Overview of Tools and Measures Investigating Vaccine Hesitancy in a Ten Year Period: A Scoping Review
Abstract
:1. Introduction
1.1. Background
1.2. Review Rationale
1.3. Review Objective
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
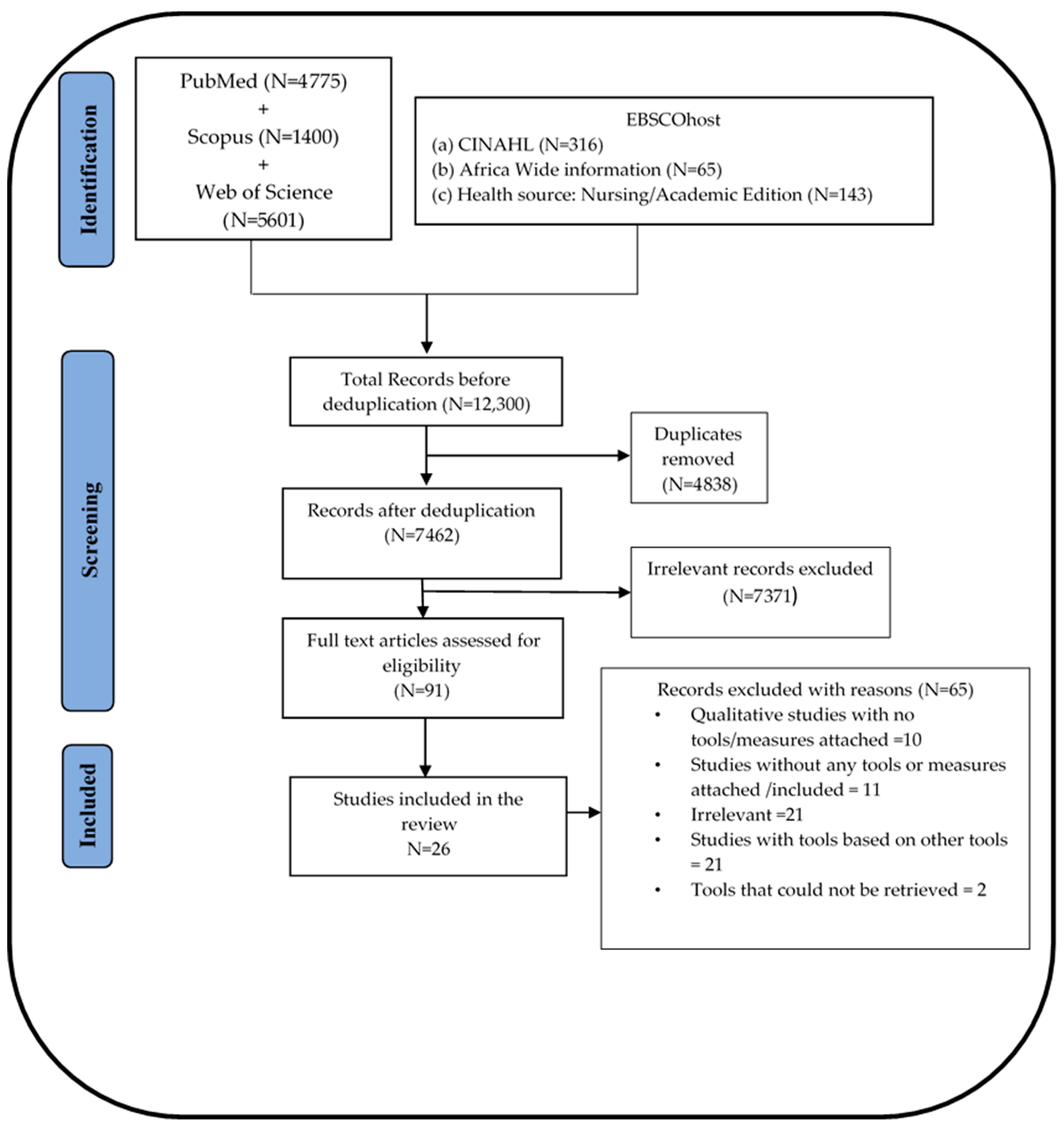
2.3. Study Selection
2.4. Data Charting
2.5. Data Synthesis and Analysis
3. Results
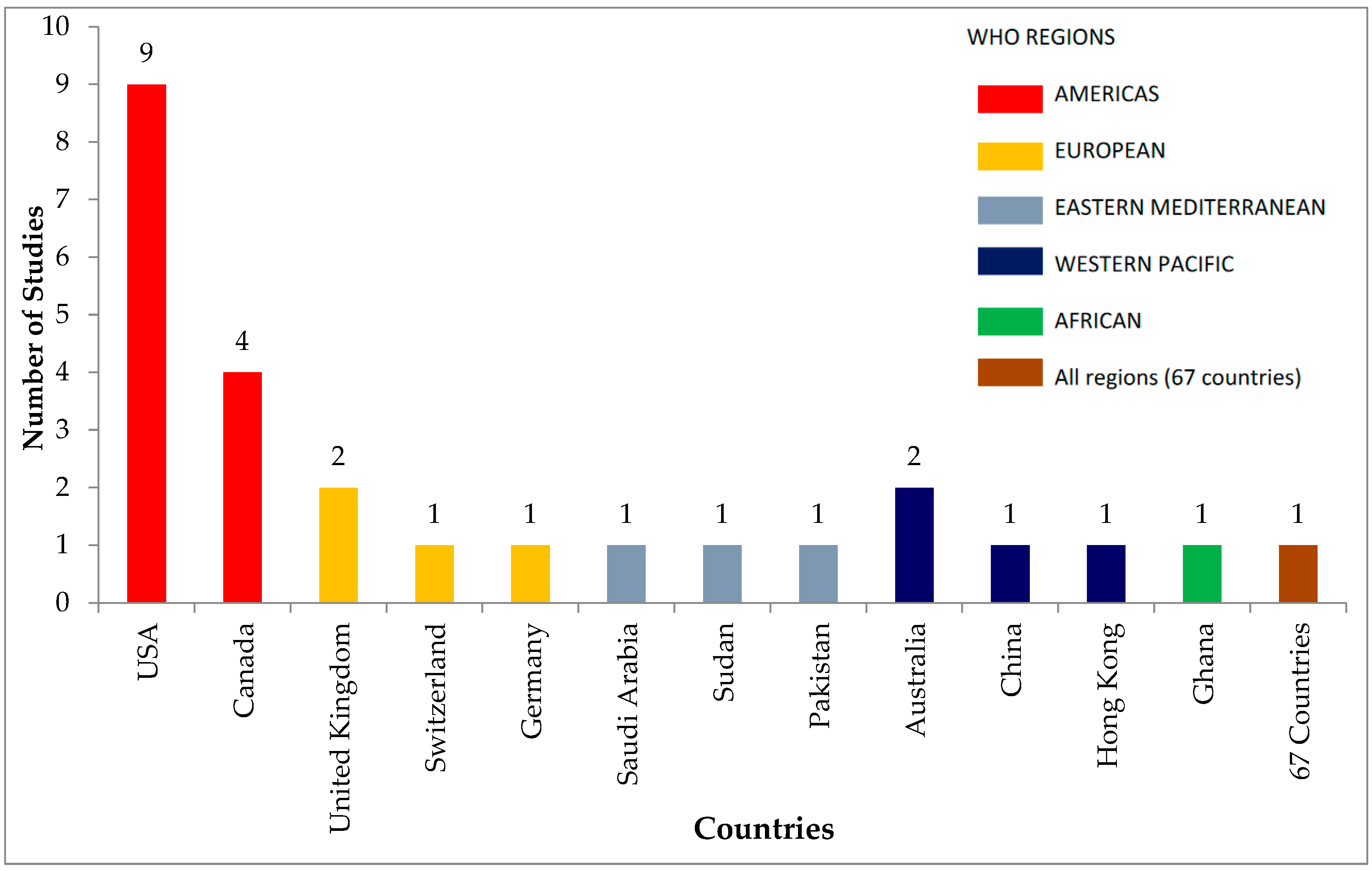
3.1. General Characteristics of Included Studies
3.2. Synopsis of Each Included Tool or Measure
3.2.1. Quantitative Tools or Measures
3.2.2. Qualitative Tools/Measures
3.2.3. Major Similarities and Differences of the Tools and Measures
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stratford, J.; MacKenzie, E.; Mockford, E. Balancing Speed and Safety: The Authorisation of COVID-19 Vaccines and Medicines. Judic. Rev. 2020, 25, 105–117. [Google Scholar] [CrossRef]
- Razai, M.S.; Oakeshott, P.; Esmail, A.; Wiysonge, C.S.; Viswanath, K.; Mills, M.C. COVID-19 vaccine hesitancy: The five Cs to tackle behavioural and sociodemographic factors. J. R. Soc. Med. 2021, 114, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Betsch, C.; Schmid, P.; Heinemeier, D.; Korn, L.; Holtmann, C.; Böhm, R. Beyond confidence: Development of a measure assessing the 5C psychological antecedents of vaccination. PLoS ONE 2018, 13, e0208601. [Google Scholar] [CrossRef] [Green Version]
- Cooper, S.; Betsch, C.; Sambala, E.Z.; Mchiza, N.; Wiysonge, C.S. Vaccine hesitancy–a potential threat to the achievements of vaccination programmes in Africa. Hum. Vaccines Immunother. 2018, 14, 2355–2357. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, N.E.; Eskola, J.; Liang, X.; Chaudhuri, M.; Dube, E.; Gellin, B.; Goldstein, S.; Larson, H.; Manzo, M.L.; Reingold, A. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef]
- World Health Organization. Understanding the behavioural and Social Drivers of Vaccine Uptake WHO Position Paper—May 2022. Weekly Epidemiological Record 2022:209–24. Available online: https://apps.who.int/iris/bitstream/handle/10665/354458/WER9720-eng-fre.pdf (accessed on 10 June 2022).
- Bloom, B.R.; Marcuse, E.; Mnookin, S. Addressing Vaccine Hesitancy. Science 2014, 344, 339. [Google Scholar] [CrossRef] [Green Version]
- Siddiqui, M.; Salmon, D.A.; Omer, S.B. Epidemiology of vaccine hesitancy in the United States. Hum. Vaccines Immunother. 2013, 9, 2643–2648. [Google Scholar] [CrossRef] [Green Version]
- Oduwole, E.O.; Pienaar, E.D.; Mahomed, H.; Wiysonge, C.S. Current tools available for investigating vaccine hesitancy: A scoping review protocol. BMJ Open 2019, 9, e033245. [Google Scholar] [CrossRef] [Green Version]
- World Health Organisation. Worldwide Measles Deaths Climb 50% from 2016 to 2019 Claiming over 207 500 Lives in 2019; World Health Organisation: Geneva, Switzerland, 2020; pp. 1–5. [Google Scholar]
- Artenstein, A.W.; Halsey, N.A.; Nowak, G.J. Fostering Further Successes in Vaccinology. Am. J. Prev. Med. 2015, 49, S303–S306. [Google Scholar] [CrossRef] [PubMed]
- Oduwole, E.O.; Mahomed, H.; Ayele, B.T.; Wiysonge, C.S. Estimating vaccine confidence levels among healthcare students and staff of a tertiary institution in South Africa: Protocol of a cross-sectional survey. BMJ Open 2021, 11, e049877. [Google Scholar] [CrossRef]
- WHO. Ten Threats to Global Health in 2019. World Health Organization. 2019, pp. 1–18. Available online: https://www.who.int/news-room/feature-stories/ten-threats-to-global-health-in-2019 (accessed on 5 March 2020).
- World Health Organization. Listings of WHO’s Response to COVID-19. World Health Organization. 2020, p. 1. Available online: https://www.who.int/news/item/29-06-2020-covidtimeline (accessed on 16 January 2021).
- The Lancet Infectious Diseases. The COVID-19 infodemic. Lancet Infect. Dis. 2020, 20, 875. [CrossRef]
- Naeem, S.; Bin Bhatti, R. The COVID-19 ‘infodemic’: A new front for information professionals. Health Info. Libr. J. 2020, 37, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Risco, A.; Mejia, C.R.; Delgado-Zegarra, J.; Del-Aguila-Arcentales, S.; Arce-Esquivel, A.A.; Valladares-Garrido, M.J.; Del Portal, M.R.; Villegas, L.F.; Curioso, W.H.; Sekar, M.C. The Peru approach against the COVID-19 infodemic: Insights and strategies. Am. J. Trop. Med. Hyg. 2020, 103, 583–586. [Google Scholar] [CrossRef]
- Xue, H.; Gong, X.; Stevens, H. COVID-19 Vaccine Fact-Checking Posts on Facebook: Observational Study. J. Med. Internet Res. 2022, 24, e38423. [Google Scholar] [CrossRef]
- Understanding Novel mRNA Vaccines|For Better|US News n.d. Available online: https://health.usnews.com/health-care/for-better/articles/understanding-novel-mrna-vaccines (accessed on 16 February 2022).
- FDA. COVID-19 Vaccines|FDA. Food Drug Adm 2021. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines (accessed on 6 March 2021).
- FDA. Emergency Use Authorization|FDA. Food Drug Adm 2020. Available online: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization (accessed on 16 January 2021).
- De Waure, C.; Quaranta, G.; Ianuale, C.; Panatto, D.; Amicizia, D.; Apprato, L.; Campanella, P.; Colotto, M.; De Meo, C.; Di Nardo, F.; et al. Knowledge, attitudes and behaviors of the Italian population towards Neisseria meningitidis, Streptococcus pneumoniae and HPV diseases and vaccinations: A cross-sectional multicentre study. Public Health 2016, 141, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Larson, H.J.; De Figueiredo, A.; Xiahong, Z.; Schulz, W.S.; Verger, P.; Johnston, I.G.; Cook, A.R.; Jones, N.S. The State of Vaccine Confidence 2016: Global Insights Through a 67-Country Survey. EBioMedicine 2016, 12, 295–301. [Google Scholar] [CrossRef] [Green Version]
- Arif, N.; Al-Jefri, M.; Bizzi, I.H.; Perano, G.B.; Goldman, M.; Haq, I.; Chua, K.L.; Mengozzi, M.; Neunez, M.; Smith, H.; et al. Fake news or weak science? Visibility and characterization of antivaccine webpages returned by google in different languages and countries. Front. Immunol. 2018, 9, 1215. [Google Scholar] [CrossRef] [Green Version]
- Gualano, M.R.; Olivero, E.; Voglino, G.; Corezzi, M.; Rossello, P.; Vicentini, C.; Bert, F.; Siliquini, R. Knowledge, attitudes and beliefs towards compulsory vaccination: A systematic review. Hum. Vaccines Immunother. 2019, 15, 918–931. [Google Scholar] [CrossRef] [Green Version]
- Dubé, E.; Laberge, C.; Guay, M.; Bramadat, P.; Roy, R.; Bettinger, J. Vaccine hesitancy: An overview. Hum. Vaccines Immunother. 2013, 9, 1763–1773. [Google Scholar] [CrossRef]
- Verger, P.; Fressard, L.; Collange, F.; Gautier, A.; Jestin, C.; Launay, O.; Raude, J.; Pulcini, C.; Peretti-Watel, P. Vaccine Hesitancy Among General Practitioners and Its Determinants During Controversies: A National Cross-sectional Survey in France. EBioMedicine 2015, 2, 891–897. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, N.E.; Dubé, E. Unpacking Vaccine Hesitancy Among Healthcare Providers. EBioMedicine 2015, 2, 792–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suryadevara, M.; Handel, A.; Bonville, C.A.; Cibula, D.A.; Domachowske, J.B. Pediatric provider vaccine hesitancy: An under-recognized obstacle to immunizing children. Vaccine 2015, 33, 6629–6634. [Google Scholar] [CrossRef] [PubMed]
- Attwell, K.; Wiley, K.E.; Waddington, C.; Leask, J.; Snelling, T. Midwives’ attitudes, beliefs and concerns about childhood vaccination: A review of the global literature. Vaccine 2018, 36, 6531–6539. [Google Scholar] [CrossRef] [PubMed]
- Paterson, P.; Meurice, F.; Stanberry, L.R.; Glismann, S.; Rosenthal, S.L.; Larson, H.J. Vaccine hesitancy and healthcare providers. Vaccine 2016, 34, 6700–6706. [Google Scholar] [CrossRef] [Green Version]
- Peterson, C.J.; Lee, B.; Nugent, K. COVID-19 Vaccination Hesitancy among Healthcare Workers—A Review. Vaccines 2022, 10, 948. [Google Scholar] [CrossRef]
- Lin, C.; Mullen, J.; Smith, D.; Kotarba, M.; Kaplan, S.J.; Tu, P. Healthcare providers’ vaccine perceptions, hesitancy, and recommendation to patients: A systematic review. Vaccines 2021, 9, 713. [Google Scholar] [CrossRef] [PubMed]
- Maltezou, H.C.; Dounias, G.; Rapisarda, V.; Ledda, C. Vaccination policies for healthcare personnel: Current challenges and future perspectives. Vaccine X 2022, 11, 100172. [Google Scholar] [CrossRef]
- Larson, H.J.; Jarrett, C.; Eckersberger, E.; Smith, D.M.D.; Paterson, P. Understanding vaccine hesitancy around vaccines and vaccination from a global perspective: A systematic review of published literature, 2007–2012. Vaccine 2014, 32, 2150–2159. [Google Scholar] [CrossRef]
- Betsch, C.; Böhm, R.; Chapman, G.B. Using Behavioral Insights to Increase Vaccination Policy Effectiveness. Policy Insights Behav. Brain Sci. 2015, 2, 61–73. [Google Scholar] [CrossRef]
- WHO. Report of the SAGE Working Group on Vaccine Hesitancy. Available online: https://www.who.int/immunization/sage/meetings/2014/october/1_Report_WORKING_GROUP_vaccine_hesitancy_final.pdf (accessed on 28 June 2019).
- Bloom, D.E. The value of vaccination. World Econ. 2005, 6, 1–8. [Google Scholar] [CrossRef]
- Hsu, J.L. A brief history of vaccines: Smallpox to the present. S. D. Med. 2013. Available online: https://pubmed.ncbi.nlm.nih.gov/23444589/ (accessed on 10 June 2022).
- Smallpox. Available online: https://www.who.int/health-topics/smallpox#tab=tab_1 (accessed on 13 July 2022).
- Hotez, P. America and Europe’s new normal: The return of vaccine-preventable diseases. Pediatr. Res. 2019, 85, 912–914. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Regional Office for the Western Pacific. Polio Eradication in the Western Pacific Region. Available online: https://apps.who.int/iris/handle/10665/206921 (accessed on 10 June 2022).
- Larson, H.J.; Jarrett, C.; Schulz, W.S.; Chaudhuri, M.; Zhou, Y.; Dube, E.; Schuster, M.; MacDonald, N.E.; Wilson, R. Measuring vaccine hesitancy: The development of a survey tool. Vaccine 2015, 33, 4165–4175. [Google Scholar] [CrossRef] [Green Version]
- Eskola, J.; Duclos, P.; Schuster, M.; MacDonald, N.E.; Liang, X.; Chaudhuri, M.; Dube, E.; Gellin, B.; Goldstein, S.; Larson, H. How to deal with vaccine hesitancy? Vaccine 2015, 33, 4215–4217. [Google Scholar] [CrossRef] [Green Version]
- Global Vaccine Action Plan. Decade of vaccine collaboration. Vaccine 2013, 31 (Suppl. 2), B5–B31. [Google Scholar] [CrossRef]
- Shapiro, G.K.; Kaufman, J.; Brewer, N.T.; Wiley, K.; Menning, L.; Leask, J.; Abad, N.; Betsch, C.; Bura, V.; Correa, G. A critical review of measures of childhood vaccine confidence. Curr. Opin. Immunol. 2021, 71, 34–45. [Google Scholar] [CrossRef]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. Theory Pract. 2005, 8, 19–32. [Google Scholar] [CrossRef] [Green Version]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef] [Green Version]
- Pham, M.T.; Rajić, A.; Greig, J.D.; Sargeant, J.M.; Papadopoulos, A.; Mcewen, S.A. A scoping review of scoping reviews: Advancing the approach and enhancing the consistency. Res. Synth. Methods 2014, 5, 371–385. [Google Scholar] [CrossRef]
- The Joanna Briggs Institute. Joanna Briggs Institute Reviewers’ Manual: 2015 Edition/Supplement Methodology for JBI Scoping Reviews; The Joanna Briggs Institute: Adelaide, Australia, 2015. [Google Scholar]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Opel, D.J.; Mangione-Smith, R.; Taylor, J.A.; Korfiatis, C.; Wiese, C.; Catz, S.; Martin, D.P. Development of a survey to identify vaccine-hesitant parents. Hum. Vaccin. 2011, 7, 419–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilkey, M.B.; Magnus, B.E.; Reiter, P.L.; McRee, A.L.; Dempsey, A.F.; Brewer, N.T. The Vaccination Confidence Scale: A brief measure of parents’ vaccination beliefs. Vaccine 2014, 32, 6259–6265. [Google Scholar] [CrossRef] [Green Version]
- Martin, L.R.; Petrie, K.J. Understanding the Dimensions of Anti-Vaccination Attitudes: The Vaccination Attitudes Examination (VAX) Scale. Ann. Behav. Med. 2017, 51, 652–660. [Google Scholar] [CrossRef]
- Luthy, K.E.; Beckstrand, R.L.; Callister, L.C. Parental Hesitation in Immunizing Children in Utah. Public Health Nurs. 2010, 27, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Meharry, P.M.; Colson, E.R.; Grizas, A.P.; Stiller, R.; Vazquez, M. Reasons why women accept or reject the trivalent inactivated influenza vaccine (TIV) during pregnancy. Matern. Child. Health J. 2013, 17, 156–164. [Google Scholar] [CrossRef]
- Quinn, S.C.; Jamison, A.M.; An, J.; Hancock, G.R.; Freimuth, V.S. Measuring vaccine hesitancy, confidence, trust and flu vaccine uptake: Results of a national survey of White and African American adults. Vaccine 2019, 37, 1168–1173. [Google Scholar] [CrossRef] [PubMed]
- Sarathchandra, D.; Navin, M.C.; Largent, M.A.; McCright, A.M. A survey instrument for measuring vaccine acceptance. Prev. Med. 2018, 109, 1–7. [Google Scholar] [CrossRef]
- Frew, P.M.; Murden, R.; Mehta, C.C.; Chamberlain, A.T.; Hinman, A.R.; Nowak, G.; Mendel, J.; Aikin, A.; Randall, L.A.; Hargreaves, A.L.; et al. Development of a US trust measure to assess and monitor parental confidence in the vaccine system. Vaccine 2019, 37, 325–332. [Google Scholar] [CrossRef]
- Dubé, E.; Gagnon, D.; Ouakki, M.; Bettinger, J.A.; Guay, M.; Halperin, S.; Wilson, K.; Graham, J.; Witteman, H.O.; MacDonald, S.; et al. Understanding vaccine hesitancy in Canada: Results of a consultation study by the Canadian Immunization Research Network. PLoS ONE 2016, 11, e0156118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, S.B.; Lum, R. Explanations for Not Receiving the Seasonal Influenza Vaccine: An Ontario Canada Based Survey. J. Health Commun. 2017, 22, 506–514. [Google Scholar] [CrossRef]
- Perez, S.; Shapiro, G.K.; Tatar, O.; Joyal-Desmarais, K.; Rosberger, Z. Development and validation of the human papillomavirus attitudes and beliefs scale in a National Canadian Sample. Sex. Transm. Dis. 2016, 43, 626–632. [Google Scholar] [CrossRef]
- Shapiro, G.K.; Holding, A.; Perez, S.; Amsel, R.; Rosberger, Z. Validation of the vaccine conspiracy beliefs scale. Papillomavirus Res. 2016, 2, 167–172. [Google Scholar] [CrossRef] [Green Version]
- Brown, K.F.; Shanley, R.; Cowley, N.A.; van Wijgerden, J.; Toff, P.; Falconer, M.; Ramsay, M.; Hudson, M.J.; Green, J.; Vincent, C.A.; et al. Attitudinal and demographic predictors of measles, mumps and rubella (MMR) vaccine acceptance: Development and validation of an evidence-based measurement instrument. Vaccine 2011, 29, 1700–1709. [Google Scholar] [CrossRef]
- Paterson, P.; Chantler, T.; Larson, H.J. Reasons for non-vaccination: Parental vaccine hesitancy and the childhood influenza vaccination school pilot programme in England. Vaccine 2018, 36, 5397–5401. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.M.; Aljaed, N.M.; Aloufi, M.E.; Jafri, S.A.; Aloufi, R.A.; Altwairqi, R.G. The prevalence of vaccine hesitancy and skipping MMR vaccine due to an autism thoughts in Saudi Arabia. Indo. Am. J. Pharm. Sci. 2019, 6, 1989–2001. [Google Scholar] [CrossRef]
- Sabahelzain, M.M.; Moukhyer, M.; Dubé, E.; Hardan, A.; van den Borne, B.; Bosma, H. Towards a further understanding of measles vaccine hesitancy in Khartoum state, Sudan: A qualitative study. PLoS ONE 2019, 14, e0213882. [Google Scholar] [CrossRef] [Green Version]
- Yousafzai, M.T.; Riaz, A.; Omer, S.B.; Husain, S.; Nisar, I.; Mahesar, W.; Imam, D.O.; Wallace, A.; Ali, A. Development and Validation of Parental Vaccine Attitudes Scale for Use in Low-income Setting. Pediatr. Infect. Dis. J. 2019, 38, e143–e148. [Google Scholar] [CrossRef] [Green Version]
- Corben, P.; Leask, J. Vaccination hesitancy in the antenatal period: A cross-sectional survey. BMC Public Health 2018, 18, 566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Buynder, P.G.; Van Buynder, J.L.; Menton, L.; Thompson, G.; Sun, J. Antigen specific vaccine hesitancy in pregnancy. Vaccine 2019, 37, 2814–2820. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, Y.; Liang, H.; Wang, Y. Reliability and validity of a survey to identify vaccine hesitancy among parents in Changxing county, Zhejiang province. Hum. Vaccin. Immunother. 2019, 15, 1092–1099. [Google Scholar] [CrossRef]
- Siu, J.Y.M.; Fung, T.K.F.; Leung, L.H.M. Social and cultural construction processes involved in HPV vaccine hesitancy among Chinese women: A qualitative study. Int. J. Equity Health 2019, 18, 147. [Google Scholar] [CrossRef]
- Wallace, A.S.; Wannemuehler, K.; Bonsu, G.; Wardle, M.; Nyaku, M.; Amponsah-Achiano, K.; Dadzie, J.F.; Sarpong, F.O.; Orenstein, W.A.; Rosenberg, E.S.; et al. Development of a valid and reliable scale to assess parents’ beliefs and attitudes about childhood vaccines and their association with vaccination uptake and delay in Ghana. Vaccine 2019, 37, 848–856. [Google Scholar] [CrossRef]
- Olufowote, J.O. Local Resistance to the Global Eradication of Polio: Newspaper Coverage of the 2003-2004 Vaccination Stoppage in Northern Nigeria. Health Commun. 2011, 26, 743–753. [Google Scholar] [CrossRef]
- Njeru, I.; Ajack, Y.; Muitherero, C.; Onyango, D.; Musyoka, J.; Onuekusi, I.; Kioko, J.; Muraguri, N.; Davis, R. Did the call for boycott by the catholic bishops affect the polio vaccination coverage in Kenya in 2015? A cross-sectional study. Pan. Afr. Med. J. 2016, 24, 120. [Google Scholar] [CrossRef]
- Oduwole, E.O.; Mahomed, H.; Laurenzi, C.A.; Larson, H.J.; Wiysonge, C.S. Point-of-care vaccinators’ perceptions of vaccine hesitancy drivers: A qualitative study from the cape metropolitan district, South Africa. Vaccine 2021, 39, 5506–5512. [Google Scholar] [CrossRef]
- Costa, J.C.; Weber, A.M.; Darmstadt, G.L.; Abdalla, S.; Victora, C.G. Religious affiliation and immunization coverage in 15 countries in Sub-Saharan Africa. Vaccine 2020, 38, 1160–1169. [Google Scholar] [CrossRef]
- Ahmed, A.; Lee, K.S.; Bukhsh, A.; Al-Worafi, Y.M.; Sarker, M.M.R.; Ming, L.C.; Khan, T.M. Outbreak of vaccine-preventable diseases in Muslim majority countries. J. Infect. Public Health 2018, 11, 153–155. [Google Scholar] [CrossRef]
- Warraich, H.J. Religious opposition to polio vaccination. Emerg. Infect. Dis. 2009, 15, 978. [Google Scholar] [CrossRef]
- Ahmad, K. Pakistan struggles to eradicate polio. Lancet Infect. Dis. 2007, 7, 247. [Google Scholar] [CrossRef]
- Oduwole, E.O.; Esterhuizen, T.M.; Mahomed, H.; Wiysonge, C.S. Estimating vaccine confidence levels among healthcare staff and students of a tertiary institution in South Africa. Vaccines 2021, 9, 1246. [Google Scholar] [CrossRef]
| First Author (Year) | Study Title | Example of Item | Major Advantage/Finding (as Reported by the Authors) | Major Disadvantage/Limitation (as Reported by the Authors) |
|---|---|---|---|---|
| Luthy, K.E. (2010) | Parental hesitation in immunizing children in Utah. | My child was delayed with immunizations because I have the following concerns about vaccine safety: | Hesitant parents have serious concerns regarding immunization safety. | The convenience sample was from one state, thus the findings cannot be generalized to other populations. |
| Brown, K.F. (2011) | Attitudinal and demographic predictors of measles, mumps and rubella (MMR) vaccine acceptance: Development and validation of an evidence-based measurement instrument. | MMR has serious side effects. | The instrument is able to elicit consistent responses at different time periods and on items which are conceptually/empirically linked, is able to discriminate between participants with differing MMR behaviors and is able to predict MMR behavior in multivariate analyses. | The study employed a retrospective design in which attitudes were measured after MMR doses were received, therefore the extent to which these attitudes incorporate retrospective justification and are able to predict future MMR behavior is yet to be ascertained. |
| Opel, D.J. (2011) | Development of a survey to identify vaccine-hesitant parents. The parent attitudes about childhood vaccines survey. | Have you ever decided not to have your child get a shot for reasons other than illness or allergy? | The final version of the PACV contains 18 items, takes 5 min or less to complete, and reads at a 6th grade level. | The total number of parents (N = 4) in the two focus groups was small and is therefore likely not representative of the larger vaccine hesitant parent population, a very heterogeneous group. |
| Meharry, P.M. (2012) | Reasons why women accept or reject the trivalent inactivated influenza vaccine (TIV) during pregnancy. | Who gave you advice about seasonal influenza and the influenza vaccine during your pregnancy? | The two-for-one benefit to mother and infant is pivotal knowledge and a predictor of future vaccination. | It was based on the experiences of 60 women during a specific time period, and although the sample had diverse cultural, educational and socio-economic backgrounds, the thematic analysis does not represent all possible responses. |
| Gilkey, M.B. (2014) | The Vaccination Confidence Scale: A brief measure of parents’ vaccination beliefs. | Vaccines are necessary to protect the health of teenagers. | Our nationally-representative sample allowed for the development of a robust tool tested with respect to demographic characteristics known to correlate with vaccination beliefs. | The primary limitation of this study was the modest number of items available for scale development. |
| Larson, H.J. (2015) | Measuring vaccine hesitancy: The development of a survey tool. | Childhood vaccines are important for my child’s health. | The Working Group developed a compendium of three different types of survey questions: core closed questions; Likert scale questions; and a set of open ended questions. | The questions identified do not address all the determinants in the Vaccine Hesitancy Matrix. |
| Suryadevara, M. (2015) | Pediatric provider vaccine hesitancy: An under-recognized obstacle to immunizing children. | Do you believe that standard immunizations are safe? | This is the first study to describe vaccine attitudes among pediatric providers attending AAP-sponsored immunization conferences. | The study population includes pediatric providers who attended AAP-sponsored conferences from a limited geographical area, and therefore the data may not be generalizable to all pediatric providers. |
| Eve Dubé (2016) | Understanding Vaccine Hesitancy in Canada: Results of a Consultation Study by the Canadian Immunization Research Network. | How prepared are you to effectively provide information about risks and benefits of vaccination? | Our findings indicate that the majority of participants—both vaccine experts and front-line vaccine providers—have the perception that vaccine rates have been declining and consider vaccine hesitancy an important issue to address in Canada. | By design, the results reported here represent the opinions of only some non-randomly selected key opinion leaders. |
| Larson, H.J. (2016) | The State of Vaccine Confidence 2016: Global Insights Through a 67-Country Survey. | Vaccines are compatible with my religious beliefs. | We find that vaccine safety sentiment is more negative in the European and the Western Pacific regions, where nine of the ten least confident countries are located. | A limitation of this survey is its generality of the survey which does not reveal whether the attitudes are related to specific vaccine(s) which an individual may have concerns about. |
| Perez, S. (2016) | Development and Validation of the Human Papillomavirus Attitudes and Beliefs Scale in a National Canadian Sample. | I feel that… the HPV vaccine will protect my son’s sexual health. | The HABS is available in both English and French for assessing HPV attitudes and beliefs. | The HABS does not capture all attitudinal items. |
| Shapiro, G.K. (2016) | Validation of the vaccine conspiracy beliefs scale. | Immunizing children is harmful and this fact is covered up. | Income, parental age, healthcare provider recommendation, and vaccine conspiracy beliefs emerged as significant predictors of parents’ willingness to vaccinate their child. | Using preselected items to develop this scale may have increased the likelihood of producing a one-dimensional scale. |
| Martin, L.R. (2017) | Understanding the Dimensions of Anti-Vaccination Attitudes: the Vaccination Attitudes Examination (VAX) Scale. | Vaccines can cause unforeseen problems in children. | The VAX scale is a short and simple tool that has demonstrated significant associations with vaccination behaviors and intentions. | No study limitation was reported by the authors of this study. |
| Meyer, S.B. (2017) | Explanations for Not Receiving the Seasonal Influenza Vaccine: An Ontario Canada Based Survey. | Why didn’t you get the seasonal flu vaccine in the last flu season? | The most cited explanation given for not receiving the seasonal influenza vaccine is related to the perceived importance of vaccination (or lack thereof). | Due to the nature of our data collection, we were unable to continue to sample until reaching saturation of themes. |
| Betsch, C. (2018) | Beyond confidence: Development of a measure assessing the 5C psychological antecedents of vaccination. | For me, it is inconvenient to receive vaccinations. | The 5C scale now offers a psychologically sound and validated measure to be used for regular global monitoring of the psychological antecedents of vaccination behavior. | A limitation of this work is that the three studies, similar to the construction studies of all other existing measures, only assess concurrent validity and not predictive validity. |
| Paul Corben (2018) | Vaccination hesitancy in the antenatal period: a cross-sectional survey. | All things considered, how much do you trust your child’s doctor? | There was no difference detected in vaccination timeliness of babies of first-time mothers and experienced mothers nor between those who considered themselves ‘not at all hesitant’ and others. | We were unable to calculate summary hesitancy and decisional conflict measures. |
| Paula M. Frew (2018) | Development of a US trust measure to assess and monitor parental confidence in the vaccine system. | Vaccines recommended for young children are safe. | [The authors] developed a parsimonious, relevant eight-item index that was able to assess vaccine confidence with a highly acceptable internal validity score. | Several sources of bias limit the ability of self-reported vaccination decisions to represent actual vaccination behavior, including recall, response, and social desirability bias. |
| Paterson, P. (2018) | Reasons for non-vaccination: Parental vaccine hesitancy and the childhood influenza vaccination school pilot programme in England | ‘‘We would be grateful if you could tell us why you decided not to vaccinate your child as part of the school immunization program” | The majority of parents interviewed illustrated a lack of perceived need for the influenza vaccine for children. | Study limitations include the possibility of sample bias, since those that took part in our study might have different views to that of the general population. |
| Sarathchandra, D. (2018) | A survey instrument for measuring vaccine acceptance. | My right to consent to medical treatment means that vaccinations should always be voluntary. | Our results indicate that vaccine acceptance is substantially eroded by conspiratorial thinking and is modestly reduced by political conservatism. | No study limitation was reported by the authors of this study. |
| Hu Yu (2019) | Reliability and validity of a survey to identify vaccine hesitancy among parents in Changxing county, Zhejiang province. | It is better for my child to develop immunity by getting sick than vaccination. | We found the concern of the vaccine efficacy was associated with under immunization, which was similar to the previous studies in other settings. | Our results might reflect current perceptions of immunizations other than perceptions at the time they were making immunization decisions, |
| Mohamed, M.M. (2019) | The prevalence of vaccine hesitancy and skipping MMR vaccine due to autism thoughts in Saudi Arabia. | Do you think that your child received too many vaccines? | Vaccination hesitant parents showed a significantly high probability that they think that healthy children don’t need to be vaccinated with MMR, and that the risk of MMR vaccine outweighs the benefit. | One of the limitations of this study was using self-reported questionnaires for collecting data which were prone to recall bias. |
| Quinn, S.C. (2019) | Measuring vaccine hesitancy, confidence, trust and flu vaccine uptake: Results of a national survey of White and African American adults. | Thinking specifically about the flu vaccine, do you think the flu vaccine is necessary? | In this article, we can distinguish between general vaccine hesitancy and vaccine hesitancy specific to the flu vaccine. | No study limitation was reported by the authors of this study. |
| Sabahelzain, M.M. (2019) | Towards a further understanding of measles vaccine hesitancy in Khartoum state, Sudan: A qualitative study. | Do you think measles vaccine hesitancy exists in Sudan? Why? | The majority of the participants agreed that the main contextual determinant is the presence of people (parents/guardians) who can be qualified as “anti-vaccination”; they mostly belong to religious groups, and they often refuse all vaccines. | This study’s findings should be interpreted within the context of the study ’s participants and areas. |
| Siu, J.Y.M. (2019) | Social and cultural construction processes involved in HPV vaccine hesitancy among Chinese women: a qualitative study. | How do you perceive the dangers of HPV? | Only a few participants knew that HPV could lead to genital warts and that HPV vaccination can also help prevent genital warts. | Our findings mostly reflect the perceptions and decision-making process of women who belong to a relatively high socioeconomic status. |
| Van Buynder, P.G. (2019) | Antigen specific vaccine hesitancy in pregnancy. | During which trimester of this pregnancy did you receive the flu vaccine? | One out of every two pregnant women surveyed accessed a pertussis vaccine booster but not an influenza vaccine. | No study limitation was reported by the authors of this study. |
| Wallace, A.S. (2019) | Development of a valid and reliable scale to assess parents’ beliefs and attitudes about childhood vaccines and their association with vaccination uptake and delay in Ghana. | People in this community have expressed concerns that a child might have a serious side effect from a vaccination. | Our study is the first to document development of a valid and reliable scale to assess caregiver attitudes and beliefs towards vaccination in a low- or middle- income country setting and show a high level of association of the scale score with child’s vaccination status. | The survey was cross-sectional, so information for the scale and for vaccination status was collected at the same time; thus, our criterion validity was limited to concurrent rather than predictive validity. |
| Yousafzai, M.T. (2019) | Development and Validation of Parental Vaccine Attitudes Scale for Use in Low-income Setting. | I should be allowed to selectively choose the vaccines which I believe my child needs. | The four-item scale addressing parental attitude toward vaccine-preventable disease salience and community benefit is sufficiently reliable, and it can predict vaccine acceptance among parents in low-income settings. | One of the limitations of this study is validation of the tool only in Pakistan, rather than in several developing countries. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oduwole, E.O.; Pienaar, E.D.; Mahomed, H.; Wiysonge, C.S. Overview of Tools and Measures Investigating Vaccine Hesitancy in a Ten Year Period: A Scoping Review. Vaccines 2022, 10, 1198. https://doi.org/10.3390/vaccines10081198
Oduwole EO, Pienaar ED, Mahomed H, Wiysonge CS. Overview of Tools and Measures Investigating Vaccine Hesitancy in a Ten Year Period: A Scoping Review. Vaccines. 2022; 10(8):1198. https://doi.org/10.3390/vaccines10081198
Chicago/Turabian StyleOduwole, Elizabeth O., Elizabeth D. Pienaar, Hassan Mahomed, and Charles S. Wiysonge. 2022. "Overview of Tools and Measures Investigating Vaccine Hesitancy in a Ten Year Period: A Scoping Review" Vaccines 10, no. 8: 1198. https://doi.org/10.3390/vaccines10081198
APA StyleOduwole, E. O., Pienaar, E. D., Mahomed, H., & Wiysonge, C. S. (2022). Overview of Tools and Measures Investigating Vaccine Hesitancy in a Ten Year Period: A Scoping Review. Vaccines, 10(8), 1198. https://doi.org/10.3390/vaccines10081198







