Oxidative Stress and Vascular Dysfunction in the Retina: Therapeutic Strategies
Abstract
1. Introduction
2. Vascular Supply of the Retina
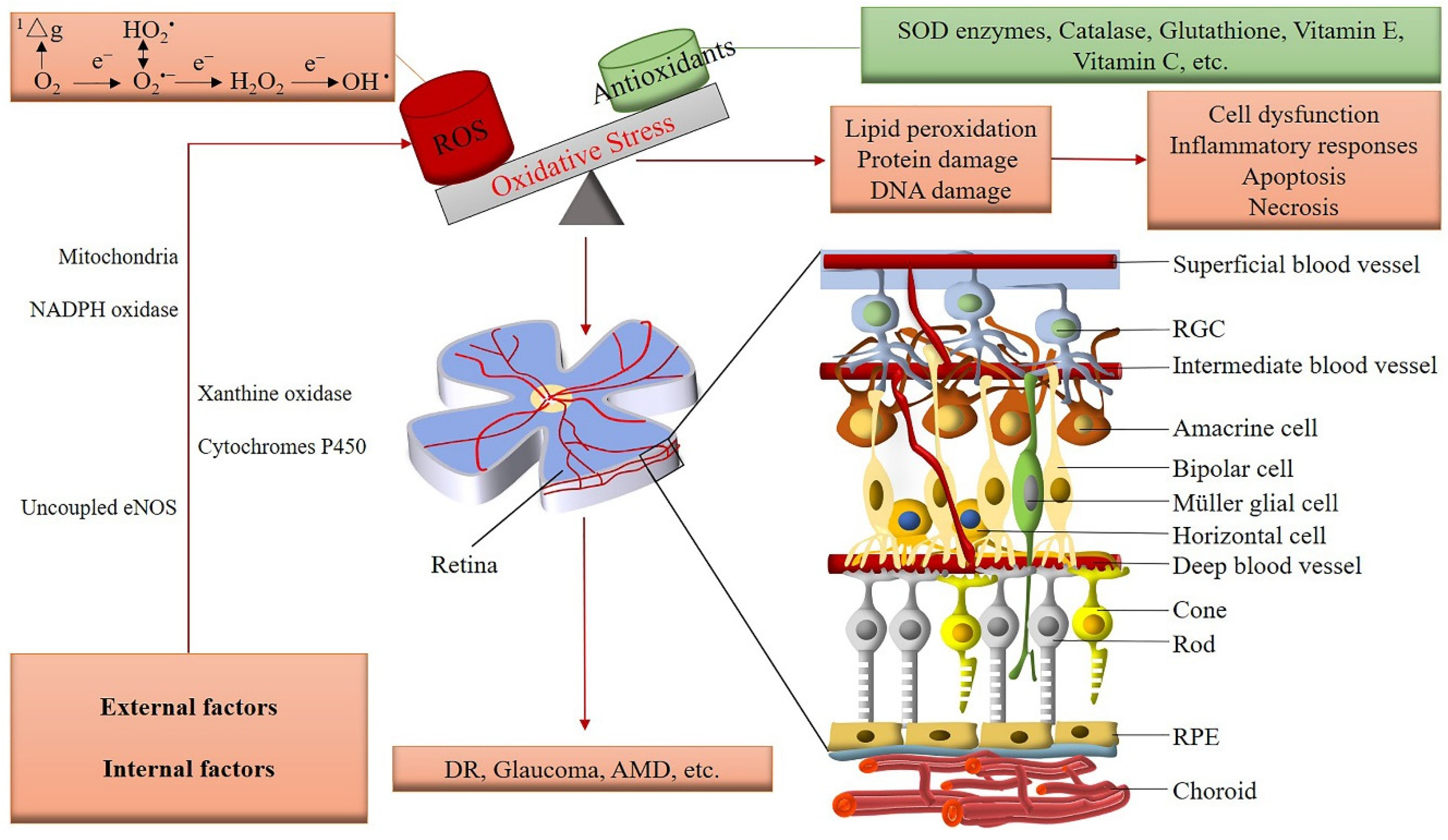
3. Regulation of the Retinal Vasculature and Involvement of ROS
4. Pathophysiological Role of ROS in the Retinal Vasculature
5. ROS and DR
6. ROS and Retinal/Ocular Ischemia
7. ROS and Glaucoma
8. AMD
9. Therapeutic Strategies to Reduce Oxidative Stress
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACE | Angiotensin-converting enzyme |
| AGEs | Advanced glycation endproducts |
| AMD | Age-related macular degeneration |
| Ang | Angiotensin |
| ApoE | Apolipoprotein E |
| AREDS | Age-Related Eye Disease Study |
| AR | Aldose reductase |
| AREs | Antioxidant response elements |
| AT1R | Angiotensin II receptor type 1 |
| AT2R | Angiotensin II receptor type 2 |
| A1 | Arginase 1 |
| A2 | Arginase 2 |
| ATP | Adenosine triphosphate |
| BH4 | Tetrahydrobiopterin |
| BH2 | 7,8-dihydrobiopterin |
| BRB | Blood-retinal barrier |
| CVD | Cardiovascular disease |
| CRAO | Central retinal artery occlusion |
| DAG | Diacylglycerol |
| DMOG | Dimethyloxalylglycine |
| DR | Diabetic retinopath |
| DUOX1 | Dual oxidase 1 |
| DUOX2 | Dual oxidase 2 |
| ETC | Electron transport chain |
| eNOS | endothelial nitric oxide synthase |
| ERK | Extracellular signal-related kinase |
| FA | Fenofibric acid |
| FH | Familial hypercholesterolemia |
| GCL | Ganglion cell layer |
| GPR91 | Succinate receptor G protein-coupled receptor-91 |
| HO-1 | Heme oxygenase-1 |
| H2O2 | Hydrogen peroxide |
| HO2• | Hydroperoxyl |
| HOC1 | Hypochlorous acid |
| HIF-1α | Hypoxia-inducible factor-1α |
| ICAM-1 | Intercellular adhesion molecule-1 |
| IL-1α | Interleukin-1 alpha |
| IL-β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| IVTA | Intravitreal injection of triamcinolone acetonide |
| I/R | Ischemia/reperfusion |
| iBRB | Inner blood-retinal barrier |
| iNOS | inducible nitric oxide synthase |
| JNK | c-Jun N-terminal kinase |
| KO | Knockout |
| LDL-C | Low-density lipoprotein cholesterol |
| L | Lutein |
| mtROS | mitochondria-derived ROS |
| mtDNA | mitochondrial DNA |
| MCP1 | Monocyte chemoattractant protein-1 |
| MD2 | Myeloid differentiation protein 2 |
| NADPH oxidase | Nicotinamide adenine dinucleotide phosphate oxidase |
| NF-κB | nuclear factor-kappa B |
| NLRP3 | Pyrin domain-containing 3 |
| NMDA | N-methyl-D-aspartate |
| NOX | NADPH oxidases |
| NO | Nitric oxide |
| NOS | nitric oxide synthase |
| nNOS | neuronal nitric oxide synthase |
| NRF2 | Nuclear factor erythroid-derived 2-related factor 2 |
| NTs | Neurotrophins |
| O2•− | Superoxide |
| O3 | Ozone |
| oBRB | Outer blood-retinal barrier |
| OCT | Octreotide |
| •OH | Hydroxyl radicals |
| ox-LDL | oxidized-LDL |
| ONOO— | Peroxynitrite |
| PARP | Poly (ADP-ribose) polymerase |
| PCSK9 | Proprotein convertase subtilisin/kexin type 9 |
| PDGF | Platelet-derived growth factor |
| PDGFRβ | Platelet-derived growth factor receptor beta |
| PKC | Protein kinase C |
| PPAR | Peroxisome proliferator-activated receptor |
| RAAS | Renin-angiotensin-aldosterone system |
| RAGE | Receptors for advanced glycation endproducts |
| RAS | Renin-angiotensin system |
| RCS | Reactive copper species |
| RGC | Retinal ganglion cell |
| RIS | Reactive iron species |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| RPE | Retinal pigment epithelium |
| ROO• | Peroxyl |
| RO• | Alkoxyl |
| RRMS | Relapsing-remitting multiple sclerosis |
| SIRT1 | NAD-dependent deacetylase sirtuin1 |
| SOD | Superoxide dismutase |
| TAGE | toxic AGEs |
| TLR4 | Toll-like receptor 4 |
| TLR2 | toll-like receptor 2 |
| TNF-α | Tumor necrosis factor-alpha |
| VEGF | vascular endothelial growth factor |
| Z | Zeaxanthin |
| 1△g | Singlet oxygen |
References
- Carrera-Julia, S.; Moreno, M.L. Antioxidant alternatives in the treatment of amyotrophic lateral sclerosis: A comprehensive review. Front. Physiol. 2020, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Kaludercic, N.; Di Lisa, F. Mitochondrial ros formation in the pathogenesis of diabetic cardiomyopathy. Front. Cardiovasc. Med. 2020, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, A.; Idelchik, M. Redox control of senescence and age-related disease. Redox Biol. 2017, 11, 91–102. [Google Scholar] [CrossRef] [PubMed]
- De Gara, L.; Foyer, C.H. Ying and yang interplay between reactive oxygen and reactive nitrogen species controls cell functions. Plant Cell Environ. 2017, 40, 459–461. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Stockwell, B.R. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 2014, 10, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Gyulkhandanyan, A.V.; Feeney, C.J. Modulation of mitochondrial membrane potential and reactive oxygen species production by copper in astrocytes. J. Neurochem. 2003, 87, 448–460. [Google Scholar] [CrossRef]
- Li, R.; Jia, Z. Defining ros in biology and medicine. React. Oxyg. Species (Apex) 2016, 1, 9–21. [Google Scholar] [CrossRef]
- Valko, M.; Izakovic, M. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem. 2004, 266, 37–56. [Google Scholar] [CrossRef]
- Munzel, T.; Camici, G.G. Impact of oxidative stress on the heart and vasculature: Part 2 of a 3-part series. J. Am. Coll. Cardiol. 2017, 70, 212–229. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef]
- Halliwell, B. Antioxidants in human health and disease. Annu. Rev. Nutr. 1996, 16, 33–50. [Google Scholar] [CrossRef] [PubMed]
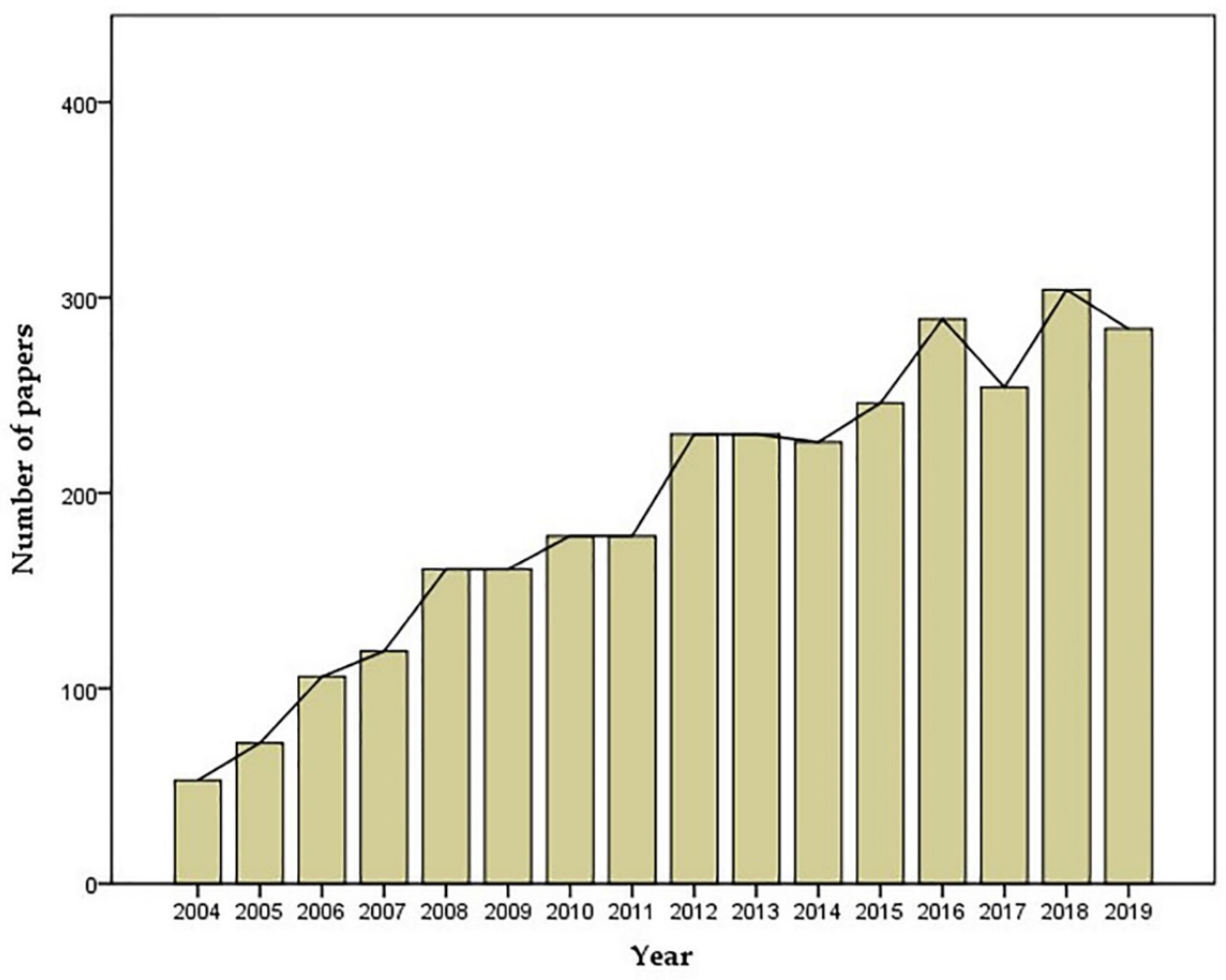
- Yearly Number of Publications Containing the Search Term “Oxidative Stress and Retina”. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 16 August 2020).
- Tsai, T.; Reinehr, S. Immune mediated degeneration and possible protection in glaucoma. Front. Neurosci. 2019, 13, 931. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Liu, Y.J. Carvedilol promotes retinal ganglion cell survival following optic nerve injury via ask1-p38 mapk pathway. CNS Neurol. Disord. Drug Targets 2019, 18, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Suzumura, A.; Kaneko, H. N-3 fatty acid and its metabolite 18-hepe ameliorate retinal neuronal cell dysfunction by enhancing müller bdnf in diabetic retinopathy. Diabetes 2020, 69, 724–735. [Google Scholar] [CrossRef]
- Rozing, M.P.; Durhuus, J.A. Age-related macular degeneration: A two-level model hypothesis. Prog. Retin. Eye Res. 2019, 76, 100825. [Google Scholar] [CrossRef]
- Gupta, M.P.; Herzlich, A.A. Retinal anatomy and pathology. Dev. Ophthalmol. 2016, 55, 7–17. [Google Scholar]
- Masland, R.H. The fundamental plan of the retina. Nat. Neurosci. 2001, 4, 877–886. [Google Scholar] [CrossRef]
- Garron, L.K. The ultrastructure of the retinal pigment epithelium with observations on the choriocapillaris and bruch’s membrane. Trans. Am. Ophthalmol. Soc. 1963, 61, 545–588. [Google Scholar]
- Gu, X.; Neric, N.J.; Crabb, J.S.; Crabb, J.W.; Bhattacharya, S.K.; Rayborn, M.E.; Hollyfield, J.G.; Bonilha, V.L. Age-related changes in the retinal pigment epithelium (rpe). PLoS ONE 2012, 7, e38673. [Google Scholar] [CrossRef]
- Kur, J.; Newman, E.A.; Chan-Ling, T. Cellular and physiological mechanisms underlying blood flow regulation in the retina and choroid in health and disease. Prog. Retin. Eye Res. 2012, 31, 377–406. [Google Scholar] [CrossRef]
- Sun, Y.; Smith, L.E.H. Retinal vasculature in development and diseases. Annu. Rev. Vis. Sci. 2018, 4, 101–122. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.Y.; Cringle, S.J.; Yu, P.K.; Balaratnasingam, C.; Mehnert, A.; Sarunic, M.V.; An, D.; Su, E.N. Retinal capillary perfusion: Spatial and temporal heterogeneity. Prog. Retin. Eye Res. 2019, 70, 23–54. [Google Scholar] [CrossRef] [PubMed]
- Giannaccini, M.; Usai, A.; Chiellini, F.; Guadagni, V.; Andreazzoli, M.; Ori, M.; Pasqualetti, M.; Dente, L.; Raffa, V. Neurotrophin-conjugated nanoparticles prevent retina damage induced by oxidative stress. Cell. Mol. Life Sci. 2018, 75, 1255–1267. [Google Scholar] [CrossRef] [PubMed]
- Caprara, C.; Thiersch, M.; Lange, C.; Joly, S.; Samardzija, M.; Grimm, C. Hif1a is essential for the development of the intermediate plexus of the retinal vasculature. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2109–2117. [Google Scholar] [CrossRef]
- Campbell, J.P.; Zhang, M.; Hwang, T.S.; Bailey, S.T.; Wilson, D.J.; Jia, Y.; Huang, D. Detailed vascular anatomy of the human retina by projection-resolved optical coherence tomography angiography. Sci. Rep. 2017, 7, 42201. [Google Scholar] [CrossRef]
- Moran, E.P.; Wang, Z.; Chen, J.; Sapieha, P.; Smith, L.E.; Ma, J.X. Neurovascular cross talk in diabetic retinopathy: Pathophysiological roles and therapeutic implications. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H738–H749. [Google Scholar] [CrossRef]
- Vecino, E.; Rodriguez, F.D.; Ruzafa, N.; Pereiro, X.; Sharma, S.C. Glia-neuron interactions in the mammalian retina. Prog. Retin. Eye Res. 2016, 51, 1–40. [Google Scholar] [CrossRef]
- Wu, S.M.; Maple, B.R. Amino acid neurotransmitters in the retina: A functional overview. Vis. Res. 1998, 38, 1371–1384. [Google Scholar] [CrossRef]
- Selvam, S.; Kumar, T.; Fruttiger, M. Retinal vasculature development in health and disease. Prog. Retin. Eye Res. 2018, 63, 1–19. [Google Scholar] [CrossRef]
- Ames, A., 3rd. Energy requirements of cns cells as related to their function and to their vulnerability to ischemia: A commentary based on studies on retina. Can. J. Physiol. Pharmacol. 1992, 70, S158–S164. [Google Scholar] [CrossRef]
- Maenhaut, N.; Boussery, K.; Delaey, C.; Van de Voorde, J. Control of retinal arterial tone by a paracrine retinal relaxing factor. Microcirculation 2007, 14, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Vanden Daele, L.; Boydens, C.; Pauwels, B.; Van de Voorde, J. The retinal relaxing factor: Update on an enigmatic regulator of the retinal circulation. Investig. Ophthalmol. Vis. Sci. 2017, 58, 1702–1708. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Funk, R.H. Blood supply of the retina. Ophthalmic. Res. 1997, 29, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Laties, A.M. Central retinal artery innervation. Absence of adrenergic innervation to the intraocular branches. Arch. Ophthalmol. 1967, 77, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Yu, D. Isolated preparations of ocular vasculature and their applications in ophthalmic research. Prog. Retin. Eye Res. 2003, 22, 135–169. [Google Scholar] [CrossRef]
- Donati, G.; Pournaras, C.J.; Munoz, J.L.; Poitry, S.; Poitry-Yamate, C.L.; Tsacopoulos, M. Nitric oxide controls arteriolar tone in the retina of the miniature pig. Investig. Ophthalmol. Vis. Sci. 1995, 36, 2228–2237. [Google Scholar]
- Konukoglu, D.; Uzun, H. Endothelial dysfunction and hypertension. Adv. Exp. Med. Biol. 2017, 956, 511–540. [Google Scholar]
- Reiner, A.; Fitzgerald, M.E.C.; Del Mar, N.; Li, C. Neural control of choroidal blood flow. Prog. Retin. Eye Res. 2018, 64, 96–130. [Google Scholar] [CrossRef]
- Yu, D.Y.; Alder, V.A.; Cringle, S.J.; Brown, M.J. Choroidal blood flow measured in the dog eye in vivo and in vitro by local hydrogen clearance polarography: Validation of a technique and response to raised intraocular pressure. Exp. Eye Res. 1988, 46, 289–303. [Google Scholar] [CrossRef]
- Alm, A.; Bill, A. The oxygen supply to the retina. Ii. Effects of high intraocular pressure and of increased arterial carbon dioxide tension on uveal and retinal blood flow in cats. A study with radioactively labelled microspheres including flow determinations in brain and some other tissues. Acta Physiol. Scand. 1972, 84, 306–319. [Google Scholar]
- Bill, A.; Sperber, G.O. Control of retinal and choroidal blood flow. Eye 1990, 4, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Schmidl, D.; Garhofer, G.; Schmetterer, L. The complex interaction between ocular perfusion pressure and ocular blood flow—Relevance for glaucoma. Exp. Eye Res. 2011, 93, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Metea, M.R.; Newman, E.A. Signalling within the neurovascular unit in the mammalian retina. Exp. Physiol. 2007, 92, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Hamel, E. Perivascular nerves and the regulation of cerebrovascular tone. J. Appl. Physiol. 2006, 100, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Meza, C.A.; La Favor, J.D.; Kim, D.H.; Hickner, R.C. Endothelial dysfunction: Is there a hyperglycemia-induced imbalance of nox and nos? Int. J. Mol. Sci. 2019, 20, 3775. [Google Scholar] [CrossRef]
- Pennathur, S.; Heinecke, J.W. Oxidative stress and endothelial dysfunction in vascular disease. Curr. Diabetes Rep. 2007, 7, 257–264. [Google Scholar] [CrossRef]
- Förstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef]
- Crabtree, M.J.; Channon, K.M. Synthesis and recycling of tetrahydrobiopterin in endothelial function and vascular disease. Nitric Oxide 2011, 25, 81–88. [Google Scholar] [CrossRef]
- Antonelli-Orlidge, A.; Smith, S.R.; D’Amore, P.A. Influence of pericytes on capillary endothelial cell growth. Am. Rev. Respir. Dis. 1989, 140, 1129–1131. [Google Scholar] [CrossRef]
- Hammes, H.P.; Lin, J.; Renner, O.; Shani, M.; Lundqvist, A.; Betsholtz, C.; Brownlee, M.; Deutsch, U. Pericytes and the pathogenesis of diabetic retinopathy. Diabetes 2002, 51, 3107–3112. [Google Scholar] [CrossRef]
- El-Danaf, R.N.; Huberman, A.D. Characteristic patterns of dendritic remodeling in early-stage glaucoma: Evidence from genetically identified retinal ganglion cell types. J. Neurosci. 2015, 35, 2329–2343. [Google Scholar] [CrossRef] [PubMed]
- Sapieha, P.; Sirinyan, M.; Hamel, D.; Zaniolo, K.; Joyal, J.S.; Cho, J.H.; Honore, J.C.; Kermorvant-Duchemin, E.; Varma, D.R.; Tremblay, S.; et al. The succinate receptor gpr91 in neurons has a major role in retinal angiogenesis. Nat. Med. 2008, 14, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Miao, F.J.P.; Lin, D.C.H.; Schwandner, R.T.; Wang, Z.; Gao, J.; Chen, J.-L.; Tian, H.; Ling, L. Citric acid cycle intermediates as ligands for orphan g-protein-coupled receptors. Nature 2004, 429, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, C.-H.; Wang, Z.; Meng, S.S.; Burnim, S.B.; SanGiovanni, J.P.; Kamenecka, T.M.; Solt, L.A.; Chen, J. Rorα modulates semaphorin 3e transcription and neurovascular interaction in pathological retinal angiogenesis. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2017, 31, 4492–4502. [Google Scholar] [CrossRef]
- Metea, M.R.; Newman, E.A. Glial cells dilate and constrict blood vessels: A mechanism of neurovascular coupling. J. Neurosci. 2006, 26, 2862–2870. [Google Scholar] [CrossRef]
- Cuenca, N.; Fernandez-Sanchez, L.; Campello, L.; Maneu, V.; De la Villa, P.; Lax, P.; Pinilla, I. Cellular responses following retinal injuries and therapeutic approaches for neurodegenerative diseases. Prog. Retin. Eye Res. 2014, 43, 17–75. [Google Scholar] [CrossRef]
- Bringmann, A.; Grosche, A.; Pannicke, T.; Reichenbach, A. Gaba and glutamate uptake and metabolism in retinal glial (muller) cells. Front. Endocrinol. (Lausanne) 2013, 4, 48. [Google Scholar] [CrossRef]
- Fu, D.; Yu, J.Y.; Connell, A.R.; Yang, S.; Hookham, M.B.; McLeese, R.; Lyons, T.J. Beneficial effects of berberine on oxidized ldl-induced cytotoxicity to human retinal muller cells. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3369–3379. [Google Scholar] [CrossRef]
- Falero-Perez, J.; Sorenson, C.M.; Sheibani, N. Cyp1b1-deficient retinal astrocytes are more proliferative and migratory and are protected from oxidative stress and inflammation. Am. J. Physiol. Cell Physiol. 2019, 316, C767–C781. [Google Scholar] [CrossRef]
- Ding, X.; Zhang, M.; Gu, R.; Xu, G.; Wu, H. Activated microglia induce the production of reactive oxygen species and promote apoptosis of co-cultured retinal microvascular pericytes. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 777–788. [Google Scholar] [CrossRef]
- Zadeh, J.K.; Ruemmler, R.; Hartmann, E.K.; Ziebart, A.; Ludwig, M.; Patzak, A.; Xia, N.; Li, H.; Pfeiffer, N.; Gericke, A. Responses of retinal arterioles and ciliary arteries in pigs with acute respiratory distress syndrome (ards). Exp. Eye Res. 2019, 184, 152–161. [Google Scholar] [CrossRef]
- Endemann, D.H.; Schiffrin, E.L. Endothelial dysfunction. J. Am. Soc. Nephrol. 2004, 15, 1983–1992. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, N.; Arora, P.; Sandhir, R. Perturbed biochemical pathways and associated oxidative stress lead to vascular dysfunctions in diabetic retinopathy. Oxid. Med. Cell. Longev. 2019, 2019, 8458472. [Google Scholar] [CrossRef] [PubMed]
- Pawlowska, E.; Szczepanska, J.; Koskela, A.; Kaarniranta, K.; Blasiak, J. Dietary polyphenols in age-related macular degeneration: Protection against oxidative stress and beyond. Oxid. Med. Cell. Longev. 2019, 2019, 9682318. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, S.; Biswas, S.; Nagrani, N.; Chu, Y.; Chakrabarti, S.; Feng, B. Glucose-induced oxidative stress and accelerated aging in endothelial cells are mediated by the depletion of mitochondrial sirts. Physiol. Rep. 2020, 8, e14331. [Google Scholar] [CrossRef]
- Dinh, Q.N.; Drummond, G.R.; Sobey, C.G.; Chrissobolis, S. Roles of inflammation, oxidative stress, and vascular dysfunction in hypertension. BioMed Res. Int. 2014, 2014, 406960. [Google Scholar] [CrossRef]
- Salminen, A.; Ojala, J.; Kaarniranta, K.; Kauppinen, A. Mitochondrial dysfunction and oxidative stress activate inflammasomes: Impact on the aging process and age-related diseases. Cell. Mol. Life Sci. 2012, 69, 2999–3013. [Google Scholar] [CrossRef]
- Schroder, K.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Lu, Q.; Chen, W.; Li, J.; Li, C.; Zheng, Z. Vitamin d3 protects against diabetic retinopathy by inhibiting high-glucose-induced activation of the ros/txnip/nlrp3 inflammasome pathway. J. Diabetes Res. 2018, 2018, 8193523. [Google Scholar] [CrossRef] [PubMed]
- Devi, T.S.; Lee, I.; Hüttemann, M.; Kumar, A.; Nantwi, K.D.; Singh, L.P. Txnip links innate host defense mechanisms to oxidative stress and inflammation in retinal muller glia under chronic hyperglycemia: Implications for diabetic retinopathy. Exp. Diabetes Res. 2012, 2012, 438238. [Google Scholar] [CrossRef] [PubMed]
- Drummond, G.R.; Sobey, C.G. Endothelial nadph oxidases: Which nox to target in vascular disease? Trends Endocrinol. Metab. 2014, 25, 452–463. [Google Scholar] [CrossRef]
- Panday, A.; Sahoo, M.K.; Osorio, D.; Batra, S. Nadph oxidases: An overview from structure to innate immunity-associated pathologies. Cell. Mol. Immunol. 2015, 12, 5–23. [Google Scholar] [CrossRef]
- Bedard, K.; Krause, K.H. The nox family of ros-generating nadph oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef]
- Lin, C.C.; Lee, I.T.; Yang, Y.L.; Lee, C.W.; Kou, Y.R.; Yang, C.M. Induction of cox-2/pge(2)/il-6 is crucial for cigarette smoke extract-induced airway inflammation: Role of tlr4-dependent nadph oxidase activation. Free Radic. Biol. Med. 2010, 48, 240–254. [Google Scholar] [CrossRef]
- Peng, J.J.; Xiong, S.Q.; Ding, L.X.; Peng, J.; Xia, X.B. Diabetic retinopathy: Focus on nadph oxidase and its potential as therapeutic target. Eur. J. Pharmacol. 2019, 853, 381–387. [Google Scholar] [CrossRef]
- Deliyanti, D.; Alrashdi, S.F.; Touyz, R.M.; Kennedy, C.R.; Jha, J.C.; Cooper, M.E.; Jandeleit-Dahm, K.A.; Wilkinson-Berka, J.L. Nox (nadph oxidase) 1, nox4, and nox5 promote vascular permeability and neovascularization in retinopathy. Hypertension 2020, 75, 1091–1101. [Google Scholar] [CrossRef]
- Appukuttan, B.; Ma, Y.; Stempel, A.; Ashander, L.M.; Deliyanti, D.; Wilkinson-Berka, J.L.; Smith, J.R. Effect of nadph oxidase 1 and 4 blockade in activated human retinal endothelial cells. Clin. Exp. Ophthalmol. 2018, 46, 652–660. [Google Scholar] [CrossRef]
- Al-Shabrawey, M.; Rojas, M.; Sanders, T.; Behzadian, A.; El-Remessy, A.; Bartoli, M.; Parpia, A.K.; Liou, G.; Caldwell, R.B. Role of nadph oxidase in retinal vascular inflammation. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3239–3244. [Google Scholar] [CrossRef]
- Brandes, R.P.; Weissmann, N.; Schroder, K. Nox family nadph oxidases: Molecular mechanisms of activation. Free Radic. Biol. Med. 2014, 76, 208–226. [Google Scholar] [CrossRef]
- Wu, M.Y.; Yiang, G.T.; Lai, T.T.; Li, C.J. The oxidative stress and mitochondrial dysfunction during the pathogenesis of diabetic retinopathy. Oxid. Med. Cell. Longev. 2018, 2018, 3420187. [Google Scholar] [CrossRef]
- Touyz, R.M.; Anagnostopoulou, A.; Rios, F.; Montezano, A.C.; Camargo, L.L. Nox5: Molecular biology and pathophysiology. Exp. Physiol. 2019, 104, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Gong, J.; Xu, Z.; Duh, E.J. Nrf2 promotes reparative angiogenesis through regulation of nadph oxidase-2 in oxygen-induced retinopathy. Free Radic. Biol. Med. 2016, 99, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Al-Shabrawey, M.; Bartoli, M.; El-Remessy, A.B.; Ma, G.; Matragoon, S.; Lemtalsi, T.; Caldwell, R.W.; Caldwell, R.B. Role of nadph oxidase and stat3 in statin-mediated protection against diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3231–3238. [Google Scholar] [CrossRef] [PubMed]
- Yokota, H.; Narayanan, S.P.; Zhang, W.; Liu, H.; Rojas, M.; Xu, Z.; Lemtalsi, T.; Nagaoka, T.; Yoshida, A.; Brooks, S.E.; et al. Neuroprotection from retinal ischemia/reperfusion injury by nox2 nadph oxidase deletion. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8123–8131. [Google Scholar] [CrossRef]
- Sahajpal, N.; Kowluru, A.; Kowluru, R.A. The regulatory role of rac1, a small molecular weight gtpase, in the development of diabetic retinopathy. J. Clin. Med. 2019, 8, 965. [Google Scholar] [CrossRef]
- Liu, X.; Du, H.; Chen, D.; Yuan, H.; Chen, W.; Jia, W.; Wang, X.; Li, X.; Gao, L. Cyclophilin d deficiency protects against the development of mitochondrial ros and cellular inflammation in aorta. Biochem. Biophys. Res. Commun. 2019, 508, 1202–1208. [Google Scholar] [CrossRef]
- Zheng, Z.; Chen, H.; Wang, H.; Ke, B.; Zheng, B.; Li, Q.; Li, P.; Su, L.; Gu, Q.; Xu, X. Improvement of retinal vascular injury in diabetic rats by statins is associated with the inhibition of mitochondrial reactive oxygen species pathway mediated by peroxisome proliferator-activated receptor gamma coactivator 1alpha. Diabetes 2010, 59, 2315–2325. [Google Scholar] [CrossRef]
- Engineer, A.; Saiyin, T.; Greco, E.R.; Feng, Q. Say no to ros: Their roles in embryonic heart development and pathogenesis of congenital heart defects in maternal diabetes. Antioxidants 2019, 8, 463. [Google Scholar] [CrossRef]
- Paige, J.S.; Jaffrey, S.R. Pharmacologic manipulation of nitric oxide signaling: Targeting nos dimerization and protein-protein interactions. Curr. Top. Med. Chem. 2007, 7, 97–114. [Google Scholar] [CrossRef]
- Beckman, J.S. Oxidative damage and tyrosine nitration from peroxynitrite. Chem. Res. Toxicol. 1996, 9, 836–844. [Google Scholar] [CrossRef]
- El-Remessy, A.B.; Abou-Mohamed, G.; Caldwell, R.W.; Caldwell, R.B. High glucose-induced tyrosine nitration in endothelial cells: Role of enos uncoupling and aldose reductase activation. Investig. Ophthalmol. Vis. Sci. 2003, 44, 3135–3143. [Google Scholar] [CrossRef] [PubMed]
- Calderon, G.D.; Juarez, O.H.; Hernandez, G.E.; Punzo, S.M.; De la Cruz, Z.D. Oxidative stress and diabetic retinopathy: Development and treatment. Eye 2017, 31, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Bourne, R.R.A.; Stevens, G.A.; White, R.A.; Smith, J.L.; Flaxman, S.R.; Price, H.; Jonas, J.B.; Keeffe, J.; Leasher, J.; Naidoo, K.; et al. Causes of vision loss worldwide, 1990–2010: A systematic analysis. Lancet Glob. Health 2013, 1, e339–e349. [Google Scholar] [CrossRef]
- Cade, W.T. Diabetes-related microvascular and macrovascular diseases in the physical therapy setting. Phys. Ther. 2008, 88, 1322–1335. [Google Scholar] [CrossRef] [PubMed]
- Heng, L.Z.; Comyn, O.; Peto, T.; Tadros, C.; Ng, E.; Sivaprasad, S.; Hykin, P.G. Diabetic retinopathy: Pathogenesis, clinical grading, management and future developments. Diabet. Med. 2013, 30, 640–650. [Google Scholar] [CrossRef]
- Gardner, T.W.; Davila, J.R. The neurovascular unit and the pathophysiologic basis of diabetic retinopathy. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 1–6. [Google Scholar] [CrossRef]
- Dehdashtian, E.; Mehrzadi, S.; Yousefi, B.; Hosseinzadeh, A.; Reiter, R.J.; Safa, M.; Ghaznavi, H.; Naseripour, M. Diabetic retinopathy pathogenesis and the ameliorating effects of melatonin; involvement of autophagy, inflammation and oxidative stress. Life Sci. 2018, 193, 20–33. [Google Scholar] [CrossRef]
- Pang, B.; Li, M.; Song, J.; Li, Q.W.; Wang, J.; Di, S.; Tong, X.L.; Ni, Q. Luo tong formula attenuates retinal inflammation in diabetic rats via inhibition of the p38mapk/nf-kappab pathway. Chin. Med. 2020, 15, 5. [Google Scholar] [CrossRef]
- Lazzara, F.; Fidilio, A.; Platania, C.B.M.; Giurdanella, G.; Salomone, S.; Leggio, G.M.; Tarallo, V.; Cicatiello, V.; De Falco, S.; Eandi, C.M.; et al. Aflibercept regulates retinal inflammation elicited by high glucose via the plgf/erk pathway. Biochem. Pharmacol. 2019, 168, 341–351. [Google Scholar] [CrossRef]
- Tarr, J.M.; Kaul, K.; Chopra, M.; Kohner, E.M.; Chibber, R. Pathophysiology of diabetic retinopathy. ISRN Ophthalmol. 2013, 2013, 343560. [Google Scholar] [CrossRef]
- Huang, C.; Zhu, H.J.; Li, H.; Li, Q.X.; Li, F.M.; Cheng, L.; Liu, Y.G. P38-mapk pathway is activated in retinopathy of microvascular disease of stz-induced diabetic rat model. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5789–5796. [Google Scholar] [PubMed]
- Atay, O.; Skotheim, J.M. Spatial and temporal signal processing and decision making by mapk pathways. J. Cell. Biol. 2017, 216, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.L.; Goldfine, I.D.; Maddux, B.A.; Grodsky, G.M. Oxidative stress and stress-activated signaling pathways: A unifying hypothesis of type 2 diabetes. Endocr. Rev. 2002, 23, 599–622. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Tonks, N.K.; Bar-Sagi, D. Inhibition of ras-induced DNA synthesis by expression of the phosphatase mkp-1. Science 1994, 266, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Asai, S.; Ohta, K.; Yoshioka, H. Mapk signaling regulates nitric oxide and nadph oxidase-dependent oxidative bursts in nicotiana benthamiana. Plant Cell 2008, 20, 1390–1406. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef]
- Ma, Y.; Zhao, Q.; Shao, Y.; Cao, M.Z.; Zhao, M.; Wang, D. Melatonin inhibits the inflammation and apoptosis in rats with diabetic retinopathy via mapk pathway. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1–8. [Google Scholar]
- Nishikawa, T.; Edelstein, D.; Brownlee, M. The missing link: A single unifying mechanism for diabetic complications. Kidney Int. Suppl. 2000, 77, S26–S30. [Google Scholar] [CrossRef]
- Hohman, T.C.; Nishimura, C.; Robison, W.G., Jr. Aldose reductase and polyol in cultured pericytes of human retinal capillaries. Exp. Eye Res. 1989, 48, 55–60. [Google Scholar] [CrossRef]
- Kinoshita, J.H. A thirty year journey in the polyol pathway. Exp. Eye Res. 1990, 50, 567–573. [Google Scholar] [CrossRef]
- Xia, P.; Kramer, R.M.; King, G.L. Identification of the mechanism for the inhibition of na+,k(+)-adenosine triphosphatase by hyperglycemia involving activation of protein kinase c and cytosolic phospholipase a2. J. Clin. Investig. 1995, 96, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Lindpaintner, K.; Jin, M.; Wilhelm, M.; Toth, M.; Ganten, D. Aspects of molecular biology and biochemistry of the cardiac renin-angiotensin system. Br. J. Clin. Pharmacol. 1989, 27, 159S–165S. [Google Scholar] [CrossRef] [PubMed]
- Danser, A.H.; Derkx, F.H.; Admiraal, P.J.; Deinum, J.; de Jong, P.T.; Schalekamp, M.A. Angiotensin levels in the eye. Investig. Ophthalmol. Vis. Sci. 1994, 35, 1008–1018. [Google Scholar]
- Steckelings, U.M.; Rompe, F.; Kaschina, E.; Unger, T. The evolving story of the raas in hypertension, diabetes and cv disease: Moving from macrovascular to microvascular targets. Fundam. Clin. Pharmacol. 2009, 23, 693–703. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Cheruvu, S.C.; Sarris, M.; Liyanage, S.S.; Lumbers, E.; Chui, J.; Wakefield, D.; McCluskey, P.J. Expression of classical components of the renin-angiotensin system in the human eye. J. Renin Angiotensin Aldosterone Syst. 2015, 16, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, R.; Kapoor, M.S.; Singh, A.; Bodakhe, S.H. Therapeutic targets of renin-angiotensin system in ocular disorders. J. Curr. Ophthalmol. 2016, 29, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Jackson, W.E.; Holmes, D.L.; Garg, S.K.; Harris, S.; Chase, H.P. Angiotensin-converting enzyme inhibitor therapy and diabetic retinopathy. Ann. Ophthalmol. 1992, 24, 99–103. [Google Scholar]
- Ozawa, Y.; Kurihara, T.; Sasaki, M.; Ban, N.; Yuki, K.; Kubota, S.; Tsubota, K. Neural degeneration in the retina of the streptozotocin-induced type 1 diabetes model. Exp. Diabetes Res. 2011, 2011, 108328. [Google Scholar] [CrossRef]
- Verma, A.; Shan, Z.; Lei, B.; Yuan, L.; Liu, X.; Nakagawa, T.; Grant, M.B.; Lewin, A.S.; Hauswirth, W.W.; Raizada, M.K.; et al. Ace2 and ang-(1–7) confer protection against development of diabetic retinopathy. Mol. Ther. 2012, 20, 28–36. [Google Scholar] [CrossRef]
- Nagai, N.; Izumi-Nagai, K.; Oike, Y.; Koto, T.; Satofuka, S.; Ozawa, Y.; Yamashiro, K.; Inoue, M.; Tsubota, K.; Umezawa, K.; et al. Suppression of diabetes-induced retinal inflammation by blocking the angiotensin ii type 1 receptor or its downstream nuclear factor-kappab pathway. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4342–4350. [Google Scholar] [CrossRef]
- Newton, A.C. Regulation of the abc kinases by phosphorylation: Protein kinase c as a paradigm. Biochem. J 2003, 370, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Shiba, T.; Inoguchi, T.; Sportsman, J.R.; Heath, W.F.; Bursell, S.; King, G.L. Correlation of diacylglycerol level and protein kinase c activity in rat retina to retinal circulation. Am. J. Physiol. 1993, 265, E783–E793. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.; Aiello, L.P.; Ishii, H.; Jiang, Z.Y.; Park, D.J.; Robinson, G.S.; Takagi, H.; Newsome, W.P.; Jirousek, M.R.; King, G.L. Characterization of vascular endothelial growth factor’s effect on the activation of protein kinase c, its isoforms, and endothelial cell growth. J. Clin. Investig. 1996, 98, 2018–2026. [Google Scholar] [CrossRef] [PubMed]
- Stitt, A.W. Ages and diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4867–4874. [Google Scholar] [CrossRef]
- Chen, B.H.; Jiang, D.Y.; Tang, L.S. Advanced glycation end-products induce apoptosis involving the signaling pathways of oxidative stress in bovine retinal pericytes. Life Sci. 2006, 79, 1040–1048. [Google Scholar] [CrossRef]
- Sato, T.; Iwaki, M.; Shimogaito, N.; Wu, X.; Yamagishi, S.; Takeuchi, M. Tage (toxic ages) theory in diabetic complications. Curr. Mol. Med. 2006, 6, 351–358. [Google Scholar] [CrossRef]
- Inagaki, Y.; Yamagishi, S.; Okamoto, T.; Takeuchi, M.; Amano, S. Pigment epithelium-derived factor prevents advanced glycation end products-induced monocyte chemoattractant protein-1 production in microvascular endothelial cells by suppressing intracellular reactive oxygen species generation. Diabetologia 2003, 46, 284–287. [Google Scholar] [CrossRef]
- Chen, Q.; Tang, L.; Xin, G.; Li, S.; Ma, L.; Xu, Y.; Zhuang, M.; Xiong, Q.; Wei, Z.; Xing, Z.; et al. Oxidative stress mediated by lipid metabolism contributes to high glucose-induced senescence in retinal pigment epithelium. Free Radic. Biol. Med. 2019, 130, 48–58. [Google Scholar] [CrossRef]
- Capozzi, M.E.; Savage, S.R.; McCollum, G.W.; Hammer, S.S.; Ramos, C.J.; Yang, R.; Bretz, C.A.; Penn, J.S. The peroxisome proliferator-activated receptor-β/δ antagonist gsk0660 mitigates retinal cell inflammation and leukostasis. Exp. Eye Res. 2020, 190, 107885. [Google Scholar] [CrossRef]
- Chang, Y.C.; Wu, W.C. Dyslipidemia and diabetic retinopathy. Rev. Diabet. Stud. 2013, 10, 121–132. [Google Scholar] [CrossRef]
- Albouery, M.; Buteau, B.; Grégoire, S.; Martine, L.; Gambert, S.; Bron, A.M.; Acar, N.; Chassaing, B.; Bringer, M.A. Impact of a high-fat diet on the fatty acid composition of the retina. Exp. Eye Res. 2020, 196, 108059. [Google Scholar] [CrossRef] [PubMed]
- Biousse, V.; Newman, N. Retinal and optic nerve ischemia. Continuum 2014, 20, 838–856. [Google Scholar] [CrossRef]
- Cugati, S.; Varma, D.D.; Chen, C.S.; Lee, A.W. Treatment options for central retinal artery occlusion. Curr. Treat. Options Neurol. 2013, 15, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Tobalem, S.; Schutz, J.S.; Chronopoulos, A. Central retinal artery occlusion - rethinking retinal survival time. BMC Ophthalmol. 2018, 18, 101. [Google Scholar] [CrossRef] [PubMed]
- Terelak-Borys, B.; Skonieczna, K.; Grabska-Liberek, I. Ocular ischemic syndrome - a systematic review. Med. Sci. Monit. Int. Med J. Exp. Clin. Res. 2012, 18, RA138–RA144. [Google Scholar] [CrossRef] [PubMed]
- Rumelt, S.; Dorenboim, Y.; Rehany, U. Aggressive systematic treatment for central retinal artery occlusion. Am. J. Ophthalmol. 1999, 128, 733–738. [Google Scholar] [CrossRef]
- Feltgen, N.; Neubauer, A.; Jurklies, B.; Schmoor, C.; Schmidt, D.; Wanke, J.; Maier-Lenz, H.; Schumacher, M. Multicenter study of the european assessment group for lysis in the eye (eagle) for the treatment of central retinal artery occlusion: Design issues and implications. Eagle study report no. 1: Eagle study report no. 1. Graefes Arch. Clin. Exp. Ophthalmol. 2006, 244, 950–956. [Google Scholar] [CrossRef]
- Schumacher, M.; Schmidt, D.; Jurklies, B.; Gall, C.; Wanke, I.; Schmoor, C.; Maier-Lenz, H.; Solymosi, L.; Brueckmann, H.; Neubauer, A.S.; et al. Central retinal artery occlusion: Local intra-arterial fibrinolysis versus conservative treatment, a multicenter randomized trial. Ophthalmology 2010, 117, 1367–1375.e1. [Google Scholar] [CrossRef]
- Zadeh, J.K.; Garcia-Bardon, A.; Hartmann, E.K.; Pfeiffer, N.; Omran, W.; Ludwig, M.; Patzak, A.; Xia, N.; Li, H.; Gericke, A. Short-time ocular ischemia induces vascular endothelial dysfunction and ganglion cell loss in the pig retina. Int. J. Mol. Sci. 2019, 20, 4685. [Google Scholar] [CrossRef]
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef]
- Gotoh, T.; Sonoki, T.; Nagasaki, A.; Terada, K.; Takiguchi, M.; Mori, M. Molecular cloning of cdna for nonhepatic mitochondrial arginase (arginase ii) and comparison of its induction with nitric oxide synthase in a murine macrophage-like cell line. FEBS Lett. 1996, 395, 119–122. [Google Scholar] [CrossRef]
- Sin, Y.Y.; Baron, G.; Schulze, A.; Funk, C.D. Arginase-1 deficiency. J. Mol. Med. (Berl.) 2015, 93, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Hein, T.W.; Wang, W.; Chang, C.I.; Kuo, L. Constitutive expression of arginase in microvascular endothelial cells counteracts nitric oxide-mediated vasodilatory function. FASEB J. 2001, 15, 1264–1266. [Google Scholar] [CrossRef] [PubMed]
- Suwanpradid, J.; Shih, M.; Pontius, L.; Yang, B.; Birukova, A.; Guttman-Yassky, E.; Corcoran, D.L.; Que, L.G.; Tighe, R.M.; MacLeod, A.S. Arginase1 deficiency in monocytes/macrophages upregulates inducible nitric oxide synthase to promote cutaneous contact hypersensitivity. J. Immunol. 2017, 199, 1827–1834. [Google Scholar] [CrossRef]
- Mahdi, A.; Tengbom, J.; Alvarsson, M.; Wernly, B.; Zhou, Z.; Pernow, J. Red blood cell peroxynitrite causes endothelial dysfunction in type 2 diabetes mellitus via arginase. Cells 2020, 9, 1712. [Google Scholar] [CrossRef]
- Romero, M.J.; Platt, D.H.; Tawfik, H.E.; Labazi, M.; El-Remessy, A.B.; Bartoli, M.; Caldwell, R.B.; Caldwell, R.W. Diabetes-induced coronary vascular dysfunction involves increased arginase activity. Circ. Res. 2008, 102, 95–102. [Google Scholar] [CrossRef]
- Zhu, M.; Goetsch, S.C.; Wang, Z.; Luo, R.; Hill, J.A.; Schneider, J.; Morris, S.M., Jr.; Liu, Z.P. Foxo4 promotes early inflammatory response upon myocardial infarction via endothelial arg1. Circ. Res. 2015, 117, 967–977. [Google Scholar] [CrossRef]
- Fouda, A.Y.; Eldahshan, W.; Narayanan, S.P.; Caldwell, R.W.; Caldwell, R.B. Arginase pathway in acute retina and brain injury: Therapeutic opportunities and unexplored avenues. Front. Pharmacol. 2020, 11, 277. [Google Scholar] [CrossRef]
- Patel, C.; Rojas, M.; Narayanan, S.P.; Zhang, W.; Xu, Z.; Lemtalsi, T.; Jittiporn, K.; Caldwell, R.W.; Caldwell, R.B. Arginase as a mediator of diabetic retinopathy. Front. Immunol. 2013, 4, 173. [Google Scholar] [CrossRef]
- Shosha, E.; Xu, Z.; Yokota, H.; Saul, A.; Rojas, M.; Caldwell, R.W.; Caldwell, R.B.; Narayanan, S.P. Arginase 2 promotes neurovascular degeneration during ischemia/reperfusion injury. Cell Death Dis. 2016, 7, e2483. [Google Scholar] [CrossRef]
- Ulbrich, F.; Lerach, T.; Biermann, J.; Kaufmann, K.B.; Lagreze, W.A.; Buerkle, H.; Loop, T.; Goebel, U. Argon mediates protection by interleukin-8 suppression via a tlr2/tlr4/stat3/nf-kappab pathway in a model of apoptosis in neuroblastoma cells in vitro and following ischemia-reperfusion injury in rat retina in vivo. J. Neurochem. 2016, 138, 859–873. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Song, Z.; Ying, S.; Yang, X.; Wu, W.; Tan, Q.; Ju, X.; Wu, W.; Zhang, X.; Qu, J.; et al. Myeloid differentiation protein 2 induced retinal ischemia reperfusion injury via upregulation of ros through a tlr4-nox4 pathway. Toxicol. Lett. 2018, 282, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Dada, T.; Mittal, D.; Mohanty, K.; Faiq, M.A.; Bhat, M.A.; Yadav, R.K.; Sihota, R.; Sidhu, T.; Velpandian, T.; Kalaivani, M.; et al. Mindfulness meditation reduces intraocular pressure, lowers stress biomarkers and modulates gene expression in glaucoma: A randomized controlled trial. J. Glaucoma 2018, 27, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarieh, M.; Flammer, J. New insights in the pathogenesis and treatment of normal tension glaucoma. Curr. Opin. Pharmacol. 2013, 13, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Tezel, G. Oxidative stress in glaucomatous neurodegeneration: Mechanisms and consequences. Prog. Retin. Eye Res. 2006, 25, 490–513. [Google Scholar] [CrossRef]
- Benoist d’Azy, C.; Pereira, B.; Chiambaretta, F.; Dutheil, F. Oxidative and anti-oxidative stress markers in chronic glaucoma: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0166915. [Google Scholar] [CrossRef]
- Gericke, A.; Mann, C.; Zadeh, J.K.; Musayeva, A.; Wolff, I.; Wang, M.; Pfeiffer, N.; Daiber, A.; Li, H.; Xia, N.; et al. Elevated intraocular pressure causes abnormal reactivity of mouse retinal arterioles. Oxid. Med. Cell. Longev. 2019, 2019, 9736047. [Google Scholar] [CrossRef]
- Salt, T.E.; Cordeiro, M.F. Glutamate excitotoxicity in glaucoma: Throwing the baby out with the bathwater? Eye 2006, 20, 730–731. [Google Scholar] [CrossRef]
- Tezel, G.; Yang, X.; Luo, C.; Peng, Y.; Sun, S.L.; Sun, D. Mechanisms of immune system activation in glaucoma: Oxidative stress-stimulated antigen presentation by the retina and optic nerve head glia. Investig. Ophthalmol. Vis. Sci. 2007, 48, 705–714. [Google Scholar] [CrossRef]
- Tezel, G.; Wax, M.B. Increased production of tumor necrosis factor-alpha by glial cells exposed to simulated ischemia or elevated hydrostatic pressure induces apoptosis in cocultured retinal ganglion cells. J. Neurosci. Off. J. Soc. Neurosci. 2000, 20, 8693–8700. [Google Scholar] [CrossRef]
- Hernandez, M.R.; Andrzejewska, W.M.; Neufeld, A.H. Changes in the extracellular matrix of the human optic nerve head in primary open-angle glaucoma. Am. J. Ophthalmol. 1990, 109, 180–188. [Google Scholar] [CrossRef]
- Garhofer, G.; Zawinka, C.; Resch, H.; Kothy, P.; Schmetterer, L.; Dorner, G.T. Reduced response of retinal vessel diameters to flicker stimulation in patients with diabetes. Br. J. Ophthalmol. 2004, 88, 887–891. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, M.; Bucala, R.; Suzuki, T.; Ohkubo, T.; Yamazaki, M.; Koike, T.; Kameda, Y.; Makita, Z. Neurotoxicity of advanced glycation end-products for cultured cortical neurons. J. Neuropathol. Exp. Neurol. 2000, 59, 1094–1105. [Google Scholar] [CrossRef]
- Tezel, G.; Luo, C. Accelerated aging in glaucoma: Immunohistochemical assessment of advanced glycation end products in the human retina and optic nerve head. Investig. Ophthalmol. Vis. Sci. 2007, 48, 1201–1211. [Google Scholar] [CrossRef]
- Thornalley, P.J. Cell activation by glycated proteins. Age receptors, receptor recognition factors and functional classification of ages. Cell. Mol. Biol. (Noisy-Le-Grand) 1998, 44, 1013–1023. [Google Scholar]
- Wang, S.; Bao, X. Hyperlipidemia, blood lipid level, and the risk of glaucoma: A meta-analysis. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1028–1043. [Google Scholar] [CrossRef]
- Ressiniotis, T.; Griffiths, P.G.; Birch, M.; Keers, S.; Chinnery, P.F. The role of apolipoprotein e gene polymorphisms in primary open-angle glaucoma. Arch. Ophthalmol. 2004, 122, 258–261. [Google Scholar] [CrossRef]
- Zetterberg, M.; Tasa, G.; Palmér, M.S.; Juronen, E.; Teesalu, P.; Blennow, K.; Zetterberg, H. Apolipoprotein e polymorphisms in patients with primary open-angle glaucoma. Am. J. Ophthalmol. 2007, 143, 1059–1060. [Google Scholar] [CrossRef]
- Al-Dabbagh, N.M.; Al-Dohayan, N.; Arfin, M.; Tariq, M. Apolipoprotein e polymorphisms and primary glaucoma in saudis. Mol. Vis. 2009, 15, 912–919. [Google Scholar]
- Mabuchi, F.; Tang, S.; Ando, D.; Yamakita, M.; Wang, J.; Kashiwagi, K.; Yamagata, Z.; Iijima, H.; Tsukahara, S. The apolipoprotein e gene polymorphism is associated with open angle glaucoma in the japanese population. Mol. Vis. 2005, 11, 609–612. [Google Scholar] [PubMed]
- Lam, C.Y.; Fan, B.J.; Wang, D.Y.; Tam, P.O.; Yung Tham, C.C.; Leung, D.Y.; Ping Fan, D.S.; Chiu Lam, D.S.; Pang, C.P. Association of apolipoprotein e polymorphisms with normal tension glaucoma in a chinese population. J. Glaucoma 2006, 15, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, Y.F.; Zhao, B.Y.; Gu, Z.Y.; Li, S.L. Apolipoprotein e gene ε4ε4 is associated with elevated risk of primary open angle glaucoma in asians: A meta-analysis. BMC Med. Genet. 2014, 15, 60. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R.W. Apolipoprotein e: Cholesterol transport protein with expanding role in cell biology. Science 1988, 240, 622–630. [Google Scholar] [CrossRef]
- Di Taranto, M.D.; Giacobbe, C.; Fortunato, G. Familial hypercholesterolemia: A complex genetic disease with variable phenotypes. Eur. J. Med. Genet. 2020, 63, 103831. [Google Scholar] [CrossRef]
- Nakashima, Y.; Plump, A.S.; Raines, E.W.; Breslow, J.L.; Ross, R. Apoe-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arterioscler. Thromb. 1994, 14, 133–140. [Google Scholar] [CrossRef]
- Omodaka, K.; Nishiguchi, K.M. Neuroprotective effect against axonal damage-induced retinal ganglion cell death in apolipoprotein e-deficient mice through the suppression of kainate receptor signaling. Brain Res. 2014, 1586, 203–212. [Google Scholar] [CrossRef]
- Yücel, I.; Akar, Y. Effect of hypercholesterolemia on inducible nitric oxide synthase expression in a rat model of elevated intraocular pressure. Vis. Res. 2005, 45, 1107–1114. [Google Scholar] [CrossRef]
- Zadeh, J.K.; Zhutdieva, M.B. Apolipoprotein e deficiency causes endothelial dysfunction in the mouse retina. Oxid. Med. Cell. Longev. 2019, 2019, 5181429. [Google Scholar] [CrossRef]
- Shibata, M.; Sugiyama, T. Changes in optic nerve head blood flow, visual function, and retinal histology in hypercholesterolemic rabbits. Exp. Eye Res. 2011, 93, 818–824. [Google Scholar] [CrossRef]
- Pennington, K.L.; DeAngelis, M.M. Epidemiology of age-related macular degeneration (amd): Associations with cardiovascular disease phenotypes and lipid factors. Eye Vis. (Lond.) 2016, 3, 34. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.L.; Su, X. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Klein, R.; Peto, T. The epidemiology of age-related macular degeneration. Am. J. Ophthalmol. 2004, 137, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Cano, M. The impact of oxidative stress and inflammation on rpe degeneration in non-neovascular amd. Prog. Retin. Eye Res. 2017, 60, 201–218. [Google Scholar] [CrossRef]
- Joachim, N.; Mitchell, P. Incidence and progression of reticular drusen in age-related macular degeneration: Findings from an older australian cohort. Ophthalmology 2014, 121, 917–925. [Google Scholar] [CrossRef]
- The age-related eye disease study system for classifying age-related macular degeneration from stereoscopic color fundus photographs: The age-related eye disease study report number 6. Am. J. Ophthalmol. 2001, 132, 668–681. [CrossRef]
- Kaarniranta, K.; Pawlowska, E. Role of mitochondrial DNA damage in ros-mediated pathogenesis of age-related macular degeneration (amd). Int. J. Mol. Sci. 2019, 20, 2374. [Google Scholar] [CrossRef]
- Strauss, O. The retinal pigment epithelium in visual function. Physiol. Rev. 2005, 85, 845–881. [Google Scholar] [CrossRef]
- Lee, J.; Giordano, S. Autophagy, mitochondria and oxidative stress: Cross-talk and redox signalling. Biochem. J. 2012, 441, 523–540. [Google Scholar] [CrossRef]
- Ershov, A.V.; Bazan, N.G. Photoreceptor phagocytosis selectively activates ppargamma expression in retinal pigment epithelial cells. J. Neurosci. Res. 2000, 60, 328–337. [Google Scholar] [CrossRef]
- Jager, R.D.; Mieler, W.F. Age-related macular degeneration. N. Engl. J. Med. 2008, 358, 2606–2617. [Google Scholar] [CrossRef] [PubMed]
- Winkler, B.S.; Boulton, M.E. Oxidative damage and age-related macular degeneration. Mol. Vis. 1999, 5, 32. [Google Scholar]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ham, W.T., Jr.; Ruffolo, J.J., Jr. Histologic analysis of photochemical lesions produced in rhesus retina by short-wave-length light. Investig. Ophthalmol. Vis. Sci. 1978, 17, 1029–1035. [Google Scholar]
- Blasiak, J.; Glowacki, S. Mitochondrial and nuclear DNA damage and repair in age-related macular degeneration. Int. J. Mol. Sci. 2013, 14, 2996–3010. [Google Scholar] [CrossRef] [PubMed]
- Provost, A.C.; Vede, L. Morphologic and electroretinographic phenotype of sr-bi knockout mice after a long-term atherogenic diet. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3931–3942. [Google Scholar] [CrossRef]
- Dithmar, S.; Curcio, C.A. Ultrastructural changes in bruch’s membrane of apolipoprotein e-deficient mice. Investig. Ophthalmol. Vis. Sci. 2000, 41, 2035–2042. [Google Scholar]
- Malek, G.; Johnson, L.V. Apolipoprotein e allele-dependent pathogenesis: A model for age-related retinal degeneration. Proc. Natl. Acad. Sci. USA 2005, 102, 11900–11905. [Google Scholar] [CrossRef]
- Rudolf, M.; Winkler, B. Increased expression of vascular endothelial growth factor associated with accumulation of lipids in bruch’s membrane of ldl receptor knockout mice. Br. J. Ophthalmol. 2005, 89, 1627–1630. [Google Scholar] [CrossRef][Green Version]
- Fernández-Robredo, P.; Recalde, S. Effect of zeaxanthin and antioxidant supplementation on vascular endothelial growth factor (vegf) expression in apolipoprotein-e deficient mice. Curr. Eye Res. 2009, 34, 543–552. [Google Scholar] [CrossRef]
- Fernandez-Robredo, P.; Moya, D. Vitamins c and e reduce retinal oxidative stress and nitric oxide metabolites and prevent ultrastructural alterations in porcine hypercholesterolemia. Investig. Ophthalmol. Vis. Sci. 2005, 46, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Mares-Perlman, J.A.; Brady, W.E. Dietary fat and age-related maculopathy. Arch. Ophthalmol. 1995, 113, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Hyman, L.; Schachat, A.P. Hypertension, cardiovascular disease, and age-related macular degeneration. Age-related macular degeneration risk factors study group. Arch. Ophthalmol. 2000, 118, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Maranville, J.C. Hdl-cholesterol levels and risk of age-related macular degeneration: A multiethnic genetic study using mendelian randomization. Int. J. Epidemiol 2017, 46, 1891–1902. [Google Scholar] [CrossRef] [PubMed]
- Colijn, J.M.; den Hollander, A.I. Increased high-density lipoprotein levels associated with age-related macular degeneration: Evidence from the eye-risk and european eye epidemiology consortia. Ophthalmology 2019, 126, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Stambolian, D. Genetic variants near timp3 and high-density lipoprotein-associated loci influence susceptibility to age-related macular degeneration. Proc. Natl. Acad. Sci. USA 2010, 107, 7401–7406. [Google Scholar] [CrossRef]
- Neale, B.M.; Fagerness, J. Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (lipc). Proc. Natl. Acad. Sci. USA 2010, 107, 7395–7400. [Google Scholar] [CrossRef]
- Fritsche, L.G.; Chen, W. Seven new loci associated with age-related macular degeneration. Nat. Genet. 2013, 45, 433–439. [Google Scholar]
- Fritsche, L.G.; Igl, W. A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nat. Genet 2016, 48, 134–143. [Google Scholar] [CrossRef]
- Rohowetz, L.J.; Kraus, J.G. Reactive oxygen species-mediated damage of retinal neurons: Drug development targets for therapies of chronic neurodegeneration of the retina. Int. J. Mol. Sci. 2018, 19, 3362. [Google Scholar] [CrossRef]
- Zhang, X.; Lai, D. Triamcinolone acetonide inhibits p38mapk activation and neuronal apoptosis in early diabetic retinopathy. Curr. Mol. Med. 2013, 13, 946–958. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, M.; Wickremasinghe, S. Diabetic retinopathy: A complex pathophysiology requiring novel therapeutic strategies. Expert Opin. Biol. Ther. 2018, 18, 1257–1270. [Google Scholar] [CrossRef] [PubMed]
- Sjølie, A.K.; Klein, R. Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (direct-protect 2): A randomised placebo-controlled trial. Lancet 2008, 372, 1385–1393. [Google Scholar] [CrossRef]
- Davis, M.D.; Sheetz, M.J. Effect of ruboxistaurin on the visual acuity decline associated with long-standing diabetic macular edema. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Gálvez, M.I. Protein kinase c inhibitors in the treatment of diabetic retinopathy. Review. Curr. Pharm Biotechnol. 2011, 12, 386–391. [Google Scholar] [CrossRef]
- Robinson, J.G.; Nedergaard, B.S. Effect of evolocumab or ezetimibe added to moderate- or high-intensity statin therapy on ldl-c lowering in patients with hypercholesterolemia: The laplace-2 randomized clinical trial. JAMA 2014, 311, 1870–1882. [Google Scholar] [CrossRef]
- Panahi, Y.; Ahmadi, Y. Curcumin as a potential candidate for treating hyperlipidemia: A review of cellular and metabolic mechanisms. J. Cell. Physiol. 2018, 233, 141–152. [Google Scholar] [CrossRef]
- Moran, E.; Ding, L. Protective and antioxidant effects of pparα in the ischemic retina. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4568–4576. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Z. Octreotide protects the mouse retina against ischemic reperfusion injury through regulation of antioxidation and activation of nf-κb. Oxid. Med. Cell. Longev. 2015, 2015, 970156. [Google Scholar] [CrossRef]
- Shimouchi, A.; Yokota, H. Neuroprotective effect of water-dispersible hesperetin in retinal ischemia reperfusion injury. Jpn. J. Ophthalmol. 2016, 60, 51–61. [Google Scholar] [CrossRef]
- Wang, C.; Ren, Y.L. Down-regulated lama4 inhibits oxidative stress-induced apoptosis of retinal ganglion cells through the mapk signaling pathway in rats with glaucoma. Cell Cycle 2019, 18, 932–948. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Stankowska, D.L. Targets of neuroprotection in glaucoma. J. Ocul. Pharmacol. Ther. 2018, 34, 85–106. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, K.A.; Harder, J.M. Jnk2 and jnk3 are major regulators of axonal injury-induced retinal ganglion cell death. Neurobiol. Dis. 2012, 46, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Prokai, L.; Prokai-Tatrai, K. Quinol-based cyclic antioxidant mechanism in estrogen neuroprotection. Proc. Natl. Acad. Sci. USA 2003, 100, 11741–11746. [Google Scholar] [CrossRef] [PubMed]
- Prokai-Tatrai, K.; Xin, H. 17β-estradiol eye drops protect the retinal ganglion cell layer and preserve visual function in an in vivo model of glaucoma. Mol. Pharm. 2013, 10, 3253–3261. [Google Scholar] [CrossRef] [PubMed]
- Kavirajan, H. Memantine: A comprehensive review of safety and efficacy. Expert Opin. Drug Saf. 2009, 8, 89–109. [Google Scholar] [CrossRef]
- Hare, W.A.; WoldeMussie, E. Efficacy and safety of memantine treatment for reduction of changes associated with experimental glaucoma in monkey, i: Functional measures. Investig. Ophthalmol. Vis. Sci. 2004, 45, 2625–2639. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Liebmann, J.M. Oral memantine for the treatment of glaucoma: Design and results of 2 randomized, placebo-controlled, phase 3 studies. Ophthalmology 2018, 125, 1874–1885. [Google Scholar] [CrossRef]
- Osborne, N.N. Recent clinical findings with memantine should not mean that the idea of neuroprotection in glaucoma is abandoned. Acta Ophthalmol. 2009, 87, 450–454. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins c and e, beta carotene, and zinc for age-related macular degeneration and vision loss: Areds report no. 8. Arch. Ophthalmol. 2001, 119, 1417–1436. [Google Scholar] [CrossRef]
- Albanes, D.; Heinonen, O.P. Alpha-tocopherol and beta-carotene supplements and lung cancer incidence in the alpha-tocopherol, beta-carotene cancer prevention study: Effects of base-line characteristics and study compliance. J. Natl. Cancer Inst. 1996, 88, 1560–1570. [Google Scholar] [CrossRef] [PubMed]
- The effect of vitamin e and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [CrossRef] [PubMed]
- Gorusupudi, A.; Nelson, K. The age-related eye disease 2 study: Micronutrients in the treatment of macular degeneration. Adv. Nutr. 2017, 8, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T. Preliminary identification of the human macular pigment. Vis. Res. 1985, 25, 1531–1535. [Google Scholar] [CrossRef]
- Seddon, J.M.; Ajani, U.A. Dietary carotenoids, vitamins a, c, and e, and advanced age-related macular degeneration. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef]
- Ma, L.; Dou, H.L. Lutein and zeaxanthin intake and the risk of age-related macular degeneration: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, 350–359. [Google Scholar] [CrossRef]
- Murthy, R.K.; Ravi, K. Lutein protects retinal pigment epithelium from cytotoxic oxidative stress. Cutan. Ocul. Toxicol. 2014, 33, 132–137. [Google Scholar] [CrossRef]
- Lo, H.M.; Tsai, Y.J. A naturally occurring carotenoid, lutein, reduces pdgf and H2O2 signaling and compromised migration in cultured vascular smooth muscle cells. J. Biomed. Sci. 2012, 19, 18. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: Areds2 report no. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar]
- Ojino, K.; Shimazawa, M. Protective effect of sun n8075, a free radical scavenger, against excessive light-induced retinal damage in mice. Biol. Pharm. Bull. 2014, 37, 424–430. [Google Scholar] [CrossRef]
- Masuda, T.; Shimazawa, M. Edaravone is a free radical scavenger that protects against laser-induced choroidal neovascularization in mice and common marmosets. Exp. Eye Res. 2016, 146, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Inokuchi, Y. Systemic administration of a free radical scavenger, edaravone, protects against light-induced photoreceptor degeneration in the mouse retina. Eur. J. Pharmacol. 2010, 642, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Laleu, B.; Gaggini, F. First in class, potent, and orally bioavailable nadph oxidase isoform 4 (nox4) inhibitors for the treatment of idiopathic pulmonary fibrosis. J. Med. Chem. 2010, 53, 7715–7730. [Google Scholar] [PubMed]
- Deliyanti, D.; Wilkinson-Berka, J. Inhibition of nox1/4 with gkt137831: A potential novel treatment to attenuate neuroglial cell inflammation in the retina. J. Neuroinflamm. 2015, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Pollock, J.D.; Williams, D.A. Mouse model of x-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat. Genet. 1995, 9, 202–209. [Google Scholar] [CrossRef]
- Altenhöfer, S.; Radermacher, K.A. Evolution of nadph oxidase inhibitors: Selectivity and mechanisms for target engagement. Antioxid. Redox Signal. 2015, 23, 406–427. [Google Scholar] [CrossRef]
- Abu-Amero, K.K.; Kondkar, A.A. Resveratrol and ophthalmic diseases. Nutrients 2016, 8, 200. [Google Scholar] [CrossRef]
- Toro, M.D.; Nowomiejska, K. Effect of resveratrol on in vitro and in vivo models of diabetic retinophathy: A systematic review. Int. J. Mol. Sci. 2019, 20, 3503. [Google Scholar] [CrossRef]
- Li, J.; Yu, S. Resveratrol prevents ros-induced apoptosis in high glucose-treated retinal capillary endothelial cells via the activation of ampk/sirt1/pgc-1α pathway. Oxid. Med. Cell. Longev. 2017, 2017, 7584691. [Google Scholar] [CrossRef]
- Luo, H.; Zhuang, J. Resveratrol delays retinal ganglion cell loss and attenuates gliosis-related inflammation from ischemia-reperfusion injury. Investig. Ophthalmol. Vis. Sci. 2018, 59, 3879–3888. [Google Scholar] [CrossRef]
- Luna, C.; Li, G. Resveratrol prevents the expression of glaucoma markers induced by chronic oxidative stress in trabecular meshwork cells. Food Chem. Toxicol. 2009, 47, 198–204. [Google Scholar] [CrossRef] [PubMed]
- López-Malo, D.; Villarón-Casares, C.A. Curcumin as a therapeutic option in retinal diseases. Antioxidants 2020, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Ran, Z.; Zhang, Y. Curcumin inhibits high glucose-induced inflammatory injury in human retinal pigment epithelial cells through the ros-pi3k/akt/mtor signaling pathway. Mol. Med. Rep. 2019, 19, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Li, W. Curcumolide reduces diabetic retinal vascular leukostasis and leakage partly via inhibition of the p38mapk/nf-kappa b signaling. Bioorg. Med. Chem. Lett. 2017, 27, 1835–1839. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: Systematic review and meta-analysis. JAMA 2007, 297, 842–857. [Google Scholar] [CrossRef] [PubMed]
- Bauersachs, J.; Widder, J.D. Reductive stress: Linking heat shock protein 27, glutathione, and cardiomyopathy? Hypertension 2010, 55, 1299–1300. [Google Scholar] [CrossRef]
- Kelsey, N.A.; Wilkins, H.M. Nutraceutical antioxidants as novel neuroprotective agents. Molecules 2010, 15, 7792. [Google Scholar] [CrossRef]
- Itoh, K.; Chiba, T. An nrf2/small maf heterodimer mediates the induction of phase ii detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Zhang, M.; Pan, H. Allicin decreases lipopolysaccharide-induced oxidative stress and inflammation in human umbilical vein endothelial cells through suppression of mitochondrial dysfunction and activation of nrf2. Cell. Physiol. Biochem. 2017, 41, 2255–2267. [Google Scholar] [CrossRef]
- Biogen Idec (2013) Tecfidera (Dimethyl Fumarate): Us Prescribing Information. Available online: http://www.tecfidera.com/pdfs/full-prescribing-information.pdf (accessed on 10 August 2020).
- Zyla, K.; Larabee, C.M. Dimethyl fumarate mitigates optic neuritis. Mol. Vis. 2019, 25, 446–461. [Google Scholar]
- Catanzaro, M.; Lanni, C. Eye-light on age-related macular degeneration: Targeting nrf2-pathway as a novel therapeutic strategy for retinal pigment epithelium. Front. Pharmacol. 2020, 11, 844. [Google Scholar] [CrossRef] [PubMed]
- Wan, P.; Su, W. Trimetazidine protects retinal ganglion cells from acute glaucoma via the nrf2/ho-1 pathway. Clin. Sci. (Lond.) 2017, 131, 2363–2375. [Google Scholar] [CrossRef] [PubMed]
- Long-Term Analysis of Dimethyl Fumarate, to Slow the Growth of Areas of Geographic Atrophy (Ladigaga). Available online: https://clinicaltrials.gov/ct2/show/NCT04292080 (accessed on 10 August 2020).

| Drug | Target | Research Type | Administration | Disease | Year | Effect | References |
|---|---|---|---|---|---|---|---|
| IVTA | p38MAPK | Animal experiment (Rat) | Intravitreal injection | DR | 2013 | Exerted neural protective effects on retinal neurons | [212] |
| Candesartan | RAAS | Clinical trial | Oral | DR | 2008 | Less severe retinopathy by the end of the trial was observed in the candesartan group | [214] |
| Ruboxistaurin | PKC β | Clinical trial | Oral | DR/Diabetic macular edema | 2009 | Ameliorated visual acuity decrease | [215] |
| GSK0660 | PPARβ/δ | Cell culture | Cell culture | DR | 2020 | PPARβ/δ inhibition mitigated inflammatory signaling events elicited by metabolic stimuli and inflammatory cytokines. | [130] |
| Evolocumab | PCSK9 LDL-C | Clinical trial | Subcutaneous injection | Hypercholes-terolemia | 2014 | Reduced LDL-C levels in patients receiving statin therapy | [217] |
| Fenofibric acid (Feno-FA) | PPARα | Animal experiment (Mouse) | Intraperitoneal injection | Retinal ischemia | 2014 | Feno-FA treatment and PPARα overexpression decreased ROS levels and protected cultured retinal cells from hypoxic cell death | [219] |
| Octreotide (OCT) | NF-κB | Animal experiment (Mouse) | Subcutaneous injection | Retinal ischemia | 2015 | Inhibition of ROS and downregulation of NF-κB and intercellular adhesion molecule-1 (ICAM-1) expression | [220] |
| Water-dispersible hesperetin (WD-Hpt) | ERK, IL-1β | Animal experiment (Mouse) | Intraperitoneal injection | Retinal ischemia | 2015 | Decreased I/R-induced ROS formation | [221] |
| 17β-estradiol | Estrogen receptor | Animal experiment (Rat) | Tropical eye drops | Glaucoma | 2014 | Demonstrated significant neuroprotective effect | [226] |
| Memantine | N-methyl-D-aspartate (NMDA) receptor | Animal experiment (Monkey) | Oral | Glaucoma | 2004 | Both safe and effective for reduction of functional changes associated with chronic ocular hypertension | [228] |
| Memantine | NMDA receptor | Clinical trial | Oral | Glaucoma | 2018 | Daily treatment with memantine 10 mg or 20 mg for 48 months did not significantly delay glaucomatous progression | [229] |
| AREDS formulation | Free radicals | Clinical trial | Oral | AMD | 2001 | Demonstrated a statistically significant odds reduction for the progression of advanced AMD | [231] |
| Lutein/ zeaxanthin | Free radicals | Clinical trial | Oral | AMD | 1994 | Most strongly associated with a reduced risk for AMD | [236] |
| Lutein/ zeaxanthin | PDGF | Cell culture | Cell culture | Vascular disease | 2012 | Lutein could inhibit PDGFR signaling, whereas zeaxanthin did not | [239] |
| AREDS2 formulation | Free radicals | Clinical trial | Oral | AMD | 2015 | Lutein and zeaxanthin could be more appropriate than beta-carotene in the AREDS type supplements | [240] |
| SUN N8075 | Free radicals | Animal experiment (Mouse) | Intraperitoneal administration | AMD | 2014 | Had protective effects on excess light-induced photoreceptor degeneration | [241] |
| Edaravone | Free radicals | Animal experiment (Mouse and common marmoset) | Intraperitoneal/ Intravenous injection | AMD | 2016 | Effective against laser-induced choroidal neovascularization (CNV) both in mice and in common marmosets | [242] |
| Edaravone | MAPKs | Animal experiment (Mouse) | Intraperitoneal/ Intravenous injection | AMD | 2010 | Significantly protected against light-induced photoreceptor degeneration at 5 days after exposure to light | [243] |
| GKT136901/ GKT137831 | NOX1/NOX4 inhibitors | Cell culture | Cell culture | Retinal vasculopathy | 2018 | Treatment with GKT136901 or GKT137831 significantly reduced ROS production and VEGFA expression by endothelial cells | [78] |
| GKT137831 | NOX1/NOX4 inhibitors | Animal experiment (Rat) | Subcutaneous injection | Retinal ischemia | 2015 | Reduced the increased leukocyte adherence to the vasculature and the pro-inflammatory phenotype of retinal immune cells | [245] |
| Resveratrol | AMPK /Sirt1/PGC-1α | Cell culture | Cell culture | DR | 2017 | Prevented ROS-induced apoptosis in high glucose-treated retinal capillary endothelial cells | [250] |
| Resveratrol | Bax-caspase-3 | Animal experiment (Rat) | Intraperitoneal injection | Glaucoma/ Retinal ischemia | 2018 | Significantly reduced retinal damage and RGC loss | [251] |
| Curcumin | PI3K/AKT/ mTOR | Cell culture | Cell culture | DR | 2018 | Increased anti-inflammatory effect to high glucose-induced inflammatory injury in human retinal pigment epithelial cells | [254] |
| Dimethyl fumarate | Nrf2 | Cell culture | Cell culture | AMD | 2020 | An efficient defensive strategy to prevent ROS-induced damage | [263] |
| Trimetazidine | Nrf2/HO-1 | Animal experiment (Mouse) | Intraperitoneal injection | Glaucoma/ Retinal ischemia | 2017 | Attenuated retinal damage and RGC death, with a decrease in ROS and inflammatory cytokine production | [264] |
| Tecfidera® | Nrt2 | Clinical trial | Oral | AMD | 2020 | Not finished yet | [265] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruan, Y.; Jiang, S.; Musayeva, A.; Gericke, A. Oxidative Stress and Vascular Dysfunction in the Retina: Therapeutic Strategies. Antioxidants 2020, 9, 761. https://doi.org/10.3390/antiox9080761
Ruan Y, Jiang S, Musayeva A, Gericke A. Oxidative Stress and Vascular Dysfunction in the Retina: Therapeutic Strategies. Antioxidants. 2020; 9(8):761. https://doi.org/10.3390/antiox9080761
Chicago/Turabian StyleRuan, Yue, Subao Jiang, Aytan Musayeva, and Adrian Gericke. 2020. "Oxidative Stress and Vascular Dysfunction in the Retina: Therapeutic Strategies" Antioxidants 9, no. 8: 761. https://doi.org/10.3390/antiox9080761
APA StyleRuan, Y., Jiang, S., Musayeva, A., & Gericke, A. (2020). Oxidative Stress and Vascular Dysfunction in the Retina: Therapeutic Strategies. Antioxidants, 9(8), 761. https://doi.org/10.3390/antiox9080761






