A Comparative Analysis of COVID-19 Vaccines Based on over 580,000 Cases from the Vaccination Adverse Event Reporting System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Extraction
2.2. Statistical Analysis
3. Results
3.1. Prevalence of Conditions in General Populations
3.2. Odds Ratios for Different Manufacturers, Gender and Age Groups
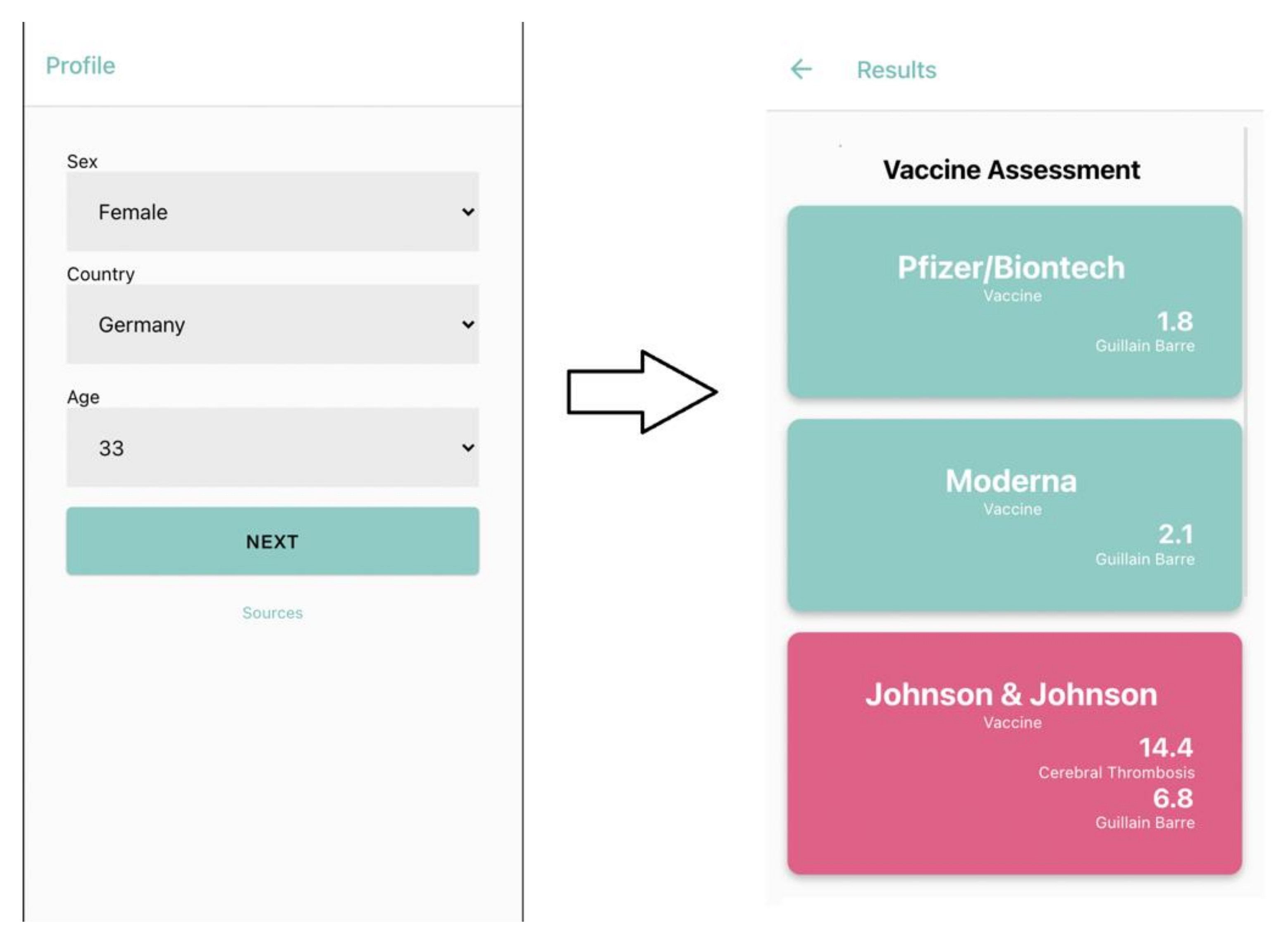
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Moderna. Clinical Study Protocol. Available online: https://www.modernatx.com/sites/default/files/mRNA-1273-P301-Protocol.pdf (accessed on 24 November 2021).
- Cines, D.B.; Bussel, J.B. SARS-CoV-2 Vaccine-Induced Immune Thrombotic Thrombocytopenia. N. Engl. J. Med. 2021, 10, 2254–2256. [Google Scholar] [CrossRef] [PubMed]
- Updated Recommendations from the Advisory Committee on Immunization Practices for Use of the Janssen (Johnson & Johnson) COVID-19 Vaccine After Reports of Thrombosis with Thrombocytopenia Syndrome Among Vaccine Recipients—United States, April 2021. Available online: https://www.cdc.gov/mmwr/volumes/70/wr/mm7017e4.htm (accessed on 24 November 2021).
- Behrouzi, R.; Punter, M. Diagnosis and Management of Cerebral Venous Thrombosis. Clin. Med. 2018, 18, 75–79. [Google Scholar] [CrossRef] [Green Version]
- Coutinho, J.M.; Zuurbier, S.M.; Aramideh, M.; Stam, J. The Incidence of Cerebral Venous Thrombosis. Stroke 2012, 43, 3375–3377. [Google Scholar] [CrossRef] [Green Version]
- Trimboli, M.; Zoleo, P.; Arabia, G.; Gambardella, A. Guillain-Barré Syndrome Following BNT162b2 COVID-19 Vaccine. Neurol. Sci. 2021, 42, 4401–4402. [Google Scholar] [CrossRef]
- Willison, H.J.; Jacobs, B.C.; van Doorn, P.A. Guillain-Barré syndrome. Lancet 2016, 388, 717–727. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0140673616003391 (accessed on 18 August 2021). [CrossRef] [Green Version]
- Jacobs, B.C.; Rothbarth, P.H.; van der Meché, F.G.A.; Herbrink, P.; Schmitz, P.I.M.; de Klerk, M.A.; Van Doorn, P.A. The spectrum of antecedent infections in Guillain-Barré syndrome. Neurology 1998, 51, 1110–1115. [Google Scholar] [CrossRef]
- Cao-Lormeau, V.-M.; Blake, A.; Mons, S.; Lastère, S.; Roche, C.; Vanhomwegen, J.; Dub, T.; Baudouin, L.; Teissier, A.; Larre, P.; et al. Guillain-Barré Syndrome outbreak associated with Zika virus infection in French Polynesia: A case-control study. Lancet 2016, 387, 1531–1539. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0140673616005626 (accessed on 18 August 2021). [CrossRef] [Green Version]
- Van den Berg, B.; van der Eijk, A.A.; Pas, S.D.; Hunter, J.G.; Madden, R.G.; Tio-Gillen, A.P.; Dalton, H.R.; Jacobs, B.C. Guillain-Barre syndrome associated with preceding hepatitis E virus infection. Neurology 2014, 82, 491–497. [Google Scholar] [CrossRef]
- McGonigal, R.; Rowan, E.G.; Greenshields, K.N.; Halstead, S.K.; Humphreys, P.D.; Rother, R.P.; Furkuawa, K.; Willison, H.J. Anti-GD1a antibodies activate complement and calpain to injure distal motor nodes of Ranvier in mice. Brain 2010, 133, 1944–1960. Available online: https://academic.oup.com/brain/article-lookup/doi/10.1093/brain/awq119 (accessed on 14 August 2021). [CrossRef] [Green Version]
- Salmon, D.A.; Proschan, M.; Forshee, R.; Gargiullo, P.; Bleser, W.; Burwen, D.R.; Cunningham, F.; Garman, P.; Greene, S.K.; Lee, G.M.; et al. Association between Guillain-Barré syndrome and influenza A (H1N1) 2009 monovalent inactivated vaccines in the USA: A meta-analysis. Lancet 2013, 381, 1461–1468. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0140673612621898 (accessed on 18 August 2021). [CrossRef]
- Martín Arias, L.H.; Sanz, R.; Sáinz, M.; Treceño, C.; Carvajal, A. Guillain-Barré syndrome and influenza vaccines: A meta-analysis. Vaccine 2015, 33, 3773–3778. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0264410X15006349 (accessed on 18 August 2021). [CrossRef]
- Das, B.B.; Moskowitz, W.B.; Taylor, M.B.; Palmer, A. Myocarditis and Pericarditis Following mRNA COVID-19 Vaccination: What Do We Know So Far? Children 2021, 8, 607. Available online: https://www.mdpi.com/2227-9067/8/7/607 (accessed on 18 August 2021). [CrossRef]
- Su, J.R.; McNeil, M.M.; Welsh, K.J.; Marquez, P.L.; Ng, C.; Yan, M.; Cano, M.V. Myopericarditis after vaccination, Vaccine Adverse Event Reporting System (VAERS), 1990–2018. Vaccine 2021, 39, 839–845. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0264410X20316145 (accessed on 16 August 2021). [CrossRef]
- Diaz, G.A.; Parsons, G.T.; Gering, S.K.; Meier, A.R.; Hutchinson, I.V.; Robicsek, A. Myocarditis and Pericarditis After Vaccination for COVID-19. JAMA 2021, 326, 1210. Available online: https://jamanetwork.com/journals/jama/fullarticle/2782900 (accessed on 7 September 2018). [CrossRef]
- Cai, C.; Peng, Y.; Shen, E.; Huang, Q.; Chen, Y.; Liu, P.; Guo, C.; Feng, Z.; Gao, L.; Zhang, X.; et al. A comprehensive analysis of the efficacy and safety of COVID-19 vaccines. Mol. Ther. 2021, 29, 2794–2805. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1525001621003956 (accessed on 24 November 2021). [CrossRef]
- Pawlowski, C.; Rincón-Hekking, J.; Awasthi, S.; Pandey, V.; Lenehan, P.; Venkatakrishnan, A.; Bade, S.; O’Horo, J.C.; Virk, A.; Swift, M.D.; et al. Cerebral Venous Sinus Thrombosis is not Significantly Linked to COVID-19 Vaccines or Non-COVID Vaccines in a Large Multi-State Health System. J Stroke Cerebrovasc. Dis. 2021, 30, 105923. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1052305721003268 (accessed on 26 November 2021). [CrossRef]
- Shasha, D.; Bareket, R.; Sikron, F.H.; Gertel, O.; Tsamir, J.; Dvir, D.; Mossinson, D.; Heymann, A.D.; Zacay, G. Real-world safety data for the Pfizer BNT162b2 SARS-CoV-2 vaccine: Historical cohort study. Clin. Microbiol. Infect. 2021, 28, 130–134. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1198743X21005383 (accessed on 26 November 2021). [CrossRef]
- Sánchez van Kammen, M.; Aguiar de Sousa, D.; Poli, S.; Cordonnier, C.; Heldner, M.R.; van de Munckhof, A.; Krzywicka, K.; van Haaps, T.; Ciccone, A.; Middeldorp, S.; et al. Characteristics and Outcomes of Patients With Cerebral Venous Sinus Thrombosis in SARS-CoV-2 Vaccine-Induced Immune Thrombotic Thrombocytopenia. JAMA Neurol. 2021, 78, 1314. Available online: https://jamanetwork.com/journals/jamaneurology/fullarticle/2784622 (accessed on 26 November 2021). [CrossRef]
- Kim, M.S.; Jung, S.Y.; Ahn, J.G.; Park, S.J.; Shoenfeld, Y.; Kronbichler, A.; Koyanagi, A.; Dragioti, E.; Tizaoui, K.; Hong, S.H.; et al. Comparative safety of mRNA COVID-19 vaccines to influenza vaccines: A pharmacovigilance analysis using WHO international database. J. Med. Virol. 2021, 94, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Szumilas, M. Explaining Odds Ratios. J. Can. Acad. Child Adolesc. Psychiatry 2010, 19, 227–229. [Google Scholar] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 12 December 2021).
- Agency for Healthcare Research and Quality (AHRQ). Available online: https://www.hcup-us.ahrq.gov/nisoverview.jsp (accessed on 11 August 2021).
- Risk/Benefit Assessment of Thrombotic Thrombocytopenic Events after Janssen COVID-19 Vaccines: Applying Evidence to Recommendation Framework. Available online: https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-04-23/06-COVID-Oliver-508.pdf (accessed on 11 August 2021).
- Janghorbani, M.; Zare, M.; Saadatnia, M.; Mousavi, S.A.; Mojarrad, M.; Asgari, E. Cerebral vein and dural sinus thrombosis in adults in Isfahan, Iran: Frequency and seasonal variation. Acta Neurol. Scand. 2007, 117, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Bousser, M.-G.; Ferro, J.M. Cerebral venous thrombosis: An update. Lancet Neurol. 2007, 6, 62–70. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1474442207700297 (accessed on 12 August 2021). [CrossRef]
- Ferro, J.M.; Correia, M.; Pontes, C.; Baptista, M.V.; Pita, F. Cerebral Vein and Dural Sinus Thrombosis in Portugal: 1980–1998. Cerebrovasc. Dis. 2001, 11, 177–182. Available online: https://www.karger.com/Article/FullText/47635 (accessed on 12 August 2021). [CrossRef]
- Mak, W.; Mok, K.Y.; Tsoi, T.H.; Cheung, R.T.F.; Ho, S.L.; Chang, C.M. Cerebral Venous Thrombosis in Hong Kong. Cerebrovasc. Dis. 2001, 11, 282–283. Available online: https://www.karger.com/Article/FullText/47652 (accessed on 12 August 2021). [CrossRef]
- Levison, L.S.; Thomsen, R.W.; Christensen, D.H.; Mellemkjær, T.; Sindrup, S.H.; Andersen, H. Guillain-Barre syndrome in Denmark: Validation of diagnostic codes and a population-based nationwide study of the incidence in a 30-year period. Clin. Epidemiol. 2019, 11, 275–283. Available online: https://www.dovepress.com/guillain-barre-syndrome-in-denmark-validation-of-diagnostic-codes-and--peer-reviewed-article-CLEP (accessed on 16 August 2021). [CrossRef] [Green Version]
- Van den Berg, B.; Walgaard, C.; Drenthen, J.; Fokke, C.; Jacobs, B.C.; van Doorn, P.A. Guillain-Barré syndrome: Pathogenesis, diagnosis, treatment and prognosis. Nat. Rev. Neurol. 2014, 10, 469–482. Available online: http://www.nature.com/articles/nrneurol.2014.121 (accessed on 16 August 2021). [CrossRef]
- Kuwabara, S. Guillain-Barre Syndrome. Drugs 2004, 64, 597–610. [Google Scholar] [CrossRef]
- Kang, M.; An, J. Viral Myocarditis. StatPearls Treasure Isl. StatPearls Publ. 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459259/ (accessed on 14 August 2021).
- Golpour, A.; Patriki, D.; Hanson, P.J.; McManus, B.; Heidecker, B. Epidemiological Impact of Myocarditis. J. Clin. Med. 2021, 10, 603. [Google Scholar] [CrossRef]
- Klein, A.; Cremer, P.; Kontzias, A.; Furqan, M.; Tubman, R.; Roy, M.; Lim-Watson, M.Z.; Magestro, M. US Database Study of Clinical Burden and Unmet Need in Recurrent Pericarditis. J. Am. Heart Assoc. 2021, 10, e018950. [Google Scholar] [CrossRef]
- Kytö, V.; Sipilä, J.; Rautava, P. Clinical Profile and Influences on Outcomes in Patients Hospitalized for Acute Pericarditis. Circulation 2014, 130, 1601–1606. [Google Scholar] [CrossRef] [Green Version]
- Imazio, M.; Cecchi, E.; Demichelis, B.; Chinaglia, A.; Ierna, S.; Demarie, D.; Ghisio, A.; Pomari, F.; Belli, R.; Trinchero, R. Myopericarditis versus viral or idiopathic acute pericarditis. Heart 2007, 94, 498–501. [Google Scholar] [CrossRef]
| Incidences per Million/Year (Citation/Source) | Publication Year | Region |
|---|---|---|
| A: Cerebral thrombosis | ||
| 2.4 [26] | 2019 | USA |
| 14.5–28.5 [27] | 2018 | USA |
| 13.2 [7] | 2012 | Netherlands |
| 12.3 [28] | 2008 | Iran |
| 2–5 [29] | 2007 | Great Britain |
| 2.2 [30] | 2001 | Portugal |
| 3.4 [31] | 2001 | Hong Kong |
| B: Guillain-Barré syndrome | ||
| 17.7 [32] | 2019 | Denmark |
| 8.1–18.9 [33] | 2014 | Worldwide |
| 13 [34] | 2004 | Worldwide |
| C: Myocarditis | ||
| 100–200 [35] | 2021 | Worldwide |
| 102–1056 [36] | 2017 | Worldwide |
| 104–400 [36] | 2017 | USA |
| D: Pericarditis | ||
| 78.3 [37] | 2021 | USA |
| 33.2 [38] | 2014 | Finland |
| 277 [39] | 2007 | Italy |
| Johnson & Johnson | Pfizer/BioNTech | Moderna | |
|---|---|---|---|
| A: Cerebral thrombosis | |||
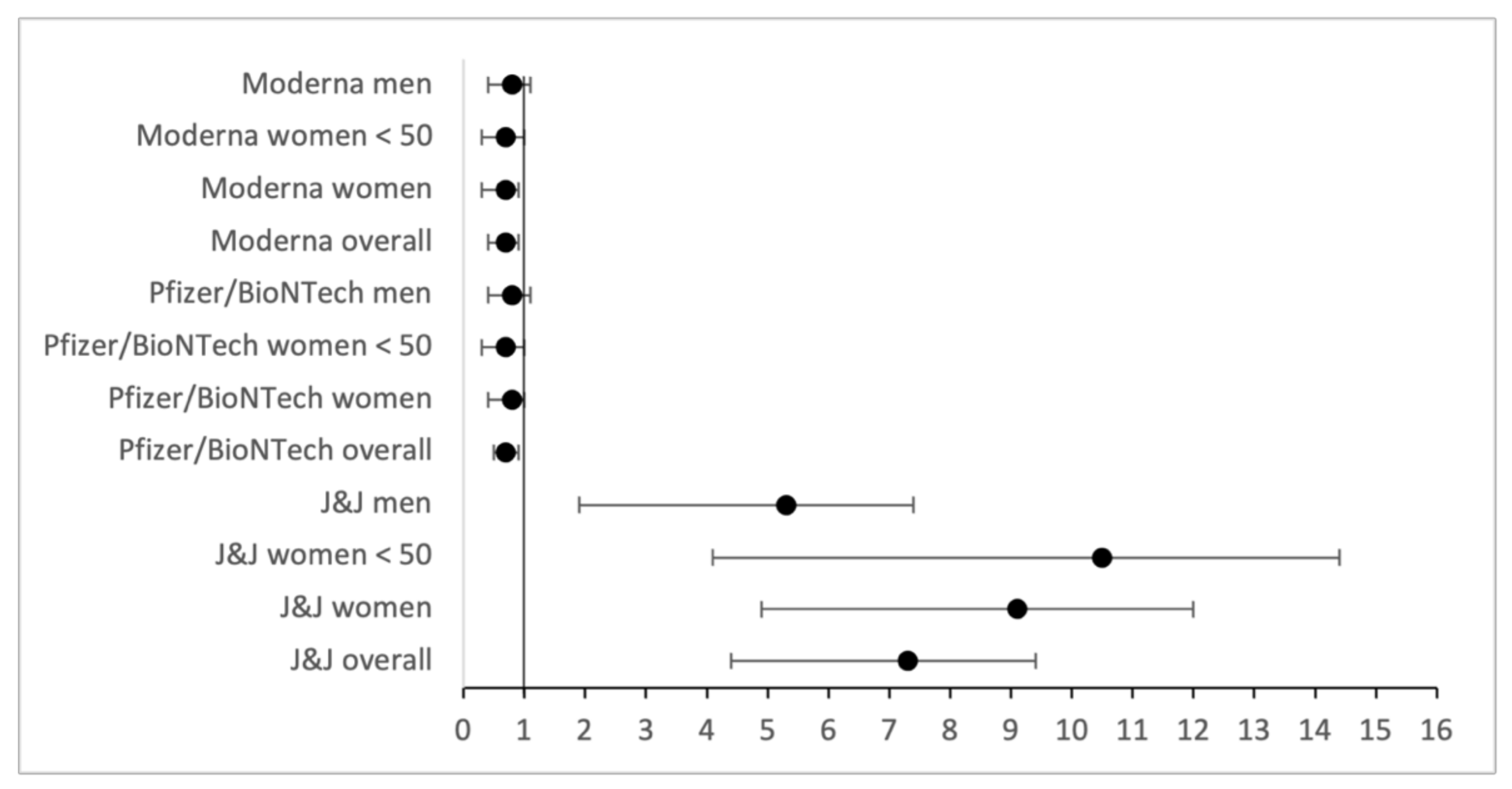
| Overall OR | 10.0 | 0.9 | 1.0 |
| OR Women | 12.5 | 1.1 | 1.0 |
| OR Women < 50 | 14.4 | 0.9 | 1.0 |
| OR Men | 7.3 | 0.6 | 1.1 |
| B: Guillain-Barré syndrome | |||
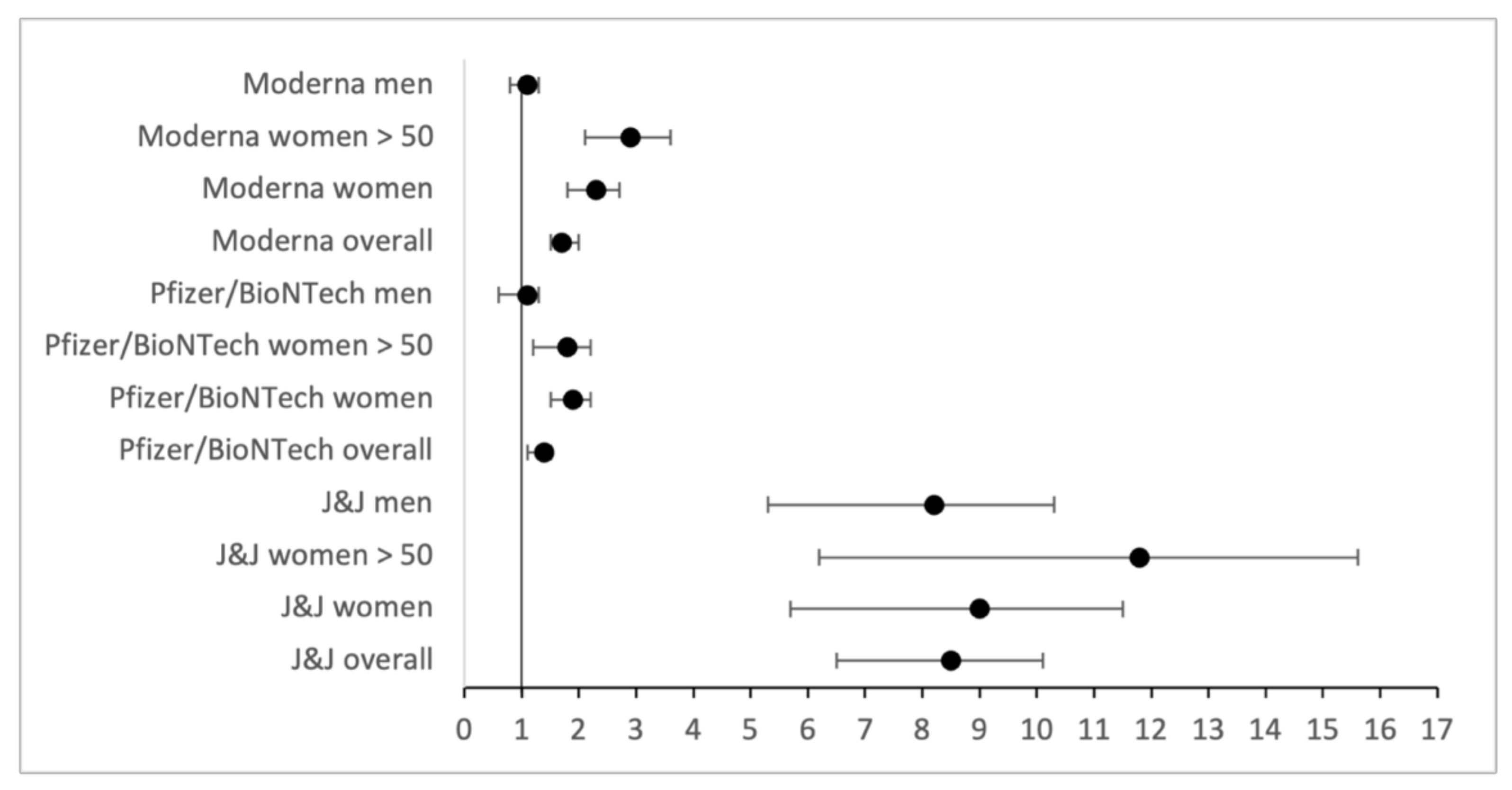
| Overall OR | 11.6 | 2.0 | 2.2 |
| OR Women | 10.2 | 2.2 | 2.6 |
| OR Men | 13.0 | 1.7 | 1.8 |
| OR Men > 50 | 17.1 | 1.9 | 2.5 |
| OR Women > 50 | 13.4 | 2.1 | 3.3 |
| C: Myocarditis | |||
| Overall OR | 0.2 | 0.7 | 0.4 |
| OR Women | 0.1 | 0.3 | 0.2 |
| OR Men | 0.2 | 1.1 | 0.7 |
| OR Men < 25 | 0.1 | 5.3 | 1.9 |
| D: Pericarditis | |||
| Overall OR | 0.6 | 0.7 | 0.5 |
| OR Women | 0.6 | 0.4 | 0.3 |
| OR Men | 0.7 | 1.0 | 0.7 |
| OR Men < 25 | 1.3 | 4.1 | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallo, K.; Goede, A.; Mura, C.; Abel, R.; Moahamed, B.; Preissner, S.; Nahles, S.; Heiland, M.; Bourne, P.E.; Preissner, R.; et al. A Comparative Analysis of COVID-19 Vaccines Based on over 580,000 Cases from the Vaccination Adverse Event Reporting System. Vaccines 2022, 10, 408. https://doi.org/10.3390/vaccines10030408
Gallo K, Goede A, Mura C, Abel R, Moahamed B, Preissner S, Nahles S, Heiland M, Bourne PE, Preissner R, et al. A Comparative Analysis of COVID-19 Vaccines Based on over 580,000 Cases from the Vaccination Adverse Event Reporting System. Vaccines. 2022; 10(3):408. https://doi.org/10.3390/vaccines10030408
Chicago/Turabian StyleGallo, Kathleen, Andrean Goede, Cameron Mura, Renata Abel, Barbara Moahamed, Saskia Preissner, Susanne Nahles, Max Heiland, Philip E. Bourne, Robert Preissner, and et al. 2022. "A Comparative Analysis of COVID-19 Vaccines Based on over 580,000 Cases from the Vaccination Adverse Event Reporting System" Vaccines 10, no. 3: 408. https://doi.org/10.3390/vaccines10030408
APA StyleGallo, K., Goede, A., Mura, C., Abel, R., Moahamed, B., Preissner, S., Nahles, S., Heiland, M., Bourne, P. E., Preissner, R., & Mallach, M. (2022). A Comparative Analysis of COVID-19 Vaccines Based on over 580,000 Cases from the Vaccination Adverse Event Reporting System. Vaccines, 10(3), 408. https://doi.org/10.3390/vaccines10030408








