Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator
Abstract
1. Introduction
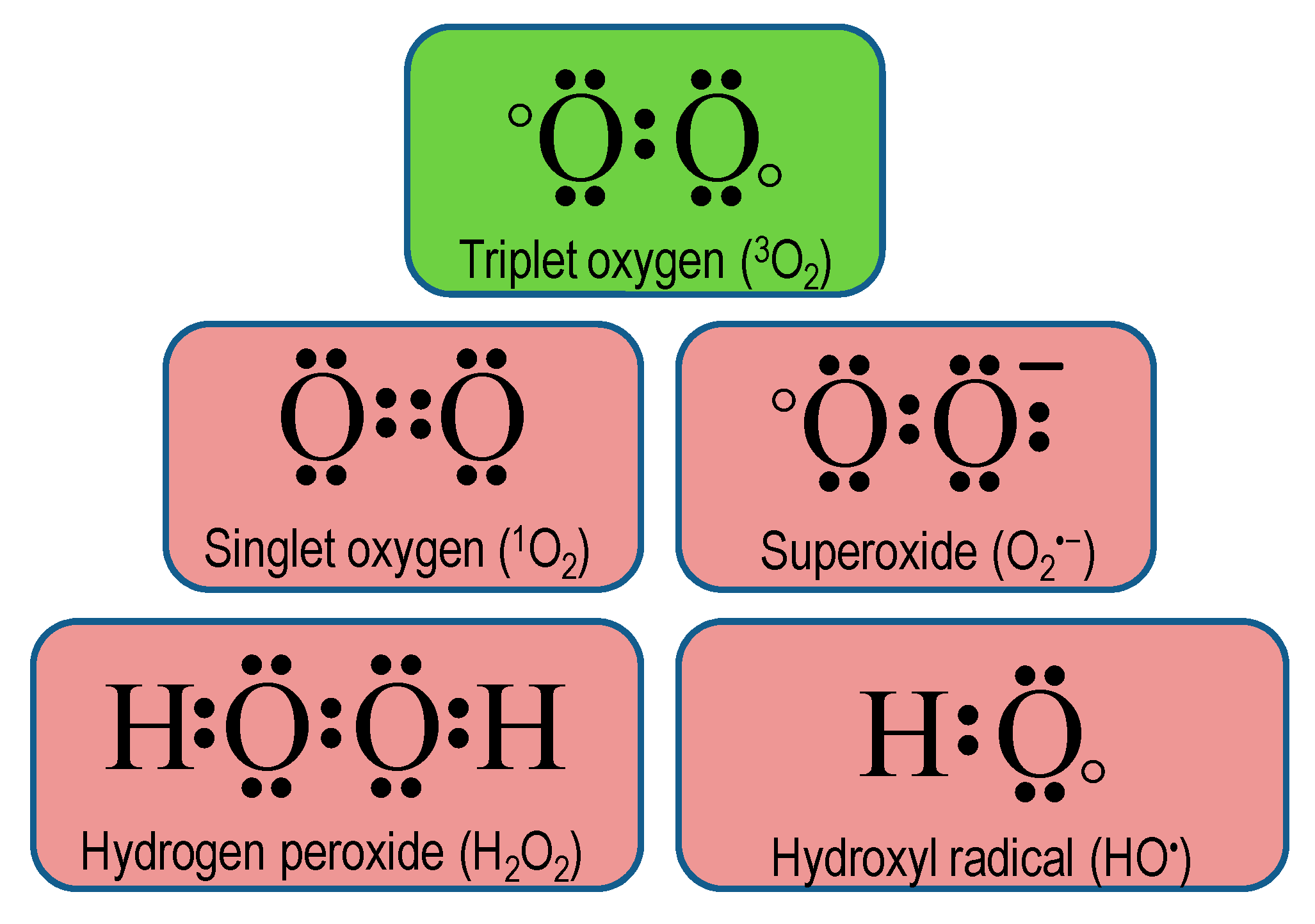
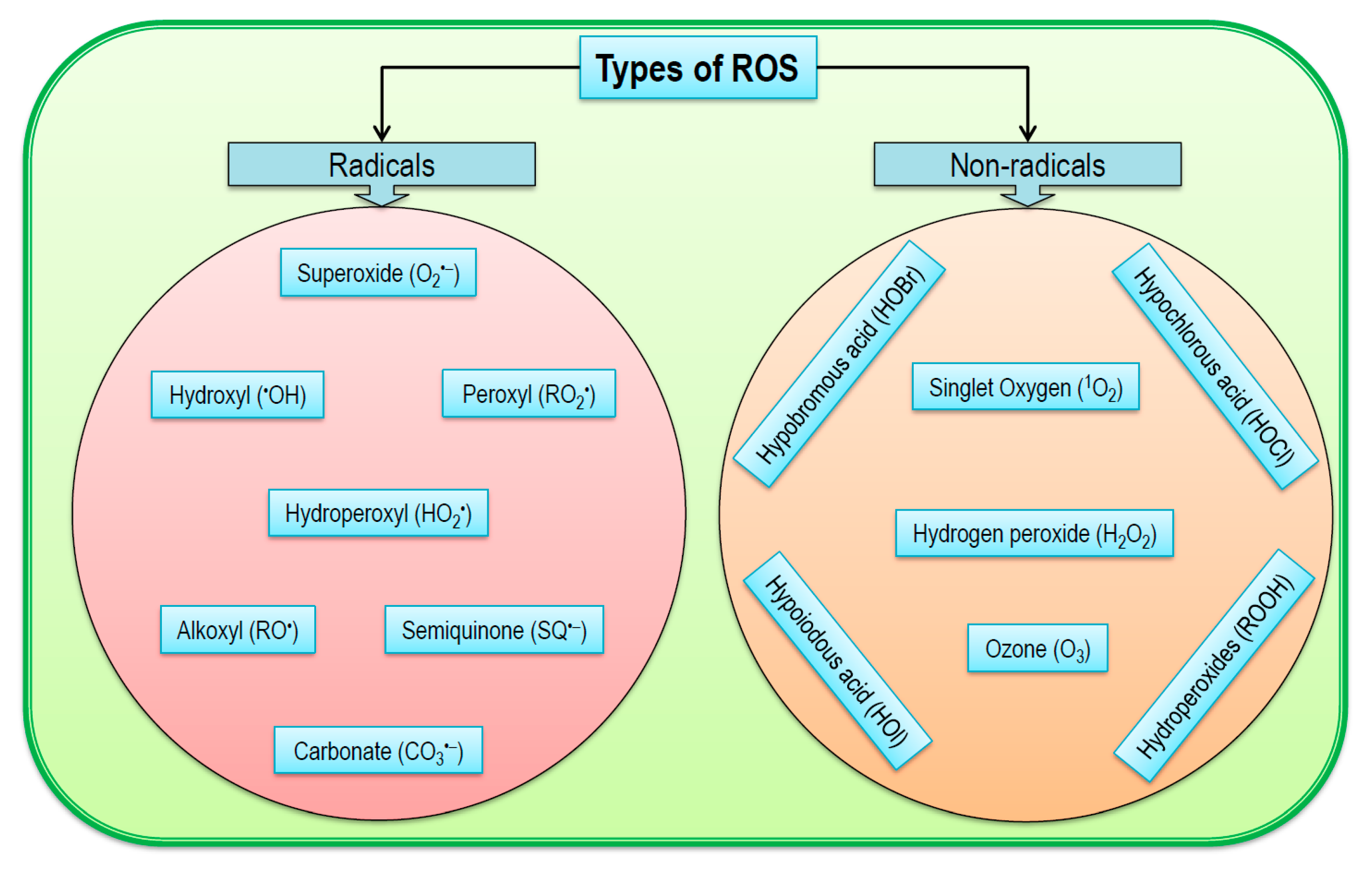
2. Chemistry of Reactive Oxygen Species
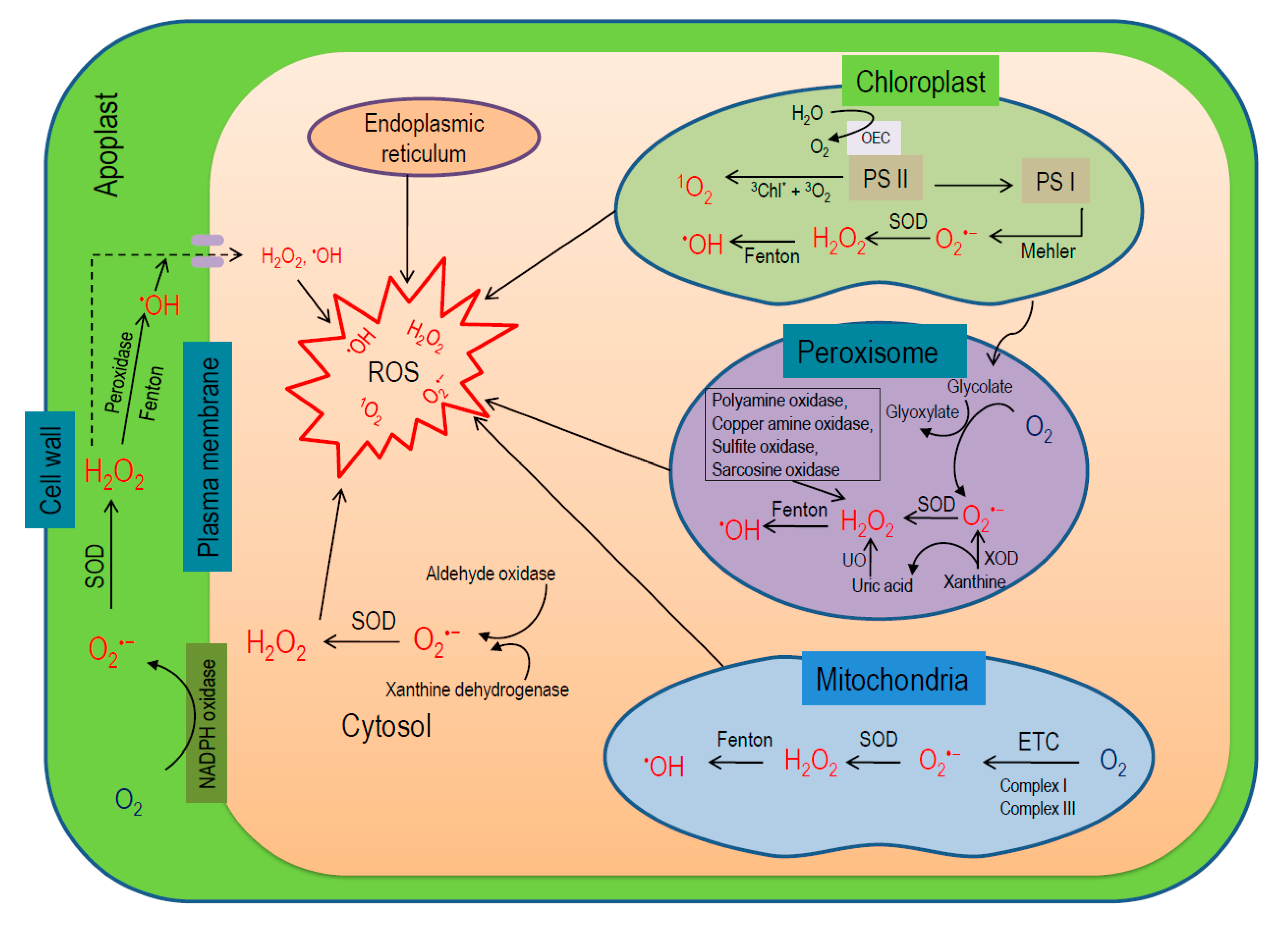
3. Localization and Processes of the Generation of ROS in Plant Cells
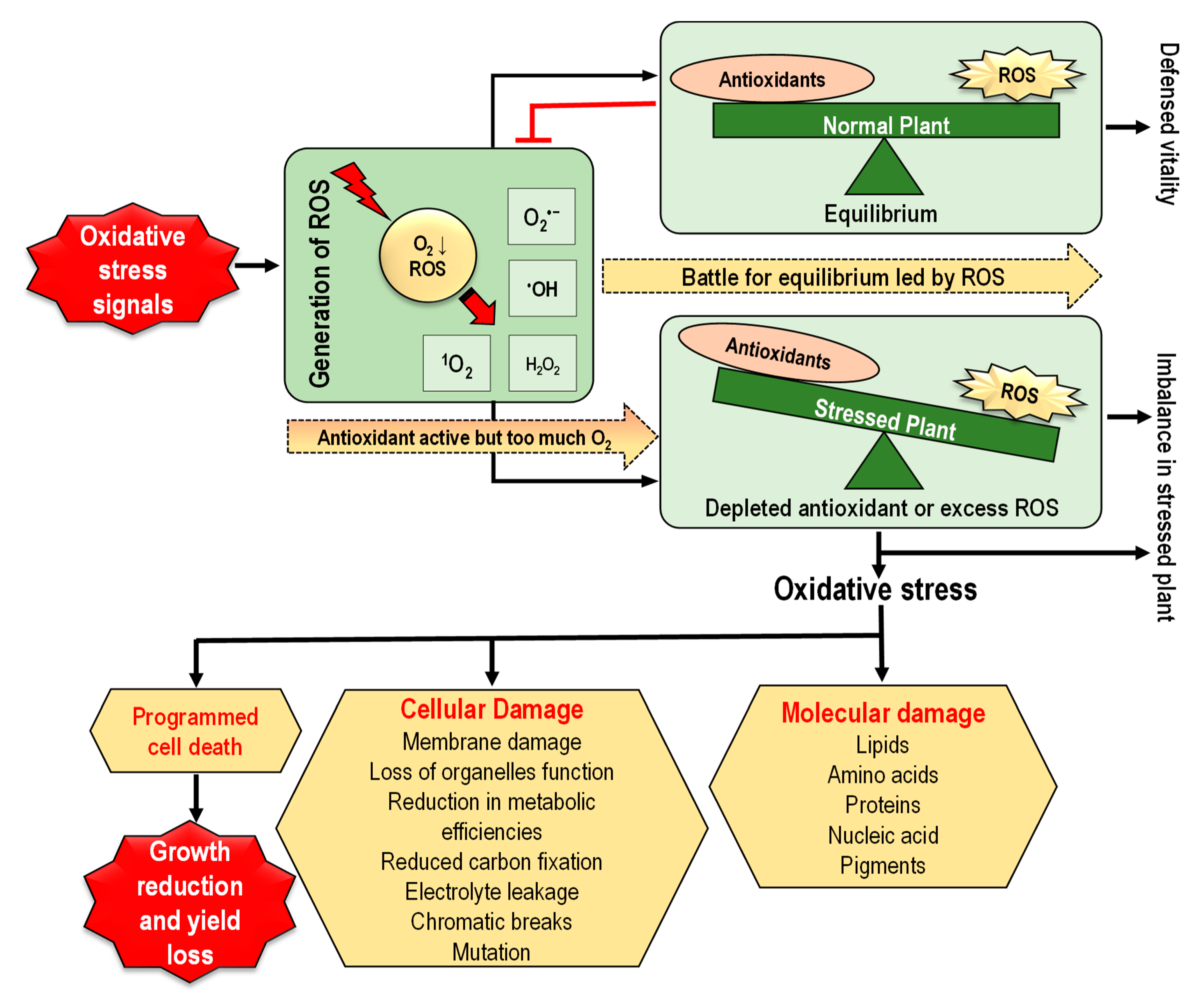
4. Oxidative Stress in Plants and Downstream Implications
5. Oxidative Stress under Abiotic Stress
5.1. Oxidative Stress under Salinity
5.2. Oxidative Stress under Water Deficit and Simulated Drought
5.3. Oxidative Stress under Metals/Metalloids Toxicity
5.4. Oxidative Stress under High Temperature
5.5. Oxidative Stress under Low Temperature
5.6. Oxidative Stress under Flooding
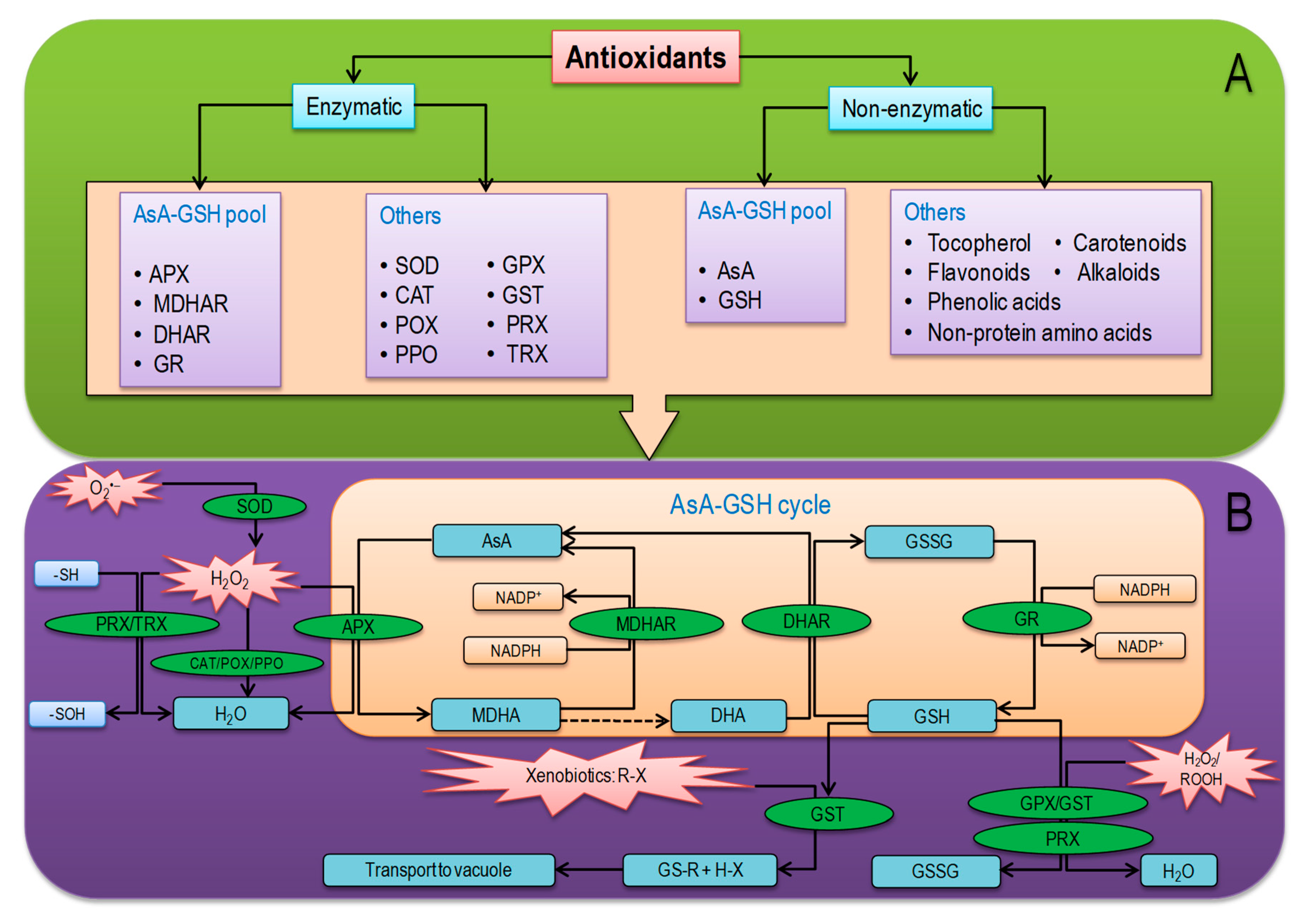
6. Overview of Plant Antioxidant Defense System
6.1. Nonenzymatic Antioxidants
6.2. Antioxidant Enzymes
7. Antioxidant Defense in Plants under Abiotic Stress: Recent Approaches
7.1. Antioxidant Defense in Plants under Salinity
7.2. Antioxidant Defense in Plants under Water Deficit and Simulated Drought
7.3. Antioxidant Defense in Plants under Toxic Metals/Metalloids
7.4. Antioxidant Defense in Plants under High Temperature
7.5. Antioxidant Defense in Plants under Low Temperature
7.6. Antioxidant Defense in Plants under Flooding
8. Revisiting ROS Signaling in Plant Defense
9. Cross-Talk of Reactive Nitrogen, Sulfur, and Carbonyl Species with ROS
9.1. Interaction between RNS and ROS
9.2. Interaction between RSS and ROS
9.3. Interaction between RCS and ROS
10. The Transgenic Approach in Enhancing Antioxidant Defense in Plants
11. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Pereira, A. Plant abiotic stress challenges from the changing environment. Front. Plant Sci. 2016, 7, 1123. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of climate change on crops adaptation and strategies to tackle its outcome: A review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [PubMed]
- Mehla, N.; Sindhi, V.; Josula, D.; Bisht, P.; Wani, S.H. An introduction to antioxidants and their roles in plant stress tolerance. In Reactive Oxygen Species and Antioxidant Systems in Plants: Role and Regulation under Abiotic Stress; Khan, M.I.R., Khan, N.A., Eds.; Springer: Singapore, 2017; pp. 1–23. [Google Scholar]
- Hasanuzzaman, M.; Bhuyan, M.; Anee, T.I.; Parvin, K.; Nahar, K.; Mahmud, J.A.; Fujita, M. Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 2019, 8, 384. [Google Scholar] [CrossRef]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Kumar, A.; Yadav, S.; Singh, I.K. Reactive oxygen species-mediated signaling during abiotic stress. Plant Gene 2019, 18, 100173. [Google Scholar] [CrossRef]
- Raja, V.; Majeed, U.; Kang, H.; Andrabi, K.I.; John, R. Abiotic stress: Interplay between ROS, hormones and MAPKs. Environ. Exp. Bot. 2017, 137, 142–157. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Hossain, M.A.; Teixeira da Silva, J.A.; Fujita, M. Plant responses and tolerance to abiotic oxidative stress: Antioxidant defense is a key factor. In Crop Stress and its Management: Perspectives and Strategies; Bandi, V., Shanker, A.K., Shanker, C., Mandapaka, M., Eds.; Springer: Berlin, Germany, 2012; pp. 261–316. [Google Scholar]
- Kaur, N.; Kaur, J.; Grewal, S.K.; Singh, I. Effect of Heat Stress on Antioxidative defense system and its amelioration by heat acclimation and salicylic acid pre-treatments in three pigeonpea genotypes. Indian J. Agric. Biochem. 2019, 32, 106–110. [Google Scholar] [CrossRef]
- Mittler, R. ROS are good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef]
- Antoniou, C.; Savvides, A.; Christou, A.; Fotopoulos, V. Unravelling chemical priming machinery in plants: The role of reactive oxygen–nitrogen–sulfur species in abiotic stress tolerance enhancement. Curr. Opin. Plant Biol. 2016, 33, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Sohag, A.A.M.; Tahjib-Ul-Arif, M.; Brestic, M.; Afrin, S.; Sakil, M.A.; Hossain, M.T.; Hossain, M.A.; Hossain, M.A. Exogenous salicylic acid and hydrogen peroxide attenuate drought stress in rice. Plant Soil Environ. 2020, 66, 7–13. [Google Scholar] [CrossRef]
- Habib, N.; Ali, Q.; Ali, S.; Javed, M.T.; Zulqurnain Haider, M.; Perveen, R.; Shahid, M.R.; Rizwan, M.; Abdel-Daim, M.M.; Elkelish, A. Use of Nitric oxide and hydrogen peroxide for better yield of wheat (Triticum aestivum L.) under water deficit conditions: Growth, osmoregulation, and antioxidative defense mechanism. Plants 2020, 9, 285. [Google Scholar] [CrossRef] [PubMed]
- Terzi, R.; Kadioglu, A.; Kalaycioglu, E.; Saglam, A. Hydrogen peroxide pretreatment induces osmotic stress tolerance by influencing osmolyte and abscisic acid levels in maize leaves. J. Plant Interact. 2014, 9, 559–565. [Google Scholar] [CrossRef]
- Fariduddin, Q.; Khan, T.A.; Yusuf, M. Hydrogen peroxide mediated tolerance to copper stress in the presence of 28-homobrassinolide in Vigna radiata. Acta Physiol. Plant. 2014, 36, 2767–2778. [Google Scholar] [CrossRef]
- Guler, N.S.; Pehlivan, N. Exogenous low-dose hydrogen peroxide enhances drought tolerance of soybean (Glycine max L.) through inducing antioxidant system. Acta Biol. Hung. 2016, 67, 169–183. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, H.; Liu, S.; Peng, X. Exogenous application of hydrogen peroxide alleviates drought stress in cucumber seedlings. S. Afr. J. Bot. 2016, 106, 23–28. [Google Scholar] [CrossRef]
- Tanou, G.; Filippou, P.; Belghazi, M.; Job, D.; Diamantidis, G.; Fotopoulos, V.; Molassiotis, A. Oxidative and nitrosative-based signaling and associated post-translational modifications orchestrate the acclimation of citrus plants to salinity stress. Plant J. 2012, 72, 585–599. [Google Scholar] [CrossRef]
- Christou, A.; Filippou, P.; Manganaris, G.A.; Fotopoulos, V. Sodium hydrosulfide induces systemic thermotolerance to strawberry plants through transcriptional regulation of heat shock proteins and aquaporin. BMC Plant Biol. 2014, 14, 1–11. [Google Scholar] [CrossRef]
- Gohari, G.; Alavi, Z.; Esfandiari, E.; Panahirad, S.; Hajihoseinlou, S.; Fotopoulos, V. Interaction between hydrogen peroxide and sodium nitroprusside following chemical priming of Ocimum basilicum L. against salt stress. Physiol. Plant. 2020, 168, 361–373. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Gill, S.S.; Alharby, H.F.; Razafindrabe, B.H.; Fujita, M. Hydrogen peroxide pretreatment mitigates cadmium-induced oxidative stress in Brassica napus L.: An intrinsic study on antioxidant defense and glyoxalase systems. Front. Plant Sci. 2017, 8, 115. [Google Scholar] [CrossRef]
- Yamasaki, H.; Ogura, M.P.; Kingjoe, K.A.; Cohen, M.F. D-Cysteine-induced rapid root abscission in the water fern Azolla pinnata: Implications for the linkage between d-amino acid and reactive sulfur species (RSS) in plant environmental responses. Antioxidants 2019, 8, 411. [Google Scholar] [CrossRef] [PubMed]
- Mailloux, J.R. Application of mitochondria-targeted pharmaceuticals for the treatment of heart disease. Curr. Pharm. Des. 2016, 22, 4763–4779. [Google Scholar] [CrossRef] [PubMed]
- Dmitrieva, V.A.; Tyutereva, E.V.; Voitsekhovskaja, O.V. Singlet oxygen in plants: Generation, detection, and signaling roles. Int. J. Mol. Sci. 2020, 21, 3237. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.B.; Hideg, E.; Krieger-Liszkay, A. Production, detection, and signaling of singlet oxygen in photosynthetic organisms. Antioxid. Redox Signal. 2013, 18, 2145–2162. [Google Scholar] [CrossRef]
- Rejeb, K.B.; Benzarti, M.; Debez, A.; Bailly, C.; Savouré, A.; Abdelly, C. NADPH oxidase-dependent H2O2 production is required for salt-induced antioxidant defense in Arabidopsis thaliana. J. Plant Physiol. 2015, 174, 5–15. [Google Scholar] [CrossRef]
- Berwal, M.K.; Ram, C. Superoxide dismutase: A stable biochemical marker for abiotic stress tolerance in higher plants. In Abiotic and Biotic Stress in Plants; De Oliveira, A., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Mhamdi, A.; Noctor, G.; Baker, A. Plant catalases: Peroxisomal redox guardians. Arch. Biochem. Biophys. 2012, 525, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Demidchik, V. Mechanisms of oxidative stress in plants: From classical chemistry to cell biology. Environ. Exp. Bot. 2015, 109, 212–228. [Google Scholar] [CrossRef]
- Maurya, A.K. Oxidative stress in crop plants. In Agronomic Crops; Hasanuzzaman, M., Ed.; Springer: Singapore, 2020; pp. 349–380. [Google Scholar]
- Kapoor, D.; Sharma, R.; Handa, N.; Kaur, H.; Rattan, A.; Yadav, P.; Gautam, V.; Kaur, R.; Bhardwaj, R. Redox homeostasis in plants under abiotic stress: Role of electron carriers, energy metabolism mediators and proteinaceous thiols. Front. Environ. Sci. 2015, 3, 13. [Google Scholar] [CrossRef]
- Nathan, C.; Ding, A. SnapShot: Reactive oxygen intermediates (ROI). Cell 2010, 140, 951. [Google Scholar] [CrossRef]
- Farnese, F.S.; Menezes-Silva, P.E.; Gusman, G.S.; Oliveira, J.A. When bad guys become good ones: The key role of reactive oxygen species and nitric oxide in the plant responses to abiotic stress. Front. Plant Sci. 2016, 7, 471. [Google Scholar] [CrossRef]
- Waszczak, C.; Carmody, M.; Kangasjärvi, J. Reactive oxygen species in plant signaling. Ann. Rev. Plant Biol. 2018, 69, 209–236. [Google Scholar] [CrossRef] [PubMed]
- Molassiotis, A.; Fotopoulos, V. Oxidative and nitrosative signaling in plants: Two branches in the same tree? Plant Signal. Behav. 2011, 6, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Koussevitzky, S.; Mittler, R.; Miller, G. ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ. 2012, 35, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Noctor, G.; Foyer, C.H. Intracellular redox compartmentation and ROS-related communication in regulation and signaling. Plant Physiol. 2016, 171, 1581–1592. [Google Scholar] [CrossRef]
- Kohli, S.K.; Khanna, K.; Bhardwaj, R.; Abd_Allah, E.F.; Ahmad, P.; Corpas, F.J. Assessment of subcellular ROS and NO metabolism in higher plants: Multifunctional signaling molecules. Antioxidants 2019, 8, 641. [Google Scholar] [CrossRef]
- Dietz, K.-J. Thiol-based peroxidases and ascorbate peroxidases: Why plants rely on multiple peroxidase systems in the photosynthesizing chloroplast? Mol. Cells 2016, 39, 20. [Google Scholar]
- Kim, C.; Dogra, V. Singlet oxygen metabolism: From genesis to signaling. Front. Plant Sci. 2019, 10, 1640. [Google Scholar]
- Müller, P.; Li, X.-P.; Niyogi, K.K. Non-photochemical quenching. A response to excess light energy. Plant Physiol. 2001, 125, 1558–1566. [Google Scholar] [CrossRef]
- Li, Z.; Wakao, S.; Fischer, B.B.; Niyogi, K.K. Sensing and responding to excess light. Ann. Rev. Plant Biol. 2009, 60, 239–260. [Google Scholar] [CrossRef]
- Krieger-Liszkay, A. Singlet oxygen production in photosynthesis. J. Exp. Bot. 2005, 56, 337–346. [Google Scholar] [CrossRef]
- Krieger-Liszkay, A.; Fufezan, C.; Trebst, A. Singlet oxygen production in photosystem II and related protection mechanism. Photosyn. Res. 2008, 98, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Flors, C.; Fryer, M.J.; Waring, J.; Reeder, B.; Bechtold, U.; Mullineaux, P.M.; Nonell, S.; Wilson, M.T.; Baker, N.R. Imaging the production of singlet oxygen in vivo using a new fluorescent sensor, Singlet Oxygen Sensor Green. J. Exp. Bot. 2006, 57, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Triantaphylidès, C.; Havaux, M. Singlet oxygen in plants: Production, detoxification and signaling. Trends Plant Sci. 2009, 14, 219–228. [Google Scholar] [CrossRef]
- Bose, J.; Rodrigo-Moreno, A.; Shabala, S. ROS homeostasis in halophytes in the context of salinity stress tolerance. J. Exp. Bot. 2014, 65, 1241–1257. [Google Scholar] [CrossRef] [PubMed]
- Kerchev, P.; Waszczak, C.; Lewandowska, A.; Willems, P.; Shapiguzov, A.; Li, Z.; Alseekh, S.; Mühlenbock, P.; Hoeberichts, F.A.; Huang, J. Lack of glycolate oxidase1, but not glycolate oxidase2, attenuates the photorespiratory phenotype of catalase2-deficient arabidopsis. Plant Physiol. 2016, 171, 1704–1719. [Google Scholar] [CrossRef] [PubMed]
- Del Río, L.A.; López-Huertas, E. ROS generation in peroxisomes and its role in cell signaling. Plant Cell Physiol. 2016, 57, 1364–1376. [Google Scholar] [CrossRef]
- Reumann, S.; Chowdhary, G.; Lingner, T. Characterization, prediction and evolution of plant peroxisomal targeting signals type 1 (PTS1s). Biochim. Biophys. Acta 2016, 1863, 790–803. [Google Scholar] [CrossRef]
- Corpas, F.J.; Del Río, L.A.; Palma, J.M. Plant peroxisomes at the crossroad of NO and H2O2 metabolism. J. Integr. Plant Biol. 2019, 61, 803–816. [Google Scholar]
- Gilroy, S.; Białasek, M.; Suzuki, N.; Górecka, M.; Devireddy, A.R.; Karpiński, S.; Mittler, R. ROS, calcium, and electric signals: Key mediators of rapid systemic signaling in plants. Plant Physiol. 2016, 171, 1606–1615. [Google Scholar] [CrossRef]
- Corpas, F.J.; González-Gordo, S.; Palma, J.M. Plant peroxisomes: A factory of reactive species. Front. Plant Sci. 2020, 11, 853. [Google Scholar] [CrossRef]
- Lisenbee, C.S.; Lingard, M.J.; Trelease, R.N. Arabidopsis peroxisomes possess functionally redundant membrane and matrix isoforms of monodehydroascorbate reductase. Plant J. 2005, 43, 900–914. [Google Scholar] [CrossRef] [PubMed]
- Leterrier, M.; Corpas, F.J.; Barroso, J.B.; Sandalio, L.M.; Luis, A. Peroxisomal monodehydroascorbate reductase. Genomic clone characterization and functional analysis under environmental stress conditions. Plant Physiol. 2005, 138, 2111–2123. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, A.; Kumar, A.; Kaur, N. ROS and oxidative burst: Roots in plant development. Plant Divers. 2020, 42, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2012. [Google Scholar] [CrossRef]
- Heyno, E.; Mary, V.; Schopfer, P.; Krieger-Liszkay, A. Oxygen activation at the plasma membrane: Relation between superoxide and hydroxyl radical production by isolated membranes. Planta 2011, 234, 35–45. [Google Scholar] [CrossRef]
- Jeevan Kumar, S.; Rajendra Prasad, S.; Banerjee, R.; Thammineni, C. Seed birth to death: Dual functions of reactive oxygen species in seed physiology. Ann. Bot. 2015, 116, 663–668. [Google Scholar] [CrossRef]
- Janků, M.; Luhová, L.; Petřivalský, M. On the origin and fate of reactive oxygen species in plant cell compartments. Antioxidants 2019, 8, 105. [Google Scholar] [CrossRef]
- Decros, G.; Baldet, P.; Beauvoit, B.; Stevens, R.; Flandin, A.; Colombié, S.; Gibon, Y.; Pétriacq, P. Get the balance right: ROS homeostasis and redox signalling in fruit. Front. Plant Sci. 2019, 10, 1091. [Google Scholar] [CrossRef]
- Paciolla, C.; Paradiso, A.; de Pinto, M.C. Cellular redox homeostasis as central modulator in plant stress response. In Redox State as a Central Regulator of Plant-Cell Stress Responses; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2016; pp. 1–23. [Google Scholar]
- Reczek, C.R.; Chandel, N.S. ROS-dependent signal transduction. Curr. Opin. Cell Biol. 2015, 33, 8–13. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef]
- Finkel, T. Signal transduction by reactive oxygen species. J. Cell Biol. 2011, 194, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Panieri, E.; Santoro, M.M. ROS signaling and redox biology in endothelial cells. Cell. Mol. Life Sci. 2015, 72, 3281–3303. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, J.A.; Bhuyan, M.H.M.B.; Anee, T.I.; Nahar, K.; Fujita, M.; Hasanuzzaman, M. Reactive oxygen species metabolism and antioxidant defense in plants under metal/metalloid stress. In Plant Abiotic Stress Tolerance; Hasanuzzaman, M., Hakeem, K., Nahar, K., Alharby, H., Eds.; Springer: Cham, Switzerland, 2019; pp. 221–257. [Google Scholar]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Abbas, G.; Shahid, M.; Saqib, M.; Farooq, A.B.U.; Hussain, M.; Murtaza, B.; Amjad, M.; Naeem, M.A.; Farooq, A. Effect of salinity on cadmium tolerance, ionic homeostasis and oxidative stress responses in conocarpus exposed to cadmium stress: Implications for phytoremediation. Ecotoxicol. Environ. Saf. 2019, 171, 146–153. [Google Scholar] [CrossRef]
- Cheng, Y.-W.; Kong, X.-W.; Wang, N.; Wang, T.-T.; Chen, J.; Shi, Z.Q. Thymol confers tolerance to salt stress by activating anti-oxidative defense and modulating Na+ homeostasis in rice root. Ecotoxicol. Environ. Saf. 2020, 188, 109894. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Mir, R.A.; Alyemeni, M.N.; Ahmad, P. Combined effects of brassinosteroid and kinetin mitigates salinity stress in tomato through the modulation of antioxidant and osmolyte metabolism. Plant Physiol. Biochem. 2020, 147, 31–42. [Google Scholar] [CrossRef]
- Abdelaal, K.A.; EL-Maghraby, L.M.; Elansary, H.; Hafez, Y.M.; Ibrahim, E.I.; El-Banna, M.; El-Esawi, M.; Elkelish, A. Treatment of sweet pepper with stress tolerance-inducing compounds alleviates salinity stress oxidative damage by mediating the physio-biochemical activities and antioxidant systems. Agronomy 2020, 10, 26. [Google Scholar] [CrossRef]
- Ahmad, P.; Ahanger, M.A.; Alam, P.; Alyemeni, M.N.; Wijaya, L.; Ali, S.; Ashraf, M. Silicon (Si) supplementation alleviates NaCl toxicity in mung bean [Vigna radiata (L.) Wilczek] through the modifications of physio-biochemical attributes and key antioxidant enzymes. J. Plant Growth Regul. 2019, 38, 70–82. [Google Scholar] [CrossRef]
- Arora, M.; Saxena, P.; Abdin, M.; Varma, A. Interaction between Piriformospora indica and Azotobacter chroococcum diminish the effect of salt stress in Artemisia annua L. by enhancing enzymatic and non-enzymatic antioxidants. Symbiosis 2020, 80, 61–73. [Google Scholar] [CrossRef]
- Lalarukh, I.; Shahbaz, M. Response of antioxidants and lipid peroxidation to exogenous application of alpha-tocopherol in sunflower (Helianthus annuus L.) under salt stress. Pak. J. Bot. 2020, 52, 75–83. [Google Scholar] [CrossRef]
- Tariq, A.; Shahbaz, M. Glycinebetaine induced modulation in oxidative defense system and mineral nutrients sesame (Sesamum indicum L.) under saline regimes. Pak. J. Bot. 2020, 52, 775–782. [Google Scholar] [CrossRef]
- Mhadhbi, H.; Fotopoulos, V.; Mylona, P.V.; Jebara, M.; Aouani, M.E.; Polidoros, A.N. Alternative oxidase 1 (AOX1) gene expression in roots of Medicago truncatulais a genotype-specific component of salt stress tolerance. J. Plant Physiol. 2013, 170, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Filippou, P.; Bouchagier, P.; Skotti, E.; Fotopoulos, V. Proline and reactive oxygen/nitrogen species metabolism is involved in the tolerant response of the invasive plant species Ailanthus altissima to drought and salinity. Environ. Exp. Bot. 2014, 97, 1–10. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Khan, M.I.R.; Al Mahmud, J.; Alam, M.M.; Fujita, M. Regulation of reactive oxygen species metabolism and glyoxalase systems by exogenous osmolytes confers thermotolerance in Brassica napus. Gesunde Pflanz. 2020, 72, 3–16. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Gill, S.S.; Fujita, M. Drought stress responses in plants, oxidative stress, and antioxidant defense. In Climate Change and Plant Abiotic Stress Tolerance; Tuteja, N., Gill, S.S., Eds.; Wiley: Weinheim, Germany, 2014; pp. 209–250. [Google Scholar]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S. Crop production under drought and heat stress: Plant responses and management options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Rahman, A.; Inafuku, M.; Oku, H.; Fujita, M. Exogenous nitric oxide donor and arginine provide protection against short-term drought stress in wheat seedlings. Physiol. Mol. Biol. Plants 2018, 24, 993–1004. [Google Scholar] [CrossRef]
- Abideen, Z.; Koyro, H.W.; Huchzermeyer, B.; Ansari, R.; Zulfiqar, F.; Gul, B. Ameliorating effects of biochar on photosynthetic efficiency and antioxidant defence of Phragmites karka under drought stress. Plant Biol. 2020, 22, 259–266. [Google Scholar] [CrossRef]
- Campos, C.N.; Ávila, R.G.; de Souza, K.R.D.; Azevedo, L.M.; Alves, J.D. Melatonin reduces oxidative stress and promotes drought tolerance in young Coffea arabica L. plants. Agric. Water Manag. 2019, 211, 37–47. [Google Scholar] [CrossRef]
- Saha, I.; De, A.K.; Sarkar, B.; Ghosh, A.; Dey, N.; Adak, M.K. Cellular response of oxidative stress when sub1A QTL of rice receives water deficit stress. Plant Sci. Today 2018, 5, 84–94. [Google Scholar] [CrossRef]
- Satish, L.; Rency, A.S.; Ramesh, M. Spermidine sprays alleviate the water deficit-induced oxidative stress in finger millet (Eleusine coracana L. Gaertn.) plants. 3 Biotech 2018, 8, 63. [Google Scholar] [CrossRef]
- Malhotra, C.; Kapoor, R.T.; Ganjewala, D.; Singh, N. Sodium silicate mediated response of antioxidative defense system in Lycopersicon esculentum mill. under water stress. Int. J. Phytomed. 2017, 9, 364–378. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Hossain, M.S.; Anee, T.I.; Parvin, K.; Fujita, M. Nitric oxide pretreatment enhances antioxidant defense and glyoxalase systems to confer PEG-induced oxidative stress in rapeseed. J. Plant Interact. 2017, 12, 323–331. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Anee, T.; Khan, M.; Fujita, M. Silicon-mediated regulation of antioxidant defense and glyoxalase systems confers drought stress tolerance in Brassica napus L. S. Afr. J. Bot. 2018, 115, 50–57. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Rahman, A.; Mahmud, J.A.; Suzuki, T.; Fujita, M. Insights into spermine-induced combined high temperature and drought tolerance in mung bean: Osmoregulation and roles of antioxidant and glyoxalase system. Protoplasma 2017, 254, 445–460. [Google Scholar] [CrossRef]
- Abbas, T.; Rizwan, M.; Ali, S.; Adrees, M.; Mahmood, A.; Zia-ur-Rehman, M.; Ibrahim, M.; Arshad, M.; Qayyum, M.F. Biochar application increased the growth and yield and reduced cadmium in drought stressed wheat grown in an aged contaminated soil. Ecotoxicol. Environ. Saf. 2018, 148, 825–833. [Google Scholar] [CrossRef]
- Liu, J.; Hasanuzzaman, M.; Wen, H.; Zhang, J.; Peng, T.; Sun, H.; Zhao, Q. High temperature and drought stress cause abscisic acid and reactive oxygen species accumulation and suppress seed germination growth in rice. Protoplasma 2019, 256, 1217–1227. [Google Scholar] [CrossRef]
- Rezayian, M.; Ebrahimzadeh, H.; Niknam, V. Nitric oxide stimulates antioxidant system and osmotic adjustment in soybean under drought stress. J. Soil Sci. Plant Nutr. 2020, 1–11. [Google Scholar] [CrossRef]
- Rady, M.M.; Belal, H.E.; Gadallah, F.M.; Semida, W.M. Selenium application in two methods promotes drought tolerance in Solanum lycopersicum plant by inducing the antioxidant defense system. Sci. Hortic. 2020, 266, 109290. [Google Scholar] [CrossRef]
- Filippou, P.; Antoniou, C.; Fotopoulos, V. Effect of drought and rewatering on the cellular status and antioxidant response of Medicago truncatula plants. Plant Signal. Behav. 2011, 6, 270–277. [Google Scholar] [CrossRef]
- Kusvuran, S.; Dasgan, H.Y. Effects of drought stress on physiological and biochemical changes in Phaseolus vulgaris L. Legume Res. 2017, 40, 55–62. [Google Scholar]
- Hasanuzzaman, M.; Alam, M.M.; Nahar, K.; Mohsin, S.M.; Bhuyan, M.B.; Parvin, K.; Hawrylak-Nowak, B.; Fujita, M. Silicon-induced antioxidant defense and methylglyoxal detoxification works coordinately in alleviating nickel toxicity in Oryza sativa L. Ecotoxicology 2019, 28, 261–276. [Google Scholar] [CrossRef]
- El-Amier, Y.; Elhindi, K.; El-Hendawy, S.; Al-Rashed, S.; Abd-ElGawad, A. Antioxidant system and biomolecules alteration in Pisum sativum under heavy metal stress and possible alleviation by 5-aminolevulinic acid. Molecules 2019, 24, 4194. [Google Scholar] [CrossRef]
- Nahar, K.; Rahman, M.; Hasanuzzaman, M.; Alam, M.M.; Rahman, A.; Suzuki, T.; Fujita, M. Physiological and biochemical mechanisms of spermine-induced cadmium stress tolerance in mung bean (Vigna radiata L.) seedlings. Environ. Sci. Pollut. Res. 2016, 23, 21206–21218. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Anee, T.I.; Fujita, M. Exogenous silicon attenuates cadmium-induced oxidative stress in Brassica napus L. by modulating AsA-GSH pathway and glyoxalase system. Front. Plant Sci. 2017, 8, 1061. [Google Scholar] [CrossRef]
- Al Mahmud, J.; Hasanuzzaman, M.; Nahar, K.; Bhuyan, M.B.; Fujita, M. Insights into citric acid-induced cadmium tolerance and phytoremediation in Brassica juncea L.: Coordinated functions of metal chelation, antioxidant defense and glyoxalase systems. Ecotoxicol. Environ. Saf. 2018, 147, 990–1001. [Google Scholar] [CrossRef]
- Gupta, D.; Pena, L.B.; Romero-Puertas, M.C.; Hernández, A.; Inouhe, M.; Sandalio, L.M. NADPH oxidases differentially regulate ROS metabolism and nutrient uptake under cadmium toxicity. Plant Cell Environ. 2017, 40, 509–526. [Google Scholar] [CrossRef]
- Kabała, K.; Zboińska, M.; Głowiak, D.; Reda, M.; Jakubowska, D.; Janicka, M. Interaction between the signaling molecules hydrogen sulfide and hydrogen peroxide and their role in vacuolar H+-ATPase regulation in cadmium-stressed cucumber roots. Physiol. Plant. 2019, 166, 688–704. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Aziz, U.; Sahli, A.A.; Alyemeni, M.N.; Ahmad, P. Combined kinetin and spermidine treatments ameliorate growth and photosynthetic inhibition in Vigna angularis by up-regulating antioxidant and nitrogen metabolism under cadmium stress. Biomolecules 2020, 10, 147. [Google Scholar] [CrossRef]
- Zaid, A.; Mohammad, F.; Fariduddin, Q. Plant growth regulators improve growth, photosynthesis, mineral nutrient and antioxidant system under cadmium stress in menthol mint (Mentha arvensis L.). Physiol. Mol. Biol. Plants 2020, 26, 25–39. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Rahman, A.; Mahmud, J.A.; Alharby, H.F.; Fujita, M. Exogenous glutathione attenuates lead-induced oxidative stress in wheat by improving antioxidant defense and physiological mechanisms. J. Plant Interact. 2018, 13, 203–212. [Google Scholar] [CrossRef]
- Yadu, B.; Chandrakar, V.; Tamboli, R.; Keshavkant, S. Dimethylthiourea antagonizes oxidative responses by up-regulating expressions of pyrroline-5-carboxylate synthetase and antioxidant genes under arsenic stress. Int. J. Environ. Sci. Technol. 2019, 16, 8401–8410. [Google Scholar] [CrossRef]
- Parvez, S.; Abbas, G.; Shahid, M.; Amjad, M.; Hussain, M.; Asad, S.A.; Imran, M.; Naeem, M.A. Effect of salinity on physiological, biochemical and photostabilizing attributes of two genotypes of quinoa (Chenopodium quinoa Willd.) exposed to arsenic stress. Ecotoxicol. Environ. Saf. 2020, 187, 109814. [Google Scholar] [CrossRef] [PubMed]
- Georgiadou, E.C.; Kowalska, E.; Patla, K.; Kulbat, K.; Smolińska, B.; Leszczyńska, J.; Fotopoulos, V. Influence of heavy metals (Ni, Cu, and Zn) on nitro-oxidative stress responses, proteome regulation and allergen production in basil (Ocimum basilicum L.) plants. Front. Plant Sci. 2018, 9, 862. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.; Roychowdhury, R.; Fujita, M. Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef]
- Fahad, S.; Hussain, S.; Saud, S.; Khan, F.; Hassan, S.; Nasim, W.; Arif, M.; Wang, F.; Huang, J. Exogenously applied plant growth regulators affect heat-stressed rice pollens. J. Agron. Crop Sci. 2016, 202, 139–150. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; He, L.; Zhou, Q.; Yu, J.; Hui, D.; Huang, D. Exogenous glutathione improves high root-zone temperature tolerance by modulating photosynthesis, antioxidant and osmolytes systems in cucumber seedlings. Sci. Rep. 2016, 6, 35424. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Perumal, R.; Jagadish, S.; Ciampitti, I.; Welti, R.; Prasad, P. Sensitivity of sorghum pollen and pistil to high-temperature stress. Plant Cell Environ. 2018, 41, 1065–1082. [Google Scholar] [CrossRef]
- Repetto, M.; Semprine, J.; Boveris, A. Lipid peroxidation: Chemical mechanism, biological implications and analytical determination. In Lipid Peroxidation; Catala, A., Ed.; InTech: Rijeka, Croatia, 2012; pp. 3–30. [Google Scholar]
- Han, Q.-H.; Huang, B.; Ding, C.-B.; Zhang, Z.-W.; Chen, Y.-E.; Hu, C.; Zhou, L.-J.; Huang, Y.; Liao, J.-Q.; Yuan, S. Effects of melatonin on anti-oxidative systems and photosystem II in cold-stressed rice seedlings. Front. Plant Sci. 2017, 8, 785. [Google Scholar] [CrossRef]
- Liu, T.; Ye, X.; Li, M.; Li, J.; Qi, H.; Hu, X. H2O2 and NO are involved in trehalose-regulated oxidative stress tolerance in cold-stressed tomato plants. Environ. Exp. Bot. 2020, 171, 103961. [Google Scholar] [CrossRef]
- Xue, M.; Guo, T.; Ren, M.; Wang, Z.; Tang, K.; Zhang, W.; Wang, M. Constitutive expression of chloroplast glycerol-3-phosphate acyltransferase from Ammopiptanthus mongolicus enhances unsaturation of chloroplast lipids and tolerance to chilling, freezing and oxidative stress in transgenic Arabidopsis. Plant Physiol. Biochem. 2019, 143, 375–387. [Google Scholar] [CrossRef]
- Loreti, E.; van Veen, H.; Perata, P. Plant responses to flooding stress. Curr. Opin. Plant Biol. 2016, 33, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Zhou, Y.; Yue, Z.; Chen, X.; Cao, X.; Xu, X.; Xing, Y.; Jiang, B.; Ai, X.; Huang, R. Changes in photosynthesis, chloroplast ultrastructure, and antioxidant metabolism in leaves of sorghum under waterlogging stress. Photosynthetica 2019, 57, 1076–1083. [Google Scholar] [CrossRef]
- Anee, T.I.; Nahar, K.; Rahman, A.; Mahmud, J.A.; Bhuiyan, T.F.; Alam, M.U.; Fujita, M.; Hasanuzzaman, M. Oxidative damage and antioxidant defense in Sesamum indicum after different waterlogging durations. Plants 2019, 8, 196. [Google Scholar] [CrossRef]
- Rasheed, R.; Iqbal, M.; Ashraf, M.A.; Hussain, I.; Shafiq, F.; Yousaf, A.; Zaheer, A. Glycine betaine counteracts the inhibitory effects of waterlogging on growth, photosynthetic pigments, oxidative defence system, nutrient composition, and fruit quality in tomato. J. Hortic. Sci. Biotechnol. 2018, 93, 385–391. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, E.J. Waterlogging induced oxidative stress and the mortality of the Antarctic plant, Deschampsia antarctica. J. Ecol. Environ. 2019, 43, 29. [Google Scholar] [CrossRef]
- Alzahrani, S.M.; Alaraidh, I.A.; Migdadi, H.; Alghamdi, S.; Khan, M.A.; Ahmad, P. Physiological, biochemical, and antioxidant properties of two genotypes of Vicia faba grown under salinity stress. Pak. J. Bot. 2019, 51, 786–798. [Google Scholar] [CrossRef]
- Hossain, M.; Alam, M.; Rahman, A.; Hasanuzzaman, M.; Nahar, K.; Al Mahmud, J.; Fujita, M. Use of iso-osmotic solution to understand salt stress responses in lentil (Lens culinaris Medik.). S. Afr. J. Bot. 2017, 113, 346–354. [Google Scholar] [CrossRef]
- Nxele, X.; Klein, A.; Ndimba, B. Drought and salinity stress alters ROS accumulation, water retention, and osmolyte content in sorghum plants. S. Afr. J. Bot. 2017, 108, 261–266. [Google Scholar] [CrossRef]
- Derbali, W.; Goussi, R.; Koyro, H.-W.; Abdelly, C.; Manaa, A. Physiological and biochemical markers for screening salt tolerant quinoa genotypes at early seedling stage. J. Plant Interact. 2020, 15, 27–38. [Google Scholar] [CrossRef]
- Han, F.; Sun, M.; He, W.; Cui, X.; Pan, H.; Wang, H.; Song, F.; Lou, Y.; Zhuge, Y. Ameliorating effects of exogenous Ca2+ on foxtail millet seedlings under salt stress. Funct. Plant Biol. 2019, 46, 407–416. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, C.; Chatzimichail, G.; Xenofontos, R.; Pavlou, J.J.; Panagiotou, E.; Christou, A.; Fotopoulos, V. Melatonin systemically ameliorates drought stress-induced damage in Medicago sativa plants by modulating nitro-oxidative homeostasis and proline metabolism. J. Pineal Res. 2017, 62, e12401. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Anwar, Y.; Hasan, M.; Iqbal, A.; Ali, M.; Alharby, H.F.; Hakeem, K.R.; Hasanuzzaman, M. Attenuation of drought stress in Brassica seedlings with exogenous application of Ca2+ and H2O2. Plants 2017, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, T.F.; Ahamed, K.U.; Nahar, K.; Al Mahmud, J.; Bhuyan, M.B.; Anee, T.I.; Fujita, M.; Hasanuzzaman, M. Mitigation of PEG-induced drought stress in rapeseed (Brassica rapa L.) by exogenous application of osmolytes. Biocatal. Agric. Biotechnol. 2019, 20, 101197. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Suzuki, T.; Fujita, M. Polyamines-induced aluminum tolerance in mung bean: A study on antioxidant defense and methylglyoxal detoxification systems. Ecotoxicology 2017, 26, 58–73. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, M.; Saleem, M.F.; Ullah, N.; Rizwan, M.; Ali, S.; Shahid, M.R.; Alamri, S.A.; Alyemeni, M.N.; Ahmad, P. Exogenously applied growth regulators protect the cotton crop from heat-induced injury by modulating plant defense mechanism. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Diao, Q.; Song, Y.; Shi, D.; Qi, H. Interaction of polyamines, abscisic acid, nitric oxide, and hydrogen peroxide under chilling stress in tomato (Lycopersicon esculentum Mill.) seedlings. Front. Plant Sci. 2017, 8, 203. [Google Scholar] [CrossRef]
- Ghanbari, F.; Sayyari, M. Controlled drought stress affects the chilling-hardening capacity of tomato seedlings as indicated by changes in phenol metabolisms, antioxidant enzymes activity, osmolytes concentration and abscisic acid accumulation. Sci. Hortic. 2018, 229, 167–174. [Google Scholar] [CrossRef]
- Luan, H.; Shen, H.; Pan, Y.; Guo, B.; Lv, C.; Xu, R. Elucidating the hypoxic stress response in barley (Hordeum vulgare L.) during waterlogging: A proteomics approach. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef]
- Nath, M.; Bhatt, D.; Bhatt, M.D.; Prasad, R.; Tuteja, N. Microbe-mediated enhancement of nitrogen and phosphorus content for crop improvement. In Crop Improvement through Microbial Biotechnology; Prasad, R., Gill, S.S., Tuteja, N., Eds.; Academic Press: Cambridge, UK, 2018; pp. 291–301. [Google Scholar]
- Laxa, M.; Liebthal, M.; Telman, W.; Chibani, K.; Dietz, K.-J. The role of the plant antioxidant system in drought tolerance. Antioxidants 2019, 8, 94. [Google Scholar] [CrossRef]
- Biczak, R. Quaternary ammonium salts with tetrafluoroborate anion: Phytotoxicity and oxidative stress in terrestrial plants. J. Hazard. Mater. 2016, 304, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Del Río, L.A.; Corpas, F.J.; López-Huertas, E.; Palma, J.M. Plant superoxide dismutases: Function under abiotic stress conditions. In Antioxidants and Antioxidant Enzymes in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 1–26. [Google Scholar]
- Fotopoulos, V.; Tanou, G.; Ziogas, V.; Molassiotis, A. Involvement of AsA/DHA and GSH/GSSG ratios in gene and protein expression and in the activation of defense mechanisms under abiotic stress conditions. In Ascorbate-Glutathione Pathway and Stress Tolerance in Plants; Anjum, N.A., Chan, M.T., Umar, S., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 265–302. [Google Scholar]
- Hasanuzzaman, M.; Bhuyan, M.; Mahmud, J.; Nahar, K.; Mohsin, S.; Parvin, K.; Fujita, M. Interaction of sulfur with phytohormones and signaling molecules in conferring abiotic stress tolerance to plants. Plant Signal. Behav. 2018, 13, e1477905. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Ascorbate and glutathione: The heart of the redox hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, D.; Singh, S.; Kumar, V.; Romero, R.; Prasad, R.; Singh, J. Antioxidant enzymes regulation in plants in reference to reactive oxygen species (ROS) and reactive nitrogen species (RNS). Plant Gene 2019, 19, 100182. [Google Scholar] [CrossRef]
- Naz, H.; Akram, N.A.; Ashraf, M. Impact of ascorbic acid on growth and some physiological attributes of cucumber (Cucumis sativus) plants under water-deficit conditions. Pak. J. Bot. 2016, 48, 877–883. [Google Scholar]
- Seminario, A.; Song, L.; Zulet, A.; Nguyen, H.T.; González, E.M.; Larrainzar, E. Drought stress causes a reduction in the biosynthesis of ascorbic acid in soybean plants. Front. Plant Sci. 2017, 8, 1042. [Google Scholar] [CrossRef]
- Kumar, S.; Thakur, P.; Kaushal, N.; Malik, J.A.; Gaur, P.; Nayyar, H. Effect of varying high temperatures during reproductive growth on reproductive function, oxidative stress and seed yield in chickpea genotypes differing in heat sensitivity. Arch. Agron. Soil Sci. 2013, 59, 823–843. [Google Scholar] [CrossRef]
- Hussain, H.A.; Men, S.; Hussain, S.; Chen, Y.; Ali, S.; Zhang, S.; Zhang, K.; Li, Y.; Xu, Q.; Liao, C. Interactive effects of drought and heat stresses on morpho-physiological attributes, yield, nutrient uptake and oxidative status in maize hybrids. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Liu, S.; Ju, J.; Xia, G. Identification of the flavonoid 3′-hydroxylase and flavonoid 3′, 5′-hydroxylase genes from Antarctic moss and their regulation during abiotic stress. Gene 2014, 543, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Di Ferdinando, M.; Brunetti, C.; Fini, A.; Tattini, M. Flavonoids as antioxidants in plants under abiotic stresses. In Abiotic Stress Responses in Plants; Ahmad, P., Prasad, M., Eds.; Springer: New York, NY, USA, 2012; pp. 159–179. [Google Scholar]
- Tiong, S.H.; Looi, C.Y.; Hazni, H.; Arya, A.; Paydar, M.; Wong, W.F.; Cheah, S.-C.; Mustafa, M.R.; Awang, K. Antidiabetic and antioxidant properties of alkaloids from Catharanthus roseus (L.) G. Don. Molecules 2013, 18, 9770–9784. [Google Scholar] [CrossRef] [PubMed]
- Vranova, V.; Rejsek, K.; Skene, K.R.; Formanek, P. Non-protein amino acids: Plant, soil and ecosystem interactions. Plant Soil 2011, 342, 31–48. [Google Scholar] [CrossRef]
- Gill, S.S.; Anjum, N.A.; Gill, R.; Yadav, S.; Hasanuzzaman, M.; Fujita, M.; Mishra, P.; Sabat, S.C.; Tuteja, N. Superoxide dismutase—mentor of abiotic stress tolerance in crop plants. Environ. Sci. Pollut. Res. 2015, 22, 10375–10394. [Google Scholar] [CrossRef]
- Jovanović, S.V.; Kukavica, B.; Vidović, M.; Morina, F.; Menckhoff, L. Class III peroxidases: Functions, localization and redox regulation of isoenzymes. In Antioxidants and Antioxidant Enzymes in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 269–300. [Google Scholar]
- Boeckx, T.; Winters, A.L.; Webb, K.J.; Kingston-Smith, A.H. Polyphenol oxidase in leaves: Is there any significance to the chloroplastic localization? J. Exp. Bot. 2015, 66, 3571–3579. [Google Scholar] [CrossRef]
- Pandey, S.; Fartyal, D.; Agarwal, A.; Shukla, T.; James, D.; Kaul, T.; Negi, Y.K.; Arora, S.; Reddy, M.K. Abiotic stress tolerance in plants: Myriad roles of ascorbate peroxidase. Front. Plant Sci. 2017, 8, 581. [Google Scholar] [CrossRef]
- García-Caparrós, P.; Hasanuzzaman, M.; Lao, M.T. Oxidative stress and antioxidant defense in plants under salinity. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; Hasanuzzaman, M., Fotopoulos, V., Nahar, K., Fujita, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 291–309. [Google Scholar]
- Couto, N.; Wood, J.; Barber, J. The role of glutathione reductase and related enzymes on cellular redox homoeostasis network. Free Radic. Biol. Med. 2016, 95, 27–42. [Google Scholar] [CrossRef]
- Bela, K.; Horváth, E.; Gallé, Á.; Szabados, L.; Tari, I.; Csiszár, J. Plant glutathione peroxidases: Emerging role of the antioxidant enzymes in plant development and stress responses. J. Plant Physiol. 2015, 176, 192–201. [Google Scholar] [CrossRef]
- Xu, J.; Xing, X.-J.; Tian, Y.-S.; Peng, R.-H.; Xue, Y.; Zhao, W.; Yao, Q.-H. Transgenic Arabidopsis plants expressing tomato glutathione S-transferase showed enhanced resistance to salt and drought stress. PLoS ONE 2015, 10, e0136960. [Google Scholar] [CrossRef]
- Christou, A.; Antoniou, C.; Christodoulou, C.; Hapeshi, E.; Stavrou, I.; Michael, C.; Fatta-Kassinos, D.; Fotopoulos, V. Stress-related phenomena and detoxification mechanisms induced by common pharmaceuticals in alfalfa (Medicago sativa L.) plants. Sci. Total Environ. 2016, 557, 652–664. [Google Scholar] [CrossRef]
- Nianiou-Obeidat, I.; Madesis, P.; Kissoudis, C.; Voulgari, G.; Chronopoulou, E.; Tsaftaris, A.; Labrou, N.E. Plant glutathione transferase-mediated stress tolerance: Functions and biotechnological applications. Plant Cell Rep. 2017, 36, 791–805. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Trivedi, P.K. Glutathione S-transferases: Role in combating abiotic stresses including arsenic detoxification in plants. Front. Plant Sci. 2018, 9, 751. [Google Scholar] [CrossRef]
- Calderón, A.; Sevilla, F.; Jiménez, A. Redox protein thioredoxins: Function under salinity, drought and extreme temperature conditions. In Antioxidants and Antioxidant Enzymes in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 123–162. [Google Scholar]
- Sevilla, F.; Jiménez, A.; Lázaro, J.J. What do the plant mitochondrial antioxidant and redox systems have to say under salinity, drought, and extreme temperature? In Reactive Oxygen Species and Oxidative Damage in Plants under Stress; Gupta, D.K., Palma, J.M., Corpas, F.J., Eds.; Springer: Cham, Switzerland, 2015; pp. 23–55. [Google Scholar]
- Pedrajas, J.R.; Bárcena, J.A. Peroxiredoxins: Types, characteristics and functions in higher plants. In Antioxidants and Antioxidant Enzymes in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 95–121. [Google Scholar]
- Liebthal, M.; Maynard, D.; Dietz, K.-J. Peroxiredoxins and redox signaling in plants. Antioxid. Redox Signal. 2018, 28, 609–624. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Nahar, K.; Anee, T.I.; Fujita, M. Glutathione in plants: Biosynthesis and physiological role in environmental stress tolerance. Physiol. Mol. Biol. Plants 2017, 23, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Cunha, J.R.; Neto, M.C.L.; Carvalho, F.E.; Martins, M.O.; Jardim-Messeder, D.; Margis-Pinheiro, M.; Silveira, J.A. Salinity and osmotic stress trigger different antioxidant responses related to cytosolic ascorbate peroxidase knockdown in rice roots. Environ. Exp. Bot. 2016, 131, 58–67. [Google Scholar] [CrossRef]
- Li, Y.; Cao, X.L.; Zhu, Y.; Yang, X.M.; Zhang, K.N.; Xiao, Z.Y.; Wang, H.; Zhao, J.H.; Zhang, L.L.; Li, G.B. Osa-miR398b boosts H2O2 production and rice blast disease-resistance via multiple superoxide dismutases. New Phytol. 2019, 222, 1507–1522. [Google Scholar] [CrossRef] [PubMed]
- Vighi, I.; Benitez, L.; Amaral, M.; Moraes, G.; Auler, P.; Rodrigues, G.; Deuner, S.; Maia, L.; Braga, E. Functional characterization of the antioxidant enzymes in rice plants exposed to salinity stress. Biol. Plant. 2017, 61, 540–550. [Google Scholar] [CrossRef]
- Zeeshan, M.; Lu, M.; Sehar, S.; Holford, P.; Wu, F. Comparison of biochemical, anatomical, morphological, and physiological responses to salinity stress in wheat and barley genotypes deferring in salinity tolerance. Agronomy 2020, 10, 127. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Akram, N.A.; Ashraf, M. Osmoprotection in plants under abiotic stresses: New insights into a classical phenomenon. Planta 2020, 251, 3. [Google Scholar] [CrossRef]
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Khan, A.; Ahmed, A.-H. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Casadesús, A.; Brockman, H.; Munné-Bosch, S. An overview of plant-based natural biostimulants for sustainable horticulture with a particular focus on moringa leaf extracts. Plant Sci. 2019, 110194. [Google Scholar] [CrossRef] [PubMed]
- Alsahli, A.; Mohamed, A.-K.; Alaraidh, I.; Al-Ghamdi, A.; Al-Watban, A.; El-Zaidy, M.; Alzahrani, S.M. Salicylic acid alleviates salinity stress through the modulation of biochemical attributes and some key antioxidants in wheat seedlings. Pak. J. Bot. 2019, 51, 1551–1559. [Google Scholar] [CrossRef]
- Ali, A.Y.A.; Ibrahim, M.E.H.; Zhou, G.; Nimir, N.E.A.; Jiao, X.; Zhu, G.; Elsiddig, A.M.I.; Suliman, M.S.E.; Elradi, S.B.M.; Yue, W. Exogenous jasmonic acid and humic acid increased salinity tolerance of sorghum. Agron. J. 2020, 112, 871–884. [Google Scholar] [CrossRef]
- Tanou, G.; Ziogas, V.; Belghazi, M.; Christou, A.; Filippou, P.; Job, D.; Fotopoulos, V.; Molassiotis, A. Polyamines reprogram oxidative and nitrosative status and the proteome of citrus plants exposed to salinity stress. Plant Cell Environ. 2014, 37, 864–885. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Qin, C.; Begum, N.; Maodong, Q.; Dong, X.X.; El-Esawi, M.; El-Sheikh, M.A.; Alatar, A.A.; Zhang, L. Nitrogen availability prevents oxidative effects of salinity on wheat growth and photosynthesis by up-regulating the antioxidants and osmolytes metabolism, and secondary metabolite accumulation. BMC Plant Biol. 2019, 19, 1–12. [Google Scholar] [CrossRef]
- Chung, Y.S.; Kim, K.-S.; Hamayun, M.; Kim, Y. Silicon confers soybean resistance to salinity stress through regulation of reactive oxygen and reactive nitrogen species. Front. Plant Sci. 2019, 10, 1725. [Google Scholar] [CrossRef]
- Christou, A.; Manganaris, G.A.; Papadopoulos, I.; Fotopoulos, V. Hydrogen sulfide induces systemic tolerance to salinity and non-ionic osmotic stress in strawberry plants through modification of reactive species biosynthesis and transcriptional regulation of multiple defence pathways. J. Exp. Bot. 2013, 64, 1953–1966. [Google Scholar] [CrossRef]
- Santander, C.; Ruiz, A.; García, S.; Aroca, R.; Cumming, J.; Cornejo, P. Efficiency of two arbuscular mycorrhizal fungal inocula to improve saline stress tolerance in lettuce plants by changes of antioxidant defense mechanisms. J. Sci. Food Agric. 2020, 100, 1577–1587. [Google Scholar] [CrossRef]
- Rady, M.M.; Kuşvuran, A.; Alharby, H.F.; Alzahrani, Y.; Kuşvuran, S. Pretreatment with proline or an organic bio-stimulant induces salt tolerance in wheat plants by improving antioxidant redox state and enzymatic activities and reducing the oxidative stress. J. Plant Growth Regul. 2019, 38, 449–462. [Google Scholar] [CrossRef]
- Rady, M.M.; Elrys, A.S.; El-Maati, M.F.A.; Desoky, E.-S.M. Interplaying roles of silicon and proline effectively improve salt and cadmium stress tolerance in Phaseolus vulgaris plant. Plant Physiol. Biochem. 2019, 139, 558–568. [Google Scholar] [CrossRef]
- Heydari, H.; Rezayian, M.; Niknam, V.; Ebrahimzadeh, H. Role of Penconazole in salt stress amelioration in Sesamum indicum L. Soil Sci. Plant Nutr. 2019, 65, 243–250. [Google Scholar] [CrossRef]
- Akram, N.A.; Iqbal, M.; Muhammad, A.; Ashraf, M.; Al-Qurainy, F.; Shafiq, S. Aminolevulinic acid and nitric oxide regulate oxidative defense and secondary metabolisms in canola (Brassica napus L.) under drought stress. Protoplasma 2018, 255, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Tian, S.; Liu, S.; Wang, W.; Sui, N. Energy dissipation and antioxidant enzyme system protect photosystem II of sweet sorghum under drought stress. Photosynthetica 2018, 56, 861–872. [Google Scholar] [CrossRef]
- Hassan, N.; Ebeed, H.; Aljaarany, A. Exogenous application of spermine and putrescine mitigate adversities of drought stress in wheat by protecting membranes and chloroplast ultra-structure. Physiol. Mol. Biol. Plants 2020, 26, 233–245. [Google Scholar] [CrossRef]
- Antoniou, C.; Xenofontos, R.; Chatzimichail, G.; Christou, A.; Kashfi, K.; Fotopoulos, V. Exploring the potential of nitric oxide and hydrogen sulfide (NOSH)-releasing synthetic compounds as novel priming agents against drought stress in Medicago sativa plants. Biomolecules 2020, 10, 120. [Google Scholar] [CrossRef]
- Gratão, P.L.; Alves, L.R.; Lima, L.W. Heavy metal toxicity and plant productivity: Role of metal scavengers. In Plant-Metal Interactions; Srivastava, S., Srivastava, A.K., Suprasanna, P., Eds.; Springer: Cham, Switzerland, 2019; pp. 49–60. [Google Scholar]
- Chakravarthi, S.; Jessop, C.E.; Bulleid, N.J. The role of glutathione in disulphide bond formation and endoplasmic-reticulum-generated oxidative stress. EMBO Rep. 2006, 7, 271–275. [Google Scholar] [CrossRef]
- Petrussa, E.; Braidot, E.; Zancani, M.; Peresson, C.; Bertolini, A.; Patui, S.; Vianello, A. Plant flavonoids—biosynthesis, transport and involvement in stress responses. Int. J. Mol. Sci. 2013, 14, 14950–14973. [Google Scholar] [CrossRef]
- Landi, M.; Tattini, M.; Gould, K.S. Multiple functional roles of anthocyanins in plant-environment interactions. Environ. Exp. Bot. 2015, 119, 4–17. [Google Scholar] [CrossRef]
- Awasthi, R.; Gaur, P.; Turner, N.C.; Vadez, V.; Siddique, K.H.; Nayyar, H. Effects of individual and combined heat and drought stress during seed filling on the oxidative metabolism and yield of chickpea (Cicer arietinum) genotypes differing in heat and drought tolerance. Crop Past. Sci. 2017, 68, 823–841. [Google Scholar] [CrossRef]
- Sarkar, J.; Chakraborty, B.; Chakraborty, U. Temperature stress induced antioxidative and biochemical changes in wheat (Triticum aestivum L.) cultivars. J. Plant Stress Physiol. 2016, 22–30. [Google Scholar] [CrossRef]
- Zandalinas, S.I.; Balfagón, D.; Arbona, V.; Gómez-Cadenas, A. Modulation of antioxidant defense system is associated with combined drought and heat stress tolerance in citrus. Front. Plant Sci. 2017, 8, 953. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Ye, L.; Wang, Y.; Zhou, X.; Yang, J.; Wang, J.; Cao, K.; Zou, Z. Melatonin increases the chilling tolerance of chloroplast in cucumber seedlings by regulating photosynthetic electron flux and the ascorbate-glutathione cycle. Front. Plant Sci. 2016, 7, 1814. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Ye, T.; Zhong, B.; Liu, X.; Chan, Z. Comparative proteomic and metabolomic analyses reveal mechanisms of improved cold stress tolerance in bermudagrass (Cynodon dactylon (L.) Pers.) by exogenous calcium. J. Integr. Plant Biol. 2014, 56, 1064–1079. [Google Scholar] [CrossRef] [PubMed]
- Wani, M.A.; Jan, N.; Qazi, H.A.; Andrabi, K.I.; John, R. Cold stress induces biochemical changes, fatty acid profile, antioxidant system and gene expression in Capsella bursa pastoris L. Acta Physiol. Plant. 2018, 40, 167. [Google Scholar] [CrossRef]
- Mohammadrezakhani, S.; Hajilou, J.; Rezanejad, F.; Zaare-Nahandi, F. Assessment of exogenous application of proline on antioxidant compounds in three citrus species under low temperature stress. J. Plant Interact. 2019, 14, 347–358. [Google Scholar] [CrossRef]
- Spanò, C.; Bottega, S.; Castiglione, M.R.; Pedranzani, H.E. Antioxidant response to cold stress in two oil plants of the genus Jatropha. Plant Soil Environ. 2017, 63, 271–276. [Google Scholar]
- Cheng, F.; Lu, J.; Gao, M.; Shi, K.; Kong, Q.; Huang, Y.; Bie, Z. Redox signaling and CBF-responsive pathway are involved in salicylic acid-improved photosynthesis and growth under chilling stress in watermelon. Front. Plant Sci. 2016, 7, 1519. [Google Scholar] [CrossRef]
- Wang, Q.J.; Sun, H.; Dong, Q.L.; Sun, T.Y.; Jin, Z.X.; Hao, Y.J.; Yao, Y.X. The enhancement of tolerance to salt and cold stresses by modifying the redox state and salicylic acid content via the cytosolic malate dehydrogenase gene in transgenic apple plants. Plant Biotechnol. J. 2016, 14, 1986–1997. [Google Scholar] [CrossRef]
- Li, W.; Mo, W.; Ashraf, U.; Li, G.; Wen, T.; Abrar, M.; Gao, L.; Liu, J.; Hu, J. Evaluation of physiological indices of waterlogging tolerance of different maize varieties in South China. Appl. Ecol. Environ. Res. 2018, 16, 2059–2072. [Google Scholar] [CrossRef]
- Qin, C.; Ahanger, M.A.; Zhou, J.; Ahmed, N.; Wei, C.; Yuan, S.; Ashraf, M.; Zhang, L. Beneficial role of acetylcholine in chlorophyll metabolism and photosynthetic gas exchange in Nicotiana benthamiana seedlings under salinity stress. Plant Biol. 2020, 22, 357–365. [Google Scholar] [CrossRef]
- Parvin, K.; Nahar, K.; Hasanuzzaman, M.; Bhuyan, M.B.; Mohsin, S.M.; Fujita, M. Exogenous vanillic acid enhances salt tolerance of tomato: Insight into plant antioxidant defense and glyoxalase systems. Plant Physiol. Biochem. 2020, 150, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Cen, H.; Wang, T.; Liu, H.; Tian, D.; Zhang, Y. Melatonin application improves salt tolerance of alfalfa (Medicago sativa L.) by enhancing antioxidant capacity. Plants 2020, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Shi, Z.; Zhang, X.; Zheng, S.; Wang, J.; Mo, J. Alleviating effects of exogenous melatonin on salt stress in cucumber. Sci. Hortic. 2020, 262, 109070. [Google Scholar] [CrossRef]
- Sehar, Z.; Masood, A.; Khan, N.A. Nitric oxide reverses glucose-mediated photosynthetic repression in wheat (Triticum aestivum L.) under salt stress. Environ. Exp. Bot. 2019, 161, 277–289. [Google Scholar] [CrossRef]
- Jiang, J.-L.; Tian, Y.; Li, L.; Yu, M.; Hou, R.-P.; Ren, X.-M. H2S alleviates salinity stress in cucumber by maintaining the Na+/K+ balance and regulating H2S metabolism and oxidative stress response. Front. Plant Sci. 2019, 10, 678. [Google Scholar] [CrossRef] [PubMed]
- Jahan, B.; AlAjmi, M.F.; Rehman, M.T.; Khan, N. Treatment of nitric oxide supplemented with nitrogen and sulfur regulates photosynthetic performance and stomatal behavior in mustard under salt stress. Physiol. Plant. 2020, 168, 490–510. [Google Scholar]
- Hamim, H.; Violita, V.; Triadiati, T.; Miftahudin, M. Oxidative stress and photosynthesis reduction of cultivated (Glycine max L.) and wild soybean (G. tomentella L.) exposed to drought and paraquat. Asian J. Plant Sci. 2017, 16, 65–77. [Google Scholar] [CrossRef][Green Version]
- Jan, N.; Majeed, U.; Andrabi, K.I.; John, R. Cold stress modulates osmolytes and antioxidant system in Calendula officinalis. Acta Physiol. Plant. 2018, 40, 73. [Google Scholar] [CrossRef]
- Chen, Z.-Y.; Wang, Y.-T.; Pan, X.-B.; Xi, Z.-M. Amelioration of cold-induced oxidative stress by exogenous 24-epibrassinolide treatment in grapevine seedlings: Toward regulating the ascorbate–glutathione cycle. Sci. Hortic. 2019, 244, 379–387. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant. Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Neill, S.; Desikan, R.; Hancock, J. Hydrogen peroxide signalling. Curr. Opin. Plant Biol. 2002, 5, 388–395. [Google Scholar] [CrossRef]
- Neill, S.J.; Desikan, R.; Clarke, A.; Hurst, R.D.; Hancock, J.T. Hydrogen peroxide and nitric oxide as signalling molecules in plants. J. Exp. Bot. 2002, 53, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- Asada, K. Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 2006, 141, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Kar, R.K. Plant responses to water stress: Role of reactive oxygen species. Plant Signal. Behav. 2011, 6, 1741–1745. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Liao, W. Hydrogen peroxide signaling in plant development and abiotic responses: Crosstalk with nitric oxide and calcium. Front. Plant Sci. 2016, 7, 230. [Google Scholar] [CrossRef]
- Janicka, M.; Reda, M.; Napieraj, N.; Kabała, K. Plant abiotic stress: Function of nitric oxide and hydrogen peroxide. In Nitric Oxide and Hydrogen Peroxide Signaling in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Swizerland, 2019; pp. 201–219. [Google Scholar]
- Savvides, A.; Ali, S.; Tester, M.; Fotopoulos, V. Chemical priming of plants against multiple abiotic stresses: Mission possible? Trends Plant Sci. 2016, 21, 329–340. [Google Scholar] [CrossRef]
- Kaur, P.; Handa, N.; Verma, V.; Bakshi, P.; Kalia, R.; Sareen, S.; Nagpal, A.; Vig, A.; Mir, B.A.; Bhardwaj, R. Cross talk among reactive oxygen, nitrogen and sulfur during abiotic stress in plants. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; Hasanuzzaman, M., Fotopoulos, V., Nahar, K., Fujita, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 857–871. [Google Scholar]
- Hancock, J.T.; Whiteman, M. Hydrogen sulfide signaling: Interactions with nitric oxide and reactive oxygen species. Ann. N. Y. Acad. Sci. 2016, 1365, 5–14. [Google Scholar] [CrossRef]
- Biswas, M.S.; Fukaki, H.; Mori, I.C.; Nakahara, K.; Mano, J.I. Reactive oxygen species and reactive carbonyl species constitute a feed-forward loop in auxin signaling for lateral root formation. Plant J. 2019, 100, 536–548. [Google Scholar] [CrossRef]
- Li, J.-T.; Qiu, Z.-B.; Zhang, X.-W.; Wang, L.-S. Exogenous hydrogen peroxide can enhance tolerance of wheat seedlings to salt stress. Acta Physiol. Plant. 2011, 33, 835–842. [Google Scholar] [CrossRef]
- Liu, Z.-J.; Guo, Y.-K.; Bai, J.-G. Exogenous hydrogen peroxide changes antioxidant enzyme activity and protects ultrastructure in leaves of two cucumber ecotypes under osmotic stress. J. Plant Growth Regul. 2010, 29, 171–183. [Google Scholar] [CrossRef]
- Zhang, X.-L.; Jia, X.-F.; Yu, B.; Gao, Y.; Bai, J.-G. Exogenous hydrogen peroxide influences antioxidant enzyme activity and lipid peroxidation in cucumber leaves at low light. Sci. Hortic. 2011, 129, 656–662. [Google Scholar] [CrossRef]
- Nawaz, F.; Majeed, S.; Ahmad, K.S.; Aqib, M.; Shehzad, M.A.; Aurangzaib, M.; Shahbaz, M. Reactive sulfur species-key regulators of abiotic stress tolerance in plants. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; Hasanuzzaman, M., Fotopoulos, V., Nahar, K., Fujita, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 685–713. [Google Scholar]
- Hasanuzzaman, M.; Oku, H.; Nahar, K.; Bhuyan, M.B.; Al Mahmud, J.; Baluska, F.; Fujita, M. Nitric oxide-induced salt stress tolerance in plants: ROS metabolism, signaling, and molecular interactions. Plant Biotechnol. Rep. 2018, 12, 77–92. [Google Scholar] [CrossRef]
- Bhuyan, M.B.; Hasanuzzaman, M.; Parvin, K.; Mohsin, S.M.; Al Mahmud, J.; Nahar, K.; Fujita, M. Nitric oxide and hydrogen sulfide: Two intimate collaborators regulating plant defense against abiotic stress. Plant Growth Regul. 2020, 1–16. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.M.; Bhuyan, M.B.; Oku, H.; Fujita, M. Exogenous nitric oxide pretreatment protects Brassica napus L. seedlings from paraquat toxicity through the modulation of antioxidant defense and glyoxalase systems. Plant Physiol. Biochem. 2018, 126, 173–186. [Google Scholar] [CrossRef]
- Bright, J.; Desikan, R.; Hancock, J.T.; Weir, I.S.; Neill, S.J. ABA-induced NO generation and stomatal closure in Arabidopsis are dependent on H2O2 synthesis. Plant J. 2006, 45, 113–122. [Google Scholar] [CrossRef]
- González, A.; de Los Ángeles Cabrera, M.; Henríquez, M.J.; Contreras, R.A.; Morales, B.; Moenne, A. Cross talk among calcium, hydrogen peroxide, and nitric oxide and activation of gene expression involving calmodulins and calcium-dependent protein kinases in Ulva compressa exposed to copper excess. Plant Physiol. 2012, 158, 1451–1462. [Google Scholar] [CrossRef]
- Zhang, A.; Jiang, M.; Zhang, J.; Ding, H.; Xu, S.; Hu, X.; Tan, M. Nitric oxide induced by hydrogen peroxide mediates abscisic acid-induced activation of the mitogen-activated protein kinase cascade involved in antioxidant defense in maize leaves. New Phytol. 2007, 175, 36–50. [Google Scholar] [CrossRef]
- Qiao, W.; Li, C.; Fan, L.-M. Cross-talk between nitric oxide and hydrogen peroxide in plant responses to abiotic stresses. Environ. Exp. Bot. 2014, 100, 84–93. [Google Scholar] [CrossRef]
- Palma, J.M.; Gupta, D.K.; Corpas, F.J. Hydrogen peroxide and nitric oxide generation in plant cells: Overview and queries. In Nitric Oxide and Hydrogen Peroxide Signaling in Higher Plants; Springer: Cham, Swizerland, 2019; pp. 1–16. [Google Scholar]
- Corpas, F.J.; Gupta, D.K.; Palma, J.M. Production sites of reactive oxygen species (ROS) in organelles from plant cells. In Reactive Oxygen Species and Oxidative Damage in Plants under Stress; Gupta, D.K., Palma, J.M., Corpas, F.J., Eds.; Springer: Cham, Swizerland, 2015; pp. 1–22. [Google Scholar]
- Corpas, F.J.; Barroso, J.B.; Palma, J.M.; Rodriguez-Ruiz, M. Plant peroxisomes: A nitro-oxidative cocktail. Redox Biol. 2017, 11, 535–542. [Google Scholar] [CrossRef]
- Radi, R. Nitric oxide, oxidants, and protein tyrosine nitration. In Proceedings of the National Academy of Sciences of the United States of America, Los Angeles, CA, USA, 12 January 2004; pp. 4003–4008. [Google Scholar]
- Liu, D.; Liu, M.; Liu, X.-L.; Cheng, X.-G.; Liang, Z.-W. Silicon priming created an enhanced tolerance in alfalfa (Medicago sativa L.) seedlings in response to high alkaline stress. Front. Plant Sci. 2018, 9, 716. [Google Scholar] [CrossRef]
- Ortega-Galisteo, A.P.; Rodríguez-Serrano, M.; Pazmiño, D.M.; Gupta, D.K.; Sandalio, L.M.; Romero-Puertas, M. S-Nitrosylated proteins in pea (Pisum sativum L.) leaf peroxisomes: Changes under abiotic stress. J. Exp. Bot. 2012, 63, 2089–2103. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.C. Reactive sulfur species: A new player in plant physiology? In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; Hasanuzzaman, M., Fotopoulos, V., Nahar, K., Fujita, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 715–728. [Google Scholar]
- Hill, B.G.; Bhatnagar, A. Protein S-glutathiolation: Redox-sensitive regulation of protein function. J. Mol. Cell. Cardiol. 2012, 52, 559–567. [Google Scholar] [CrossRef]
- Tao, L.; English, A.M. Protein S-glutathiolation triggered by decomposed S-nitrosoglutathione. Biochemistry 2004, 43, 4028–4038. [Google Scholar] [CrossRef] [PubMed]
- Pajares, M.; Jiménez-Moreno, N.; Dias, I.H.; Debelec, B.; Vucetic, M.; Fladmark, K.E.; Basaga, H.; Ribaric, S.; Milisav, I.; Cuadrado, A. Redox control of protein degradation. Redox Biol. 2015, 6, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Jia, H.; Wang, J.; Cao, Q.; Wen, Z. Hydrogen sulfide is involved in maintaining ion homeostasis via regulating plasma membrane Na+/H+ antiporter system in the hydrogen peroxide-dependent manner in salt-stress Arabidopsis thaliana root. Protoplasma 2014, 251, 899–912. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Rahman, A.; Suzuki, T.; Fujita, M. Polyamine and nitric oxide crosstalk: Antagonistic effects on cadmium toxicity in mung bean plants through upregulating the metal detoxification, antioxidant defense and methylglyoxal detoxification systems. Ecotoxicol. Environ. Saf. 2016, 126, 245–255. [Google Scholar] [CrossRef]
- Gupta, V.; Carroll, K.S. Sulfenic acid chemistry, detection and cellular lifetime. Biochim. Biophys. Acta 2014, 1840, 847–875. [Google Scholar] [CrossRef]
- Ono, K.; Akaike, T.; Sawa, T.; Kumagai, Y.; Wink, D.A.; Tantillo, D.J.; Hobbs, A.J.; Nagy, P.; Xian, M.; Lin, J. Redox chemistry and chemical biology of H2S, hydropersulfides, and derived species: Implications of their possible biological activity and utility. Free Radic. Biol. Med. 2014, 77, 82–94. [Google Scholar] [CrossRef]
- Akaike, T.; Ida, T.; Wei, F.-Y.; Nishida, M.; Kumagai, Y.; Alam, M.M.; Ihara, H.; Sawa, T.; Matsunaga, T.; Kasamatsu, S. Cysteinyl-tRNA synthetase governs cysteine polysulfidation and mitochondrial bioenergetics. Nat. Commun. 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Mano, J.I.; Biswas, M.; Sugimoto, K. Reactive carbonyl species: A missing link in ROS signaling. Plants 2019, 8, 391. [Google Scholar] [CrossRef]
- Yalcinkaya, T.; Uzilday, B.; Ozgur, R.; Turkan, I. The roles of reactive carbonyl species in induction of antioxidant defence and ROS signalling in extreme halophytic model Eutrema parvulum and glycophytic model Arabidopsis thaliana. Environ. Exp. Bot. 2019, 160, 81–91. [Google Scholar] [CrossRef]
- Mano, J.I.; Kanameda, S.; Kuramitsu, R.; Matsuura, N.; Yamauchi, Y. Detoxification of reactive carbonyl species by glutathione transferase Tau isozymes. Front. Plant Sci. 2019, 10, 487. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.M.; Ye, W.; Matsushima, D.; Rhaman, M.S.; Munemasa, S.; Okuma, E.; Nakamura, Y.; Biswas, M.S.; Mano, J.I.; Murata, Y. Reactive carbonyl species function as signal mediators downstream of H2O2 production and regulate [Ca2+] cyt elevation in ABA signal pathway in Arabidopsis guard cells. Plant Cell Physiol. 2019, 60, 1146–1159. [Google Scholar] [CrossRef] [PubMed]
- Kaur, C.; Singla-Pareek, S.L.; Sopory, S.K. Glyoxalase and methylglyoxal as biomarkers for plant stress tolerance. Crit. Rev. Plant Sci. 2014, 33, 429–456. [Google Scholar] [CrossRef]
- Saito, R.; Yamamoto, H.; Makino, A.; Sugimoto, T.; Miyake, C. Methylglyoxal functions as Hill oxidant and stimulates the photoreduction of O2 at photosystem I: A symptom of plant diabetes. Plant Cell Environ. 2011, 34, 1454–1464. [Google Scholar] [CrossRef]
- Hao, Q.; Maret, W. Aldehydes release zinc from proteins. A pathway from oxidative stress/lipid peroxidation to cellular functions of zinc. FEBS J. 2006, 273, 4300–4310. [Google Scholar] [CrossRef]
- Kiranmai, K.; Lokanadha Rao, G.; Pandurangaiah, M.; Nareshkumar, A.; Amaranatha Reddy, V.; Lokesh, U.; Venkatesh, B.; Anthony Johnson, A.; Sudhakar, C. A novel WRKY transcription factor, MuWRKY3 (Macrotyloma uniflorum Lam. Verdc.) enhances drought stress tolerance in transgenic groundnut (Arachis hypogaea L.) plants. Front. Plant Sci. 2018, 9, 346. [Google Scholar] [CrossRef]
- Sun, X.; Wang, P.; Jia, X.; Huo, L.; Che, R.; Ma, F. Improvement of drought tolerance by overexpressing MdATG18a is mediated by modified antioxidant system and activated autophagy in transgenic apple. Plant Biotechnol. J. 2018, 16, 545–557. [Google Scholar] [CrossRef]
- Wang, K.; Zhong, M.; Wu, Y.-h.; Bai, Z.-y.; Liang, Q.-y.; Liu, Q.-l.; Pan, Y.-z.; Zhang, L.; Jiang, B.-b.; Jia, Y. Overexpression of a chrysanthemum transcription factor gene DgNAC1 improves the salinity tolerance in chrysanthemum. Plant Cell Rep. 2017, 36, 571–581. [Google Scholar] [CrossRef]
- Shafi, A.; Pal, A.K.; Sharma, V.; Kalia, S.; Kumar, S.; Ahuja, P.S.; Singh, A.K. Transgenic potato plants overexpressing SOD and APX exhibit enhanced lignification and starch biosynthesis with improved salt stress tolerance. Plant Mol. Biol. Rep. 2017, 35, 504–518. [Google Scholar] [CrossRef]
- Sapara, K.K.; Khedia, J.; Agarwal, P.; Gangapur, D.R.; Agarwal, P.K. SbMYB15 transcription factor mitigates cadmium and nickel stress in transgenic tobacco by limiting uptake and modulating antioxidative defence system. Funct. Plant Biol. 2019, 46, 702–714. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dubey, A.K.; Kumar, V.; Ansari, M.A.; Narayan, S.; Kumar, S.; Pandey, V.; Shirke, P.A.; Pande, V.; Sanyal, I. Over-expression of chickpea glutaredoxin (CaGrx) provides tolerance to heavy metals by reducing metal accumulation and improved physiological and antioxidant defence system. Ecotoxicol. Environ. Saf. 2020, 192, 110252. [Google Scholar] [CrossRef] [PubMed]
- Karkute, S.; Krishna, R.; Ansari, W.; Singh, B.; Singh, P.; Singh, M.; Singh, A. Heterologous expression of the AtDREB1A gene in tomato confers tolerance to chilling stress. Biol. Plant. 2019, 63, 268–277. [Google Scholar] [CrossRef]
- Che, Y.; Zhang, N.; Zhu, X.; Li, S.; Wang, S.; Si, H. Enhanced tolerance of the transgenic potato plants overexpressing Cu/Zn superoxide dismutase to low temperature. Sci. Hortic. 2020, 261, 108949. [Google Scholar] [CrossRef]
- Wang, L.; Gao, J.; Zhang, Z.; Liu, W.; Cheng, P.; Mu, W.; Su, T.; Chen, S.; Chen, F.; Jiang, J. Overexpression of CmSOS1 confers waterlogging tolerance in Chrysanthemum. J. Integr. Plant Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Hou, L.; Xiao, P.; Guo, Y.; Deyholos, M.K.; Liu, X. VvWRKY30, a grape WRKY transcription factor, plays a positive regulatory role under salinity stress. Plant Sci. 2019, 280, 132–142. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, N.; Yang, J.; Guo, H.; Liu, Z.; Zheng, X.; Li, S.; Xiang, F. The salt-induced transcription factor GmMYB84 confers salinity tolerance in soybean. Plant Sci. 2020, 291, 110326. [Google Scholar] [CrossRef]
- Duan, F.; Ding, J.; Lee, D.; Lu, X.; Feng, Y.; Song, W. Overexpression of SoCYP85A1, a spinach cytochrome p450 gene in transgenic tobacco enhances root development and drought stress tolerance. Front. Plant Sci. 2017, 8, 1909. [Google Scholar] [CrossRef]
- Xia, Z.; Xu, Z.; Wei, Y.; Wang, M. Overexpression of the maize sulfite oxidase increases sulfate and GSH levels and enhances drought tolerance in transgenic tobacco. Front. Plant Sci. 2018, 9, 298. [Google Scholar] [CrossRef]
- Dubey, A.K.; Kumar, N.; Kumar, A.; Ansari, M.A.; Ranjan, R.; Gautam, A.; Sahu, N.; Pandey, V.; Behera, S.K.; Mallick, S. Over-expression of CarMT gene modulates the physiological performance and antioxidant defense system to provide tolerance against drought stress in Arabidopsis thaliana L. Ecotoxicol. Environ. Saf. 2019, 171, 54–65. [Google Scholar] [CrossRef]
- Tan, Y.; Wang, L. MpDGK2, a novel diacylglycerol kinase from Malus prunifolia, confers drought stress tolerance in transgenic Arabidopsis. Plant Mol. Biol. Rep. 2020, 1–9. [Google Scholar] [CrossRef]
- Kumar, S.; Khare, R.; Trivedi, P.K. Arsenic-responsive high-affinity rice sulphate transporter, OsSultr1;1, provides abiotic stress tolerance under limiting sulphur condition. J. Hazard. Mater. 2019, 373, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, W.; Wang, W.; Zhang, G.; Liu, Y.; Wang, Y.; Wang, W. Wheat F-box protein gene TaFBA1 is involved in plant tolerance to heat stress. Front. Plant Sci. 2018, 9, 521. [Google Scholar] [CrossRef]
- Huo, L.; Sun, X.; Guo, Z.; Jia, X.; Che, R.; Sun, Y.; Zhu, Y.; Wang, P.; Gong, X.; Ma, F. MdATG18a overexpression improves basal thermotolerance in transgenic apple by decreasing damage to chloroplasts. Hortic. Res. 2020, 7, 1–15. [Google Scholar] [CrossRef]
- Lv, Y.; Fu, S.; Chen, S.; Zhang, W.; Qi, C. Ethylene response factor BnERF2-like (ERF2. 4) from Brassica napus L. enhances submergence tolerance and alleviates oxidative damage caused by submergence in Arabidopsis thaliana. Crop J. 2016, 4, 199–211. [Google Scholar] [CrossRef]
- Phukan, U.J.; Jeena, G.S.; Tripathi, V.; Shukla, R.K. MaRAP2-4, a waterlogging-responsive ERF from Mentha, regulates bidirectional sugar transporter AtSWEET10 to modulate stress response in Arabidopsis. Plant Biotechnol. J. 2018, 16, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Luan, H.; Guo, B.; Shen, H.; Pan, Y.; Hong, Y.; Lv, C.; Xu, R. Overexpression of barley transcription factor HvERF2.11 in Arabidopsisenhances plant waterlogging tolerance. Int. J. Mol. Sci. 2020, 21, 1982. [Google Scholar] [CrossRef] [PubMed]

| Plant Species/Genotypes | Stress Condition | Oxidative Stress Status | References |
|---|---|---|---|
| Salinity | |||
| Triticum aestivum | 150 mM NaCl; 20 d applied on alternate days | 62.11% and 63.78% increase in H2O2 and O2•−, respectively. 44% increase in lipid peroxidation. | [74] |
| Vicia faba cv. ILB-4347 and Hassawi-3 | 150 mM NaCl | 90%, 66% and 84% increase in H2O2, MDA, and EL, respectively in ILB-4347. 128%, 92%, and 96% increase in H2O2, MDA, and EL, respectively, in Hassawi-3. | [124] |
| Oryza sativa | 150 mM NaCl; 72 h | 2-fold higher ROS level in root tissues. Sharp increases in lipid peroxidation, EL, H2O2, and O2•− in leaf tissue. | [71] |
| Solanum lycopersicum | 100 mM NaCl | 157%, 176%, 158%, and 94% increased O2•−, H2O2, EL and MDA content. | [72] |
| Vigna radiate | 100 mM NaCl | A 2-fold increase in the levels of H2O2, MDA, EL, and O2•−. | [74] |
| Ocimum basilicum | 50 mM NaCl; sprinkling in 2 d intervals till harvest | Significant increase in H2O2 and MDA contents. | [21] |
| Lens culinaris | 100 mM NaCl; 3 d | Higher accumulation of MDA and H2O2 content by 139% and 37%, respectively. | [125] |
| Sorghum bicolor | 100 mM NaCl, 3 d intervals for 16 d | H2O2 content increased by 149% in leaf while 38% in roots. MDA content increased by 106% in leaf while 116% for roots. | [126] |
| Chenopodium quinoa cv. Q1, Q2, Q3, and Q4 | 100, 300, and 500 mM NaCl; 14 d | MDA and H2O2 increased in a concentration-dependent manner in all cultivars. Under 100 mM NaCl, the salt-sensitive cultivars (Q1, Q2, and Q3) showed the highest accumulation of H2O2 and MDA. Under 300 and 500 mM NaCl, cv. Q4 exhibited the least increase in MDA. | [127] |
| Panicum italicum | 1% NaCl | 1.5-fold increase in H2O2 and a 3-fold increase in MDA levels. | [128] |
| Water Deficit and Simulated Drought | |||
| Zea mays cv. Run Nong 35, Wan Dan 13 and Dong Dan 0 | Drought (80%, 60%, and 40% FC) | Increased ROS accumulation and membrane damage. Enhanced level of O2•−, H2O2, EL, LOX, and TBARS were found in all cultivars. | [129] |
| Medicago sativa | Water deficit, 7 d | Dramatically increased (by 5-fold) H2O2 content. Increased NO content (by 15%) compared with control. | [130] |
| Brassica napus Binasarisha-3 | Osmotic stress (10% and 20% PEG), 48 h | Both levels of H2O2 and MDA were upregulated significantly, with the highest value in 20% PEG. | [89] |
| B. napus cv. Bulbul-98 | Water deficit (30% FC) | Increased EL by 2-folds with membrane damage. Significant increase in H2O2 content. | [131] |
| S. lycopersicum mill. cv. Pusa 120 | Drought (withheld irrigation), 6 d | Increased lipid peroxidation (MDA content) and EL (39%). | [88] |
| V. radiata cv. BARI Mung-2 | Osmotic stress (5% PEG), 48 h | 74% and 84% increase in H2O2 and O2•− compared to control. 62% increase in LOX activity. | [91] |
| S. bicolor cv. Sugargraze | Water deficit, 16 d | 113% increase in H2O2 content. Increased MDA content by 94% and 98% in leaf and root, respectively. A drastic loss in cell viability. | [126] |
| T. aestivum | Drought (35% FC) | 31%, 25%, and 38% increase in TBARS, EL, and H2O2 contents, respectively, compared to control. | [92] |
| B. napus cv. BINA Sharisha-3 | Osmotic stress (10% and 20% PEG), 48 h | 123% and 93% increased MDA and H2O2 content over control. | [90] |
| O. sativa, sub1A quantitative trait loci (sub1A QTL) | Drought (withdrawing irrigation), 8 d | Increased O2•−, H2O2 and MDA content by 1.8-, 2.1-, and 1.66-folds, respectively. | [86] |
| Eleusine coracana L. Gaertn. | Drought (75% water deficit condition), 3 w | Increased EL and H2O2 content. | [87] |
| B. rapa cv. BARI Sharisha-15 | Osmotic stress (20% PEG), 2 d | 82% and 131% increased MDA and H2O2 content over control. Overproduction of toxic O2•−. | [132] |
| Coffea arabica L. | Drought (40% water holding capacity), 20 d | Increased MDA content. | [85] |
| O. sativa var. japonica. cv. Nipponbare | Osmotic stress (20% PEG-6000), 5 d | 16% and 23% increased MDA and O2•− accumulation comparing control. 1-fold enhanced H2O2 generation over control. | [93] |
| Phragmites karka | Drought (40% water holding capacity), 35 d | Increased MDA content by 22%. | [84] |
| S. lycopersicum cv. Login 935 | Water deficit (60% FC), 20 d | 83%, 37%, and 75% increased MDA, H2O2, and O2•− content compared to control. | [95] |
| Glycine max | Osmotic stress (15% PEG), 3 w | 47% declined EL, while LOX activity enhanced by 38%. | [94] |
| Toxic Metals/Metalloids | |||
| O. sativa cv. BRRI dhan54 | 0.25 and 0.5 mM NiSO4.7H2O, 3 d | Increased contents of MDA increased (by 172% and 199%). H2O2 (by 28% and 35%) and LOX activity (by 38% and 73%) under 0.25 mM and 0.5 mM Ni-stress, respectively. | [98] |
| B. juncea cv. BARI Sharisha-11 | 0.5 and 1.0 mM CdCl2, 3 d | Enhanced MDA content by 35% and 66%, H2O2 content by 43% and 54%, and LOX activity by 69% and 108% under 0.5 and 1.0 mM Cd stress, respectively. | [102] |
| B. napus BINA Sharisha-3 | 0.5 and 1.0 mM CdCl2, 2 d | Increased MDA contents by 56% and 133%, and H2O2 contents by 38% and 70% in 0.5 and 1.0 mM Cd stress, respectively. | [101] |
| V. radiata cv. BARI Mung-2 | 1.0 and 1.5 mM CdCl2, 2 d | Increased MDA level by 85% and 177%, H2O2 content by 73% and 127% and O2•– generation rate by 69% and 120% under 1.0 and 1.5 mM Cd stresses, respectively. | [100] |
| T. aestivum cv. Pradip | 0.5 and 1.0 mM Pb(NO3)2, 2 d | MDA content increased by 58% and 179% and H2O2 levels by 41% and 95% under both levels of Pb stress. | [107] |
| V. radiata cv. BARI mung-2 | 0.5 mM AlCl3, 2 and 3 d | H2O2, O2•− generation rate, and LOX activity increased by 83%, 110%, and 72%, which increased the lipid peroxidation by 97%. | [133] |
| B. juncea cv. BARI Sharisha-11 | 0.15 and 0.3 mM K2CrO4, 5 d | Increased TBARS content (by 30% and 65%), H2O2 (by 24% and 46%), and LOX activity (by 68% and 101%) under both levels of Cr stress, respectively. | [104] |
| Extreme Temperature | |||
| Cucumis sativus | 35 ± 1 °C, 7 days | Increased MDA content (by 60.6%) and O2− (by 79.9%). | [113] |
| S. bicolor | 36/26 °C and 39/29 °C, until 7 d after full anthesis | O2•− content increased 2 to 4-fold in pollen and 1 to 2.3-folds in the pistil. | [114] |
| Gossypium hirsutum | 45/30 ± 2 °C, 120 d | Enhanced MDA content by 0.78%, affecting fiber quality and boll weight. | [134] |
| O. sativa | 38 °C, 5 d | H2O2 accumulation increased 1-fold. | [93] |
| O. sativa cv. DM You 6188) | 12 °C, for 6 d | Enhanced MDA content and EL by 180% and 49%, respectively. | [116] |
| L. esculentum | 4 °C, 24 h | Enhanced H2O2 content by 32%. | [135] |
| L. esculentum. cv. C.H Falat | 3 °C, 6 h, 6 d | Enhanced H2O2 content (by 2-fold) and EL (by 20%). | [136] |
| Solanum lycopersicum L. | 15/8 °C day/night, 24 and 48 h | Increased MDA and H2O2 by 62% and 34%, respectively. | [117] |
| Waterlogging | |||
| S. bicolor cvs. JN01 and JZ31 | Waterlogged soil, 12 d | 2.45-fold higher MDA content in WL-sensitive JZ31, but 1.8-fold higher in WL-tolerant JN01. | [120] |
| Hordeum vulgare cvs. TF57 and TF58 | Waterlogged soil, 21 d | MDA content and O2•– generation rate σ were markedly increased in WL-sensitive TF57, but slightly increased in WL-tolerant TF58 genotype. | [137] |
| S. lycopersicum cv. Roma | Waterlogged soil, 15 d | 54% and 208% higher MDA and H2O2 contents, respectively. | [122] |
| Deschampsia Antarctica | Waterlogged soil, 7 d | 84% and 52% higher MDA and H2O2 contents. | [123] |
| Sesamum indicum cv. BARI Til-4 | Waterlogged soil, 2, 4, 6, and 8 d | Both MDA and H2O2 increased in a duration-dependent manner 39% and 62% higher MDA and H2O2 content after 8 days of WL. | [121] |
| Antioxidants | Reactions Catalyzed | Catalytic Reaction Sites |
|---|---|---|
| Nonenzymatic | ||
| Ascorbic acid | Scavenges O2•–, H2O2, •OH, and 1O2 | Chloroplast, peroxisomes, cytosol, mitochondria, apoplast |
| Glutathione | Scavenges H2O2, •OH, and 1O2 | Chloroplast, peroxisomes, cytosol, mitochondria, apoplast |
| Tocopherol | Scavenges •OH, 1O2, ROO• and ROOH | Thylakoid membrane of chloroplast |
| Carotenoids | Scavenges mainly 1O2 | Chloroplast |
| Flavonoids | Scavenges O2•–, H2O2, and 1O2 | Chloroplast, vacuole |
| Phenolic acids | Scavenges O2•−, •OH, ROO•, and ONOO– | Cell wall |
| Alkaloids | Scavenges O2•–, •OH, H2O2, and 1O2 | Vacuole |
| Nonprotein amino acids | Scavenges O2•–, H2O2, and 1O2 | Cytosol, mitochondria, cell wall |
| Enzymatic | ||
| Superoxide dismutase (SOD; EC 1.15.1.1) | 2O2•− + 2H+→ O2 + H2O2 | Chloroplast, peroxisomes, cytosol, mitochondria, apoplast |
| Catalase (CAT; EC 1.11.1.6) | 2H2O2 → 2H2O + O2 | Peroxisomes |
| Peroxidases (POX; EC 1.11.1.7) | 2PhOH + H2O2→ 2PhO• + 2H2O 2PhO• → cross-linked substances PhO• + Asc → PhOH + MDHA PhO• + MDHA → PhOH + DHA | Cell wall, apoplast, vacuole |
| Polyphenol oxidase (PPO; EC 1.14.18.1) | PhOH + O2 → Catechols Catechols + O2 → Q + H2O | Thylakoid membrane of chloroplast, cytosol, vacuole |
| Ascorbate peroxidase (APX; EC 1.11.1.11) | H2O2 + AsA → 2H2O + MDHA | Chloroplast, peroxisomes, cytosol, mitochondria, apoplast |
| Monodehydroascorbate reductase (MDHAR; EC 1.6.5.4) | MDHA + NAD(P)H → AsA + NAD(P)+ | Chloroplast, cytosol, mitochondria |
| Dehydroascorbate reductase (DHAR; EC 1.8.5.1) | 2GSH + DHA → GSSG + AsA | Chloroplast, cytosol, mitochondria |
| Glutathione reductase (GR; EC 1.6.4.2) | GSSG + NADPH + H+ → GSH + NADP+ | Chloroplast, cytosol, mitochondria |
| Glutathione peroxidase (GPX; EC 1.11.1.9) | H2O2 + GSH → H2O + GSSG | Cytosol, mitochondria |
| Glutathione S-transferase (GST; EC 2.5.1.18) | R-X + GSH → GS-R + H-X | Chloroplast, cytosol, mitochondria |
| Peroxiredoxins (PRX; EC 1.11.1.15) | H2O2 + PRX-S– → OH– + PRX-SOH PRX-SOH + GSH → PRX-SSG + H2O PRX-SSG + GSH → PRX-S– + GSSG | Cytosol, chloroplasts, mitochondria, nucleus, extracellular spaces |
| Thioredoxin (TRX; EC 1.8. 1.9) | TRX-RS2 + NADPH + H+→ TRX-R(SH)2 + NADP+ | Chloroplast, cytosol, mitochondria |
| Plant Species | Stress Conditions | Antioxidant Defense | References |
|---|---|---|---|
| Salinity | |||
| Triticum aestivum | 100 mM NaCl; 20 d | Nitrogen supplementations increased the activity of SOD, CAT, GR, MDHAR, and DHAR by 2-folds and APX 3-folds, respectively, compared to untreated. | [182] |
| Nicotiana benthamiana | 150 mM NaCl; 15 d | Acetylcholine application increased SOD by 1-fold and POD by 2-folds. | [208] |
| Solanum lycopersicum | 150 mM NaCl; 5 d | Vanillic acid increased AsA/DHA, GSH/GSSG, MDHAR, GR, GST, SOD, and CAT by 161%, 90%, 18%, 53%, 87%, 43%, 105%, respectively. | [209] |
| Medicago sativa | 250 mM NaCl; 2 w | Melatonin increased the activities of CAT, POX, and Cu/Zn-SOD. | [210] |
| Cucumis sativus | 150 mM NaCl; 3 d | Melatonin increased CAT, SOD, POD, and APX by 23%, 29%, 15%, and 16%, respectively. | [211] |
| T. aestivum | 100 mM NaCl; 20 d | Sodium nitroprusside (SNP) and glucose solely increased Cys and GSH content by 86% and 79%, and 19% and 18%, respectively, whereas SOD, CAT, APX, and GR increased by 75% and 65%, 49% and 37%, 97% and 57%, and 60% and 57%, respectively. Combined SNP and glucose application increased the activity of these antioxidant enzymes (SOD, CAT, APX, and GR by 138%, 61%, 271%, 127% and 44%, 17%, 119%, 23%, respectively, compared to the control and glucose-treated plants. | [212] |
| C. sativus | 200 mM NaCl; 7 d | H2S increased ASA content by 42.6% and GR activity by 9.1%. Reversed decreased SOD and POD activity. | [213] |
| Brassica juncea | 100 mM NaCl; 15 d | Nitric oxide increased SOD, CAT, APX, and GR activity by 91%, 33%, 114%, and 49%, respectively. | [214] |
| Water Deficit and Simulated Drought | |||
| Zea mays cv. Run Nong 35, Wan Dan 13 and Dong Dan 80 | Mild drought (80% FC), moderate drought (60% FC), and severe drought (40% FC) | Increased activities of APX, MDHAR, and DHAR by 24%, 13%, and 29% in Dong Dan. 80% and 16%, 11%, and 10% in Wan Dan 13, respectively, under severe drought. Higher SOD activity as well as AsA and DHA contents under moderate and severe drought in both maize hybrids. | [129] |
| Glycine max and G. tomentella | Water deficit, flowering stage, 12 d | A substantially increased SOD and GR enzymes activities with the highest value during 8th day of stress treatment in G. max. A gradual increase in GR activity till the end of drought treatment was observed in G. tomentella. | [215] |
| Brassica napus cv. Binasarisha-3 | Osmotic stress (10% and 20% PEG), 48 h | MDHAR activity was higher under 10% PEG. DHAR activity increased under both stress level. GR and GST activity was higher by 26% and 23% and 25% and 31% at both stress level, respectively. | [89] |
| Phaseolus vulgaris cv. Bn-150 (drought-tolerant) and Bn-16 (drought-sensitive) | Moderate drought (50% FC) and severe drought (0% FC), 14 d | Significantly increased total phenolic contents of Bn-150 by 223% and 265%, respectively, under moderate and severe drought. SOD, CAT, APX, and GR activities were increased in tolerant genotypes (Bn-150) than the sensitive one (Bn-16). | [97] |
| Vigna radiata cv. BARI Mung-2 | Osmotic stress (5% PEG), 48 h. | Decreased AsA/DHA ratio by 54%. Increased APX and GR by 20% and 42%, respectively. Reduced CAT and MDHAR activity by 13% and 26%, respectively. | [91] |
| B. napus cv. Binasarisha-3 | Osmotic stress (10% and 20% PEG), 48 h | Moderate stress increased AsA content, GPX, and GST activity but reduced CAT activity, whereas severe stress enhanced APX activity but reduced MDHAR, DHAR, and GR activities. Both levels of stress increased GSH and GSSG contents by 31% and 26%; and 83% and 225%, respectively, compared to control. | [90] |
| B. rapa cv. BARI Sharisha-15 | Osmotic stress (20% PEG), 2 d | AsA and GSH contents increased by 10% and 72%, respectively. APX, GR, CAT, and GPX activity increased by 23%, 81%, 29%, and 26%, respectively. | [132] |
| Z. mays cv. Xida 889 and Xida 319 | Drought (50% FC), 15 d | SOD and total antioxidant activities increased, whereas CAT, APX, and POD activity declined. GSH content increased by 17% and 28% in Xida 319 and Xida 889, respectively, compared to control. | [150] |
| Oryza sativa var. japonica cv. Nipponbare | Osmotic stress (20% PEG-6000), 5 d | Decreased SOD, APX, and CAT activities, but POD activity increased by 59% compared to control. | [93] |
| S. lycopersicum cv. Login 935 | Drought stress (60% FC), 20 d | Enhanced SOD, CAT, and APX activities by 110%, 66%, and 77%, respectively. Increased AsA, GSH, and α tocopherol contents by 81%, 93%, and 103%, respectively. | [95] |
| G. max | Osmotic stress (5%, 10%, and 15% PEG), 3 w | Highest activities of CAT, APX, and PPO were observed at mild osmotic stress (5% PEG), whereas increased SOD and POX activities were found at severe osmotic stress (15% PEG). Total phenol and tocopherol contents increased by 51%, 32%, and 44%, and 26%, 26%, and 21% at three levels of osmotic stress intensities, respectively, compared to control. | [94] |
| Toxic Metals/Metalloids | |||
| Brassica napus Cv. BINA Sharisha-3 | 0.5 and 1.0 mM CdCl2, 2 d | Reduction of AsA content, whereas higher GSSG content and GST activity. APX and GR activity increased, but CAT, MDHAR, and DHAR activity reduced. | [101] |
| V. radiata cv. BARI mung-2 | 0.5 mM AlCl3, 2 and 3 d | AsA content reduced, but GSH and GSSG increased. The activity of SOD, GST, GPX, APX, and GR increased, but MDHAR, DHAR, and CAT decreased. | [133] |
| Oryza sativa cv. BRRI dhan54 | 0.25 and 0.5 mM NiSO4⋅7H2O, 3 d | 64% lower AsA and 146% higher GSH content at 0.5 mM Ni stress. APX, MDHAR, DHAR, and GR activities increased by 114%, 116%, 31%, and 104% at 0.5 mM Ni stress, respectively. | [98] |
| Pisum sativum | 100 µM NiCl2, 3 d | GSH accumulation increased by 5-fold. SOD activity increased by 14-fold, CAT and APX activities both by 6-fold, and GR activity by almost 3-fold. | [99] |
| P. sativum | 100 µM CdCl2, 3 d | GSH accumulation increased by 3-fold and GSSG by 2-fold. SOD activity increased by 10-fold, CAT and APX activities both by 8-fold, and GR activity by almost 4-fold. | [99] |
| B. juncea cv. BARI Sharisha-11 | 0.5 and 1.0 mM CdCl2, 3 d | 42% lower AsA and 200% higher DHA at severe stress, whereas 44% and 72% higher GSSG content under mild and severe stress, respectively. 44% higher SOD and 31% higher GPX activity at severe stress. | [102] |
| T. aestivum cv. Pradip | 0.5 and 1.0 mM Pb(NO3)2, 2 d | APX activity increased, but MDHAR and DHAR decreased; GR increased initially and then declined. 35% higher SOD, 44% higher GST along with 31% lower CAT, and 28% lower GPX activities were reported. | [107] |
| Extreme Temperature | |||
| Cicer arietinum (sensitive genotype: ICC14183, ICC5912; tolerant genotypes: ICCV07110, ICCV92944) | 30/20, 35/25, 40/30, and 45/35 °C; 2 d for flower and 8 d for three leaves stage | Reduced APX (by 38–49% and 43–50% at 40/30 °C) and GR (by 30–46% and 44–49% at 45/35 °C) activity as well as AsA (by 13–18% and 28–32% at 40/30 °C), and GSH (by 24–33% and 37–44% at 45/35 °C) content in sensitive genotypes. | [149] |
| Cucumis sativus | 35 ± 1 °C; 7 d | Improved SOD (by 16.6%), CAT (by 13%), APX (by 25.2%), GR (by 14.4%), and POD (by 35.4%) activity. | [113] |
| Sorghum bicolor | 36/26 and 39/29 °C; until 7 d after full anthesis | In pollen, decreased the SOD (58–87%), CAT (44–56%), and POX (36–60%) activity. In pistil, decreased the SOD (59–77%), CAT (35–60%), and POX (42–78%) activity. | [114] |
| Gossypium hirsutum | 45/30 ± 2 °C; 120 d | Enhanced SOD and CAT activity. | [134] |
| O. sativa | 38 °C; 5 d | Decreased the activity of SOD and CAT. Enhanced POD (by 32.1%) activity. | [93] |
| O. sativa cv. DM You 6188 | 12 °C; 6 d | Enhanced SOD (by 1.4%), CAT (by 1.58%), and GSH/GSSG (by 2.42-fold). | [116] |
| Calendula officinalis | 4 °C; 24, 48, 72, 96, and 120 h | Elevated GR (161%), SOD (46%), and APX (82%) activity at 120 h. | [216] |
| Capsella bursa-pastoris | 10 °C; 24, 48, 72, 96, and 120 h | Elevated GR (70%), POD (79%), and CAT (70%) activity at 120 h. | [202] |
| Citrus reticulata | 1, −1, and −3 °C; 3 h | Enhanced CAT (1.35-fold) and APX (2-fold) activities. | [203] |
| Vitis vinifera | 5 °C; 6, 12, 24, 48, and 72 h | Elevated GR (20.26%), DHAR (7.64%), and MDHAR (16.60%) activities with increased AsA (12.13%), DHA (7.89%), and GSH (56.09%) contents. | [217] |
| Waterlogging | |||
| S. bicolor cv. JN01 and JZ31 | Waterlogged soil, 12 d | Increased SOD (by 1.38- and 1.5-fold) and CAT (by 1.43- and 1.36-fold) in JN01 and JZ31, respectively. | [120] |
| Hordeum vulgare cvs. TF57 and TF58 | Waterlogged soil, 21 d | SOD, POD, and CAT activities increased in both WL-sensitive TF57 and WL-tolerant TF58 genotype. | [137] |
| S. lycopersicum cv. Roma | Waterlogged soil, 15 d | AsA content reduced by 31%. SOD, CAT, and POD activities increased by 7%, 33%, and 57%, respectively, compared with control samples. | [122] |
| Deschampsia antarctica | Waterlogged soil, 7 d | Increment of CAT activity by 91%. | [123] |
| Sesamum indicum cv. BARI Til-4 | Waterlogged soil, 2, 4, 6, and 8 d | GSH and GSSG increased by 45% and 150%, respectively, whereas AsA content decreased by 38% after 8 d WL. APX and MDHAR activity increased by 61% and 55%, but DHAR and GR activity reduced by 59% and 23%, respectively, after 8 d WL. | [121] |
| Plant Species | Stress Condition | H2O2 Treatments | Positive Effects | References |
|---|---|---|---|---|
| Triticum aestivum cv. Zhengmai No. 004 | 150 mM NaCl; 2 d | Cotreatment; 0.05 µM, 2 d | Decreased MDA content and O2•− generation. Increased GSH and carotene content by 21% and 33%, respectively. Increased SOD, POD, CAT, and APX activity. Increased growth and biomass. | [229] |
| Cucumis sativus cv. Jinchun no. 4 and Lvfeng no. 6 | Osmotic stress; (10% PEG 6000); 2 d | Pretreatment as spraying; 1.5 mM | Decreased MDA and H2O2 content. Increased AsA and GSH content. Increased activity of GPX, CAT, APX, GR, MDHAR, and DHAR. | [230] |
| C. sativus cv. Jinchun no. 4 | Low light; 100 mol m−2 s−1; 144 h | Pretreatment as spraying; 1.5 mM | Decreased O2•−, H2O2, and MDA content. Increased CAT, SOD, APX, GR, MDHAR, and DHAR activity. | [231] |
| Vigna radiata L. Wilczek) cv. SML-668 | Cu, (CuSO4·5H2O); 50 and 100 mg kg−1 of soil. | Spraying; 2.5 mM | Increased relative water content (RWC) and SPAD value. Increased Pro content. Enhanced activity of SOD and CAT. Increased growth. | [16] |
| Zea mays | Osmotic stress (3% PEG 6000), 12 h | Pretreatment; 10 mM, 6 h | Decreased water loss, MDA, and H2O2 content. Increased levels of soluble sugars and proline. Increased Put, Spd, and Spm content by 72%, 106%, and 68%, respectively, over control. | [15] |
| Glycine max cv. Merrill 537 | Drought; withholding irrigation, 4 and 7 d | Foliar spray; 1 mM, 3 d | Improved water status, pigment content, and alleviated lipid peroxidation. Decreased MDA and H2O2 content. Increased activity of SOD (by 93% and 190%), CAT (by 49% and 120%), APX (by 106% and 194%), and GR (by 31% and 229%) on day 4 and 7 of the drought period, respectively, over control. | [17] |
| C. sativus L. | Drought; 60 ± 5% FC | Spraying; 1.5 mM (100 mL pot−1) | Decreased MDA content and ROS (O2•−, H2O2) generation. Increased activity of SOD and POD. Increased soluble sugar and proline content. Increased chl and RWC. | [18] |
| Brassica napus cv. Binasarisha-3 | Cd; 0.5 mM (mild) and 1.0 mM (severe) CdCl2; 2 d | Pretreatment; 50 µM, 24 h | Decreased MDA content (by 23% and 25%) under both Cd toxicity levels, respectively. Decreased H2O2 content. Increased AsA (by 32% and 30%); GSH content (by 38% and 25%) under both Cd stresses, respectively. Enhanced GSH/GSSG ratio. Increased activity of APX (by 40% and 39%), DHAR (by 77% and 67%), GR (by 36% and 79%), GST (by 44% and 43%), CAT (by 79% and 47%), and glyoxalase II (by 47% and 55%) under both Cd stresses, respectively. Enhanced GPX activity (by 40%) under severe stress and glyoxalase I activity (by 35%) under mild stress. | [22] |
| T. aestivum cv. Fsd-2008 and S-24 | Drought; withholding irrigation, 6 w | Seed priming; 1.5 mM, 16 h | Decreased MDA and H2O2 content. Enhanced the activity of SOD, CAT, and POD. Increased photosynthetic pigments. Increased GB and Pro content. | [14] |
| Oryza sativa cv. BRRI dhan29 | Osmotic stress (15% PEG-6000) | Foliar spray; 5 and 10 mM | Decreased MDA and H2O2 content. Increased activity of CAT and GPX. Protected photosynthetic pigments. | [13] |
| Stress Condition and Duration | Studied Plant | Source Plant | Gene | Impact on Antioxidant Defense Systems | References |
|---|---|---|---|---|---|
| Salinity | |||||
| 0, 50, 100, and 150 mM NaCl; 7 and 15 d | Solanum tuberosum | Potentilla atrosanguinea and Rheum australe | PaSOD and RaAPX | Enzyme activities are enhanced in transgenic plants as of SOD by 2–6-fold in PaSOD and 1–3-fold in double transgenic plants (DTP); APX by 5–11-fold in APX and 4–8 fold in DTP. | [265] |
| 100, 200, and 400 mM; 1, 5, 10, 15 d | Chrysanthemum | Dendronthema grandiform | DgNAC1 | Enzyme activities are enhanced in transgenic plants as of SOD by 2-, CAT by 2-, and POD by 3-folds. | [264] |
| 150 mM NaCl; 3, 6, 9, 12, and 24 h | Arabidopsis thaliana | Vitis vinifera | VvWRKY30 | Enzyme activities are significantly enhanced in transgenic plants, i.e., POD, CAT, and SOD. | [271] |
| 150 or 200 mM NaCl, till germination | Glycine max | Glycine max | GmMYB84 | Enzyme activities are significantly enhanced in transgenic plants, i.e., SOD, POD, and CAT. | [272] |
| Water Deficit and Simulated Drought | |||||
| Osmotic stress (20% PEG); 1, 3, 6 12, 24, and 48 h | Nicotiana tabacum | Spinacia oleracea | SoCYP85A1 | Overexpressed lines improve the activity of POD by 1.3–1.5 and SOD by 1.36–1.39-fold | [273] |
| Withholding water for 14 and 21 d | Malus domestica | Malus domestica | MdATG18a | Enzyme activities are enhanced in transgenic plants as of CAT and POD by 1.57–2.05-fold in overexpressed lines. | [263] |
| Withholding water till the wilting stage | Arachis hypogaea | Macrotyloma uniflorum Lam. Verdc. | MuWRKY3 | Enzyme activities are enhanced in transgenic plants as of SOD by 3–5 and APX by 3-7–fold | [262] |
| Osmotic stress (15% PEG); 60 d | N. tabacum | Zea mays | ZmSO | Overexpressed lines increase the activity of GSH 64% and 88%. | [274] |
| Osmotic stress (15% and 25% PEG); 7 d | A. thaliana | Cicer arietinum | CaMT | Enzyme activities are enhanced in transgenic plants as of APX 488%, POD 135%, GPX 134%, and GRX 186%. | [275] |
| Withholding water for 12 d | A. thaliana | Malus prunifolia | MpDGK2 | Enzyme activities are enhanced in transgenic plants, i.e., CAT, APX, and POD. | [276] |
| Toxic Metals/Metalloids | |||||
| As(III) (5 and 10 μM (NaAsO2), As(V) (50 and 100 μM (Na2HAsO4), Cd (30 and 50 μM (CdCl2) and Cr (K2Cr2O7) | A. thaliana | Oryza sativa | OsSultr1;1 | Enzyme activity is enhanced in transgenic plants, i.e., GSH with As(III) toxicity. | [277] |
| 300 μM CdCl2 and 300 μM NiCl2.6H2O; 1, 12, 24, and 48 h | N. tabacum | Salicornia brachiate | SbMYB15 | Enzyme activities are enhanced in transgenic plants as of CAT and SOD and also increase the expression of MnSOD at 100 μM (1.69-fold) and 300 μM (3.2-fold) of CdCl2 and CAT1 by 62.19- and 9.8-fold at 100 and 300 μM. | [266] |
| Extreme Temperature | |||||
| 45 °C; 0.5, 1, 2, 3, 6, 9, 12, and 24 h | N. tabacum | Triticum aestivum | TaFBA1 | Enzyme activities are enhanced in transgenic plants, i.e., SOD, POD, and APX, while CAT activity was decreased under heat stress. | [278] |
| 48 °C; 6 h | M. domestica | M. domestica | MdATG18a | Enzyme activities are enhanced in transgenic plants, i.e., SOD, POD, CAT, AsA, and GSH, whereas a decreased ratio of GSH/GSSG was reported. | [279] |
| 4 °C; 5 d | Solanum lycopersicum | A. thaliana | AtDREB1A | Enzyme activities are enhanced in transgenic plants as of SOD by 29.49% and CAT by 21.34%. | [268] |
| 4 °C; 6, 12, 24, 36, and 48 h | S. tuberosum | S. tuberosum | StSOD1 | Enzyme activities are enhanced in transgenic plants as of SOD by 1.38, POD by 1.24, and CAT by 1.37 folds. | [269] |
| Waterlogging | |||||
| 2 cm waterlogging; 3, 6, 12, 24, and 72 h | A. thaliana | Brassica napus | BnERF2.4 | Enzyme activities are enhanced in transgenic plants, i.e., SOD, POD, and CAT. | [280] |
| Soil–atmosphere interface for 1 w | A. thaliana | Mentha arvensis | MaRAP2-4 | Enzyme activities are enhanced in transgenic plants, i.e., CAT, GPX, and SOD. | [281] |
| 5 cm waterlogging; 24 and 48 h | A. thaliana | Dioscorea alata | DaAPX | Enzyme activity is enhanced in transgenic plants, i.e., APX but no significant effect on CAT. | [217] |
| 3 cm standing water; 14 d | Chrysanthemum morifolium | Chrysanthemum morifolium | CmSOS1 | Overexpressed plants enhance the activities of SOD and CAT by 171%. | [270] |
| 3.0 cm above the nutritional substance surface; 2 weeks | A. thaliana | Hordeum vulgare | HvERF2.11 | Enzyme activities are enhanced in transgenic plants as of SOD by 55%, 48%, and 45%, POD by 64%, 65%, and 70%, CAT by 2.2%, 2.1%, and 2.1%, alcohol dehydrogenases by 2.1-, 2.3-, and 1.9-fold in three transgenic lines, respectively. | [282] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants 2020, 9, 681. https://doi.org/10.3390/antiox9080681
Hasanuzzaman M, Bhuyan MHMB, Zulfiqar F, Raza A, Mohsin SM, Mahmud JA, Fujita M, Fotopoulos V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants. 2020; 9(8):681. https://doi.org/10.3390/antiox9080681
Chicago/Turabian StyleHasanuzzaman, Mirza, M.H.M. Borhannuddin Bhuyan, Faisal Zulfiqar, Ali Raza, Sayed Mohammad Mohsin, Jubayer Al Mahmud, Masayuki Fujita, and Vasileios Fotopoulos. 2020. "Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator" Antioxidants 9, no. 8: 681. https://doi.org/10.3390/antiox9080681
APA StyleHasanuzzaman, M., Bhuyan, M. H. M. B., Zulfiqar, F., Raza, A., Mohsin, S. M., Mahmud, J. A., Fujita, M., & Fotopoulos, V. (2020). Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants, 9(8), 681. https://doi.org/10.3390/antiox9080681












