SARS-CoV-2 Antibody Response against Mild-to-Moderate Breakthrough COVID-19 in Home Isolation Setting in Thailand
Abstract
:1. Introduction
2. Materials and Methods
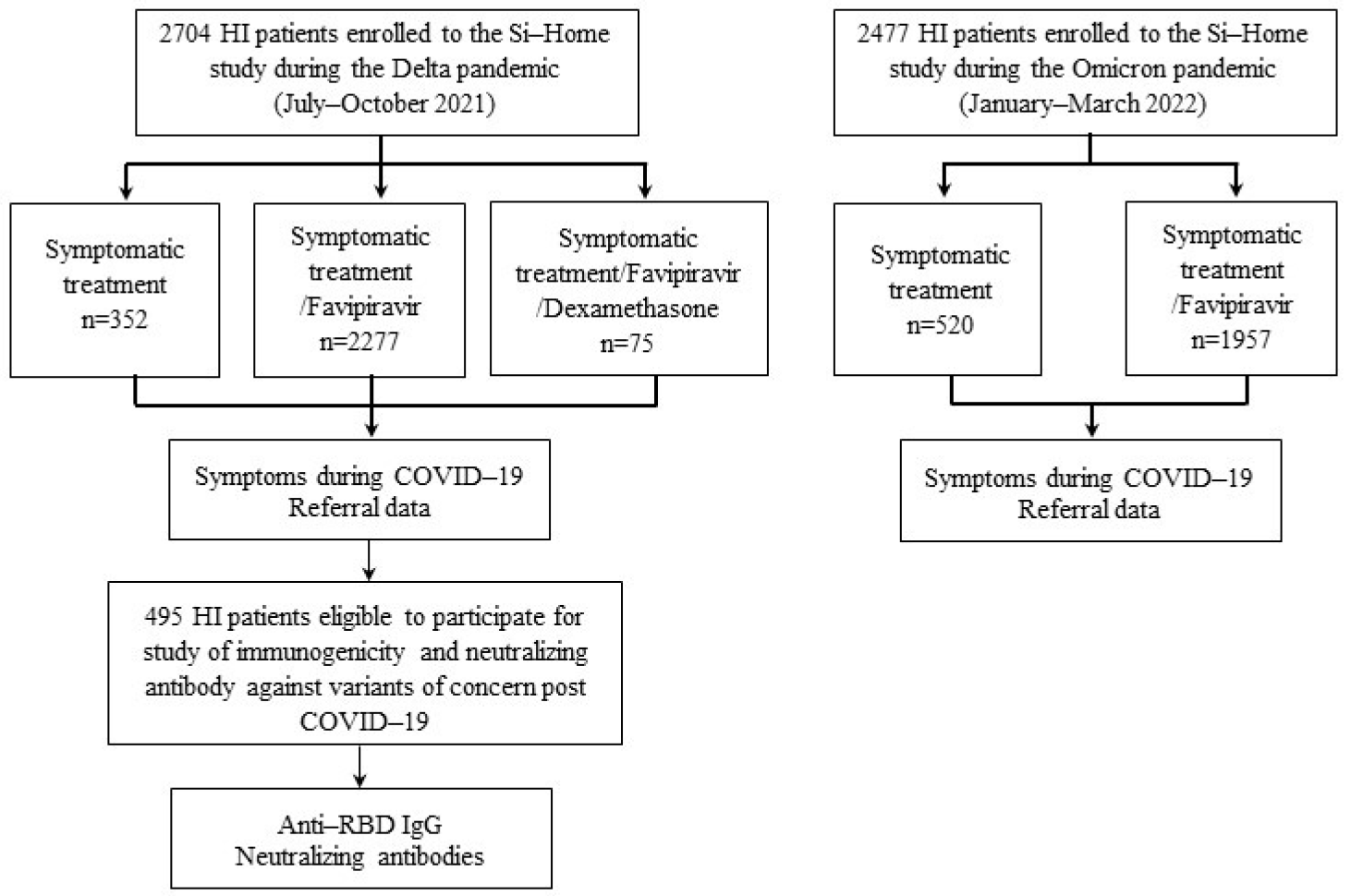
2.1. Study Population
2.2. Patient Selection and Procedures
2.3. Outcome Measures
2.4. Diagnosis of COVID-19
2.5. Serological Assays
2.6. Statistical Analysis
3. Results
3.1. Demographic and Clinical Data
3.2. Rehospitalized COVID-19 Patients
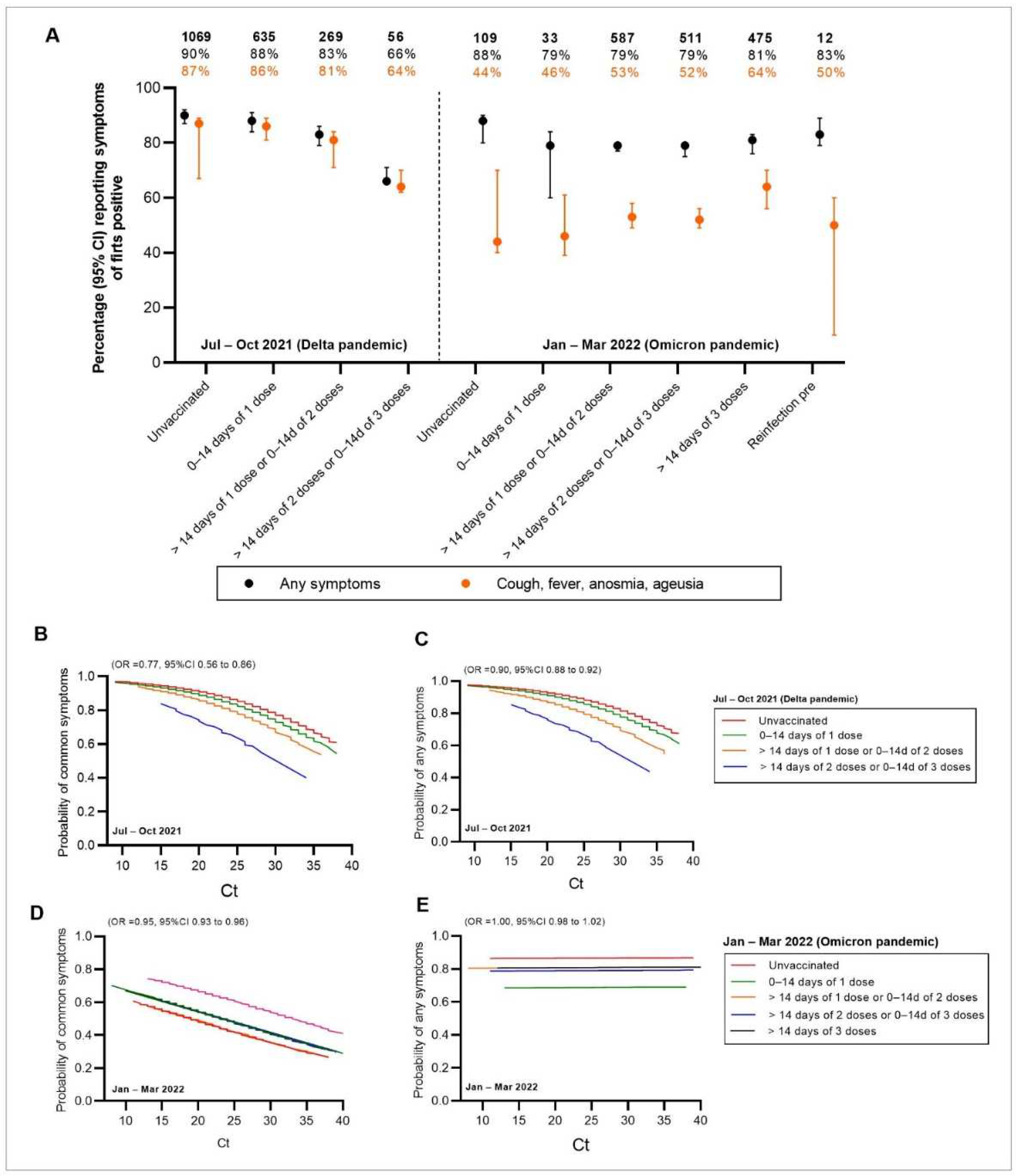
3.3. Number of Symptoms and Fatigue Scores during Home Isolation
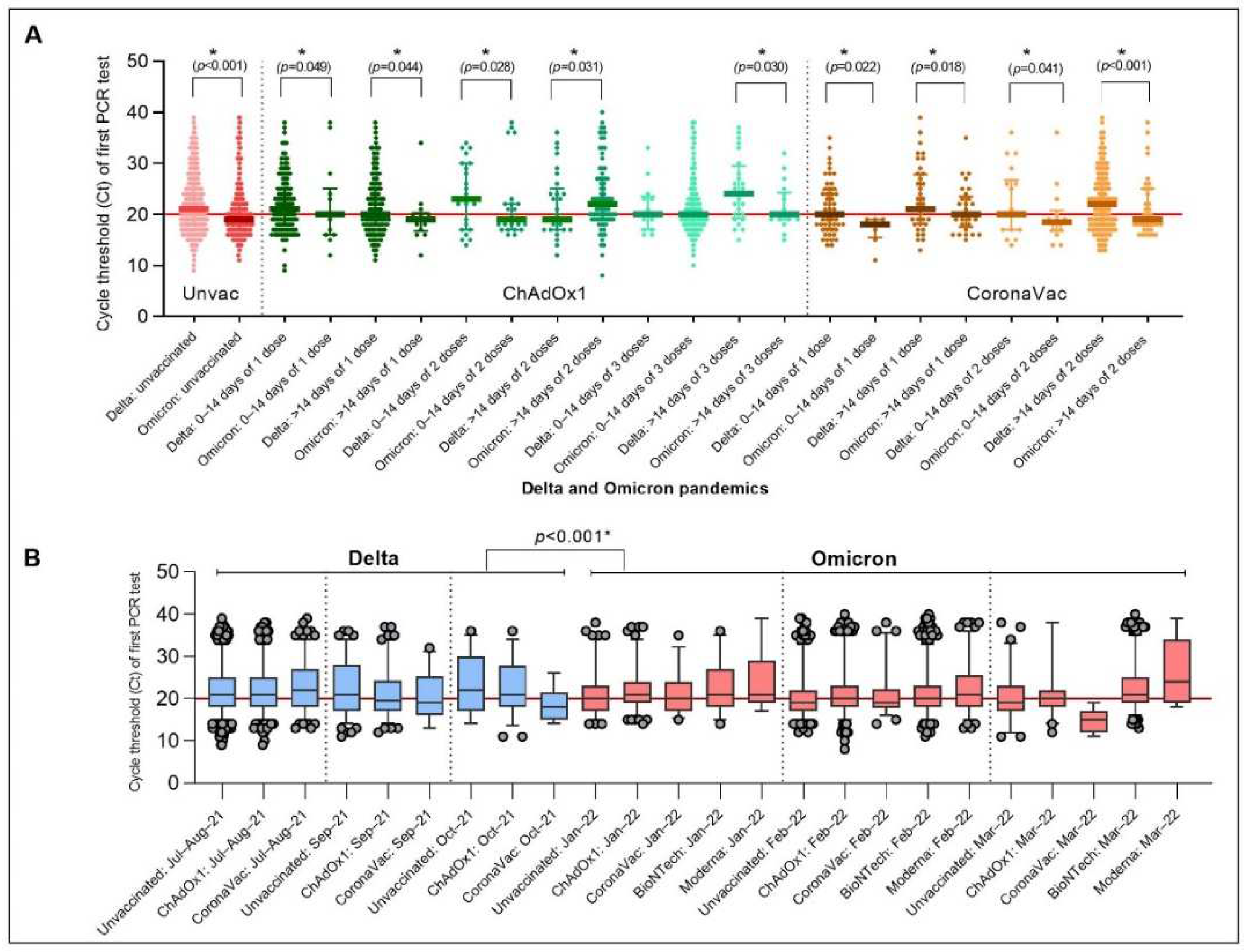
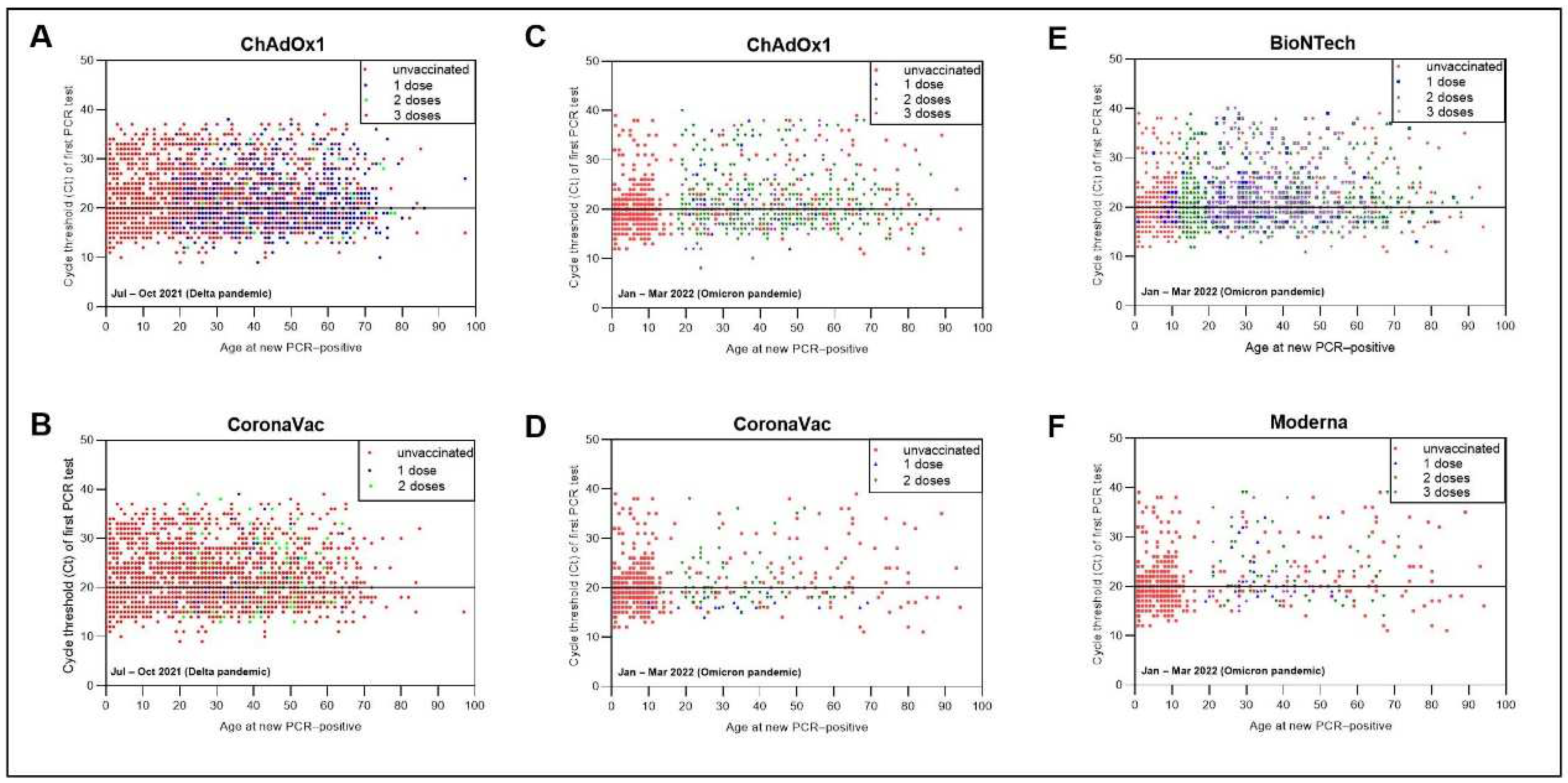
3.4. Clinical Manifestations and Viral Burden
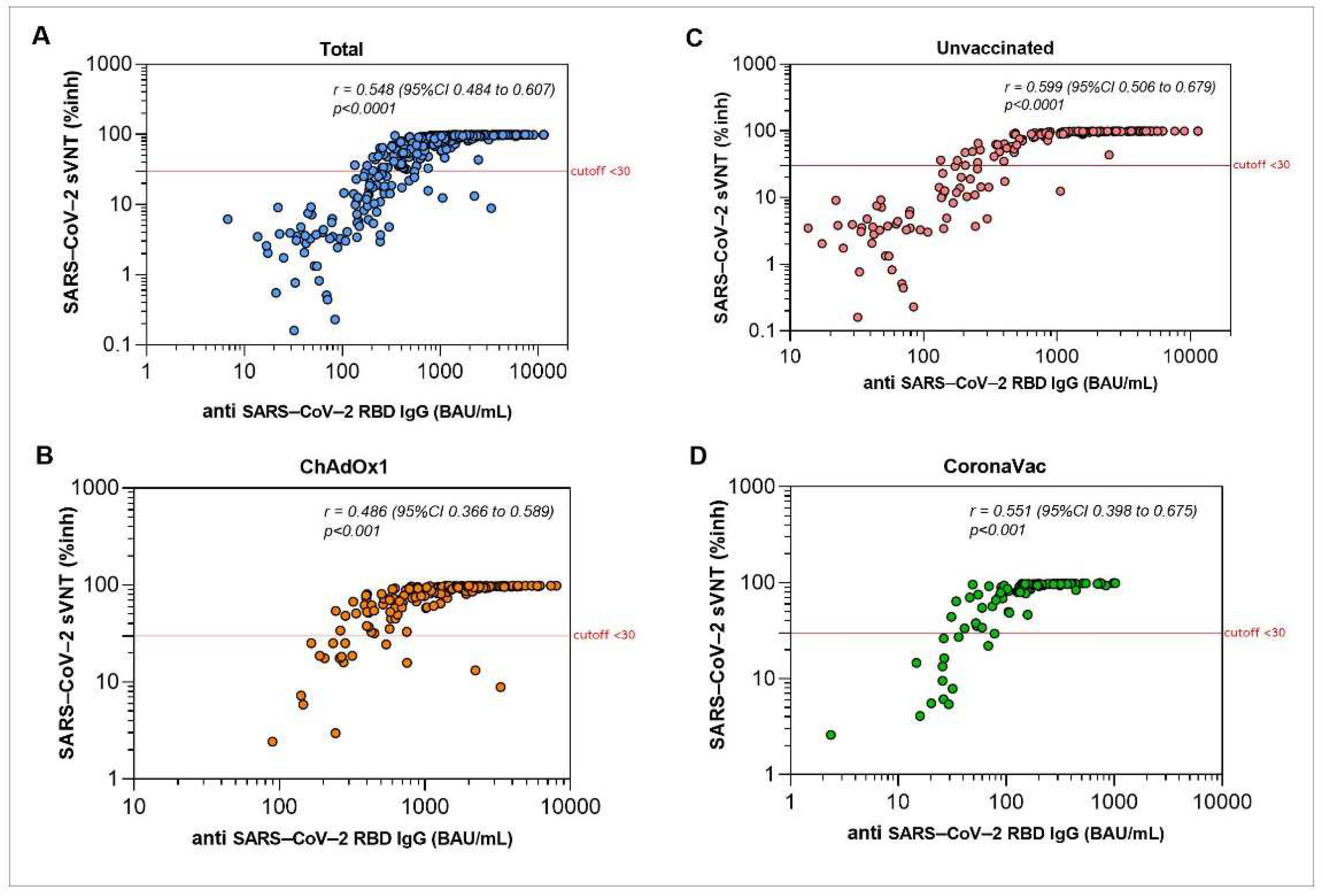
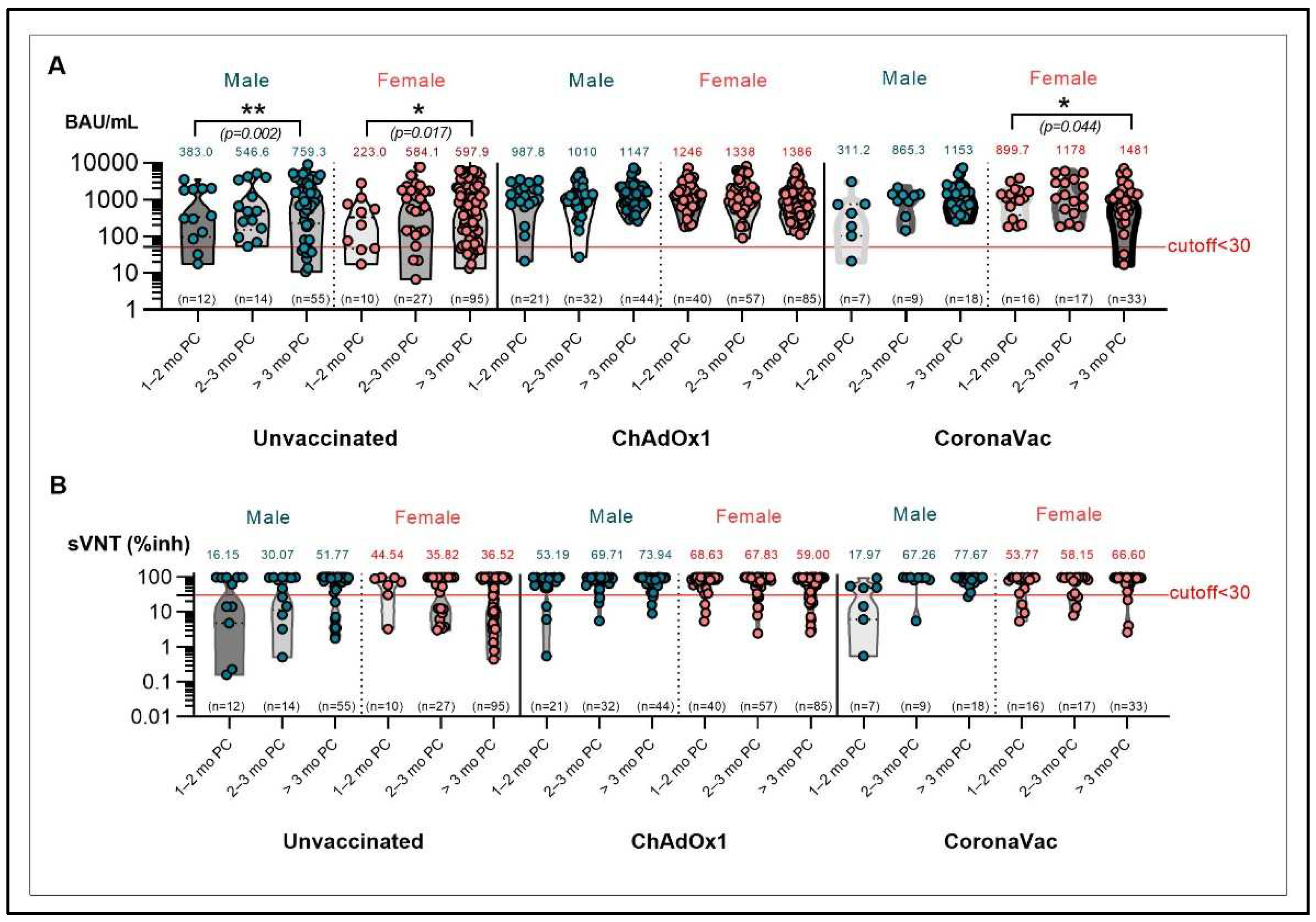
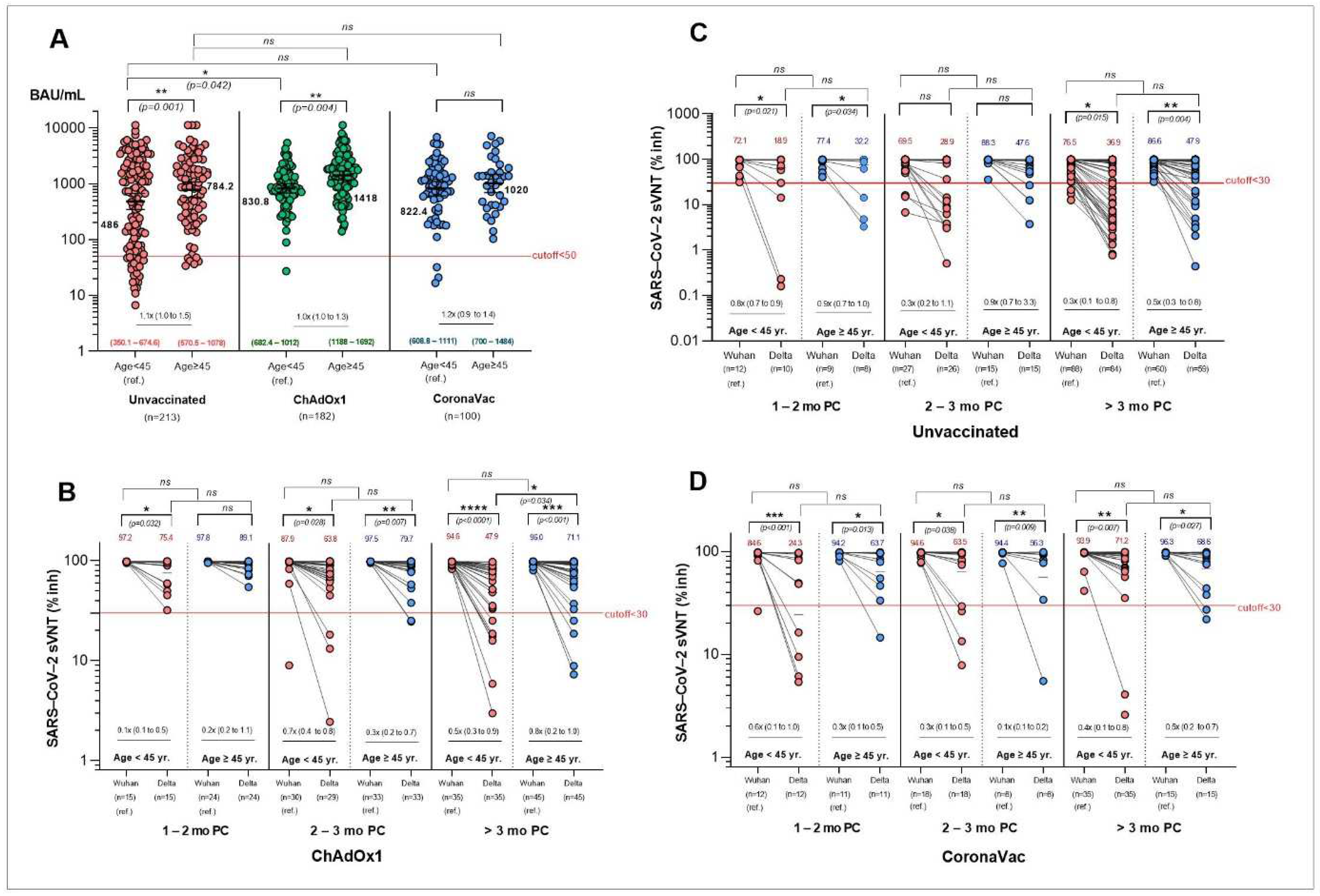
3.5. Immune Responses against SARS-CoV-2 Variants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Suntronwong, N.; Yorsaeng, R.; Puenpa, J.; Auphimai, C.; Thongmee, T.; Vichaiwattana, P.; Kanokudom, S.; Duangchinda, T.; Chantima, W.; Pakchotanon, P.; et al. COVID-19 Breakthrough Infection after Inactivated Vaccine Induced Robust Antibody Responses and Cross-Neutralization of SARS-CoV-2 Variants, but Less Immunity against Omicron. Vaccines 2022, 10, 391. [Google Scholar] [CrossRef] [PubMed]
- Escandón, K.; Rasmussen, A.L.; Bogoch, I.I.; Murray, E.J.; Escandón, K.; Popescu, S.V.; Kindrachuk, J. COVID-19 false dichotomies and a comprehensive review of the evidence regarding public health, COVID-19 symptomatology, SARS-CoV-2 transmission, mask wearing, and reinfection. BMC Infect. Dis. 2021, 21, 710. [Google Scholar] [CrossRef] [PubMed]
- Angkasekwinai, N.; Sewatanon, J.; Niyomnaitham, S.; Phumiamorn, S.; Sukapirom, K.; Sapsutthipas, S.; Sirijatuphat, R.; Wittawatmongkol, O.; Senawong, S.; Mahasirimongkol, S.; et al. Comparison of safety and immunogenicity of CoronaVac and ChAdOx1 against the SARS-CoV-2 circulating variants of concern (Alpha, Delta, Beta) in Thai healthcare workers. Vaccine X 2022, 10, 100153. [Google Scholar] [CrossRef] [PubMed]
- Mascellino, M.T.; Di Timoteo, F.; De Angelis, M.; Oliva, A. Overview of the Main Anti-SARS-CoV-2 Vaccines: Mechanism of Action, Efficacy and Safety. Infect. Drug Resist. 2021, 14, 3459–3476. [Google Scholar] [CrossRef]
- Darraj, M.A.; Al-Mekhlafi, H.M. Prospective Evaluation of Side-Effects Following the First Dose of Oxford/AstraZeneca COVID-19 Vaccine among Healthcare Workers in Saudi Arabia. Vaccines 2022, 10, 223. [Google Scholar] [CrossRef]
- World Health Organization Thailand. COVID-19—WHO Thailand Situation Reports. Available online: https://www.who.int/thailand/emergencies/novel-coronavirus-2019/situation-reports (accessed on 8 June 2022).
- Servellita, V.; Syed, A.M.; Morris, M.K.; Brazer, N.; Saldhi, P.; Garcia-Knight, M.; Sreekumar, B.; Khalid, M.M.; Ciling, A.; Chen, P.-Y.; et al. Neutralizing immunity in vaccine breakthrough infections from the SARS-CoV-2 Omicron and Delta variants. Cell 2022, 185, 1539–1548. [Google Scholar] [CrossRef]
- Feikin, D.R.; Abu-Raddad, L.J.; Andrews, N.; Davies, M.-A.; Higdon, M.M.; Orenstein, W.A.; Patel, M.K. Assessing vaccine effectiveness against severe COVID-19 disease caused by omicron variant. Report from a meeting of the World Health Organization. Vaccine 2022, 40, 3516–3527. [Google Scholar] [CrossRef]
- Department of Disease Control. Corona Virus Disease (COVID-19). Available online: https://ddc.moph.go.th/covid19-dashboard/ (accessed on 27 June 2022).
- Manosuthi, W.; Prasanchaimontri, I.; Niyomnaitham, S.; Sirijatuphat, R.; Charoenpong, L.; Copeland, K.; Cressey, T.R.; Mokmued, P.; Chokephaibulkit, K. Pharmacokinetics of favipiravir in adults with mild COVID-19 in Thailand. medRxiv 2022. [Google Scholar] [CrossRef]
- Udwadia, Z.F.; Singh, P.; Barkate, H.; Patil, S.; Rangwala, S.; Pendse, A.; Kadam, J.; Wu, W.; Caracta, C.F.; Tandon, M. Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial. Int. J. Infect. Dis. 2021, 103, 62–71. [Google Scholar] [CrossRef]
- Terpos, E.; Ntanasis-Stathopoulos, I.; Elalamy, I.; Kastritis, E.; Sergentanis, T.N.; Politou, M.; Psaltopoulou, T.; Gerotziafas, G.; Dimopoulos, M.A. Hematological findings and complications of COVID-19. Am. J. Hematol. 2020, 95, 834–847. [Google Scholar] [CrossRef] [Green Version]
- Vangeel, L.; Chiu, W.; De Jonghe, S.; Maes, P.; Slechten, B.; Raymenants, J.; André, E.; Leyssen, P.; Neyts, J.; Jochmans, D. Remdesivir, Molnupiravir and Nirmatrelvir remain active against SARS-CoV-2 Omicron and other variants of concern. Antivir. Res. 2022, 198, 105252. [Google Scholar] [CrossRef] [PubMed]
- Angkasekwinai, N.; Niyomnaitham, S.; Sewatanon, J.; Phumiamorn, S.; Sukapirom, K.; Senawong, S.; Mahasirimongkol, S.; Toh, Z.Q.; Umrod, P.; Somporn, T.; et al. The immunogenicity against variants of concern and reactogenicity of four COVID-19 booster vaccinations following CoronaVac or ChAdOx1 nCoV-19 primary series. medRxiv 2022. [Google Scholar] [CrossRef]
- Lu, L.; Mok, B.W.; Chen, L.L.; Chan, J.M.; Tsang, O.T.; Lam, B.H.; Chuang, V.W.; Chu, A.W.; Chan, W.M.; Ip, J.D.; et al. Neutralization of SARS-CoV-2 Omicron variant by sera from BNT162b2 or Coronavac vaccine recipients. Clin. Infect. Dis. 2021, ciab1041. [Google Scholar] [CrossRef]
- Wolter, N.; Jassat, W.; Walaza, S.; Welch, R.; Moultrie, H.; Groome, M.; Amoako, D.G.; Everatt, J.; Bhiman, J.N.; Scheepers, C.; et al. Early assessment of the clinical severity of the SARS-CoV-2 omicron variant in South Africa: A data linkage study. Lancet 2022, 399, 437–446. [Google Scholar] [CrossRef]
- Torjesen, I. Covid-19: Omicron may be more transmissible than other variants and partly resistant to existing vaccines, scientists fear. BMJ 2021, 375, n2943. [Google Scholar] [CrossRef] [PubMed]
- Sui, Y.; Bekele, Y.; Berzofsky, J.A. Potential SARS-CoV-2 Immune Correlates of Protection in Infection and Vaccine Immunization. Pathogens 2021, 10, 138. [Google Scholar] [CrossRef] [PubMed]
- Madhi, S.A.; Kwatra, G.; Myers, J.E.; Jassat, W.; Dhar, N.; Mukendi, C.K.; Nana, A.J.; Blumberg, L.; Welch, R.; Ngorima-Mabhena, N.; et al. Population Immunity and Covid-19 Severity with Omicron Variant in South Africa. N. Engl. J. Med. 2022, 386, 1314–1326. [Google Scholar] [CrossRef]
- Tegally, H.; Moir, M.; Everatt, J.; Giovanetti, M.; Scheepers, C.; Wilkinson, E.; Subramoney, K.; Moyo, S.; Amoako, D.G.; Baxter, C.; et al. Continued Emergence and Evolution of Omicron in South Africa: New BA.4 and BA.5 lineages. medRxiv 2022. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention; U.S. Department of Health & Human Services. Quarantine and Isolation. Available online: https://www.cdc.gov/coronavirus/2019-ncov/your-health/quarantine-isolation.html (accessed on 8 June 2022).
- Manabe, T.; Kambayashi, D.; Akatsu, H.; Kudo, K. Favipiravir for the treatment of patients with COVID-19: A systematic review and meta-analysis. BMC Infect. Dis. 2021, 21, 489. [Google Scholar] [CrossRef]
- Platten, M.; Hoffmann, D.; Grosser, R.; Wisplinghoff, F.; Wisplinghoff, H.; Wiesmüller, G.; Schildgen, O.; Schildgen, V. SARS-CoV-2, CT-Values, and Infectivity-Conclusions to Be Drawn from Side Observations. Viruses 2021, 13, 1459. [Google Scholar] [CrossRef]
- Sirijatuphat, R.; Suputtamongkol, Y.; Angkasekwinai, N.; Horthongkham, N.; Chayakulkeeree, M.; Rattanaumpawan, P.; Koomanachai, P.; Assanasen, S.; Rongrungruang, Y.; Chierakul, N.; et al. Epidemiology, clinical characteristics, and treatment outcomes of patients with COVID-19 at Thailand’s university-based referral hospital. BMC Infect. Dis. 2021, 21, 382. [Google Scholar] [CrossRef] [PubMed]
- Aranha, C.; Patel, V.; Bhor, V.; Gogoi, D. Cycle threshold values in RT-PCR to determine dynamics of SARS-CoV-2 viral load: An approach to reduce the isolation period for COVID-19 patients. J. Med. Virol. 2021, 93, 6794–6797. [Google Scholar] [CrossRef] [PubMed]
- Infantino, M.; Pieri, M.; Nuccetelli, M.; Grossi, V.; Lari, B.; Tomassetti, F.; Calugi, G.; Pancani, S.; Benucci, M.; Casprini, P.; et al. The WHO International Standard for COVID-19 serological tests: Towards harmonization of anti-spike assays. Int. Immunopharmacol. 2021, 100, 108095. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C. The Chalder Fatigue Scale (CFQ 11). Occup. Med. 2015, 65, 86. [Google Scholar] [CrossRef] [Green Version]
- Surapat, B.; Kobpetchyok, W.; Kiertiburanakul, S.; Arnuntasupakul, V. Use of Favipiravir for the Treatment of Coronavirus Disease 2019 in the Setting of Hospitel. Int. J. Clin. Pract. 2022, 2022, 1–8. [Google Scholar] [CrossRef]
- Hassanipour, S.; Arab-Zozani, M.; Amani, B.; Heidarzad, F.; Fathalipour, M.; Martinez-de-Hoyo, R. The efficacy and safety of Favipiravir in treatment of COVID-19: A systematic review and meta-analysis of clinical trials. Sci. Rep. 2021, 11, 11022. [Google Scholar] [CrossRef]
- Sentis, C.; Billaud, G.; Bal, A.; Frobert, E.; Bouscambert, M.; Destras, G.; Josset, L.; Lina, B.; Morfin, F.; Gaymard, A.; et al. SARS-CoV-2 Omicron Variant, Lineage BA.1, Is Associated with Lower Viral Load in Nasopharyngeal Samples Compared to Delta Variant. Viruses 2022, 14, 919. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.-M.; et al. Covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Nyberg, T.; Ferguson, N.M.; Nash, S.G.; Webster, H.H.; Flaxman, S.; Andrews, N.; Hinsley, W.; Bernal, J.L.; Kall, M.; Bhatt, S.; et al. Comparative analysis of the risks of hospitalisation and death associated with SARS-CoV-2 omicron (B.1.1.529) and delta (B.1.617.2) variants in England: A cohort study. Lancet 2022, 399, 1303–1312. [Google Scholar] [CrossRef]
- Wratil, P.R.; Stern, M.; Priller, A.; Willmann, A.; Almanzar, G.; Vogel, E.; Feuerherd, M.; Cheng, C.-C.; Yazici, S.; Christa, C.; et al. Three exposures to the spike protein of SARS-CoV-2 by either infection or vaccination elicit superior neutralizing immunity to all variants of concern. Nat. Med. 2022, 28, 496–503. [Google Scholar] [CrossRef]
- Mehandru, S.; Merad, M. Pathological sequelae of long-haul COVID. Nat. Immunol. 2022, 23, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Elderdery, A.Y.; Elkhalifa, A.M.E.; Alsrhani, A.; Zawbaee, K.I.; Alsurayea, S.M.; Escandarani, F.K.; Alhamidi, A.H.; Idris, H.M.E.; Abbas, A.M.; Shalabi, M.G.; et al. Complete Blood Count Alterations of COVID-19 Patients in Riyadh, Kingdom of Saudi Arabia. J. Nanomater. 2022, 2022, 1–6. [Google Scholar] [CrossRef]
- Pozdnyakova, O.; Connell, N.T.; Battinelli, E.M.; Connors, J.M.; Fell, G.; Kim, A.S. Clinical significance of CBC and WBC morphology in the diagnosis and clinical course of COVID-19 infection. Am. J. Clin. Pathol. 2021, 155, 364–375. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, N.; Zhao, D.; Zhang, J.; Hu, Z.; Tao, Z. Clinical Characteristics of COVID-19 Patients Infected by the Omicron Variant of SARS-CoV-2. Front. Med. 2022, 9, 912367. [Google Scholar] [CrossRef] [PubMed]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 2022, 23, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Montazersaheb, S.; Hosseiniyan Khatibi, S.M.; Hejazi, M.S.; Tarhriz, V.; Farjami, A.; Ghasemian Sorbeni, F.; Farahzadi, R.; Ghasemnejad, T. COVID-19 infection: An overview on cytokine storm and related interventions. Virol. J. 2022, 19, 92. [Google Scholar] [CrossRef]
- Santa Cruz, A.; Mendes-Frias, A.; Oliveira, A.I.; Dias, L.; Matos, A.R.; Carvalho, A.; Capela, C.; Pedrosa, J.; Castro, A.G.; Silvestre, R. Interleukin-6 Is a Biomarker for the Development of Fatal Severe Acute Respiratory Syndrome Coronavirus 2 Pneumonia. Front. Immunol. 2021, 12, 613422. [Google Scholar] [CrossRef]
- Kaaijk, P.; Olivo Pimentel, V.; Emmelot, M.E.; Poelen, M.C.M.; Cevirgel, A.; Schepp, R.M.; den Hartog, G.; Reukers, D.F.M.; Beckers, L.; van Beek, J.; et al. Children and Adults With Mild COVID-19: Dynamics of the Memory T Cell Response up to 10 Months. Front. Immunol. 2022, 13, 817876. [Google Scholar] [CrossRef]
- Zuo, J.; Dowell, A.C.; Pearce, H.; Verma, K.; Long, H.M.; Begum, J.; Aiano, F.; Amin-Chowdhury, Z.; Hoschler, K.; Brooks, T.; et al. Robust SARS-CoV-2-specific T cell immunity is maintained at 6 months following primary infection. Nat. Immunol. 2021, 22, 620–626. [Google Scholar] [CrossRef]
- Gao, Y.; Cai, C.; Grifoni, A.; Müller, T.R.; Niessl, J.; Olofsson, A.; Humbert, M.; Hansson, L.; Österborg, A.; Bergman, P.; et al. Ancestral SARS-CoV-2-specific T cells cross-recognize the Omicron variant. Nat. Med. 2022, 28, 472–476. [Google Scholar] [CrossRef]
- Chamnanphon, M.; Pongpanich, M.; Suttichet, T.B.; Jantarabenjakul, W.; Torvorapanit, P.; Putcharoen, O.; Sodsai, P.; Phokaew, C.; Hirankarn, N.; Chariyavilaskul, P.; et al. Host genetic factors of COVID-19 susceptibility and disease severity in a Thai population. J. Hum. Genet. 2022, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, Y.; Ishigame, H.; Saijo, S.; Nakae, S. Functional specialization of interleukin-17 family members. Immunity 2011, 34, 149–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pappu, R.; Ramirez-Carrozzi, V.; Sambandam, A. The interleukin-17 cytokine family: Critical players in host defence and inflammatory diseases. Immunology 2011, 134, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Cavanaugh, A.M.; Spicer, K.B.; Thoroughman, D.; Glick, C.; Winter, K. Reduced risk of reinfection with SARS-CoV-2 after COVID-19 vaccination—Kentucky, May–June 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1081–1083. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Dattani, S.; Roser, M. Coronavirus Pandemic (COVID-19). Available online: https://ourworldindata.org/coronavirus (accessed on 8 June 2022).
- Kalin, A.; Javid, B.; Knight, M.; Inada-Kim, M.; Greenhalgh, T. Direct and indirect evidence of efficacy and safety of rapid exercise tests for exertional desaturation in COVID-19: A rapid systematic review. Syst. Rev. 2021, 10, 77. [Google Scholar] [CrossRef]

| Characteristics | July–October 2021 (Delta) | January–March 2022 (Omicron) | p *,§ | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All Patients (n = 2704) | S (n = 352) | Favi (n = 2277) | Favi/Dexa (n = 75) | p *,† | All Patients (n = 2477) | S (n = 520) | Favi (n = 1957) | p *,‡ | |||||||||
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | ||||
| Female sex | 1451 | 53.7 | 176 | 50.0 | 1235 | 54.2 | 40 | 53.3 | 0.332 | 1446 | 58.4 | 340 | 65.4 | 1106 | 56.5 | 0.020 | 0.001 |
| Age, year, mean (SD) | 33.8 | (11.6) | 15.7 | (9.4) | 36.0 | (8.7) | 52.5 | (6.4) | <0.001 | 31.3 | (12.3) | 36.0 | (15.4) | 30.1 | (11.2) | <0.001 | <0.001 |
| <25 | 955 | 35.3 | 275 | 78.1 | 677 | 29.7 | 3 | 4.0 | <0.001 | 1015 | 41.0 | 115 | 22.1 | 900 | 46.0 | <0.001 | <0.001 |
| 25–60 | 1470 | 54.4 | 75 | 21.3 | 1350 | 59.3 | 45 | 60.0 | 1220 | 49.3 | 360 | 69.2 | 860 | 43.9 | |||
| >60 | 279 | 10.3 | 2 | 0.6 | 250 | 11.0 | 27 | 36.0 | 242 | 9.8 | 45 | 8.7 | 197 | 10.1 | |||
| Body weight, kg, mean (SD) | 57.5 | (22.0) | 38.3 | (21.4) | 59.7 | (20.9) | 69.7 | (13.2) | 0.043 | 55.8 | (22.4) | 61.6 | (16.7) | 54.2 | (23.4) | 0.034 | 0.007 |
| Presence of comorbidities | |||||||||||||||||
| Diabetes mellitus | 162 | 6.0 | 1 | 0.3 | 151 | 6.6 | 10 | 13.3 | <0.001 | 150 | 6.1 | 34 | 6.5 | 116 | 5.9 | 0.604 | 0.922 |
| Hypertension | 313 | 11.6 | 7 | 2.0 | 283 | 12.4 | 23 | 30.7 | <0.001 | 513 | 20.7 | 116 | 22.3 | 397 | 20.3 | 0.312 | <0.001 |
| Dyslipidemia | 123 | 4.5 | 2 | 0.6 | 113 | 5.0 | 8 | 10.7 | 0.017 | 108 | 4.4 | 16 | 3.1 | 92 | 4.7 | 0.107 | 0.742 |
| Obesity | 23 | 0.9 | 0 | 0.0 | 23 | 1.0 | 0 | 0.0 | 0.114 | 41 | 1.7 | 7 | 1.3 | 34 | 1.7 | 0.534 | 0.009 |
| Malignancy | 22 | 0.8 | 0 | 0.0 | 21 | 0.9 | 1 | 1.3 | 0.176 | 25 | 1.0 | 2 | 0.4 | 23 | 1.2 | 0.109 | 0.458 |
| Neurologic disease | 7 | 0.3 | 0 | 0.0 | 7 | 0.3 | 0 | 0.0 | 0.288 | 271 | 10.9 | 78 | 15.0 | 193 | 9.9 | 0.001 | <0.001 |
| Heart disease | 33 | 1.2 | 1 | 0.3 | 31 | 1.4 | 1 | 1.3 | 0.230 | 38 | 1.5 | 2 | 0.4 | 36 | 1.8 | 0.016 | 0.332 |
| Lung disease | 51 | 1.9 | 1 | 0.3 | 30 | 1.3 | 20 | 26.7 | <0.001 | na | na | na | |||||
| Kidney disease | 14 | 0.5 | 1 | 0.3 | 13 | 0.6 | 0 | 0.0 | 0.641 | 10 | 0.4 | 1 | 0.2 | 9 | 0.5 | 0.392 | 0.546 |
| Others | 336 | 12.4 | 33 | 9.4 | 292 | 12.8 | 11 | 14.7 | 0.158 | 753 | 30.4 | 162 | 31.2 | 591 | 30.2 | 0.674 | <0.001 |
| Presenting symptoms | |||||||||||||||||
| Asymptomatic infection | 390 | 14.4 | 103 | 29.3 | 286 | 12.6 | 1 | 1.3 | <0.001 | 969 | 39.1 | 224 | 43.1 | 745 | 38.1 | 0.038 | <0.001 |
| Fever/history of fever | 1250 | 46.2 | 133 | 37.8 | 1074 | 47.2 | 43 | 57.3 | 0.001 | 267 | 10.8 | 23 | 4.4 | 244 | 12.5 | <0.001 | <0.001 |
| BT ¶ (°C), median (IQR) ‖ | 36.6 | (0.7) | 36.3 | (0.6) | 36.6 | (0.7) | 37.0 | (0.6) | 0.015 | 36.8 | (0.5) | 36.7 | (0.4) | 36.9 | (0.5) | 0.149 | <0.001 |
| <37.5 | 2474 | 95.2 | 318 | 97.0 | 2094 | 95.3 | 62 | 83.8 | 0.002 | 2107 | 88.8 | 489 | 95.5 | 1618 | 86.9 | 0.015 | <0.001 |
| 37.5–38.0 | 120 | 4.6 | 10 | 3.0 | 99 | 4.5 | 11 | 14.9 | 244 | 10.3 | 21 | 4.1 | 223 | 12.0 | |||
| >38.0 | 6 | 0.2 | 0 | 0.0 | 5 | 0.2 | 1 | 1.4 | 23 | 1.0 | 2 | 0.4 | 21 | 1.1 | |||
| Cough | 1642 | 60.7 | 152 | 43.2 | 1425 | 62.6 | 65 | 86.7 | <0.001 | 1181 | 47.7 | 242 | 46.5 | 939 | 48.0 | 0.558 | <0.001 |
| Sore throat | 1038 | 38.4 | 81 | 23.0 | 918 | 40.3 | 39 | 52.0 | 0.010 | 1181 | 47.7 | 242 | 46.5 | 939 | 48.0 | 0.558 | <0.001 |
| Rhinorrhea | 419 | 15.5 | 47 | 13.4 | 358 | 15.7 | 14 | 18.7 | 0.383 | 626 | 25.3 | 143 | 27.5 | 483 | 24.7 | 0.189 | <0.001 |
| Productive sputum | 537 | 19.9 | 46 | 13.1 | 465 | 20.4 | 26 | 34.7 | 0.029 | na | na | na | |||||
| Loss of taste | 312 | 11.5 | 23 | 6.5 | 274 | 12.0 | 15 | 20.0 | 0.010 | 43 | 1.7 | 12 | 2.3 | 31 | 1.6 | 0.261 | <0.001 |
| Loss of smell | 821 | 30.4 | 66 | 18.8 | 725 | 31.8 | 30 | 40.0 | 0.017 | 43 | 1.7 | 12 | 2.3 | 31 | 1.6 | 0.261 | <0.001 |
| Dyspnea | 305 | 11.3 | 5 | 1.4 | 271 | 11.9 | 29 | 38.7 | 0.012 | 24 | 1.0 | 2 | 0.4 | 22 | 1.1 | 0.126 | <0.001 |
| Myalgia | 282 | 10.4 | 11 | 3.1 | 249 | 10.9 | 22 | 29.3 | 0.005 | 206 | 8.3 | 52 | 10.0 | 154 | 7.9 | 0.118 | 0.009 |
| Diarrhea | 126 | 4.7 | 11 | 3.1 | 108 | 4.7 | 7 | 9.3 | <0.001 | 63 | 2.5 | 10 | 1.9 | 53 | 2.7 | 0.312 | <0.001 |
| Nausea/vomiting | 59 | 2.2 | 2 | 0.6 | 49 | 2.2 | 8 | 10.7 | 0.038 | 37 | 1.5 | 5 | 1.0 | 32 | 1.6 | 0.260 | 0.067 |
| Others | 1549 | 57.3 | 168 | 47.7 | 1328 | 58.3 | 53 | 70.7 | <0.001 | 191 | 7.7 | 36 | 6.9 | 155 | 7.9 | 0.449 | <0.001 |
| Clinical features at the time of admission | |||||||||||||||||
| Time from symptom onset to PCR diagnosis, median (IQR), days | 1.9 | (1.6) | 2.1 | (1.9) | 1.9 | (1.6) | 1.9 | (1.5) | 0.394 | 2.0 | (1.1) | 2.1 | (1.0) | 2.0 | (1.1) | 0.167 | 0.049 |
| Time from symptom onset to admission, median (IQR), days | 5.1 | (2.4) | 5.6 | (2.6) | 5.0 | (2.3) | 5.2 | (2.0) | 0.337 | 2.8 | (1.6) | 3.2 | (2.0) | 2.6 | (1.4) | 0.032 | 0.021 |
| Cycle threshold ** | |||||||||||||||||
| Cycle threshold, median (IQR) | 21.0 | (7.8) | 24.1 | (9.4) | 20.7 | (7.4) | 18.5 | (4.3) | 0.001 | 19.0 | (5.7) | 21.4 | (6.9) | 19.7 | (5.3) | 0.001 | <0.001 |
| <20 | 1118 | 43.1 | 88 | 25.9 | 979 | 45.0 | 51 | 68.9 | <0.001 | 1162 | 49.4 | 201 | 38.7 | 961 | 52.4 | <0.001 | <0.001 |
| 20–30 | 1218 | 47.0 | 189 | 55.6 | 1008 | 46.3 | 21 | 28.4 | 974 | 41.4 | 240 | 46.2 | 734 | 40.0 | |||
| >30 | 255 | 9.8 | 63 | 18.5 | 190 | 8.7 | 2 | 2.7 | 216 | 9.2 | 78 | 15.0 | 138 | 7.5 | |||
| Envelope (E), median (IQR) | 17.5 | (8.4) | 20.9 | (9.9) | 17.3 | (8.0) | 14.3 | (4.7) | 0.012 | 18.0 | (5.4) | 17.8 | (5.1) | 19.0 | (6.7) | 0.121 | <0.001 |
| RNA-dependent RNA polymerase (RdRp), median (IQR) | 22.3 | (8.2) | 25.9 | (9.6) | 22.0 | (7.7) | 19.7 | (4.7) | <0.001 | 19.3 | (5.4) | 20.3 | (6.9) | 19.1 | (5.0) | 0.220 | <0.001 |
| Referred back to the hospital, yes | 89 | 3.3 | 3 | 0.9 | 61 | 2.7 | 25 | 33.3 | <0.001 | 43 | 1.7 | 5 | 1.0 | 38 | 1.9 | 0.128 | <0.001 |
| Dead, yes | 5 | 0.2 | 0 | 0.0 | 0 | 0.0 | 5 | 6.7 | <0.001 | 2 | 0.1 | 0 | 0.0 | 2 | 0.1 | 0.466 | 0.308 |
| Characteristics | July–October 2021 (Delta) | January–March 2022 (Omicron) | p *,§ | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All Patients (n = 89) | Alive (n = 84) | Dead (n = 5) | p *,† | All Patients (n = 43) | Alive (n = 41) | Dead (n = 2) | p *,‡ | ||||||||
| n | % | n | % | n | % | n | % | n | % | n | % | ||||
| Male sex | 44 | 49.4 | 43 | 51.2 | 1 | 20.0 | 0.175 | 20 | 46.5 | 18 | 43.9 | 2 | 100.0 | 0.121 | 0.753 |
| Age, year, median (IQR) | 55.0 | (24.0) | 55.5 | (24.5) | 54.0 | (5.0) | 0.617 | 33.0 | (14.0) | 30.5 | (12.0) | 81.0 | (8.5) | 0.073 | <0.001 |
| <18 | 5 | 5.6 | 5 | 6.0 | 0 | 0.0 | 0.017 | 25 | 58.1 | 25 | 61.0 | 0 | 0.0 | 0.022 | 0.697 |
| 18–44 | 26 | 29.2 | 26 | 31.0 | 0 | 0.0 | 4 | 9.3 | 4 | 9.8 | 0 | 0.0 | |||
| 45–64 | 37 | 41.6 | 32 | 38.1 | 5 | 100.0 | 3 | 7.0 | 3 | 7.3 | 0 | 0.0 | |||
| ≥65 | 21 | 23.6 | 21 | 25.0 | 0 | 0.0 | 11 | 25.6 | 9 | 22.0 | 2 | 100.0 | |||
| Presence of comorbidities | |||||||||||||||
| Diabetes mellitus | 23 | 25.8 | 21 | 25.0 | 2 | 40.0 | 0.457 | 7 | 16.3 | 7 | 17.1 | 0 | 0.0 | na | <0.001 |
| Hypertension | 28 | 31.5 | 26 | 31.0 | 2 | 40.0 | 0.672 | 10 | 23.3 | 10 | 24.4 | 0 | 0.0 | na | <0.001 |
| Dyslipidemia | 12 | 13.5 | 12 | 14.3 | 0 | 0.0 | 0.364 | 6 | 14.0 | 6 | 14.6 | 0 | 0.0 | na | <0.001 |
| Heart disease | na | na | na | 3 | 7.0 | 3 | 7.3 | 0 | 0.0 | na | <0.002 | ||||
| Others | 31 | 34.8 | 28 | 33.3 | 3 | 60.0 | 0.224 | 2 | 4.7 | 2 | 4.9 | 0 | 0.0 | na | 0.258 |
| Presenting symptoms of entering HI ¶ | |||||||||||||||
| Asymptomatic infection | 9 | 10.1 | 9 | 10.7 | 0 | 0.0 | 0.440 | 32 | 74.4 | 30 | 73.2 | 2 | 100.0 | 0.396 | 0.020 |
| Fever/history of fever | 49 | 55.1 | 46 | 54.8 | 3 | 60.0 | 0.464 | 27 | 62.8 | 25 | 61.0 | 2 | 100.0 | 0.530 | 0.003 |
| BT ‖ (°C), median (IQR) ** | 36.8 | (0.5) | 36.8 | (0.5) | 36.8 | (0.2) | 0.156 | 36.7 | (0.9) | 36.7 | (0.9) | - | - | na | |
| >38.0 | 9 | 10.1 | 8 | 9.5 | 1 | 20.0 | 0.429 | 2 | 4.7 | 2 | 4.9 | 0 | 0.0 | na | 0.660 |
| Cough | 61 | 68.5 | 58 | 69.0 | 3 | 60.0 | 0.672 | 20 | 46.5 | 18 | 43.9 | 2 | 100.0 | 0.258 | 0.533 |
| URI †† | 45 | 50.6 | 43 | 51.2 | 2 | 40.0 | 0.489 | 21 | 48.8 | 21 | 51.2 | 0 | 0.0 | 0.111 | 0.174 |
| Loss of taste/smell | 22 | 24.7 | 21 | 25.0 | 1 | 20.0 | 0.201 | 1 | 2.3 | 1 | 2.4 | 0 | 0.0 | 0.793 | 0.008 |
| Dyspnea | 53 | 59.6 | 48 | 57.1 | 5 | 100.0 | 0.013 | 2 | 4.7 | 2 | 4.9 | 0 | 0.0 | 0.706 | <0.001 |
| Muscle aches | 23 | 25.8 | 21 | 25.0 | 2 | 40.0 | 0.101 | 8 | 18.6 | 8 | 19.5 | 0 | 0.0 | 0.399 | 0.925 |
| Diarrhea | 13 | 14.6 | 11 | 13.1 | 2 | 40.0 | 0.098 | 2 | 4.7 | 2 | 4.9 | 0 | 0.0 | 0.706 | 0.219 |
| Nausea/vomiting | 10 | 11.2 | 10 | 11.9 | 1 | 20.0 | 0.413 | 6 | 14.0 | 6 | 14.6 | 0 | 0.0 | 0.483 | 0.372 |
| Chest radiograph on referral date | |||||||||||||||
| Pneumonia detected in chest radiograph | 51 | 57.3 | 48 | 57.1 | 3 | 60.0 | 0.638 | 12 | 27.9 | 12 | 29.3 | 0 | 0.0 | na | 0.158 |
| Hematological, median (IQR) | |||||||||||||||
| WBC ‡‡ (×103/µL) | 7.7 | (7.4) | 7.7 | (7.4) | 7.4 | (7.2) | 0.785 | 6.3 | (4.0) | 6.3 | (3.7) | 8.8 | (9.8) | 0.061 | 0.012 |
| Lymphocytes (×103/µL) | 0.8 | (0.8) | 0.9 | (0.4) | 0.8 | (0.8) | 0.038 | 1.8 | (2.1) | 1.8 | (2.0) | 2.1 | (2.6) | 0.914 | <0.001 |
| <1 × 103/uL | 35 | 39.3 | 32 | 38.1 | 3 | 60.0 | 0.592 | 12 | 27.9 | 11 | 26.8 | 1 | 50.0 | 0.492 | 0.001 |
| Neutrophil (×103/µL) | 6.1 | (7.0) | 6.4 | (7.4) | 6.1 | (7.0) | 0.584 | 3.4 | (2.5) | 3.4 | (2.6) | 6.1 | (7.3) | 0.047 | <0.001 |
| Creatinine (mg/dL) | 0.8 | (0.4) | 0.8 | (0.4) | 1.0 | (1.6) | 0.068 | 0.8 | (0.8) | 0.8 | (0.9) | 0.8 | (0.3) | 0.047 | <0.001 |
| eGFR §§ (mL/min/1.73 m2) | 84.5 | (37.5) | 84.5 | (38.2) | 56.4 | (87.1) | 0.080 | 67.0 | (64.5) | 67.0 | (64.5) | - | - | na | 0.139 |
| AST ¶¶ (U/L) | 47.5 | (27.5) | 47.0 | (26.0) | 59.0 | (146.0) | 0.043 | 30.0 | (17.0) | 30.0 | (17.0) | 36.0 | - | na | <0.001 |
| >40 | 35 | 39.3 | 32 | 38.1 | 3 | 60.0 | 0.056 | 10 | 23.3 | 10 | 24.4 | 0 | 0.0 | 0.552 | <0.001 |
| ALT ¶¶ (U/L) | 41.5 | (32.5) | 42.0 | (32.0) | 19.0 | (39.0) | 0.337 | 18.0 | (8.0) | 17.5 | (8.0) | 22.0 | - | na | <0.001 |
| >40 | 29 | 32.6 | 28 | 33.3 | 1 | 20.0 | 0.511 | 3 | 7.0 | 3 | 7.3 | 0 | 0.0 | 0.771 | <0.001 |
| C-reactive protein (mg/L) | 72.1 | (65.2) | 72.5 | (64.0) | 64.1 | (54.7) | 0.154 | 3.4 | (9.2) | 3.4 | (8.7) | 67.6 | (29.3) | 0.035 | 0.008 |
| <10 | 6 | 9.5 | 4 | 6.7 | 2 | 66.7 | 0.005 | 31 | 73.8 | 30 | 75.0 | 1 | 50.0 | 0.114 | <0.001 |
| 10–100 | 40 | 63.5 | 40 | 66.7 | 0 | 0.0 | 8 | 19.0 | 8 | 20.0 | 0 | 0.0 | |||
| >100 | 17 | 27.0 | 16 | 26.7 | 1 | 33.3 | 3 | 7.1 | 2 | 5.0 | 1 | 50.0 | |||
| Procalcitonin (ng/mL) | 0.1 | (0.3) | 0.1 | (0.2) | 0.5 | (1.5) | 0.085 | 0.1 | (0.3) | 0.1 | (0.3) | 0.1 | (0.2) | 0.439 | <0.001 |
| >0.05 | 40 | 83.3 | 36 | 81.8 | 4 | 100.0 | 0.097 | 9 | 69.2 | 7 | 63.6 | 2 | 100.0 | 0.305 | 0.257 |
| D-dimer, median (ng/mL) | 2671.9 | (1008.9) | 2552.1 | (1067.1) | 4308.0 | (2872.6) | 0.059 | 1347.0 | (4588.5) | 1297.0 | (2723.0) | 7633.0 | - | na | 0.029 |
| <500 | 13 | 29.5 | 13 | 31.7 | 0 | 0.0 | 0.215 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0.117 | 0.099 |
| 501–3000 | 25 | 56.8 | 23 | 56.1 | 2 | 66.7 | 5 | 62.5 | 5 | 71.4 | 0 | 0.0 | |||
| >3000 | 6 | 13.6 | 5 | 12.2 | 1 | 33.3 | 3 | 37.5 | 2 | 28.6 | 1 | 100.0 | |||
| Cycle Threshold ‖‖ (viral load at the time of entering HI) | |||||||||||||||
| Nucleocapsid (N), median (IQR) | 19.1 | (5.4) | 19.2 | (5.2) | 16.1 | (3.7) | 0.846 | 18.9 | (3.9) | 18.9 | (3.2) | 18.2 | (5.6) | 0.729 | 0.023 |
| <20 | 57 | 64.0 | 53 | 63.1 | 4 | 80.0 | 0.714 | 31 | 72.1 | 30 | 73.2 | 1 | 50.0 | 0.567 | 0.414 |
| 20–30 | 28 | 31.5 | 27 | 32.1 | 1 | 20.0 | 9 | 20.9 | 8 | 19.5 | 1 | 50.0 | |||
| >30 | 4 | 4.5 | 4 | 4.8 | 0 | 0.0 | 3 | 7.0 | 3 | 7.3 | 0 | 0.0 | |||
| Vaccine status | |||||||||||||||
| Unvaccinated | 58 | 65.2 | 54 | 64.3 | 4 | 80.0 | 0.408 | 29 | 67.4 | 27 | 65.9 | 2 | 100.0 | 0.603 | 0.134 |
| 1 dose | 10 | 11.2 | 9 | 10.7 | 1 | 20.0 | 9 | 20.9 | 9 | 22.0 | 0 | 0.0 | |||
| ≥2 doses | 21 | 23.6 | 21 | 25.0 | 0 | 0.0 | 5 | 11.6 | 5 | 12.2 | 0 | 0.0 | |||
| ORcrude (95% CI) *** | 0.309 (0.189–0.504) | 0.131 (0.052–0.334) | |||||||||||||
| ORage and sex adjusted | 0.142 (0.016–0.265) | 0.109 (0.011–0.318) | |||||||||||||
| ORfully adjusted | 0.299 (0.012–7.643) | 0.105 (0.005–0.294) | |||||||||||||
| Characteristics | n % | Number of Symptoms † (0–12) | Fatigue Score ‡ (0–27) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RR § | 95% CI ¶ | p *,‖ | aRR ** | 95% CI | p *,‖ | RR | 95% CI | p *,†† | aRR | 95% CI | p *,†† | |||
| Female sex | 2194 | 58.4 | 1.02 | (0.94, 1.10) | 0.650 | 1.03 | (0.96, 1.11) | 0.361 | 1.13 | (0.99, 1.30) | 0.071 | 1.15 | (1.00, 1.31) | 0.049 * |
| Age, year, median (range) | 31 (17–47) | 1.00 | (1.00, 1.00) | 0.966 | 1.00 | (1.00, 1.00) | 0.146 | 1.00 | (0.99, 1.00) | 0.458 | 1.00 | (0.99, 1.00) | 0.292 | |
| Comorbidity | ||||||||||||||
| Hypertension | 769 | 20.5 | 1.05 | (0.95, 1.16) | 0.322 | 1.04 | (0.93, 1.16) | 0.488 | 0.97 | (0.81, 1.15) | 0.708 | - | - | - |
| Dyslipidemia | 231 | 6.2 | 1.01 | (0.89, 1.15) | 0.890 | 1.07 | (0.93, 1.22) | 0.353 | 0.97 | (0.77, 1.22) | 0.781 | 1.11 | (0.86, 1.42) | 0.427 |
| Diabetes mellitus | 311 | 8.3 | 0.99 | (0.87, 1.13) | 0.914 | 0.95 | (0.83, 1.09) | 0.491 | 0.83 | (0.65, 1.05) | 0.115 | 0.81 | (0.63, 1.04) | 0.104 |
| Asthma/COPD ‡‡ | 97 | 4.8 | 0.99 | (0.85, 1.15) | 0.882 | 0.99 | (0.86, 1.14) | 0.882 | 0.84 | (0.64, 1.10) | 0.211 | 0.85 | (0.65, 1.11) | 0.232 |
| Chronic heart disease | 65 | 1.7 | 1.07 | (0.83, 1.39) | 0.592 | 1.07 | (0.84, 1.37) | 0.568 | 1.22 | (0.77, 1.93) | 0.39 | 1.30 | (0.83, 2.04) | 0.252 |
| Severity of initial illness | 102 | 2.7 | 1.34 | (1.11, 1.61) | 0.002 * | 1.22 | (1.02, 1.45) | 0.030 * | 1.46 | (0.95, 1.75) | 0.051 | 1.43 | (1.37, 1.72) | 0.039 * |
| Immunosuppression §§, median (range) | 8.89 (7.94–9.53) | 1.05 | (1.03, 1.08) | <0.001 * | 1.04 | (1.01, 1.08) | 0.025 * | 1.04 | (1.00, 1.10) | 0.074 | 1.09 | (1.02, 1.16) | 0.015 * | |
| Neutralizing antibody titers ¶¶, median (range) | 4.58 (4.55–4.59) | 1.22 | (1.08, 1.37) | 0.001 * | 1.22 | (1.05, 1.42) | 0.009 * | 1.18 | (0.80, 1.20) | 0.876 | 1.14 | (0.66, 1.19) | 0.324 | |
| Vaccinated, yes | 2577 | 68.6 | 0.81 | (0.76, 0.87) | <0.001 * | 0.78 | (0.72, 0.84) | <0.001 * | 0.85 | (0.75, 0.97) | 0.014 * | 0.84 | (0.73, 0.96) | 0.011 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mongkolsucharitkul, P.; Surawit, A.; Pumeiam, S.; Sookrung, N.; Tungtrongchitr, A.; Phisalprapa, P.; Sayabovorn, N.; Srivanichakorn, W.; Washirasaksiri, C.; Auesomwang, C.; et al. SARS-CoV-2 Antibody Response against Mild-to-Moderate Breakthrough COVID-19 in Home Isolation Setting in Thailand. Vaccines 2022, 10, 1131. https://doi.org/10.3390/vaccines10071131
Mongkolsucharitkul P, Surawit A, Pumeiam S, Sookrung N, Tungtrongchitr A, Phisalprapa P, Sayabovorn N, Srivanichakorn W, Washirasaksiri C, Auesomwang C, et al. SARS-CoV-2 Antibody Response against Mild-to-Moderate Breakthrough COVID-19 in Home Isolation Setting in Thailand. Vaccines. 2022; 10(7):1131. https://doi.org/10.3390/vaccines10071131
Chicago/Turabian StyleMongkolsucharitkul, Pichanun, Apinya Surawit, Sureeporn Pumeiam, Nitat Sookrung, Anchalee Tungtrongchitr, Pochamana Phisalprapa, Naruemit Sayabovorn, Weerachai Srivanichakorn, Chaiwat Washirasaksiri, Chonticha Auesomwang, and et al. 2022. "SARS-CoV-2 Antibody Response against Mild-to-Moderate Breakthrough COVID-19 in Home Isolation Setting in Thailand" Vaccines 10, no. 7: 1131. https://doi.org/10.3390/vaccines10071131
APA StyleMongkolsucharitkul, P., Surawit, A., Pumeiam, S., Sookrung, N., Tungtrongchitr, A., Phisalprapa, P., Sayabovorn, N., Srivanichakorn, W., Washirasaksiri, C., Auesomwang, C., Sitasuwan, T., Chaisathaphol, T., Tinmanee, R., Chayakulkeeree, M., Phoompoung, P., Tangjittipokin, W., Senawong, S., Sanpawitayakul, G., Muangman, S., ... on behalf of the Siriraj Population Health and Nutrition Research (SPHERE) Group. (2022). SARS-CoV-2 Antibody Response against Mild-to-Moderate Breakthrough COVID-19 in Home Isolation Setting in Thailand. Vaccines, 10(7), 1131. https://doi.org/10.3390/vaccines10071131







