1. Introduction
Infection in humans can occur through the ingestion of undercooked meat and contaminated water, resulting in severe symptoms depending on the host’s immune status. In the case of immunocompromised individuals, pregnant women, and the elderly, it can cause congenital toxoplasmosis, resulting in hydrocephalus and microcephaly, and in severe cases miscarriage [
1]. The current treatment options for
T. gondii infection involve co-administration of pyrimethamine and sulfadiazine. Other drugs such as hydroxynaphthoquinone (atovaquone) and azithromycin could also be used, but there are major issues with these drugs. Pathogen drug resistance is a recurring theme frequently reported from various diseases and toxoplasmosis is no exception. This and the severe side effects, poor patient tolerability, and therapeutic ineffectiveness against bradyzoites of
T. gondii are several of the obstacles that must be surmounted [
1]. Vaccines are widely considered to be the most cost-effective healthcare intervention, and successful vaccines can benefit public health, which ultimately translates to economic growth [
2]. For these reasons, developing a vaccine to protect against toxoplasmosis is an urgent need.
Vaccinia virus has been used extensively as a vaccine platform against numerous diseases such as smallpox. Currently, genetically modified vaccinia virus-based vaccines such as the modified vaccinia Ankara (MVA) are frequently used in heterologous prime-boost immunization strategies and have proven to be highly effective. To date, only a few studies have investigated the vaccine efficacies of recombinant vaccinia virus against toxoplasmosis. Roque-Resndiz et al. [
3] reported that ROP2-expressing MVA vaccines were capable of inducing
T. gondii-specific antibodies and that their immunization prolonged the survival of mice upon challenge infection with the RH strain. Heterologous immunization strategies involving MVA vaccines were also successful. In comparison to homologous immunization, heterologously immunizing GRA4-expressing DNA and MVA vaccines elicited greater brain cyst reduction and promoted longer survival of immunized mice after challenge infecting 20,000 PLK strains [
4]. Similar findings were observed in another comparative study. Heterologously immunizing mice with SAG1-expressing adenovirus and MVA vaccines induced greater protection than homologous adenovirus-vectored vaccines [
5].
The rhoptry proteins of
T. gondii are parasitic components required for proliferation and host cell invasion [
6,
7]. Several studies have assessed the efficacy of ROP4 antigens using various vaccine platforms such as virus-like particles (VLPs) [
8] or subunit vaccines [
9]. Furthermore, we have recently investigated the efficacy of ROP4-expressing recombinant baculovirus (rBV) vaccines and the resulting immune responses in mice [
10]. Nevertheless, there are inherent problems with the baculovirus-based vaccines. First and foremost, repeated vaccination with the baculovirus vaccines can result in the development of baculovirus-specific antibodies in hosts and subsequent neutralization of the baculovirus upon re-entry [
11,
12]. Another issue is the rapid degradation of baculoviral particles by the host complement system, thus limiting their application as vaccines [
12,
13]. To address these limitations of our previous works, we generated a ROP4-expressing recombinant vaccinia virus (rVV) vaccine as not a single study has investigated their efficacy in animal models. Orally administered vaccines are highly sought after due to their mucosal and systemic immunity-inducing effects [
14]. Given that
T. gondii is orally acquired in hosts, mucosal immunity is of importance since the intestinal mucosa acts as the initial site of infection for
T. gondii and many other pathogens. Here, we investigated the resulting mucosal immunity induced by the ROP4-rVVs against
T. gondii infection, which has yet to be reported. Our findings demonstrated that the ROP4-rVV vaccines elicited a robust cellular and humoral response that protected mice upon lethal
T. gondii infection.
2. Materials and Methods
2.1. Animals and Ethics
Six-week-old female BALB/c mice purchased from NARA Biotech (Seoul, Korea) were used for this study. The animals were housed in a specific-pathogen-free facility with easy access to food and water. All animal experiment protocols were approved and performed in accordance with the guidelines of Kyung Hee University IACUC (permit number: KHUASP (SE) 20-648).
2.2. Parasite, Cells, and Antibodies
Parasites and cells used in this study were maintained as described earlier (Kang et al., 2020b). T. gondii ME49 strain was maintained by serial passaging in BALB/c mice. Mice were sacrificed and ME49 cysts were purified from the brain tissues of infected mice. CV-1 and Vero cells were cultured in T175 flasks with Dulbecco’s Modified Eagle’s Medium (ThermoFisher, Waltham, MA, USA) supplemented with 1% penicillin/streptomycin and 10% fetal bovine serum (FBS) at 37 °C. Horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG and IgA secondary antibodies were purchased from Southern Biotech (Birmingham, AL, USA).
2.3. Generation of Recombinant Vaccinia Virus (rVV)
The rhoptry protein 4 (ROP4) gene was amplified by polymerase chain reaction (PCR) and cloned into pRB21 vectors with StuI/HindIII restriction enzymes [
15]. Prior to transfection with the recombinant pRB21 plasmid containing the ROP4 gene using Cellfectine II reagent (Invitrogen, Waltham, MA, USA), CV-1 cells were infected with the nonplaque-forming vRB12 mutant as previously described [
16]. Infected cells were harvested with a scraper once morphological changes and other cytopathic effects were detected from approximately 70% of the seeded cells and centrifuged to pellet the cells. Supernatants were aspirated and cells were subjected to repeated freeze–thaw cycles that involved freezing at −80 °C in a deep freezer for 30 min immediately followed by thawing at 37 °C in a water bath a total of 3 times. After this procedure, cells were sonicated and centrifuged at 6000 rpm, 20 min, 4 °C. Supernatants were removed and pellets were resuspended in fresh DMEM media. To further purify the rVVs, resuspended rVV-containing cell pellets were carefully stacked on top of 36% sucrose gradient. After centrifugation at 20,000 rpm, 30 min, 4 °C, a distinct clear band was observed and this fraction was carefully collected. After resuspending the band fraction containing the rVV in 0.1 M PBS, protein concentration was determined using the QuantiPro BCA Assay Kit (Sigma-Aldrich, St Louis, MA, USA).
2.4. Quantification of rVVs by Plaque Assay
Vero cells were used to quantify rVV titers. In brief, confluent monolayers of Vero cells were cultured in 12-well plates in DMEM media supplemented with 1% penicillin/streptomycin and 10% FBS. Upon reaching 90–100% confluence, cells were infected with the serially diluted rVVs (102, 103, 104 dilutions in serum-free media) and incubated at 37 °C, 5% CO2 for 1 h. Virus inoculum was removed after 1 h and overlay media (1% noble agar and 2% FBS in DMEM media) were gently added into each well. Plates were incubated at 37 °C for 4 days and enlarged viral plaques were counted to determine rVV titers.
2.5. Immunization and Challenge Infection
A total of 18 mice were subdivided into 3 groups (n = 6 per group): unimmunized (Naïve), unimmunized mice that were challenge-infected (Naïve+Cha), and rVV-immunized mice. Mice were primed and boost-immunized with 5 × 103 pfu/100 μL of ROP4-rVVs through the oral route by oral gavage at 4-week intervals. Four weeks after the boost immunization, mice were orally challenge-infected with a 50 LD50 dose (2000 cysts) of ME49 cysts. Mice were monitored daily to assess changes in bodyweight and survival rates. Mice that lost more than 20% of their initial bodyweight were humanely euthanized. At 16 days post-infection (dpi), all the mice were sacrificed for sample collection and immunological assays were performed.
2.6. Sample Collection and Preparation
Samples were collected as previously described [
10]. Four weeks after each immunization, blood samples of mice were collected by retro-orbital plexus puncture. Sera were separated from the blood via centrifugation at 5000 RPM for 10 min. Sera were diluted (1:100 dilution in PBS) and used as primary antibodies for ELISA. After sacrificing all of the mice at 16 dpi, brain tissues and other mucosal samples were acquired. Briefly, 100 μL of 0.1 M PBS was added per 0.1 g of fecal sample. Vaginal samples were collected by washing the vaginal canals with 200 μL of 0.1 M PBS. Intestinal samples were harvested by longitudinally sectioning the intestines and immersing the tissues in 500 μL of 0.1 M PBS. Mucosal samples were incubated at 37 °C for 1 h then centrifuged at 500 rpm for 10 min. Supernatants were collected and samples were stored at −20 °C until use. The brain tissues were used to quantify cyst burden in mice. Spleens and mesenteric lymph nodes were carefully harvested for immunological assays. All the samples were processed on an individual basis.
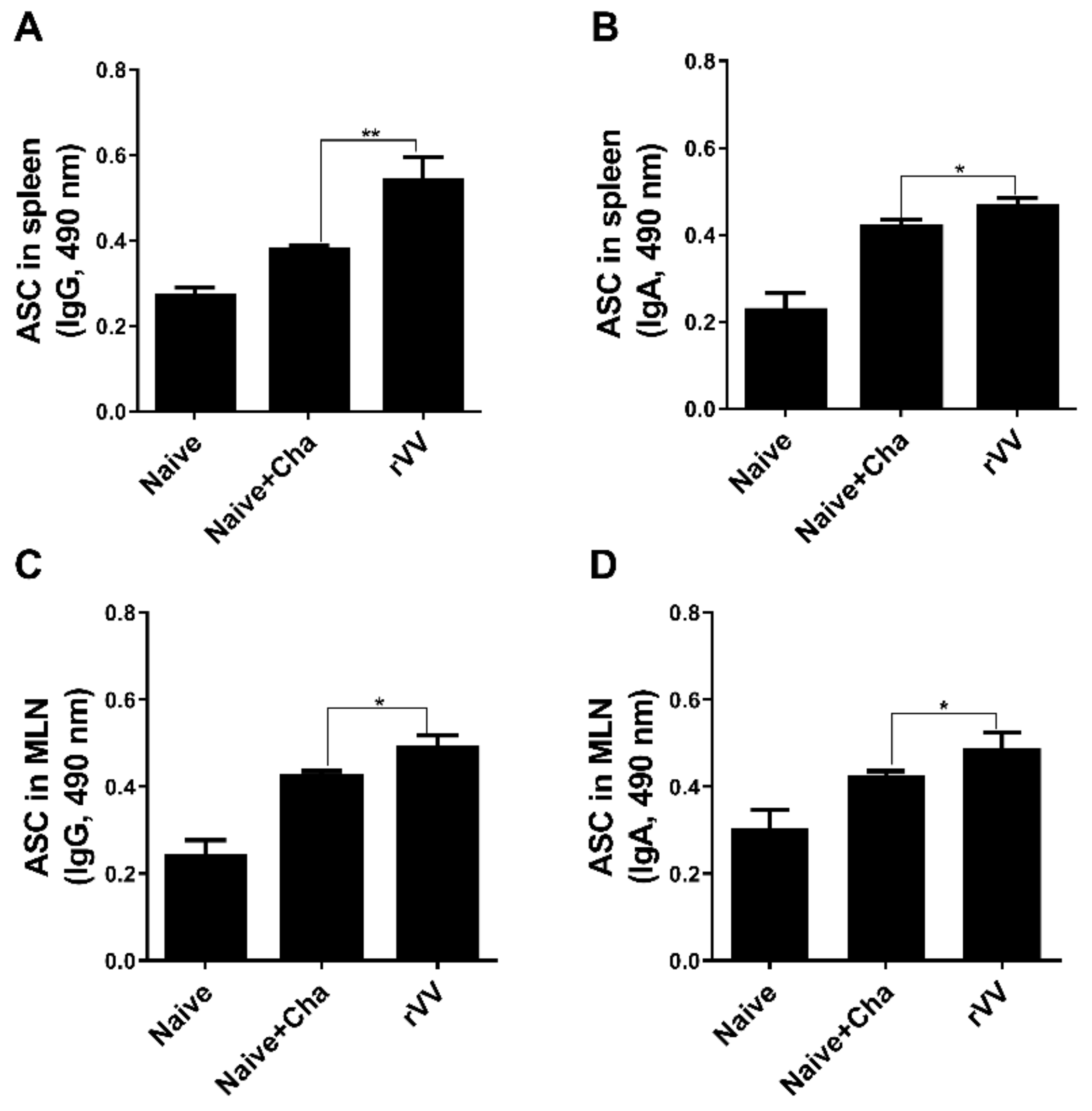
2.7. Enzyme-Linked Immunosorbent Assay (ELISA) and Antibody-Secreting Cell (ASC) Responses
T. gondii-specific serum antibody responses were determined as previously described [
8,
10]. To evaluate ASC induction, splenic and mesenteric lymph nodes (MLNs) were collected from mice. Single cell suspensions of splenocytes and MLN cells were prepared as previously described [
8]. After RBC lysis, splenocytes and MLN cells were seeded into 96-well immunoplates coated with
T. gondii ME49 whole antigen (4 μg/mL). Plates were incubated for 5 days at 37 °C. Supernatants were removed and HRP-conjugated anti-mouse IgG or IgA antibodies were added into each well. Plates were incubated for 1 h at 37 °C, and 100 μL of OPD substrate dissolved in citrate substrate buffer were inoculated into each well. Reactions were stopped using dilute H
2SO
4 and OD
490 values were measured.
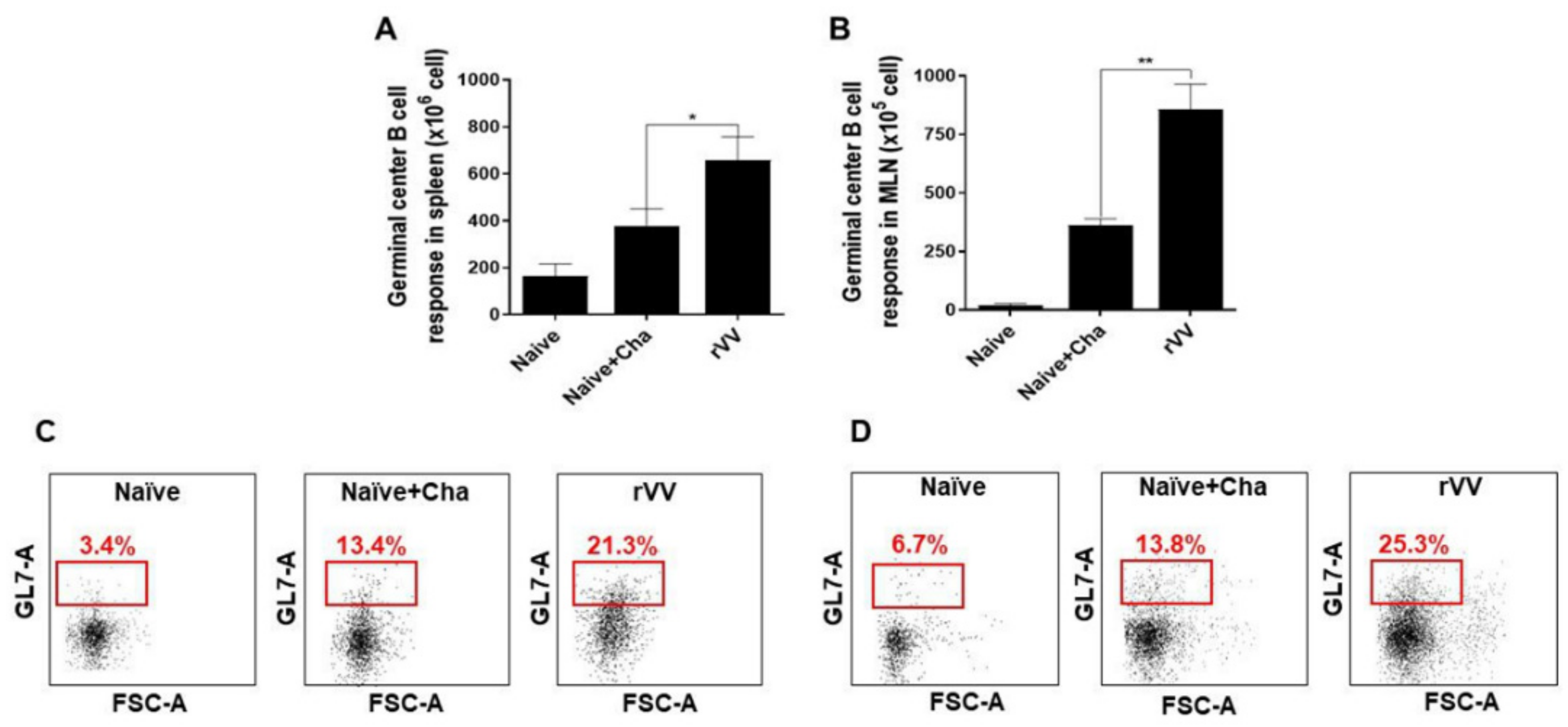
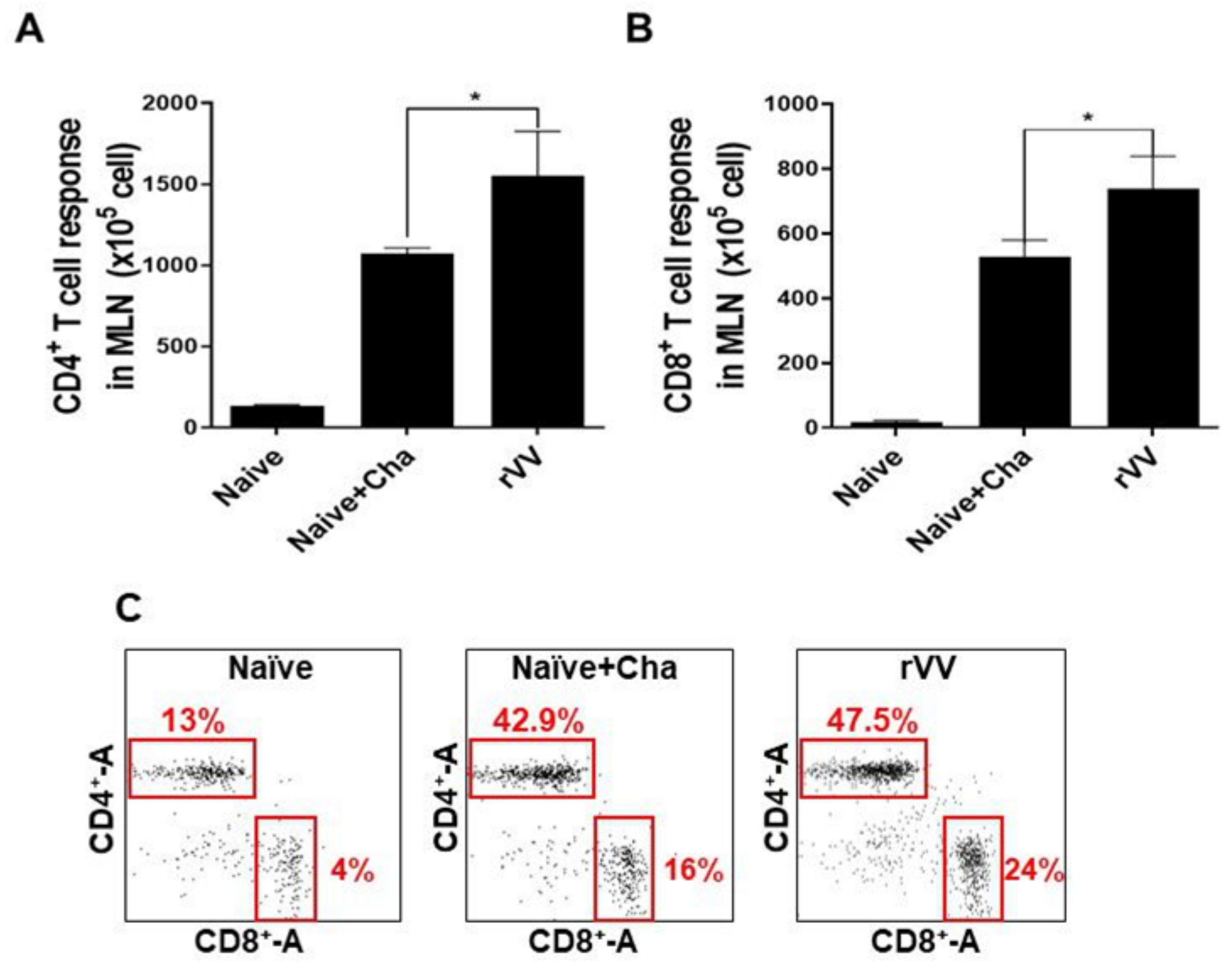
2.8. Flow Cytometry and Immune Cell Population Assessment
Flow cytometry was performed on splenocytes and MLN cells as previously described [
17]. Splenocytes (1 × 10
6 cells/mouse) and MLN cells (1 × 10
5 cells/mouse) were stimulated with
T. gondii ME49 antigen (2 μg/mL) at 37 °C with 5% CO
2 for 2 h. Stimulated cells were stained with fluorophore-conjugated antibodies purchased from BD Biosciences (Franklin Lakes, NJ, USA) and Invitrogen (Waltham, MA, USA). CD4
+ T cells, CD8
+ T cells, and germinal center B (GC B) cell populations were measured using the following antibodies: CD3 (PE- Cy7), CD4 (FITC), CD8 (PE), GL7 (PE), and B220 (FITC). All staining procedures were performed according to the manufacturer’s protocol. Stained cells were acquired by Accuri C6 flow cytometer and analyzed with the C6 Accuri software (BD Biosciences, Franklin Lakes, NJ, USA). For GC B cells, B220
+ lung cells and splenocyte populations were gated, and from this population those positive for GL7 were gated. For CD4
+ and CD8
+ T cells, CD3-positive single cell populations were gated and from this population CD4
+ and CD8
+ cells were determined.
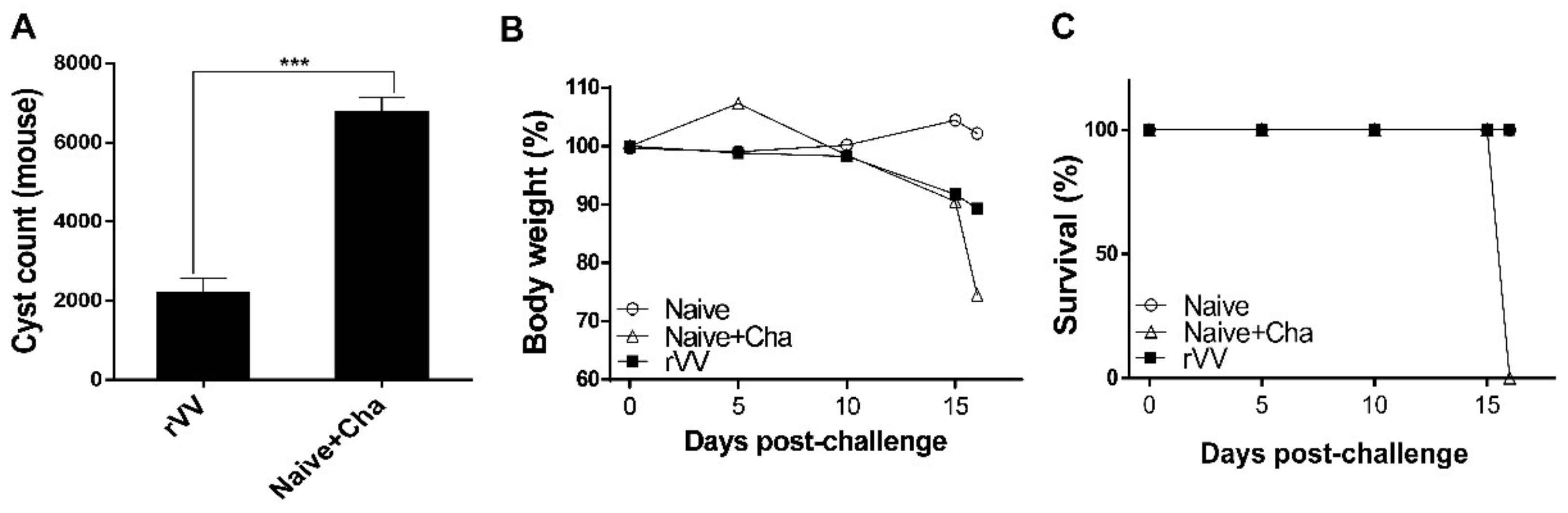
2.9. Inflammatory Cytokine Production and Cyst Burden in the Brain
Brain tissues of individual mice were subdivided into left and right hemispheres and processed separately for inflammatory cytokine assessment and cyst count, respectively. The left hemispheres were homogenized in 500 μL of PBS and then centrifuged at 10,000 rpm for 10 min. Supernatants were carefully collected and stored at −80 °C until use. Pro-inflammatory cytokines in these supernatants were measured using IFN-γ and IL-6 BD OptEIA ELISA kits (BD Biosciences, Franklin Lakes, NJ, USA). All experiments were performed according to the manufacturer’s instructions and standard curves were generated to calculate the cytokine concentrations. The right hemispheres of the brains were used to quantify cyst burdens in challenge-infected mice following the method previously described [
18]. Tissues were homogenized in 500 μL of PBS and centrifuged at 12,100 rpm for 20 min through Percoll density gradient media (BD Biosciences, Franklin Lakes, NJ, USA). The
T. gondii cyst layer was carefully collected and repeatedly washed with PBS. Cysts from each sample were counted 3 times from 3 different fields of view under a microscope (Leica DMi8, Leica, Wetzlar, Germany).
2.10. Statistical Analysis
All statistical analyses were performed using Graphpad Prism version 6 (San Diego, CA, USA). A one-way ANOVA with Tukey’s post hoc test or 2-way ANOVA with Bonferroni’s post hoc test was performed to assess the statistical significance between the means. Data are presented as mean ± SD and statistical significance is denoted with asterisks. p values of * p < 0.05, ** p < 0.01, *** p < 0.001, and **** p < 0.0001 were considered statistically significant.
4. Discussion
Oral vaccines are highly desired for several reasons. They are easy to administer with excellent patient compliance since pain infliction is virtually non-existent. Since these vaccines do not require the presence of highly trained medical personnel for proper administration, it is an efficient mass vaccination strategy [
19]. Nevertheless, the major obstacle with mucosal immunization via the oral route is the need to traverse through the highly acidic environment of the stomach. Consequently, antigenic epitopes are prone to degradation and this substantially lessens the efficacy of the vaccines [
19]. While the ROP4-rVV vaccine used in the present study was subjected to such detrimental effects, they were immunogenic and conferred protection against a lethal dose of
T. gondii ME49 challenge infection in mice.
Most of the immunological findings reported here are similar to the protective efficacy induced by ROP4-rBV vaccines as described in our previous study, although several differences in immunological parameters were noted [
10]. Consistent with our previous study, a remarkable increase in GC B cells was observed from both the spleens and MLNs of mice. However, discrepancies were observed in the T cell populations. In the oral ROP4-rBV immunized mice, differences were not significant compared to the control groups, while such differences were observed in the ROP4-rVV immunized mice. Differences were also detected in terms of brain inflammatory cytokine production. While significant IL-6 reductions were not observed in our earlier works with the ROP4-rBVs, drastic IL-6 and IFN-γ reductions were observed in the current study. Although cyst counts were comparable for the two vectored vaccines in our works, bodyweight reductions were less severe when mice were immunized with the ROP4-rVVs. Given these results, the protection induced by the ROP4-rVV vaccine appears to be superior to that of the rBV vaccine expressing an identical antigen, thereby suggesting that rVV vectors serve as potent inducers of mucosal immunity in mice. In support of this notion, one study comparing the efficacies of MVA-based vaccines against rBV vaccines has reported similar findings. MVA vaccines expressing the VP2 antigen of African horse sickness virus conferred better protection in mice than rBV vaccines expressing the same antigen at an identical dosage and number of immunizations [
20].
Antibodies are functionally required for mounting optimal protection against pathogens. Our findings revealed that immunization with the ROP4-rVV elicited significantly enhanced antibody responses in sera and also in the mucosal tissues. Mucosal antibodies, particularly the IgA subclass, are of importance. These antibodies deter the attachment of microorganisms to the mucosal surface [
21]. Since the intestinal epithelial cells are the site of infection for
T. gondii, mounting a strong first line of defense in these regions is important. The presence of these vaccine-induced mucosal antibodies, especially IgA, is crucial as they will act to limit parasitic entry into the host cells. Evidently,
T. gondii-specific secretory IgA antibodies collected from patients were reported to have successfully inhibited the
T. gondii infection of enterocytes [
22]. As exemplified using these clinical samples, the ROP4-specific mucosal IgA elicited by the ROP4-rVV vaccine may have protected mice against challenge infection by limiting the parasitic entry into intestinal epithelial cells. Similarly, pro-inflammatory cytokines such as IFN-γ are essential for resistance against
T. gondii, as their production by T cells is required to inhibit tachyzoite proliferation in the brain [
23,
24]. Nevertheless, this comes at the expense of neuronal damage. For instance, in pneumococcal meningitis mice models, reduced neuronal damages were observed from IFN-γ ablated mice whereas brain pathologies were much more severe in the wild-type mice [
25]. Therefore, a balance in inflammatory cytokines that enable parasitic inhibition while inflicting minimal neuronal damage is necessary.
There were several discrepancies between our findings and those of another study. Surprisingly, immunizing C57BL/6 mice with the recombinant vaccinia virus vaccine (LC16m0 strain) expressing the GRA4 antigen of
T. gondii failed to elicit antigen-specific antibody responses even after 3 immunizations at 3-week intervals [
4]. Contrary to these results, profound antibody responses were observed in the sera and mucosal tissues of the ROP4-rVV immunized mice. Similar to our results, mice immunized with the ROP2-expressing recombinant MVA vaccine also developed high titers of antigen-specific antibody responses [
3]. Identifying the exact factor accountable for such differences is unknown and difficult to discern, but it is possible that antigen candidates and the strain of the vaccinia virus may be involved. LC16m8 strain, which is derived from the LC16m0 strain, is categorized under 3rd generation vaccinia virus vaccines along with the MVA strain. However, their method of production and the extent of their attenuation greatly differs. Consequently, different strains of these 3rd generation vaccines were reported to possess differing virus properties, which include replication competency in mammalian cells, pro-inflammatory gene induction, and others.
Vaccinia virus dissemination into the ovaries of infected mice following intravenous and intraperitoneal inoculation has been reported, but such a phenomenon was not observed when the viruses were injected via a subcutaneous route [
26]. Intranasally inoculating vaccinia virus resulted in uncontrolled virus replication in the murine lungs, which ultimately led to their death [
27]. However, such issues were not observed from our study or by others [
28] when vaccinia viruses were inoculated via oral routes, thus implying that the oral route of administration could be an effective way for developing vaccinia virus-based vaccine candidates. As previously mentioned, antigen degradation and efficacy loss is one detriment associated with oral vaccines. To overcome this issue and improve the efficacy of orally administered mucosal vaccines, alternative strategies could be employed. One such method would be immunization through the sublingual/buccal tissues outlining the oral cavity. Evidently, sublingual/buccal immunization with the MVA-based human immunodeficiency virus 1 (HIV-1) elicited potent mucosal immune responses and protected rhesus macaques upon challenge infection [
29]. Another strategy would be to employ heterologous immunization as described through earlier MVA-based
T. gondii vaccine studies [
4,
5].
One major limitation of this study is the lack of data confirming long-term protection. In our study, all mice were sacrificed at an identical time point to enable accurate comparison of immunological parameters between groups. Thus, long-term protection could not be evaluated. Therefore, interpreting the findings illustrated in
Figure 7B would be speculative. For example, based on the downward trend of bodyweight, mice could either recover and survive for months or succumb to death via delayed disease progression. As such, further studies assessing this must be conducted.