Ocular Complications Following Vaccination for COVID-19: A One-Year Retrospective
Abstract
1. Introduction
2. Materials and Methods
3. Adverse Ocular Events: Patient Overview
4. Eyelid
5. Orbit
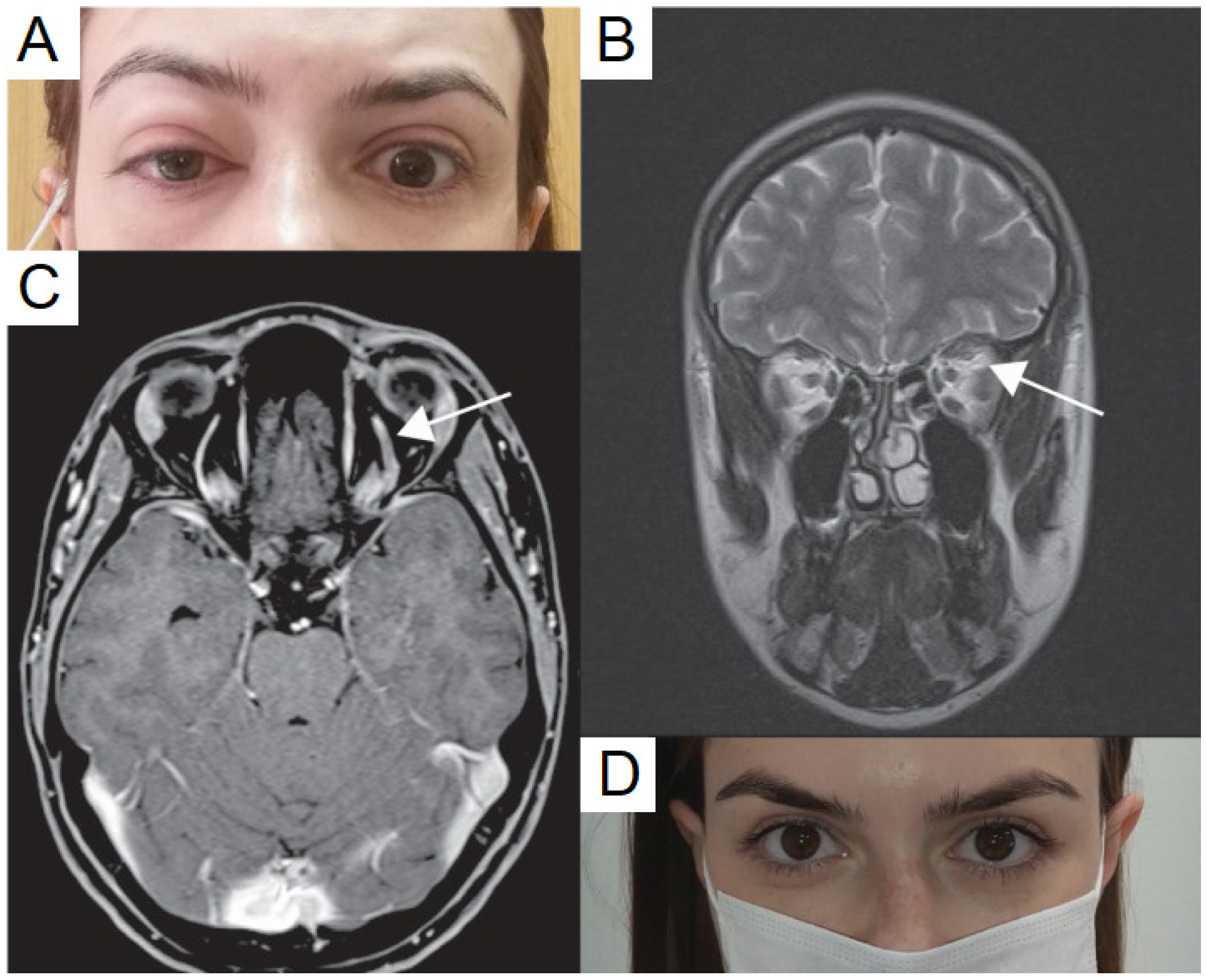
5.1. Superior Ophthalmic Vein Thrombosis
5.2. Tolosa-Hunt Syndrome
5.3. Mechanisms
6. Uveitis
6.1. Uveitis Flares
6.2. Choroiditis
6.3. Vogt–Koyanagi–Harada Disease
6.4. Acute Retinal Necrosis
6.5. Acute Zonal Occult Outer Retinopathy
6.6. Mechanisms
7. Cornea
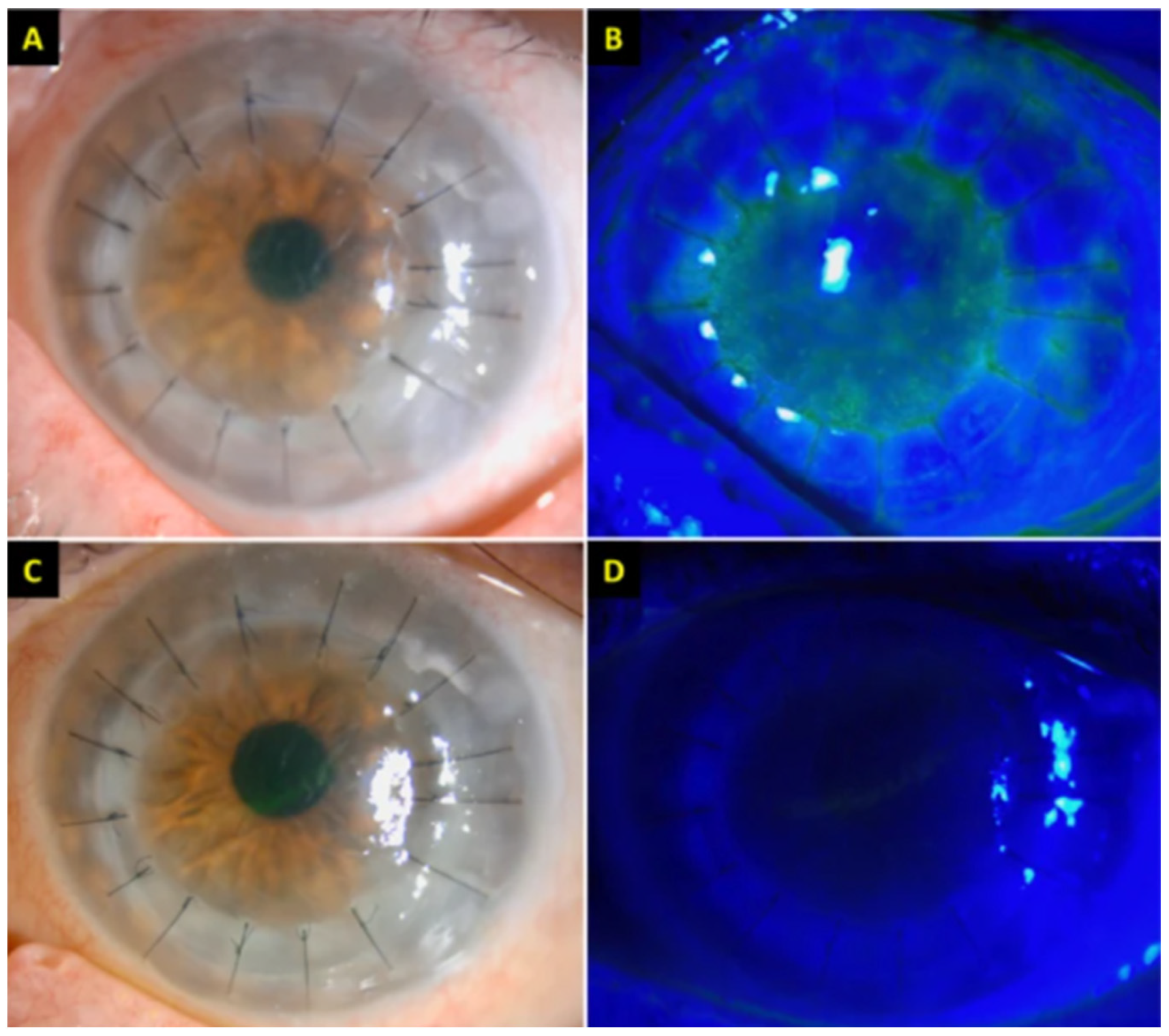
7.1. Graft Rejection
7.2. Corneal Melting
7.3. Mechanisms
8. Retina
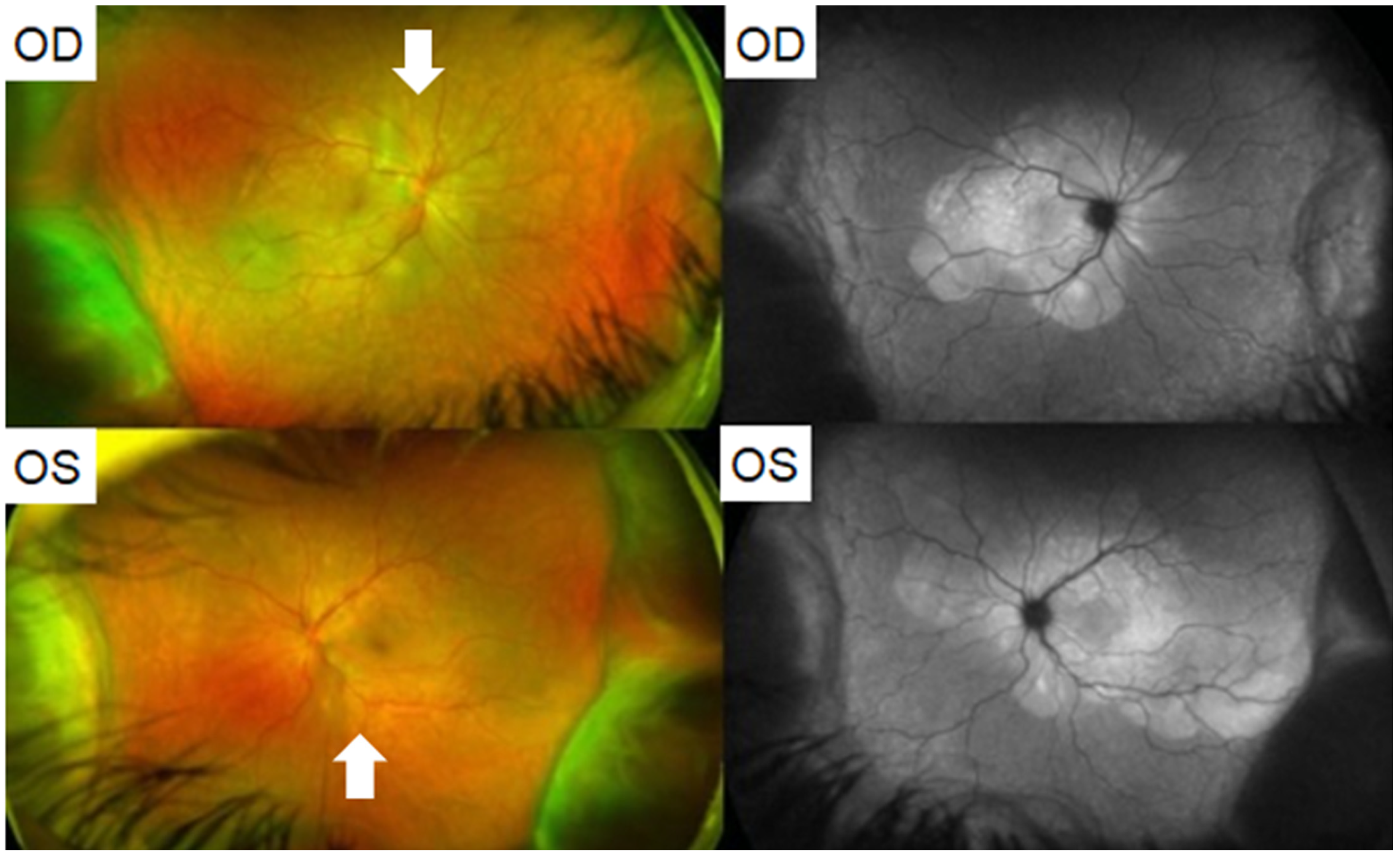
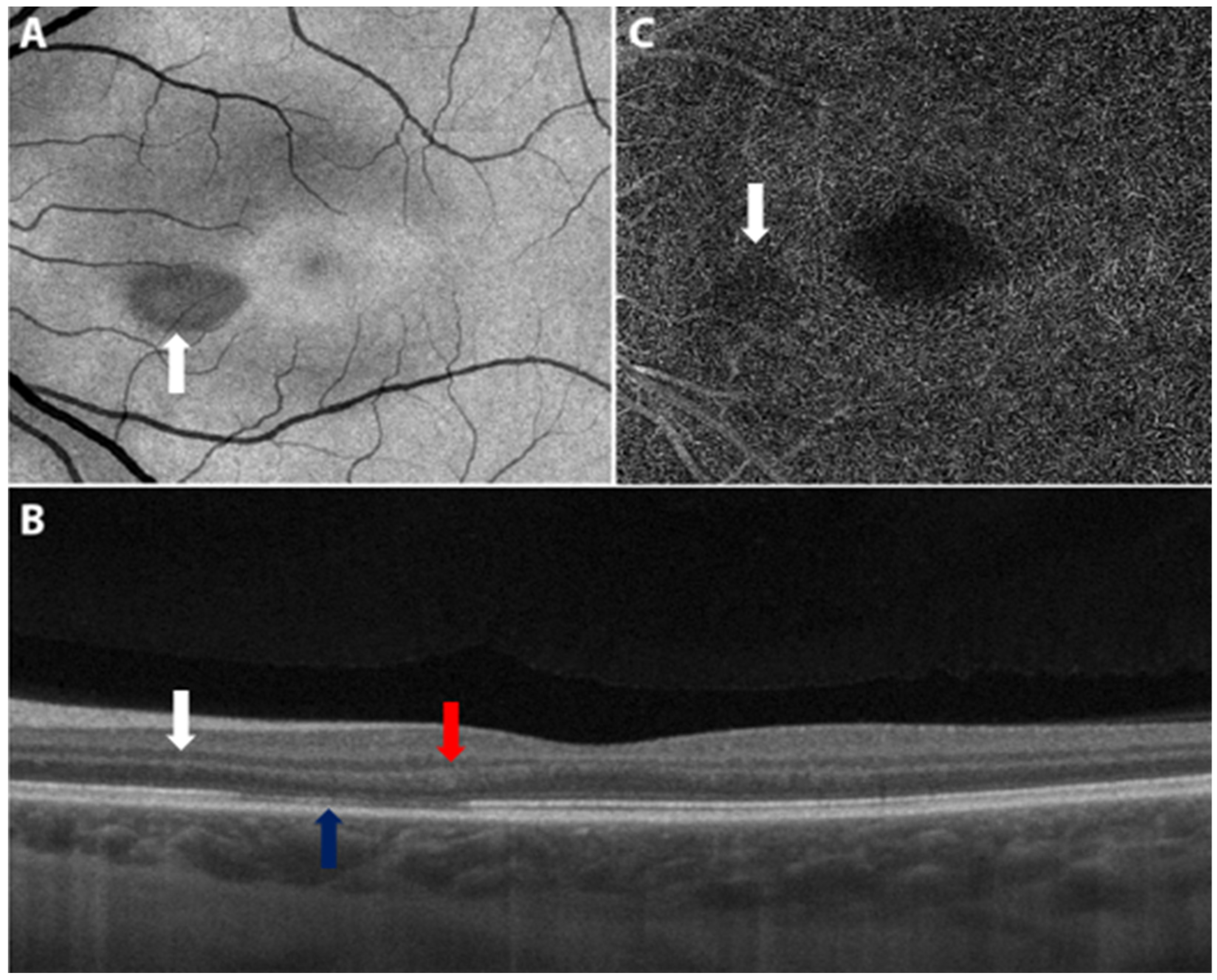
8.1. Acute Macular Neuroretinopathy
8.2. Paracentral Acute Middle Maculopathy
8.3. Other Reports
8.4. Mechanisms
9. Vascular
9.1. Central and Hemi-Retinal Vein Occlusion
9.2. Mechanisms
10. Neuro-Ophthalmology
10.1. Optic Neuritis
10.2. Other Reports
10.3. Mechanisms
11. Ocular Motility Disorders
11.1. Ocular Gaze Palsies
11.2. Ocular Gaze Palsies
12. Other Reports
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saied, S.M.; Saied, E.M.; Kabbash, I.A.; Abdo, S.A.E. Vaccine hesitancy: Beliefs and barriers associated with COVID-19 vaccination among Egyptian medical students. J. Med. Virol. 2021, 93, 4280–4291. [Google Scholar] [CrossRef]
- Kadali, R.A.K.; Janagama, R.; Peruru, S.R.; Racherla, S.; Tirumala, R.; Madathala, R.R.; Gajula, V. Adverse effects of COVID-19 messenger RNA vaccines among pregnant women: A cross-sectional study on healthcare workers with detailed self-reported symptoms. Am. J. Obs. Gynecol. 2021, 225, 458–460. [Google Scholar] [CrossRef]
- Meo, S.A.; Fahad Al-Jassir, F.; Al-Qahtani, S.; Albarrak, R.; Usmani, A.M.; Klonoff, D.C. Effect of Pfizer/BioNTech and Oxford/AstraZeneca vaccines against COVID-19 morbidity and mortality in real-world settings at countrywide vaccination campaign in Saudi Arabia. Eur. Rev. Med. Pharm. Sci. 2021, 25, 7185–7191. [Google Scholar] [CrossRef]
- Meo, S.A.; Bukhari, I.A.; Akram, J.; Meo, A.S.; Klonoff, D.C. COVID-19 vaccines: Comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur. Rev. Med. Pharm. Sci. 2021, 25, 1663–1669. [Google Scholar] [CrossRef]
- Singh, S.; Garcia, G.; Shah, R.; Kramerov, A.A.; Wright, R.E., 3rd; Spektor, T.M.; Ljubimov, A.V.; Arumugaswami, V.; Kumar, A. SARS-CoV-2 and its beta variant of concern infect human conjunctival epithelial cells and induce differential antiviral innate immune response. Ocul. Surf. 2021, 23, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.; Queiruga-Pineiro, J.; Lozano-Sanroma, J.; Alcalde, I.; Gallar, J.; Fernandez-Vega Cueto, L.; Alfonso, J.F.; Quiros, L.M.; Merayo-Lloves, J. Small fiber neuropathy in the cornea of Covid-19 patients associated with the generation of ocular surface disease. Ocul. Surf. 2021, 23, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, Y.; Musch, D.C.; Wei, N.; Qi, X.; Ding, G.; Li, X.; Li, J.; Song, L.; Zhang, Y.; et al. Progression of Myopia in School-Aged Children After COVID-19 Home Confinement. JAMA Ophthalmol. 2021, 139, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Rokohl, A.C.; Grajewski, R.S.; Matos, P.A.W.; Kopecky, A.; Heindl, L.M.; Cursiefen, C. Ocular Involvement in COVID-19: Conjunctivitis and More. Klin. Monbl. Augenheilkd. 2021, 238, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, L.; Pacella, F.; Pacella, E.; Tiscione, G.; Oliva, A.; Violi, F. Conjunctivitis and COVID-19: A meta-analysis. J. Med. Virol. 2020, 92, 1413–1414. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, A.S.; Farford, B.A.; Forouhi, M.; Abdin, R.; Salinas, M. The Ocular Manifestations of COVID-19 through Conjunctivitis. Cureus 2020, 12, e12218. [Google Scholar] [CrossRef]
- Elhusseiny, A.M.; Eleiwa, T.K.; Yacoub, M.S.; George, J.; ElSheikh, R.H.; Haseeb, A.; Kwan, J.; Elsaadani, I.A.; Abo Shanab, S.M.; Solyman, O.; et al. Relationship between screen time and dry eye symptoms in pediatric population during the COVID-19 pandemic. Ocul. Surf. 2021, 22, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Eleiwa, T.K.; Elmaghrabi, A.; Helal, H.G.; Abdelrahman, S.N.; ElSheikh, R.H.; Elhusseiny, A.M. Phlyctenular Keratoconjunctivitis in a Patient with COVID-19 Infection. Cornea 2021, 40, 1502–1504. [Google Scholar] [CrossRef] [PubMed]
- Eleiwa, T.; Abdelrahman, S.N.; ElSheikh, R.H.; Elhusseiny, A.M. Orbital inflammatory disease associated with COVID-19 infection. J. AAPOS 2021, 25, 232–234. [Google Scholar] [CrossRef] [PubMed]
- Eleiwa, T.K.; Gaier, E.D.; Haseeb, A.; ElSheikh, R.H.; Sallam, A.B.; Elhusseiny, A.M. Adverse Ocular Events following COVID-19 Vaccination. Inflamm. Res. 2021, 70, 1005–1009. [Google Scholar] [CrossRef]
- Ng, X.L.; Betzler, B.K.; Ng, S.; Chee, S.P.; Rajamani, L.; Singhal, A.; Rousselot, A.; Pavesio, C.E.; Gupta, V.; de Smet, M.D.; et al. The Eye of the Storm: COVID-19 Vaccination and the Eye. Ophthalmol. Ther. 2021, 11, 81–100. [Google Scholar] [CrossRef] [PubMed]
- Sen, M.; Honavar, S.G. After the Storm: Ophthalmic Manifestations of COVID-19 Vaccines. Indian J. Ophthalmol. 2021, 69, 3398–3420. [Google Scholar] [CrossRef] [PubMed]
- Austria, Q.M.; Lelli, G.J.; Segal, K.L.; Godfrey, K.J. Transient Eyelid Edema following COVID-19 Vaccination. Ophthalmic. Plast Reconstr. Surg. 2021, 37, 501–502. [Google Scholar] [CrossRef]
- Mazzatenta, C.; Piccolo, V.; Pace, G.; Romano, I.; Argenziano, G.; Bassi, A. Purpuric lesions on the eyelids developed after BNT162b2 mRNA COVID-19 vaccine: Another piece of SARS-CoV-2 skin puzzle? J. Eur. Acad. Derm. Venereol. 2021, 35, e543–e545. [Google Scholar] [CrossRef]
- Bayas, A.; Menacher, M.; Christ, M.; Behrens, L.; Rank, A.; Naumann, M. Bilateral superior ophthalmic vein thrombosis, ischaemic stroke, and immune thrombocytopenia after ChAdOx1 nCoV-19 vaccination. Lancet 2021, 397, e11. [Google Scholar] [CrossRef]
- Panovska-Stavridis, I.; Pivkova-Veljanovska, A.; Trajkova, S.; Lazarevska, M.; Grozdanova, A.; Filipche, V. A Rare Case of Superior Ophthalmic Vein Thrombosis and Thrombocytopenia following ChAdOx1 nCoV-19 Vaccine against SARS-CoV-2. Mediterr. J. Hematol. Infect Dis. 2021, 13, e2021048. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.Y.; Burda, K.; Teklemariam, E.; Athar, K. Tolosa-Hunt Syndrome Presenting after COVID-19 Vaccination. Cureus 2021, 13, e16791. [Google Scholar] [CrossRef] [PubMed]
- Dutta, P.; Anand, K. Tolosa-Hunt Syndrome: A Review of Diagnostic Criteria and Unresolved Issues. J. Curr. Ophthalmol. 2021, 33, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Hyer, R.; McGuire, D.K.; Xing, B.; Jackson, S.; Janssen, R. Safety of a two-dose investigational hepatitis B vaccine, HBsAg-1018, using a toll-like receptor 9 agonist adjuvant in adults. Vaccine 2018, 36, 2604–2611. [Google Scholar] [CrossRef] [PubMed]
- Schultz, N.H.; Sorvoll, I.H.; Michelsen, A.E.; Munthe, L.A.; Lund-Johansen, F.; Ahlen, M.T.; Wiedmann, M.; Aamodt, A.H.; Skattor, T.H.; Tjonnfjord, G.E.; et al. Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. 2021, 384, 2124–2130. [Google Scholar] [CrossRef]
- Owatanapanich, S.; Wanlapakorn, N.; Tangsiri, R.; Poovorawan, Y. Measles-mumps-rubella vaccination induced thrombocytopenia: A case report and review of the literature. Southeast Asian J. Trop. Med. Public Health 2014, 45, 1053–1057. [Google Scholar]
- Kashyape, S.S.; Kashyape, P.S. Thrombocytopenia following MMR vaccination. Indian Pediatr. 2005, 42, 80–82. [Google Scholar]
- Forrest, J.M.; Honeyman, M.C.; Lovric, V.A. Rubella vaccination and thrombocytopenia. Aust. N. Z. J. Med. 1974, 4, 352–355. [Google Scholar] [CrossRef]
- Bartos, H.R. Thrombocytopenia associated with rubella vaccination. N. Y. State J. Med. 1972, 72, 499. [Google Scholar]
- Yamamoto, Y.; Ohara, Y.; Iwai, A.; Hara, R.; Matsuki, T.; Fukushima, K.; Oshitani, Y.; Kagawa, H.; Tsujino, K.; Yoshimura, K.; et al. Influenza Vaccination-associated Acute Thrombocytopenia and Diffuse Alveolar Hemorrhage. Intern. Med. 2020, 59, 1633–1637. [Google Scholar] [CrossRef]
- Moulis, G.; Sailler, L.; Sommet, A.; Lapeyre-Mestre, M.; Adoue, D. Prevalence of pneumococcal vaccination in adult immune thrombocytopenia patients exposed to rituximab or to splenectomy. Eur. J. Haematol. 2013, 90, 438–439. [Google Scholar] [CrossRef]
- Meindersma, T.E. Thrombocytopenia after vaccination against smallpox. Ned. Milit. Geneeskd. Tijdschr. 1961, 14, 199–205. [Google Scholar]
- McCrae, K.R. Thrombotic thrombocytopenia due to SARS-CoV-2 vaccination. Clevel. Clin. J. Med. 2021. [Google Scholar] [CrossRef]
- Lee, E.J.; Cines, D.B.; Gernsheimer, T.; Kessler, C.; Michel, M.; Tarantino, M.D.; Semple, J.W.; Arnold, D.M.; Godeau, B.; Lambert, M.P.; et al. Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination. Am. J. Hematol. 2021, 96, 534–537. [Google Scholar] [CrossRef]
- Lai, K.Y.; Au, S.Y.; Fong, K.M. Thrombotic Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. 2021, 385, e11. [Google Scholar] [CrossRef]
- Greinacher, A.; Thiele, T.; Warkentin, T.E.; Weisser, K.; Kyrle, P.A.; Eichinger, S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. 2021, 384, 2092–2101. [Google Scholar] [CrossRef]
- Bitzan, M.; Zieg, J. Influenza-associated thrombotic microangiopathies. Pediatr. Nephrol. 2018, 33, 2009–2025. [Google Scholar] [CrossRef]
- ElSheikh, R.H.; Haseeb, A.; Eleiwa, T.K.; Elhusseiny, A.M. Acute Uveitis following COVID-19 Vaccination. Ocul. Immunol. Inflamm. 2021, 29, 1207–1209. [Google Scholar] [CrossRef]
- Jain, A.; Kalamkar, C. COVID-19 vaccine-associated reactivation of uveitis. Indian J. Ophthalmol. 2021, 69, 2899–2900. [Google Scholar] [CrossRef]
- Mudie, L.I.; Zick, J.D.; Dacey, M.S.; Palestine, A.G. Panuveitis following Vaccination for COVID-19. Ocul. Immunol. Inflamm. 2021, 29, 1–2. [Google Scholar] [CrossRef]
- Renisi, G.; Lombardi, A.; Stanzione, M.; Invernizzi, A.; Bandera, A.; Gori, A. Anterior uveitis onset after bnt162b2 vaccination: Is this just a coincidence? Int. J. Infect. Dis. 2021, 110, 95–97. [Google Scholar] [CrossRef]
- Ishay, Y.; Kenig, A.; Tsemach-Toren, T.; Amer, R.; Rubin, L.; Hershkovitz, Y.; Kharouf, F. Autoimmune phenomena following SARS-CoV-2 vaccination. Int. Immunopharmacol. 2021, 99, 107970. [Google Scholar] [CrossRef]
- Herbort, C.P., Jr.; Papasavvas, I. Effect of SARS-CoV-2 mRNA vaccination on ocular herpes simplex and varicella-zoster virus reactivation: Should preventive antiviral treatment be given in known herpes patients. J. Ophthalmic. Inflamm. Infect. 2021, 11, 33. [Google Scholar] [CrossRef]
- Rabinovitch, T.; Ben-Arie-Weintrob, Y.; Hareuveni-Blum, T.; Shaer, B.; Vishnevskia-Dai, V.; Shulman, S.; Newman, H.; Biadsy, M.; Masarwa, D.; Fischer, N.; et al. Uveitis following the BNT162b2 mRNA vaccination against SARS-CoV-2 infection: A possible association. Retina 2021, 41, 2462–2471. [Google Scholar] [CrossRef]
- Goyal, M.; Murthy, S.I.; Annum, S. Bilateral Multifocal Choroiditis following COVID-19 Vaccination. Ocul. Immunol. Inflamm. 2021, 29, 1–5. [Google Scholar] [CrossRef]
- Pan, L.; Zhang, Y.; Cui, Y.; Wu, X. Bilateral uveitis after inoculation with COVID-19 vaccine: A case report. Int. J. Infect. Dis. 2021, 113, 116–118. [Google Scholar] [CrossRef]
- Read, R.W.; Rao, N.A.; Cunningham, E.T. Vogt-Koyanagi-Harada disease. Curr. Opin. Ophthalmol. 2000, 11, 437–442. [Google Scholar] [CrossRef]
- Read, R.W.; Holland, G.N.; Rao, N.A.; Tabbara, K.F.; Ohno, S.; Arellanes-Garcia, L.; Pivetti-Pezzi, P.; Tessler, H.H.; Usui, M. Revised diagnostic criteria for Vogt-Koyanagi-Harada disease: Report of an international committee on nomenclature. Am. J. Ophthalmol. 2001, 131, 647–652. [Google Scholar] [CrossRef]
- Rao, N.A.; Gupta, A.; Dustin, L.; Chee, S.P.; Okada, A.A.; Khairallah, M.; Bodaghi, B.; Lehoang, P.; Accorinti, M.; Mochizuki, M.; et al. Frequency of distinguishing clinical features in Vogt-Koyanagi-Harada disease. Ophthalmology 2010, 117, 591–599. [Google Scholar] [CrossRef]
- Harada, Y.; Bhat, P.; Munk, M.R.; Goldstein, D.A. Changes in Scleral Architecture in Chronic Vogt-Koyanagi-Harada Disease. Ocul. Immunol. Inflamm. 2017, 25, 85–92. [Google Scholar] [CrossRef]
- Papasavvas, I.; Herbort, C.P., Jr. Reactivation of Vogt-Koyanagi-Harada disease under control for more than 6 years, following anti-SARS-CoV-2 vaccination. J. Ophthalmic. Inflamm. Infect. 2021, 11, 21. [Google Scholar] [CrossRef]
- Saraceno, J.J.F.; Souza, G.M.; Dos Santos Finamor, L.P.; Nascimento, H.M.; Belfort, R., Jr. Vogt-Koyanagi-Harada Syndrome following COVID-19 and ChAdOx1 nCoV-19 (AZD1222) vaccine. Int. J. Retin. Vitr. 2021, 7, 49. [Google Scholar] [CrossRef]
- Mishra, S.B.; Mahendradas, P.; Kawali, A.; Sanjay, S.; Shetty, R. Reactivation of varicella zoster infection presenting as acute retinal necrosis post COVID 19 vaccination in an Asian Indian male. Eur. J. Ophthalmol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Maleki, A.; Look-Why, S.; Manhapra, A.; Foster, C.S. COVID-19 Recombinant mRNA Vaccines and Serious Ocular Inflammatory Side Effects: Real or Coincidence? J. Ophthalmic. Vis. Res. 2021, 16, 490–501. [Google Scholar] [CrossRef]
- Holt, H.D.; Hinkle, D.M.; Falk, N.S.; Fraunfelder, F.T.; Fraunfelder, F.W. Human papilloma virus vaccine associated uveitis. Curr. Drug Saf. 2014, 9, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Heydari-Kamjani, M.; Vante, I.; Uppal, P.; Demory Beckler, M.; Kesselman, M.M. Uveitis Sarcoidosis Presumably Initiated after Administration of Shingrix Vaccine. Cureus 2019, 11, e4920. [Google Scholar] [CrossRef] [PubMed]
- Fraunfelder, F.W.; Suhler, E.B.; Fraunfelder, F.T. Hepatitis B vaccine and uveitis: An emerging hypothesis suggested by review of 32 case reports. Cutan. Ocul. Toxicol. 2010, 29, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Esmaeli-Gutstein, B.; Winkelman, J.Z. Uveitis associated with varicella virus vaccine. Am. J. Ophthalmol. 1999, 127, 733–734. [Google Scholar] [CrossRef]
- Benage, M.; Fraunfelder, F.W. Vaccine-Associated Uveitis. Mo. Med. 2016, 113, 48–52. [Google Scholar]
- Aguirre, G.; Carmichael, L.; Bistner, S. Corneal endothelium in viral induced anterior uveitis. Ultrastructural changes following canine adenovirus type 1 infection. Arch. Ophthalmol. 1975, 93, 219–224. [Google Scholar] [CrossRef]
- Sawant, O.B.; Singh, S.; Wright, R.E., 3rd; Jones, K.M.; Titus, M.S.; Dennis, E.; Hicks, E.; Majmudar, P.A.; Kumar, A.; Mian, S.I. Prevalence of SARS-CoV-2 in human post-mortem ocular tissues. Ocul. Surf. 2021, 19, 322–329. [Google Scholar] [CrossRef]
- Koo, E.H.; Eghrari, A.O.; Dzhaber, D.; Shah, A.; Fout, E.; Dubovy, S.; Maestre-Mesa, J.; Miller, D. Presence of SARS-CoV-2 Viral RNA in Aqueous Humor of Asymptomatic Individuals. Am. J. Ophthalmol. 2021, 230, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Nance, K.D.; Meier, J.L. Modifications in an Emergency: The Role of N1-Methylpseudouridine in COVID-19 Vaccines. ACS Cent. Sci. 2021, 7, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Phylactou, M.; Li, J.O.; Larkin, D.F.P. Characteristics of endothelial corneal transplant rejection following immunisation with SARS-CoV-2 messenger RNA vaccine. Br. J. Ophthalmol. 2021, 105, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Crnej, A.; Khoueir, Z.; Cherfan, G.; Saad, A. Acute corneal endothelial graft rejection following COVID-19 vaccination. J. Fr. Ophtalmol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Wasser, L.M.; Roditi, E.; Zadok, D.; Berkowitz, L.; Weill, Y. Keratoplasty Rejection After the BNT162b2 messenger RNA Vaccine. Cornea 2021, 40, 1070–1072. [Google Scholar] [CrossRef]
- Rallis, K.I.; Ting, D.S.J.; Said, D.G.; Dua, H.S. Corneal graft rejection following COVID-19 vaccine. Eye 2021. [Google Scholar] [CrossRef]
- Abousy, M.; Bohm, K.; Prescott, C.; Bonsack, J.M.; Rowhani-Farid, A.; Eghrari, A.O. Bilateral EK Rejection After COVID-19 Vaccine. Eye Contact Lens 2021, 47, 625–628. [Google Scholar] [CrossRef]
- Nioi, M.; D’Aloja, E.; Fossarello, M.; Napoli, P.E. Dual Corneal-Graft Rejection after mRNA Vaccine (BNT162b2) for COVID-19 during the First Six Months of Follow-Up: Case Report, State of the Art and Ethical Concerns. Vaccines 2021, 9, 1274. [Google Scholar] [CrossRef] [PubMed]
- Parmar, D.P.; Garde, P.V.; Shah, S.M.; Bhole, P.K. Acute graft rejection in a high-risk corneal transplant following COVID-19 vaccination: A case report. Indian J. Ophthalmol. 2021, 69, 3757–3758. [Google Scholar] [CrossRef]
- Ravichandran, S.; Natarajan, R. Corneal graft rejection after COVID-19 vaccination. Indian J. Ophthalmol. 2021, 69, 1953–1954. [Google Scholar] [CrossRef] [PubMed]
- Thiem, U.; Borchhardt, K. Vitamin D in solid organ transplantation with special emphasis on kidney transplantation. Vitam. Horm. 2011, 86, 429–468. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.M.; Shane, E. Vitamin D in organ transplantation. Osteoporos. Int. 2011, 22, 2107–2118. [Google Scholar] [CrossRef]
- Courbebaisse, M.; Souberbielle, J.C.; Thervet, E. Potential nonclassical effects of vitamin D in transplant recipients. Transplantation 2010, 89, 131–137. [Google Scholar] [CrossRef]
- DeLuca, H.F.; Zierold, C. Mechanisms and functions of vitamin D. Nutr. Rev. 1998, 56, S4–S10. [Google Scholar] [CrossRef]
- Lemire, J. 1,25-Dihydroxyvitamin D3—A hormone with immunomodulatory properties. Z. Rheumatol. 2000, 59 (Suppl. 1), 24–27. [Google Scholar] [CrossRef]
- Khan, T.A.; Sidhu, N.; Khan, L.; Sen, S.; Hussain, N.; Tandon, R.; Gupta, N. Bilateral Immune-Mediated Keratolysis after Immunization with SARS-CoV-2 Recombinant Viral Vector Vaccine. Cornea 2021, 40, 1629–1632. [Google Scholar] [CrossRef]
- Steinemann, T.L.; Koffler, B.H.; Jennings, C.D. Corneal allograft rejection following immunization. Am. J. Ophthalmol. 1988, 106, 575–578. [Google Scholar] [CrossRef]
- Vignapiano, R.; Vicchio, L.; Favuzza, E.; Cennamo, M.; Mencucci, R. Corneal Graft Rejection after Yellow Fever Vaccine: A Case Report. Ocul. Immunol. Inflamm. 2021, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Wertheim, M.S.; Keel, M.; Cook, S.D.; Tole, D.M. Corneal transplant rejection following influenza vaccination. Br. J. Ophthalmol. 2006, 90, 925. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Solomon, A.; Frucht-Pery, J. Bilateral simultaneous corneal graft rejection after influenza vaccination. Am. J. Ophthalmol. 1996, 121, 708–709. [Google Scholar] [CrossRef]
- Fabre, J.W.; Milton, A.D.; Spencer, S.; Settaf, A.; Houssin, D. Regulation of alloantigen expression in different tissues. Transpl. Proc. 1987, 19, 45–49. [Google Scholar]
- Steger, B.; Curnow, E.; Cheeseman, R.; Romano, V.; Kaye, A.; Jones, M.; Kaye, S.; National Health Service, B.; Transplant Ocular Tissue Advisory, G.; Contributing, O. Sequential Bilateral Corneal Transplantation and Graft Survival. Am. J. Ophthalmol. 2016, 170, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, A.A.; Pappuru, R.R.; Sarraf, D.; Le, P.P.; McCannel, C.A.; Sobrin, L.; Goldstein, D.A.; Honowitz, S.; Walsh, A.C.; Sadda, S.R.; et al. Acute macular neuroretinopathy: Long-term insights revealed by multimodal imaging. Retina 2012, 32, 1500–1513. [Google Scholar] [CrossRef]
- Bos, P.J.; Deutman, A.F. Acute macular neuroretinopathy. Am. J. Ophthalmol. 1975, 80, 573–584. [Google Scholar] [CrossRef]
- Bhavsar, K.V.; Lin, S.; Rahimy, E.; Joseph, A.; Freund, K.B.; Sarraf, D.; Cunningham, E.T., Jr. Acute macular neuroretinopathy: A comprehensive review of the literature. Surv. Ophthalmol. 2016, 61, 538–565. [Google Scholar] [CrossRef]
- Monson, B.K.; Greenberg, P.B.; Greenberg, E.; Fujimoto, J.G.; Srinivasan, V.J.; Duker, J.S. High-speed, ultra-high-resolution optical coherence tomography of acute macular neuroretinopathy. Br. J. Ophthalmol. 2007, 91, 119–120. [Google Scholar] [CrossRef][Green Version]
- Bohler, A.D.; Strom, M.E.; Sandvig, K.U.; Moe, M.C.; Jorstad, O.K. Acute macular neuroretinopathy following COVID-19 vaccination. Eye 2021, 1–2. [Google Scholar] [CrossRef]
- Mambretti, M.; Huemer, J.; Torregrossa, G.; Ullrich, M.; Findl, O.; Casalino, G. Acute Macular Neuroretinopathy following Coronavirus Disease 2019 Vaccination. Ocul. Immunol. Inflamm. 2021, 29, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Hodge, C. Comment on: Acute macular neuroretinopathy following COVID-19 vaccination. Eye 2021. [Google Scholar] [CrossRef] [PubMed]
- Pichi, F.; Aljneibi, S.; Neri, P.; Hay, S.; Dackiw, C.; Ghazi, N.G. Association of Ocular Adverse Events with Inactivated COVID-19 Vaccination in Patients in Abu Dhabi. JAMA Ophthalmol. 2021, 139, 1131–1135. [Google Scholar] [CrossRef] [PubMed]
- Book, B.A.J.; Schmidt, B.; Foerster, A.M.H. Bilateral Acute Macular Neuroretinopathy after Vaccination against SARS-CoV-2. JAMA Ophthalmol. 2021, 139, e212471. [Google Scholar] [CrossRef]
- Druke, D.; Pleyer, U.; Hoerauf, H.; Feltgen, N.; Bemme, S. Acute macular neuroretinopathy (AMN) following COVID-19 vaccination. Am. J. Ophthalmol. Case Rep. 2021, 24, 101207. [Google Scholar] [CrossRef]
- Valenzuela, D.A.; Groth, S.; Taubenslag, K.J.; Gangaputra, S. Acute macular neuroretinopathy following Pfizer-BioNTech COVID-19 vaccination. Am. J. Ophthalmol. Case Rep. 2021, 24, 101200. [Google Scholar] [CrossRef]
- Virgo, J.; Mohamed, M. Paracentral acute middle maculopathy and acute macular neuroretinopathy following SARS-CoV-2 infection. Eye 2020, 34, 2352–2353. [Google Scholar] [CrossRef] [PubMed]
- Nemiroff, J.; Kuehlewein, L.; Rahimy, E.; Tsui, I.; Doshi, R.; Gaudric, A.; Gorin, M.B.; Sadda, S.; Sarraf, D. Assessing Deep Retinal Capillary Ischemia in Paracentral Acute Middle Maculopathy by Optical Coherence Tomography Angiography. Am. J. Ophthalmol. 2016, 162, 121–132.e121. [Google Scholar] [CrossRef] [PubMed]
- Sarraf, D.; Rahimy, E.; Fawzi, A.A.; Sohn, E.; Barbazetto, I.; Zacks, D.N.; Mittra, R.A.; Klancnik, J.M., Jr.; Mrejen, S.; Goldberg, N.R.; et al. Paracentral acute middle maculopathy: A new variant of acute macular neuroretinopathy associated with retinal capillary ischemia. JAMA Ophthalmol. 2013, 131, 1275–1287. [Google Scholar] [CrossRef]
- Rahimy, E.; Sarraf, D. Paracentral acute middle maculopathy spectral-domain optical coherence tomography feature of deep capillary ischemia. Curr. Opin. Ophthalmol. 2014, 25, 207–212. [Google Scholar] [CrossRef]
- Rahimy, E.; Kuehlewein, L.; Sadda, S.R.; Sarraf, D. Paracentral Acute Middle Maculopathy: What We Knew Then and What We Know Now. Retina 2015, 35, 1921–1930. [Google Scholar] [CrossRef]
- Vinzamuri, S.; Pradeep, T.G.; Kotian, R. Bilateral paracentral acute middle maculopathy and acute macular neuroretinopathy following COVID-19 vaccination. Indian J. Ophthalmol. 2021, 69, 2862–2864. [Google Scholar] [CrossRef]
- Fowler, N.; Mendez Martinez, N.R.; Pallares, B.V.; Maldonado, R.S. Acute-onset central serous retinopathy after immunization with COVID-19 mRNA vaccine. Am. J. Ophthalmol. Case Rep. 2021, 23, 101136. [Google Scholar] [CrossRef]
- Khochtali, S.; Krifa, H.; Zina, S.; Khairallah, M.; Jelliti, B.; Abroug, N.; Khairallah, M. Multimodal Imaging of Acute Foveolitis following COVID-19 Vaccination. Ocul. Immunol. Inflamm. 2021, 1–4. [Google Scholar] [CrossRef]
- Shah, P.; Zaveri, J.S.; Haddock, L.J. Acute Macular Neuroretinopathy following the Administration of an Influenza Vaccination. Ophthalmic. Surg. Lasers Imaging Retin. 2018, 49, e165–e168. [Google Scholar] [CrossRef]
- Liu, J.C.; Nesper, P.L.; Fawzi, A.A.; Gill, M.K. Acute macular neuroretinopathy associated with influenza vaccination with decreased flow at the deep capillary plexus on OCT angiography. Am. J. Ophthalmol. Case Rep. 2018, 10, 96–100. [Google Scholar] [CrossRef]
- Giacuzzo, C.; Eandi, C.M.; Kawasaki, A. Bilateral acute macular neuroretinopathy following COVID-19 infection. Acta Ophthalmol. 2021, 100, 611–612. [Google Scholar] [CrossRef]
- Premkumar, M.; Bhujade, H.; Karki, T.; Chaluvashetty, S.B.; Kaur, H.; Duseja, A.K.; Singh, V. New Portal Vein Thrombosis in Cirrhosis-is the thrombophilia exacerbated due to Vaccine or COVID-19? J. Clin. Exp. Hepatol. 2021. [Google Scholar] [CrossRef]
- John, N.A.; John, J.; Kamble, P.; Singhal, A.; Daulatabad, V.; Vamshidhar, I.S. COVID 19 vaccine in patients of hypercoagulable disorders: A clinical perspective. Horm. Mol. Biol. Clin. Investig. 2021. [Google Scholar] [CrossRef]
- Tomerak, S.; Khan, S.; Almasri, M.; Hussein, R.; Abdelati, A.; Aly, A.; Salameh, M.A.; Saed Aldien, A.; Naveed, H.; Elshazly, M.B.; et al. Systemic inflammation in COVID-19 patients may induce various types of venous and arterial thrombosis: A systematic review. Scand. J. Immunol. 2021, 94, e13097. [Google Scholar] [CrossRef]
- Bialasiewicz, A.A.; Farah-Diab, M.S.; Mebarki, H.T. Central retinal vein occlusion occurring immediately after 2nd dose of mRNA SARS-CoV-2 vaccine. Int. Ophthalmol. 2021, 41, 3889–3892. [Google Scholar] [CrossRef]
- Endo, B.; Bahamon, S.; Martinez-Pulgarin, D.F. Central retinal vein occlusion after mRNA SARS-CoV-2 vaccination: A case report. Indian J. Ophthalmol. 2021, 69, 2865–2866. [Google Scholar] [CrossRef]
- Tanaka, H.; Nagasato, D.; Nakakura, S.; Tanabe, H.; Nagasawa, T.; Wakuda, H.; Imada, Y.; Mitamura, Y.; Tabuchi, H. Exacerbation of branch retinal vein occlusion post SARS-CoV2 vaccination: Case reports. Medicine 2021, 100, e28236. [Google Scholar] [CrossRef]
- Goyal, M.; Murthy, S.I.; Srinivas, Y. Unilateral retinal vein occlusion in a young, healthy male following Sputnik V vaccination. Indian J. Ophthalmol. 2021, 69, 3793–3794. [Google Scholar] [CrossRef]
- Elnahry, A.G.; Asal, Z.B.; Shaikh, N.; Dennett, K.; Abd Elmohsen, M.N.; Elnahry, G.A.; Shehab, A.; Vytopil, M.; Ghaffari, L.; Athappilly, G.K.; et al. Optic neuropathy after COVID-19 vaccination: A report of two cases. Int. J. Neurosci. 2021, 1–7. [Google Scholar] [CrossRef]
- Pawar, N.; Maheshwari, D.; Ravindran, M.; Padmavathy, S. Ophthalmic complications of COVID-19 vaccination. Indian J. Ophthalmol. 2021, 69, 2900–2902. [Google Scholar] [CrossRef]
- Leber, H.M.; Sant′Ana, L.; Konichi da Silva, N.R.; Raio, M.C.; Mazzeo, T.; Endo, C.M.; Nascimento, H.; de Souza, C.E. Acute Thyroiditis and Bilateral Optic Neuritis following SARS-CoV-2 Vaccination with CoronaVac: A Case Report. Ocul. Immunol. Inflamm. 2021, 29, 1–7. [Google Scholar] [CrossRef]
- Barnaba, V. Viruses, hidden self-epitopes and autoimmunity. Immunol. Rev. 1996, 152, 47–66. [Google Scholar] [CrossRef]
- Hull, T.P.; Bates, J.H. Optic neuritis after influenza vaccination. Am. J. Ophthalmol. 1997, 124, 703–704. [Google Scholar] [CrossRef]
- McCoy, L.; Tsunoda, I.; Fujinami, R.S. Multiple sclerosis and virus induced immune responses: Autoimmunity can be primed by molecular mimicry and augmented by bystander activation. Autoimmunity 2006, 39, 9–19. [Google Scholar] [CrossRef]
- Miller, S.D.; Vanderlugt, C.L.; Begolka, W.S.; Pao, W.; Yauch, R.L.; Neville, K.L.; Katz-Levy, Y.; Carrizosa, A.; Kim, B.S. Persistent infection with Theiler’s virus leads to CNS autoimmunity via epitope spreading. Nat. Med. 1997, 3, 1133–1136. [Google Scholar] [CrossRef]
- Nahill, S.R.; Welsh, R.M. High frequency of cross-reactive cytotoxic T lymphocytes elicited during the virus-induced polyclonal cytotoxic T lymphocyte response. J. Exp. Med. 1993, 177, 317–327. [Google Scholar] [CrossRef]
- Reyes-Capo, D.P.; Stevens, S.M.; Cavuoto, K.M. Acute abducens nerve palsy following COVID-19 vaccination. J. AAPOS 2021, 25, 302–303. [Google Scholar] [CrossRef]
- Kawtharani, A.A.; Fakhry, B.; Serhan, A. Longitudinal extensive transverse myelitis with sixth nerve palsy post ChAdOx1 nCov-19 vaccine: A case report and literature review. World J. Adv. Res. Rev. 2021. [Google Scholar]
- Pappaterra, M.C.; Rivera, E.J.; Oliver, A.L. Transient Oculomotor Palsy Following the Administration of the Messenger RNA-1273 Vaccine for SARS-CoV-2 Diplopia Following the COVID-19 Vaccine. J. Neuroophthalmol. 2021. [Google Scholar] [CrossRef]
- Manea, M.M.; Dragos, D.; Enache, I.; Sirbu, A.G.; Tuta, S. Multiple cranial nerve palsies following COVID-19 vaccination-Case report. Acta Neurol. Scand. 2021, 145, 257–259. [Google Scholar] [CrossRef]
- Essrani, R.; Essrani, R.K.; Mehershahi, S.; Lohana, A.K.; Sudhakaran, A. Oculomotor Nerve Palsy after Influenza Vaccine in Inflammatory Bowel Disease. Cureus 2018, 10, e3759. [Google Scholar] [CrossRef]
- Roger, H.; Poursines, Y.; Roger, J.; Pujol, R. Guillain-Barre syndrome with paralysis of the two external oculomotor muscles after smallpox vaccination. Rev. Otoneuroophtalmol. 1953, 25, 425–427. [Google Scholar]
- Larmande, A.; Margaillan, A. Oculomotor paralysis & jennerian vaccination. Rev. Otoneuroophtalmol. 1956, 28, 431. [Google Scholar]
- Kim, B.Y.; Yung, Y.; Kim, G.S.; Park, H.R.; Lee, J.J.; Song, P.; Cho, J.Y. Complete Oculomotor Palsy after Influenza Vaccination in a Young Healthy Adult: A Case Report. Case Rep. Neurol. 2021, 13, 35–39. [Google Scholar] [CrossRef]
- De Almeida, D.F.; Teodoro, A.T.; Radaeli Rde, F. Transient oculomotor palsy after influenza vaccination: Short report. ISRN Neurol. 2011, 2011, 849757. [Google Scholar] [CrossRef]
- Grewal, D.S.; Zeid, J.L. Isolated abducens nerve palsy following neonatal hepatitis B vaccination. J. AAPOS 2014, 18, 75–76. [Google Scholar] [CrossRef]
- Bourtoulamaiou, A.; Yadav, S.; Nayak, H. Benign Recurrent Sixth (Abducens) Nerve Palsy following Measles-Mumps-Rubella Vaccination. Case Rep. Pediatr. 2015, 2015, 734516. [Google Scholar] [CrossRef]
- Pereima, R.R.; Bonatti, R.; Crotti, F.; Furtado, J.M.; Lopes, M.H.; Yamamoto, J.H.; Kreuz, A.C. Ocular Adverse Events following Yellow Fever Vaccination: A Case Series. Ocul. Immunol. Inflamm. 2021, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Jumroendararasame, C.; Panyakorn, S.; Othong, R.; Jumroendararasame, A.; Srimanan, W.; Tipparut, K. Transient visual field loss after COVID-19 vaccination: Experienced by ophthalmologist, case report. Am. J. Ophthalmol. Case Rep. 2021, 24, 101212. [Google Scholar] [CrossRef] [PubMed]
- Santovito, L.S.; Pinna, G. Acute reduction of visual acuity and visual field after Pfizer-BioNTech COVID-19 vaccine 2nd dose: A case report. Inflamm. Res. 2021, 70, 931–933. [Google Scholar] [CrossRef] [PubMed]
| Author | Age | Sex | Vaccine | Time from Vaccine to Symptom (days) | Presenting VA | Side | Manifestations |
|---|---|---|---|---|---|---|---|
| Eyelid | |||||||
| Austria et al., 2021 | 32 | F | BNT162b2, #NR | 1 to 2 | NR | NR | Unilateral upper greater than lower eyelid edema and erythema without other systemic or ocular findings on exam. |
| 43 | F | BNT162b2, #NR | |||||
| 43 | F | BNT162b2, #NR | |||||
| Mazzatena et al., 2021 | 67 | F | BNT162b2, #1 | 10 | NR | OD and OS | Ecchymotic lesions on the upper eyelids. Lesions were moderately itchy. |
| 44 | F | BNT162b2, #2 | 21 | NR | OD and OS | Purpuric lesions bilaterally. Lesions were circumscribed on the upper eyelid and totally asymptomatic. | |
| 63 | M | BNT162b2, #2 | 21 | NR | OD and OS | Purpuric lesions bilaterally. Lesions were circumscribed on the upper eyelid and totally asymptomatic. | |
| Orbit | |||||||
| Bayas et al., 2021 | 55 | F | AZD1222, #1 | 10 | 20/140 | OD | Bilateral conjunctival congestion, retroorbital pain, and diplopia. MRI showed bilateral superior ophthalmic vein thrombosis. |
| 20/140 | OS | ||||||
| Chuang et al., 2021 | 45 | M | NR | 7 | NR | OS | Progressive ptosis and decreased vision OS, diplopia, and examination with APD and complete ophthalmoplegia. CT and MRI with left cavernous sinus thrombosis. Pt diagnosed with Tolosa-Hunt syndrome. |
| Panovska-Stavridis et al., 2021 | 29 | F | AZD1222, #1 | 10 | NR | OS | Left orbital swelling, severe headache, and blurred vision OS. Labs showed thrombocytopenia of 18 × 1019/L. MRI demonstrated central filling defects and a diagnosis of superior ophthalmic vein thrombosis was made. |
| Cornea | |||||||
| De la Presa et al., 2021 | 27 | F | Moderna Vaccine, #1 | 15 | 20/20 | OD | Redness and irritation with 1+ conjunctival hyperemia and an irregular temporal epithelial rejection line in a patient post LR-CLAL 4 years earlier. A diagnosis of acute unilateral graft rejection was made. |
| Abousy et al., 2021 | 73 | F | BNT162b2, #2 | 4 | 20/200 | OD | Vision loss with corneal thickening with Descemet folds bilaterally in a patient with DSEK 8 years previously, consistent with acute bilateral graft rejection. |
| 20/40 | OS | ||||||
| Crnej et al., 2021 | 71 | M | BNT162b2, #1 | 7 | 20/125 | OD | Painless decrease in right eye vision with conjunctival injection and diffuse corneal edema 5 months post-DMEK, diagnosed as acute unilateral graft rejection. |
| Khan et al., 2021 | 48 | M | AZD1222, #1 | 21 | LP | OD | Vision loss, bilateral lid edema, diffuse conjunctival and ciliary congestion, corneal melting and perforation with diffuse corneal haze, uveal tissue prolapse, bilateral massive choroidal detachment on B-scan ultrasonography. |
| LP | OS | ||||||
| Nioi et al., 2021 | 44 | F | BNT162b2, #1 | 13 | CF | OS | Blurry vision, eye redness and discomfort OS. Examination with ciliary injection, diffuse corneal edema, keratic precipitates, Descemet folds, anterior chamber cells, consistent with acute unilateral graft rejection. |
| Papasavvas et al., 2021 | 69 | F | BNT162b2, #1 | 10 | 20/30 | OD | Excruciating pain in the left V1 dermatome with a small dendrite in the supero-temporal cornea. Diagnosis of HZO was made. |
| 73 | F | BNT162b2, #3 | 16 | 20/40 | OD | Excruciating pain in the right V1 dermatome without dendrite formation. Vitreous cells present. Diagnosis of HZO was made. | |
| 72 | F | Moderna Vaccine, #1 | 13 | 20/63 | OS | Excruciating pain in the left V1 dermatome with conjunctival chemosis but no corneal or AC changes. 10 days later with AC uveitis with cell, flare, KP, and Descemet folds. Diagnosis of HZO was made. | |
| Parmar et al., 2021 | 35 | M | AZD1222, #1 | 2 | CF | OS | Decreased vision in a patient post-repeat PKP 6 months previously after original PKP 3 years earlier. Exam with graft edema more prominent in the lower half as well as KPs and AC reaction. Diagnosis of acute unilateral graft rejection was made. |
| Phylactou et al. 2021 | 66 | F | BNT162b2, #1 | 7 | 20/125 | OD | Acute-onset right eye blurred vision, redness, and photophobia with conjunctival injection, diffuse corneal edema, fine KP, 1+ AC cells 21 days post-DMEK, diagnosed as acute unilateral graft rejection. |
| 83 | F | BNT162b2, #2 | 21 | 20/80 | OD | Acute-onset bilateral blurred vision, pain, photophobia and red with bilateral circumcorneal injection, KP, and AC inflammation, 6 (OD) and 3 (OS) years post-DMEK, diagnosed as acute bilateral graft rejection. | |
| 20/40 | OS | ||||||
| Rallis et al., 2021 | 68 | F | BNT162b2, #1 | 3 | CF | OS | Vision loss OS with conjunctival hyperemia, diffuse corneal punctate staining and graft edema, and KP 3 months post-redo PKP for failed DSAEK, diagnosed as acute unilateral graft rejection. Pre-existing OD graft was intact. |
| Ravichandran and Natarajan 2021 | 62 | M | AZD1222, #1 | 21 | NR | NR | Right eye decreased vision and congestion, with an advancing Kodadoust rejection line and corneal graft edema, 2 years post-PKP. Diagnosed as acute unilateral graft rejection. |
| Wasser et al., 2021 | 73 | M | BNT162b2, #1 | 13 | 20/200 | OS | Eye discomfort OS and vision loss with ciliary injection, corneal edema, Descemet folds, and KP 2 years after re-graft for PKP performed 44 years earlier. Diagnosed as acute unilateral graft rejection. |
| 56 | M | BNT162b2, #1 | 12 | CF | OD | Blurred vision and redness OD, with diffuse corneal edema, KP, and AC cells 25 years post-PKP, diagnosed as acute unilateral graft rejection. Pre-existing OS graft from PKP 7 years earlier was intact. | |
| Uveitis | |||||||
| ElSheikh et al., 2021 | 18 | F | Sinopharm, #2 | 5 | 20/40 | OD | Bilateral acute uveitis with 2+ AC flare OU and 1+ cell OU and hyperreflective dots in the AC in a patient with juvenile idiopathic arthritis. |
| 20/120 | OS | ||||||
| Goyal et al., 2021 | 34 | M | AZD1222, #1 | 4 | 20/120 | OD | Ocular pain followed by nasal redness OS and a floater OD progressing to severe vision loss. Fundus exam with multiple bilateral oval lesions at the level of the choroid with serous detachments, consistent with bilateral multifocal choroiditis. |
| 20/20 | OS | ||||||
| Herbort and Papasavvas 2021 | 53 | M | Moderna Vaccine, #2 | 5 | NR | OD | Severe flare-up of pre-existing herpes-keratouveitis OD inactive for 18 months without treatment. Pt presented with numerous KPs, elevated IOP to 41 mmHg. |
| Ishay et al., 2021 | 28 | M | BNT162b2, #1 | 10 | NR | OS | Pain, redness, and blurred vision OS in a patient with Behçet’s disease on colchicine twice daily. Examination revealed severe panuveitis. |
| Jain and Kalamkar 2021 | 27 | M | AZD1222, #1 | 2 | 20/20 | OS | Pain, redness and severe circumcorneal congestion OS with 2+ AC cells and non-granulomatous KP in a patient with juvenile idiopathic arthritis and one previous episode of bilateral uveitis. Acute uveitis was diagnosed. |
| Koong et al., 2021 | 54 | M | BNT162b2, #1 | 1 | 20/80 | OD | Acute bilateral, sequential blurring of vision with bilateral areas of subretinal fluid with dot-blot hemorrhages on examination. OCT with bilateral serous neurosensory retinal detachments. ICGA confirmed diagnosis of VKH. |
| 20/160 | OS | ||||||
| Maleki et al., 2021 | 33 | F | Moderna Vaccine, #2 | 10 | 20/20 | OD | Bilateral photopsia and progressive nasal field defect OS. OCT with outer layer segmental disruption OS. Elevated ESR and CRP. Diagnosis of acute zonal occult outer retinopathy (AZOOR) was made. |
| 20/20 | OS | ||||||
| Mishra et al., 2021 | 71 | M | AZD1222, #1 | 10 | CF | OD | Reactivation of VZV presenting with panuveitis OD, circumcorneal congestion, multiple fine keratic precipitates, anterior chamber cells and flare, vitritis, and widespread acute retinal necrosis. |
| Mudie et al., 2021 | 43 | F | BNT162b2, #2 | 3 | 20/500 | OD | Bilateral substantial vision loss, eye pain and redness, and photophobia, with 3–4+ AC cell and 2–3+ vitreous cell. OCT with significant choroidal thicnening, FA with mild peripheral vascular leakage. Diagnosis of panuveitis was made. |
| 20/500 | OS | ||||||
| Pan et al., 2021 | 50 | F | Unspecified inactivated Vero cell-based vaccine approved in China | 5 | 20/33 | OD | Bilateral blurred vision with pale, blurry optic disc, absent foveal reflex, macular edema, and fluorescein angiography consistent with bilateral choroiditis. |
| 20/66 | OS | ||||||
| Papasavvas and Herbort 2021 | 43 | F | BNT162b2, #2 | 42 | 20/20 | OD | Reactivation of pre-existing VKH disease with significant anterior segment inflammation OU, and 3–4 mutton-fat KP OD. OCT showed retinal folds and subretinal fluid. Multiple hypofluorescent dark dots present on ICGA. |
| 20/20 | OS | ||||||
| Rabinovitch et al., 2021 | 43 | F | BNT162b2, #1 | 2 | 20/25 | OD | Redness, pain, blurred vision. 3+ cell and 1+ flare and fibrin on exam. Diagnosis of anterior uveitis was made. |
| 34 | M | BNT162b2, #1 | 4 | 20/32 | OD | Redness and pain. 1+ cell and non-granulomatous KPs on exam. Diagnosis of anterior uveitis was made. | |
| 34 | F | BNT162b2, #1 | 1 | 20/50 | OS | Redness, pain, and photophobia. 2+ cell and non-granulomatous KPs on exam. Diagnosis of anterior uveitis was made. | |
| 53 | M | BNT162b2, #1 | 13 | 20/25 | OS | Pain only. 0.5+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 64 | M | BNT162b2, #1 | 15 | 20/25 | OS | Redness, pain, and photophobia. 0.5+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 68 | M | BNT162b2, #1 | 5 | 20/200 | OD | Redness and pain. 1+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 61 | F | BNT162b2, #1 | 12 | 20/25 | OD | Pain and photophobia. 2+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 65 | F | BNT162b2, #1 | 3 | 20/80 | OD | Redness, pain, photophobia, and blurred vision. 2+ cell and 2+ flare on exam. Diagnosis of anterior uveitis was made. | |
| 78 | M | BNT162b2, #2 | 3 | 20/25 | OS | Redness, pain, and blurred vision. 2+ cell and 2+ flare with posterior synechiae on exam. Diagnosis of anterior uveitis was made. | |
| 59 | M | BNT162b2, #2 | 8 | 20/32 | OS | Pain, photophobia, and blurred vision. 2+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 72 | M | BNT162b2, #2 | 16 | 20/80 | OD | Redness only. 1+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 51 | M | BNT162b2, #2 | 2 | 20/50 | OS | Redness and pain. 2+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 42 | F | BNT162b2, #2 | 20 | 20/25 | OD | Pain and blurred vision bilaterally. 2+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 20/25 | OS | ||||||
| 74 | M | BNT162b2, #2 | 7 | 20/40 OD | OD | Pain only. 1+ cell and 2+ flare on exam. Diagnosis of anterior uveitis was made. | |
| 39 | M | BNT162b2, #2 | 5 | 20/32 | OD | Blurred vision with defect and photopsia. Outer retinal changes on exam. Diagnosis of MEWDS was made. | |
| 64 | F | BNT162b2, #2 | 6 | 20/25 | OD | Photophobia only. 1+ flare on exam. Diagnosis of anterior uveitis was made. | |
| 50 | F | BNT162b2, #2 | 2 | 20/25 | OS | Pain only. 1+ cell on exam. Diagnosis of anterior uveitis was made. | |
| 23 | F | BNT162b2, #2 | 2 | 20/25 | OD | Redness, blurred vision, and photophobia bilaterally. 1+ cell and 1+ flare on exam. Diagnosis of anterior uveitis was made. | |
| 20/25 | OS | ||||||
| 36 | M | BNT162b2, #2 | 1 | 20/80 | OS | Redness, photophobia, and blurred vision. 3+ cell and 3+ flare with non-granulomatous KPs on exam. Diagnosis of anterior uveitis was made. | |
| 41 | M | BNT162b2, #2 | 2 | 20/50 | OD | Redness, photophobia, and blurred vision. 2+ cell and 2+ flare on exam. Diagnosis of anterior uveitis was made. | |
| 28 | F | BNT162b2, #2 | 30 | 20/32 | OS | Blurred vision, visual field defect, and photopsia. Outer retinal changes on exam. Diagnosis of MEWDS was made. | |
| Renisi et al., 2021 | 23 | M | BNT162b2, #2 | 14 | 20/40 | OS | Pain and photophobia OS with perikeratic and conjunctival hyperemia, posterior synechiae, AC cells, and KP. Diagnosis of anterior uveitis was made. |
| Saraceno et al., 2021 | 62 | F | AZD1222, #1 | 2 | 20/600 | OD | Acute bilateral loss of vision with mild 2+ AC cell and 1+ vitreous cell OU. Fundus examination revealed a serous retinal detachment OU. OCT revealed the same and subretinal hyperreflective dots. Diagnosis of VKH was made. |
| 20/200 | OS | ||||||
| Retina | |||||||
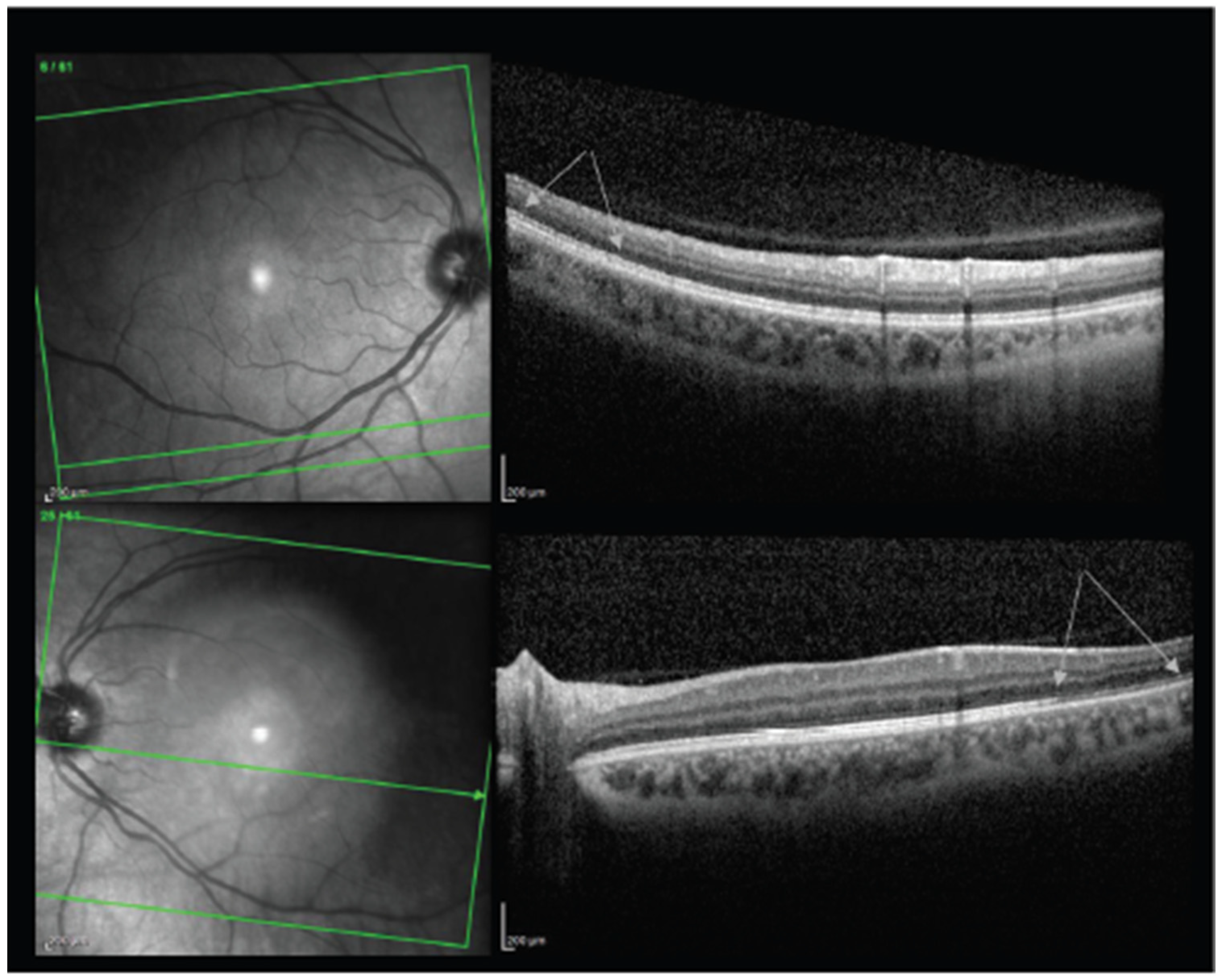
| Bøhler et al., 2021 | 27 | F | AZD1222, #1 | 2 | 20/20 | OS | Left eye paracentral scotoma with a teardrop-shaped macular lesion nasal to the fovea on ophthalmoscopy, diagnosed as unilateral AMN. |
| Book et al., 2021 | 21 | F | AZD1222, #1 | 3 | 20/16 | OD | Bilateral paracentral scotomas with underlying circumscribed paracentral dark lesions on exam, OCT with outer plexiform layer thickening and discontinuity, diagnosed as bilateral AMN. |
| 20/16 | OS | ||||||
| Chen et al., 2021 | 21 | F | BNT162b2, #1 | 3 | 20/20 | OS | Paracentral scotomas OS with barely visible oval parafoveal lesions on fundus exam. Infrared imaging revealed hypo-reflective lesions consistent with left AMN. |
| Drüke et al., 2021 | 23 | F | AZD1222, #1 | 1 | 20/20 | OD | Development of bilateral paracentral scotomas. Fundus photography revealed a subtle brownish rimmed lesion parafoveally OD and blurred lesion nasal to the macula OS. IR and OCT imaging confirmed a diagnosis of AMN. |
| 20/20 | OS | ||||||
| Fowler et al., 2021 | 33 | M | BNT162b2, #1 | 3 | 20/63 | OD | Blurry vision OD with swollen macula, central foveal thickness (CFT) of 457 μm on OCT, and macular serous detachment of the neurosensory retina on FA. OCTA confirmed a diagnosis of central serous retinopathy. |
| Khochtali et al., 2021 | 24 | F | BNT162b2, #1 | 5 | 20/40 | OS | Foveolitis with 2+ vitreous cell, diffuse retinal vascular leakage, faint foveal hyperfluorescence and late phase hypofluorescence of the foveal lesion, and granular hyperreflective specks in the inner nuclear layer. |
| Mambretti et al., 2021 | 22 | F | AZD1222, #1 | 2 | 20/20 | OD | Acute paracentral scotoma OD with barely visible parafoveal lesions on fundus exam. OCT was consistent with AMN. |
| 28 | F | AZD1222, #1 | 2 | 20/20 | OD | Acute paracentral scotoma OD with OCT consistent with AMN. | |
| Michel et al., 2021 | 21 | F | AZD1222, #1 | 2 | 20/20 | OS | Acute-onset of 4 central scotomas OS, well-demarcated dark oval-shaped areas surrounding the left fovea on infrared imaging. OCT with multifocal highly reflective lesions and with ellipsoid and interdigitation zone disruption consistent with AMN. |
| Pichi et al., 2021 | NR | NR | Sinopharm, #NR | 5 | 20/400 | OS | Acute vision loss OS, with OCT showing hyperreflectivity of the outer plexiform, Henle fiber, and outer nuclear layers. A diagnosis of AMN was made. |
| NR | NR | Sinopharm, #NR | 0 | 20/30 | OS | Tachycardia, systolic hypertension (210 mm Hg), and inferior scotoma OS 20 min after vaccination. Fundus examination revealed a suprafoveal dot hemorrhage. A diagnosis of PAMM was made. | |
| Subramony et al., 2021 | 22 | F | Moderna Vaccine, #2 | 10 | 20/70 | OD | Progressive painless vision loss OD and no vision changes OS, but macula-off inferotemporal retinal detachment OD and small macula-on temporal retinal detachment OS. |
| 20/20 | OS | ||||||
| Valenzuela et al., 2021 | 20 | F | BNT162b2, #2 | 2 | 20/20 | OD | Development of bilateral paracentral scotomas and shimmering lights. Fundus exam was unrevealing, but OCT demonstrated corresponding parafoveal foci of hyperreflectivity. Diagnosis of AMN was made. |
| 20/20 | OS | ||||||
| Vinzamuri et al., 2021 | 35 | M | AZD1222, #2 | NR | 20/20 | OD | Visual disturbance, OCT with hyperreflective lesions involving the nerve fiber layer, ganglion cell layer and outer plexiform layer; diagnosed as PAMM and AMN. |
| 20/20 | OS | ||||||
| Vascular | |||||||
| Bialasiewicz et al., 2021 | 50 | M | BNT162b2, #2 | 0 | CF | OD | Immediate bilateral retrobulbar pain, red eye, and vision loss. Examination and OCT revealed a hemorrhagic CRVO with ischemic areas and cystoid macular edema. |
| CF | OS | ||||||
| Endo et al., 2021 | 52 | M | BNT162b2, #1 | 14 | 20/20 | OS | Sudden blurred vision OS with minimal dot hemorrhages in the upper quadrants, dilated tortuous veins in four quadrants, and disperse exudates. FA was consistent with non-ischemic CRVO. |
| Goyal et al., 2021 | 28 | M | Sputnik V, #2 | 11 | 20/30 | OD | Visual disturbance with fundus examination revealing superior hemi-retinal vein occlusion with severe cystoid macular edema. |
| Tanaka et al., 2021 | 71 | F | BNT162b2, #2 | 1 | 20/30 | OS | Vision loss, with examination and OCT showing superior temporal BRVO and secondary macular edema with previously resolved inferior temporal BRVO. |
| 72 | M | BNT162b2, #1 | 1 | 20/25 | OD | Vision loss, with examination and OCT showing recurrence of previously resolved superior temporal BRVO and macular edema. | |
| Neuro-Ophthalmology | |||||||
| Elnahry et al., 2021 | 69 | F | BNT162b2, #2 | 16 | CF | OD | Blurry vision OU with immediate OS clearing but persistent blurring OD. Examination with optic nerve head edema (OD > OS) and RAPD OD on exam. RNFL imaging confirmed a diagnosis of central nervous system inflammatory syndrome with neuroretinitis. |
| 20/20 | OS | ||||||
| 32 | F | AZD1222, #1 | 4 | 20/30 | OS | Blurred vision with superior field defect OS. Examination revealed left optic disc swelling and RAPD with decreased RNFL thickness. MRI was diagnostic of left optic neuritis. | |
| Leber et al., 2021 | 32 | F | Corona Vac, #2 | 0 | 20/200 | OS | Rapidly progressive worsening vision and pain with EOM OS. Examination revealed RAPD OS and disc swelling OD and OS. Labs revealed thyroiditis and MRI revealed bilateral optic neuritis. |
| 20/20 | OD | ||||||
| Maleki et al., 2021 | 79 | F | BNT162b2, #2 | 2 | 20/1250 | OD | Bilateral sudden loss of vision, OD > OS, with 3+ afferent pupillary defect OD. OCT, FA, and ICG consistent with generalized disc pallor OD and inferior pallor OS, consistent with bilateral arteritic anterior ischemic optic neuropathy (AAION). |
| 20/40 | OS | ||||||
| Pawar et al., 2021 | 28 | F | NR | 21 | 20/120 | OS | Sudden vision loss OS, with examination revealing mild blurring of the optic disc margin. MRI was consistent with optic neuritis. |
| Ocular Motility | |||||||
| Eleiwa et al., 2021 | 46 | M | AZD1222, #2 | 3 | NR | OD | Torsional, binocular diplopia. A diagnosis of right trochlear (4th cranial) nerve palsy was made. |
| Kawtharani et al., 2021 | 37 | F | AZD1222, #1 | NR | NR | OS | Left eye esotropia diagnosed as abducens (6th cranial) nerve palsy. |
| Manea et al., 2021 | 29 | M | BNT162b2, #1 | 6 | NR | OS | Multiple cranial neuropathies, namely incomplete oculomotor (3rd cranial), abducens (6th cranial), and facial (7th cranial) nerve palsy. |
| Pawar et al., 2021 | 23 | M | NR | 6 | NR | OS | Acute esotropia OS in a patient with previous recurrent abducens (6th cranial) nerve palsy following chickenpox. Normal fundus examination and MRI. |
| 24 | F | NR | 21 | NR | OD | Diplopia and squinting bilaterally, with examination revealing restricted elevation of both eyes. MRI and neurological examination were otherwise normal. Pt was diagnosed with bilateral vertical gaze palsy. | |
| NR | OS | ||||||
| 44 | M | NR | 28 | NR | OS | Acute abducens (6th cranial) nerve palsy OS. Normal fundus examination and MRI otherwise. | |
| Pappaterra et al., 2021 | 81 | M | Moderna Vaccine, #1 | 1 | 20/30 | OS | Acute bilateral oblique diplopia. Examination revealed limited adduction and infraduction OS only. Diagnosis of oculomotor (3rd cranial) nerve palsy was made. |
| Pereira et al., 2021 | 65 | M | AZD1222, #NR | 3 | 20/20 | OD | Sudden-onset painless binocular diplopia, with examination revealing esotropia OD of 12 PD and severe abduction deficit. Diagnosis of right abducens (6th cranial) nerve palsy was made. |
| Reyes-Capo et al., 2021 | 59 | F | BNT162b2, #1 | 2 | 20/25 | OD | Acute binocular diplopia and painless, horizontal diplopia, and new right esotropia and abduction deficits OD only. Pt was diagnosed with abducens (6th cranial) nerve palsy. |
| Other | |||||||
| Pichi et al., 2021 | NR | NR | NR | 7 | 20/20 | OD | Bilateral eye redness and pain, with examination demonstrating significant scleral hyperemia with positive phenylephrine test results. No AC cell or flare was present. A diagnosis of scleritis was made. |
| 20/20 | OS | ||||||
| Santovito and Pinna 2021 | NR | M | BNT162b2, #2 | NR | NR | OD | Sudden darkening of visual field and reduction of visual acuity preceded hours earlier by unilateral headache and succeeded by confusion and nausea. |
| NR | OS | ||||||
| Jumroendararasame et al., 2021 | 42 | M | Corona Vac, #2 | 0 | 20/20 | OD | Immediate blurred vision centrally followed by obscuring of the left visual field. Examination and OCT imaging were unremarkable. Authors proposed acute vasospasm as the underlying cause. |
| 20/20 | OS | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haseeb, A.A.; Solyman, O.; Abushanab, M.M.; Abo Obaia, A.S.; Elhusseiny, A.M. Ocular Complications Following Vaccination for COVID-19: A One-Year Retrospective. Vaccines 2022, 10, 342. https://doi.org/10.3390/vaccines10020342
Haseeb AA, Solyman O, Abushanab MM, Abo Obaia AS, Elhusseiny AM. Ocular Complications Following Vaccination for COVID-19: A One-Year Retrospective. Vaccines. 2022; 10(2):342. https://doi.org/10.3390/vaccines10020342
Chicago/Turabian StyleHaseeb, Abid A., Omar Solyman, Mokhtar M. Abushanab, Ahmed S. Abo Obaia, and Abdelrahman M. Elhusseiny. 2022. "Ocular Complications Following Vaccination for COVID-19: A One-Year Retrospective" Vaccines 10, no. 2: 342. https://doi.org/10.3390/vaccines10020342
APA StyleHaseeb, A. A., Solyman, O., Abushanab, M. M., Abo Obaia, A. S., & Elhusseiny, A. M. (2022). Ocular Complications Following Vaccination for COVID-19: A One-Year Retrospective. Vaccines, 10(2), 342. https://doi.org/10.3390/vaccines10020342






