Benign Fasciculation Syndrome and Migraine Aura without Headache: Possible Rare Side Effects of the BNT162b2 mRNA Vaccine? A Case Report and a Potential Hypothesis
Abstract
1. Introduction
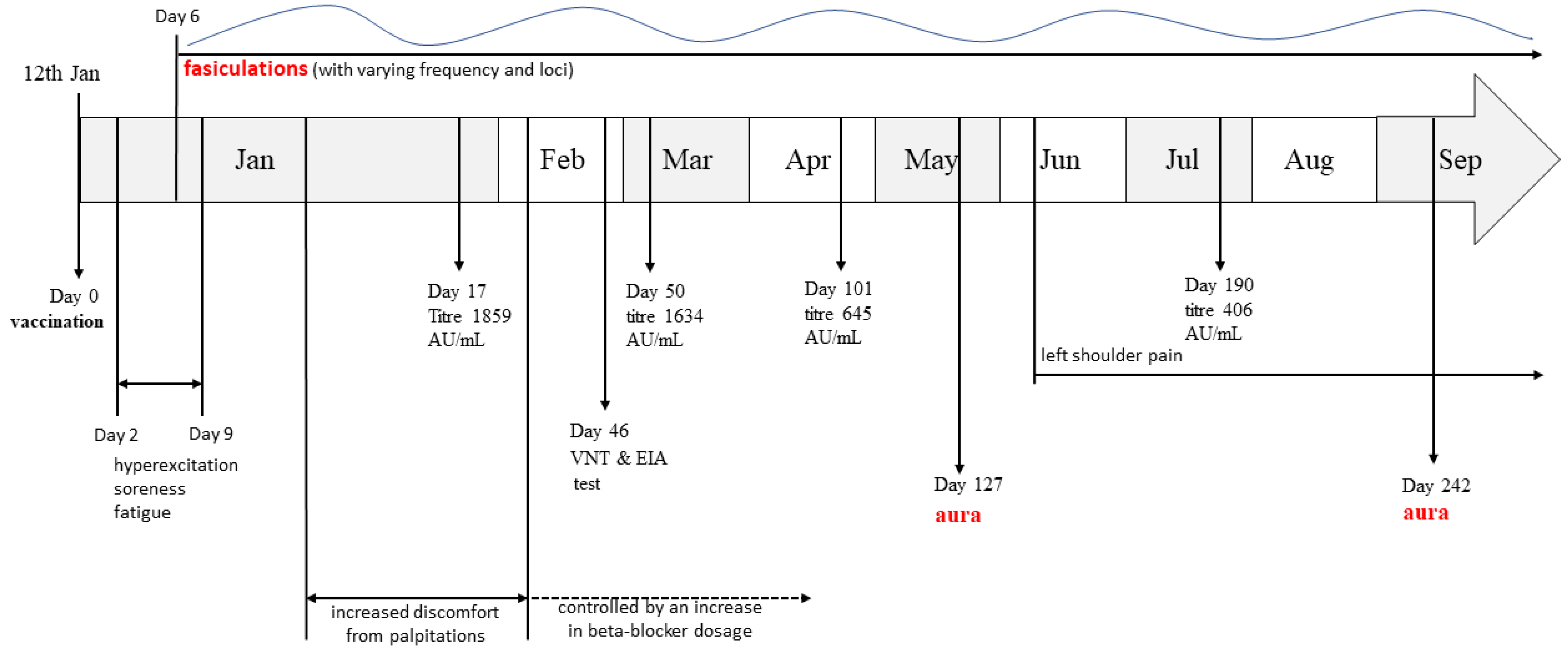
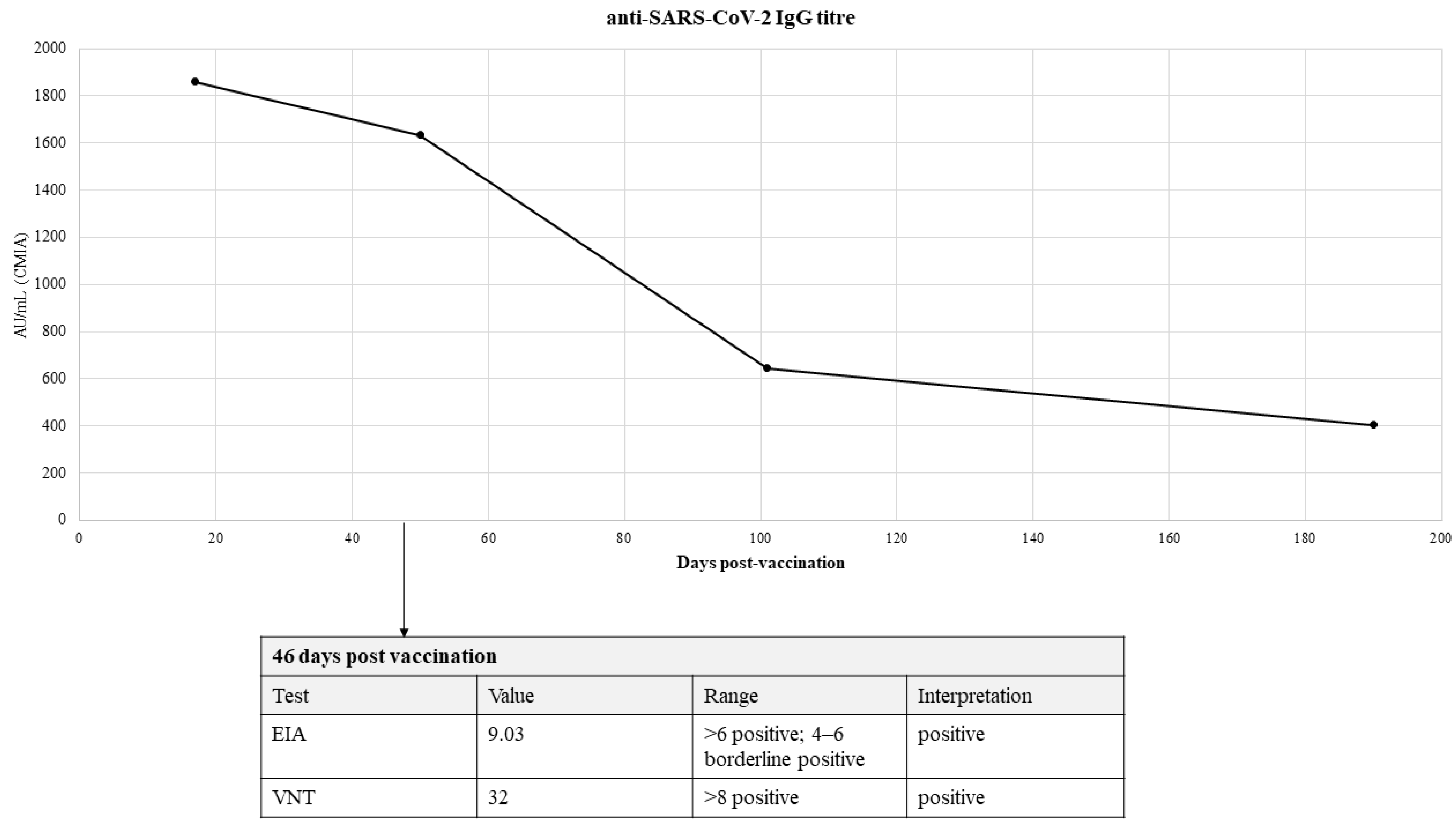
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of interest
References
- Tavilani, A.; Abbasi, E.; Kian Ara, F.; Darini, A.; Asefy, Z. COVID-19 vaccines: Current evidence and considerations. Metab. Open 2021, 12, 100124. Available online: https://linkinghub.elsevier.com/retrieve/pii/S2589936821000487 (accessed on 15 December 2021). [CrossRef]
- Adhisivam, B.; Bhat, B.V. Dancing Fascicles—A New Signal Related to Hepatitis B Immunization. Indian J. Pediatr. 2015, 82, 387. Available online: http://link.springer.com/10.1007/s12098-014-1632-z (accessed on 15 December 2021). [CrossRef] [PubMed][Green Version]
- Jeon, M.; Kim, J.; Oh, C.E.; Lee, J.-Y. Adverse Events Following Immunization Associated with Coronavirus Disease 2019 Vaccination Reported in the Mobile Vaccine Adverse Events Reporting System. J. Korean Med. Sci. 2021, 36, e114. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8093606 (accessed on 15 December 2021). [CrossRef]
- Finsterer, J.; Scorza, F.A.; Scorza, C.A. Post SARS-CoV-2 vaccination Guillain-Barre syndrome in 19 patients. Clinics 2021, 28, 76. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8478139 (accessed on 15 December 2021). [CrossRef] [PubMed]
- Ismail, I.I.; Salama, S. A systematic review of cases of CNS demyelination following COVID-19 vaccination. J. Neuroimmunol. 2022, 362, 577765. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0165572821002927 (accessed on 15 December 2021). [CrossRef] [PubMed]
- García-Estrada, C.; Gómez-Figueroa, E.; Alban, L.; Arias-Cárdenas, A. Optic neuritis after COVID-19 vaccine application. Clin. Exp. Neuroimmunol. 2021, 00, 1–3. Available online: https://onlinelibrary.wiley.com/doi/10.1111/cen3.12682 (accessed on 15 December 2021). [CrossRef]
- Cauchi, M.; Ball, H.; Ben-Shlomo, Y.; Robertson, N. Interpretation of vaccine associated neurological adverse events: A methodological and historical review. J. Neurol. 2021, 269, 493–503. Available online: https://link.springer.com/10.1007/s00415-021-10747-8 (accessed on 15 December 2021). [CrossRef] [PubMed]
- Walter, A.; Kraemer, M. A neurologist’s rhombencephalitis after comirnaty vaccination. A change of perspective. Neurol. Res. Pract. 2021, 8, 356. Available online: https://neurolrespract.biomedcentral.com/articles/10.1186/s42466-021-00156-7 (accessed on 15 December 2021). [CrossRef] [PubMed]
- Ng, J.; Chaudhuri, K.R.; Tan, E. Functional Neurological Disorders and COVID-19 Vaccination. Ann. Neurol. 2021, 90, 328. Available online: https://onlinelibrary.wiley.com/doi/10.1002/ana.26160 (accessed on 15 December 2021). [CrossRef] [PubMed]
- Goss, A.L.; Samudralwar, R.D.; Das, R.R.; Nath, A. ANA Investigates: Neurological Complications of COVID-19 Vaccines. Ann. Neurol. 2021, 89, 856–857. Available online: https://onlinelibrary.wiley.com/doi/10.1002/ana.26065 (accessed on 15 December 2021). [CrossRef]
- Thakur, K.T.; Epstein, S.; Bilski, A.; Balbi, A.; Boehme, A.K.; Brannagan, T.H.; Wesley, S.F.; Riley, C.S. Neurologic Safety Monitoring of COVID-19 Vaccines. Neurology 2021, 97, 767–775. Available online: http://www.neurology.org/lookup/doi/10.1212/WNL.0000000000012703 (accessed on 15 December 2021).
- Lu, L.; Xiong, W.; Mu, J.; Zhang, Q.; Zhang, H.; Zou, L.; Li, W.; He, L.; Sander, J.W.; Zhou, D. The potential neurological effect of the COVID-19 vaccines: A review. Acta Neurol. Scand. 2021, 144, 3–12. Available online: https://onlinelibrary.wiley.com/doi/10.1111/ane.13417 (accessed on 15 December 2021). [CrossRef]
- Leite, M.A.A.; Orsini, M.; De Freitas, M.R.G.; Pereira, J.S.; Gobbi, F.H.P.; Bastos, V.H.; de Castro Machado, D.; Machado, S.; Arrias-Carrion, O.; de Souza, J.A.; et al. Another perspective on fasciculations: When is it not caused by the classic form of amyotrophic lateral sclerosis or progressive spinal atrophy? Neurol. Int. 2014, 6, 47–51. Available online: http://www.pagepress.org/journals/index.php/ni/article/view/5208 (accessed on 8 December 2021). [CrossRef]
- Filippakis, A.; Jara, J.; Ventura, N.; Scala, S.; Scopa, C.; Ruthazer, R.; Karakis, I.; Srinivasan, J.; Russell, J.A.; Ho, D.T. A prospective study of benign fasciculation syndrome and anxiety. Muscle Nerve 2018, 58, 852–854. Available online: https://onlinelibrary.wiley.com/doi/10.1002/mus.26193 (accessed on 8 December 2021). [CrossRef]
- Shah, D.R.; Dilwali, S.; Friedman, D.I. Current Aura without Headache. Curr. Pain Headache Rep. 2018, 22, 77. Available online: http://link.springer.com/10.1007/s11916-018-0725-1 (accessed on 9 December 2021). [CrossRef] [PubMed]
- Olesen, J. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808. Available online: http://journals.sagepub.com/doi/10.1177/0333102413485658 (accessed on 5 December 2021).
- Huzly, D.; Panning, M.; Smely, F.; Enders, M.; Komp, J.; Steinmann, D. Validation and performance evaluation of a novel interferon-γ release assay for the detection of SARS-CoV-2 specific T-cell response. medRxiv 2021, medRxiv:21260316. Available online: http://medrxiv.org/content/early/2021/07/22/2021.07.17.21260316.abstract (accessed on 15 December 2021).
- De Carvalho, M.; Kiernan, M.C.; Swash, M. Fasciculation in amyotrophic lateral sclerosis: Origin and pathophysiological relevance. J. Neurol. Neurosurg. Psychiatry 2017, 88, 773–779. Available online: https://jnnp.bmj.com/lookup/doi/10.1136/jnnp-2017-315574 (accessed on 2 December 2021). [CrossRef] [PubMed]
- De Carvalho, M.; Miranda, P.C.; de Lourdes Sales Luís, M.; Ducla-Soares, E. Neurophysiological features of fasciculation potentials evoked by transcranial magnetic stimulation in amyotrophic lateral sclerosis. J. Neurol. 2000, 247, 189–194. Available online: http://link.springer.com/10.1007/s004150050561 (accessed on 3 December 2021). [CrossRef]
- Montalvo, A.; Swash, M.; de Carvalho, M. Benign fasciculations: A follow-up study with electrophysiological studies. Muscle Nerve 2021, 64, 670–675. Available online: https://onlinelibrary.wiley.com/doi/10.1002/mus.27411 (accessed on 15 December 2021). [CrossRef]
- Paliwal, V.K.; Garg, R.K.; Gupta, A.; Tejan, N. Neuromuscular presentations in patients with COVID-19. Neurol. Sci. 2020, 41, 3039–3056. Available online: https://link.springer.com/10.1007/s10072-020-04708-8 (accessed on 15 December 2021). [CrossRef]
- Waliszewska-Prosół, M.; Budrewicz, S. The unusual course of a migraine attack during COVID-19 infection—Case studies of three patients. J. Infect. Public Health 2021, 14, 903–905. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1876034121001180 (accessed on 15 December 2021). [CrossRef]
- Consoli, S.; Dono, F.; Evangelista, G.; D’Apolito, M.; Travaglini, D.; Onofrj, M.; Bonanni, L. Status migrainosus: A potential adverse reaction to Comirnaty (BNT162b2, BioNtech/Pfizer) COVID-19 vaccine—A case report. Neurol. Sci. 2021, 1–4. Available online: https://link.springer.com/10.1007/s10072-021-05741-x (accessed on 14 December 2021).
- Rattanawong, W.; Akaratanawat, W.; Tepmongkol, S.; Chutinet, A.; Tantivatana, J.; Suwanwela, N.C. Acute prolonged motor aura resembling ischemic stroke after COVID-19 vaccination (CoronaVac): The first case report. J. Headache Pain 2021, 22, 93. Available online: https://thejournalofheadacheandpain.biomedcentral.com/articles/10.1186/s10194-021-01311-w (accessed on 14 December 2021). [CrossRef]
- Cantarelli Rodrigues, T.; Hidalgo, P.F.; Skaf, A.Y.; Serfaty, A. Subacromial-subdeltoid bursitis following COVID-19 vaccination: A case of shoulder injury related to vaccine administration (SIRVA). Skeletal Radiol. 2021, 50, 2293–2297. Available online: https://link.springer.com/10.1007/s00256-021-03803-x (accessed on 15 December 2021). [CrossRef]
- Lui, D.T.W.; Lee, K.K.; Lee, C.H.; Lee, A.C.H.; Hung, I.F.N.; Tan, K.C.B. Development of Graves’ Disease After SARS-CoV-2 mRNA Vaccination: A Case Report and Literature Review. Front Public Heal 2021, 9. Available online: https://www.frontiersin.org/articles/10.3389/fpubh.2021.778964/full (accessed on 15 December 2021). [CrossRef] [PubMed]
- Weintraub, M.A.; Ameer, B.; Sinha Gregory, N. Graves Disease Following the SARS-CoV-2 Vaccine: Case Series. J. Investig. Med. High Impact Case Rep. 2021, 9, 232470962110633. Available online: http://journals.sagepub.com/doi/10.1177/23247096211063356 (accessed on 15 December 2021). [CrossRef] [PubMed]
- Vera-Lastra, O.; Ordinola Navarro, A.; Cruz Domiguez, M.P.; Medina, G.; Sánchez Valadez, T.I.; Jara, L.J. Two Cases of Graves’ Disease Following SARS-CoV-2 Vaccination: An Autoimmune/Inflammatory Syndrome Induced by Adjuvants. Thyroid 2021, 31, 1436–1439. Available online: https://www.liebertpub.com/doi/10.1089/thy.2021.0142 (accessed on 15 December 2021). [CrossRef]
- Mateu-Salat, M.; Urgell, E.; Chico, A. SARS-CoV-2 as a trigger for autoimmune disease: Report of two cases of Graves’ disease after COVID-19. J. Endocrinol. Investig. 2020, 43, 1527–1528. Available online: https://link.springer.com/10.1007/s40618-020-01366-7 (accessed on 2 December 2021).
- Ricov, M.; Salai, G.; Sigur, V.; Seric, A.; Basic-Jukic, N. Magnesium Homeostasis Disorder in Patients with Chronic Kidney Disease. Acta Med. Croat 2021, 75, 41–51. Available online: https://hrcak.srce.hr/259262 (accessed on 1 December 2021).
- Schwalfenberg, G.K.; Genuis, S.J. The Importance of Magnesium in Clinical Healthcare. Scientifica 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Cañas, C.A. The triggering of post-COVID-19 autoimmunity phenomena could be associated with both transient immunosuppression and an inappropriate form of immune reconstitution in susceptible individuals. Med. Hypotheses 2020, 145, 110345. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0306987720325172 (accessed on 7 December 2021). [CrossRef] [PubMed]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Grgurevic, L.; Novak, R.; Hrkac, S.; Salai, G.; Grazio, S. Post-COVID-19 exacerbation of fibrodysplasia ossificans progressiva with multiple flare-ups and extensive heterotopic ossification in a 45-year-old female patient. Rheumatol. Int. 2021, 41, 1495–1501. Available online: https://link.springer.com/10.1007/s00296-021-04911-6 (accessed on 1 December 2021). [CrossRef]
| 149 Days Post Vaccination | ||
|---|---|---|
| Test | Value | Reference Range |
| Thyroid stimulating hormone | 2.43 | 0.35–4.9 |
| T4 | 103 | 62–150 |
| Anti-Tg | 33.5 | Positive > 4.1 |
| Anti-TPO | 10.05 | Positive > 5.61 |
| PTH | 4.84 | 1.6–7.2 |
| 25-hidroxy vitamin D | 74 | Normal > 75 nmol/L |
| Sodium | 139 | 137–146 mmol/L |
| Potassium | 4.3 | 3.9–5.1 mmol/L |
| Ionic calcium | 1.21 | 1.11–1.32 mmol/L |
| Total magnesium | 0.91 | 0.65–1.05 mmol/L |
| Inorganic phosphorous | 1.25 | 0.79–1.42 mmol/L |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salai, G.; Bilic, E.; Primorac, D.; Lakusic, D.M.; Bilic, H.; Lazibat, I.; Grgurevic, L. Benign Fasciculation Syndrome and Migraine Aura without Headache: Possible Rare Side Effects of the BNT162b2 mRNA Vaccine? A Case Report and a Potential Hypothesis. Vaccines 2022, 10, 117. https://doi.org/10.3390/vaccines10010117
Salai G, Bilic E, Primorac D, Lakusic DM, Bilic H, Lazibat I, Grgurevic L. Benign Fasciculation Syndrome and Migraine Aura without Headache: Possible Rare Side Effects of the BNT162b2 mRNA Vaccine? A Case Report and a Potential Hypothesis. Vaccines. 2022; 10(1):117. https://doi.org/10.3390/vaccines10010117
Chicago/Turabian StyleSalai, Grgur, Ervina Bilic, Dragan Primorac, Darija Mahovic Lakusic, Hrvoje Bilic, Ines Lazibat, and Lovorka Grgurevic. 2022. "Benign Fasciculation Syndrome and Migraine Aura without Headache: Possible Rare Side Effects of the BNT162b2 mRNA Vaccine? A Case Report and a Potential Hypothesis" Vaccines 10, no. 1: 117. https://doi.org/10.3390/vaccines10010117
APA StyleSalai, G., Bilic, E., Primorac, D., Lakusic, D. M., Bilic, H., Lazibat, I., & Grgurevic, L. (2022). Benign Fasciculation Syndrome and Migraine Aura without Headache: Possible Rare Side Effects of the BNT162b2 mRNA Vaccine? A Case Report and a Potential Hypothesis. Vaccines, 10(1), 117. https://doi.org/10.3390/vaccines10010117








