Alterations in the Antioxidant Enzyme Activities in the Neurodevelopmental Rat Model of Schizophrenia Induced by Glutathione Deficiency during Early Postnatal Life
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Treatment
2.2. Chemicals and Reagents
2.3. Preparation of Tissue Homogenates
2.4. Determination of Total Superoxide Dismutase (SOD) Activity
2.5. Determination of Catalase (CAT) Activity
2.6. Determination of Glutathione Peroxidase (GPx) Activity
2.7. Determination of Glutathione Disulfide Reductase (GR) Activity
2.8. Determination of Reactive Oxygen Species (ROS) Level
2.9. Determination of the Concentration of MDA
2.10. Determination of Total GSH and Cys Levels in the Striatum
2.11. Determination of Met Level in the Striatum
2.12. Determination of Bound Sulfane Sulfur
2.13. Determination of Protein Content
2.14. Statistics
3. Results
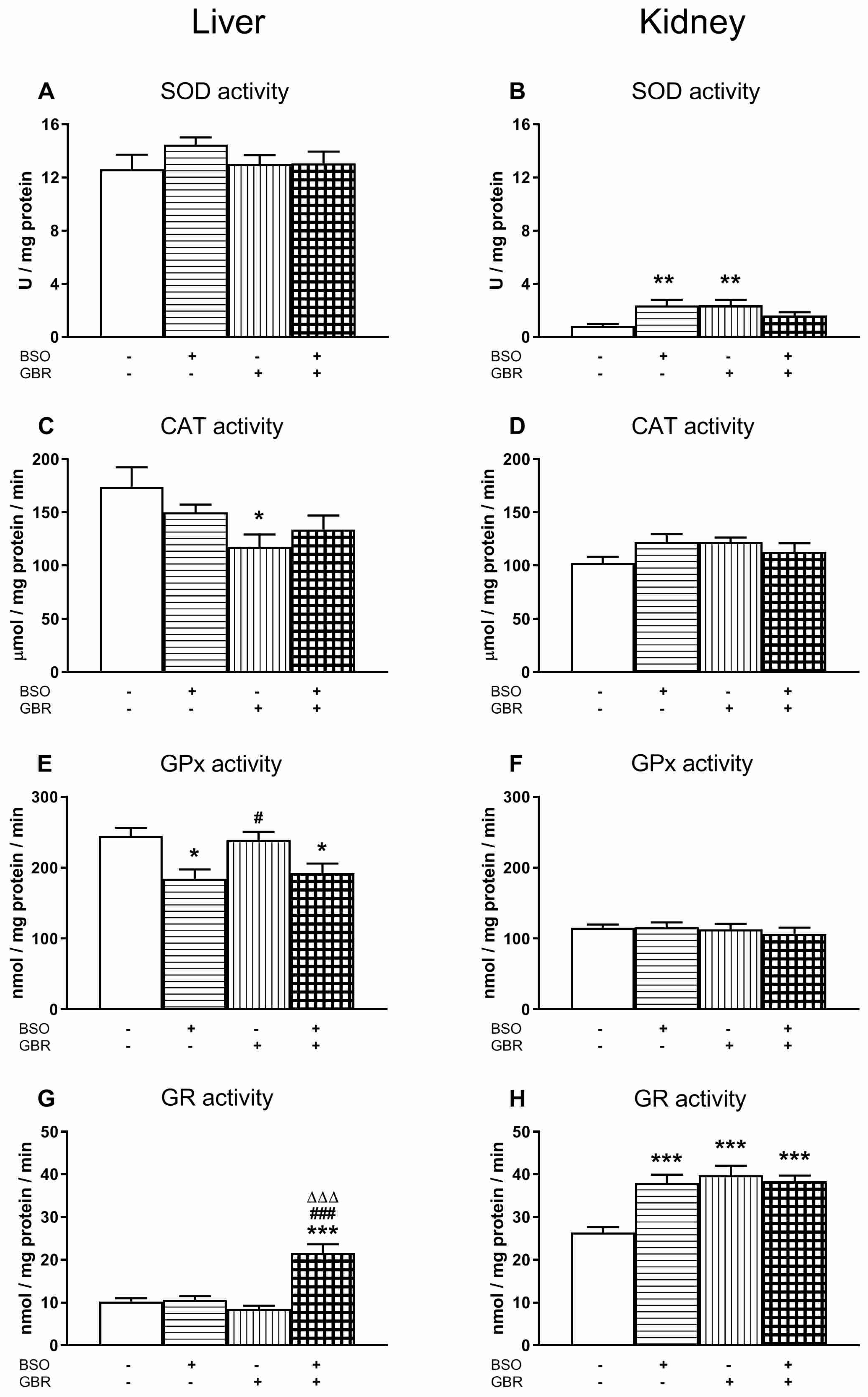
3.1. The Effects of Chronic Treatment with BSO and GBR 12909 on the Enzymatic Activities of Superoxide Dismutase (SOD), Catalase (CAT), Glutathione Peroxidase (GPx) and Glutathione Disulfide Reductase (GR) in the Liver and Kidney of 16-Day-Old Rats
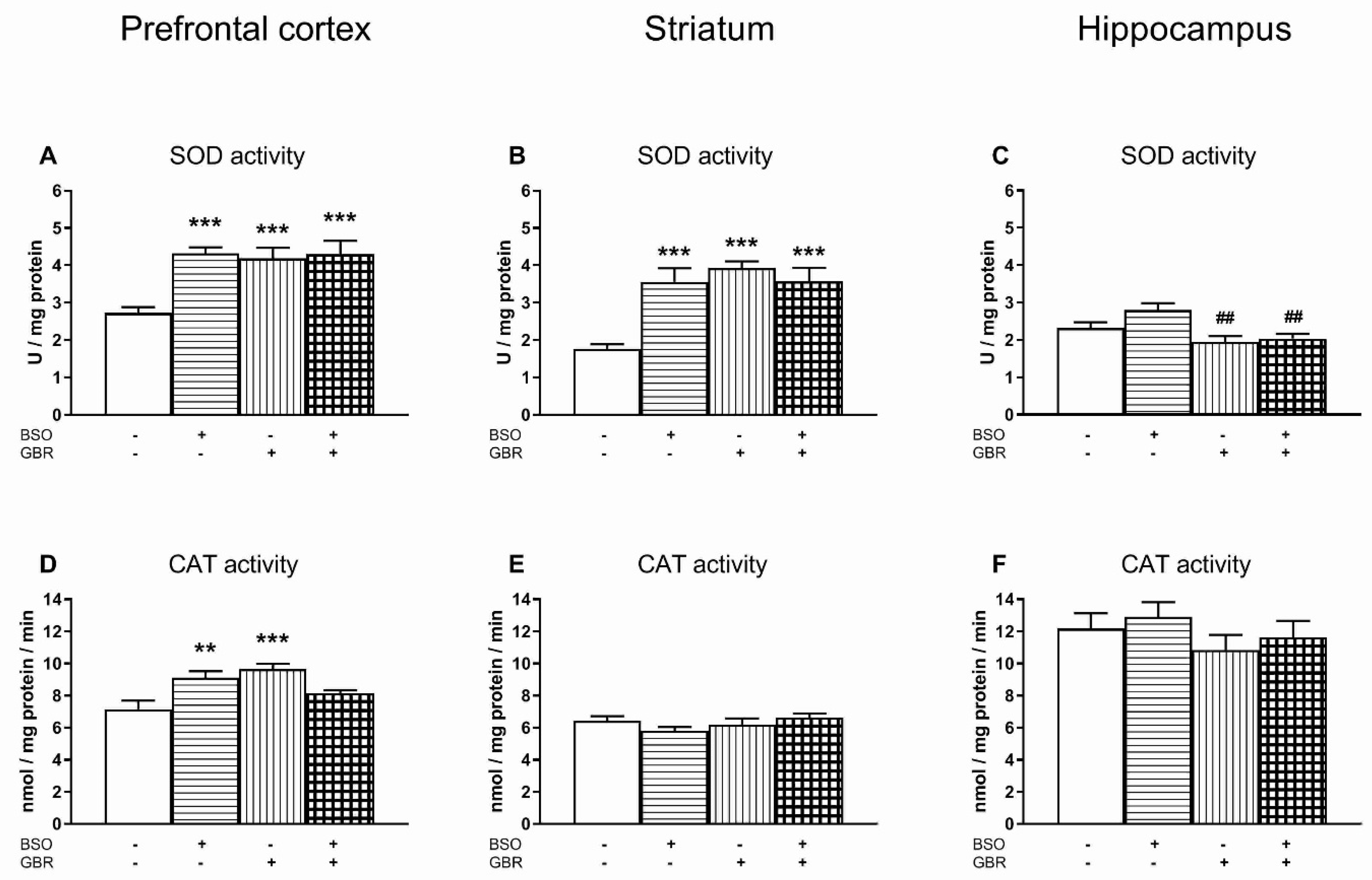
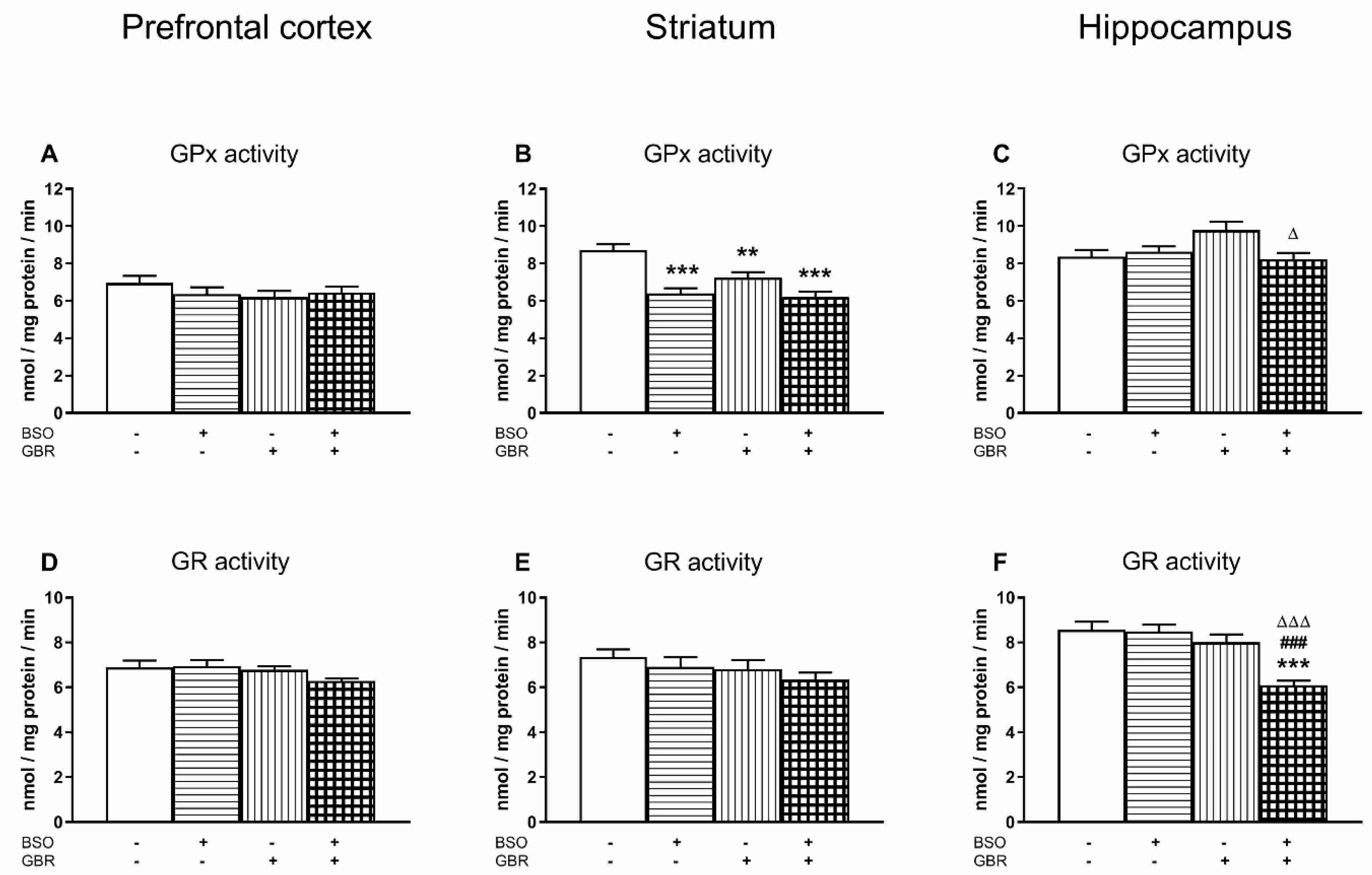
3.2. The Effects of Chronic Treatment with BSO and GBR 12909 on the Enzymatic Activities of Superoxide Dismutase (SOD), Catalase (CAT), Glutathione Peroxidase (GPx) and Glutathione Disulfide Reductase (GR) in the Selected Brain Structures of 16-Day-Old Rats
3.3. The Effects of Chronic Administration of BSO and GBR 12909 on the Total Levels of ROS in the Liver, Kidney and the Selected Brain Structures
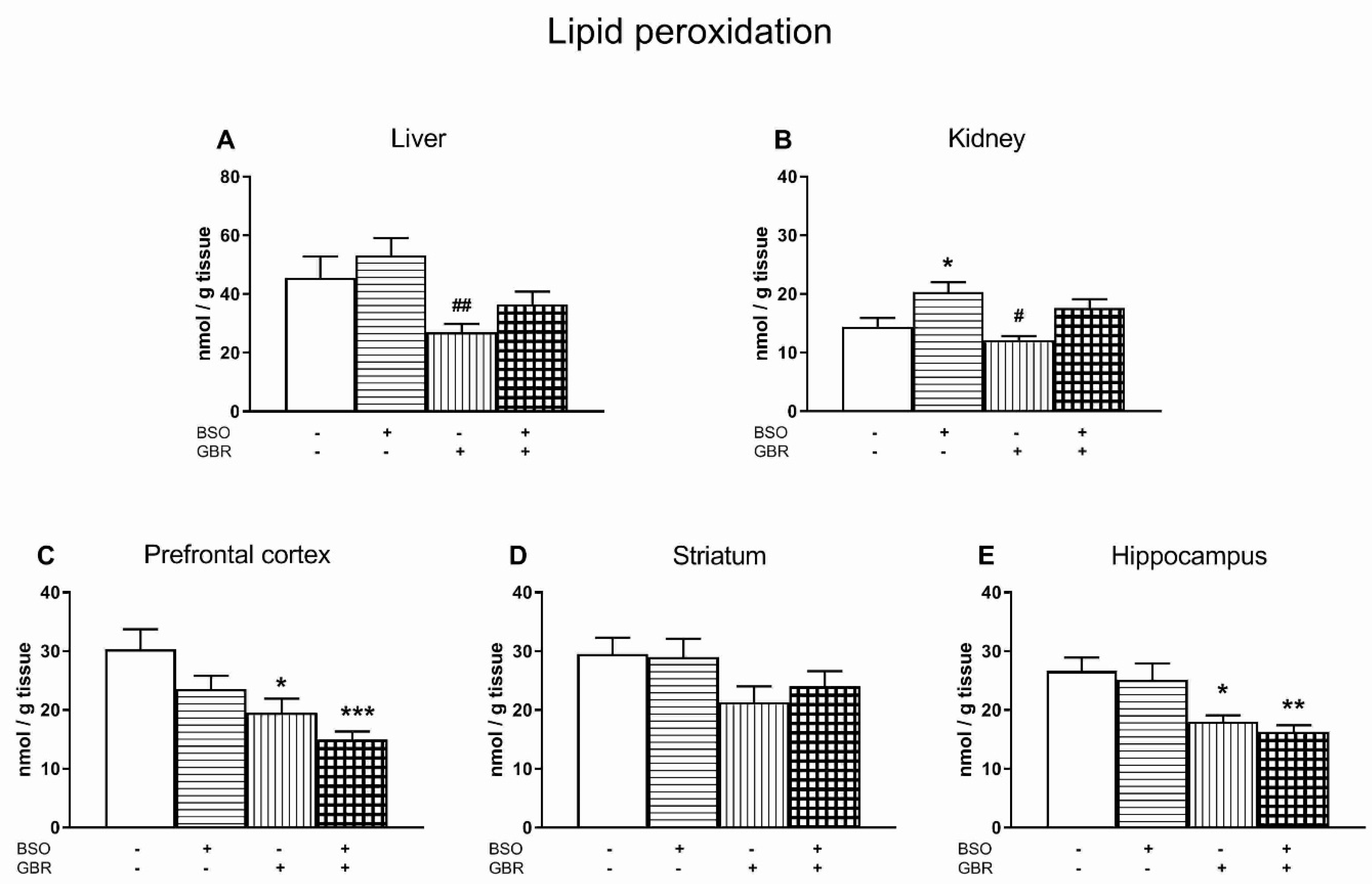
3.4. The Effects of Chronic Administration of BSO and GBR 12909 on Lipid Peroxidation in the Liver, Kidney and the Selected Brain Structures
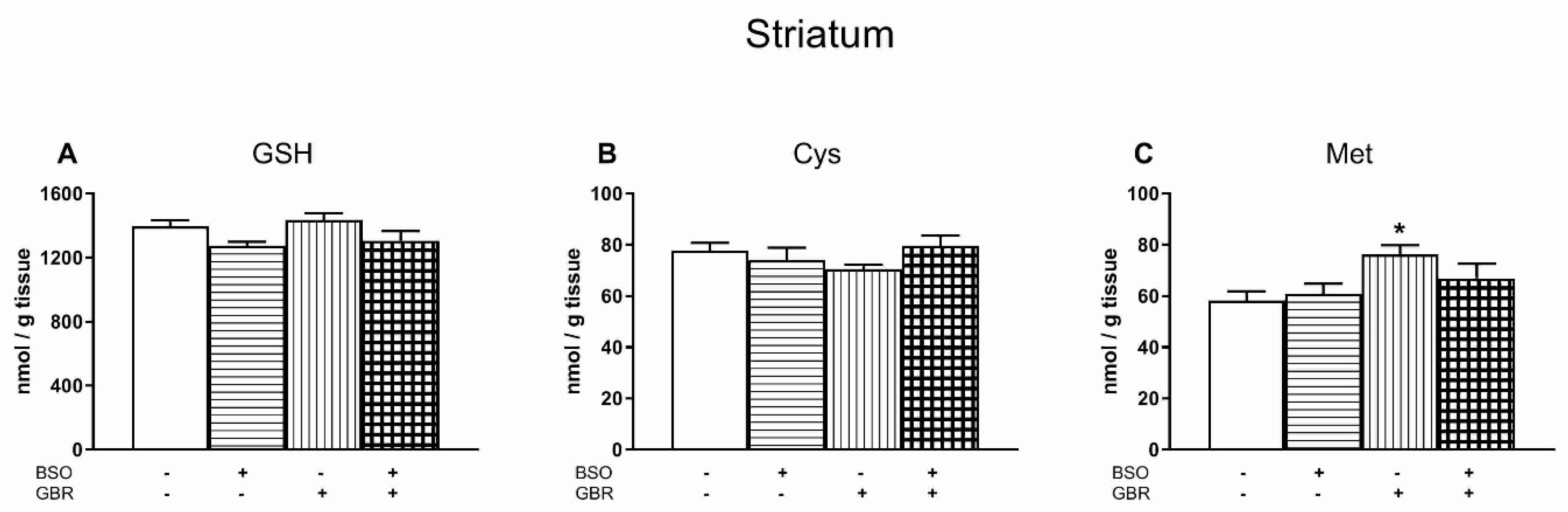
3.5. The Effects of Chronic Treatment with BSO and GBR 12909 on the Levels of GSH, Cys and Met in the Striatum (STR)
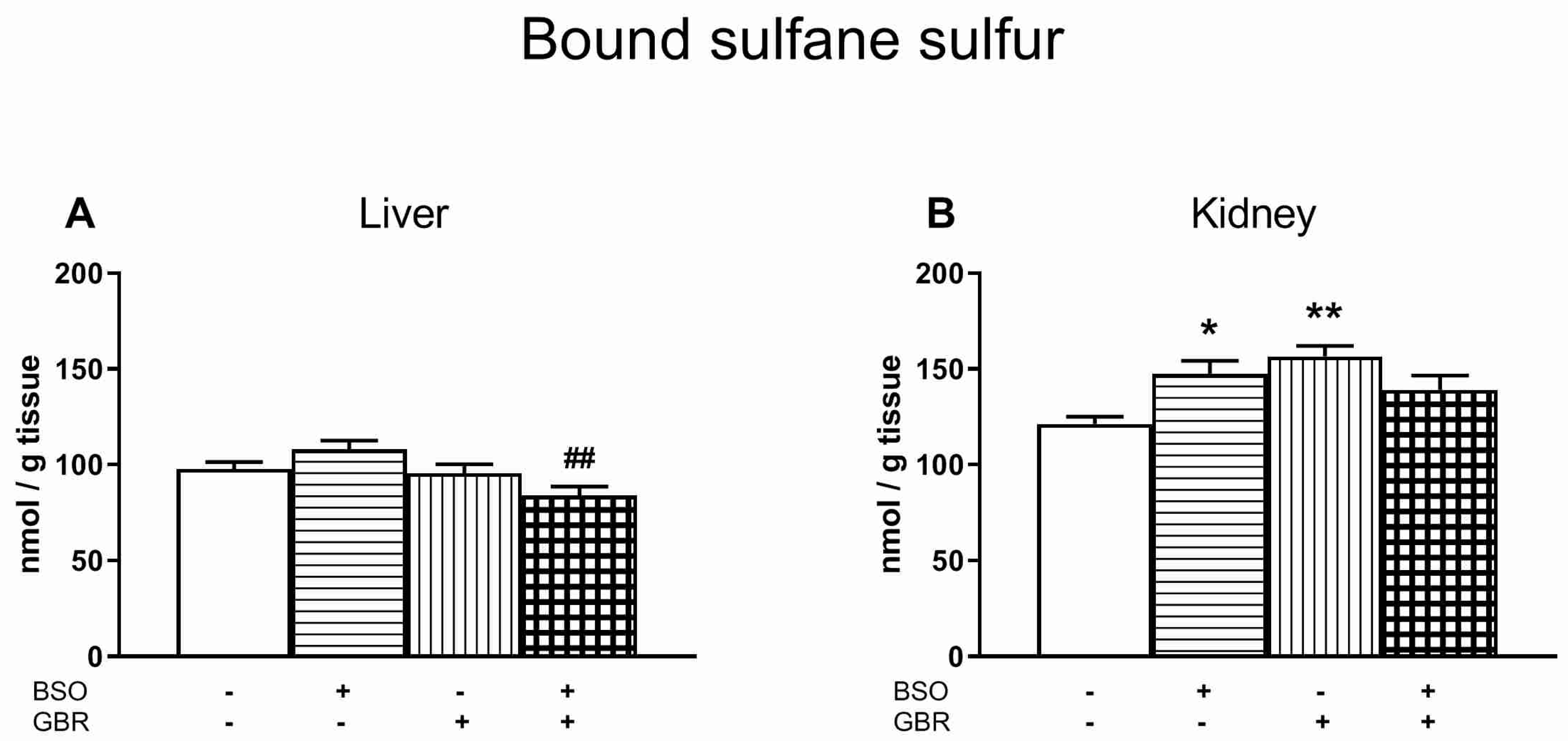
3.6. The Effects of Chronic Treatment with BSO and GBR 12909 on the Boud Sulfane Sulfur Level in the Liver and Kidney
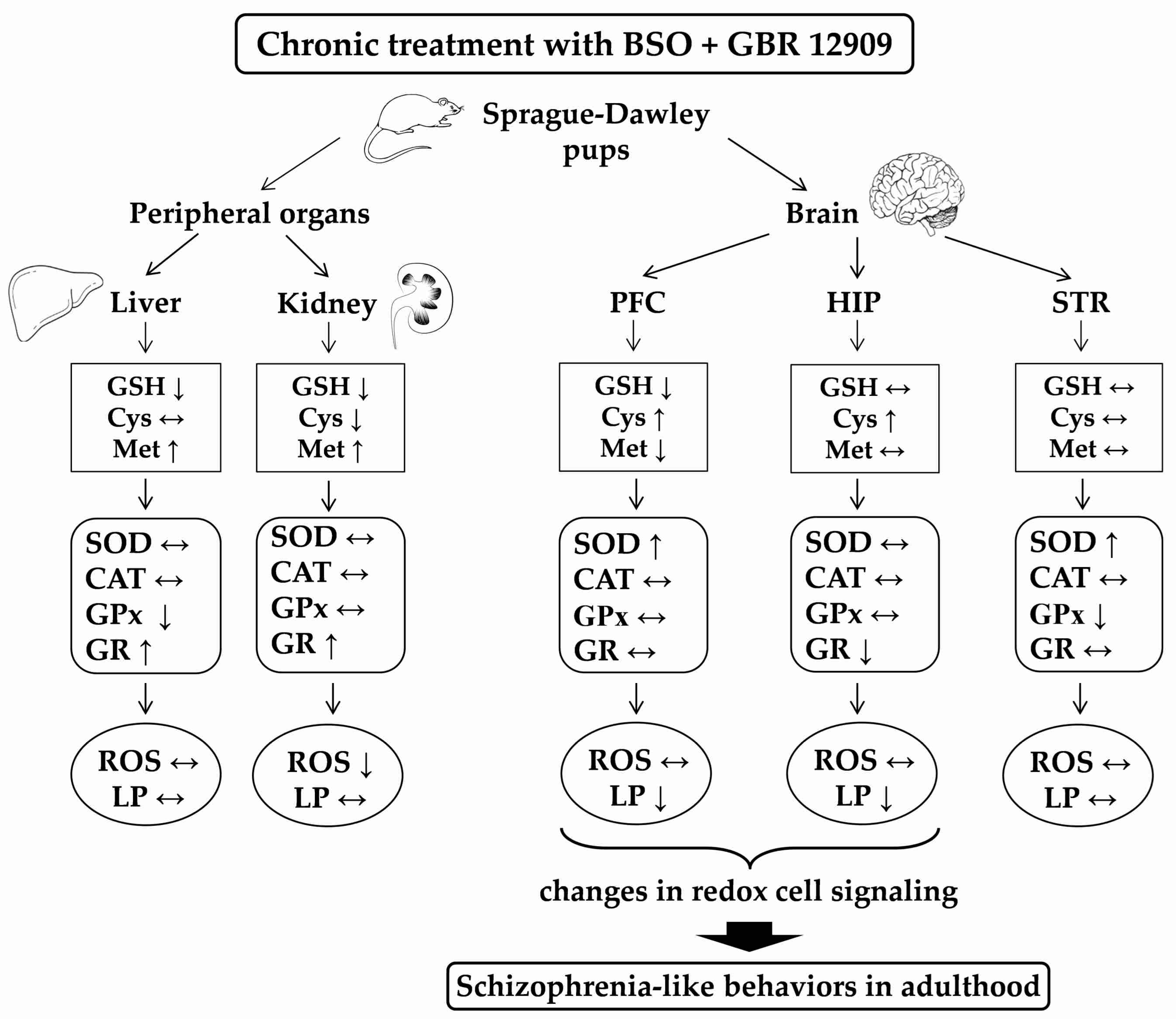
4. Discussion
4.1. Basic Activity of Antioxidant Enzymes and Production of ROS in the Studied Tissues
4.2. Potential Cause-Effect Relationships between Concentrations of GSH, Met, ROS and Lipid Peroxidation Products and the Antioxidant Enzymes Activities in the Liver, Kidney and Brain of Rats Treated with BSO and GBR 12909, Alone and in Combination
4.2.1. Liver
4.2.2. Kidney
4.2.3. Brain
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mahadik, S.P.; Mukherjee, S. Free radical pathology and antioxidant defense in schizophrenia: A review. Schizophr. Res. 1996, 19, 1–17. [Google Scholar] [CrossRef]
- Mukerjee, S.; Mahadik, S.P.; Scheffer, R.; Correnti, E.E.; Kelkar, H. Impaired antioxidant defense at the onset of psychosis. Schizophr. Res. 1996, 19, 19–26. [Google Scholar] [CrossRef]
- Ng, F.; Berk, M.; Dean, O.; Bush, A.I. Oxidative stress in psychiatric disorders: Evidence base and therapeutic implications. Int. J. Neuropsychopharmacol. 2008, 11, 851–876. [Google Scholar] [CrossRef] [PubMed]
- Bitanihirwe, B.K.; Woo, T.U. Oxidative stress in schizophrenia: An integrated approach. Neurosci. Biobehav. Rev. 2011, 35, 878–893. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.K.; Keshavan, M.S. Antioxidants, redox signaling, and pathophysiology in schizophrenia: An integrative view. Antioxid. Redox Signal. 2011, 15, 2011–2035. [Google Scholar] [CrossRef] [PubMed]
- Kemp, M.; Go, Y.M.; Jones, D.P. Nonequilibrium thermodynamics of thiol/disulfide redox systems: A perspective on redox systems biology. Free Radic. Biol. Med. 2008, 44, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Radical-free biology of oxidative stress. Am. J. Physiol. Cell Physiol. 2008, 295, C849–C868. [Google Scholar] [CrossRef]
- Yao, J.K.; Leonard, S.; Reddy, R. Altered glutathione redox state in schizophrenia. Dis. Markers 2006, 22, 83–93. [Google Scholar] [CrossRef]
- Do, K.Q.; Trabesinger, A.H.; Kirsten-Krüger, M.; Lauer, C.J.; Dydak, U.; Hell, D.; Holsboer, F.; Boesiger, P.; Cuénod, M. Schizophrenia: Glutathione deficit in cerebrospinal fluid and prefrontal cortex in vivo. Eur. J. Neurosci. 2000, 12, 3721–3728. [Google Scholar] [CrossRef]
- Matsuzawa, D.; Hashimoto, K. Magnetic resonance spectroscopy study of the antioxidant defense system in schizophrenia. Antioxid. Redox Signal. 2011, 15, 2057–2065. [Google Scholar] [CrossRef]
- Gawryluk, J.W.; Wang, J.F.; Andreazza, A.C.; Shao, L.; Young, L.T. Decreased levels of glutathione, the major brain antioxidant, in post-mortem prefrontal cortex from patients with psychiatric disorders. Int. J. Neuropsychopharmacol. 2011, 14, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.M.; Pradhan, S.; Coughlin, J.M.; Trivedi, A.; DuBois, S.L.; Crawford, J.L.; Sedlak, T.W.; Nucifora, F.C., Jr.; Nestadt, G.; Nucifora, L.G.; et al. Assessing brain metabolism with 7-T proton magnetic resonance spectroscopy in patients with first-episode sychosis. JAMA Psychiatry 2019, 76, 314–323. [Google Scholar] [CrossRef]
- Das, T.K.; Javadzadeh, A.; Dey, A.; Sabesan, P.; Théberge, J.; Radua, J.; Palaniyappan, L. Antioxidant defense in schizophrenia and bipolar disorder: A meta-analysis of MRS studies of anterior cingulate glutathione. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 91, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Tosic, M.; Ott, J.; Barral, S.; Bovet, P.; Deppen, P.; Gheorghita, F.; Matthey, M.L.; Parnas, J.; Preisig, M.; Saraga, M.; et al. Schizophrenia and oxidative stress: Glutamate cysteine ligase modifier as a susceptibility gene. Am. J. Hum. Genet. 2006, 79, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Gysin, R.; Kraftsik, R.; Sandell, J.; Bovet, P.; Chappuis, C.; Conus, P.; Deppen, P.; Preisig, M.; Ruiz, V.; Steullet, P.; et al. Impaired glutathione synthesis in schizophrenia: Convergent genetic and functional evidence. Proc. Natl. Acad. Sci. USA 2007, 104, 16621–16626. [Google Scholar] [CrossRef] [PubMed]
- Gysin, R.; Kraftsik, R.; Boulat, O.; Bovet, P.; Conus, P.; Comte-Krieger, E.; Polari, A.; Steullet, P.; Preisig, M.; Teichmann, T.; et al. Genetic dysregulation of glutathione synthesis predicts alteration of plasma thiol redox status in schizophrenia. Antioxid. Redox Signal. 2011, 15, 2003–2010. [Google Scholar] [CrossRef]
- Matsuzawa, D.; Obata, T.; Shirayama, Y.; Nonaka, H.; Kanazawa, Y.; Yoshitome, E.; Takanashi, J.; Matsuda, T.; Shimizu, E.; Ikehira, H.; et al. Negative correlation between brain glutathione level and negative symptoms in schizophrenia: A 3T 1H-MRS study. PLoS ONE 2008, 3, e1944. [Google Scholar] [CrossRef]
- Shukitt-Hale, B.; Erat, S.A.; Joseph, J.A. Spatial learning and memory deficits induced by dopamine administration with decreased glutathione. Free Radic. Biol. Med. 1998, 24, 1149–1158. [Google Scholar] [CrossRef]
- Almaguer-Melian, W.; Cruz-Aguado, R.; Bergado, J.A. Synaptic plasticity is impaired in rats with a low glutathione content. Synapse 2000, 38, 369–374. [Google Scholar] [CrossRef]
- Cruz-Aguado, R.; Almaguer-Melian, W.; Díaz, C.M.; Lorigados, L.; Bergado, J. Behavioral and biochemical effects of glutathione depletion in the rat brain. Brain Res. Bull. 2001, 55, 327–333. [Google Scholar] [CrossRef]
- Castagné, V.; Cuénod, M.; Do, K.Q. An animal model with relevance to schizophrenia: Sex-dependent cognitive deficits in osteogenic disorder-Shionogi rats induced by glutathione synthesis and dopamine uptake inhibition during development. Neuroscience 2004, 123, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Castagné, V.; Rougemont, M.; Cuénod, M.; Do, K.Q. Low brain glutathione and ascorbic acid associated with dopamine uptake inhibition during rat’s development induce long-term cognitive deficit: Relevance to schizophrenia. Neurobiol. Dis. 2004, 15, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Cabungcal, J.H.; Preissmann, D.; Delseth, C.; Cuénod, M.; Do, K.Q.; Schenk, F. Transitory glutathione deficit during brain development induces cognitive impairment in juvenile and adult rats: Relevance to schizophrenia. Neurobiol. Dis. 2007, 26, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Do, K.Q.; Cabungcal, J.H.; Frank, A.; Steullet, P.; Cuénod, M. Redox dysregulation, neurodevelopment, and schizophrenia. Curr. Opin. Neurobiol. 2009, 19, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Steullet, P.; Cabungcal, J.H.; Coyle, J.; Didriksen, M.; Gill, K.; Grace, A.A.; Hensch, T.K.; LaMantia, A.S.; Lindemann, L.; Maynard, T.M.; et al. Oxidative stress-driven parvalbumin interneuron impairment as a common mechanism in models of schizophrenia. Mol. Psychiatr. 2017, 22, 936–943. [Google Scholar] [CrossRef]
- Steullet, P.; Cabungcal, J.H.; Kulak, A.; Kraftsik, R.; Chen, Y.; Dalton, T.P.; Cuénod, M.; Do, K.Q. Redox dysregulation affects the ventral but not dorsal hippocampus: Impairment of parvalbumin neurons, gamma oscillations, and related behaviors. J. Neurosci. 2010, 30, 2547–2558. [Google Scholar] [CrossRef]
- Lorenc-Koci, E. Dysregulation of glutathione synthesis in psychiatric disorders. In Studies on Psychiatric Disorders. Oxidative Stress in Applied Basic Research and Clinical Practice, 1st ed.; Dietrich-Muszalska, A., Chauhan, V., Grignon, S., Eds.; Springer: New York, NY, USA; Heidelberg, Germany; Dordrecht, The Netherlands; London, UK, 2015; pp. 269–299. [Google Scholar]
- Kulak, A.; Cuénod, M.; Do, K.Q. Behavioral phenotyping of glutathione-deficient mice: Relevance to schizophrenia and bipolar disorder. Behav. Brain Res. 2012, 226, 563–570. [Google Scholar] [CrossRef]
- Dalton, T.P.; Dieter, M.Z.; Yang, Y.; Shertzer, H.G.; Nebert, D.W. Knockout of the mouse glutamate cysteine ligase catalytic subunit (Gclc) gene: Embryonic lethal when homozygous, and proposed model for moderate glutathione deficiency when heterozygous. Biochem. Biophys. Res. Commun. 2000, 279, 324–329. [Google Scholar] [CrossRef]
- Dalton, T.P.; Chen, Y.; Schneider, S.N.; Nebert, D.W.; Shertzer, H.G. Genetically altered mice to evaluate glutathione homeostasis in health and disease. Free Radic. Biol. Med. 2004, 37, 1511–1526. [Google Scholar] [CrossRef]
- Yang, Y.; Dieter, M.Z.; Chen, Y.; Shertzer, H.G.; Nebert, D.W.; Dalton, T.P. Initial characterization of the glutamate-cysteine ligase modifier subunit Gclm(-/-) knockout mouse. Novel model system for a severely compromised oxidative stress response. J. Biol. Chem. 2002, 277, 49446–49452. [Google Scholar] [CrossRef]
- Broquist, H.P. Buthionine sulfoximine, an experimental tool to induce glutathione deficiency: Elucidation of glutathione and ascorbate in their role as antioxidants. Nutr. Rev. 1992, 50, 110–111. [Google Scholar] [CrossRef] [PubMed]
- Masukawa, T.; Sai, M.; Tochino, Y. Methods for depleting brain glutathione. Life Sci. 1989, 44, 417–424. [Google Scholar] [CrossRef]
- Górny, M.; Wnuk, A.; Kamińska, A.; Kamińska, K.; Chwatko, G.; Bilska-Wilkosz, A.; Iciek, M.; Kajta, M.; Rogóż, Z.; Lorenc-Koci, E. Glutathione deficiency and alterations in the sulfur amino acids homeostasis during early postnatal development as potential triggering factors for schizophrenia-like behavior in adult rats. Molecules 2019, 24, 4253. [Google Scholar] [CrossRef]
- Holmström, K.M.; Finkel, T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat. Rev. Mol. Cell Biol. 2014, 15, 411–421. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS sources in physiological and pathological conditions. Oxid. Med. Cell Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.J.; Forman, H.J.; Sevanian, A. Oxidants as stimulators of signal transduction. Free Radic. Biol. Med. 1997, 22, 269–285. [Google Scholar] [CrossRef]
- D’Autréaux, B.; Toledano, M.B. ROS as signalling molecules: Mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 2007, 8, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Maiorino, M.; Ursini, F. Signaling functions of reactive oxygen species. Biochemistry 2010, 49, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Ursini, F.; Maiorino, M. An overview of mechanisms of redox signaling. J. Mol. Cell Cardiol. 2014, 73, 2–9. [Google Scholar] [CrossRef]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef]
- Olguín-Albuerne, M.; Morán, J. Redox signaling mechanisms in nervous system development. Antioxid. Redox Signal. 2018, 28, 1603–1625. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.; Muñoz-Palma, E.; González-Billault, C. From birth to death: A role for reactive oxygen species in neuronal development. Semin. Cell Dev. Biol. 2018, 80, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Weidinger, A.; Kozlov, A.V. Biological activities of reactive oxygen and nitrogen species: Oxidative stress versus signal transduction. Biomolecules 2015, 5, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Bald, E.; Głowacki, R. 2-Chloro-1-methylquinolinium tetrafluoroborate as an effective and thiol specific uv-tagging reagent for liquid chromatography. J. Liq. Chrom. Rel. Technol. 2001, 24, 1323–1339. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Bondy, S.C.; Guo, S.X. Effect of ethanol treatment on indices of cumulative oxidative stress. Eur. J. Pharmacol. 1994, 270, 349–355. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohshi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Bald, E.; Chwatko, G.; Głowacki, R.; Kuśmierek, K. Analysis of plasma thiols by high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. A 2004, 1032, 109–115. [Google Scholar] [CrossRef]
- Kamińska, A.; Olejarz, P.; Borowczyk, K.; Głowacki, R.; Chwatko, G. Simultaneous determination of total homocysteine, cysteine, glutathione and N-acetylcysteine in brain homogenates by HPLC. J. Sep. Sci. 2018, 41, 3241–3249. [Google Scholar] [CrossRef]
- Borowczyk, K.; Chwatko, G.; Kubalczyk, P.; Jakubowski, H.; Kubalska, J.; Głowacki, R. Simultaneous determination of methionine and homocysteine by on-column derivatization with o-phtaldialdehyde. Talanta 2016, 161, 917–924. [Google Scholar] [CrossRef]
- Ogasawara, Y.; Isoda, S.; Tanabe, S. Tissue and subcellular distribution of bound and acid labile sulfur, and the enzymic capacity for sulfide production in the rat. Biol. Pharm. Bull. 1994, 17, 1535–1542. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Mavelli, I.; Rigo, A.; Federico, R.; Ciriolo, M.R.; Rotilio, G. Superoxide dismutase, glutathione peroxidase and catalase in developing rat brain. Biochem. J. 1982, 204, 535–540. [Google Scholar] [CrossRef]
- Ho, Y.S.; Magnenat, J.L.; Bronson, R.T.; Cao, J.; Gargano, M.; Sugawara, M.; Funk, C.D. Mice deficient in cellular glutathione peroxidase develop normally and show no increased sensitivity to hyperoxia. J. Biol. Chem. 1997, 272, 16644–16651. [Google Scholar] [CrossRef] [PubMed]
- Dringen, R. Metabolism and functions of glutathione in brain. Prog. Neurobiol. 2000, 62, 649–671. [Google Scholar] [CrossRef]
- Ulusu, N.N.; Sahilli, M.; Avci, A.; Canbolat, O.; Ozansoy, G.; Ari, N.; Bali, M.; Stefek, M.; Stolc, S.; Gajdosik, A.; et al. Pentose phosphate pathway, glutathione-dependent enzymes and antioxidant defense during oxidative stress in diabetic rodent brain and peripheral organs: Effects of stobadine and vitamin E. Neurochem. Res. 2003, 28, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Mori, N.; Yasutake, A.; Hirayama, K. Comparative study of activities in reactive oxygen species production/defense system in mitochondria of rat brain and liver, and their susceptibility to methylmercury toxicity. Arch Toxicol. 2007, 81, 769–776. [Google Scholar] [CrossRef]
- Dasuri, K.; Zhang, L.; Keller, J.N. Oxidative stress, neurodegeneration, and the balance of protein degradation and protein synthesis. Free Radic. Biol. Med. 2013, 62, 170–185. [Google Scholar] [CrossRef]
- Mailloux, R.J. Teaching the fundamentals of electron transfer reactions in mitochondria and the production and detection of reactive oxygen species. Redox Biol. 2015, 4, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Brand, M.D. Mitochondrial generation of superoxide and hydrogen peroxide as the source of mitochondrial redox signaling. Free Radic. Biol. Med. 2016, 100, 14–31. [Google Scholar] [CrossRef] [PubMed]
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.; Núñez, M.T.; González-Billault, C. Contribution of NADPH oxidase to the establishment of hippocampal neuronal polarity in culture. J. Cell Sci. 2015, 128, 2989–2995. [Google Scholar] [CrossRef] [PubMed]
- Nayernia, Z.; Jaquet, V.; Krause, K.H. New insights on NOX enzymes in the central nervous system. Antioxid. Redox Signal. 2014, 20, 2815–2837. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Bardaweel, S.K.; Gul, M.; Alzweiri, M.; Ishaqat, A.; ALSamat, H.A.; Bashatwah, R.M. Reactive oxygen species: The dual role in physiological and pathological conditions of the human body. Euroasian J. Med. 2018, 50, 193–201. [Google Scholar] [CrossRef]
- Le Belle, J.E.; Orozco, N.M.; Paucar, A.A.; Saxe, J.P.; Mottahedeh, J.; Pyle, A.D.; Wu, H.; Kornblum, H.I. Proliferative neural stem cells have high endogenous ROS levels that regulate self-renewal and neurogenesis in a PI3K/Akt-dependant manner. Cell Stem Cell. 2011, 8, 59–71. [Google Scholar] [CrossRef]
- Atmaca, G. Antioxidant effects of sulfur-containing amino acids. Yonsei Med. J. 2004, 45, 776–788. [Google Scholar] [CrossRef]
- Bin, P.; Huang, R.; Zhou, X. Oxidation resistance of the sulfur amino acids: Methionine and cysteine. Biomed. Res. Int. 2017, 9584932. [Google Scholar] [CrossRef]
- Martínez, Y.; Li, X.; Liu, G.; Bin, P.; Yan, W.; Más, D.; Valdivié, M.; Hu, C.A.; Ren, W.; Yin, Y. The role of methionine on metabolism, oxidative stress, and diseases. The role of methionine on metabolism, oxidative stress, and diseases. Amino Acids 2017, 49, 2091–2098. [Google Scholar] [CrossRef]
- Čolović, M.B.; Vasić, V.M.; Djurić, D.M.; Krstić, D.Z. Sulphur-containing amino acids: Protective role against free radicals and heavy metals. Curr. Med. Chem. 2018, 25, 324–335. [Google Scholar] [CrossRef]
- Brunell, D.; Weissbach, H.; Hodder, P.; Brot, N. A high-throughput screening compatible assay for activators and inhibitors of methionine sulfoxide reductase A. Assay Drug Dev. Technol. 2010, 8, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.L.; Mosoni, L.; Berlett, B.S.; Stadtman, E.R. Methionine residues as endogenous antioxidants in proteins. Proc. Natl. Acad. Sci. USA 1996, 93, 15036–15040. [Google Scholar] [CrossRef]
- Levine, R.L.; Berlett, B.S.; Moskovitz, J.; Mosoni, L.; Stadtman, E.R. Methionine residues may protect proteins from critical oxidative damage. Mech. Ageing Dev. 1999, 107, 323–332. [Google Scholar] [CrossRef]
- Lu, C.; Yao, S.; Lin, N. Studies on reactions of oxidizing sulfur-sulfur three-electron-bond complexes and reducing alpha-amino radicals derived from OH reaction with methionine in aqueous solution. Biochim. Biophys. Acta 2001, 1525, 89–96. [Google Scholar] [CrossRef]
- Xu, Y.Y.; Du, F.; Meng, B.; Xie, G.H.; Cao, J.; Fan, D.; Yu, H. Hepatic overexpression of methionine sulfoxide reductase A reduces atherosclerosis in apolipoprotein E-deficient mice. J. Lipid Res. 2015, 56, 1891–1900. [Google Scholar] [CrossRef]
- Weissbach, H.; Resnick, L.; Brot, N. Methionine sulfoxide reductases: History and cellular role in protecting against oxidative damage. Biochim. Biophys. Acta 2005, 1703, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Boschi-Muller, S.; Gand, A.; Branlant, G. The methionine sulfoxide reductases: Catalysis and substrate specificities. Arch. Biochem. Biophys. 2008, 474, 266–273. [Google Scholar] [CrossRef]
- Cudic, P.; Joshi, N.; Sagher, D.; Williams, B.T.; Stawikowski, M.J.; Weissbach, H. Identification of activators of methionine sulfoxide reductases A and B. Biochem. Biophys. Res. Commun. 2016, 469, 863–867. [Google Scholar] [CrossRef]
- Costa, M.Z.; Da Silva, T.M.; Flores, N.P.; Schmitz, F.; Da Silva Scherer, E.B.; Viau, C.M.; Saffi, J.; Barschak, A.G.; De Souza Wyse, A.T.; Spanevello, R.M.; et al. Methionine and methionine sulfoxide alter parameters of oxidative stress in the liver of young rats: In vitro and in vivo studies. Mol. Cell Biochem. 2013, 384, 21–28. [Google Scholar] [CrossRef]
- Campbell, K.; Vowinckel, J.; Keller, M.A.; Ralser, M. Methionine metabolism alters oxidative stress resistance via the pentose phosphate pathway. Antioxid. Redox Signal. 2016, 24, 543–547. [Google Scholar] [CrossRef]
- Singh, I. Mammalian peroxisomes: Metabolism of oxygen and reactive oxygen species. Ann. N. Y. Acad. Sci. 1996, 804, 612–627. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, J.T.; Brosnan, M.E. The sulfur-containing amino acids: An overview. J. Nutr. 2006, 136, 1636S–1640S. [Google Scholar] [CrossRef] [PubMed]
- Hinchman, C.A.; Ballatori, N. Glutathione-degrading capacities of liver and kidney in different species. Biochem. Pharmacol. 1990, 40, 131–135. [Google Scholar] [CrossRef]
- Włodek, P.; Sokołowska, M.; Smoleński, O.; Włodek, L. The γ-glutamyltransferase activity and non-protein sulfhydryl compounds levels in rat kidney of different age groups. Acta Biochim. Pol. 2002, 49, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Aebi, S.; Lauterburg, B.H. Divergent effects of intravenous GSH and cysteine on renal and hepatic GSH. Am. J. Physiol. 1992, 263, R348–R352. [Google Scholar] [CrossRef]
- Everett, S.A.; Folkes, L.K.; Wardman, P.; Asmus, K.D. Free-radical repair by a novel perthiol: Reversible hydrogen transfer and perthiyl radical formation. Free Radic. Res. 1994, 20, 387–400. [Google Scholar] [CrossRef]
- Anglada, J.M.; Crehuet, R.; Adhikari, S.; Francisco, J.S.; Xia, Y. Reactivity of hydropersulfides toward the hydroxyl radical unraveled: Disulfide bond cleavage, hydrogen atom transfer, and proton-coupled electron transfer. Phys. Chem. Chem. Phys. 2018, 20, 4793–4804. [Google Scholar] [CrossRef]
- Iciek, M.; Bilska-Wilkosz, A.; Górny, M. Sulfane sulfur—New findings on an old topic. Acta Biochim. Pol. 2019, 66, 533–544. [Google Scholar] [CrossRef]
- Bilska, A.; Dudek, M.; Iciek, M.; Kwiecień, I.; Sokołowska-Jeżewicz, M.; Filipek, B.; Włodek, L. Biological actions of lipoic acid associated with sulfane sulfur metabolism. Pharmacol. Rep. 2008, 60, 225–232. [Google Scholar]
- Chauvin, J.R.; Griesser, M.; Pratt, D.A. Hydropersulfides: H-atom transfer agents par excellence. J. Am. Chem. Soc. 2017, 139, 6484–6493. [Google Scholar] [CrossRef]
- Brannan, R.G. Reactive sulfur species act as prooxidants in liposomal and skeletal muscle model systems. J. Agric. Food Chem. 2010, 58, 3767–3771. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk-Pachel, D.; Iciek, M.; Wydra, K.; Nowak, E.; Górny, M.; Filip, M.; Włodek, L.; Lorenc-Koci, E. Cysteine metabolism and oxidative processes in the rat liver and kidney after acute and repeated cocaine treatment. PLoS ONE 2016, 11, e0147238. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.E. Di-γ-glutamylcystine as a substrate for glutathione reductase. Biochim. Biophys. Acta 1971, 242, 36–38. [Google Scholar] [CrossRef]
- Anderson, M.E.; Meister, A. Transport and direct utilization of gamma-glutamylcyst(e)ine for glutathione synthesis. Proc. Natl. Acad. Sci. USA 1983, 80, 707–711. [Google Scholar] [CrossRef] [PubMed]
- McBean, G.J.; Aslan, M.; Griffiths, H.R.; Torrão, R.C. Thiol redox homeostasis in neurodegenerative disease. Redox Biol. 2015, 5, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.D.; Sbodio, J.I.; Snyder, S.H. Cysteine metabolism in neuronal redox homeostasis. Trends Pharmacol. Sci. 2018, 39, 513–524. [Google Scholar] [CrossRef]
- McBean, G.J. The transsulfuration pathway: A source of cysteine for glutathione in astrocytes. Amino Acids 2012, 42, 199–205. [Google Scholar] [CrossRef]
- Kimura, H. Physiological role of hydrogen sulfide and polysulfide in the central nervous system. Neurochem. Int. 2013, 63, 492–497. [Google Scholar] [CrossRef]
- Camarero, J.; Sanchez, V.; O’Shea, E.; Green, A.R.; Colado, M. Studies, using in vivo microdialysis, on the effect of the dopamine uptake inhibitor GBR 12909 on 3,4-methylenedioxymethamphetamine (‘ecstasy’)-induced dopamine release and free radical formation in the mouse striatum. J. Neurochem. 2002, 81, 961–972. [Google Scholar] [CrossRef]
- Rougemont, M.; Do, K.Q.; Castagné, V. New model of glutathione deficit during development: Effect on lipid peroxidation in the rat brain. J. Neurosci. Res. 2002, 70, 774–783. [Google Scholar] [CrossRef]
| Tissue | Experimental Groups | ROS (nmol/g Tissue) | ANOVA Results |
|---|---|---|---|
| Peripheral Organs | |||
| Liver | control BSO GBR BSO + GBR | 48.6 ± 7.8 67.9 ± 9.0 38.7 ± 3.7 # 53.2 ± 3.6 | Effect of BSO F(1,24) = 6.789, p < 0.05 Effect of GBR F(1,24) = 3.556, p = 0.07 Interaction F(1,24) = 0.133, NS |
| Kidney | control BSO GBR BSO + GBR | 89.8 ± 9.7 49.7 ± 3.1 *** 38.0 ± 5.5 *** 42.8 ± 6.1 *** | Effect of BSO F(1,28) = 7.320, p < 0.05 Effect of GBR F(1,28) = 19.979, p < 0.001 Interaction F(1,28) = 11.776, p < 0.01 |
| Brain Structures | |||
| Prefrontal Cortex | control BSO GBR BSO + GBR | 38.7 ± 6.3 36.3 ± 6.6 39.6 ± 7.2 38.4 ± 7.6 | Effect of BSO F(1,24) = 0.073, NS Effect of GBR F(1,24) = 0.045, NS Interaction F(1,24) = 0.008, NS |
| Hippocampus | control BSO GBR BSO + GBR | 24.8 ± 1.1 23.2 ± 1.6 19.9 ± 1.1 23.8 ± 1.0 | Effect of BSO F(1,24) = 0.946, NS Effect of GBR F(1,24) = 3.246, p = 0.08 Interaction F(1,24) = 4.957, p < 0.05 |
| Striatum | control BSO GBR BSO + GBR | 28.2 ± 5.4 19.7 ± 1.9 18.4 ± 2.6 20.8 ± 2.9 | Effect of BSO F(1,24) = 0.768, NS Effect of GBR F(1,24) = 1.583, NS Interaction F(1,24) = 2.517, NS |
| Tissue | Experimental Groups | GSH | Cys | Met |
|---|---|---|---|---|
| Peripheral Organs | ||||
| Liver | control BSO GBR BSO + GBR | 100% ↓ by 66.7% ↑ by 18.7% ↓ by 76.4% | 100% ↔ ↓ tendency ↔ | 100% ↑ by 114% ↑ by 315% ↑ by 381.5% |
| Kidney | control BSO GBR BSO + GBR | 100% ↓ by 49% ↔ ↓ by 30% | 100% ↓ by 43% ↔ ↓ by 41% | 100% ↑ by 32% ↑ tendency ↑ by 27% |
| Brain Structures | ||||
| Prefrontal cortex | control BSO GBR BSO + GBR | 100% ↓ by 7% ↔ ↓ by 7% | 100% ↑ by 47.5% ↑ by 28.5% ↑ by 42.2% | 100% ↓ by 18.5% ↑ tendency ↓ by 19.7% |
| Hippocampus | control BSO GBR BSO + GBR | 100% ↔ ↑ by 8.5% ↔ | 100% ↑ by 34.9% ↑ tendency ↑ by 30.9% | 100% ↑ by 37.5% ↑ by 62.7% ↔ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Górny, M.; Bilska-Wilkosz, A.; Iciek, M.; Hereta, M.; Kamińska, K.; Kamińska, A.; Chwatko, G.; Rogóż, Z.; Lorenc-Koci, E. Alterations in the Antioxidant Enzyme Activities in the Neurodevelopmental Rat Model of Schizophrenia Induced by Glutathione Deficiency during Early Postnatal Life. Antioxidants 2020, 9, 538. https://doi.org/10.3390/antiox9060538
Górny M, Bilska-Wilkosz A, Iciek M, Hereta M, Kamińska K, Kamińska A, Chwatko G, Rogóż Z, Lorenc-Koci E. Alterations in the Antioxidant Enzyme Activities in the Neurodevelopmental Rat Model of Schizophrenia Induced by Glutathione Deficiency during Early Postnatal Life. Antioxidants. 2020; 9(6):538. https://doi.org/10.3390/antiox9060538
Chicago/Turabian StyleGórny, Magdalena, Anna Bilska-Wilkosz, Małgorzata Iciek, Marta Hereta, Kinga Kamińska, Adrianna Kamińska, Grażyna Chwatko, Zofia Rogóż, and Elżbieta Lorenc-Koci. 2020. "Alterations in the Antioxidant Enzyme Activities in the Neurodevelopmental Rat Model of Schizophrenia Induced by Glutathione Deficiency during Early Postnatal Life" Antioxidants 9, no. 6: 538. https://doi.org/10.3390/antiox9060538
APA StyleGórny, M., Bilska-Wilkosz, A., Iciek, M., Hereta, M., Kamińska, K., Kamińska, A., Chwatko, G., Rogóż, Z., & Lorenc-Koci, E. (2020). Alterations in the Antioxidant Enzyme Activities in the Neurodevelopmental Rat Model of Schizophrenia Induced by Glutathione Deficiency during Early Postnatal Life. Antioxidants, 9(6), 538. https://doi.org/10.3390/antiox9060538







