Ubiquinone Supplementation with 300 mg on Glycemic Control and Antioxidant Status in Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial
Abstract
1. Introduction
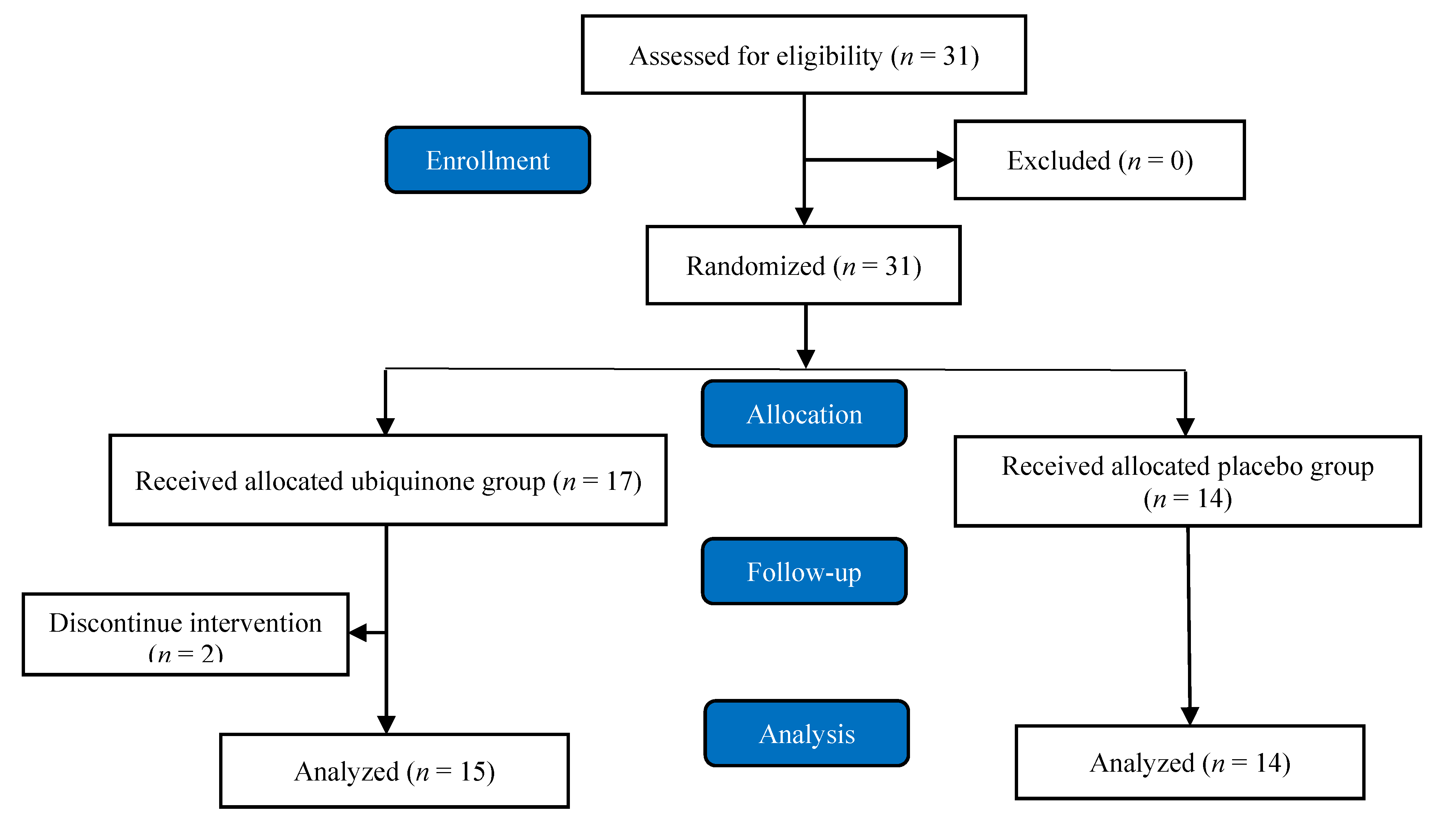
2. Materials and Methods
2.1. Study Design and Participants’ Inclusion and Exclusion Criteria
2.2. Supplementation
2.3. Demographic Data Collection and Anthropometric Assessments
2.4. Blood Sample Collection
2.5. Hematological Measurements
2.6. Oxidative Stress and Antioxidant Capacity
2.7. Ubiquinone Status Measurements
2.8. Statistical Analyses
3. Results
3.1. Baseline Subjects’ Characteristics
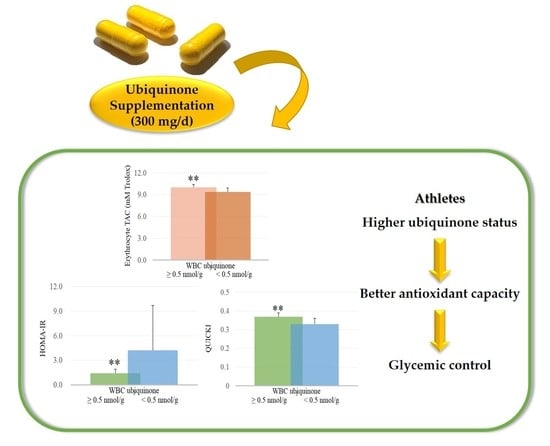
3.2. Ubiquinone Status, Glycemic Profile, Oxidative Stress, and Antioxidant Capacity
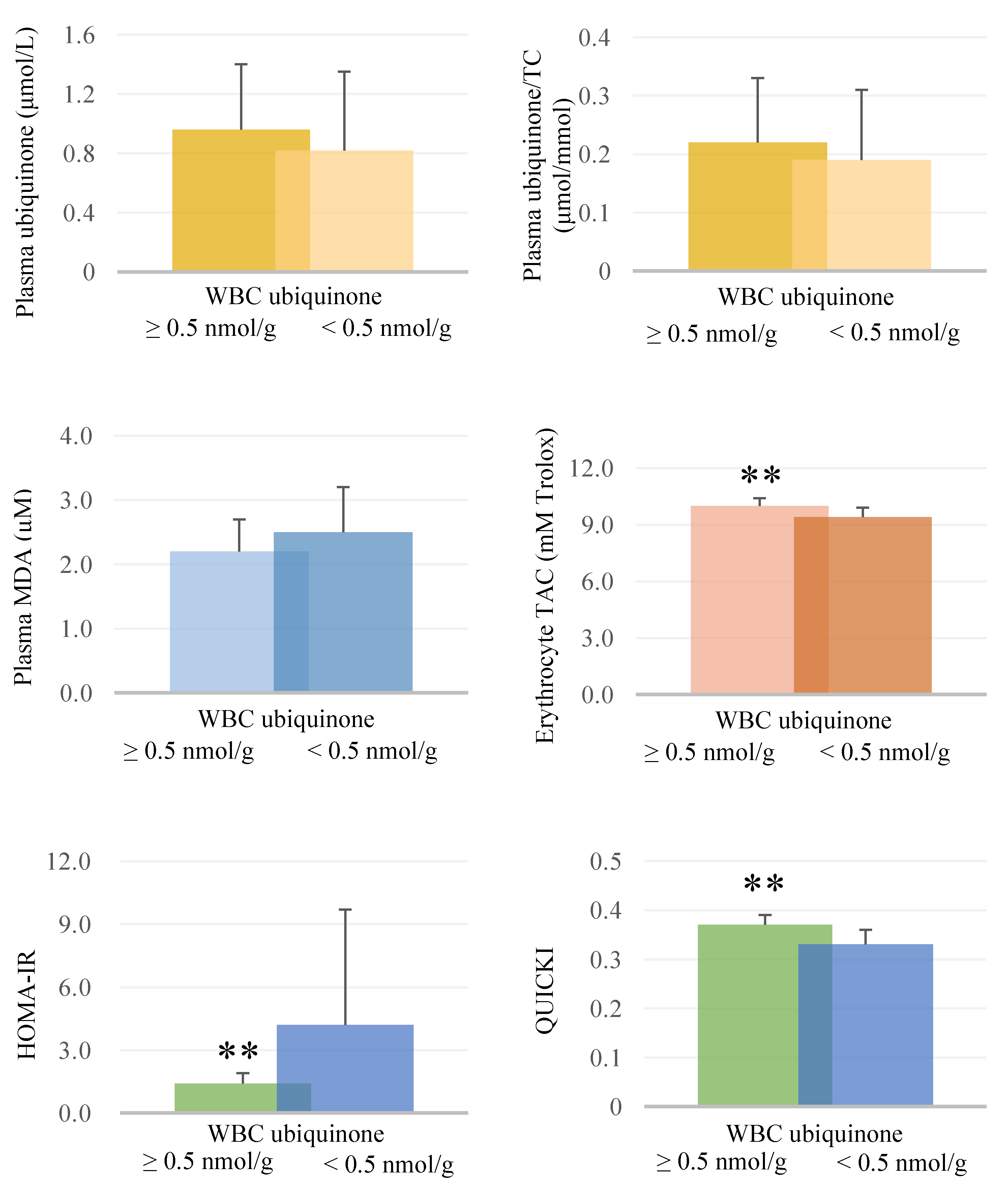
3.3. Correlations between Ubiquinone Status and Oxidative Stress, Total Antioxidant Capacity, and Glycemic Parameters after Supplementation
3.4. Correlations between Oxidative Stress, Antioxidant Capacity, and Glycemic Parameters after Supplementation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gomez-Cabrera, M.C.; Domenech, E.; Viña, J. Moderate exercise is an antioxidant: Upregulation of antioxidant genes by training. Free Radic. Biol. Med. 2008, 44, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Pattwell, D.M.; McArdle, A.; Morgan, J.E.; Patridge, T.A.; Jackson, M.J. Release of reactive oxygen and nitrogen species from contracting skeletal muscle cells. Free Radic. Biol. Med. 2004, 37, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Pingitore, A.; Lima, G.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Mason, S.A.; Trewin, A.J.; Parker, L.; Wadley, G.D. Antioxidant supplements and endurance exercise: Current evidence and mechanistic insights. Redox Biol. 2020, 101471. [Google Scholar] [CrossRef] [PubMed]
- Enriquez, J.A.; Lenaz, G. Coenzyme q and the respiratory chain: Coenzyme q pool and mitochondrial supercomplexes. Mol. Syndromol. 2014, 5, 119–140. [Google Scholar] [CrossRef]
- Ernster, L.; Dallner, G. Biochemical, physiological and medical aspects of ubiquinone function. Biochim. Biophys. Acta 1995, 1271, 195–204. [Google Scholar] [CrossRef]
- Littarru, G.P.; Tiano, L. Bioenergetic and antioxidant properties of coenzyme Q10: Recent developments. Mol. Biotechnol. 2007, 37, 31–37. [Google Scholar] [CrossRef]
- Medelson, S.D. Nutritional supplements and metabolic syndeome. In Metabolic Syndrome and Psychiatric Illness; Elsevier Inc.: Amsterdam, The Netherlands, 2008; pp. 141–183. [Google Scholar]
- Sarmiento, A.; Diaz-Castro, J.; Pulido-Moran, M.; Kajarabille, N.; Guisado, R.; Ochoa, J.J. Coenzyme Q10 supplementation and exercise in healthy humans: A systematic review. Curr. Drug. Metab. 2016, 17, 345–358. [Google Scholar] [CrossRef]
- Ho, C.C.; Tseng, C.Y.; Chen, H.W.; Chiu, Y.W.; Tsai, M.C.; Chang, P.S.; Lin, P.T. Coenzyme Q10 status, glucose parameters, and antioxidative capacity in college athletes. J. Int. Soc. Sports. Nutr. 2020, 17, 5. [Google Scholar] [CrossRef]
- Mondazzi, L.; Arcelli, E. Glycemic index in sport nutrition. J. Am. Coll. Nutr. 2009, 28 (Suppl. 4), 455S–463S. [Google Scholar] [CrossRef]
- Walton, P.; Rhodes, E.C. Glycaemic index and optimal performance. Sports Med. 1997, 23, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.J.; Tseng, Y.F.; Yen, C.H.; Lin, P.T. Effects of coenzyme Q10 supplementation (300 mg/d) on antioxidation and anti-inflammation in coronary artery disease patients during statins therapy: A randomized, placebo-controlled trial. Nutr. J. 2013, 12, 142. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Huang, Y.C.; Cheng, S.B.; Huang, Y.T.; Lin, P.T. Effects of coenzyme Q10 supplementation on antioxidant capacity and inflammation in hepatocellular carcinoma patients after surgery: A randomized, placebo-controlled trial. Nutr. J. 2016, 15, 85. [Google Scholar] [CrossRef] [PubMed]
- Dagur, P.K.; McCoy, J.P., Jr. Collection, Storage, and Preparation of Human Blood Cells. Curr. Protoc. Cytom. 2015, 73, 5.1.1–5.1.16. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative insulin sensitivity check index: A simple, accurate method for assessing insulin sensitivity in humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Tatum, V.L.; Changchit, C.; Chow, C.K. Measurement of malondialdehyde by high performance liquid chromatography with fluorescence detection. Lipids 1990, 25, 226–229. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Littarru, G.P.; Mosca, F.; Fattorini, D.; Bompadre, S. Method to Assay Coenzyme Q10 in Blood Plasma or Blood Serum. U.S. Patent 7303921, 4 December 2007. Available online: https://patentimages.storage.googleapis.com/pdfs/6aadab0bc819eac3443b/US7303921.pdf (accessed on 8 November 2010).
- Emami, A. The Impact of Pre-Cooling and CoQ10 Supplementation on Mediators of Inflammatory Cytokines in Elite Swimmers. Nutr. Cancer 2020, 72, 41–51. [Google Scholar] [CrossRef]
- Emami, A.; Bazargani-Gilani, B. Effect of oral CoQ10 supplementation along with precooling strategy on cellular response to oxidative stress in elite swimmers. Food Funct. 2018, 9, 4384–4393. [Google Scholar] [CrossRef]
- Emami, A.; Tofighi, A.; Asri-Rezaei, S.; Bazargani-Gilani, B. Effect of Short-term Coenzyme Q10 Supplementation and Precooling on Serum Endogenous Antioxidant Enzymes of Elite Swimmers. J. Strength Cond. Res. 2018, 32, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Kon, M.; Tanabe, K.; Akimoto, T.; Kimura, F.; Tanimura, Y.; Shimizu, K.; Okamoto, T.; Kono, I. Reducing exercise-induced muscular injury in kendo athletes with supplementation of coenzyme Q10. Br. J. Nutr. 2008, 100, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Nagato, S.; Sakuraba, K.; Morio, K.; Sawaki, K. Short-term Ubiquinol-10 supplementation alleviates tissue damage in muscle and fatigue caused by strenuous exercise in male distance runners. Int. J. Vitam. Nutr. Res. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alf, D.; Schmidt, M.E.; Siebrecht, S.C. Ubiquinol supplementation enhances peak power production in trained athletes: A double-blind, placebo controlled study. J. Int. Soc. Sports Nutr. 2013, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Sarmiento, A.; Diaz-Castro, J.; Pulido-Moran, M.; Moreno-Fernandez, J.; Kajarabille, N.; Chirosa, I.; Guisado, I.M.; Chirosa, L.J.; Guisado, R.; Ochoa, J.J. Short-term ubiquinol supplementation reduces oxidative stress associated with strenuous exercise in healthy adults: A randomized trial. Biofactors 2016, 42, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Orlando, P.; Silvestri, S.; Galeazzi, R.; Antonicelli, R.; Marcheggiani, F.; Cirilli, I.; Bacchetti, T.; Tiano, L. Effect of ubiquinol supplementation on biochemical and oxidative stress indexes after intense exercise in young athletes. Redox Rep. 2018, 23, 136–145. [Google Scholar] [CrossRef]
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef]
- Bhagavan, H.N.; Chopra, R.K. Plasma coenzyme Q10 response to oral ingestion of coenzyme Q10 formulations. Mitochondrion 2007, 7, S78–S88. [Google Scholar] [CrossRef]
- López-Lluch, G.; Pozo-Cruz, J.D.; Sánchez-Cuesta, A.; Cortés-Rodríguez, A.B.; Navas, P. Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutrition 2019, 57, 133–140. [Google Scholar] [CrossRef]
- Kras, K.A.; Hoffman, N.; Roust, L.R.; Benjamin, T.R.; De Filippis, E.A.; Katsanos, C.S. Adenosine Triphosphate Production of Muscle Mitochondria after Acute Exercise in Lean and Obese Humans. Med. Sci. Sports Exerc. 2019, 51, 445–453. [Google Scholar] [CrossRef]
- Diaz-Castro, J.; Moreno-Fernandez, J.; Chirosa, I.; Chirosa, L.J.; Guisado, R.; Ochoa, J.J. Beneficial Effect of Ubiquinol on Hematological and Inflammatory Signaling during Exercise. Nutrients 2020, 12, 424. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Ciccarelli, M.; Trimarco, B.; Iaccarino, G. Physical activity ameliorates cardiovascular health in elderly subjects: The functional role of the β adrenergic system. Front. Physiol. 2013, 4, 209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, C. Vasoprotection by dietary supplements and exercise: Role of TNFα signaling. Exp. Diabetes Res. 2012, 2012, 972679. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.L.; McMillin, S.L.; Weyrauch, L.A.; Witczak, C.A. Regulation of Skeletal Muscle Glucose Transport and Glucose Metabolism by Exercise Training. Nutrients 2019, 11, 2432. [Google Scholar] [CrossRef]
- Evans, J.L. Antioxidants: Do they have a role in the treatment of insulin resistance? Indian J. Med. Res. 2007, 125, 355–372. [Google Scholar]
- Jiménez-Maldonado, A.; Virgen-Ortiz, A.; Melnikov, V.; Rodríguez-Hernández, A.; Gamboa-Domínguez, A.; Montero, S.; Muñiz-Murguía, J.; Lemus, M.; Roces de Álvarez-Buylla, E. Effect of moderate and high intensity chronic exercise on the pancreatic islet morphometry in healthy rats: BDNF receptor participation. Islets 2017, 9, 1–10. [Google Scholar] [CrossRef]
- Powers, S.K.; Nelson, W.B.; Hudson, M.B. Exercise-induced oxidative stress in humans: Cause and consequences. Free Radic. Biol. Med. 2011, 51, 942–950. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Watts, G.F.; Playford, D.A.; Burke, V.; Croft, K.D. Coenzyme Q10 improves blood pressure and glycaemic control: A controlled trial in subjects with type 2 diabetes. Eur. J. Clin. Nutr. 2002, 56, 1137–1142. [Google Scholar] [CrossRef]
- Kolahdouz Mohammadi, R.; Hosseinzadeh-Attar, M.J.; Eshraghian, M.R.; Nakhjavani, M.; Khorami, E.; Esteghamati, A. The effect of coenzyme Q10 supplementation on metabolic status of type 2 diabetic patients. Minerva Gastroenterol. Dietol. 2013, 59, 231–236. [Google Scholar]
- Yen, C.H.; Chu, Y.J.; Lee, B.J.; Lin, Y.C.; Lin, P.T. Effect of liquid ubiquinol supplementation on glucose, lipids, and antioxidant capacity in type 2 diabetes patients: A double-blind, randomized, placebo-controlled trial. Br. J. Nutr. 2018, 120, 57–63. [Google Scholar] [CrossRef]
- Amin, M.M.; Asaad, G.F.; Abdel Salam, R.M.; El-Abhar, H.S.; Arbid, M.S. Novel CoQ10 antidiabetic mechanisms underlie its positive effect: Modulation of insulin and adiponectine receptors, tyrosine kinase, PI3K, glucose transporters, sRAGE and visfatin in insulin resistant/diabetic rats. PLoS ONE 2014, 9, e89169. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Montagnana, M.; Salvagno, G.L.; Franchini, M.; Guidi, G.C. Glycaemic control in athletes. Int. J. Sports Med. 2008, 29, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Tarperi, C.; Montagnana, M.; Danese, E.; Salvagno, G.L.; Schena, F. Challenges of diagnosing diabetes in endurance athletes. J. Clin. Pathol. 2018, 71, 945–946. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.A.; Murphy, M.P. Animal and human studies with the mitochondria-targeted antioxidant MitoQ. Ann. N. Y. Acad. Sci. 2010, 1201, 96–103. [Google Scholar] [CrossRef]
- Williamson, J.; Hughes, C.M.; Cobley, J.N.; Davison, G.W. The mitochondria-targeted antioxidant MitoQ, attenuates exercise-induced mitochondrial DNA damage. Redox Biol. 2020, 36, 101673. [Google Scholar] [CrossRef]
- Siciliano, G.; Chico, L.; Gerfo, A.L.; Simoncini, C.; Schirinzi, E.; Ricci, G. Exercise-Related Oxidative Stress as Mechanism to Fight Physical Dysfunction in Neuromuscular Disorders. Front. Physiol. 2020, 11, 451. [Google Scholar] [CrossRef]
- Bouitbir, J.; Charles, A.L.; Rasseneur, L.; Dufour, S.; Piquard, F.; Geny, B.; Zoll, J. Atorvastatin treatment reduces exercise capacities in rats: Involvement of mitochondrial impairments and oxidative stress. J. Appl. Physiol. 2011, 111, 1477–1483. [Google Scholar] [CrossRef]
- Thirupathi, A.; Pinho, R.A.; Chang, Y.Z. Physical exercise: An inducer of positive oxidative stress in skeletal muscle aging. Life Sci. 2020, 252, 117630. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef]
- Pravst, I.; Zmitek, K.; Zmitek, J. Coenzyme Q10 contents in foods and fortification strategies. Crit. Rev. Food Sci. Nutr. 2010, 50, 269–280. [Google Scholar] [CrossRef]
| Characteristics | Ubiquinone (n = 15) | Placebo (n = 14) | p Value |
|---|---|---|---|
| Age (years) | 19.9 ± 1.3 (20.0) 1 | 19.6 ± 1.3 (19.0) | 0.56 |
| Sports | 0.87 | ||
| Soccer (n, %) | 9 (60.0%) | 7 (50.0%) | |
| Males/Females (n) | 4/5 | 5/2 | 0.36 |
| Taekwondo (n, %) | 6 (40.0%) | 7 (50.0%) | |
| Males/Females (n) | 4/2 | 7/1 | 0.56 |
| Body mass index (kg/m2) | 22.4 ± 1.8 (22.8) | 22.1 ± 2.8 (22.2) | 0.76 |
| Waist-hip ratio | 0.79 ± 0.04 (0.79) | 0.79 ± 0.05 (0.78) | 0.76 |
| Systolic blood pressure (mmHg) | 116.4 ± 13.6 (114.0) | 125.2 ± 13.9 (120.0) | 0.10 |
| Diastolic blood pressure (mmHg) | 67.9 ± 9.3 (65.0) | 70.4 ± 7.4 (70.0) | 0.45 |
| Hematology | |||
| Albumin (g/L) | 49.9 ± 3.0 (51.0) | 50.1 ± 2.6 (49.0) | 0.84 |
| Blood urea nitrogen (mmol/L) | 5.2 ± 1.2 (5.4) | 5.1 ± 1.3 (5.0) | 0.93 |
| Creatinine (μmol/L) | 68.6 ± 11.4 (71.7) | 84.0 ± 14.1 (85.7) | 0.39 |
| GOT (U/L) | 20.5 ± 6.9 (21.0) | 20.0 ± 3.9 (19.0) | 0.83 |
| GPT (U/L) | 17.6 ± 11.2 (13.0) | 15.9 ± 5.0 (16.0) | 0.65 |
| Uric acid (μmol/L) | 350.9 ± 77.3 (380.7) | 374.8 ± 65.4 (398.5) | 0.43 |
| TC (mmol/L) | 4.4 ± 0.7 (4.7) | 4.4 ± 0.9 (4.2) | 1.00 |
| Triglyceride (mmol/L) | 0.69 ± 0.30 (0.55) | 0.84 ± 0.81 (0.62) | 0.97 |
| LDL-C (mmol/L) | 2.4 ± 0.6 (2.5) | 2.6 ± 0.8 (2.4) | 0.46 |
| HDL-C (mmol/L) | 1.9 ± 0.3 (1.9) | 1.6 ± 0.4 (1.6) | 0.07 |
| Parameters | Ubiquinone (n = 15) | Placebo (n = 14) | p Value |
|---|---|---|---|
| Ubiquinone status | |||
| Plasma ubiquinone (μmol/L)-Baseline | 0.57 ± 0.18 (0.53) 1 | 0.58 ± 0.16 (0.54) | 0.92 |
| Plasma ubiquinone (μmol/L)-12 weeks | 1.14 ± 0.54 (1.06) * | 0.60 ± 0.20 (0.60) | <0.01 |
| Plasma ubiquinone/TC (μmol/mmol)-Baseline | 0.13 ± 0.03 (0.13) | 0.13 ± 0.04 (0.13) | 0.66 |
| Plasma ubiquinone/TC (μmol/mmol)-12 weeks | 0.27 ± 0.12 (0.26) * | 0.14 ± 0.04 (0.14) | <0.01 |
| WBC ubiquinone (nmol/g)-Baseline | 0.42 ± 0.32 (0.42) | 0.31 ± 0.21 (0.30) | 0.28 |
| WBC ubiquinone (nmol/g)-12 weeks | 0.75 ± 0.71 (0.48) * | 0.55 ± 0.43 (0.49) * | 0.45 |
| Glycemic profile | |||
| Fasting glucose (mmol/L)-Baseline | 4.7 ± 0.5 (4.7) | 5.0 ± 0.3 (4.9) | 0.10 |
| Fasting glucose (mmol/L)-12 weeks | 5.0 ± 1.0 (4.8) | 4.7 ± 0.4 (4.7) | 0.46 |
| HbA1c (%)-Baseline | 5.4 ± 0.4 (5.4) | 5.5 ± 0.2 (5.6) | 0.38 |
| HbA1c (%)-12 weeks | 5.3 ± 0.4 (5.3) † | 5.4 ± 0.3 (5.4) * | 0.55 |
| Oxidative stress | |||
| Plasma MDA (μM)-Baseline | 2.5 ± 0.5 (2.5) | 2.3 ± 0.4 (2.4) | 0.31 |
| Plasma MDA (μM)-12 weeks | 2.4 ± 0.7 (2.4) | 2.4 ± 0.7 (2.3) | 1.00 |
| Erythrocyte MDA (nmol/mg protein)-Baseline | 3.7 ± 0.3 (3.7) | 3.8 ± 0.3 (3.8) | 0.34 |
| Erythrocyte MDA (nmol/mg protein)-12 weeks | 3.6 ± 0.3 (3.6) | 4.1 ± 0.5 (4.0) * | <0.01 |
| Antioxidant capacity | |||
| Serum TAC (mM Trolox)-Baseline | 5.7 ± 0.4 (5.8) | 5.7 ± 0.3 (5.8) | 0.53 |
| Serum TAC (mM Trolox)-12 weeks | 5.2 ± 0.4 (5.2) * | 5.2 ± 0.2 (5.2) * | 0.88 |
| Erythrocyte TAC (mM Trolox)-Baseline | 10.4 ± 0.5 (10.5) | 10.7 ± 0.6 (10.6) | 0.16 |
| Erythrocyte TAC (mM Trolox)-12 weeks | 9.7 ± 0.5 (9.8) * | 9.7 ± 0.6 (9.8) * | 0.90 |
| Parameters | Plasma Ubiquinone (μmol/L) | Plasma Ubiquinone/TC (μmol/mmol) | WBC Ubiquinone (nmol/g) |
|---|---|---|---|
| Correlation Coefficients 1 | |||
| Oxidative stress | |||
| Plasma MDA (μM) | −0.35 † | −0.40 * | −0.15 |
| Erythrocyte MDA (nmol/mg protein) | −0.46 * | −0.43 * | −0.44 ** |
| Antioxidant capacity | |||
| Serum TAC (mM Trolox) | 0.27 | 0.23 | 0.57 ** |
| Erythrocyte TAC (mM Trolox) | −0.09 | −0.10 | 0.19 |
| Glycemic parameters | |||
| Fasting glucose (mmol/L) | −0.02 | 0.01 | −0.09 |
| HbA1c (%) | −0.23 | −0.19 | −0.46 * |
| HOMA-IR | −0.15 | −0.19 | −0.67 ** |
| QUICKI | 0.15 | 0.19 | 0.67 ** |
| Parameters | Plasma MDA (μM) | Erythrocyte MDA (nmol/mg Protein) | Serum TAC (mM Trolox) | Erythrocyte TAC (mM Trolox) |
|---|---|---|---|---|
| Correlation Coefficients 1 | ||||
| Fasting glucose (mmol/L) | 0.06 | −0.03 | −0.05 | 0.18 |
| HbA1c (%) | 0.28 | 0.40 * | −0.25 | −0.14 |
| HOMA-IR | 0.22 | 0.13 | −0.44 * | −0.04 |
| QUICKI | −0.22 | −0.13 | 0.44 * | 0.04 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, C.-C.; Chang, P.-S.; Chen, H.-W.; Lee, P.-F.; Chang, Y.-C.; Tseng, C.-Y.; Lin, P.-T. Ubiquinone Supplementation with 300 mg on Glycemic Control and Antioxidant Status in Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Antioxidants 2020, 9, 823. https://doi.org/10.3390/antiox9090823
Ho C-C, Chang P-S, Chen H-W, Lee P-F, Chang Y-C, Tseng C-Y, Lin P-T. Ubiquinone Supplementation with 300 mg on Glycemic Control and Antioxidant Status in Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Antioxidants. 2020; 9(9):823. https://doi.org/10.3390/antiox9090823
Chicago/Turabian StyleHo, Chien-Chang, Po-Sheng Chang, Hung-Wun Chen, Po-Fu Lee, Yun-Chi Chang, Ching-Yu Tseng, and Ping-Ting Lin. 2020. "Ubiquinone Supplementation with 300 mg on Glycemic Control and Antioxidant Status in Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial" Antioxidants 9, no. 9: 823. https://doi.org/10.3390/antiox9090823
APA StyleHo, C.-C., Chang, P.-S., Chen, H.-W., Lee, P.-F., Chang, Y.-C., Tseng, C.-Y., & Lin, P.-T. (2020). Ubiquinone Supplementation with 300 mg on Glycemic Control and Antioxidant Status in Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Antioxidants, 9(9), 823. https://doi.org/10.3390/antiox9090823