Cudratricusxanthone O Inhibits H2O2-Induced Cell Damage by Activating Nrf2/HO-1 Pathway in Human Chondrocytes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Materials and Isolation of Compounds
2.3. Cell Culture
2.4. Cell Viability and Coefficient Assays
2.5. Western Blot Analysis
2.6. Cytosolic and Nuclear Protein Extraction
2.7. Measurement of ROS Generation
2.8. RT-qPCR Analysis
2.9. Annexin V-FITC/PI Apoptosis Assay
2.10. Statistical Analysis
3. Results
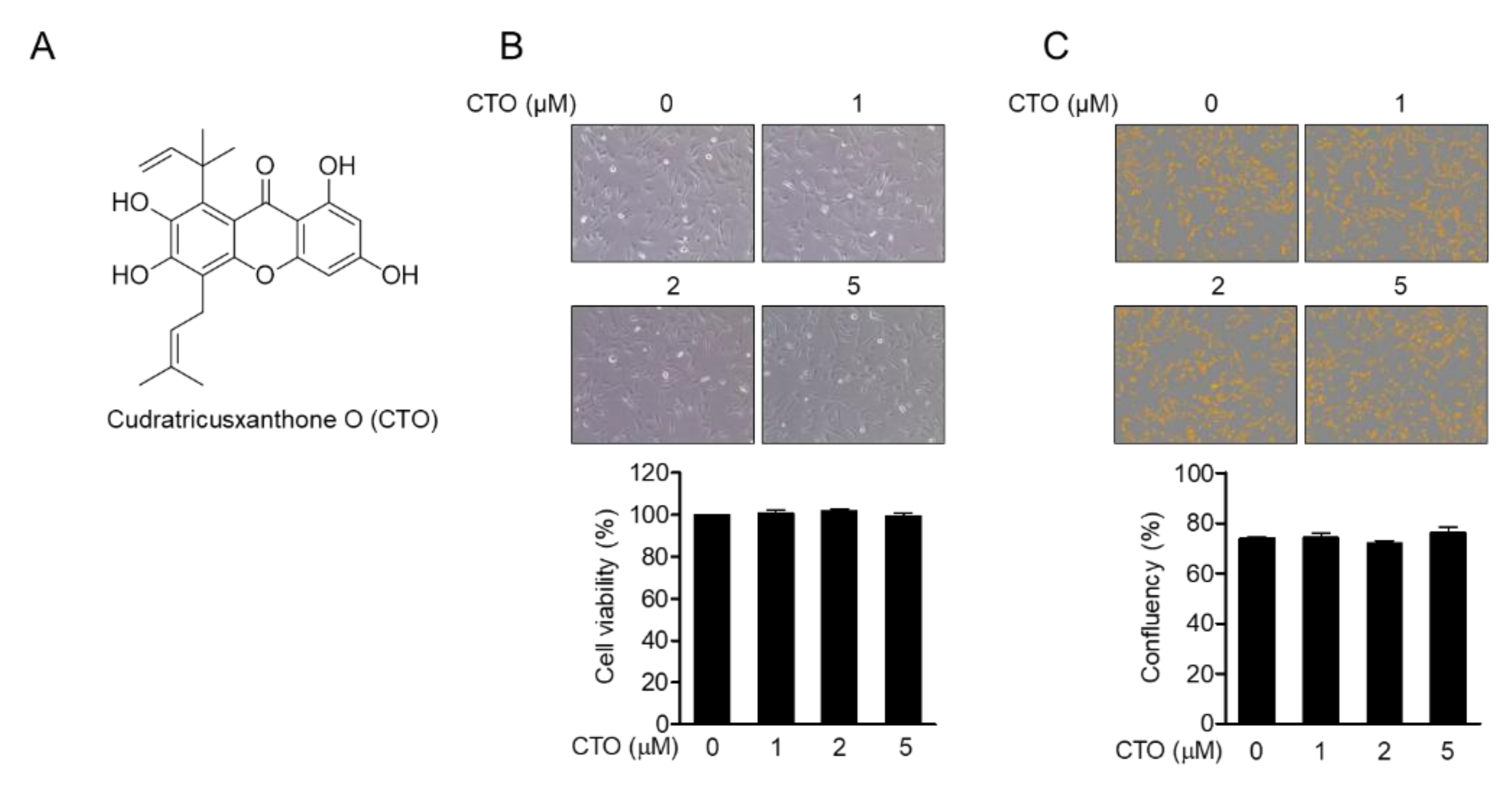
3.1. CTO Is Not Cytotoxic to SW1353 Chondrocytes
3.2. CTO Induces HO-1 Expression by Promotion of Nrf2 Translocation
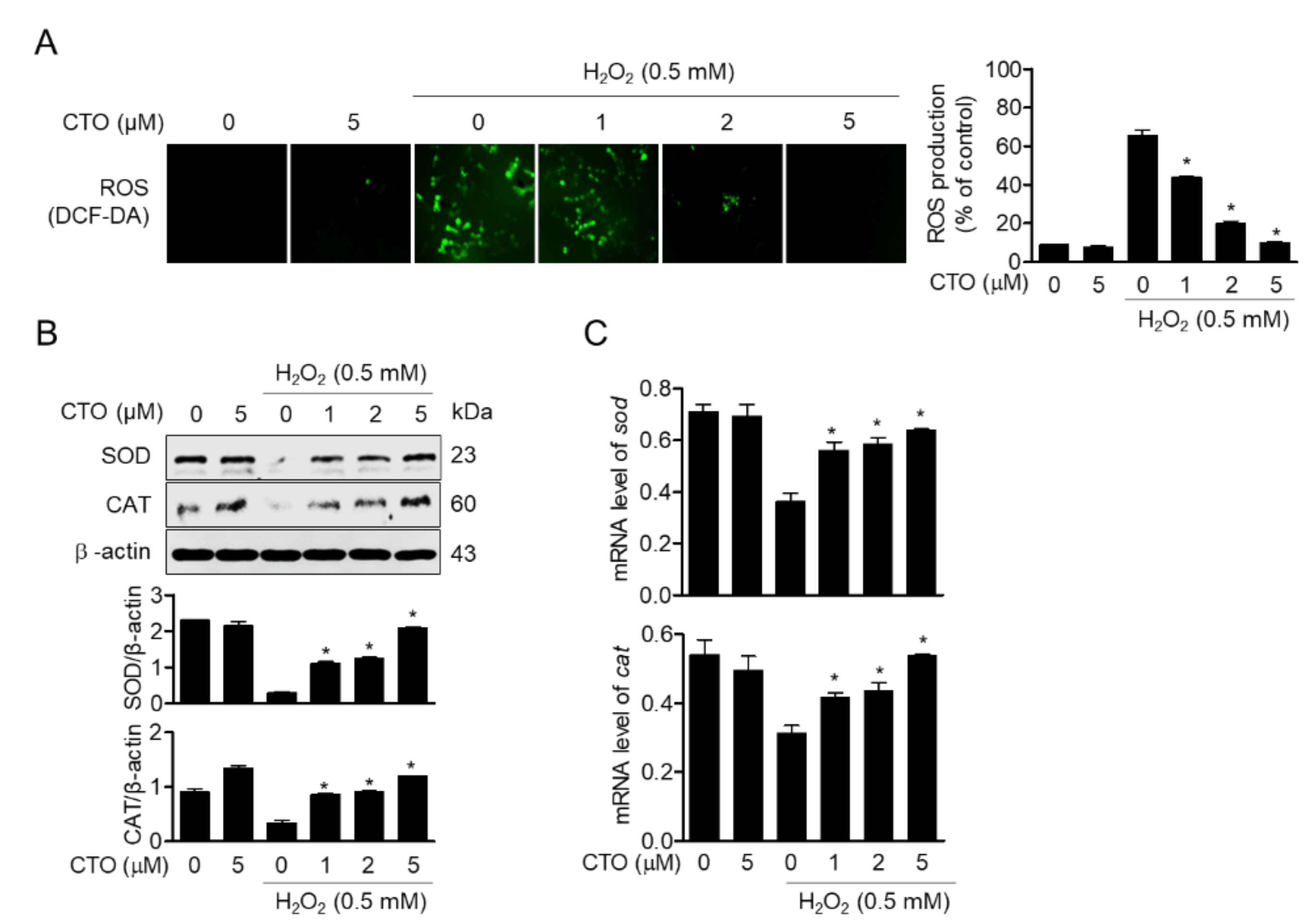
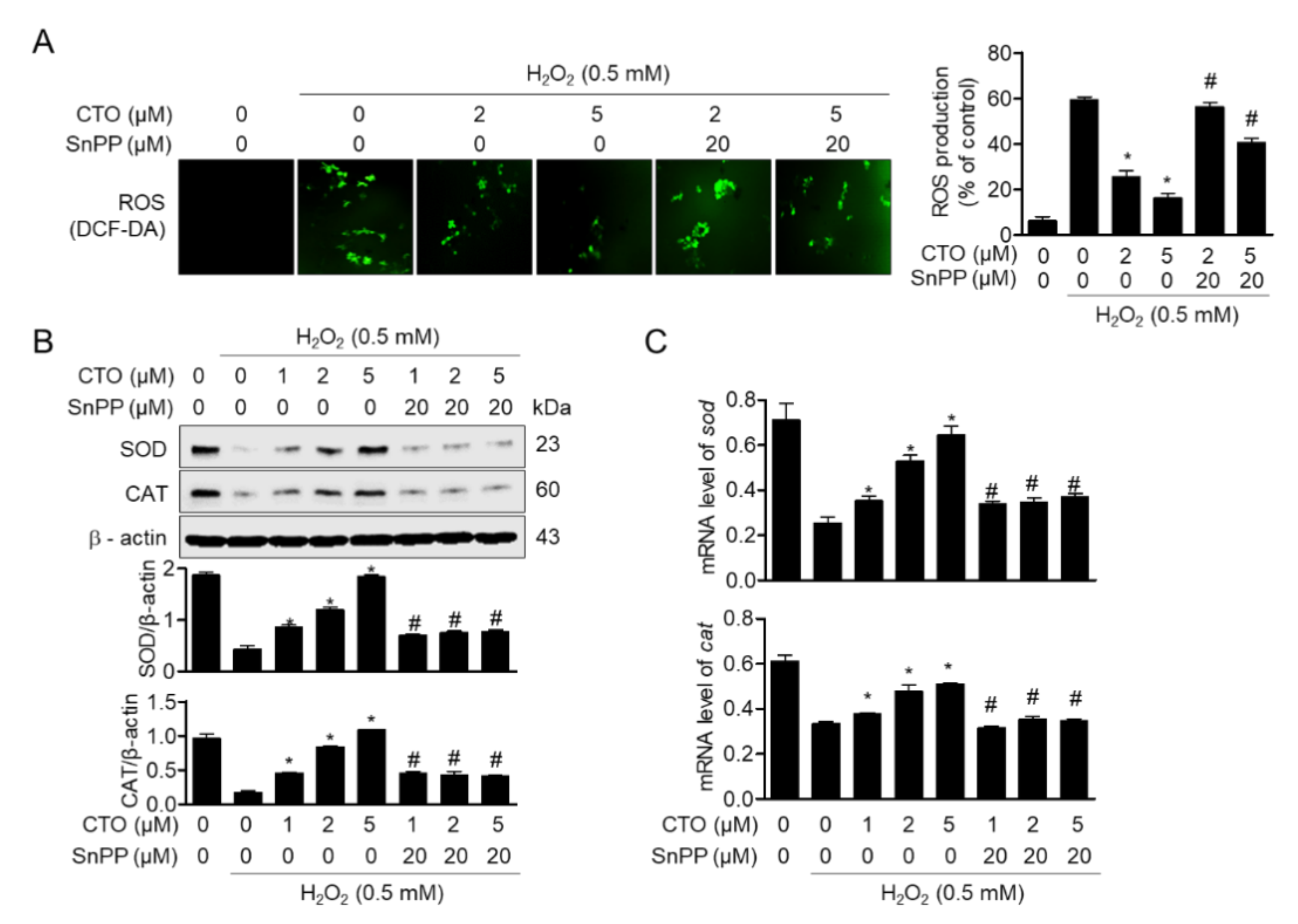
3.3. CTO Suppresses ROS Production by Inducing SOD and CAT in SW1353 Cells
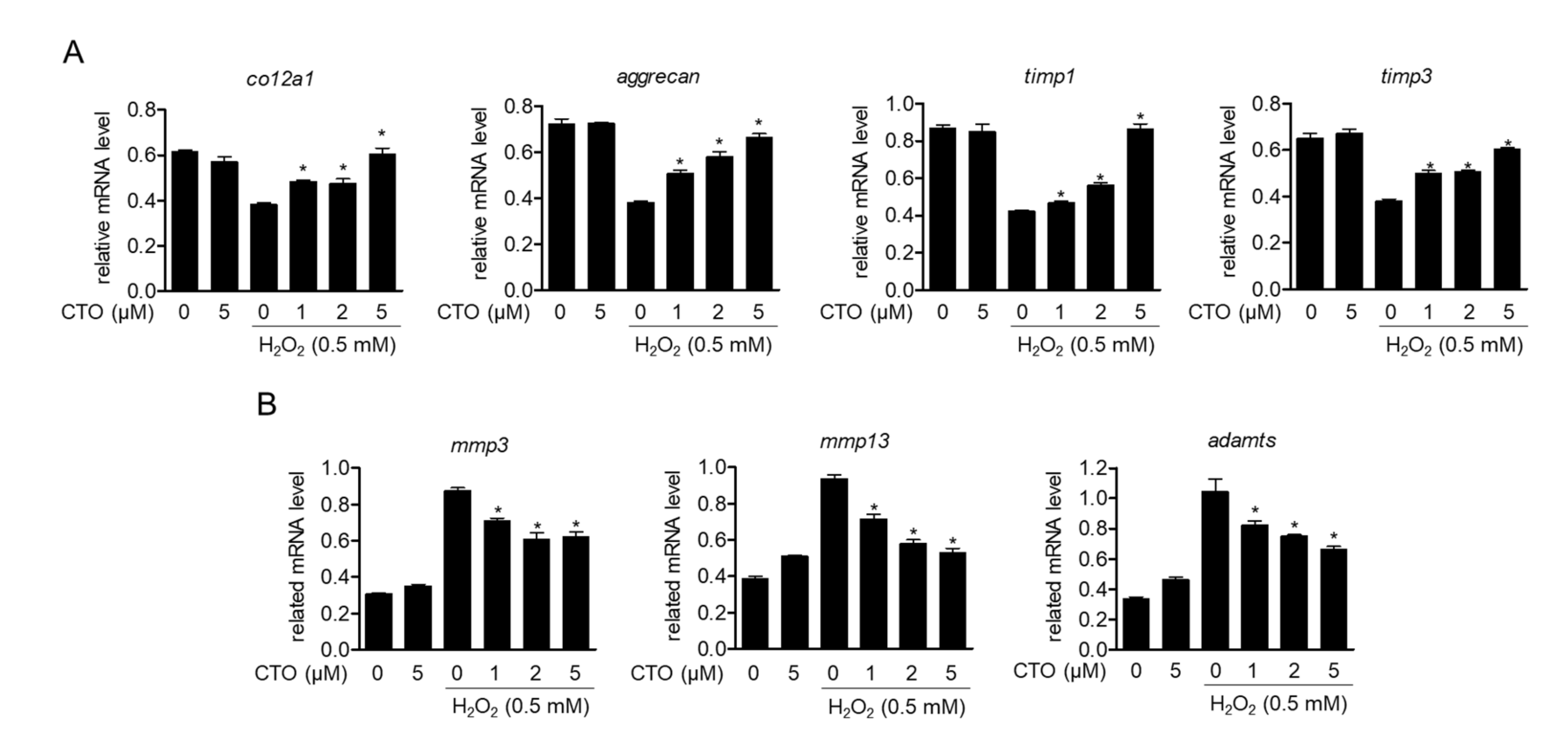
3.4. CTO Up-Regulates Chondrocytes-Specific Genes but Inhibits the Expression of MMPs in H2O2 Treated SW1353 Cells
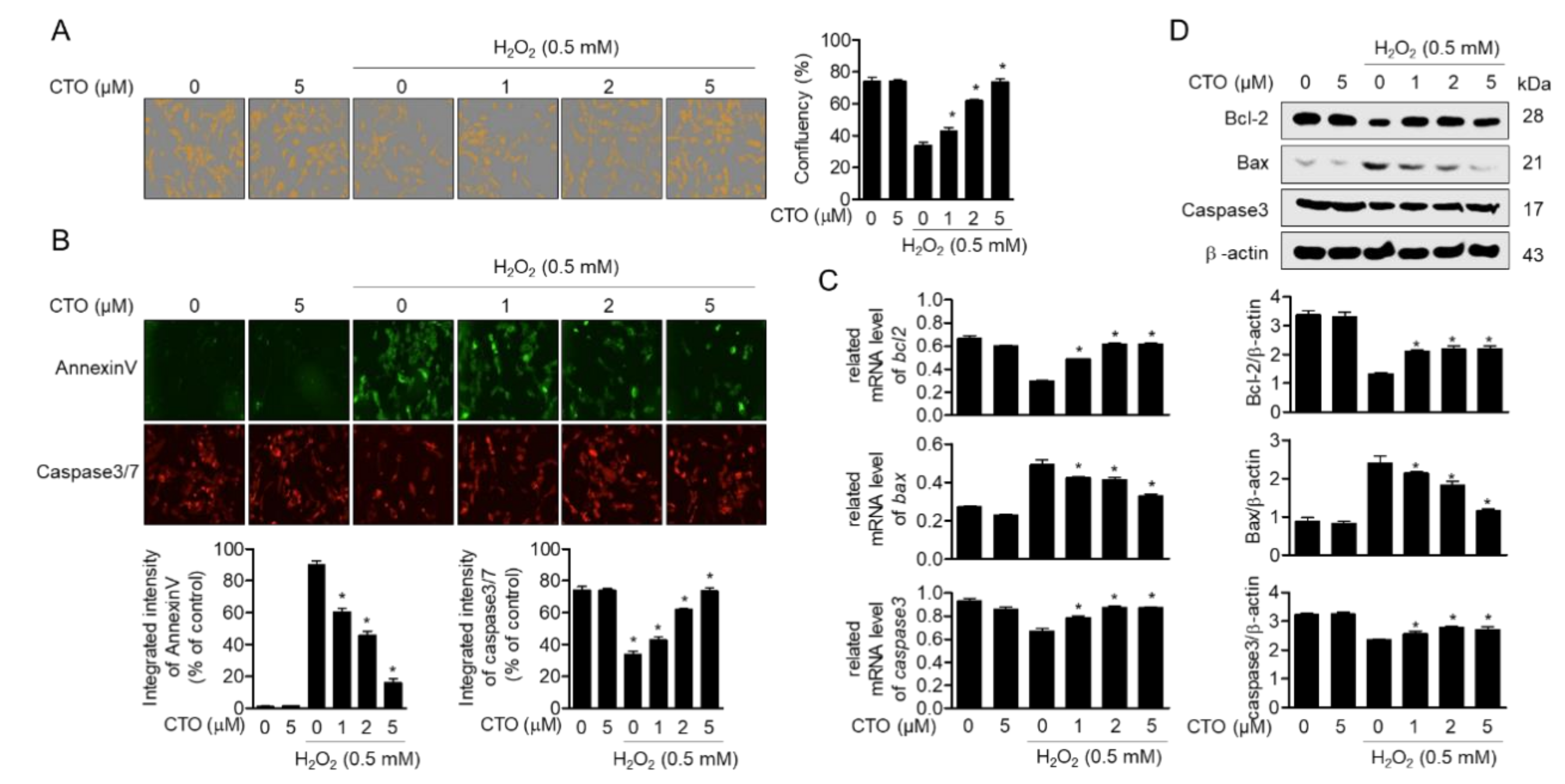
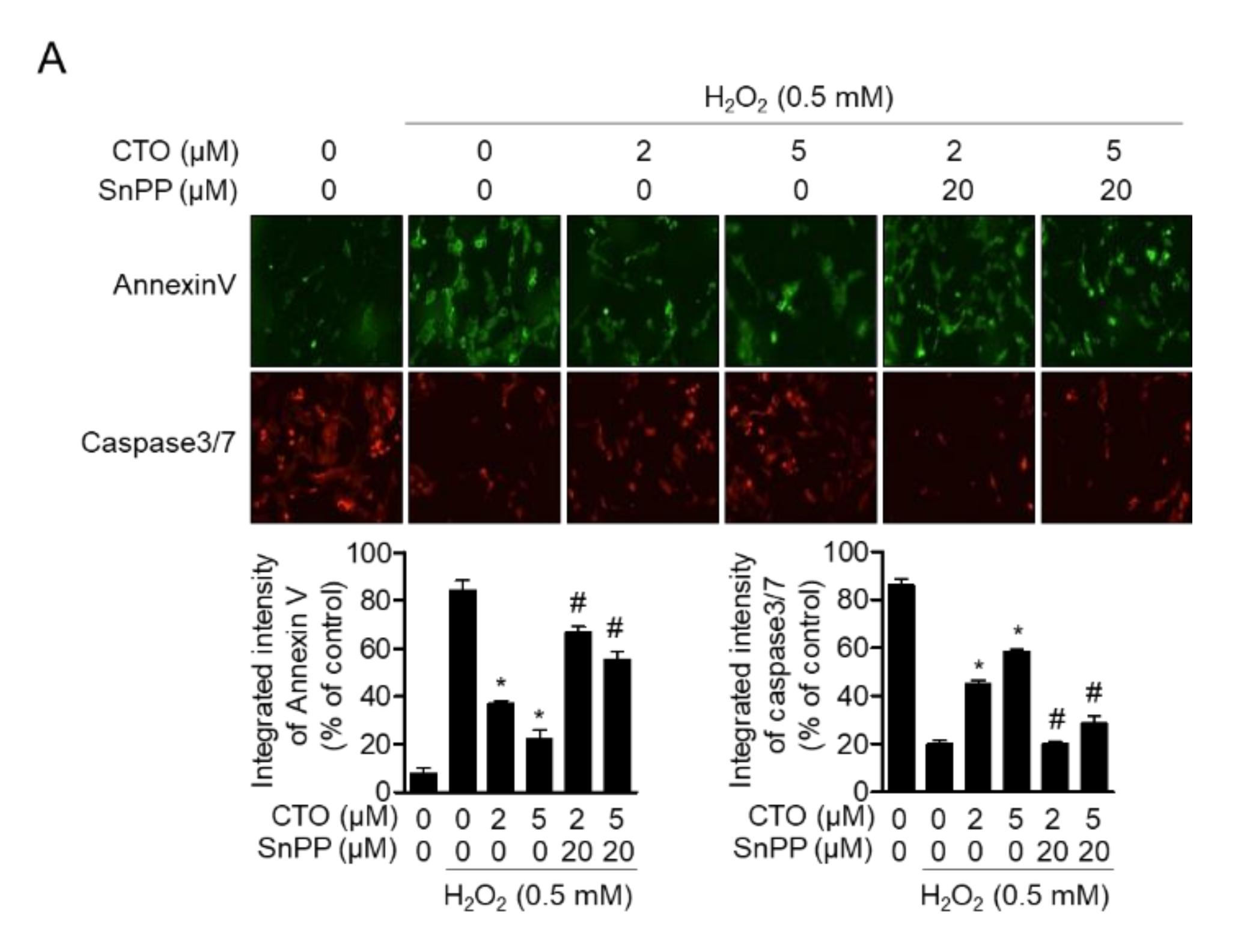
3.5. CTO Inhibits Apoptotic Pathway Induced by Treatment with H2O2 in SW1353 Cells
3.6. Upregulated HO-1 by Pre-Treatment with CTO Protects SW1353 Cells from ROS Production Induced by H2O2
3.7. Induced HO-1 by CTO Regulates Chondrocytes-Specific Genes and Expression of mmps in H2O2 Treated SW1353 Cell
3.8. Enhanced HO-1 by CTO Shows Protective Effect from Apoptosis Induced by H2O2 in SW1353 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.J. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res. 2017, 5, 16044. [Google Scholar] [CrossRef] [PubMed]
- Felson, D.T.; Anderson, J.J.; Naimark, A. The prevalence of knee osteoarthritis in the elderly. The framingham osteoarthritis study. Arthritis Rheum. 1987, 30, 914–918. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Hayami, T.; Funaki, H.; Yaoeda, K. Expression of the cartilage derived anti-angiogenic factor chondromodulin-I decreases in the early stage of experimental osteoarthritis. J. Rheumatol. 2003, 30, 2207–2217. [Google Scholar]
- Pacifici, M.; Koyama, E.; Iwamoto, M.; Gentili, C. Development of articular cartilage: What do we know about it and how may it occur? Connect Tissue Res. 2000, 41, 175–184. [Google Scholar] [CrossRef]
- Mendes, A.F.; Carvalho, A.P.; Caramona, M.M.; Lopes, M.C. Diphenyleneiodonium inhibits NF-kappaB activation and iNOS expression induced by IL-1beta: Involvement of reactive oxygen species. Mediators Inflamm. 2001, 10, 209–215. [Google Scholar] [CrossRef] [Green Version]
- Ndisang, J.F. Synergistic interaction between heme oxygenase (HO) and nuclear-factor E2-related factor-2 (Nrf2) against oxidative stress in cardiovascular related diseases. Curr. Pharm. Des. 2017, 23, 1465–1470. [Google Scholar] [CrossRef]
- Lee, D.H.; Park, J.S.; Lee, Y.S.; Sung, S.H.; Lee, Y.H.; Bae, S.H. The hypertension drug, verapamil, activates Nrf2 by promoting p62-dependent autophagic Keap1 degradation and prevents acetaminophen-induced cytotoxicity. BMB Rep. 2017, 50, 91–96. [Google Scholar] [CrossRef]
- Jaramillo, M.C.; Zhang, D.D. The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Genes Dev. 2013, 27, 2179–2191. [Google Scholar] [CrossRef] [Green Version]
- Ryter, S.W.; Tyrrell, R.M. The heme synthesis and degradation pathways: Role in oxidant sensitivity. Heme oxygenase has both pro- and antioxidant properties. Free Radic. Biol. Med. 2000, 28, 289–309. [Google Scholar] [CrossRef]
- Takahashi, T.; Morita, K.; Akagi, R.; Sassa, S. Heme oxygenase-1: A novel therapeutic target in oxidative tissue injuries. Curr. Med. Chem. 2004, 11, 1545–1561. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Lu, F.; Latham, C.; Zander, D.S.; Visner, G.A. Heme oxygenase-1 expression in human lungs with cystic fibrosis and cytoprotective effects against Pseudomonas aeruginosa in vitro. Am. J. Respir. Crit. Care Med. 2004, 170, 633–640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hiep, N.T.; Kwon, J.; Kim, D.W.; Hwang, B.Y.; Lee, H.J.; Mar, W.; Lee, D. Isoflavones with neuroprotective activities from fruits of Cudrania tricuspidata. Phytochemistry 2015, 111, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ha, H.; Lee, J.K.; Seo, C.s.; Lee, N.h.; Jung, D.Y.; Park, S.J.; Shin, H.K. The fruits of Cudrania tricuspidata suppress development of atopic dermatitis in NC/Nga mice. Phytother. Res. 2012, 26, 594–599. [Google Scholar] [CrossRef]
- Han, X.H.; Hong, S.S.; Jin, Q.; Li, D.; Kim, H.K.; Lee, J.; Kwon, S.H.; Lee, D.; Lee, C.K.; Lee, M.K. Prenylated and benzylated flavonoids from the fruits of Cudrania tricuspidata. J. Nat. Prod. 2008, 72, 164–167. [Google Scholar] [CrossRef]
- Tian, Y.H.; Kim, H.C.; Cui, J.M.; Kim, Y.C. Hepatoprotective constituents of Cudrania tricuspidata. Arch. Pharm. Res. 2005, 28, 44–48. [Google Scholar] [CrossRef]
- Jeong, G.S.; An, R.B.; Pae, H.O.; Chung, H.T.; Yoon, K.H.; Kang, D.G.; Lee, H.S.; Kim, Y.C. Cudratricusxanthone A protects mouse hippocampal cells against glutamate-induced neurotoxicity via the induction of heme oxygenase-1. Planta Med. 2008, 74, 1368–1373. [Google Scholar] [CrossRef]
- Quang, T.H.; Ngan, N.T.; Yoon, C.S.; Cho, K.H.; Kang, D.G.; Lee, H.S.; Kim, Y.C.; Oh, H. Protein tyrosine phosphatase 1B inhibitors from the roots of Cudrania tricuspidata. Molecules 2015, 20, 11173–11183. [Google Scholar] [CrossRef]
- Jeong, G.S.; Lee, D.S.; Kim, Y.C. Cudratricusxanthone A from Cudrania Tricuspidata suppresses pro-Inflammatory mediators through expression of anti-inflammatory Heme oxygenase-1 in RAW264.7 macrophages. Int. Immunopharmacol. 2009, 9, 241–246. [Google Scholar] [CrossRef]
- Xiaolei, X.; Xudan, L.; Yingchun, Y.; Jianyi, H.; Mengqi, J.; Yue, H.; Xiaotong, L.; Li, L.; Hailun, G. Resveratrol Exerts Anti-Osteoarthritic Effect by Inhibiting TLR4/NF-κB Signaling Pathway via the TLR4/Akt/FoxO1 Axis in IL-1β-Stimulated SW1353 Cells. Drug Des. Dev. Ther. 2020, 14, 2079–2090. [Google Scholar]
- Huang, T.C.; Chang, W.T.; Hu, Y.C.; Hsieh, B.S.; Cheng, H.L.; Yen, J.H.; Chiu, P.R.; Chang, K.L. Zinc Protects Articular Chondrocytes through Changes in Nrf2-Mediated Antioxidants, Cytokines and Matrix Metalloproteinases. Nutrients 2018, 10, 471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, C.; Jeong, J.W.; Lee, D.S.; Yim, M.J.; Lee, J.M.; Han, M.H.; Kim, S.; Kim, H.S.; Kim, G.Y.; Park, E.K.; et al. Sargassum serratifolium Extract Attenuates Interleukin-1_-Induced Oxidative Stress and Inflammatory Response in Chondrocytes by Suppressing the Activation of NF-kB, p38 MAPK, and PI3K/Akt. Int. J. Mol. Sci. 2018, 19, 2308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, G.U.; Lee, J.Y.; Kang, H.; Kim, T.Y.; Park, J.Y.; Hong, E.Y.; Shin, Y.H.; Jung, S.H.; Chang, H.B.; Kim, Y.H.; et al. Inhibition of Osteoarthritis-RelatedMolecules by Isomucronulatol 7-O-β-D-glucoside and Ecliptasaponin A in IL-1β-Stimulated Chondrosarcoma Cell Model. Molecules 2018, 23, 2807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, W.H.; Ku, S.K.; Kim, T.I.; Kim, E.N.; Park, E.K.; Jeong, G.S.; Bae, J.S. Inhibitory effects of cudratricusxanthone O on particulate matter-induced pulmonary injury. Int. J. Environ. Health Res. 2019, 1–14. [Google Scholar] [CrossRef]
- Cai, D.; Yin, S.; Yang, J.; Jiang, Q.; Cao, W. Histone deacetylase inhibition activates Nrf2 and protects against osteoarthritis. Arthritis Res. Ther. 2015, 17, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takada, T.; Miyaki, S.; Ishitobi, H.; Hirai, Y.; Nakasa, T.; Igarashi, K.; Lotz, M.K.; Ochi, M. Bach1 deficiency reduces severity of osteoarthritis through upregulation of heme oxygenase-1. Arthritis Res. Ther. 2015, 17, 285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jakus, V. The role of free radicals, oxidative stress and antioxidant systems in diabetic vascular disease. Bratislavské Lekárske Listy 2000, 101, 541–551. [Google Scholar]
- Kang, K.W.; Lee, S.J.; Kim, S.G. Molecular mechanism of Nrf2 activation by oxidative stress. Antioxid. Redox. Sign. 2005, 7, 1664–1673. [Google Scholar] [CrossRef]
- Wu, P.S.; Yen, J.H.; Kou, M.C.; Wu, M.J. “Luteolin and apigenin attenuate 4-hydroxy-2-nonenal-mediated cell death through modulation of UPR, Nrf2-ARE and MAPK pathways in PC12 cells. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, O.K.; Robertson, D.L.; Fagan, T.F. Different regulatory mechanisms modulate the expression of a dinoflagellate iron-superoxide dismutase. J. Biol. Chem. 2001, 276, 19989–19993. [Google Scholar] [CrossRef] [Green Version]
- Poole, A.R.; Kobayashi, M.; Yasuda, T.; Laverty, S.; Mwale, F.; Kojima, T.; Sakai, T.; Wahl, C.; El-Maadawy, S.; Webb, G.; et al. Type II collagen degradation and its regulation in articular cartilage in osteoarthritis. Ann. Rheum. Dis. 2002, 61, 78–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandya, S.; Achan, M.A.; Sudhakaran, P.R. Multiple matrix metalloproteinases in type II collagen induced arthritis. Indian J. Clin. Biochem. 2009, 24, 42–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mancini, F.; Nannarone, S.; Buratta, S.; Ferrara, G.; Stabile, A.M.; Vuerich, M. Effects of xylazine and dexmedetomidine on equine articular chondrocytes in vitro. Vet. Anaesth. Analg. 2017, 44, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Robertson, J.D.; Gogvadze, V.; Zhivotovsky, B.; Orrenius, S. Cytochrome c release from mitochondria proceeds by a two-step process. Proc. Natl. Acad. Sci. USA 2002, 99, 1259–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Multiple pathways of cytochrome c release from mitochondria in apoptosis. Biochim. Biophys. Acta. 2006, 1757, 639–647. [Google Scholar] [CrossRef] [Green Version]
- Kiraz, Y.; Adan, A.; Kartal Yandim, M.; Baran, Y. Major apoptotic mechanisms and genes involved in apoptosis. Tumour Biol. 2016, 37, 8471–8486. [Google Scholar] [CrossRef] [Green Version]
- Zhao, R.; Feng, J.; He, G. Hypoxia increases Nrf2-induced HO-1 expression via the PI3K/Akt pathway. Front. Biosci. 2016, 21, 385–396. [Google Scholar]


| Target Gene | Sequence | |
|---|---|---|
| col2a1 | Forward (5′-3′) | TGGACGCCATGAAGGTTTTCT |
| Reverse (3′-5′) | TGGGAGCCAGATTGTCATCTC | |
| aggrecan | Forward (5′-3′) | GAAGTGGCGTCCAAACCAA |
| Reverse (3′-5′) | CGTTCCATTCACCCCTCTCA | |
| timp1 | Forward (5′-3′) | AATTCCGACCTCGTCATCAG |
| Reverse (3′-5′) | GTTGTGGGACCTGTGGAAGT | |
| mmp3 | Forward (5′-3′) | GGT GTG GAG TTC CTG ATG TTG |
| Reverse (3′-5′) | AGC CTG GAG AAT GTG AGT GG | |
| mmp13 | Forward (5′-3′) | TCA GGA AAC CAG GTC TGG AG |
| Reverse (3′-5′) | TGA CGC GAA CAA TAC GGT TA | |
| adamts | Forward (5′-3′) | TTCCACGGCAGTGGTCTAAAG |
| Reverse (3′-5′) | CCACCAGGCTAACTGAATTACG | |
| sod | Forward (5′-3′) | GGGAGATGGCCCAACTACTG |
| Reverse (3′-5′) | CCAGTTGACATCGAACCGTT | |
| cat | Forward (5′-3′) | ATGGTCCATGCTCTCAAACC |
| Reverse (3′-5′) | CAGGTCATCCAATAGGAAGG | |
| bcl2 | Forward (5′-3′) | AGG CTG GGA TGC CTT TGT GG |
| Reverse (3′-5′) | GGG CAG GCA TGT TGA CTT CAC | |
| bax | Forward (5′-3′) | TTCTGACGGCAACTTCAACTGG |
| Reverse (3′-5′) | AGGAAGTCCAATGTCCAGCC | |
| caspase3 | Forward (5′-3′) | GGAATTGATGCGTGATGTT |
| Reverse (3′-5′) | TGGCTCAGAAGCACACAAAC | |
| gapdh | Forward (5′-3′) | ACCCAGAAGACTGTGGATGG |
| Reverse (3′-5′) | CACATTGGGGGTAGGAACAC | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, E.-N.; Lee, H.-S.; Jeong, G.-S. Cudratricusxanthone O Inhibits H2O2-Induced Cell Damage by Activating Nrf2/HO-1 Pathway in Human Chondrocytes. Antioxidants 2020, 9, 788. https://doi.org/10.3390/antiox9090788
Kim E-N, Lee H-S, Jeong G-S. Cudratricusxanthone O Inhibits H2O2-Induced Cell Damage by Activating Nrf2/HO-1 Pathway in Human Chondrocytes. Antioxidants. 2020; 9(9):788. https://doi.org/10.3390/antiox9090788
Chicago/Turabian StyleKim, Eun-Nam, Hyun-Su Lee, and Gil-Saeng Jeong. 2020. "Cudratricusxanthone O Inhibits H2O2-Induced Cell Damage by Activating Nrf2/HO-1 Pathway in Human Chondrocytes" Antioxidants 9, no. 9: 788. https://doi.org/10.3390/antiox9090788
APA StyleKim, E.-N., Lee, H.-S., & Jeong, G.-S. (2020). Cudratricusxanthone O Inhibits H2O2-Induced Cell Damage by Activating Nrf2/HO-1 Pathway in Human Chondrocytes. Antioxidants, 9(9), 788. https://doi.org/10.3390/antiox9090788




