Durability of Humoral Responses after the Second Dose of mRNA BNT162b2 Vaccine in Residents of a Long Term Care Facility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. SARS-CoV-2 Antibody Assays
2.3. Statistical Analysis
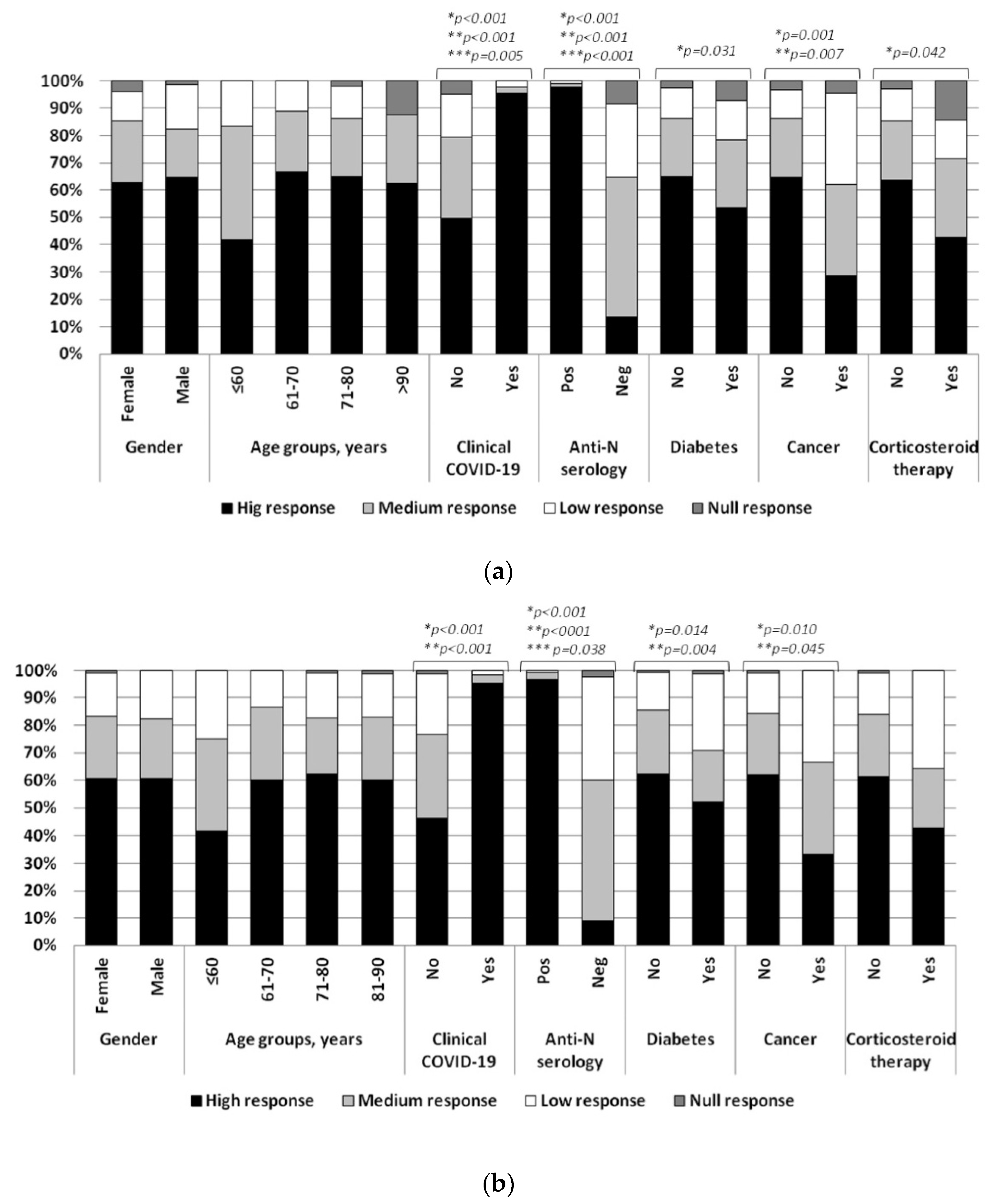
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Girolamo, G.; Bellelli, G.; Bianchetti, A.; Starace, F.; Zanetti, O.; Zarbo, C.; Micciolo, R. Older People Living in Long-Term Care Facilities and Mortality Rates During the COVID-19 Pandemic in Italy: Preliminary Epidemiological Data and Lessons to Learn. Front. Psychiatry 2020, 11, 586524. [Google Scholar] [CrossRef]
- Dorrucci, M.; Minelli, G.; Boros, S.; Manno, V.; Prati, S.; Battaglini, M.; Corsetti, G.; Andrianou, X.; Riccardo, F.; Fabiani, M.; et al. Excess Mortality in Italy During the COVID-19 Pandemic: Assessing the Differences Between the First and the Second Wave, Year 2020. Front. Public Health 2021, 9, 669209. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.M.; Guo, G.; Gibson, D.S.; Bjourson, A.J.; Rai, T.S. Role of Senescence and Aging in SARS-CoV-2 Infection and COVID-19 Disease. Cells 2021, 10, 3367. [Google Scholar] [CrossRef] [PubMed]
- Romero Starke, K.; Reissig, D.; Petereit-Haack, G.; Schmauder, S.; Nienhaus, A.; Seidler, A. The isolated effect of age on the risk of COVID-19 severe outcomes: A systematic review with meta-analysis. BMJ Glob. Health 2021, 6, e006434. [Google Scholar] [CrossRef] [PubMed]
- Bencivenga, L.; Rengo, G.; Varricchi, G. Elderly at time of COronaVIrus disease 2019 (COVID-19): Possible role of immunosenescence and malnutrition. GeroScience 2020, 42, 1089–1092. [Google Scholar] [CrossRef]
- Hand, J.; Rose, E.B.; Salinas, A.; Lu, X.; Sakthivel, S.K.; Schneider, E.; Watson, J.T. Severe Respiratory Illness Outbreak Associated with Human Coronavirus NL63 in a Long-Term Care Facility. Emerg. Infect. Dis. 2018, 24, 1964–1966. [Google Scholar] [CrossRef]
- Lansbury, L.E.; Brown, C.S.; Nguyen-Van-Tam, J.S. Influenza in long-term care facilities. Influenza Other Respir. Viruses 2017, 11, 356–366. [Google Scholar] [CrossRef]
- McMichael, T.M.; Currie, D.W.; Clark, S.; Pogosjans, S.; Kay, M.; Schwartz, N.G.; Lewis, J.; Baer, A.; Kawakami, V.; Lukoff, M.D.; et al. Epidemiology of Covid-19 in a Long-Term Care Facility in King County, Washington. N. Engl. J. Med. 2020, 382, 2005–2011. [Google Scholar] [CrossRef]
- Patel, M.C.; Chaisson, L.H.; Borgetti, S.; Burdsall, D.; Chugh, R.K.; Hoff, C.R.; Murphy, E.B.; Murskyj, E.A.; Wilson, S.; Ramos, J.; et al. Asymptomatic SARS-CoV-2 Infection and COVID-19 Mortality During an Outbreak Investigation in a Skilled Nursing Facility. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020, 71, 2920–2926. [Google Scholar] [CrossRef]
- Ladhani, S.N.; Jeffery-Smith, A.; Patel, M.; Janarthanan, R.; Fok, J.; Crawley-Boevey, E.; Vusirikala, A.; Fernandez Ruiz De Olano, E.; Perez, M.S.; Tang, S.; et al. High prevalence of SARS-CoV-2 antibodies in care homes affected by COVID-19: Prospective cohort study, England. EClinicalMedicine 2020, 28, 100597. [Google Scholar] [CrossRef]
- Wajnberg, A.; Mansour, M.; Leven, E.; Bouvier, N.M.; Patel, G.; Firpo-Betancourt, A.; Mendu, R.; Jhang, J.; Arinsburg, S.; Gitman, M.; et al. Humoral response and PCR positivity in patients with COVID-19 in the New York City region, USA: An observational study. Lancet Microbe 2020, 1, e283–e289. [Google Scholar] [CrossRef]
- Deeks, J.J.; Dinnes, J.; Takwoingi, Y.; Davenport, C.; Spijker, R.; Taylor-Phillips, S.; Adriano, A.; Beese, S.; Dretzke, J.; Ferrante di Ruffano, L.; et al. Antibody tests for identification of current and past infection with SARS-CoV-2. Cochrane Database Syst. Rev. 2020, 6, CD013652. [Google Scholar] [CrossRef]
- Graham, N.S.N.; Junghans, C.; McLaren, R.; Randell, P.; Lang, N.; Ladhani, S.N.; Sharp, D.J.; Sanderson, F. High rates of SARS-CoV-2 seropositivity in nursing home residents. J. Infect. 2021, 82, 282–327. [Google Scholar] [CrossRef]
- Hall, V.J.; Foulkes, S.; Charlett, A.; Atti, A.; Monk, E.J.M.; Simmons, R.; Wellington, E.; Cole, M.J.; Saei, A.; Oguti, B.; et al. SARS-CoV-2 infection rates of antibody-positive compared with antibody-negative health-care workers in England: A large, multicentre, prospective cohort study (SIREN). Lancet 2021, 397, 1459–1469. [Google Scholar] [CrossRef]
- Nace, D.A.; Kip, K.E.; Mellors, J.W.; Peck Palmer, O.M.; Shurin, M.R.; Mulvey, K.; Crandall, M.; Sobolewski, M.D.; Enick, P.N.; McCormick, K.D.; et al. Antibody Responses After mRNA-Based COVID-19 Vaccination in Residential Older Adults: Implications for Reopening. J. Am. Med. Dir. Assoc. 2021, 22, 1593–1598. [Google Scholar] [CrossRef]
- Caimi, B.; Franzetti, M.; Velleca, R.; Lai, A.; Gatti, A.; Luigi, R.; D’Orso, M.; Pregliasco, F.; Balotta, C.; Calicchio, G. Sero-Survey on Long-Term Care Facility Residents Reveals Increased Risk of Sub-Optimal Antibody Response to BNT162B2: Implications for Breakthrough Prevention. BMC Geriatr. 2022, 22, 191. [Google Scholar] [CrossRef]
- Bergwerk, M.; Gonen, T.; Lustig, Y.; Amit, S.; Lipsitch, M.; Cohen, C.; Mandelboim, M.; Levin, E.G.; Rubin, C.; Indenbaum, V.; et al. Covid-19 Breakthrough Infections in Vaccinated Health Care Workers. N. Engl. J. Med. 2021, 385, 1474–1484. [Google Scholar] [CrossRef]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Boccuto, A.; Dragoni, F.; Bergna, A.; Ventura, C.D.; Giammarino, F.; Saladini, F.; Pezzati, L.; Zehender, G.; Zazzi, M.; Vicenti, I.; et al. Decreased neutralization of the Eta SARS-CoV-2 variant by sera of previously infected and uninfected vaccinated individuals. J. Infect. 2022, 84, 94–118. [Google Scholar] [CrossRef]
- Dolscheid-Pommerich, R.; Bartok, E.; Renn, M.; Kümmerer, B.M.; Schulte, B.; Schmithausen, R.M.; Stoffel-Wagner, B.; Streeck, H.; Saschenbrecker, S.; Steinhagen, K.; et al. Correlation between a quantitative anti-SARS-CoV-2 IgG ELISA and neutralization activity. J. Med. Virol. 2022, 94, 388–392. [Google Scholar] [CrossRef]
- Demonbreun, A.R.; Sancilio, A.; Velez, M.P.; Ryan, D.T.; Saber, R.; Vaught, L.A.; Reiser, N.L.; Hsieh, R.R.; D’Aquila, R.T.; Mustanski, B.; et al. Comparison of IgG and neutralizing antibody responses after one or two doses of COVID-19 mRNA vaccine in previously infected and uninfected individuals. EClinicalMedicine 2021, 38, 101018. [Google Scholar] [CrossRef]
- Đaković Rode, O.; Bodulić, K.; Zember, S.; Cetinić Balent, N.; Novokmet, A.; Čulo, M.; Rašić, Ž.; Mikulić, R.; Markotić, A. Decline of Anti-SARS-CoV-2 IgG Antibody Levels 6 Months after Complete BNT162b2 Vaccination in Healthcare Workers to Levels Observed Following the First Vaccine Dose. Vaccines 2022, 10, 153. [Google Scholar] [CrossRef]
- Levin, E.G.; Lustig, Y.; Cohen, C.; Fluss, R.; Indenbaum, V.; Amit, S.; Doolman, R.; Asraf, K.; Mendelson, E.; Ziv, A.; et al. Waning Immune Humoral Response to BNT162b2 Covid-19 Vaccine over 6 Months. N. Engl. J. Med. 2021, 385, e84. [Google Scholar] [CrossRef]
- Infantino, M.; Pieri, M.; Nuccetelli, M.; Grossi, V.; Lari, B.; Tomassetti, F.; Calugi, G.; Pancani, S.; Benucci, M.; Casprini, P.; et al. The WHO International Standard for COVID-19 serological tests: Towards harmonization of anti-spike assays. Int. Immunopharmacol. 2021, 100, 108095. [Google Scholar] [CrossRef]
- Dimeglio, C.; Migueres, M.; Mansuy, J.M.; Saivin, S.; Miedougé, M.; Chapuy-Regaud, S.; Izopet, J. Antibody titers and breakthrough infections with Omicron SARS-CoV-2. J. Infect. In Press. 2022. [Google Scholar] [CrossRef]
- Dimeglio, C.; Herin, F.; Martin-Blondel, G.; Miedougé, M.; Izopet, J. Antibody titers and protection against a SARS-CoV-2 infection. J. Infect. 2022, 84, 248–288. [Google Scholar] [CrossRef]
- Ciabattini, A.; Nardini, C.; Santoro, F.; Garagnani, P.; Franceschi, C.; Medaglini, D. Vaccination in the elderly: The challenge of immune changes with aging. Semin. Immunol. 2018, 40, 83–94. [Google Scholar] [CrossRef]
- Salvagno, G.L.; Henry, B.M.; Pighi, L.; De Nitto, S.; Gianfilippi, G.; Lippi, G. The pronounced decline of anti-SARS-CoV-2 spike trimeric IgG and RBD IgG in baseline seronegative individuals six months after BNT162b2 vaccination is consistent with the need for vaccine boosters. Clin. Chem. Lab. Med. 2022, 60, e29–e31. [Google Scholar] [CrossRef]
- Naaber, P.; Tserel, L.; Kangro, K.; Sepp, E.; Jürjenson, V.; Adamson, A.; Haljasmägi, L.; Rumm, A.P.; Maruste, R.; Kärner, J.; et al. Dynamics of antibody response to BNT162b2 vaccine after six months: A longitudinal prospective study. Lancet Reg. Health. Eur. 2021, 10, 100208. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifoni, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Vanshylla, K.; Di Cristanziano, V.; Kleipass, F.; Dewald, F.; Schommers, P.; Gieselmann, L.; Gruell, H.; Schlotz, M.; Ercanoglu, M.S.; Stumpf, R.; et al. Kinetics and correlates of the neutralizing antibody response to SARS-CoV-2 infection in humans. Cell Host Microbe 2021, 29, 917–929.e4. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.; Stec, M.; Rewane, A.; Landay, A.; Cloherty, G.; Moy, J. SARS-CoV-2 Antibody Responses in Infection-Naive or Previously Infected Individuals After 1 and 2 Doses of the BNT162b2 Vaccine. JAMA Netw. Open 2021, 4, e2119741. [Google Scholar] [CrossRef] [PubMed]
- Tarkowski, M.; de Jager, W.; Schiuma, M.; Covizzi, A.; Lai, A.; Gabrieli, A.; Corbellino, M.; Bergna, A.; Ventura, C.D.; Galli, M.; et al. Anti-SARS-CoV-2 Immunoglobulin Isotypes, and Neutralization Activity Against Viral Variants, According to BNT162b2-Vaccination and Infection History. Front. Immunol. 2021, 12, 793191. [Google Scholar] [CrossRef] [PubMed]
- Collier, D.A.; Ferreira, I.A.T.M.; Kotagiri, P.; Datir, R.P.; Lim, E.Y.; Touizer, E.; Meng, B.; Abdullahi, A.; Baker, S.; Dougan, G.; et al. Age-related immune response heterogeneity to SARS-CoV-2 vaccine BNT162b2. Nature 2021, 596, 417–422. [Google Scholar] [CrossRef]
- Barron, E.; Bakhai, C.; Kar, P.; Weaver, A.; Bradley, D.; Ismail, H.; Knighton, P.; Holman, N.; Khunti, K.; Sattar, N.; et al. Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: A whole-population study. Lancet Diabetes Endocrinol. 2020, 8, 813–822. [Google Scholar] [CrossRef]
- McGurnaghan, S.J.; Weir, A.; Bishop, J.; Kennedy, S.; Blackbourn, L.A.K.; McAllister, D.A.; Hutchinson, S.; Caparrotta, T.M.; Mellor, J.; Jeyam, A.; et al. Risks of and risk factors for COVID-19 disease in people with diabetes: A cohort study of the total population of Scotland. Lancet Diabetes Endocrinol. 2021, 9, 82–93. [Google Scholar] [CrossRef]
- Yelin, I.; Katz, R.; Herzel, E.; Berman-Zilberstein, T.; Ben-Tov, A.; Kuint, J.; Gazit, S.; Patalon, T.; Chodick, G.; Kishony, R. Associations of the BNT162b2 COVID-19 vaccine effectiveness with patient age and comorbidities. medRxiv 2021. [Google Scholar] [CrossRef]
- Ali, H.; Alterki, A.; Sindhu, S.; Alahmad, B.; Hammad, M.; Al-Sabah, S.; Alghounaim, M.; Jamal, M.H.; Aldei, A.; Mairza, M.J.; et al. Robust Antibody Levels in Both Diabetic and Non-Diabetic Individuals After BNT162b2 mRNA COVID-19 Vaccination. Front. Immunol. 2021, 12, 752233. [Google Scholar] [CrossRef]
| 1st Time Point | 2nd Time Point | |||||
|---|---|---|---|---|---|---|
| p Value | OR * | 95% CI ** | p Value | OR | 95% CI | |
| Age, per 10 year higher | 0.007 | 2.460 | 1.13–6.14 | 0.136 | 2.630 | 1.13–6.14 |
| Gender, females vs. males | 0.264 | 0.571 | 0.21–1.53 | 0.722 | 0.834 | 0.31–2.27 |
| Positive N serology | 0.000 | 0.016 | 0.004–0.067 | 0.000 | 0.007 | 0.001–0.049 |
| Diabetes mellitus | 0.426 | 1.431 | 0.59–3.46 | 0.013 | 3.080 | 1.27–7.48 |
| Cancer | 0.231 | 2.198 | 0.61–7.98 | 0.835 | 0.869 | 0.23–3.26 |
| Corticosteroid therapy | 0.252 | 2.394 | 0.54–10.67 | 0.142 | 3.384 | 0.67–17.26 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, A.; Caimi, B.; Franzetti, M.; Bergna, A.; Velleca, R.; Gatti, A.; Rossi, P.L.; D’Orso, M.; Pregliasco, F.; Balotta, C.; et al. Durability of Humoral Responses after the Second Dose of mRNA BNT162b2 Vaccine in Residents of a Long Term Care Facility. Vaccines 2022, 10, 446. https://doi.org/10.3390/vaccines10030446
Lai A, Caimi B, Franzetti M, Bergna A, Velleca R, Gatti A, Rossi PL, D’Orso M, Pregliasco F, Balotta C, et al. Durability of Humoral Responses after the Second Dose of mRNA BNT162b2 Vaccine in Residents of a Long Term Care Facility. Vaccines. 2022; 10(3):446. https://doi.org/10.3390/vaccines10030446
Chicago/Turabian StyleLai, Alessia, Barbara Caimi, Marco Franzetti, Annalisa Bergna, Rossella Velleca, Antonella Gatti, Pier Luigi Rossi, Marco D’Orso, Fabrizio Pregliasco, Claudia Balotta, and et al. 2022. "Durability of Humoral Responses after the Second Dose of mRNA BNT162b2 Vaccine in Residents of a Long Term Care Facility" Vaccines 10, no. 3: 446. https://doi.org/10.3390/vaccines10030446
APA StyleLai, A., Caimi, B., Franzetti, M., Bergna, A., Velleca, R., Gatti, A., Rossi, P. L., D’Orso, M., Pregliasco, F., Balotta, C., & Calicchio, G. (2022). Durability of Humoral Responses after the Second Dose of mRNA BNT162b2 Vaccine in Residents of a Long Term Care Facility. Vaccines, 10(3), 446. https://doi.org/10.3390/vaccines10030446






