV-TIV) |
|---|---|---|---|
| Events | |||
| GP consultations | 2631.35 | 2595.77 | −35.58 |
| Hospitalizations due to influenza | 239.00 | 235.77 | −3.23 |
| Deaths due to influenza hospitalization | 19.00 | 18.74 | −0.26 |
| Hospitalizations due to pneumonia | 21.00 | 20.72 | −0.28 |
| Deaths due to pneumonia hospitalization | 8.00 | 7.89 | −0.11 |
| Hospitalizations due to RD | 40.00 | 39.46 | −0.54 |
| Deaths due to RD hospitalization | 2.00 | 1.97 | −0.03 |
| Hospitalizations due to HD | 73.00 | 72.01 | −0.99 |
| Deaths due to HD hospitalization | 8.00 | 7.89 | −0.11 |
| Vaccine doses | 1,035,895 | 1,035,895 | 0.00 |
| Costs | |||
| GP consultations | EUR 81,572 | EUR 80,469 | EUR −1103 |
| Hospitalizations due to influenza | EUR 710,464 | EUR 701,189 | EUR −9275 |
| Deaths due to influenza hospitalization | EUR 121,552 | EUR 119,934 | EUR −1617 |
| Hospitalizations due to pneumonia | EUR 128,213 | EUR 126,508 | EUR −1705 |
| Deaths due to pneumonia hospitalization | EUR 90,401 | EUR 89,189 | EUR −1211 |
| Hospitalizations due to RD | EUR 99,125 | EUR 97,841 | EUR −1285 |
| Deaths due to RD hospitalization | EUR 2731 | EUR 2697 | EUR −34 |
| Hospitalizations due to HD | EUR 256,048 | EUR 252,687 | EUR −3361 |
| Deaths due to HD Hospitalization | EUR 62,303 | EUR 61,471 | EUR −831 |
| Vaccine doses | EUR 2,668,465 | EUR 7,968,557 | EUR 5,300,092 |
| Total | EUR 8,018,570 | EUR 13,301,618 | EUR 5,283,047 |
| QALYs (Total) | 1,265,456.95 | 1,265,457.15 | 0.20 |
| ICER (EUR/QALY) | 26,403,007 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tavares, D.; Mouriño, H.; Rodríguez, C.A.; Saborido, C.M. Cost Effectiveness of Quadrivalent Versus Trivalent Inactivated Influenza Vaccines for the Portuguese Elderly Population. Vaccines 2022, 10, 1285. https://doi.org/10.3390/vaccines10081285
Tavares D, Mouriño H, Rodríguez CA, Saborido CM. Cost Effectiveness of Quadrivalent Versus Trivalent Inactivated Influenza Vaccines for the Portuguese Elderly Population. Vaccines. 2022; 10(8):1285. https://doi.org/10.3390/vaccines10081285
Chicago/Turabian StyleTavares, Diana, Helena Mouriño, Cristina Antón Rodríguez, and Carlos Martín Saborido. 2022. "Cost Effectiveness of Quadrivalent Versus Trivalent Inactivated Influenza Vaccines for the Portuguese Elderly Population" Vaccines 10, no. 8: 1285. https://doi.org/10.3390/vaccines10081285
APA StyleTavares, D., Mouriño, H., Rodríguez, C. A., & Saborido, C. M. (2022). Cost Effectiveness of Quadrivalent Versus Trivalent Inactivated Influenza Vaccines for the Portuguese Elderly Population. Vaccines, 10(8), 1285. https://doi.org/10.3390/vaccines10081285






