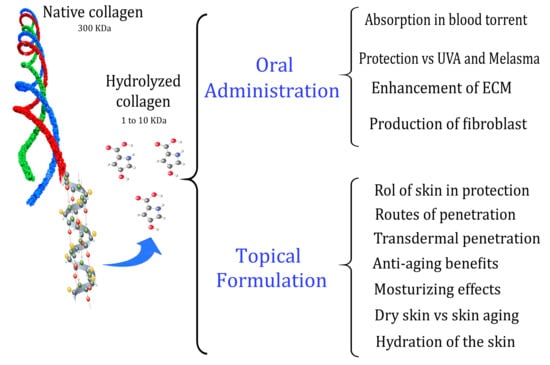
Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation
Abstract
1. Introduction
2. Topical Formulation
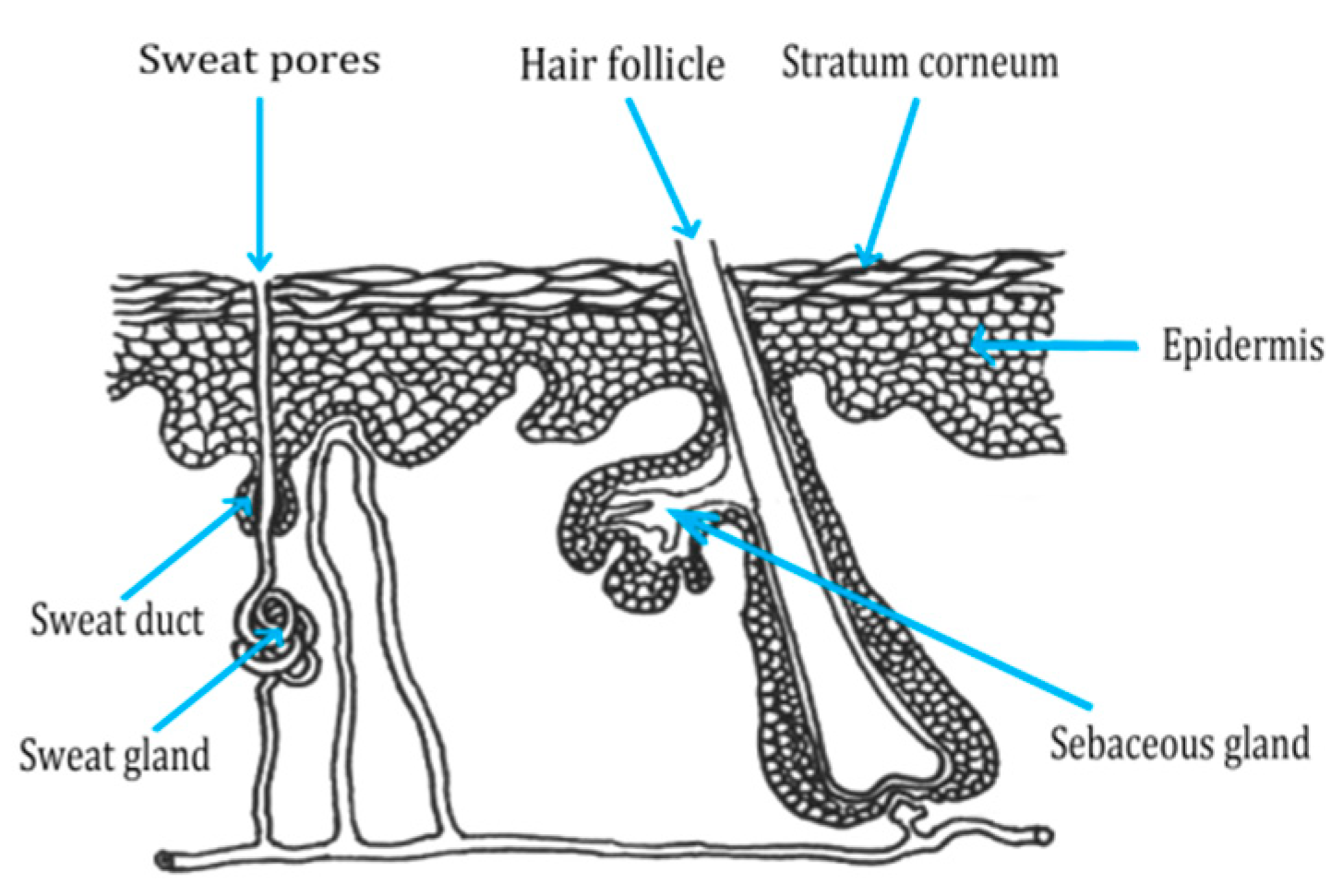
2.1. Role of the Skin in Protection
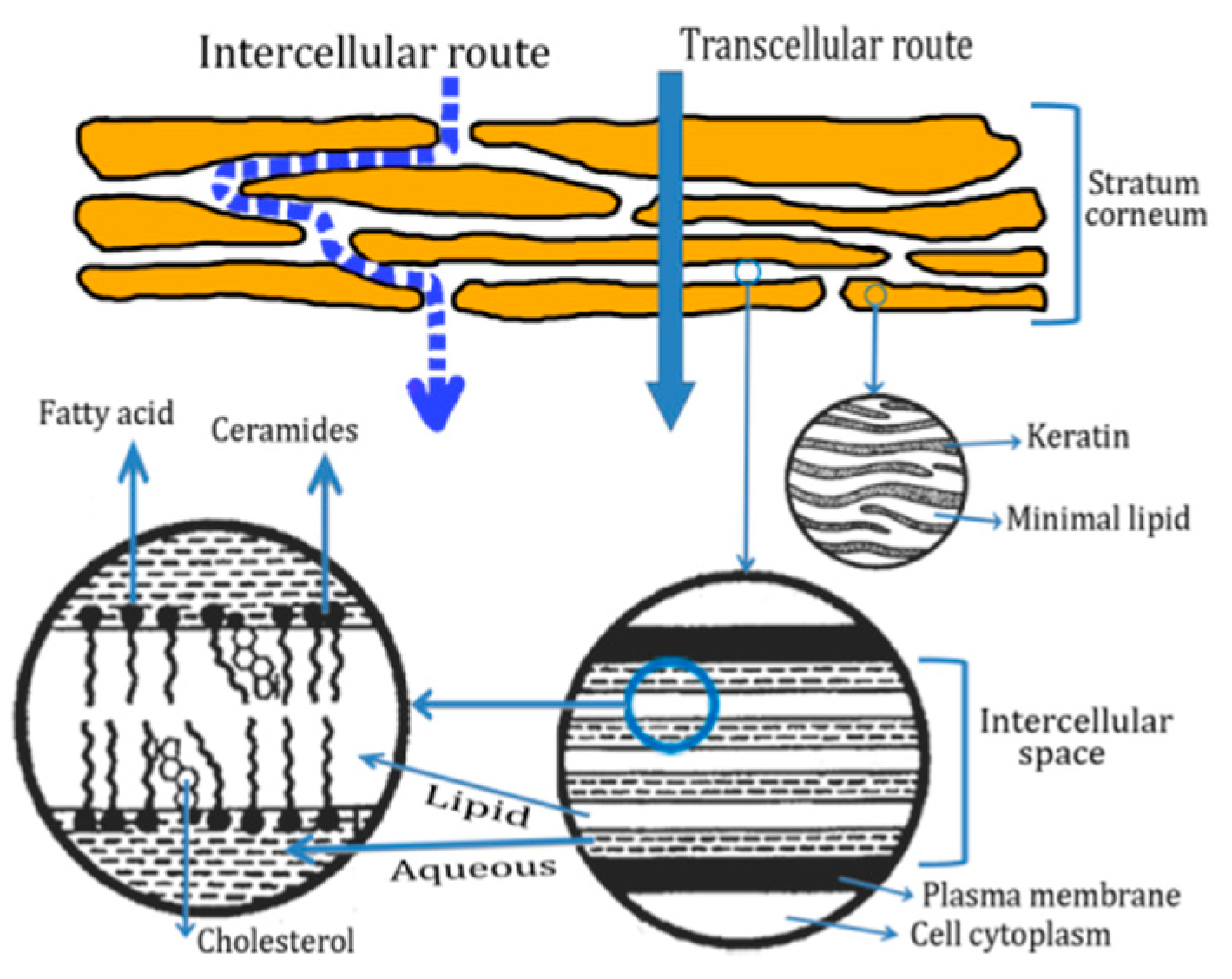
2.2. Routes of Peptide Penetration onto the Skin
2.3. Transdermal Penetration of HC on the Skin
2.4. Anti-Aging Benefits
2.5. Creams and Lotions with Mosturizing Action
2.6. Dry Skin as a Trigger for Skin Aging
2.7. Hydration of the Skin
3. Role of Hydrolyzed Collagen as an Antioxidant Ingredient
4. Oral Administration of HC
4.1. Absorption of HC into the Blood Torrent
4.2. Improvement of Skin Properties
4.3. Protection of the Skin Against UV and Melasma
4.4. Enhancement of Fibroblast Production and Extracellular Matrix of the Skin
5. Combination of Both Topical and Oral Intake
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| UVA | Ultraviolet A |
| UVB | Ultraviolet B |
| SC | Stratum Corneum |
| ECM | Extracellular Matrix |
| NMF | Natural Moisturizing Factor |
| TEWL | Transepidermal Water Loss |
| MMP | Matrix Metalloproteinases |
| kDa | KiloDalton |
| HC | Hydrolyzed Collagen |
References
- Sies, H. Oxidative Stress: Introductory Remarks; Academic Press: London, UK, 1985; pp. 1–8. [Google Scholar]
- Shin, J.-W.; Kwon, S.-H.; Choi, J.-Y.; Na, J.-I.; Huh, C.-H.; Choi, H.-R.; Park, K.-C. Molecular Mechanisms of Dermal Aging and Antiaging Approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef] [PubMed]
- Fuller, B. Role of PGE-2 and Other Inflammatory Mediators in Skin Aging and Their Inhibition by Topical Natural Anti-Inflammatories. Cosmetics 2019, 6, 6. [Google Scholar] [CrossRef]
- Levakov, A.; Vuckovic, N.; Dolai, M.; Kacanski, M.M.; Bozanic, S. Age-related skin changes. Med. Pregl. 2012, 65, 191–195. [Google Scholar] [CrossRef]
- Bocheva, G.; Slominski, R.M.; Slominski, A.T. Neuroendocrine Aspects of Skin Aging. Int. J. Mol. Sci. 2019, 20, 2798. [Google Scholar] [CrossRef] [PubMed]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Ageing Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Imokawa, G. Melanocyte Activation Mechanisms and Rational Therapeutic Treatments of Solar Lentigos. Int. J. Mol. Sci. 2019, 20, 3666. [Google Scholar] [CrossRef]
- Krutmann, J. How the sun ages our skin. The dermis as the driving force. Hautarzt Z. Dermatol. Venerol. Verwandte Geb. 2011, 62, 588–590. [Google Scholar] [CrossRef]
- Wenk, J.; Brenneisen, P.; Meewes, C.; Wlaschek, M.; Peters, T.; Blaudschun, R.; Ma, W.; Kuhr, L.; Schneider, L.; Scharffetter-Kochanek, K. UV-induced oxidative stress and photoaging. Curr. Probl. Dermatol. 2001, 29, 83–94. [Google Scholar]
- Naidoo, K.; Birch-Machin, M.A. Oxidative Stress and Ageing: The Influence of Environmental Pollution, Sunlight and Diet on Skin. Cosmetics 2017, 4, 4. [Google Scholar] [CrossRef]
- Rembiesa, J.; Ruzgas, T.; Engblom, J.; Holefors, A. The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims. Cosmetics 2018, 5, 4. [Google Scholar] [CrossRef]
- Wlaschek, M.; Tantcheva-Poor, I.; Naderi, L.; Ma, W.; Schneider, L.A.; Razi-Wolf, Z.; Schuller, J.; Scharffetter-Kochanek, K. Solar UV irradiation and dermal photoaging. J. Photochem. Photobiol. B Biol. 2001, 63, 41–51. [Google Scholar] [CrossRef]
- Avila Rodríguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef]
- Eastoe, J.E.; Leach, A.A. Chemical constitution of gelatin. In The Science and Technology of Gelatin; Ward, A.G., Courts, A., Eds.; Academic Press Inc.: London, UK, 1977; pp. 73–108. [Google Scholar]
- Li, G.Y.; Fukunaga, S.; Takenouchi, K.; Nakamura, F. Comparative study of the physiological properties of collagen, gelatin and collagen hydrolysate as cosmetic materials. Int. J. Cosmet. Sci. 2005, 27, 101–106. [Google Scholar] [CrossRef]
- Oshimura, E.; Sakamoto, K. Chapter 19—Amino Acids, Peptides, and Proteins. In Cosmetic Science and Technology; Sakamoto, K., Lochhead, R.Y., Maibach, H.I., Yamashita, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 285–303. [Google Scholar]
- Holmes, R.; Kirk, S.; Tronci, G.; Yang, X.; Wood, D. Influence of telopeptides on the structural and physical properties of polymeric and monomeric acid-soluble type I collagen. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 77, 823–827. [Google Scholar] [CrossRef]
- Daliri, E.B.-M.; Oh, D.H.; Lee, B.H. Bioactive Peptides. Foods 2017, 6, 32. [Google Scholar] [CrossRef]
- Langmaier, F.; Mládek, M.; Kolomazník, K.; Sukop, S. Isolation of elastin and collagen polypeptides from long cattle tendons as raw material for the cosmetic industry. Int. J. Cosmet. Sci. 2002, 24, 273–279. [Google Scholar] [CrossRef]
- Liu, D.; Nikoo, M.; Boran, G.; Zhou, P.; Regenstein, J.M. Collagen and gelatin. Annu. Rev. Food Sci. Technol. 2015, 6, 527–557. [Google Scholar] [CrossRef]
- Frei, V.; Perrier, E.; Orly, I.; Huc, A.; Augustin, C.; Damour, O. Activation of fibroblast metabolism in a dermal and skin equivalent model: A screening test for activity of peptides. Int. J. Cosmet. Sci. 1998, 20, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Schagen, S.K.; Zampeli, V.A.; Makrantonaki, E.; Zouboulis, C.C. Discovering the link between nutrition and skin aging. Derm. Endocrinol. 2012, 4, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Kligman, A. The future of cosmeceuticals: An interview with Albert Kligman, MD, PhD. Interview by Zoe Diana Draelos. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. 2005, 31, 890–891. [Google Scholar]
- Wertz, P.W. Lipids and the Permeability and Antimicrobial Barriers of the Skin. J. Lipids 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, V.T.; Ganju, P.; Ramkumar, A.; Grover, R.; Gokhale, R.S. Multifaceted pathways protect human skin from UV radiation. Nat. Chem. Biol. 2014, 10, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Wertz, P.W. Lipids and barrier function of the skin. Acta Derm. Venereol. Suppl. 2000, 208, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Feingold, K.R. Thematic review series: Skin lipids. The role of epidermal lipids in cutaneous permeability barrier homeostasis. J. Lipid Res. 2007, 48, 2531–2546. [Google Scholar] [CrossRef]
- Jones, L.A.; Smith, A.M. Tactile sensory system: Encoding from the periphery to the cortex. Wiley Interdiscip. Rev. Syst. Biol. Med. 2014, 6, 279–287. [Google Scholar] [CrossRef]
- Benson, H.A. Transdermal drug delivery: Penetration enhancement techniques. Curr. Drug Deliv. 2005, 2, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Barry, B.W. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2001, 14, 101–114. [Google Scholar] [CrossRef]
- Mojumdar, E.H.; Pham, Q.D.; Topgaard, D.; Sparr, E. Skin hydration: Interplay between molecular dynamics, structure and water uptake in the stratum corneum. Sci. Rep. 2017, 7, 15712. [Google Scholar] [CrossRef] [PubMed]
- Chvapli, M.; Eckmayer, Z. Role of proteins in cosmetics. Int. J. Cosmet. Sci. 1985, 7, 41–49. [Google Scholar] [CrossRef] [PubMed]
- CIR. 7 Final Report on the Safety Assessment of Hydrolyzed Collagen. Cosmetic Ingredient Review. J. Am. Coll. Toxicol. 1985, 4, 199–221. [Google Scholar] [CrossRef]
- CIR. Annual review of cosmetic ingredient safety assessments-2004/2005. Int. J. Toxicol. 2006, 25, 1–89. [Google Scholar] [CrossRef] [PubMed]
- CIR. Safety Assessment of Skin and Connective Tissue-Derived Proteins and Peptides as Used in Cosmetics; Cosmetic Ingredient Review: Washington, DC, USA, 2017; pp. 1–28. [Google Scholar]
- Lintner, K. Peptides and proteins. In Cosmetic Dermatology, Products and Procedures; Draelos, Z.D., Ed.; Wiley-Blackwell: West Sussex, UK, 2010; pp. 292–301. [Google Scholar]
- Ranade, V.V. Drug delivery systems. 6. Transdermal drug delivery. J. Clin. Pharmacol. 1991, 31, 401–418. [Google Scholar] [CrossRef] [PubMed]
- Gorouhi, F.; Maibach, H.I. Role of topical peptides in preventing or treating aged skin. Int. J. Cosmet. Sci. 2009, 31, 327–345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Pan, D.; Dong, Y.; Su, W.; Su, H.; Wei, X.; Yang, C.; Jing, L.; Tang, X.; Li, X.; et al. Transdermal permeation effect of collagen hydrolysates of deer sinew on mouse skin, ex vitro, and antioxidant activity, increased type I collagen secretion of percutaneous proteins in NIH/3T3 cells. J. Cosmet. Dermatol. 2019. [Google Scholar] [CrossRef]
- Secchi, G. Role of protein in cosmetics. Clin. Dermatol. 2008, 26, 321–325. [Google Scholar] [CrossRef]
- Sionkowska, A.; Skrzyński, S.; Śmiechowski, K.; Kołodziejczak, A. The review of versatile application of collagen. Polym. Adv. Technol. 2017, 28, 4–9. [Google Scholar] [CrossRef]
- Chai, H.J.; Li, J.H.; Huang, H.N.; Li, T.L.; Chan, Y.L.; Shiau, C.Y.; Wu, C.J. Effects of sizes and conformations of fish-scale collagen peptides on facial skin qualities and transdermal penetration efficiency. J. Biomed. Biotechnol. 2010, 2010, 757301. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.J.; Al-Bader, T.; Kerrigan, D.; Hickey, S.; Laloeuf, A.; Rawlings, A.V. Synergistic action of a triple peptide complex on an essential extra-cellular matrix protein exhibits significant anti-aging benefits. J. Cosmet. Dermatol. 2010, 9, 108–116. [Google Scholar] [CrossRef]
- Trookman, N.S.; Rizer, R.L.; Ford, R.; Ho, E.; Gotz, V. Immediate and Long-term Clinical Benefits of a Topical Treatment for Facial Lines and Wrinkles. J. Clin. Aesthet. Derm. 2009, 2, 38–43. [Google Scholar]
- Swatschek, D.; Schatton, W.; Kellermann, J.; Muller, W.E.; Kreuter, J. Marine sponge collagen: Isolation, characterization and effects on the skin parameters surface-pH, moisture and sebum. Eur. J. Pharm. Biopharm. Off. J. Arb. Pharm. Verfahr. 2002, 53, 107–113. [Google Scholar] [CrossRef]
- Berardesca, E.; Abril, E.; Serio, M.; Cameli, N. Effects of topical gluco-oligosaccharide and collagen tripeptide F in the treatment of sensitive atopic skin. Int. J. Cosmet. Sci. 2009, 31, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Lupo, M.P.; Cole, A.L. Cosmeceutical peptides. Dermatol. Ther. 2007, 20, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Bode, W.; Fernandez-Catalan, C.; Grams, F.; Gomis-Ruth, F.X.; Nagase, H.; Tschesche, H.; Maskos, K. Insights into MMP-TIMP interactions. Ann. N. Y. Acad. Sci. 1999, 878, 73–91. [Google Scholar] [CrossRef]
- Xhauflaire-Uhoda, E.; Fontaine, K.; Piérard, G.E. Kinetics of moisturizing and firming effects of cosmetic formulations. Int. J. Cosmet. Sci. 2008, 30, 131–138. [Google Scholar] [CrossRef]
- Sethi, A.; Kaur, T.; Malhotra, S.; Gambhir, M. Moisturizers: The slippery road. Indian J. Dermatol. 2016, 61, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Flynn, T.C.; Petros, J.; Clark, R.E.; Viehman, G.E. Dry skin and moisturizers. Clin. Dermatol. 2001, 19, 387–392. [Google Scholar] [CrossRef]
- Kraft, J.N.; Lynde, C.W. Moisturizers: What they are and a practical approach to product selection. Ski. Ther. Lett. 2005, 10, 1–8. [Google Scholar]
- Spada, F.; Barnes, T.M.; Greive, K.A. Skin hydration is significantly increased by a cream formulated to mimic the skin’s own natural moisturizing systems. Clin. Cosmet. Investig. Dermatol. 2018, 11, 491–497. [Google Scholar] [CrossRef]
- Ciszek, A. Variability of skin pH after the use of different collagen gels. J. Cosmet. Dermatol. 2017, 16, 531–536. [Google Scholar] [CrossRef]
- Sandilands, A.; Sutherland, C.; Irvine, A.D.; McLean, W.H. Filaggrin in the frontline: Role in skin barrier function and disease. J. Cell Sci. 2009, 122, 1285–1294. [Google Scholar] [CrossRef]
- Zanna, N. Defeating dry skin: Treatments, topical ingredients and the role of nutrition. J. Aesthet. Nurs. 2015, 4, 478–485. [Google Scholar] [CrossRef]
- Duplan, H.; Nocera, T. Skin hydration and hydrating products. Ann. Dermatol. Venereol. 2018, 145, 376–384. [Google Scholar] [CrossRef]
- Gioia, F.; Celleno, L. The dynamics of transepidermal water loss (TEWL) from hydrated skin. Ski. Res. Technol. 2002, 8, 178–186. [Google Scholar] [CrossRef]
- Bernengo, J.-C.; de Rigal, J. Physical Methods to Measure Stratum Corneum Water Content In Vivo. In Agache’s Measuring the Skin: Non-Invasive Investigations, Physiology, Normal Constants; Humbert, P., Fanian, F., Maibach, H.I., Agache, P., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 299–340. [Google Scholar]
- Milani, M.; Sparavigna, A. The 24-hour skin hydration and barrier function effects of a hyaluronic 1%, glycerin 5%, and Centella asiatica stem cells extract moisturizing fluid: An intra-subject, randomized, assessor-blinded study. Clin. Cosmet. Investig. Dermatol. 2017, 10, 311–315. [Google Scholar] [CrossRef]
- Voegeli, R.; Gierschendorf, J.; Summers, B.; Rawlings, A.V. Facial skin mapping: From single point bio-instrumental evaluation to continuous visualization of skin hydration, barrier function, skin surface pH, and sebum in different ethnic skin types. Int. J. Cosmet. Sci. 2019, 41, 411–424. [Google Scholar] [CrossRef]
- Guzmán-Alonso, M.; Cortazár, T.M. Water content at different skin depths and the influence of moisturizing formulations. Househ. Pers. Care Today 2016, 11, 35–40. [Google Scholar]
- Bielfeldt, S.; Schoder, V.; Ely, U.; Van Der Pol, A.; De Sterke, J.; Wilhelm, K.-P. Assessment of human stratum corneum thickness and its barrier properties by in-vivo confocal Raman spectroscopy. Int. J. Cosmet. Sci. 2009, 31, 479–480. [Google Scholar] [CrossRef]
- Verdier-Sevrain, S.; Bonte, F. Skin hydration: A review on its molecular mechanisms. J. Cosmet. Dermatol. 2007, 6, 75–82. [Google Scholar] [CrossRef]
- Gniadecka, M.; Faurskov Nielsen, O.; Christensen, D.H.; Wulf, H.C. Structure of water, proteins, and lipids in intact human skin, hair, and nail. J. Investig. Dermatol. 1998, 110, 393–398. [Google Scholar] [CrossRef]
- Waller, J.M.; Maibach, H.I. Age and skin structure and function, a quantitative approach (II): Protein, glycosaminoglycan, water, and lipid content and structure. Ski. Res. Technol. 2006, 12, 145–154. [Google Scholar] [CrossRef]
- Qassem, M.; Kyriacou, P. Review of Modern Techniques for the Assessment of Skin Hydration. Cosmetics 2019, 6, 19. [Google Scholar] [CrossRef]
- Egawa, M. In vivo simultaneous measurement of urea and water in the human stratum corneum by diffuse-reflectance near-infrared spectroscopy. Ski. Res. Technol. 2009, 15, 195–199. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Q.; Mao, G.; Conroy, O.; Pyatski, Y.; Fevola, M.J.; Cula, G.O.; Maitra, P.; Mendelsohn, R.; Flach, C.R. Novel confocal Raman microscopy method to investigate hydration mechanisms in human skin. Skin. Res. Technol. 2019, 25, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Yeo, W.H.; Liu, Y.; Rogers, J.A. Epidermal differential impedance sensor for conformal skin hydration monitoring. Biointerphases 2012, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Webb, R.C.; Bonifas, A.P.; Behnaz, A.; Zhang, Y.; Yu, K.J.; Cheng, H.; Shi, M.; Bian, Z.; Liu, Z.; Kim, Y.-S.; et al. Ultrathin conformal devices for precise and continuous thermal characterization of human skin. Nat. Mater. 2013, 12, 938–944. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Shi, Y.; Webb, R.C.; Ma, Y.; Bastien, P.; Crawford, K.E.; Wang, A.; Feng, X.; Manco, M.; Kurniawan, J.; et al. Multimodal epidermal devices for hydration monitoring. Microsyst. Amp Nanoeng. 2017, 3, 17014. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.-A.; Ahn, J.-W.; Chun, I.-K.; Kim, H.-J. Development of a Method for the Determination of Human Skin Moisture Using a Portable Near-Infrared System. Anal. Chem. 2001, 73, 4964–4971. [Google Scholar] [CrossRef] [PubMed]
- Fluhr, J.W.; Darlenski, R. Transepidermal Water Loss (TEWL). In Non Invasive Diagnostic Techniques in Clinical Dermatology; Berardesca, E., Maibach, H.I., Wilhelm, K.-P., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 353–356. [Google Scholar]
- Lopez-Alarcon, C.; Denicola, A. Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal. Chim. Acta 2013, 763, 1–10. [Google Scholar] [CrossRef]
- Hermund, D.B. Antioxidant properties of seaweed-derived substances. In Bioactive Seaweeds for Food Applications, Natural Ingredients for Healty Diets; Qin, Y., Ed.; Academic Press: London, UK, 2018; pp. 201–222. [Google Scholar]
- Silva, S.; Ferreira, M.; Oliveira, A.S.; Magalhães, C.; Sousa, M.E.; Pinto, M.; Sousa Lobo, J.M.; Almeida, I.F. Evolution of the use of antioxidants in anti-ageing cosmetics. Int. J. Cosmet. Sci. 2019, 41, 378–386. [Google Scholar] [CrossRef]
- Esfandi, R.; Walters, M.E.; Tsopmo, A. Antioxidant properties and potential mechanisms of hydrolyzed proteins and peptides from cereals. Heliyon 2019, 5, e01538. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Wijesekara, I. Development and biological activities of marine-derived bioactive peptides: A review. J. Funct. Foods 2010, 2, 1–9. [Google Scholar] [CrossRef]
- Hettiarachchy, N.S.; Glenn, K.C.; Gnanasambandam, R.; Johnson, M.G. Natural Antioxidant Extract from Fenugreek (Trigonella foenumgraecum) for Ground Beef Patties. J. Food Sci. 1996, 61, 516–519. [Google Scholar] [CrossRef]
- León-López, A.; Fuentes-Jiménez, L.; Hernández-Fuentes, A.D.; Campos-Montiel, R.G.; Aguirre-Álvarez, G. Hydrolysed Collagen from Sheepskins as a Source of Functional Peptides with Antioxidant Activity. Int. J. Mol. Sci. 2019, 20, 3931. [Google Scholar] [CrossRef] [PubMed]
- Mendis, E.; Rajapakse, N.; Byun, H.G.; Kim, S.K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.J.; Jung, W.K.; Byun, H.G.; Kim, S.K. Protective effect of an antioxidative peptide purified from gastrointestinal digests of oyster, Crassostrea gigas against free radical induced DNA damage. Bioresour. Technol. 2008, 99, 3365–3371. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Mendis, E.; Jung, W.K.; Je, J.Y.; Kim, S.K. Purification of a radical scavenging peptide from fermented mussel sauce and its antioxidant properties. Food Res. Int. 2005, 38, 175–182. [Google Scholar] [CrossRef]
- Kim, S.Y.; Je, J.Y.; Kim, S.K. Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J. Nutr. Biochem. 2007, 18, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Je, J.Y.; Qian, Z.J.; Lee, S.H.; Byun, H.G.; Kim, S.K. Purification and antioxidant properties of bigeye tuna (Thunnus obesus) dark muscle peptide on free radical-mediated oxidative systems. J. Med. Food 2008, 11, 629–637. [Google Scholar] [CrossRef]
- Slizyte, R.; Mozuraityte, R.; Martinez-Alvarez, O.; Falch, E.; Fouchereau-Peron, M.; Rustad, T. Functional, bioactive and antioxidative properties of hydrolysates obtained from cod (Gadus morhua) backbones. Process Biochem. 2009, 44, 668–677. [Google Scholar] [CrossRef]
- Thinsilakul, Y.; Benjakul, S.; Shahidi, F. Antioxidative activity of protein hydrolysate from round scad muscle using alcalase and flavourzyme. J. Food Biochem. 2007, 31, 266–287. [Google Scholar] [CrossRef]
- Amarowicz, R.; Shahidi, F. Antioxidant activity of peptide fractions of capelin protein hydrolysates. Food Chem. 1997, 58, 355–359. [Google Scholar] [CrossRef]
- Wu, H.C.; Chen, H.M.; Shiau, C.Y. Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res. Int. 2003, 36, 949–957. [Google Scholar] [CrossRef]
- Cho, S.-S.; Hyo-ku, L.; Yeon, Y.C.; Kim, M.J.; Soo, S.E.; Eun-Hwa, S.; Myoung-Gun, C.; Lim, J.D. Isolation and characterization of bioactive peptides from Hwangtae (yellowish dried Alaska pollack) protein hydrolysate. J. Food Sci. Nutr. 2008, 13, 196–203. [Google Scholar]
- Jun, S.-Y.; Park, P.-J.; Jung, W.-K.; Kim, S.-K. Purification and characterization of an antioxidative peptide from enzymatic hydrolysate of yellowfin sole (Limanda aspera) frame protein. Eur. Food Res. Technol. 2004, 219, 20–26. [Google Scholar]
- Klompong, V.; Benjakul, S.; Yachai, M.; Visessanguan, W.; Shahidi, F.; Hayes, K.D. Amino acid composition and antioxidative peptides from protein hydrolysates of yellow stripe Trevally (Selaroides leptolepis). J. Food Sci. 2009, 74, C126–C133. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Wijesekara, I.; Park, E.Y.; Matsumura, Y.; Nakamura, Y.; Sato, K. Proteins and peptides as antioxidants. In Bioactive Food Proteins and Peptides, Applications in Human Health; Hettiarachchy, N.S., Sato, K., Marshall, M.R., Kannan, A., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 97–116. [Google Scholar]
- Marini, A. Beauty from the inside. Does it really work? Hautarzt Z. Dermatol. Venerol. Verwandte Geb. 2011, 62, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Draelos, Z.D. Aging skin: The role of diet: Facts and controversies. Clin. Dermatol. 2013, 31, 701–706. [Google Scholar] [CrossRef]
- Taeymans, J.; Clarys, P.; Barel, A.O. Use of Food Supplements as Nutricosmetics in Health and Fitness: A Review. In Handbook of Cosmetic Science and Technology, 4th ed.; Barel, A.O., Paye, M., Maibach, H.I., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 583–596. [Google Scholar]
- Iwai, K.; Hasegawa, T.; Taguchi, Y.; Morimatsu, F.; Sato, K.; Nakamura, Y.; Higashi, A.; Kido, Y.; Nakabo, Y.; Ohtsuki, K. Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates. J. Agric. Food Chem. 2005, 53, 6531–6536. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Morifuji, M.; Ohara, H.; Matsumoto, H.; Takeuchi, Y.; Sato, K. Hydroxyproline-containing dipeptides and tripeptides quantified at high concentration in human blood after oral administration of gelatin hydrolysate. Int. J. Food Sci. Nutr. 2010, 61, 52–60. [Google Scholar] [CrossRef]
- Ohara, H.; Matsumoto, H.; Ito, K.; Iwai, K.; Sato, K. Comparison of quantity and structures of hydroxyproline-containing peptides in human blood after oral ingestion of gelatin hydrolysates from different sources. J. Agric. Food Chem. 2007, 55, 1532–1535. [Google Scholar] [CrossRef]
- Yazaki, M.; Ito, Y.; Yamada, M.; Goulas, S.; Teramoto, S.; Nakaya, M.A.; Ohno, S.; Yamaguchi, K. Oral Ingestion of Collagen Hydrolysate Leads to the Transportation of Highly Concentrated Gly-Pro-Hyp and Its Hydrolyzed Form of Pro-Hyp into the Bloodstream and Skin. J. Agric. Food Chem. 2017, 65, 2315–2322. [Google Scholar] [CrossRef] [PubMed]
- Skov, K.; Oxfeldt, M.; Thogersen, R.; Hansen, M.; Bertram, H.C. Enzymatic Hydrolysis of a Collagen Hydrolysate Enhances Postprandial Absorption Rate-A Randomized Controlled Trial. Nutrients 2019, 11, 1064. [Google Scholar] [CrossRef]
- López-Morales, C.A.; Vázquez-Leyva, S.; Vallejo-Castillo, L.; Carballo-Uicab, G.; Muñoz-García, L.; Herbert-Pucheta, J.E.; Zepeda-Vallejo, L.G.; Velasco-Velázquez, M.; Pavón, L.; Pérez-Tapia, S.M.; et al. Determination of Peptide Profile Consistency and Safety of Collagen Hydrolysates as Quality Attributes. J. Food Sci. 2019, 84, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Mizushige, T.; Jinno, S.; Sugihara, F.; Inoue, N.; Tanaka, H.; Kabuyama, Y. Absorption and metabolism of orally administered collagen hydrolysates evaluated by the vascularly perfused rat intestine and liver in situ. Biomed. Res. (Tokyo Jpn.) 2018, 39, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Hayasaka, F.; Deguchi, K.; Okudera, T.; Furusawa, T.; Sakai, Y. Absorption and plasma kinetics of collagen tripeptide after peroral or intraperitoneal administration in rats. Biosci. Biotechnol. Biochem. 2015, 79, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Genovese, L.; Corbo, A.; Sibilla, S. An Insight into the Changes in Skin Texture and Properties following Dietary Intervention with a Nutricosmeceutical Containing a Blend of Collagen Bioactive Peptides and Antioxidants. Ski. Pharmacol. Physiol. 2017, 30, 146–158. [Google Scholar] [CrossRef]
- Inoue, N.; Sugihara, F.; Wang, X. Ingestion of bioactive collagen hydrolysates enhance facial skin moisture and elasticity and reduce facial ageing signs in a randomised double-blind placebo-controlled clinical study. J. Sci. Food Agric. 2016, 96, 4077–4081. [Google Scholar] [CrossRef]
- Sugihara, F.; Inoue, N. Clinical effects of collagen hydrolysates ingestion on UV-induced pigmented spots of human skin: A preliminary study. Health Sci. 2012, 28, 153–156. [Google Scholar]
- Ito, N.; Seki, S.; Ueda, F. Effects of Composite Supplement Containing Collagen Peptide and Ornithine on Skin Conditions and Plasma IGF-1 Levels-A Randomized, Double-Blind, Placebo-Controlled Trial. Mar Drugs 2018, 16, 482. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H. Clinical effects of fish type I collagen hydrolysate on skin properties. ITE Lett. Batter. New Technol. Med. 2006, 7, 386–390. [Google Scholar]
- Schwartz, S.R.; Park, J. Ingestion of BioCell Collagen®, a novel hydrolyzed chicken sternal cartilage extract; enhanced blood microcirculation and reduced facial aging signs. Clin. Interv. Aging 2012, 7, 267–273. [Google Scholar] [PubMed]
- Petersen Vitello Kalil, C.L.; Campos, V.; Cignachi, S.; Favaro Izidoro, J.; Prieto Herman Reinehr, C.; Chaves, C. Evaluation of cutaneous rejuvenation associated with the use of ortho-silicic acid stabilized by hydrolyzed marine collagen. J. Cosmet. Dermatol. 2018, 17, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Koyama, Y.; Nomura, Y. Effects of collagen peptide ingestion on UV-B-induced skin damage. Biosci. Biotechnol. Biochem. 2009, 73, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Gui, M.; Kan, J.; Qu, D.; Chen, Y.; Luo, R.; Liu, Y.; Du, J. Instrumental Evaluation of the Depigmenting Efficacy of an Oral Supplementation Containing Peptides and Chrysanthemum Extract for the Treatment of Melasma. Cosmetics 2017, 4, 42. [Google Scholar] [CrossRef]
- Lee, H.-J.; Jang, H.-L.; Ahn, D.-K.; Kim, H.-J.; Jeon, H.Y.; Seo, D.B.; Lee, J.-H.; Choi, J.K.; Kang, S.-S. Orally administered collagen peptide protects against UVB-induced skin aging through the absorption of dipeptide forms, Gly-Pro and Pro-Hyp. Biosci. Biotechnol. Biochem. 2019, 83, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Varani, J.; Voorhees, J.J. Looking older: Fibroblast collapse and therapeutic implications. Arch. Dermatol. 2008, 144, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Edgar, S.; Hopley, B.; Genovese, L.; Sibilla, S.; Laight, D.; Shute, J. Effects of collagen-derived bioactive peptides and natural antioxidant compounds on proliferation and matrix protein synthesis by cultured normal human dermal fibroblasts. Sci. Rep. 2018, 8, 10474. [Google Scholar] [CrossRef] [PubMed]
- Rippa, A.L.; Kalabusheva, E.P.; Vorotelyak, E.A. Regeneration of Dermis: Scarring and Cells Involved. Cells 2019, 8, 607. [Google Scholar] [CrossRef] [PubMed]
- Asai, T.T.; Oikawa, F.; Yoshikawa, K.; Inoue, N.; Sato, K. Food-Derived Collagen Peptides, Prolyl-Hydroxyproline (Pro-Hyp), and Hydroxyprolyl-Glycine (Hyp-Gly) Enhance Growth of Primary Cultured Mouse Skin Fibroblast Using Fetal Bovine Serum Free from Hydroxyprolyl Peptide. Int. J. Mol. Sci. 2019, 21, 229. [Google Scholar] [CrossRef]
- Marcos-Garcés, V.; Molina Aguilar, P.; Bea Serrano, C.; García Bustos, V.; Benavent Seguí, J.; Ferrández Izquierdo, A.; Ruiz-Saurí, A. Age-related dermal collagen changes during development, maturation and ageing—A morphometric and comparative study. J. Anat. 2014, 225, 98–108. [Google Scholar] [CrossRef]
- Proksch, E.; Schunck, M.; Zague, V.; Segger, D.; Degwert, J.; Oesser, S. Oral intake of specific bioactive collagen peptides reduces skin wrinkles and increases dermal matrix synthesis. Ski. Pharmacol. Physiol. 2014, 27, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kielty, C.M.; Sherratt, M.J.; Shuttleworth, C.A. Elastic fibres. J. Cell Sci. 2002, 115, 2817–2828. [Google Scholar] [PubMed]
- Zague, V.; de Freitas, V.; da Costa Rosa, M.; de Castro, G.A.; Jaeger, R.G.; Machado-Santelli, G.M. Collagen hydrolysate intake increases skin collagen expression and suppresses matrix metalloproteinase 2 activity. J. Med. Food 2011, 14, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Pei, X.; Zhang, Z.; Wang, N.; Wang, J.; Li, Y. The protective effects of long-term oral administration of marine collagen hydrolysate from chum salmon on collagen matrix homeostasis in the chronological aged skin of Sprague-Dawley male rats. J. Food Sci. 2010, 75, H230–H238. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, Y.; Song, H.; He, J.; Li, G.; Zheng, Y.; Li, B. Collagen peptides promote photoaging skin cell repair by activating the TGF-beta/Smad pathway and depressing collagen degradation. Food Funct. 2019, 10, 6121–6134. [Google Scholar] [CrossRef]
- Borumand, M.; Sibilla, S. Daily consumption of the collagen supplement Pure Gold Collagen® reduces visible signs of aging. Clin. Interv. Aging 2014, 9, 1747–1758. [Google Scholar] [CrossRef]
- Asserin, J.; Lati, E.; Shioya, T.; Prawitt, J. The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: Evidence from an ex vivo model and randomized, placebo-controlled clinical trials. J. Cosmet. Dermatol. 2015, 14, 291–301. [Google Scholar] [CrossRef]
- Maia Campos, P.; Melo, M.O.; Siqueira Cesar, F.C. Topical application and oral supplementation of peptides in the improvement of skin viscoelasticity and density. J. Cosmet. Dermatol. 2019, 18, 1693–1699. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguirre-Cruz, G.; León-López, A.; Cruz-Gómez, V.; Jiménez-Alvarado, R.; Aguirre-Álvarez, G. Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation. Antioxidants 2020, 9, 181. https://doi.org/10.3390/antiox9020181
Aguirre-Cruz G, León-López A, Cruz-Gómez V, Jiménez-Alvarado R, Aguirre-Álvarez G. Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation. Antioxidants. 2020; 9(2):181. https://doi.org/10.3390/antiox9020181
Chicago/Turabian StyleAguirre-Cruz, Gabriel, Arely León-López, Verónica Cruz-Gómez, Rubén Jiménez-Alvarado, and Gabriel Aguirre-Álvarez. 2020. "Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation" Antioxidants 9, no. 2: 181. https://doi.org/10.3390/antiox9020181
APA StyleAguirre-Cruz, G., León-López, A., Cruz-Gómez, V., Jiménez-Alvarado, R., & Aguirre-Álvarez, G. (2020). Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation. Antioxidants, 9(2), 181. https://doi.org/10.3390/antiox9020181