EGFR-Based Targeted Therapy for Colorectal Cancer—Promises and Challenges
Abstract
:1. Introduction
2. Receptors Used for Targeted Therapy
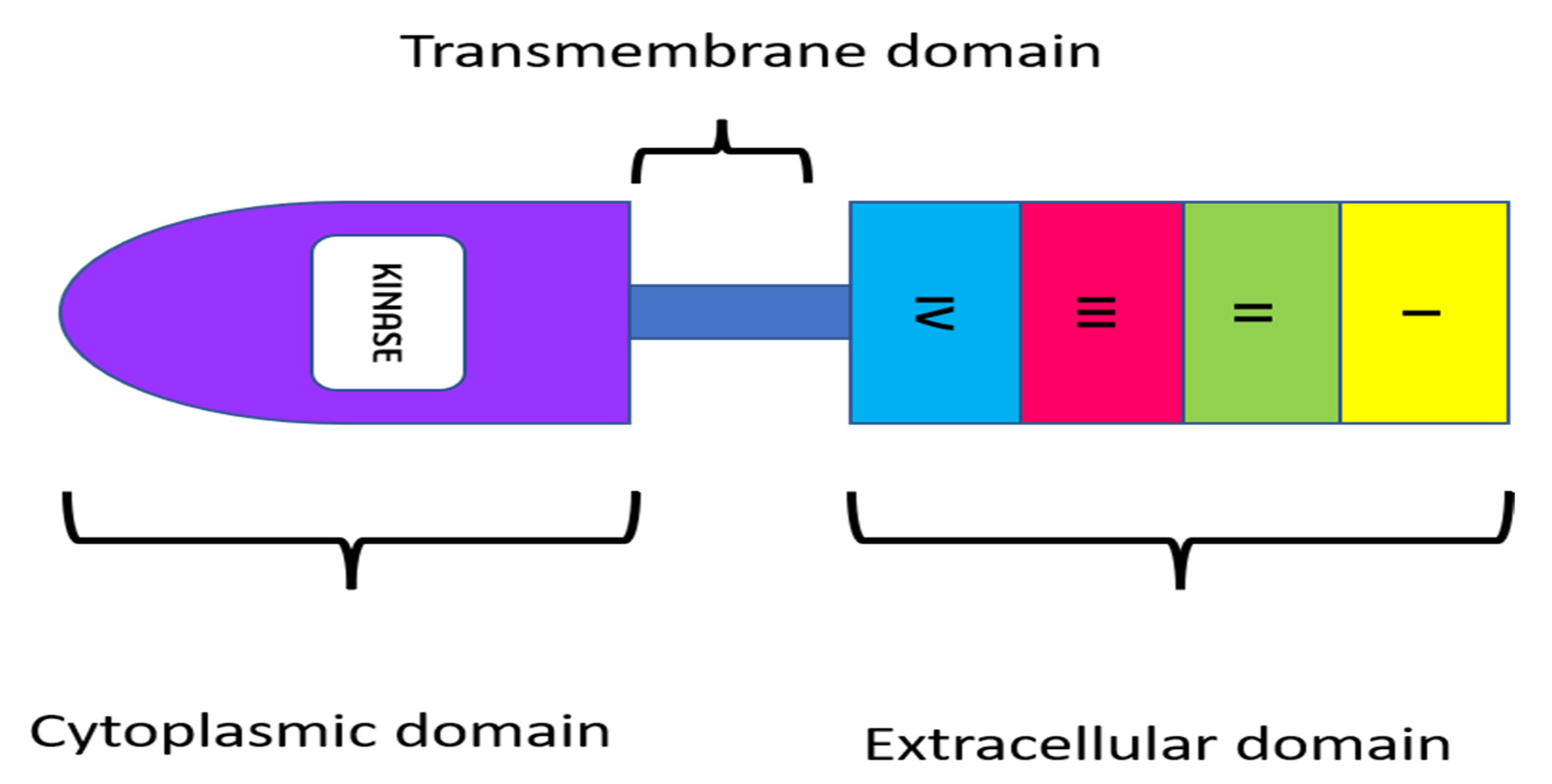
- EGFR: The epidermal growth factor receptor (EGFR) is a receptor tyrosine kinase belonging to the ErbB family of proteins. Ligand binding is required to activate the tyrosine kinase domain. This activates signaling pathways responsible for cell proliferation, angiogenesis migration, continued existence, and adhesion. Since these pathways are essential for the survival of cancer cells, EGFR is a valuable target in the treatment of colorectal carcinoma metastases [8].
- VEGFR: The vascular endothelial growth factor receptor (VEGFR) is a tyrosine kinase receptor. Binding of the ligand vascular endothelial growth factor (VEGF) to this receptor leads to the activation of the receptor and promotes vasculogenesis and angiogenesis [9]. VEGF overexpression is observed in 40–60% of colorectal cancers and is related to cancer recurrence and decreased survival [10].
- FGFR: In several essential physiological mechanisms such as homeostasis of tissue metabolism, embryonic development, endocrine function, and wound repair, angiogenesis fibroblast growth factor (FGFR) signaling pathways are crucially significant [11]. Therapy against FGFR2 and its specific isoforms are being considered as novel treatment options for colorectal cancer patients. By administering shRNA to bind FGFR2, CRC development, invasion, and migration can be reduced [12].
- HER 2: The type I transmembrane glycoprotein human epidermal growth factor receptor 2 (HER2) is involved in signaling pathways that control cell proliferation, survival, and apoptosis in breast cancer. In 20–25% of breast cancer patients, the HER2 gene is amplified, which is connected to an aggressive phenotype and worse prognosis [13]. The efficacy of HER2-targeted therapy is comparable to that of developing therapeutic options for metastatic colorectal cancer, such as immunotherapy with checkpoint inhibitors and BRAF-directed therapy [14].
- TGF-β: The signaling pathway that is activated by transforming growth factor-beta (TGF-β) is crucial in the regulation of tissue development, proliferation, differentiation, apoptosis, and homeostasis [15]. TGF-β is also a powerful regulator of cell adhesion, motility, and the composition of the extracellular matrix, all of which are implicated in tumor invasion and metastasis. However, TGF-β signaling also stimulates angiogenesis and immunosuppression. TGF-β signaling breakdown in colorectal cancer cells promotes tumor growth in the early stages, whereas the stimulation thereof may enhance cancer invasion and metastasis. Thus, while TGF-β may be used as a target in nanotherapeutic methods, its dual roles in enhancing and suppressing tumorigenesis require it to be treated in a careful and highly selective manner [16].
3. Significance of EGFR as a Target
4. Monoclonal Antibodies for EGFR Targeted Therapy
Cetuximab
- Cetuximab binds to the second (L2) EGFR domain and consequently blocks downstream signaling by triggering receptor internalization and blocks the interaction between ligand and receptor [36].
- Through antibody-dependent cell-mediated cytotoxicity (ADCC), cetuximab directs cytotoxic immune effector cells toward EGFR-expressing tumor cells, potentially contributing to its antitumoral impact [37].
- Cetuximab causes a G1 cell cycle arrest by increasing the cell cycle inhibitor p27kip1 and suppressing proliferating cell nuclear antigen (PCNA) [38].
- Cetuximab inhibits angiogenesis by restricting the production of pro-angiogenic factors such as interleukin-8, vascular endothelial growth factor (VEGF), and basic fibroblast growth factor (FGF) [39].
- Induction of apoptosis by cetuximab is mediated through two processes: (a) Increased expression of pro-apoptotic proteins such as BAX, caspase-3, caspase-8, and caspase-9 and (b) inactivation of Bcl-2, which is an anti-apoptotic protein [39].
5. EGFR Targeted Therapy Using Cetuximab
5.1. Gold Nanoparticles
5.2. Iron Oxide Nanoparticles
5.3. Polymeric Nanoparticles
5.4. Protein Nanoparticles
5.5. Liposomes
5.6. Micelles
5.7. Carbon Nanotubes
5.8. Quantum Dots
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xie, Y.-H.; Chen, Y.-X.; Fang, J.-Y. Comprehensive review of targeted therapy for colorectal cancer. Signal Transduct. Target. Ther. 2020, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Gulbake, A.; Jain, A.; Jain, A.; Jain, A.; Jain, S.K. Insight to drug delivery aspects for colorectal cancer. World J. Gastroenterol. 2016, 22, 582–599. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Ritter, G.; Nishikawa, H. Antibody-based therapy in colorectal cancer. Immunotherapy 2013, 5, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Fay, F.; Scott, C.J. Antibody-targeted nanoparticles for cancer therapy. Immunotherapy 2011, 3, 381–394. [Google Scholar] [CrossRef]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active Targeting Strategies Using Biological Ligands for Nanoparticle Drug Delivery Systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Zhou, Y.; Liu, L.; Xu, Y.; Chen, Q.; Wang, Y.; Wu, S.; Deng, Y.; Zhang, J.; Shao, A. Nanoparticle-Based Drug Delivery in Cancer Therapy and Its Role in Overcoming Drug Resistance. Front. Mol. Biosci. 2020, 7, 193. [Google Scholar] [CrossRef]
- Iqbal, J.; Anwar, F.; Afridi, S. Targeted Drug Delivery Systems and Their Therapeutic Applications in Cancer and Immune Pathological Conditions. Infect. Disord. Drug Targets. 2017, 17, 149–159. [Google Scholar] [CrossRef]
- Piawah, S.; Venook, A.P. Targeted therapy for colorectal cancer metastases: A review of current methods of molecularly targeted therapy and the use of tumor biomarkers in the treatment of metastatic colorectal cancer. Cancers 2019, 125, 4139–4147. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Banerjee, S.; Flores-Rozas, H. Monoclonal antibodies for targeted therapy in colorectal cancer. Cancer Biol. Ther. 2010, 9, 563–571. [Google Scholar] [CrossRef] [Green Version]
- Luo, H.; Zhang, T.; Cheng, P.; Li, D.; Ogorodniitchouk, O.; Lahmamssi, C.; Wang, G.; Lan, M. Therapeutic implications of fibroblast growth factor receptor inhibitors in a combination regimen for solid tumors. Oncol. Lett. 2020, 20, 2525–2536. [Google Scholar] [CrossRef]
- Dariya, B.; Merchant, N.; Aliya, S.; Alam, A.; Nagaraju, G.P. EGFR and FGFR in Growth and Metastasis of Colorectal Cancer; Springer: Singapore, 2018. [Google Scholar]
- Chen, F.; Ma, K.; Madajewski, B.; Zhuang, L.; Zhang, L.; Rickert, K.; Marelli, M.; Yoo, B.; Turker, M.Z.; Overholtzer, M.; et al. Ultrasmall targeted nanoparticles with engineered antibody fragments for imaging detection of HER2-overexpressing breast cancer. Nat. Commun. 2018, 9, 4141. [Google Scholar] [CrossRef] [Green Version]
- Siena, S.; Sartore-Bianchi, A.; Marsoni, S.; Hurwitz, H.I.; McCall, S.J.; Penault-Llorca, F.; Srock, S.; Bardelli, A.; Trusolino, L. Targeting the human epidermal growth factor receptor 2 (HER2) oncogene in colorectal cancer. Ann. Oncol. 2018, 29, 1108–1119. [Google Scholar] [CrossRef]
- Itatani, Y.; Kawada, K.; Sakai, Y. Transforming Growth Factor-β Signaling Pathway in Colorectal Cancer and Its Tumor Microenvironment. Int. J. Mol. Sci. 2019, 20, 5822. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Pasche, B. TGF-β signaling alterations and susceptibility to colorectal cancer. Hum. Mol. Genet. 2007, 16, R14–R20. [Google Scholar] [CrossRef] [Green Version]
- Milane, L.; Duan, Z.; Amiji, M. Development of EGFR-targeted polymer blend nanocarriers for combination paclitaxel/lonidamine delivery to treat multi-drug resistance in human breast and ovarian tumor cells. Mol. Pharm. 2011, 8, 185–203. [Google Scholar] [CrossRef] [Green Version]
- Vale, C.L.; Tierney, J.F.; Fisher, D.; Adams, R.A.; Kaplan, R.; Maughan, T.S.; Parmar, M.K.; Meade, A.M. Does anti-EGFR therapy improve outcome in advanced colorectal cancer? A systematic review and meta-analysis. Cancer Treat Rev. 2012, 38, 618–625. [Google Scholar] [CrossRef]
- Koyen Malashevich, A.; Nabeta, G.; Bienfait, S.; Schlafstein, A.; Yu, D.; Daddacha, W. EGFR Role in Cancer: A Potential Therapeutic Target. In Role of Tyrosine Kinases in Gastrointestinal Malignancies; Springer: Singapore, 2018; pp. 225–234. [Google Scholar]
- Krasinskas, A.M. EGFR Signaling in Colorectal Carcinoma. Patholog. Res. Int. 2011, 2011, 932932. [Google Scholar] [CrossRef]
- Chang, S.S.; Califano, J. Current status of biomarkers in head and neck cancer. J. Surg. Oncol. 2008, 97, 640–643. [Google Scholar] [CrossRef]
- Spano, J.P.; Fagard, R.; Soria, J.C.; Rixe, O.; Khayat, D.; Milano, G. Epidermal growth factor receptor signaling in colorectal cancer: Preclinical data and therapeutic perspectives. Ann. Oncol. 2005, 16, 189–194. [Google Scholar] [CrossRef]
- Pabla, B.; Bissonnette, M.; Konda, V.J. Colon cancer and the epidermal growth factor receptor: Current treatment paradigms, the importance of diet, and the role of chemoprevention. World J. Clin. Oncol. 2015, 6, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.H.; Wang, Y.Z.; Tu, J.; Liu, C.W.; Yuan, Y.J.; Lin, R.; He, W.L.; Cai, S.R.; He, Y.L.; Ye, J.N. Anti-EGFR therapy in metastatic colorectal cancer: Mechanisms and potential regimens of drug resistance. Gastroenterol. Rep. 2020, 8, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Dawson, J.P.; Berger, M.B.; Lin, C.C.; Schlessinger, J.; Lemmon, M.A.; Ferguson, K.M. Epidermal growth factor receptor dimerization and activation require ligand-induced conformational changes in the dimer interface. Mol. Cell Biol. 2005, 25, 7734–7742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wee, P.; Wang, Z. Epidermal Growth Factor Receptor Cell Proliferation Signaling Pathways. Cancers 2017, 9, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, R.; Weihua, Z. Rethink of EGFR in Cancer With Its Kinase Independent Function on Board. Front. Oncol. 2019, 9, 800. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, E.J.; Grady, W.M.; Lieberman, D.; Seufferlein, T.; Sung, J.J.; Boelens, P.G.; van de Velde, C.J.H.; Watanabe, T. Colorectal cancer. Nat. Rev. Dis. Primers. 2015, 1, 15065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seshacharyulu, P.; Ponnusamy, M.P.; Haridas, D.; Jain, M.; Ganti, A.K.; Batra, S.K. Targeting the EGFR signaling pathway in cancer therapy. Expert Opin. Ther. Targets 2012, 16, 15–31. [Google Scholar] [CrossRef] [Green Version]
- El Guerrab, A.; Bamdad, M.; Kwiatkowski, F.; Bignon, Y.-J.; Penault-Llorca, F.; Aubel, C. Anti-EGFR monoclonal antibodies and EGFR tyrosine kinase inhibitors as combination therapy for triple-negative breast cancer. Oncotarget 2016, 7, 73618–73637. [Google Scholar] [CrossRef] [Green Version]
- Burgess, A.W.; Cho, H.S.; Eigenbrot, C.; Ferguson, K.M.; Garrett, T.P.; Leahy, D.J.; Lemmon, M.A.; Sliwkowski, M.X.; Ward, C.W.; Yokoyama, S. An open-and-shut case? Recent insights into the activation of EGF/ErbB receptors. Mol. Cell 2003, 12, 541–552. [Google Scholar] [CrossRef]
- Frattini, M.; Saletti, P.; Molinari, F.; De Dosso, S. EGFR signaling in colorectal cancer: A clinical perspective. Gastrointest. Cancer Targets Ther. 2015, 2015, 21. [Google Scholar] [CrossRef] [Green Version]
- You, B.; Chen, E.X. Anti-EGFR monoclonal antibodies for treatment of colorectal cancers: Development of cetuximab and panitumumab. J. Clin. Pharmacol. 2012, 52, 128–155. [Google Scholar] [CrossRef]
- Bou-Assaly, W.; Mukherji, S. Cetuximab (Erbitux). Am. J. Neuroradiol. 2010, 31, 626–627. [Google Scholar] [CrossRef] [Green Version]
- Ebisumoto, K.; Okami, K.; Hamada, M.; Maki, D.; Sakai, A.; Saito, K.; Shimizu, F.; Kaneda, S.; Iida, M. Cetuximab with radiotherapy as an alternative treatment for advanced squamous cell carcinoma of the temporal bone. Auris Nasus Larynx 2018, 45, 637–639. [Google Scholar] [CrossRef]
- Van Krieken, J.H.; Jung, A.; Kirchner, T.; Carneiro, F.; Seruca, R.; Bosman, F.T.; Quirke, P.; Fléjou, J.F.; Plato Hansen, T.; de Hertogh, G.; et al. KRAS mutation testing for predicting response to anti-EGFR therapy for colorectal carcinoma: Proposal for an European quality assurance program. Virchows Arch. 2008, 453, 417–431. [Google Scholar] [CrossRef] [Green Version]
- Hanck-Silva, G.; Fatori Trevizan, L.N.; Petrilli, R.; de Lima, F.T.; Eloy, J.O.; Chorilli, M. A Critical Review of Properties and Analytical/Bioanalytical Methods for Characterization of Cetuximab. Crit. Rev. Anal. Chem. 2020, 50, 125–135. [Google Scholar] [CrossRef]
- Okuyama, K.; Suzuki, K.; Naruse, T.; Tsuchihashi, H.; Yanamoto, S.; Kaida, A.; Miura, M.; Umeda, M.; Yamashita, S. Prolonged cetuximab treatment promotes p27(Kip1)-mediated G1 arrest and autophagy in head and neck squamous cell carcinoma. Sci. Rep. 2021, 11, 5259. [Google Scholar] [CrossRef]
- Martinelli, E.; De Palma, R.; Orditura, M.; De Vita, F.; Ciardiello, F. Anti-epidermal growth factor receptor monoclonal antibodies in cancer therapy. Clin. Exp. Immunol. 2009, 158, 1–9. [Google Scholar] [CrossRef]
- Tseng, S.H.; Chou, M.Y.; Chu, I.M. Cetuximab-conjugated iron oxide nanoparticles for cancer imaging and therapy. Int. J. Nanomed. 2015, 10, 3663–3685. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.; Liang, X. Progress in research on gold nanoparticles in cancer management. Medicine 2019, 98, e15311. [Google Scholar] [CrossRef]
- Sun, M.; Wang, T.; Li, L.; Li, X.; Zhai, Y.; Zhang, J.; Li, W. The Application of Inorganic Nanoparticles in Molecular Targeted Cancer Therapy: EGFR Targeting. Front. Pharmacol. 2021, 12, 702445. [Google Scholar] [CrossRef]
- Leve, F.; Bonfim, D.P.; Fontes, G.; Morgado-Díaz, J.A. Gold nanoparticles regulate tight junctions and improve cetuximab effect in colon cancer cells. Nanomed. 2019, 14, 1565–1578. [Google Scholar] [CrossRef] [PubMed]
- El Hallal, R.; Lyu, N.; Wang, Y. Effect of Cetuximab-Conjugated Gold Nanoparticles on the Cytotoxicity and Phenotypic Evolution of Colorectal Cancer Cells. Molecules 2021, 26, 567. [Google Scholar] [CrossRef]
- Cho, Y.S.; Yoon, T.J.; Jang, E.S.; Soo Hong, K.; Young Lee, S.; Ran Kim, O.; Park, C.; Kim, Y.J.; Yi, G.C.; Chang, K. Cetuximab-conjugated magneto-fluorescent silica nanoparticles for in vivo colon cancer targeting and imaging. Cancer Lett. 2010, 299, 63–71. [Google Scholar] [CrossRef]
- Othman, M.H.; Zayed, G.M.; Ali, U.F.; Abdellatif, A.A.H. Colon-specific tablets containing 5-fluorouracil microsponges for colon cancer targeting. Drug Dev. Ind. Pharm. 2020, 46, 2081–2088. [Google Scholar] [CrossRef] [PubMed]
- Sabra, R.; Billa, N.; Roberts, C.J. Cetuximab-conjugated chitosan-pectinate (modified) composite nanoparticles for targeting colon cancer. Int. J. Pharm. 2019, 572, 118775. [Google Scholar] [CrossRef]
- Maya, S.; Sarmento, B.; Lakshmanan, V.-K.; Menon, D.; Jayakumar, R. Actively Targeted Cetuximab Conjugated γ-Poly(glutamic acid)-Docetaxel Nanomedicines for Epidermal Growth Factor Receptor Over Expressing Colon Cancer Cells. J. Biomed. Nanotechnol. 2014, 10, 1416–1428. [Google Scholar] [CrossRef]
- Duwa, R.; Banstola, A.; Emami, F.; Jeong, J.-H.; Lee, S.; Yook, S. Cetuximab conjugated temozolomide-loaded poly (lactic-co-glycolic acid) nanoparticles for targeted nanomedicine in EGFR overexpressing cancer cells. J. Drug Deliv. Sci. Technol. 2020, 60, 101928. [Google Scholar] [CrossRef]
- Ye, Z.; Zhang, Y.; Liu, Y.; Liu, Y.; Tu, J.; Shen, Y. EGFR Targeted Cetuximab-Valine-Citrulline (vc)-Doxorubicin Immunoconjugates-Loaded Bovine Serum Albumin (BSA) Nanoparticles for Colorectal Tumor Therapy. Int. J. Nanomed. 2021, 16, 2443–2459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, X.; Cheng, L.; Yuan, J.; Zhong, Z. SP94 peptide mediating highly specific and efficacious delivery of polymersomal doxorubicin hydrochloride to hepatocellular carcinoma in vivo. Colloids Surf. B: Biointerfaces. 2021, 197, 111399. [Google Scholar] [CrossRef]
- Zalba, S.; Contreras, A.M.; Haeri, A.; Ten Hagen, T.L.; Navarro, I.; Koning, G.; Garrido, M.J. Cetuximab-oxaliplatin-liposomes for epidermal growth factor receptor targeted chemotherapy of colorectal cancer. J. Control Release. 2015, 210, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Du, Y.; Liang, X.; Sun, T.; Xue, H.; Tian, J.; Jin, Z. EGFR-targeted liposomal nanohybrid cerasomes: Theranostic function and immune checkpoint inhibition in a mouse model of colorectal cancer. Nanoscale 2018, 10, 16738–16749. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.H.; Luo, T.Y.; Chiang, P.F.; Yao, C.J.; Lin, W.J.; Peng, C.L.; Shieh, M.J. EGFR-targeted micelles containing near-infrared dye for enhanced photothermal therapy in colorectal cancer. J. Control Release 2017, 258, 196–207. [Google Scholar] [CrossRef]
- Lee, P.C.; Chiou, Y.C.; Wong, J.M.; Peng, C.L.; Shieh, M.J. Targeting colorectal cancer cells with single-walled carbon nanotubes conjugated to anticancer agent SN-38 and EGFR antibody. Biomaterials 2013, 34, 8756–8765. [Google Scholar] [CrossRef]
- Hashemkhani, M.; Demirci, G.; Bayir, A.; Muti, A.; Sennaroglu, A.; Hadi, L.; Yaghini, E.; Loizidou, M.; MacRobert, A.; Acar, H. Cetuximab-Ag2S Quantum Dots for Targeted Imaging and Combination ALA-based Photodynamic/Chemotherapy of Colorectal Cancer. Nanoscale 2021. [Google Scholar] [CrossRef]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [Green Version]
- Masood, F. Polymeric nanoparticles for targeted drug delivery system for cancer therapy. Mater. Sci. Eng. 2016, 60, 569–578. [Google Scholar] [CrossRef]
- Abdellatif, A.A.H.; Ibrahim, M.A.; Amin, M.A.; Maswadeh, H.; Alwehaibi, M.N.; Al-Harbi, S.N.; Alharbi, Z.A.; Mohammed, H.A.; Mehany, A.B.M.; Saleem, I. Cetuximab Conjugated with Octreotide and Entrapped Calcium Alginate-beads for Targeting Somatostatin Receptors. Sci. Rep. 2020, 10, 4736. [Google Scholar] [CrossRef]
- Jain, A.; Singh, S.K.; Arya, S.K.; Kundu, S.C.; Kapoor, S. Protein Nanoparticles: Promising Platforms for Drug Delivery Applications. ACS Biomater. Sci. Eng. 2018, 4, 3939–3961. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Hu, Y.; Pan, L. Cetuximab-modified doxorubicin bovine serum albumin nanoparticles for the treatment of epidermal growth factor receptor-overexpressing colon cancer cells. Mater. Express. 2021, 11, 1619–1627. [Google Scholar] [CrossRef]
- Mochida, Y.; Cabral, H.; Kataoka, K. Polymeric micelles for targeted tumor therapy of platinum anticancer drugs. Expert. Opin. Drug Deliv. 2017, 14, 1423–1438. [Google Scholar] [CrossRef] [PubMed]
- Debnath, S.K.; Srivastava, R. Drug Delivery With Carbon-Based Nanomaterials as Versatile Nanocarriers: Progress and Prospects. Front. Nanotechnol. 2021, 3, 15. [Google Scholar] [CrossRef]
- Kumar, S.; Rani, R.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.-H. Carbon nanotubes: A novel material for multifaceted applications in human healthcare. Chem. Soc. Rev. 2017, 46, 158–196. [Google Scholar] [CrossRef]
- Gil, H.M.; Price, T.W.; Chelani, K.; Bouillard, J.-S.G.; Calaminus, S.D.J.; Stasiuk, G.J. NIR-quantum dots in biomedical imaging and their future. iScience 2021, 24, 102189. [Google Scholar] [CrossRef]


| Name of the Antibody | Antibody Type | Disease | Molecular Weight | Mechanism of Action | Market Status | Side Effects |
|---|---|---|---|---|---|---|
| Cetuximab (Erbitux) | Chimeric antibodyIgG1 | Head and neck cancer Metastatic colorectal cancer | 145.7816 kDa | (1) Binds to domain III of EGFR receptor and prevents the conformational change required for EGFR activation (2) Induce apoptosis and decrease matrix metalloproteinase and VGEF | Marketed | rash, itching, dry or cracked skin, nail changes, headache, diarrhea, nausea, vomiting, upset stomach, weight loss, weakness, and respiratory, skin, and mouth infections. |
| Panitumumab (Vectibix) | Humanan tibody IgG2 | Metastatic colorectal cancer | 147 kDa | (1) Binds to domain III of EGFR receptor and prevents the conformational change required for EGFR activation | Marketed | Skin reactions, Fatigue, General deterioration, Abdominal pain, Nausea, Diarrhea, Vomiting, Swelling in hands or feet, Cough, Dry skin, Inflammation of the bed of the fingernails, Eye irritation |
| Carriers | Formulation | Method of Antibody conjugation/coating | Drug | Target cells/Animal models | Disease/Application | Significance |
|---|---|---|---|---|---|---|
| Gold nanoparticles | Tetrachloroauric acid, sodium citrate, and cetuximab | Physical adsorption | - | Caco-2, HT-29 and HCT-116 | Colorectal cancer | Tight junction modulation by Gold nanoparticles aid in drug delivery. Improved cell death mediated by cetuximab [43]. |
| Gold nanoparticles | Tetrachloroauric acid, sodium citrate, s 5,50 -dithiobis-(2-nitrobenzoic acid), 7-mercapto4-methyl coumarin, 2,3,5,6-tetrafluoro-4-mercaptobenzoic acid, and cetuximab | Physical adsorption | - | HT-29 | Colorectal cancer | Expression of cell surface biomarkers such as MCAM, HER3, and EpCAM with more heterogenicity [44]. |
| Magneto fluorescent silica nanoparticles | Polyvinylpyrrolidone, ferrite, 2- [methoxy- (polyethyleneoxy)propyl] trimethoxysilane, (3-trimethoxysilil) propyl diethylene triamine, and cetuximab antibody fragment | Physical adsorption | - | HCT 116, H520 cells, HT 29, SW620, and BALB/c nude male mice | Colorectal cancer | Considerable MRI signal changes Local concentration of MFSN-Ctx amplified by an external magnetic field [45]. |
| Ca-alginate-beads | sodium alginate, calcium chloride, Octreotide, and Cetuximab | solvent evaporation method | Octreotide | MCF-7, HepG-2, and HCT-116 | Breast cancer, Hepatocellular carcinoma, and colorectal cancer | Target a specific area in Gastro-Intestinal Tract Specifically penetrates somatostatin expressing cells and releases octreotide [46]. |
| Chitosan-Pectin nanoparticles | Chitosan, pectin, curcumin and cetuximab | EDC–NHS chemistry | Curcumin | Caco-2, and HCT-116. | Colorectal cancer | EGFR overexpressing cancer cell line had stronger anti-cancer activity Cetuximab enhanced the nanoparticle uptake [47]. |
| Γ Poly (glutamic Acid) nanoparticles | Γ Poly (glutamic Acid), chitosan, docetaxel, rhodamine-123, and cetuximab. | EDC–NHS chemistry | Docetaxel | HT-29, IEC-6, and Swiss Albino mice | Colorectal cancer | A two-fold increase in nanoparticle uptake in EGFR+ve cells increased cytotoxicity antiproliferative activity due to cell cycle arrest at G2/M phase [48]. |
| PLGA nanoparticles | Poly (lactic-co-glycolic acid), Temozolomide, and cetuximab. | EDC–NHS chemistry | Temozolomide | U-87MG, SK-Mel 28, and SW480 | Brain cancer, Skin cancer, and Colorectal cancer | Improved cellular uptake Increased cytotoxicity Increased apoptotic impact Upregulation of γ-H2A [49]. |
| BSA nanoparticles | MC-Val-Cit-PAB-PNP, Doxorubicin, and cetuximab | Direct coupling | Doxorubicin | RKO, d LS174, and BALB/c nude mice | Colorectal cancer | Increased the duration of doxorubicin uptake into cells Improved accumulation of the drug in tumors Lowered systemic toxicity [50]. |
| BSA nanoparticles | BSA, doxorubicin, and cetuximab | Direct coupling | Doxorubicin | RKO and LS174 t | Colorectal cancer | Significant cytotoxic activity of doxorubicin in EGFR overexpressing cells with increased selectivity and low toxicity [51]. |
| Liposomes | Oxaliplatin, and Fab of cetuximab | Maleimide chemistry | Oxaliplatin | HCT-116, HT-29, SW-480, SW-620, and nude mice | Colorectal cancer | Effective delivery of intracellular L-OH Resistance to L-OH was reversed when targeted liposomes are used Fab’ ligand targeted liposomes were more efficacious in antitumor activity than cetuximab targeted liposomes [52]. |
| Cerosomes | 1,2-Distearoyl-sn-glycero-3- phosphoethanolamine-n-[poly(ethylene glycol)]-hydroxy succi- nimide, cetuximab, and porphyrin | Direct Coupling | Porphyrin | CT26-fLuc, and Balb/c mice | Colorectal cancer | Preferential accumulation of EGFR-CPIG at tumor locations The combination of EPR effect and the active tumor targeting capability of EGFR increase the tumour targeting capacity [53]. |
| Micelles | ε-caprolactone, Methoxy poly ethylene glycol, Poly ethylene glycol monoethyl ether maleimide, (DTPA dianhydride, and cetuximab | Chemical modification (Thiolation) | IR-780 iodide (Near infrared dye) | HCT-116, SW-620, and nude mice | Colorectal cancer | Highest contrast of NIRF signals was obtained between HCT-116 and SW-620 tumors. Increased delivery of IR-780 and enhanced photo thermal therapy was observed in HCT-116 tumors (EGFR +ve ) [54]. |
| Carbon nanotubes | Carbon nanotubes, SWNT-COOH, NH2-PEGNH2, and cetuximab | EDC–NHS chemistry | SN38 (Topoisomerase I inhibitor), and Pyrene 38 | HCT116, HT29, and SW-620 | Colorectal cancer | The targeting ability and delivery of SN38 by Cetuximab conjugated SWNT25/py38 was enhanced in EGFR +ve colorectal cancer cell line. Clatherin-dependent endocytosis was responsible for uptake of SWNT25/py38 [55]. |
| Quantum Dots | Silver nitrate,5-aminolevulinic acid hydrochloride, 2-Mercaptopropionic acid, Sodium sulfide, And cetuximab | EDC–NHS chemistry | ALA | HCT116, HT29, and SW-480 | Colorectal cancer | The targeting ability of quantum dots was observed to increase with increased EGFR expression and there is a strong intracellular NIR signals [56]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janani, B.; Vijayakumar, M.; Priya, K.; Kim, J.H.; Prabakaran, D.S.; Shahid, M.; Al-Ghamdi, S.; Alsaidan, M.; Othman Bahakim, N.; Hassan Abdelzaher, M.; et al. EGFR-Based Targeted Therapy for Colorectal Cancer—Promises and Challenges. Vaccines 2022, 10, 499. https://doi.org/10.3390/vaccines10040499
Janani B, Vijayakumar M, Priya K, Kim JH, Prabakaran DS, Shahid M, Al-Ghamdi S, Alsaidan M, Othman Bahakim N, Hassan Abdelzaher M, et al. EGFR-Based Targeted Therapy for Colorectal Cancer—Promises and Challenges. Vaccines. 2022; 10(4):499. https://doi.org/10.3390/vaccines10040499
Chicago/Turabian StyleJanani, Balakarthikeyan, Mayakrishnan Vijayakumar, Kannappan Priya, Jin Hee Kim, D. S. Prabakaran, Mohammad Shahid, Sameer Al-Ghamdi, Mohammed Alsaidan, Nasraddin Othman Bahakim, Mohammad Hassan Abdelzaher, and et al. 2022. "EGFR-Based Targeted Therapy for Colorectal Cancer—Promises and Challenges" Vaccines 10, no. 4: 499. https://doi.org/10.3390/vaccines10040499
APA StyleJanani, B., Vijayakumar, M., Priya, K., Kim, J. H., Prabakaran, D. S., Shahid, M., Al-Ghamdi, S., Alsaidan, M., Othman Bahakim, N., Hassan Abdelzaher, M., & Ramesh, T. (2022). EGFR-Based Targeted Therapy for Colorectal Cancer—Promises and Challenges. Vaccines, 10(4), 499. https://doi.org/10.3390/vaccines10040499








