Safety Messaging Boosts Parental Vaccination Intention for Children Ages 5–11
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Design
2.1.1. Intervention Texts
2.1.2. Randomization
2.2. Participants and Sample Size
2.3. Data Collection
2.4. Outcome Variables
2.4.1. Primary Outcome Variable 1: Parental Vaccination Intention
2.4.2. Primary Outcome Variable 2: Children-Oriented COVID-19 Vaccine Hesitancy Scale
2.4.3. Important Covariates
2.5. Statistical Analysis
3. Results
3.1. Background Characteristics of the Participants
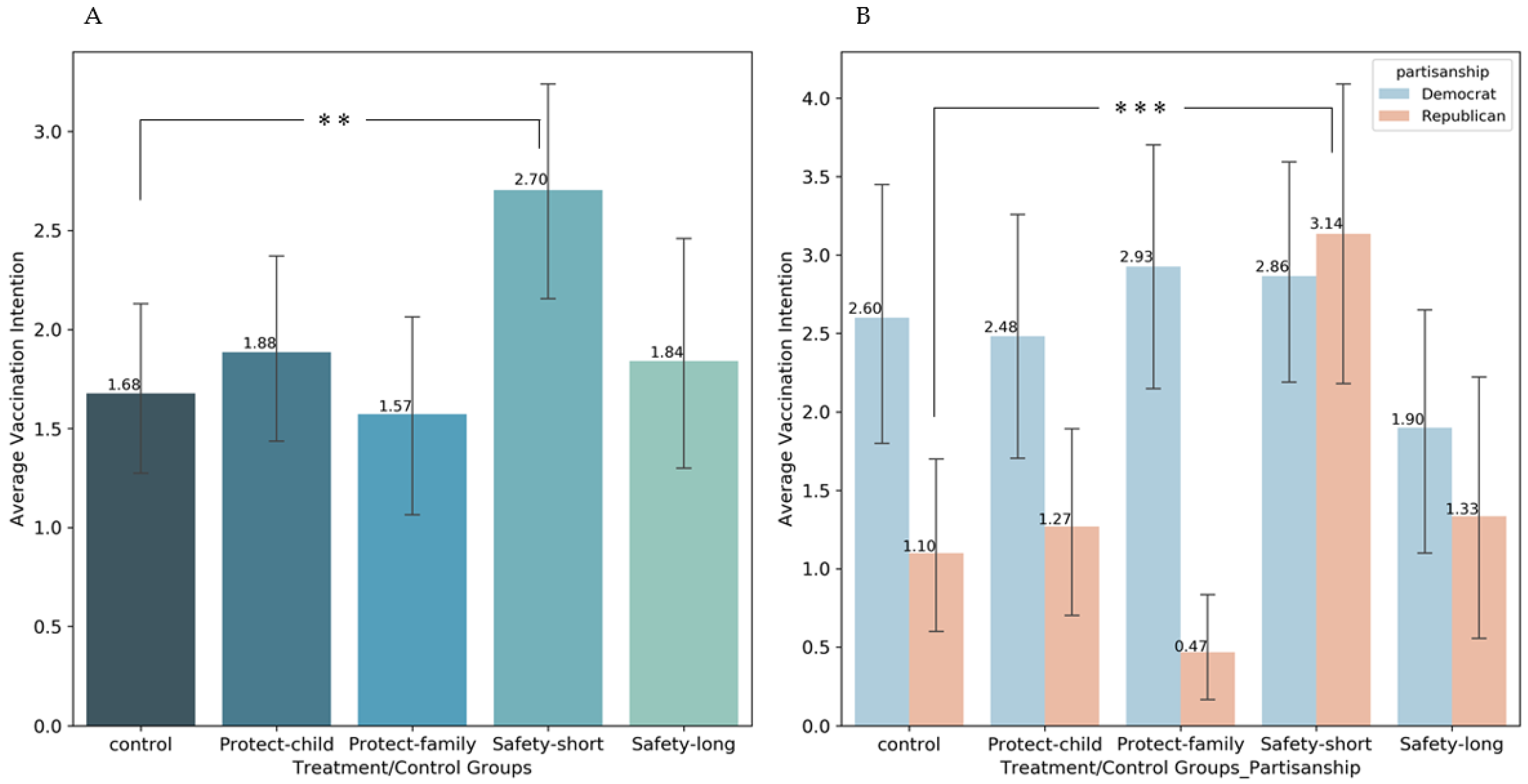
3.2. Parental Vaccination Intention
3.3. Mediation Analysis on Vaccine Hesitancy
3.4. Partisanship Heterogeneity Effects
4. Discussion
- (1)
- The mechanisms of all the interesting heterogeneity effects are not fully explored, and the explanations are therefore not thorough. The limited size (mainly due to difficulties in data collection: we could recruit only 15–20 effective participants per day according to the basic screening rules of CloudResearch, as the proportion of participants with unvaccinated children ages 5–11 is low) and sample representativeness may also lead to our inability to consummately address the mechanism problems. For instance, although the partisanship difference is intriguing, we need to acknowledge that this heterogeneity effect needs to be further addressed by more statistical evidence in larger samples. Further work is needed to identify the detailed mechanisms of the attitude change process.
- (2)
- The external validity of these information interventions is still pending study. Vaccination intention does not necessarily equate to vaccination behavior. Despite the high correlation between intention and behavior [48], there are still gaps between them [49], and the long-term effects of information interventions still await future study. It will be interesting and meaningful for future researchers to test these interventions in the field to assess the real-world power of vaccine messaging.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Flaxman, S.; Whittaker, C.; Semenova, E.; Rashid, T.; Parks, R.; Blenkinsop, A.; Unwin, H.J.T.; Mishra, S.; Bhatt, S.; Gurdasani, D.; et al. COVID-19 is a leading cause of death in children and young people ages 0–19 years in the United States. medRxiv 2022. [Google Scholar] [CrossRef]
- Ludvigsson, J.F. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID-19. Acta Paediatr. 2021, 110, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Selden, T.M.; Berdahl, T.A.; Fang, Z. The Risk of Severe COVID-19 Within Households of School Employees and School-Age Children: Study examines how often persons at risk of severe COVID-19 were connected to schools, either as employees or by living in the same households as school employees or school-age children. Health Affairs 2020, 39, 2002–2009. [Google Scholar] [PubMed]
- Walter, E.B.; Talaat, K.R.; Sabharwal, C.; Gurtman, A.; Lockhart, S.; Paulsen, G.C.; Barnett, E.D.; Muñoz, F.M.; Maldonado, Y.; Pahud, B.A.; et al. Evaluation of the BNT162b2 COVID-19 vaccine in children 5 to 11 years of age. N. Engl. J. Med. 2022, 386, 35–46. [Google Scholar] [CrossRef]
- Hause, A.M.; Baggs, J.; Marquez, P.; Myers, T.R.; Gee, J.; Su, J.R.; Zhang, B.; Thompson, D.; Shimabukuro, T.T.; Shay, D.K. COVID-19 vaccine safety in children aged 5–11 years—United States, November 3–December 19 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 1755. [Google Scholar] [CrossRef]
- Capponi, M.; Pulvirenti, F.; Cinicola, B.L.; Brindisi, G.; Conti, M.G.; Colaiocco, G.; de Castro, G.; Guido, C.A.; Duse, M.; Midulla, F.; et al. Short-Term Side Effects and SARS-CoV-2 Infection after COVID-19 Pfizer–BioNTech Vaccine in Children Aged 5–11 Years: An Italian Real-World Study. Vaccines 2022, 10, 1056. [Google Scholar] [CrossRef]
- Su, J.R.; Vaccine Safety Team. COVID-19 Vaccine Safety Updates: Primary Series in Children and Adolescents Ages 5–11 and 12–15 Years, and Booster Doses in Adolescents Ages 16–24 Years; CDC: Atlanta, GA, USA, 2022. [Google Scholar]
- Centers for Disease Control and Prevention. COVID Data Tracker; US Department of Health and Human Services, CDC: Atlanta, GA, USA, 2022. Available online: https://covid.cdc.gov/covid-data-tracker (accessed on 6 July 2022).
- Sadaf, A.; Richards, J.L.; Glanz, J.; Salmon, D.A.; Omer, S.B. A systematic review of interventions for reducing parental vaccine refusal and vaccine hesitancy. Vaccine 2013, 31, 4293–4304. [Google Scholar] [CrossRef]
- Troiano, G.; Nardi, A. Vaccine hesitancy in the era of COVID-19. Public Health 2021, 194, 245–251. [Google Scholar] [CrossRef]
- Kates, J.; Tolbert, J.; Rouw, A. Update on COVID-19 Vaccination of 5–11 Year Olds in the U.S.; KFF: Oakland, CA, USA, 2022; Available online: https://www.kff.org/coronavirus-covid-19/issue-brief/update-on-covid-19-vaccination-of-5-11-year-olds-in-the-u-s/ (accessed on 19 July 2022).
- Szilagyi, P.G.; Shah, M.D.; Delgado, J.R.; Thomas, K.; Vizueta, N.; Cui, Y.; Vangala, S.; Shetgiri, R.; Kapteyn, A. Parents’ intentions and perceptions about COVID-19 vaccination for their children: Results from a national survey. Pediatrics 2021, 148, 1–11. [Google Scholar] [CrossRef]
- Ruggiero, K.M.; Wong, J.; Sweeney, C.F.; Avola, A.; Auger, A.; Macaluso, M.; Reidy, P. Parents’ intentions to vaccinate their children against COVID-19. J. Pediatric Health Care 2021, 35, 509–517. [Google Scholar] [CrossRef]
- Goldman, R.D.; Yan, T.D.; Seiler, M.; Cotanda, C.P.; Brown, J.C.; Klein, E.J.; Hoeffe, J.; Gelernter, R.; Hall, J.E.; Davis, A.L.; et al. Caregiver willingness to vaccinate their children against COVID-19: Cross sectional survey. Vaccine 2020, 38, 7668–7673. [Google Scholar] [CrossRef]
- Teasdale, C.A.; Borrell, L.N.; Kimball, S.; Rinke, M.L.; Rane, M.; Fleary, S.A.; Nash, D. Plans to vaccinate children for coronavirus disease 2019: A survey of United States parents. J. Pediatrics 2021, 237, 292–297. [Google Scholar] [CrossRef]
- Yılmaz, M.; Sahin, M.K. Parents’ willingness and attitudes concerning the COVID-19 vaccine: A cross-sectional study. Int. J. Clin. Pract. 2021, 75, e14364. [Google Scholar] [CrossRef]
- Yigit, M.; Ozkaya-Parlakay, A.; Senel, E. Evaluation of COVID-19 vaccine refusal in parents. Pediatric Infect. Dis. J. 2021, 40, e134–e136. [Google Scholar] [CrossRef]
- Zhang, M.X.; Lin, X.Q.; Chen, Y.; Tung, T.H.; Zhu, J.S. Determinants of parental hesitancy to vaccinate their children against COVID-19 in China. Expert Rev. Vaccines 2021, 20, 1339–1349. [Google Scholar] [CrossRef]
- Montalti, M.; Rallo, F.; Guaraldi, F.; Bartoli, L.; Po, G.; Stillo, M.; Perrone, P.; Squillace, L.; Dallolio, L.; Pandolfi, P.; et al. Would parents get their children vaccinated against SARS-CoV-2? Rate and predictors of vaccine hesitancy according to a survey over 5000 families from Bologna, Italy. Vaccines 2021, 9, 366. [Google Scholar] [CrossRef]
- Rossen, I.; Hurlstone, M.J.; Lawrence, C. Going with the grain of cognition: Applying insights from psychology to build support for childhood vaccination. Front. Psychol. 2016, 7, 1483. [Google Scholar] [CrossRef] [Green Version]
- Pennings, S.; Symons, X. Persuasion, not coercion or incentivisation, is the best means of promoting COVID-19 vaccination. J. Med. Ethics 2021, 47, 709–711. [Google Scholar] [CrossRef]
- Lewandowsky, S.; Cook, J.; Ecker, U.; Albarracin, D.; Amazeen, M.; Kendou, P.; Lombardi, D.; Newman, E.; Pennycook, G.; Porter, E.; et al. The Debunking Handbook 2020; Databrary: New York, NY, USA, 2020. [Google Scholar] [CrossRef]
- Janz, N.K.; Becker, M.H. The health belief model: A decade later. Health Educ. Q. 1984, 11, 1–47. [Google Scholar] [CrossRef] [Green Version]
- Champion, V.L.; Skinner, C.S. The health belief model. In Health Behavior and Health Education: Theory, Research, and Practice; Jossey-Bass: Hoboken, NJ, USA, 2008; pp. 45–65. [Google Scholar]
- Smith, L.E.; Amlôt, R.; Weinman, J.; Yiend, J.; Rubin, G.J. A systematic review of factors affecting vaccine uptake in young children. Vaccine 2017, 35, 6059–6069. [Google Scholar] [CrossRef] [Green Version]
- Prentice-Dunn, S.; Rogers, R.W. Protection motivation theory and preventive health: Beyond the health belief model. Health Educ. Res. 1986, 1, 153–161. [Google Scholar] [CrossRef]
- Milne, S.; Sheeran, P.; Orbell, S. Prediction and intervention in health-related behavior: A meta-analytic review of protection motivation theory. J. Appl. Soc. Psychol. 2000, 30, 106–143. [Google Scholar] [CrossRef]
- Gidengil, C.; Chen, C.; Parker, A.M.; Nowak, S.; Matthews, L. Beliefs around childhood vaccines in the United States: A systematic review. Vaccine 2019, 37, 6793–6802. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.R.; Freeman, A.L.; Marteau, T.M.; van der Linden, S. Effect of information about COVID-19 vaccine effectiveness and side effects on behavioural intentions: Two online experiments. Vaccines 2021, 9, 379. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.J.; Salmon, D.A.; Marcuse, E.K. Building Trust to Achieve Confidence in COVID-19 Vaccines. JAMA Netw. Open 2020, 3, e2025672. [Google Scholar] [CrossRef] [PubMed]
- Kitta, A.; Goldberg, D.S. The significance of folklore for vaccine policy: Discarding the deficit model. Crit. Public Health 2017, 27, 506–514. [Google Scholar] [CrossRef]
- Petty, R.E.; Cacioppo, J.T. The elaboration likelihood model of persuasion. In Communication and Persuasion; Springer: New York, NY, USA, 1986; pp. 1–24. [Google Scholar]
- Petty, R.E.; Barden, J.; Wheeler, S.C. The elaboration likelihood model of persuasion: Developing health promotions for sustained behavioral change. In Emerging Theories in Health Promotion Practice and Research; Jossey-Bass/Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Chiang, K.P.; Jackson, A. Health literacy and its outcomes: Application and extension of elaboration likelihood model. Int. J. Healthc. Manag. 2013, 6, 152–157. [Google Scholar] [CrossRef]
- Chen, S.; Chaiken, S. The heuristic-systematic model in its broader context. In Dual-Process Theories in Social Psychology; The Guilford Press: New York, NY, USA, 2019. [Google Scholar]
- Todorov, A.; Chaiken, S.; Henderson, M.D. The heuristic-systematic model of social information processing. The persuasion handbook: Developments in theory and practice. J. Personal. Soc. Psychol. 2002, 23, 195–211. [Google Scholar]
- Chaiken, S. Heuristic versus systematic information processing and the use of source versus message cues in persuasion. J. Personal. Soc. Psychol. 1980, 39, 752. [Google Scholar] [CrossRef]
- Ashworth, M.; Thunström, L.; Cherry, T.L.; Newbold, S.C.; Finnoff, D.C. Emphasize personal health benefits to boost COVID-19 vaccination rates. Proc. Natl. Acad. Sci. USA 2021, 118, e2108225118. [Google Scholar] [CrossRef]
- Motta, M.; Sylvester, S.; Callaghan, T.; Lunz-Trujillo, K. Encouraging COVID-19 vaccine uptake through effective health communication. Front. Political Sci. 2021, 3. [Google Scholar] [CrossRef]
- Palm, R.; Bolsen, T.; Kingsland, J.T. The effect of frames on COVID-19 vaccine resistance. Front. Political Sci. 2021, 3, 661257. [Google Scholar] [CrossRef]
- Loomba, S.; de Figueiredo, A.; Piatek, S.J.; de Graaf, K.; Larson, H.J. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat. Hum. Behav. 2021, 5, 337–348. [Google Scholar] [CrossRef]
- Shapiro, G.K.; Tatar, O.; Dube, E.; Amsel, R.; Knauper, B.; Naz, A.; Perez, S.; Rosberger, Z. The vaccine hesitancy scale: Psychometric properties and validation. Vaccine 2018, 36, 660–667. [Google Scholar] [CrossRef]
- Grotta, A.; Bellocco, R. A review of mediation analysis in Stata: Principles, methods and applications. In Proceedings of the Italian Stata Users Group Meeting, Firenze, Italy, 14 November 2013. [Google Scholar]
- Fridman, A.; Gershon, R.; Gneezy, A. COVID-19 and vaccine hesitancy: A longitudinal study. PLoS ONE 2021, 16, e0250123. [Google Scholar] [CrossRef]
- Limaye, R.J.; Sauer, M.; Truelove, S.A. Politicizing public health: The powder keg of rushing COVID-19 vaccines. Hum. Vaccines Immunother. 2021, 17, 1662–1663. [Google Scholar] [CrossRef]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Decis. Processes 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Godin, G.; Kok, G. The theory of planned behavior: A review of its applications to health-related behaviors. Am. J. Health Promot. 1996, 11, 87–98. [Google Scholar] [CrossRef]
- Lambooij, M.S.; Harmsen, I.A.; Veldwijk, J.; de Melker, H.; Mollema, L.; van Weert, Y.W.; de Wit, G.A. Consistency between stated and revealed preferences: A discrete choice experiment and a behavioural experiment on vaccination behaviour compared. BMC Med. Res. Methodol. 2015, 15, 19. [Google Scholar] [CrossRef] [Green Version]
- Bronchetti, E.T.; Huffman, D.B.; Magenheim, E. Attention, intentions, and follow-through in preventive health behavior: Field experimental evidence on flu vaccination. J. Econ. Behav. Organ. 2015, 116, 270–291. [Google Scholar] [CrossRef] [Green Version]
| Condition | n | Content | Reference | Length in Words |
|---|---|---|---|---|
| Control | 47 | A placeholder, no extra information on vaccination. | - | 112 |
| Protect-child | 55 | Information about how COVID-19 vaccines could prevent the children from adverse outcomes of contracting the virus. | Ashworth et al. (2021) [38] | 137 |
| Protect-family | 44 | Information about how COVID-19 vaccines could protect the whole family by preventing the child from infection. | Ashworth et al. (2021) [38] | 130 |
| Safety-short | 57 | Short, concise information about the lower doses (and thus, a lower side effect rate) of pediatric COVID-19 vaccines and the rigorous processes of approval. | U.S. FDA’s webpage | 152 |
| Safety-long | 40 | Long, detailed information about the lower doses (and thus, a lower side effect rate) of pediatric COVID-19 vaccines and the rigorous processes of approval. | U.S. FDA’s webpage | 359 |
| Title 1 | Mean | SD | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. COVID-19 vaccines are important for my child’s health. | 2.74 | 1.42 | |||||||||
| 2. Getting a COVID-19 vaccine is a good way to protect my child/children from the disease. | 2.81 | 1.40 | 0.90 * | ||||||||
| 3. COVID-19 vaccines are effective for children. | 2.88 | 1.30 | 0.81 * | 0.83 * | |||||||
| 4. Having my child vaccinated is important for the health of others in my community. | 2.91 | 1.44 | 0.85 * | 0.85 * | 0.80 * | ||||||
| 5. Children’s COVID-19 vaccines offered by the government program in my community are beneficial. | 2.84 | 1.38 | 0.86 * | 0.91 * | 0.85 * | 0.84 * | |||||
| 6. The information I receive about COVID-19 vaccines from the vaccine program is reliable and trustworthy. | 2.93 | 1.33 | 0.76 * | 0.78 * | 0.78 * | 0.76 * | 0.79 * | ||||
| 7. Generally I do what my doctor or health care provider recommends about COVID-19 vaccines for my child/children. | 3.23 | 1.27 | 0.51 * | 0.53 * | 0.54 * | 0.47 * | 0.55 * | 0.48 * | |||
| 8. COVID-19 vaccines carry more risks than influenza vaccines. | 3.49 | 1.21 | −0.29 * | −0.32 * | −0.26 * | −0.27 * | −0.30 * | −0.26 * | −0.05 | ||
| 9. I am concerned about serious adverse effects of children’s COVID-19 vaccines. | 3.94 | 1.21 | −0.30 * | −0.33 * | −0.29 * | −0.27 * | −0.28 * | −0.29 * | −0.06 | 0.65 * |
| Characteristics | N | % | |
|---|---|---|---|
| Sociodemographic characteristics | |||
| Gender | |||
| Male | 78 | 32.1 | |
| Female | 163 | 67.08 | |
| Others | 2 | 0.82 | |
| Age group, years | |||
| 18–34 | 116 | 47.74 | |
| 35–54 | 118 | 48.56 | |
| 55 or above | 9 | 3.7 | |
| Partisanship identity | |||
| Strong Republican | 28 | 11.52 | |
| Republican | 46 | 18.93 | |
| Independent Leaning Republican | 27 | 11.11 | |
| Independent | 47 | 19.34 | |
| Independent Leaning Democrat | 33 | 13.58 | |
| Democrat | 49 | 20.16 | |
| Strong Democrat | 13 | 5.35 | |
| Ethnicity | |||
| White | 179 | 73.66 | |
| Hispanic | 15 | 6.17 | |
| Black | 19 | 7.82 | |
| Asian | 21 | 8.64 | |
| Others | 9 | 3.7 | |
| Highest level of education | |||
| Some high school or less | 0 | 0 | |
| High school graduate | 35 | 14.4 | |
| Completed some college, but no degree | 42 | 17.28 | |
| Associate’s degree | 31 | 12.76 | |
| Bachelor’s degree | 102 | 41.98 | |
| Master’s or professional degree | 27 | 11.11 | |
| Doctorate degree | 6 | 2.47 | |
| Number of people in the household | |||
| 1 | 1 | 0.41 | |
| 2 | 29 | 11.93 | |
| 3 | 68 | 27.98 | |
| 4 | 94 | 38.68 | |
| 5 | 37 | 15.23 | |
| 6 or more | 14 | 5.76 | |
| Annual household income | |||
| $25,000 or less | 26 | 10.7 | |
| $25,000–$35,000 | 25 | 10.29 | |
| $35,000–$45,000 | 29 | 11.93 | |
| $45,000–$55,000 | 29 | 11.93 | |
| $55,000–$70,000 | 52 | 21.4 | |
| $70,000–$85,000 | 23 | 9.47 | |
| $85,000–$100,000 | 15 | 6.17 | |
| $100,000–$120,000 | 13 | 5.35 | |
| $120,000–$140,000 | 12 | 4.94 | |
| $140,000 or more | 19 | 7.82 | |
| History of COVID-19 and COVID-19 vaccination | |||
| Own status of COVID-19 vaccination | |||
| I am fully vaccinated and had got a booster (3rd shot). | 69 | 28.4 | |
| I am fully vaccinated but had not gotten a booster. | 68 | 27.98 | |
| I am partially vaccinated (with one shot). | 9 | 3.7 | |
| I am not vaccinated. | 97 | 39.92 | |
| Ever experienced any side effects or not | |||
| Yes | 68 | 46.58 | |
| No | 78 | 53.42 | |
| Severity of the vaccination side effect experiences | |||
| Very severe | 9 | 13.24 | |
| Severe | 8 | 11.76 | |
| Moderate | 25 | 36.76 | |
| Mild | 22 | 32.35 | |
| Very mild | 4 | 5.88 | |
| Child ever tested positive for COVID-19 | |||
| No | 124 | 82.67 | |
| Yes | 26 | 17.33 | |
| Youngest child (if more than one child in the family) | |||
| No | 66 | 70.97 | |
| Yes | 27 | 29.03 | |
| Oldest child (if more than one child in the family) | |||
| No | 70 | 75.27 | |
| Yes | 23 | 24.73 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, Z.; Liu, L.; Li, D.; Wu, S.J.; Zhai, X. Safety Messaging Boosts Parental Vaccination Intention for Children Ages 5–11. Vaccines 2022, 10, 1205. https://doi.org/10.3390/vaccines10081205
Cui Z, Liu L, Li D, Wu SJ, Zhai X. Safety Messaging Boosts Parental Vaccination Intention for Children Ages 5–11. Vaccines. 2022; 10(8):1205. https://doi.org/10.3390/vaccines10081205
Chicago/Turabian StyleCui, Zhihan, Lu Liu, Dan Li, Sherry Jueyu Wu, and Xinyue Zhai. 2022. "Safety Messaging Boosts Parental Vaccination Intention for Children Ages 5–11" Vaccines 10, no. 8: 1205. https://doi.org/10.3390/vaccines10081205
APA StyleCui, Z., Liu, L., Li, D., Wu, S. J., & Zhai, X. (2022). Safety Messaging Boosts Parental Vaccination Intention for Children Ages 5–11. Vaccines, 10(8), 1205. https://doi.org/10.3390/vaccines10081205







