Seroprevalence of Varicella in Pregnant Women and Newborns in a Region of Colombia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design and Data Sources
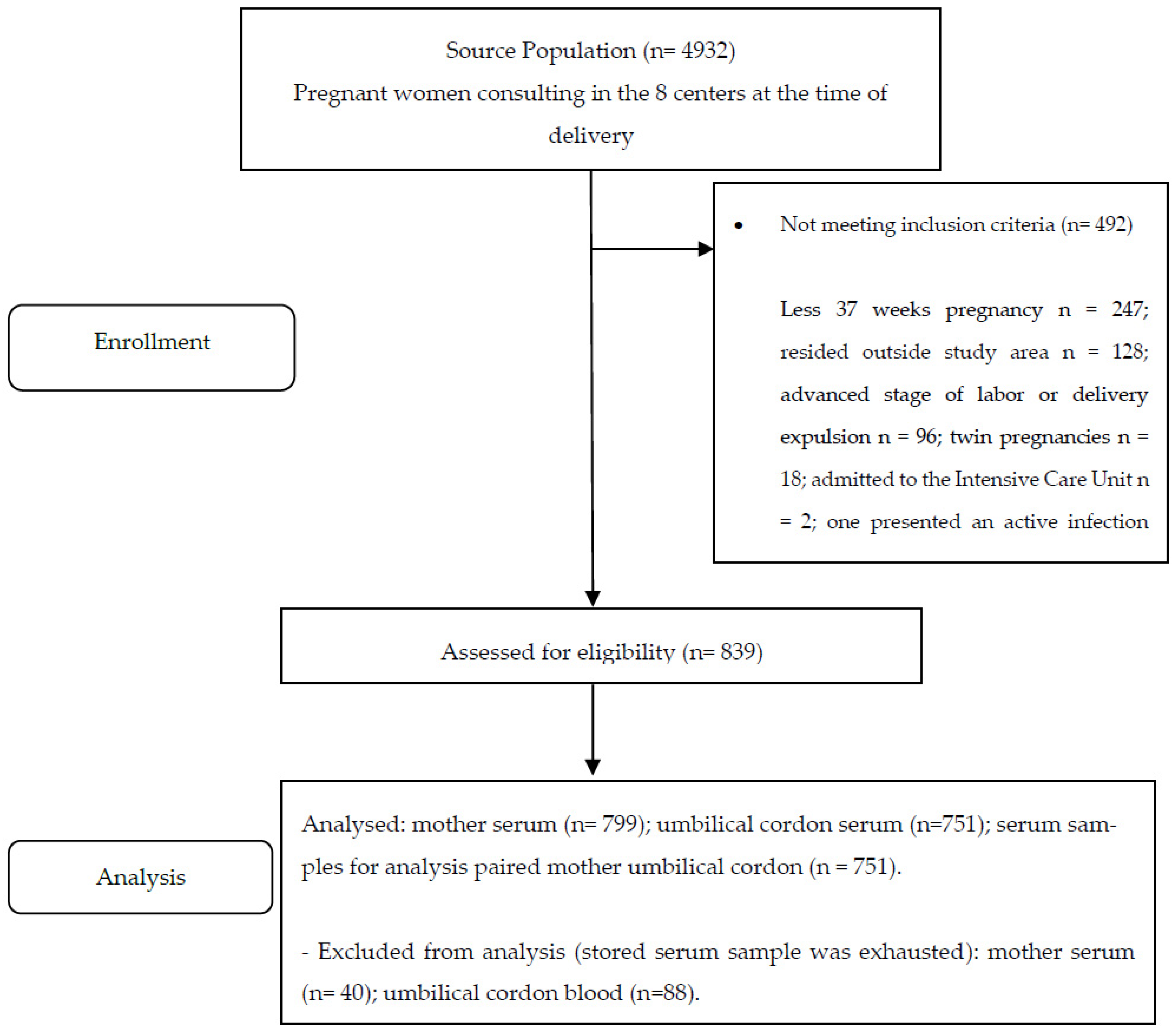
2.2. Population and Sample
2.3. Inclusion and Exclusion Criteria
2.4. Lab Tests
2.5. Statistical Analysis
3. Results
3.1. Description of the Study Population
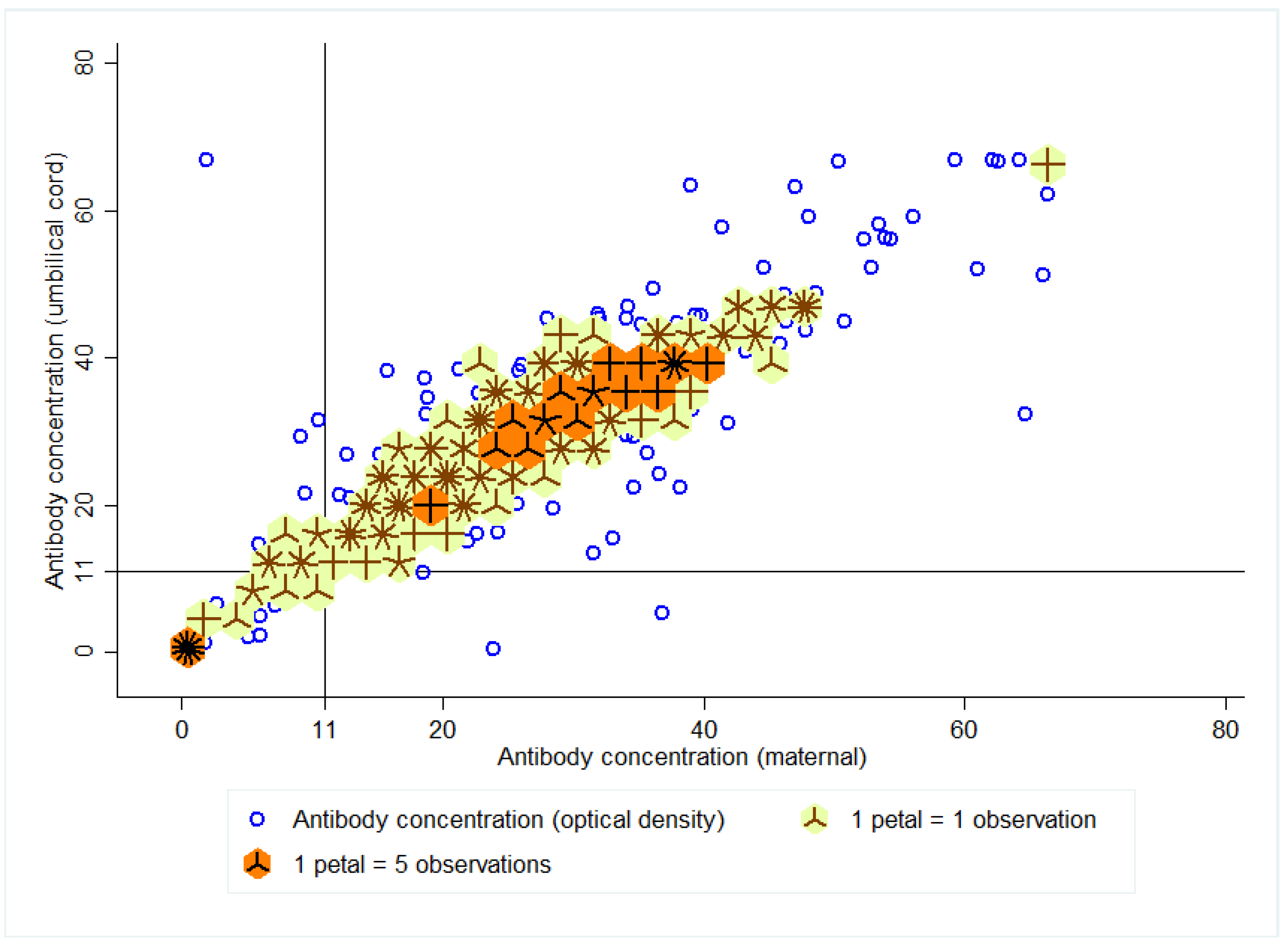
3.2. Seroprevalence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mueller, N.H.; Gilden, D.H.; Cohrs, R.J.; Mahalingam, R.; Nagel, M.A. Varicella Zoster Virus Infection: Clinical Features, Molecular Pathogenesis of Disease, and Latency. Neurol. Clin. 2008, 26, 675–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plotkin, S.A. Vaccines: Past, present and future. Nat. Med. 2005, 11, S5–S11. [Google Scholar] [CrossRef] [PubMed]
- Seward, J.F.; Zhang, J.X.; Maupin, T.J.; Mascola, L.; Jumaan, A.O. Contagiousness of Varicella in Vaccinated Cases. JAMA 2004, 292, 704–708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lloreda-Garciaa, J.M.; Martínez-Ferrándeza, C.; Gil-Sáncheza, S.; Ibáñez-Micó, S. Congenital varicella syndrome and neonatal herpes zoster. Enferm. Infecc. Microbiol. Clin. 2013, 31, 699–706. [Google Scholar] [CrossRef]
- Lamont, R.F.; Sobel, J.D.; Carrington, D.; Mazaki-Tovi, S.; Kusanovic, J.P.; Vaisbuch, E.; Romero, R. Varicella-zoster virus (chickenpox) infection in pregnancy. BJOG Int. J. Obstet. Gynaecol. 2011, 118, 1155–1162. [Google Scholar] [CrossRef] [Green Version]
- O’Grady, K.-A.; Merianos, A.; Patel, M.; Gilbert, L. High seroprevalence of antibodies to varicella zoster virus in adult women in a tropical climate. Trop. Med. Int. Health 2000, 5, 732–736. [Google Scholar] [CrossRef]
- Bollaerts, K.; Montes, M.R.; Heininger, U.; Hens, N.; Souverain, A.; Verstraeten, T.; Hartwig, S. A systematic review of varicella seroprevalence in European countries before universal childhood immunization: Deriving incidence from seroprevalence data. Epidemiol. Infect. 2017, 145, 2666–2677. [Google Scholar] [CrossRef] [Green Version]
- Arlant, L.H.F.; Garcia, M.C.P.; Aguero, M.L.A.; Cashat, M.; Parellada, C.I.; Wolfson, L.J. Burden of varicella in Latin America and the Caribbean: Findings from a systematic literature review. BMC Public Health 2019, 19, 528. [Google Scholar] [CrossRef]
- Plans, P.; Costa, J.; Espuñes, J.; Plasència, A.; Salleras, L. Prevalence of varicella-zoster antibodies in pregnant women in Catalonia (Spain). Rationale for varicella vaccination of women of childbearing age. BJOG Int. J. Obstet. Gynaecol. 2007, 114, 1122–1127. [Google Scholar] [CrossRef]
- Premathilake, I.P.; Aluthbaduge, P.; Senanayake, C.P.; Jayalatharachchi, R.; Gamage, S.; Jayamaha, J. Susceptibility for varicella and factors associated with immunity among pregnant women in a tertiary care hospital in Sri Lanka-a cross-sectional study. BMC Infect. Dis. 2019, 19, 356. [Google Scholar] [CrossRef]
- Daulagala, S.W.P.L.; Noordeen, F.; Fara, M.M.F.; Rathnayake, C.; Gunawardana, K. Exposure rate of VZV among women attending antenatal care clinic in Sri Lanka—A cross sectional study. BMC Infect. Dis. 2017, 17, 625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guido, M.; Tinelli, A.; De Donno, A.; Quattrocchi, M.; Malvasi, A.; Campilongo, F.; Zizza, A. Susceptibility to varicella-zoster among pregnant women in the province of Lecce, Italy. J. Clin. Virol. 2012, 53, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Clemens, S.A.; Azevedo, T.; Fonseca, J.C.; Silva, A.C.; Da Silveira, T.R.; Clemens, R. Seroepidemiología de la varicela en Brasil: Resultados de un estudio prospectivo transversal. Rev. Chil. Pediatría 2001, 72, 538–547. [Google Scholar] [CrossRef]
- Rubio, M.; Castro, J.; Guz, E.; Tanaka, J. Seroprevalence of hepatitis A and chickenpox in Bogotá. Rev. Panam. Infectología 2000, 4. Available online: https://encolombia.com/medicina/revistas-medicas/infectologia/voli-41/revistapanadeinfev4-1-investigaseroprevaha/ (accessed on 20 October 2021).
- Torruella, J.I.; Soto, R.G. Chickenpox seroprevalence among healthcare and social assistance workers in Catalonia (Spain). Arch. Prev. Riesgos Labor. 2017, 20, 158–166. [Google Scholar] [CrossRef]
- Ballester, R.; Porras, M.-I. El significado histórico de las encuestas de seroprevalencia como tecnología de laboratorio aplicada a las campañas de inmunización. El caso de la poliomielitis en España. Asclepio 2009, 61, 55–80. [Google Scholar] [CrossRef] [PubMed]
- Daulagala, S.W.P.L.; Noordeen, F. Epidemiology and factors influencing varicella infections in tropical countries including Sri Lanka. Virus Dis. 2018, 29, 277–284. [Google Scholar] [CrossRef]
- Ibrahim, E.G.; Wahed, W.Y.A.; Eid, H.M.; Deeb, W.S. Seroprevalence of varicella-zoster virus among pregnant women in Fayoum Governorate, Egypt. J. Egypt. Public Health Assoc. 2019, 94, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Völker, F.; Cooper, P.; Bader, O.; Uy, A.; Zimmermann, O.; Lugert, R.; Groß, U. Prevalence of pregnancy-relevant infections in a rural setting of Ghana. BMC Pregnancy Childbirth 2017, 17, 172. [Google Scholar] [CrossRef] [Green Version]
- Technical and Operational Guidelines for the application of the reinforcement of the varicella vaccine. In The Vaccination Scheme of the Expanded Program of Immunizations-PAI; Ministerio de Salud de Colombia: Bogotá, Colombia, June 2019; Volume 8.
- Hincapié-Palacio, D.; Hoyos, M.C.; Ochoa, J.; Montoya, N.; García, D.; Osorio, E. Effect of maternal immunization against pertussis in Medellin and the metropolitan area, Colombia, 2016–2017. Vaccine 2018, 36, 3984–3991. [Google Scholar] [CrossRef] [PubMed]
- Wutzler, P.; Bonanni, P.; Burgess, M.; Gershon, A.; Safadi, M.; Casabona, G. Varicella vaccination—The global experience. Expert Rev. Vaccines 2017, 16, 833–843. [Google Scholar] [CrossRef] [Green Version]
- Habib, A.; Prymula, R.; Carryn, S.; Esposito, S.; Henry, O.; Ravault, S.; Usonis, V.; Wysocki, J.; Gillard, P.; Povey, M. Correlation of protection against varicella in a randomized Phase III varicella-containing vaccine efficacy trial in healthy infants. Vaccine 2021, 39, 3445–3454. [Google Scholar] [CrossRef] [PubMed]
- Cortés-Reyes, É.; Rubio-Romero, J.A.; Gaitán-Duarte, H. Métodos estadísticos de evaluación de la concordancia y la reproducibilidad de pruebas diagnósticas. Rev. Colomb. Obstet. Ginecol. 2010, 61, 247–255. [Google Scholar] [CrossRef] [Green Version]
- Parker, R.; Scott, C.; Inácio, V.; Stevens, N.T. Using multiple agreement methods for continuous repeated measures data: A tutorial for practitioners. BMC Med. Res. Methodol. 2020, 20, 1–14. [Google Scholar] [CrossRef]
- Karunajeewa, H.; Siebert, D.; Hammond, R.; Garland, S.; Kelly, H. Seroprevalence of varicella zoster virus, parvovirus B19 and Toxoplasma gondii in a Melbourne obstetric population: Implications for management. Aust. N. Z. J. Obstet. Gynaecol. 2001, 41, 23–28. [Google Scholar] [CrossRef]
- Van Der Zwet, W.C.; Vandenbroucke-Grauls, C.M.; van Elburg, R.; Cranendonk, A.; Zaaijer, H.L. Neonatal antibody titers against varicella-zoster virus in relation to gestational age, birth weight, and maternal titer. Pediatrics 2002, 109, 79–85. [Google Scholar] [CrossRef]
- Leineweber, B.; Grote, V.; Schaad, B.; Heininger, U. Transplacentally acquired Immunoglobulin G antibodies against measles, mumps, rubella and varicella-zoster virus in preterm and full term newborns. Pediatr. Infect. Dis. J. 2004, 23, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Vojgani, Y.; Zarei, S.; Rajaei, S.; Chamani-Tabriz, L.; Ghaemimanesh, F.; Mohammadinia, N.; Jeddi-Tehrani, M. Sero-Prevalence of Antibodies against Varicella Zoster Virus in Children under Seven-Years Old in 2012 in Tehran, Iran. Iran. J. Public Health 2014, 43, 1569–1575. [Google Scholar] [PubMed]
- Masuet-Aumatell, C.; Ramon-Torrell, J.M.; Casanova-Rituerto, A.; Banqué-Navarro, M.; Dávalos-Gamboa, M.D.R.; Montaño-Rodríguez, S.L. Seroprevalence of varicella-zoster virus infection in children from Cochabamba: Tropical or temperate pattern? Trop. Med. Int. Health 2012, 18, 296–302. [Google Scholar] [CrossRef]
- Hoseini, S.G.; Kelishadi, R.; Kasaeian, A.; Ataei, B.; Yaran, M.; Motlagh, M.E.; Heshmat, R.; Ardalan, G.; Safari, O.; Qorbani, M.; et al. Seroprevalence and Risk Factors of Varicella Zoster Infection in Iranian Adolescents: A Multilevel Analysis; The CASPIAN-III Study. PLoS ONE 2016, 11, e0158398. [Google Scholar] [CrossRef]
- Parente, S.; Moriello, N.S.; Maraolo, A.E.; Tosone, G. Management of chickenpox in pregnant women: An Italian perspective. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
| Pregnant Women | Total n (%) | Seropositive n (%) | Seronegative n (%) | Equivocal n (%) |
| Age (years) | ||||
| 13 to 22 | 362 (45.3) | 298 (43.7) | 60 (57.7) | 4 (30.8) |
| 23 to 32 | 341 (42.7) | 299 (43.8) | 36 (34.6) | 6 (46.2) |
| 33 to 43 | 96 (12.0) | 85 (12.5) | 8 (7.7) | 3 (23.1) |
| Area | ||||
| Urban | 760 (95.1) | 651 (95.5) | 98 (94.2) | 11 (84.6) |
| Rural | 39 (4.9) | 31 (4.5) | 6 (5.8) | 2 (15.4) |
| Educational level | ||||
| Less than 11 years | 612 (76.6) | 527 (77.3) | 77 (74.0) | 8 (61.5) |
| 11 years or more | 185 (23.1) | 154 (22.6) | 26 (25.0) | 5 (38.5) |
| No data | 2 (0.3) | 1 (0.1) | 1 (1.0) | 0 (0.00) |
| Socioeconomic status | ||||
| Low | 715 (89.5) | 613 (89.9) | 90 (86.5) | 12 (92.3) |
| High | 35 (4.4) | 29 (4.2) | 6 (5.8) | 0 (0.0) |
| No data | 49 (6.1) | 40 (5.9) | 8 (7.7) | 1 (7.7) |
| Social security in health | ||||
| Contributory | 391 (48.94) | 325 (47.65) | 58 (55.8) | 8 (61.5) |
| Subsidized | 408 (51.06) | 357 (52.35) | 46 (44.2) | 5 (38.5) |
| Overcrowding | ||||
| Yes | 26 (3.2) | 22 (3.2) | 4 (3.8) | 0 (0.0) |
| No | 773 (96.8) | 660 (96.8) | 100 (96.2) | 13 (100.0) |
| Number of pregnancies | ||||
| 1 | 382 (47.8) | 319 (46.8) | 55 (52.9) | 8 (61.5) |
| 2 | 220 (27.5) | 194 (28.4) | 24 (23.1) | 2 (15.2) |
| 3 or more | 197 (24.7) | 169 (24.8) | 25 (24.0) | 3 (24.0) |
| Gestational week at delivery | ||||
| 37 | 149 (18.7) | 124 (18.2) | 21 (20.3) | 4 (30.8) |
| 38 | 183 (22.9) | 154 (22.6) | 27 (26.0) | 2 (15.4) |
| 39 | 227 (28.4) | 193 (28.3) | 30 (28.8) | 4 (30.8) |
| 40 | 203 (25.4) | 181 (26.5) | 20 (19.2) | 2 (15.4) |
| 41 | 37 (4.6) | 30 (4.4) | 6 (5.7) | 1 (7.6) |
| Maternal morbidity | ||||
| Yes | 72 (9.0) | 65 (9.5) | 6 (5.8) | 1 (7.7) |
| No | 727 (91.0) | 617 (90.5) | 98 (94.2) | 12 (92.3) |
| Total | 799 (100.0) | 682(85.4) | 104 (12.3) | 13 (1.6) |
| Newborns | Total n (%) | Seropositive n (%) | Seronegative n (%) | Equivocal n (%) |
| Sex | ||||
| Male | 392 (52.2) | 341 (51.9) | 47 (52.8) | 4 (80.0) |
| Female | 359 (47.8) | 316 (48.1) | 42 (47.2) | 1 (20.0) |
| Birth weight (grams) | ||||
| 3000 or more | 483(64.3) | 425(64.7) | 55(61.8) | 3(60.0) |
| Less than 3000 | 268(35.7) | 232(35.3) | 34(38.2) | 2(40.0) |
| Total | 751 (100.00) | 657 (87.5) | 89 (11.8) | 5 (0.7) |
| Variable | Pregnant Woman | Newborn | |||||
|---|---|---|---|---|---|---|---|
| Incidence Rate Ratio (IRR) | 95% CI | Percentage Change in Seropositivity | Incidence Rate Ratio (IRR) | 95% CI | Percentage Change in Seropositivity | ||
| Area of residence | Urban | Reference | - | Reference | - | ||
| Rural * | 0.4 | 0.2–0.7 | −60 (−77 to −30) | 0.5 | 0.3–0.9 | −50 (−70 to −10) | |
| Socioeconomic status | Low | Reference | - | Reference | - | ||
| High * | 0.4 | 0.2–0.7 | −60 (−77 to −30) | 0.5 | 0.3–0.9 | −50 (−70 to −10) | |
| Gestational week at delivery | 37 | Reference | - | Reference | - | ||
| 38 | 1.2 | 0.7–1.8 | 20 (−30 to 80) | 1.1 | 0.8–1.7 | 10 (−20 to 70) | |
| 39 | 1.3 | 0.9–2.1 | 30 (−10 to 110) | 1.1 | 0.8–1.8 | 10 (−20 to 80) | |
| 40 | 1.4 | 0.9–2.2 | 40 (−10 to 120) | 1.3 | 0.8–2.0 | 30 (−20 to 100) | |
| 41 | 0.5 * | 0.3–1.0 | −50 (−70 to 0) | 0.6 | 0.3–1.1 | −40 (−70 to 10) | |
| Mother’s age (in years) | <18 | Reference | - | Reference | - | ||
| 18 to 34 | 1.5 | 0.9–2.5 | 50 (−10 to 150) | 1.4 | 0.9–2.1 | 40 (−10 to 110) | |
| >35 | 0.6 | 0.3–1.1 | −40 (−10 to 10) | 0.7 | 0.4–1.3 | −30 (−60 to 30) | |
| Educational level | <11 years | Reference | - | Reference | - | ||
| ≥11 years * | 0.6 | 0.4–0.8 | −40 (−60 to −20) | 0.6 | 0.5–0.8 | −40 (−50 to −20) | |
| Birth weight (grams) | ≥3000 | - | - | Reference | - | ||
| <3000 | - | - | - | 0. 8 | 0.6–1.0 | −20 (−40 to 0) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenis-Ballesteros, V.; Ochoa, J.; Hincapié-Palacio, D.; León-Álvarez, A.; Vargas-Restrepo, F.; Ospina, M.C.; Buitrago-Giraldo, S.; Díaz, F.J.; Gonzalez-Ortíz, D. Seroprevalence of Varicella in Pregnant Women and Newborns in a Region of Colombia. Vaccines 2022, 10, 52. https://doi.org/10.3390/vaccines10010052
Lenis-Ballesteros V, Ochoa J, Hincapié-Palacio D, León-Álvarez A, Vargas-Restrepo F, Ospina MC, Buitrago-Giraldo S, Díaz FJ, Gonzalez-Ortíz D. Seroprevalence of Varicella in Pregnant Women and Newborns in a Region of Colombia. Vaccines. 2022; 10(1):52. https://doi.org/10.3390/vaccines10010052
Chicago/Turabian StyleLenis-Ballesteros, Viviana, Jesús Ochoa, Doracelly Hincapié-Palacio, Alba León-Álvarez, Felipe Vargas-Restrepo, Marta C. Ospina, Seti Buitrago-Giraldo, Francisco J. Díaz, and Denise Gonzalez-Ortíz. 2022. "Seroprevalence of Varicella in Pregnant Women and Newborns in a Region of Colombia" Vaccines 10, no. 1: 52. https://doi.org/10.3390/vaccines10010052
APA StyleLenis-Ballesteros, V., Ochoa, J., Hincapié-Palacio, D., León-Álvarez, A., Vargas-Restrepo, F., Ospina, M. C., Buitrago-Giraldo, S., Díaz, F. J., & Gonzalez-Ortíz, D. (2022). Seroprevalence of Varicella in Pregnant Women and Newborns in a Region of Colombia. Vaccines, 10(1), 52. https://doi.org/10.3390/vaccines10010052






