Identification of Naturally Processed Epitope Region Using Artificial APC Expressing a Single HLA Class I Allotype and mRNA of HCMV pp65 Antigen Fragments
Abstract
1. Introduction
2. Materials and Methods
2.1. PBMCs and Ethics Statement
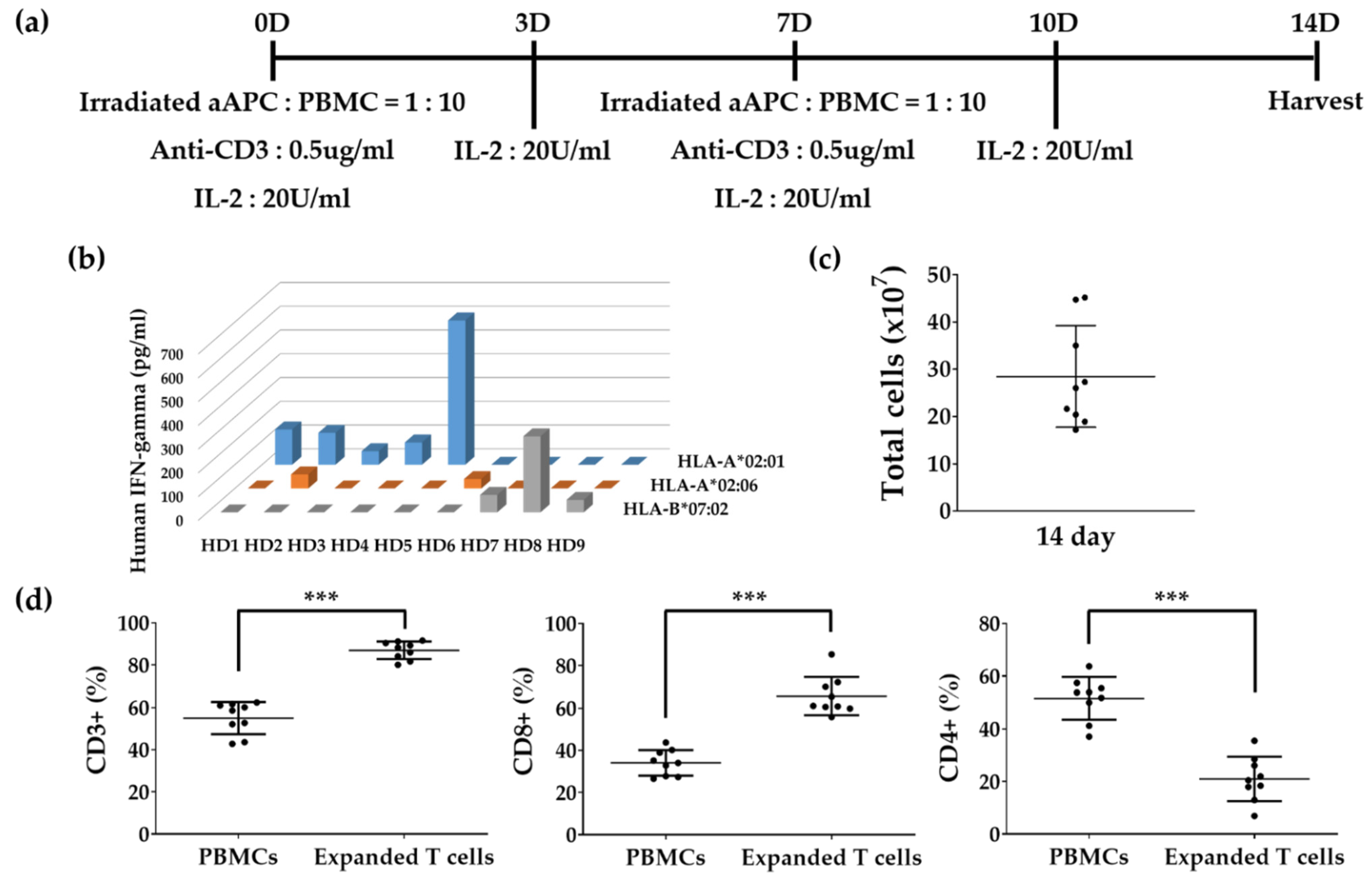
2.2. In Vitro Expansion of T Cells
2.3. Transcriptionally Active PCR (TAP) for pp65 Antigen
2.4. In Vitro Transcription of pp65 Antigen mRNA from TAP Products
2.5. Electroporation of mRNA to aAPCs Expressing a Single HLA Class I Allotype
2.6. IFN-γ ELISA
2.7. Flow Cytometry
2.8. Statistical Analysis
3. Results
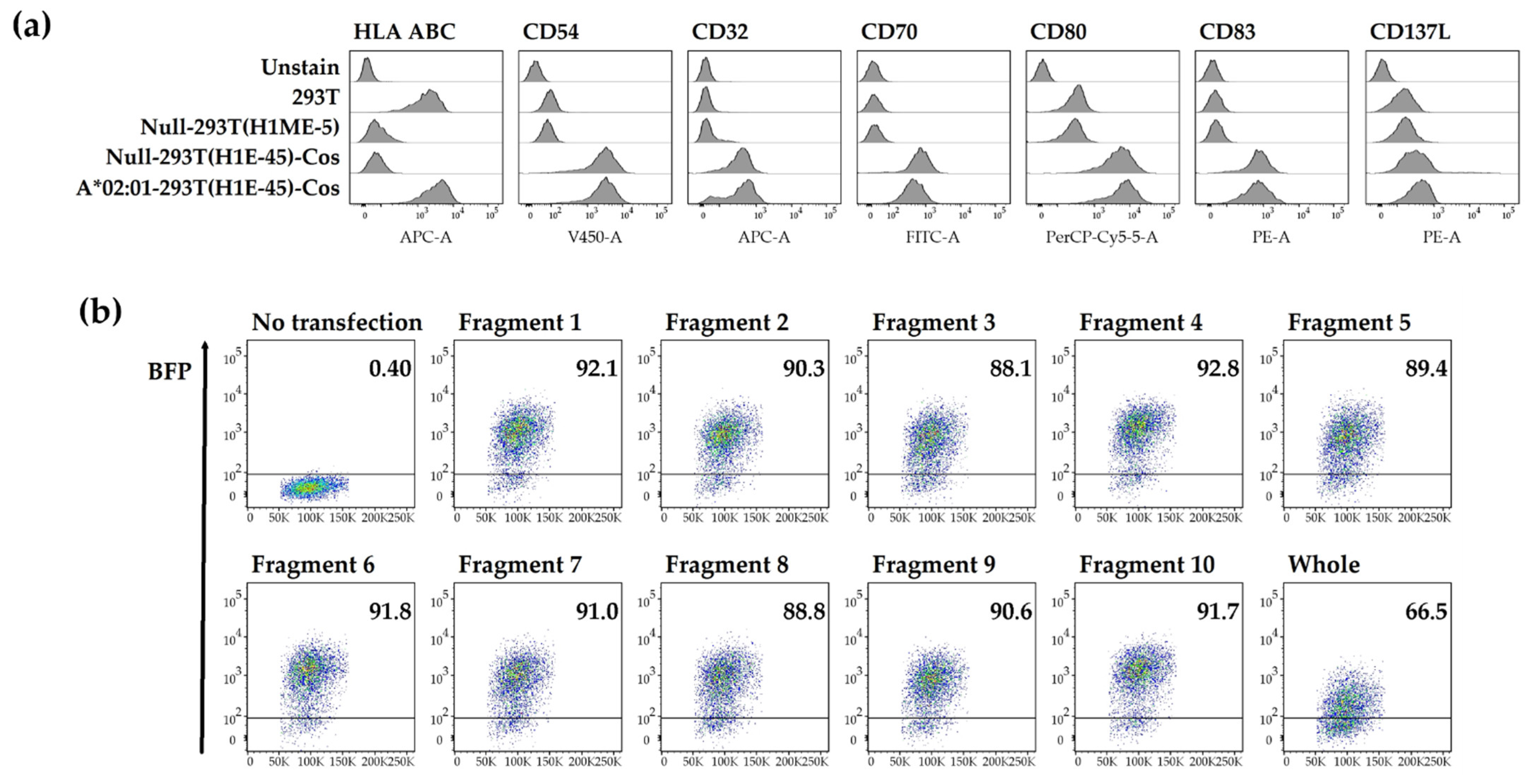
3.1. Generation of mRNA including HCMV pp65 Antigens
3.2. Artificial Antigen-Presenting Cells Expressing a Single HLA Class I Allotype and pp65 Antigen
3.3. In Vitro Expansion of T Cells to Identify Epitopes
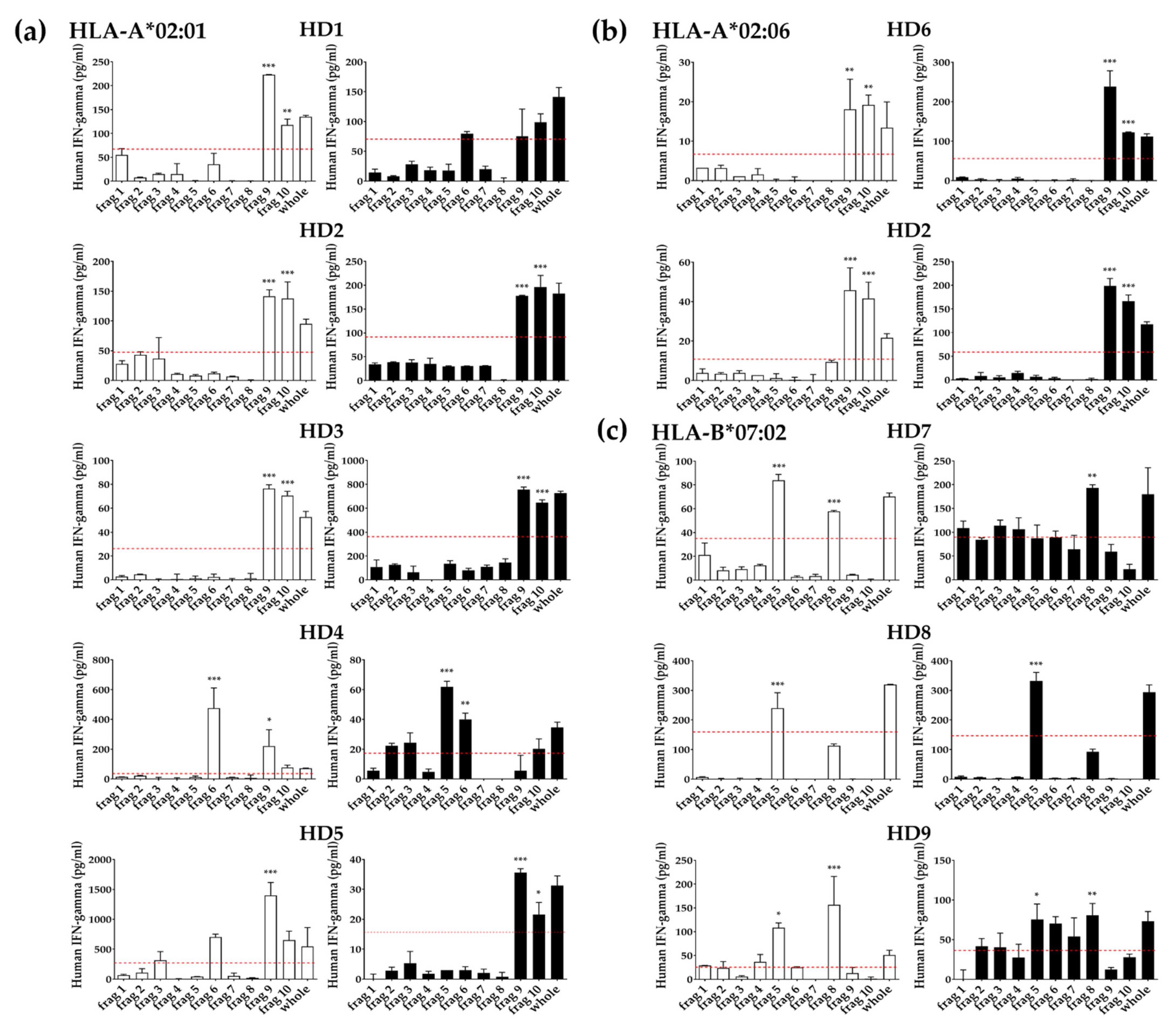
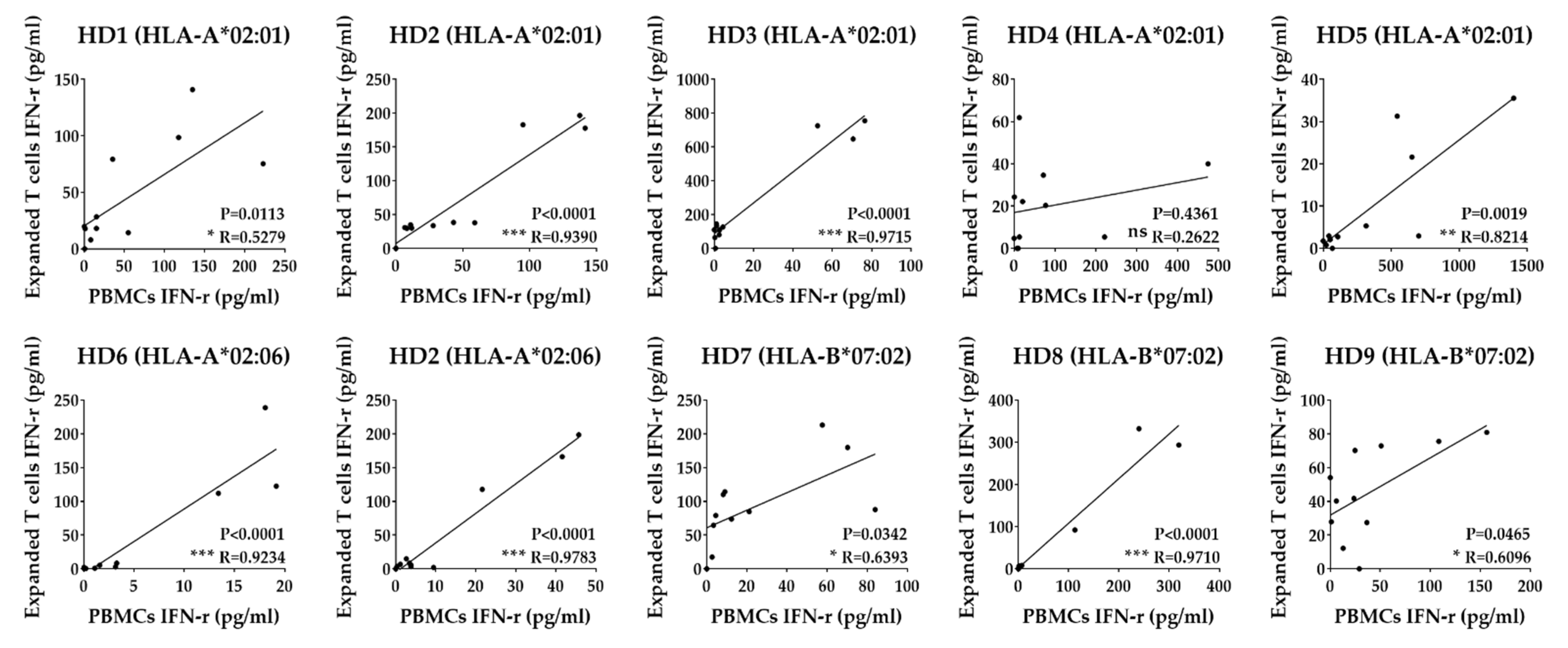
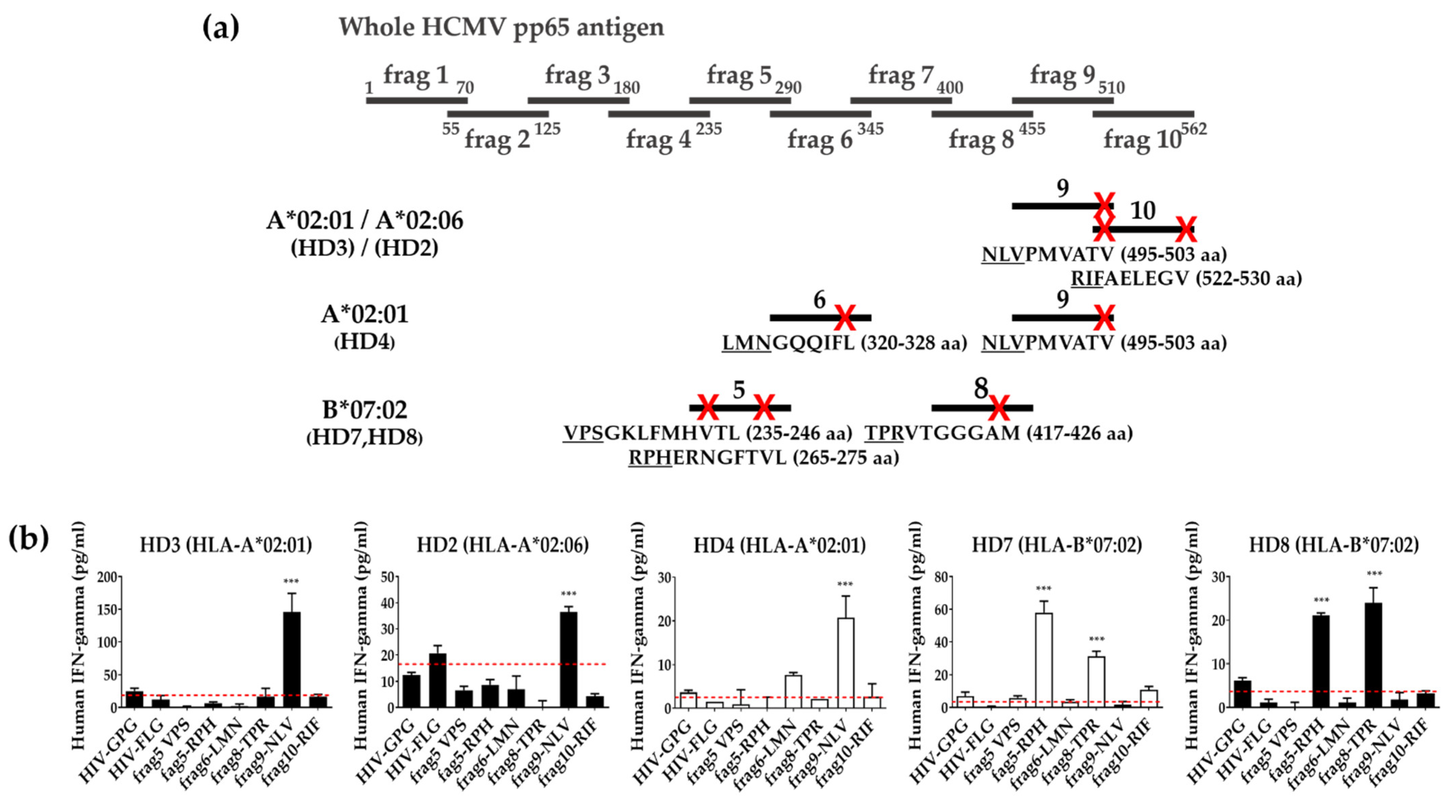
3.4. Identification of T Cell Epitope Region Using aAPCs
3.5. Identification of Epitopes within Fragment Antigens
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Klein, J.; Sato, A. The HLA system. First of two parts. N. Engl. J. Med. 2000, 343, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Pamer, E.; Cresswell, P. Mechanisms of MHC class I—Restricted antigen processing. Annu. Rev. Immunol. 1998, 16, 323–358. [Google Scholar] [CrossRef] [PubMed]
- Tiercy, J.M. Molecular basis of HLA polymorphism: Implications in clinical transplantation. Transpl. Immunol. 2002, 9, 173–180. [Google Scholar] [CrossRef]
- Sidney, J.; Peters, B.; Sette, A. Epitope prediction and identification—Adaptive T cell responses in humans. Semin. Immunol. 2020, 50, 101418. [Google Scholar] [CrossRef] [PubMed]
- Soethout, E.C.; Meiring, H.D.; Jong, A.P.D.; Els, C.A.V. Identifying the epitope-specific T cell response to virus infections. Vaccine 2007, 25, 3200–3203. [Google Scholar] [CrossRef] [PubMed]
- Bakker, A.H.; Hoppes, R.; Linnemann, C.; Toebes, M.; Rodenko, B.; Berkers, C.R.; Hadrup, S.R.; van Esch, W.J.E.; Heemskerk, M.H.M.; Ovaa, H.; et al. Conditional MHC class I ligands and peptide exchange technology for the human MHC gene products HLA-A1, -A3, -A11, and -B7. Proc. Natl. Acad. Sci. USA 2008, 105, 3825–3830. [Google Scholar] [CrossRef]
- Engelhard, V.H. The contributions of mass spectrometry to understanding of immune recognition by T lymphocytes. Int. J. Mass Spectrom. 2007, 259, 32–39. [Google Scholar] [CrossRef]
- Barbosa, C.R.; Bettencourt, P.J. Harnessing the power of CD8+ T-cells: Identification and validation of peptides bound to major histocompatibility complex class I by immunopeptidomics. In Vaccinology and Methods in Vaccine Research; Elsevier: Amsterdam, The Netherlands, 2022; pp. 133–161. [Google Scholar]
- Robbins, P.F.; el-Gamil, M.; Li, Y.F.; Topalian, S.L.; Rivoltini, L.; Sakaguchi, K.; Appella, E.; Kawakami, Y.; Rosenberg, S.A. Cloning of a new gene encoding an antigen recognized by melanoma-specific HLA-A24-restricted tumor-infiltrating lymphocytes. J. Immunol. 1995, 154, 5944–5950. [Google Scholar]
- Speiser, D.E.; Baumgaertner, P.; Voelter, V.; Devevre, E.; Barbey, C.; Rufer, N.; Romero, P. Unmodified self antigen triggers human CD8 T cells with stronger tumor reactivity than altered antigen. Proc. Natl. Acad. Sci. USA 2008, 105, 3849–3854. [Google Scholar] [CrossRef]
- Ho, O.; Green, W.R. Alternative translational products and cryptic T cell epitopes: Expecting the unexpected. J. Immunol. 2006, 177, 8283–8589. [Google Scholar] [CrossRef]
- Strobel, I.; Berchtold, S.; Götze, A.; Schulze, U.; Schuler, G.; Steinkasserer, A. Human dendritic cells transfected with either RNA or DNA encoding influenza matrix protein M1 differ in their ability to stimulate cytotoxic T lymphocytes. Gene Ther. 2000, 7, 2028–2035. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Kim, E.K.; Han, J.Y.; Cho, H.W.; Sohn, H.J.; Kim, S.Y.; Kim, T.G. Fusion of the Human Cytomegalovirus pp65 antigen with both ubiquitin and ornithine decarboxylase additively enhances antigen presentation to CD8(+) T cells in human dendritic cells. Hum. Gene Ther. 2010, 21, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Kim, N.; Sohn, H.; Lee, S.K.; Oh, S.T.; Lee, H.; Cho, H.; Yim, H.W.; Jung, S.E.; Park, G.; et al. Long-term Outcome of Extranodal NK/T Cell Lymphoma Patients Treated With Postremission Therapy Using EBV LMP1 and LMP2a-specific CTLs. Mol. Ther. 2015, 23, 1401–1409. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Yoon, J.S.; Sohn, H.J.; Kim, C.K.; Paik, S.Y.; Hong, Y.K.; Kim, T.G. Direct vaccination with pseudotype baculovirus expressing murine telomerase induces anti-tumor immunity comparable with RNA-electroporated dendritic cells in a murine glioma model. Cancer Lett. 2007, 250, 276–283. [Google Scholar] [CrossRef]
- Meirvenne, S.V.; Straetman, L.; Heirman, C.; Dullaers, M.; Greef, C.D.; Tendeloo, V.V.; Thielemans, K. Efficient genetic modification of murine dendritic cells by electroporation with mRNA. Cancer Gene Ther. 2002, 9, 787–797. [Google Scholar] [CrossRef]
- Britten, C.M.; Meyer, R.G.; Graf, C.; Huber, C.; Wölfel, T. Identification of T cell epitopes by the use of rapidly generated mRNA fragments. J. Immunol. Methods 2005, 299, 165–175. [Google Scholar] [CrossRef]
- Liang, X.; Teng, A.; Braun, D.M.; Felgner, J.; Wang, Y.; Baker, S.I.; Chen, S.; Zelphati, O.; Felgner, P.L. Transcriptionally active polymerase chain reaction (TAP): High throughput gene expression using genome sequence data. J. Biol. Chem. 2002, 277, 3593–3598. [Google Scholar] [CrossRef]
- Doucet, J.D.; Gauchat, D.; Lapointe, R. Identification of T-cell epitopes by a novel mRNA PCR-based epitope chase technique. J. Immunother. 2011, 34, 183–186. [Google Scholar] [CrossRef]
- Gandhi, M.K.; Khanna, R. Human cytomegalovirus: Clinical aspects, immune regulation, and emerging treatments. Lancet Infect. Dis. 2004, 4, 725–738. [Google Scholar] [CrossRef]
- Broers, A.E.C.; Holt, R.V.D.; Esser, J.W.J.V.; Gratama, J.; Henzen-Logmans, S.; Kuenen-Boumeester, V.; Löwenberg, B.; Cornelissen, J. Increased transplant-related morbidity and mortality in CMV-seropositive patients despite highly effective prevention of CMV disease after allogeneic T-cell-depleted stem cell transplantation. Blood 2000, 95, 2240–2245. [Google Scholar] [CrossRef]
- Kondo, E.; Akatsuka, Y.; Kuzushima, K.; Tsujimura, K.; Asakura, S.; Tajima, K.; Kagami, Y.; Kodera, Y.; Tanimoto, M.; Morishima, Y.; et al. Identification of novel CTL epitopes of CMV-pp65 presented by a variety of HLA alleles. Blood 2004, 103, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Kern, F.; Bunde, T.; Faulhaber, N.; Kiecker, F.; Khatamzas, E.; Rudawski, I.; Pruss, A.; Gratama, J.; Volkmer-Engert, R.; Ewert, R.; et al. Cytomegalovirus (CMV) phosphoprotein 65 makes a large contribution to shaping the T cell repertoire in CMV-exposed individuals. J. Infect. Dis. 2002, 185, 1709–1716. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.N.; Kollen, W.J.; Trivedi, D.; Selvakumar, A.; Dupont, B.; Sadelain, M.; O’Reilly, R.J. A panel of artificial APCs expressing prevalent HLA alleles permits generation of cytotoxic T cells specific for both dominant and subdominant viral epitopes for adoptive therapy. J. Immunol. 2009, 183, 2837–2850. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.H.; Sohn, H.J.; Lee, H.J.; Cho, H.I.; Kim, T.G. Antigen Presentation by Individually Transferred HLA Class I Genes in HLA-A, HLA-B, HLA-C Null Human Cell Line Generated Using the Multiplex CRISPR-Cas9 System. J. Immunother. 2017, 40, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Hyun, S.J.; Sohn, H.; Lee, H.; Lee, S.; Kim, S.; Sohn, D.; Hong, C.; Choi, H.; Cho, H.; Kim, T. Comprehensive Analysis of Cytomegalovirus pp65 Antigen-Specific CD8(+) T Cell Responses According to Human Leukocyte Antigen Class I Allotypes and Intraindividual Dominance. Front. Immunol. 2017, 8, 1591. [Google Scholar] [CrossRef]
- Hyun, Y.S.; Jo, H.; Lee, Y.; Kim, S.; Baek, I.; Sohn, H.; Cho, H.; Kim, T. Comprehensive Analysis of CD4(+) T Cell Responses to CMV pp65 Antigen Restricted by Single HLA-DR, -DQ, and -DP Allotype Within an Individual. Front. Immunol. 2020, 11, 602014. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, S.; Tan, S.; Zheng, B.; Gao, G.F. Revival of the identification of cytotoxic T-lymphocyte epitopes for immunological diagnosis, therapy and vaccine development. Exp. Biol. Med. 2011, 236, 253–267. [Google Scholar] [CrossRef]
- Leen, A.M.; Christin, A.; Khalil, M.; Weiss, H.; Gee, A.P.; Brenner, M.K.; Heslop, H.E.; Rooney, C.M.; Bollard, C.M. Identification of hexon-specific CD4 and CD8 T-cell epitopes for vaccine and immunotherapy. J. Virol. 2008, 82, 546–554. [Google Scholar] [CrossRef]
- Regis, D.P.; Dobaño, C.; Quiñones-Olson, P.; Liang, X.; Graber, N.L.; Stefaniak, M.E.; Campo, J.J.; Carucci, D.J.; Roth, D.A.; He, H.; et al. Transcriptionally active PCR for antigen identification and vaccine development: In vitro genome-wide screening and in vivo immunogenicity. Mol. Biochem. Parasitol. 2008, 158, 32–45. [Google Scholar] [CrossRef][Green Version]
- Zhao, Y.; Zheng, Z.; Cohen, C.J.; Gattinoni, L.; Palmer, D.C.; Restifo, N.P.; Rosenberg, S.A.; Morgan, R.A. High-efficiency transfection of primary human and mouse T lymphocytes using RNA electroporation. Mol. Ther. 2006, 13, 151–159. [Google Scholar] [CrossRef]
- Sylwester, A.W.; Mitchell, B.L.; Edgar, J.B.; Taormina, C.; Pelte, C.; Ruchti, F.; Sleath, P.R.; Grabstein, K.H.; Hosken, N.A.; Kern, F.; et al. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J. Exp. Med. 2005, 202, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Weekes, M.P.; Wills, M.R.; Mynard KI, M.; Carmichael, A.J.; Sissons, J.P. The memory cytotoxic T-lymphocyte (CTL) response to human cytomegalovirus infection contains individual peptide-specific CTL clones that have undergone extensive expansion in vivo. J. Virol. 1999, 73, 2099–2108. [Google Scholar] [CrossRef] [PubMed]
- Appay, V.; Dunbar, P.R.; Callan, M.; Klenerman, P.; Gillespie, G.M.A.; Papagno, L.; Ogg, G.S.; King, A.; Lechner, F.; Spina, C.A.; et al. Memory CD8+ T cells vary in differentiation phenotype in different persistent virus infections. Nat. Med. 2002, 8, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Waller, E.C.; Day, E.; Sissons, J.G.; Wills, M.R. Dynamics of T cell memory in human cytomegalovirus infection. Med. Microbiol. Immunol. 2008, 197, 83–96. [Google Scholar] [CrossRef]
- Arenz, M.; Herzog-Hauff, S.; Büschenfelde, K.H.M.Z.; Löhr, H.F. Antigen-independent in vitro expansion of T cells does not affect the T cell receptor V beta repertoire. J. Mol. Med. 1997, 75, 678–686. [Google Scholar] [CrossRef]
- Anthony, D.D.; Lehmann, P.V. T-cell epitope mapping using the ELISPOT approach. Methods 2003, 29, 260–269. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pyo, H.-S.; Hong, C.-H.; Choi, H.; Baek, I.-C.; Kim, T.-G. Identification of Naturally Processed Epitope Region Using Artificial APC Expressing a Single HLA Class I Allotype and mRNA of HCMV pp65 Antigen Fragments. Vaccines 2022, 10, 787. https://doi.org/10.3390/vaccines10050787
Pyo H-S, Hong C-H, Choi H, Baek I-C, Kim T-G. Identification of Naturally Processed Epitope Region Using Artificial APC Expressing a Single HLA Class I Allotype and mRNA of HCMV pp65 Antigen Fragments. Vaccines. 2022; 10(5):787. https://doi.org/10.3390/vaccines10050787
Chicago/Turabian StylePyo, Hong-Seon, Cheol-Hwa Hong, Haeyoun Choi, In-Cheol Baek, and Tai-Gyu Kim. 2022. "Identification of Naturally Processed Epitope Region Using Artificial APC Expressing a Single HLA Class I Allotype and mRNA of HCMV pp65 Antigen Fragments" Vaccines 10, no. 5: 787. https://doi.org/10.3390/vaccines10050787
APA StylePyo, H.-S., Hong, C.-H., Choi, H., Baek, I.-C., & Kim, T.-G. (2022). Identification of Naturally Processed Epitope Region Using Artificial APC Expressing a Single HLA Class I Allotype and mRNA of HCMV pp65 Antigen Fragments. Vaccines, 10(5), 787. https://doi.org/10.3390/vaccines10050787





