Missed Opportunities for Vaccination and Associated Factors among Children Attending Primary Health Care Facilities in Cape Town, South Africa: A Pre-Intervention Multilevel Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Setting
2.3. Study Population
2.4. Sample Size Estimation
2.5. Sampling
2.6. Data Collection
2.7. Variables
- Level 1 (individual-level factors). These included child-related factors (such as age of child, sex of child, birth order and birth weight) and caregiver-related factors (such as caregiver relationship with child, marital status, mothers’ attendance of antenatal care, level of education, employment status, mode of transport to health facility, duration of transport to health facility and recent exposure to immunisation messages);
- Level 2 (health-facility-level factors): These included facility-related factors, such as facility size (clinic vs. community health centre (CHC)), location (sub-district), number of staff, patient load, vaccine availability or stock-outs and immunisation scheduling.
2.8. Data Analysis
3. Results
3.1. Participants’ Characteristics
3.2. Facility Characteristics
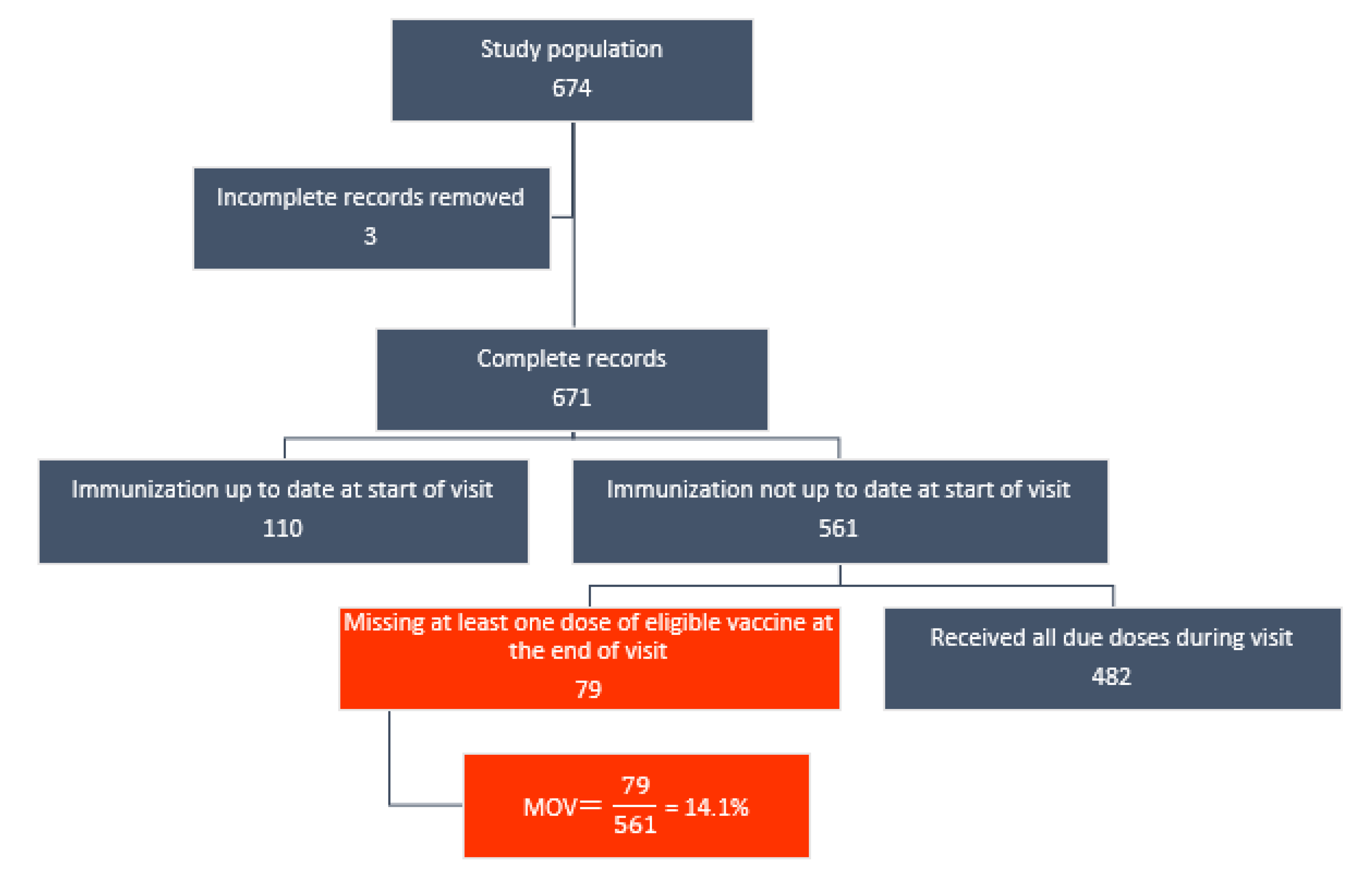
3.3. Prevalence of MOV
3.4. Dose-Specific MOV Prevalence
3.5. Measures of Association (Fixed Effects)
3.6. Measures of Variations (Random Effects)
4. Discussion
4.1. Main Findings
4.2. Implications for Immunisation Practice and Quality Improvement
4.3. Implications for Broader Policy and Practice
4.4. Limitations and Strengths
4.5. Implications for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wilder-Smith, A.; Longini, I.; Zuber, P.L.; Bärnighausen, T.; Edmunds, W.J.; Dean, N.; Spicher, V.M.; Benissa, M.R.; Gessner, B.D. The public health value of vaccines beyond efficacy: Methods, measures and outcomes. BMC Med. 2017, 15, 138. [Google Scholar] [CrossRef] [PubMed]
- Bloom, D.E.; Canning, D.; Weston, M. The value of vaccination. World Econ. 2005, 6, 15. [Google Scholar]
- Maurice, J.M.; Davey, S. State of the World’s Vaccines and Immunization; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- World Health Organization. Immunization Coverage. Available online: http://www.who.int/mediacentre/factsheets/fs378/en/ (accessed on 31 October 2017).
- GBD 2016 Mortality Collaborators. Global, regional, and national under-5 mortality, adult mortality, age-specific mortality, and life expectancy, 1970–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1084–1150. [Google Scholar]
- Kaslow, D.C.; Kalil, J.; Bloom, D.; Breghi, G.; Colucci, A.M.; De Gregorio, E.; Madhavan, G.; Meier, G.; Seabrook, R.; Xu, X. The role of vaccines and vaccine decision-making to achieve the goals of the Grand Convergence in public health. Vaccine 2017, 35, A10–A15. [Google Scholar] [CrossRef]
- Ozawa, S.; Mirelman, A.; Stack, M.L.; Walker, D.G.; Levine, O.S. Cost-effectiveness and economic benefits of vaccines in low- and middle-income countries: A systematic review. Vaccine 2012, 31, 96–108. [Google Scholar] [CrossRef]
- Sartori, A.M.; de Soarez, P.C.; Novaes, H.M.; Amaku, M.; de Azevedo, R.S.; Moreira, R.C.; Pereira, L.M.; Ximenes, R.A.; Martelli, C.M. Cost-effectiveness analysis of universal childhood hepatitis A vaccination in Brazil: Regional analyses according to the endemic context. Vaccine 2012, 30, 7489–7497. [Google Scholar] [CrossRef]
- Nnaji, C.A.; Owoyemi, A.J.; Amaechi, U.A.; Wiyeh, A.B.; Ndwandwe, D.E.; Wiysonge, C.S. Taking stock of global immunisation coverage progress: The gains, the losses and the journey ahead. Int. Health 2020, 13, 653–657. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO/UNICEF Estimates of National Immunization Coverage, Updated July 2020. Available online: https://apps.who.int/immunization_monitoring/globalsummary/timeseries/tswucoveragedtp3.html (accessed on 11 January 2022).
- Ndwandwe, D.; Nnaji, C.A.; Mashunye, T.; Uthman, O.A.; Wiysonge, C.S. Incomplete vaccination and associated factors among children aged 12–23 months in South Africa: An analysis of the South African demographic and health survey 2016. Hum. Vaccines Immunother. 2021, 17, 247–254. [Google Scholar] [CrossRef]
- Nyasulu, J.; Pandya, H. The effects of coronavirus disease 2019 pandemic on the South African health system: A call to maintain essential health services. Afr. J. Prim. Health Care Fam. Med. 2020, 12, e1–e5. [Google Scholar] [CrossRef]
- Mbunge, E. Effects of COVID-19 in South African health system and society: An explanatory study. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 1809–1814. [Google Scholar] [CrossRef]
- Siedner, M.J.; Kraemer, J.D.; Meyer, M.J.; Harling, G.; Mngomezulu, T.; Gabela, P.; Dlamini, S.; Gareta, D.; Majozi, N.; Ngwenya, N.; et al. Access to primary healthcare during lockdown measures for COVID-19 in rural South Africa: A longitudinal cohort study. medRxiv 2020. [Google Scholar] [CrossRef]
- Siedner, M.J.; Kraemer, J.D.; Meyer, M.J.; Harling, G.; Mngomezulu, T.; Gabela, P.; Dlamini, S.; Gareta, D.; Majozi, N.; Ngwenya, N.; et al. Access to primary healthcare during lockdown measures for COVID-19 in rural South Africa: An interrupted time series analysis. BMJ Open 2020, 10, e043763. [Google Scholar] [CrossRef] [PubMed]
- Jacob, N.; Coetzee, D. Missed opportunities for immunisation in health facilities in Cape Town, South Africa. S. Afr. Med. J. 2015, 105, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Mothiba, T.M.; Tladi, F.M. Challenges faced by professional nurses when implementing the Expanded Programme on Immunisation at rural clinics in Capricorn District, Limpopo. Afr. J. Prim. Health Care Fam. Med. 2016, 8, e1–e5. [Google Scholar] [CrossRef] [PubMed]
- Wiysonge, C.S.; Ngcobo, N.J.; Jeena, P.M.; Madhi, S.A.; Schoub, B.D.; Hawkridge, A.; Shey, M.S.; Hussey, G.D. Advances in childhood immunisation in South Africa: Where to now? Programme managers’ views and evidence from systematic reviews. BMC Public Health 2012, 12, 578. [Google Scholar] [CrossRef] [PubMed]
- Burnett, R.J.; Mmoledi, G.; Ngcobo, N.J.; Dochez, C.; Seheri, L.M.; Mphahlele, M.J. Impact of vaccine stock-outs on infant vaccination coverage: A hospital-based survey from South Africa. Int. Health 2018, 10, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, S.S.; Jansen, H.; Robertson, S.E.; Evans, P.; Kim-Farley, R.J. Studies of missed opportunities for immunization in developing and industrialized countries. Bull. World Health Organ. 1993, 71, 549. [Google Scholar]
- Nnaji, C.A.; Ndwandwe, D.; Lesosky, M.; Mahomed, H.; Wiysonge, C.S. Tackling missed opportunities for vaccination in a new era of immunisation. Lancet 2021, 398, 21. [Google Scholar] [CrossRef]
- World Health Organization. Methodology for the Assessment of Missed Opportunities for Vaccination; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Sridhar, S.; Maleq, N.; Guillermet, E.; Colombini, A.; Gessner, B.D. A systematic literature review of missed opportunities for immunization in low- and middle-income countries. Vaccine 2014, 32, 6870–6879. [Google Scholar] [CrossRef]
- Uthman, O.A.; Sambala, E.Z.; Adamu, A.A.; Ndwandwe, D.; Wiyeh, A.B.; Olukade, T.; Bishwajit, G.; Yaya, S.; Okwo-Bele, J.-M.; Wiysonge, C.S. Does it really matter where you live? A multilevel analysis of factors associated with missed opportunities for vaccination in sub-Saharan Africa. Hum. Vaccines Immunother. 2018, 14, 2397–2404. [Google Scholar] [CrossRef]
- Sambala, E.Z.; Uthman, O.A.; Adamu, A.A.; Ndwandwe, D.; Wiyeh, A.B.; Olukade, T.; Bishwajit, G.; Yaya, S.; Okwo-Bele, J.-M.; Wiysonge, C.S. Mind the Gap: What explains the education-related inequality in missed opportunities for vaccination in sub-Saharan Africa? Compositional and structural characteristics. Hum. Vaccines Immunother. 2018, 14, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Adedokun, S.T.; Uthman, O.A.; Adekanmbi, V.T.; Wiysonge, C.S. Incomplete childhood immunization in Nigeria: A multilevel analysis of individual and contextual factors. BMC Public Health 2017, 17, 236. [Google Scholar] [CrossRef] [PubMed]
- Etana, B.; Deressa, W. Factors associated with complete immunization coverage in children aged 12–23 months in Ambo Woreda, Central Ethiopia. BMC Public Health 2012, 12, 566. [Google Scholar] [CrossRef] [PubMed]
- Yismaw, A.E.; Assimamaw, N.T.; Bayu, N.H.; Mekonen, S.S. Incomplete childhood vaccination and associated factors among children aged 12–23 months in Gondar city administration, Northwest, Ethiopia 2018. BMC Res. Notes 2019, 12, 241. [Google Scholar] [CrossRef] [PubMed]
- Zida-Compaore, W.I.C.; Ekouevi, D.K.; Gbeasor-Komlanvi, F.A.; Sewu, E.K.; Blatome, T.; Gbadoe, A.D.; Agbere, D.A.; Atakouma, Y. Immunization coverage and factors associated with incomplete vaccination in children aged 12 to 59 months in health structures in Lome. BMC Res. Notes 2019, 12, 84. [Google Scholar] [CrossRef]
- Adamu, A.A.; Uthman, O.A.; Gadanya, M.A.; Adetokunboh, O.O.; Wiysonge, C.S. A multilevel analysis of the determinants of missed opportunities for vaccination among children attending primary healthcare facilities in Kano, Nigeria: Findings from the pre-implementation phase of a collaborative quality improvement programme. PLoS ONE 2019, 14, e0218572. [Google Scholar] [CrossRef]
- Adetokunboh, O.; Iwu-Jaja, C.J.; Nnaji, C.A.; Ndwandwe, D. Missed opportunities for vaccination in Africa. Curr. Opin. Immunol. 2021, 71, 55–61. [Google Scholar] [CrossRef]
- Ogbuanu, I.U.; Li, A.J.; Anya, B.-P.M.; Tamadji, M.; Chirwa, G.; Chiwaya, K.W.; Djalal, M.E.-H.; Cheikh, D.; Machekanyanga, Z.; Okeibunor, J.; et al. Can vaccination coverage be improved by reducing missed opportunities for vaccination? Findings from assessments in Chad and Malawi using the new WHO methodology. PLoS ONE 2019, 14, e0210648. [Google Scholar] [CrossRef]
- Okeibunor, J.C.; Ogbuanu, I.; Blanche, A.; Chiwaya, K.; Chirwa, G.; Machekanyanga, Z.; Mihigo, R.; Zawaira, F. Towards a Strategy for Reducing Missed Opportunities for Vaccination in Malawi: Implications of a Qualitative Health Facility Assessment. J. Immunol. Sci. 2018, 7, 46–54. [Google Scholar] [CrossRef]
- Magadzire, B.P.; Joao, G.; Shendale, S.; Ogbuanu, I.U. Reducing missed opportunities for vaccination in selected provinces of Mozambique: A study protocol. Gates Open Res. 2017, 1, 5. [Google Scholar] [CrossRef]
- Li, A.J.; Peiris, T.S.R.; Sanderson, C.; Nic Lochlainn, L.; Mausiry, M.; da Silva, R.B.J.B.M.; Ogbuanu, I.U. Opportunities to improve vaccination coverage in a country with a fledgling health system: Findings from an assessment of missed opportunities for vaccination among health center attendees-Timor Leste, 2016. Vaccine 2019, 37, 4281–4290. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, C.A.; Yach, D.; de Beer, Z.J. Missed opportunities for immunisation at hospitals in the western Cape—A reappraisal. S. Afr. Med. J. 1994, 84, 149–152. [Google Scholar] [PubMed]
- Ndwandwe, D.; Nnaji, C.A.; Wiysonge, C.S. The Magnitude and Determinants of Missed Opportunities for Childhood Vaccination in South Africa. Vaccines 2020, 8, 705. [Google Scholar] [CrossRef] [PubMed]
- Nnaji, C.A.; Wiysonge, C.S.; Lesosky, M.; Mahomed, H.; Ndwandwe, D. COVID-19 and the Gaping Wounds of South Africa’s Suboptimal Immunisation Coverage: An Implementation Research Imperative for Assessing and Addressing Missed Opportunities for Vaccination. Vaccines 2021, 9, 691. [Google Scholar] [CrossRef] [PubMed]
- Western Cape Provincial Department of Health. Cape Metro District Health Plan 2018–2020. Available online: http://www.health.gov.za/DHP/docs/DHP2018-21/Western_Cape/Metro_District_Health_Plan.pdf (accessed on 31 January 2020).
- Burnett, R.J.; Dlamini, N.R.; Meyer, J.C.; Fernandes, L.; Motloung, B.R.; Ndlovu, T.H.; Simango, H.A.; Kibuuka, D.K.; Dochez, C.; Montwedi, D.N.; et al. Progress towards obtaining valid vaccination coverage data in South Africa. S. Afr. J. Sci. 2019, 115, 1–4. [Google Scholar] [CrossRef]
- Adamu, A.A.; Sarki, A.M.; Uthman, O.A.; Wiyeh, A.B.; Gadanya, M.A.; Wiysonge, C.S. Prevalence and dynamics of missed opportunities for vaccination among children in Africa: Applying systems thinking in a systematic review and meta-analysis of observational studies. Expert Rev. Vaccines 2019, 18, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Biswas, T. How to calculate sample size for different study designs in medical research? Indian J. Psychol. Med. 2013, 35, 121. [Google Scholar] [CrossRef] [PubMed]
- Israel, G.D. Determining Sample Size; EDIS Gainesville, Institute of Food and Agriculture Sciences, University of Florida Cooperative Extension Service: Gainesville, FL, USA, 1992. [Google Scholar]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Velandia-González, M.; Trumbo, S.P.; Díaz-Ortega, J.L.; Bravo-Alcántara, P.; Danovaro-Holliday, M.C.; Dietz, V.; Ruiz-Matus, C. Lessons learned from the development of a new methodology to assess missed opportunities for vaccination in Latin America and the Caribbean. BMC Int. Health Hum. Rights 2015, 15, 5. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, H.; Browne, W.; Rasbash, J. Partitioning Variation in Multilevel Models. Underst. Stat. 2002, 1, 223–231. [Google Scholar] [CrossRef]
- Courgeau, D.; Baccaini, B. Multilevel analysis in the social sciences. Popul. Engl. Sel. 1998, 10, 39–71. [Google Scholar]
- Spiegelhalter, D.J.; Best, N.G.; Carlin, B.P.; Van Der Linde, A. Bayesian measures of model complexity and fit. J. R. Stat. Soc. Ser. B (Stat. Methodol.) 2002, 64, 583–639. [Google Scholar] [CrossRef]
- Browne, W.J. MCMC Estimation in MLwiN, version 2.31; Centre for Multilevel Modelling, University of Bristol: Bristol, UK, 2014. [Google Scholar]
- Leckie, G.; Charlton, C. Runmlwin: A Program to Run the MLwiN Multilevel Modeling Software from within Stata. J. Stat. Softw. 2013, 52, 1–40. [Google Scholar] [CrossRef]
- Stata Statistical Software, version 14; StataCorp: College Station, TX, USA, 2015.
- Brugha, R. Missed opportunities for immunizations at curative and preventive health care visits. Trans. R. Soc. Trop. Med. Hyg. 1995, 89, 698. [Google Scholar] [CrossRef]
- Anderson, E.L. Recommended solutions to the barriers to immunization in children and adults. Mol. Med. 2014, 111, 344–348. [Google Scholar]
- Ekhaguere, O.A.; Oluwafemi, R.O.; Oyo-Ita, A.; Mamlin, B.; Bondich, P.; Mendonca, E.A.; Rollins, A.L. Determinants of Long Immunization Clinic Wait Times in a Sub-Saharan African Country. Glob. Pediatr. Health 2021, 8, X211028211–X212333794. [Google Scholar] [CrossRef]
- Kaufman, J.; Ryan, R.; Walsh, L.; Horey, D.; Leask, J.; Robinson, P.; Hill, S. Face-to-face interventions for informing or educating parents about early childhood vaccination. Cochrane Database Syst. Rev. 2018, 5, Cd010038. [Google Scholar] [CrossRef]
- Saeterdal, I.; Lewin, S.; Austvoll-Dahlgren, A.; Glenton, C.; Munabi-Babigumira, S. Interventions aimed at communities to inform and/or educate about early childhood vaccination. Cochrane Database Syst. Rev. 2014, 11, Cd010232. [Google Scholar] [CrossRef]
- Boland, L.; Kryworuchko, J.; Saarimaki, A.; Lawson, M.L. Parental decision making involvement and decisional conflict: A descriptive study. BMC Pediatr. 2017, 17, 146. [Google Scholar] [CrossRef]
- Otsuka-Ono, H.; Hori, N.; Ohta, H.; Uemura, Y.; Kamibeppu, K. A childhood immunization education program for parents delivered during late pregnancy and one-month postpartum: A randomized controlled trial. BMC Health Serv. Res. 2019, 19, 798. [Google Scholar] [CrossRef]
- World Health Organization. Summary of the April 2016 Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE); World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Abbas, K.; Procter, S.R.; van Zandvoort, K.; Clark, A.; Funk, S.; Mengistu, T.; Hogan, D.; Dansereau, E.; Jit, M.; Flasche, S.; et al. Routine childhood immunisation during the COVID-19 pandemic in Africa: A benefit–risk analysis of health benefits versus excess risk of SARS-CoV-2 infection. Lancet Glob. Health 2020, 8, E1264–E1272. [Google Scholar] [CrossRef]
- Department of Health. Quality Improvement—The Key to Providing Improved Quality of Care. 2012. Available online: https://www.idealhealthfacility.org.za/docs/guidelines/Quality%20Improvement%20Guide%202012.pdf (accessed on 25 June 2020).
- Department of Health. National Core Standards for Health Establishments in South Africa. 2011. Available online: http://rhap.org.za/wp-content/uploads/2014/05/National-Core-Standards-2011-1.pdf (accessed on 25 June 2020).
| Age | Vaccine Offered |
|---|---|
| Birth | BCG, OPV (0) |
| 6 Weeks | OPV (1), RV (1), DTaP-IPV-Hib-HepB (1), PCV (1) |
| 10 Weeks | DTaP-IPV-HIB-HepB (2) |
| 14 Weeks | RV (2), DTaP-IPV-Hib-HepB (3), PCV (2) |
| 6 Months | Measles (1) |
| 9 Months | PCV (3) |
| 12 Months | Measles (2) |
| 18 Months | DTaP-IPV-Hib-HepB (4) |
| 6 Years | Td (1) |
| 9 Years | HPV (1), HPV (2) (2 doses, 6 months apart) * |
| 12 Years | Td (2) |
| Variables | Number of Children (%) | MOV Prevalence (%) | p-Value# |
|---|---|---|---|
| All children | 561 | 79 (14.1) | |
| Sub-district | |||
| Southern | 99 (17.8) | 9 (9.1) | |
| Western | 38 (6.8) | 5 (13.2) | |
| Tygerberg | 240 (43.2) | 32 (13.3) | |
| M/Plain | 89 (16.0) | 15 (16..9) | |
| Khayelitsha | 90 (16.2) | 17 (18.9) | 0.346 |
| Facility type | |||
| CHC | 269 (48.4) | 36 (13.4) | |
| Clinic | 287 (51.6) | 42 (14.6) | 0.671 |
| Facility ownership | |||
| Province | 213 (38.2) | 29 (13.6) | |
| City | 345 (61.8) | 49 (14.4) | 0.902 |
| Vaccine stock-out in the past 3 months | |||
| Yes | 125 (22.4) | 17 (13.6) | |
| No | 432 (77.6) | 61 (14.1) | 0.883 |
| Vaccine cold-chain challenges in the past months | |||
| Yes | 32 (5.7) | 2 (6.3) | |
| No | 526 (94.3) | 76 (14.5) | 0.194 |
| Number of health workers | |||
| Fewer than 50 | 349 (62.8) | 56 (16,1) | |
| 50 or more | 207 (37.2) | 10 (10.63) | 0.075 |
| Immunisation waiting time | |||
| Less than 30 min | 147 (30.4) | 14 (9.5) | |
| 30 min or longer | 336 (69.6) | 51 (15.1) | 0.070 |
| Child-level factors | |||
| Age of child | Mean (SD): 7.5 (1.2) months | ||
| 0–11 months | 420 (75.3) | 62 (14.8) | |
| 12–23 months | 138 (24.7) | 16 (11.6) | 0.148 |
| Sex of child | |||
| Female | 280 (49.9) | 33 (11.8) | |
| Male | 281 (50.1) | 45 (16.0) | 0.148 |
| Birth order | |||
| 1st–3rd order | 501 (89.3) | 67 (13.4) | |
| 4th+ order | 60 (10.7) | 11 (18.3) | 0.294 |
| Birth size | |||
| Large | 141 (25.4) | 23 (16.3) | |
| Average | 351 (63.2) | 47 (13.9) | |
| Small | 63 (11.4) | 7 (11.1) | 0.556 |
| Reason for visit | |||
| Vaccination | 445 (79.3) | 55 (12.4) | |
| Non-vaccination | 116 (20.7) | 23 (19.8) | 0.038 |
| Time of visit | |||
| Morning | 342 (61.7) | 48 (14.0) | |
| Afternoon | 212 (38.3) | 30 (14.2) | 0.970 |
| Caregiver-related factors | |||
| Caregiver age | Mean (SD): 29.3 (6.9) years | ||
| 18–24 years | 153 (27.4) | 24 (15.7) | |
| 25–34 years | 281 (50.4) | 38 (13.5) | |
| 35+ years | 124 (22.2) | 16 (12.9) | 0.764 |
| Level of education | |||
| Primary | 26 (4.6) | 7 (26.9) | |
| Post-primary | 535 (95.4) | 71 (13.3) | 0.049 |
| Relationship to child | |||
| Mother | 519 (92.5) | 71 (13.7) | |
| Other relation | 42 (7.3) | 7 (16.7) | 0.591 |
| Marital status | |||
| Not Married | 369 (65.8) | 56 (15.2) | |
| Married | 192 (34.2) | 22 (11.5) | 0.227 |
| Maternal antenatal care | |||
| Attended | 546 | 75 (13.7) | |
| Never attended | 6 | 3 (50.0) | 0.034 |
| Employment status | |||
| Employed | 138 (30.0) | 19 (13.8) | |
| Unemployed | 423 (70.0) | 59 (14.0) | 0.958 |
| Means of transport | |||
| Own vehicle | 61 (11.0) | 3 (4.9) | |
| Public transport | 178 (32.0) | 21 (11.8) | |
| Walk | 318 (57.0) | 54 (17.0) | 0.027 |
| Child immunisation message in the last 3 months | |||
| Yes | 341 (61.3) | 36 (10.6) | |
| No | 215 (38.7) | 42 (19.5) | 0.003 |
| Child immunisation decision making | |||
| Both parents | 117 (20.1) | 10 (8.6) | |
| Not both parents | 444 (79.1) | 65 (15.3) | 0.060 |
| Age of Eligibility | Vaccine Antigen (Dose) | Number Eligible | MOV (%) |
| All doses | 561 | 79 (14.1) | |
| Birth | BCG | 555 | 9 (1.6) |
| OPV (0) | 555 | 15 (2.7) | |
| 6 weeks | OPV (1) | 520 | 34 (6.5) |
| RV (1) | 520 | 3 (0.6) | |
| DtaP-IPV-Hib-HepB (1) | 520 | 3 (0.6) | |
| PCV (1) | 520 | 5 (1.0) | |
| 10 weeks | DtaP-IPV-Hib-HepB (2) | 439 | 6 (1.4) |
| 14 weeks | RV (2) | 381 | 14 (3.7) |
| PCV (2) | 381 | 12 (3.2) | |
| DtaP-IPV-Hib-HepB (3) | 381 | 10 (2.6) | |
| 6 months | Measles (1) | 287 | 12 (4.2) |
| 9 months | PCV (3) | 205 | 6 (2.9) |
| 12 months | Measles (2) | 137 | 13 (9.5) |
| 18 months | DtaP-IPV-Hib-HepB (4) | 59 | 5 (8.5) |
| Model 1 a OR (95% CrI) | Model 2 b OR (95% CrI) | Model 3 c OR (95% CrI) | Model 4 d OR (95% CrI) | |
|---|---|---|---|---|
| INDIVIDUAL-LEVEL FACTORS | ||||
| Age of child | ||||
| ref | ref | ||
| 0.58 (0.27–1.25) | 0.61 (0.28–1.35) | ||
| Sex of child | ||||
| ref | ref | ||
| 0.63 (0.35–1.14) | 0.70 (0.39–1.28) | ||
| Birth order | ||||
| ref | ref | ||
| 1.83 (0.71–4.74) | 2.05 (0.79–5.35) | ||
| Birth size | ||||
| ref | ref | ||
| 0.89 (0.58–1.37) | 0.74 (0.47–1.16) | ||
| Reason for visit | ||||
| ref | ref | ||
| 0.55 (0.61–2.11) | 0.69 (0.27–1.76) | ||
| Time of visit | ||||
| Ref | Ref | ||
| 1.13 (0.61–2.11) | 1.10 (0.58–2.07) | ||
| CAREGIVER-LEVEL FACTORS | ||||
| Maternal age | ||||
| ref | ref | ||
| 0.73 (0.45–1.16) | 0.69 (0.43–1.11) | ||
| Maternal education | ||||
| ref | ref | ||
| 3.03 (1.00–9.2) | 3.53 (1.13–11.03) | ||
| Marital status | ||||
| ref | ref | ||
| 0.92 (0.45–1.86) | 1.03 (0.50–2.14) | ||
| Maternal antenatal care | ||||
| ||||
| 0.51 (0.95–2.70) | 0.57 (0.11–3.01) | ||
| Maternal employment | ||||
| ref | ref | ||
| 1.29 (0.65–2.57) | 1.33 (0.66–2.68) | ||
| Means of transport to facility | ||||
| ref | ref | ||
| 1.49 (0.89–2.50) | 1.51 (0.88–2.60) | ||
| Child immunisation message in the last 3 months | ||||
| ref | ref | ||
| 0.45 (0.25–0.84) | 0.46 (0.25–0.87) | ||
| Child immunisation decision making | ||||
| ref | ref | ||
| 0.28 (0.10–0.80) | 0.21 (0.07–0.62) | ||
| FACILITY-LEVEL FACTORS | ||||
| Facility type | ||||
| ref | ref | ||
| 0.71 (0.31–1.62) | 0.72 (0.27–1.94) | ||
| Facility ownership | ||||
| ref | ref | ||
| 1.95 (1.18–3.23) | 1.71 (0.90–3.24) | ||
| Vaccine cold-chain disruption in the last 3 months | ||||
| ref | ref | ||
| 0.57 (0.12–2.72) | 0.18 (0.02–1.71) | ||
| Vaccine stock-out in the last 3 months | ||||
| ref | ref | ||
| 1.35 (0.42–4.30) | 1.73 (0.42–7.16) | ||
| Number of health workers | ||||
| ref | ref | ||
| 0.33 (0.02–0.13) | 0.18 (0.06–0.61) | ||
| Random-effect estimates | ||||
| 0.49 (0.20–1.15) | 0.49 (0.18–1.34) | 0.15 (0.01–11.42) | 0.54 (0.01–4.27) |
| 12.85 | 12.87 | 4.46 | 14.27 |
| 1.94 | 1.94 | 1.45 | 2.03 |
| Model fit statistics | ||||
| 445.87 | 365.26 | 447.38 | 366.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nnaji, C.A.; Wiysonge, C.S.; Adamu, A.A.; Lesosky, M.; Mahomed, H.; Ndwandwe, D. Missed Opportunities for Vaccination and Associated Factors among Children Attending Primary Health Care Facilities in Cape Town, South Africa: A Pre-Intervention Multilevel Analysis. Vaccines 2022, 10, 785. https://doi.org/10.3390/vaccines10050785
Nnaji CA, Wiysonge CS, Adamu AA, Lesosky M, Mahomed H, Ndwandwe D. Missed Opportunities for Vaccination and Associated Factors among Children Attending Primary Health Care Facilities in Cape Town, South Africa: A Pre-Intervention Multilevel Analysis. Vaccines. 2022; 10(5):785. https://doi.org/10.3390/vaccines10050785
Chicago/Turabian StyleNnaji, Chukwudi A., Charles S. Wiysonge, Abdu A. Adamu, Maia Lesosky, Hassan Mahomed, and Duduzile Ndwandwe. 2022. "Missed Opportunities for Vaccination and Associated Factors among Children Attending Primary Health Care Facilities in Cape Town, South Africa: A Pre-Intervention Multilevel Analysis" Vaccines 10, no. 5: 785. https://doi.org/10.3390/vaccines10050785
APA StyleNnaji, C. A., Wiysonge, C. S., Adamu, A. A., Lesosky, M., Mahomed, H., & Ndwandwe, D. (2022). Missed Opportunities for Vaccination and Associated Factors among Children Attending Primary Health Care Facilities in Cape Town, South Africa: A Pre-Intervention Multilevel Analysis. Vaccines, 10(5), 785. https://doi.org/10.3390/vaccines10050785







