Vitamin C and Cardiovascular Disease: An Update
Abstract
1. Physiology of Vitamin C
2. Genetics and Diet
3. Pharmacokinetics and Bioavailability of Vitamin C
4. Cardiovascular Effects of Vitamin C: Pre-Clinical Evidence
5. Vitamin C and Cardiac Disease
6. Vitamin C and Vascular Disease
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kyle:, R.A.; Shampo, M.A. Albert Szent-Gyorgy—Nobel laureate. Mayo Clin. Proc. 2000, 75, 722. [Google Scholar] [CrossRef]
- Szent-Gyorgyi, A. The Identification of Vitamin C. Science 1938, 87, 214–215. [Google Scholar] [CrossRef] [PubMed]
- Zilva, S.S. The Antiscorbutic Fraction of Lemon Juice. I. Biochem. J. 1924, 18, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, K.J. The discovery of vitamin C. Ann. Nutr. Metab. 2012, 61, 259–264. [Google Scholar] [CrossRef]
- De Tullio, M.C. Beyond the antioxidant: The double life of vitamin C. Subcell. Biochem. 2012, 56, 49–65. [Google Scholar]
- Frei, B.; England, L.; Ames, B.N. Ascorbate is an outstanding antioxidant in human blood plasma. Proc. Natl. Acad. Sci. USA 1989, 86, 6377–6381. [Google Scholar] [CrossRef]
- Frei, B.; Stocker, R.; England, L.; Ames, B.N. Ascorbate: The most effective antioxidant in human blood plasma. Adv. Exp. Med. Biol. 1990, 264, 155–163. [Google Scholar]
- Wyckelsma, V.L.; Venckunas, T.; Brazaitis, M.; Gastaldello, S.; Snieckus, A.; Eimantas, N.; Baranauskiene, N.; Subocius, A.; Skurvydas, A.; Paasuke, M.; et al. Vitamin C and E Treatment Blunts Sprint Interval Training-Induced Changes in Inflammatory Mediator-, Calcium-, and Mitochondria-Related Signaling in Recreationally Active Elderly Humans. Antioxidants 2020, 9, 879. [Google Scholar] [CrossRef]
- Jacob, R.A.; Sotoudeh, G. Vitamin C function and status in chronic disease. Nutr. Clin. Care 2002, 5, 66–74. [Google Scholar] [CrossRef]
- Van den Berg, J.J.; Kuypers, F.A.; Roelofsen, B.; den Kamp, J.A.O. The cooperative action of vitamins E and C in the protection against peroxidation of parinaric acid in human erythrocyte membranes. Chem. Phys. Lipids 1990, 53, 309–320. [Google Scholar] [CrossRef]
- Johnston, C.S.; Meyer, C.G.; Srilakshmi, J.C. Vitamin C elevates red blood cell glutathione in healthy adults. Am. J. Clin. Nutr. 1993, 58, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Englard, S.; Seifter, S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986, 6, 365–406. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Schellhorn, H.E. New developments and novel therapeutic perspectives for vitamin C. J. Nutr. 2007, 137, 2171–2184. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef]
- Warner, M.J.; Kamran, M.T. Anemia, Iron Deficiency; StatPearls: Treasure Island, FL, USA, 2020. [Google Scholar]
- Sourabh, S.; Bhatia, P.; Jain, R. Favourable improvement in haematological parameters in response to oral iron and vitamin C combination in children with Iron Refractory Iron Deficiency Anemia (IRIDA) phenotype. Blood Cells Mol. Dis. 2019, 75, 26–29. [Google Scholar] [CrossRef]
- Hunt, J.R.; Gallagher, S.K.; Johnson, L.K. Effect of ascorbic acid on apparent iron absorption by women with low iron stores. Am. J. Clin. Nutr. 1994, 59, 1381–1385. [Google Scholar] [CrossRef]
- Davidsson, L.; Galan, P.; Kastenmayer, P.; Cherouvrier, F.; Juillerat, M.A.; Hercberg, S.; Hurrell, R.F. Iron bioavailability studied in infants: The influence of phytic acid and ascorbic acid in infant formulas based on soy isolate. Pediatr. Res. 1994, 36, 816–822. [Google Scholar] [CrossRef]
- Linster, C.L.; Van Schaftingen, E. Vitamin C. Biosynthesis, recycling and degradation in mammals. FEBS J. 2007, 274, 1–22. [Google Scholar] [CrossRef]
- Drouin, G.; Godin, J.R.; Page, B. The genetics of vitamin C loss in vertebrates. Curr. Genom. 2011, 12, 371–378. [Google Scholar] [CrossRef]
- Grollman, A.P.; Lehninger, A.L. Enzymic synthesis of L-ascorbic acid in different animal species. Arch. Biochem. Biophys. 1957, 69, 458–467. [Google Scholar] [CrossRef]
- Bublitz, C.; Lehninger, A.L. The enzymic conversion of L-gulonate to L-as-corbate by rat-liver enzymes. Biochim. Biophys. Acta 1959, 32, 290–291. [Google Scholar] [CrossRef]
- Chatterjee, I.B.; Chatterjee, G.C.; Ghosh, N.C.; Ghosh, J.J.; Guha, B.C. Biological synthesis of L-ascorbic acid in animal tissues: Conversion of L-gulonolactone into L-ascorbic acid. Biochem. J. 1960, 74, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Nishikimi, M.; Yagi, K. Molecular basis for the deficiency in humans of gulonolactone oxidase, a key enzyme for ascorbic acid biosynthesis. Am. J. Clin. Nutr. 1991, 54, 1203S–1208S. [Google Scholar] [CrossRef] [PubMed]
- Nishikimi, M.; Kawai, T.; Yagi, K. Guinea pigs possess a highly mutated gene for L-gulono-gamma-lactone oxidase, the key enzyme for L-ascorbic acid biosynthesis missing in this species. J. Biol. Chem. 1992, 267, 21967–21972. [Google Scholar]
- Pollock, J.I.; Mullin, R.J. Vitamin C biosynthesis in prosimians: Evidence for the anthropoid affinity of Tarsius. Am. J. Phys. Anthropol. 1987, 73, 65–70. [Google Scholar] [CrossRef]
- Birney, E.C.; Jenness, R.; Ayaz, K.M. Inability of bats to synthesise L-ascorbic acid. Nature 1976, 260, 626–628. [Google Scholar] [CrossRef]
- Milton, K.; Jenness, R. Ascorbic acid content of neotropical plant parts available to wild monkeys and bats. Experientia 1987, 43, 339–342. [Google Scholar] [CrossRef]
- Institute of Medicine (US). Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Hampl, J.S.; Taylor, C.A.; Johnston, C.S. Vitamin C deficiency and depletion in the United States: The Third National Health and Nutrition Examination Survey, 1988 to 1994. Am. J. Public Health 2004, 94, 870–875. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Carroll, M.D.; Ford, E.S.; Lacher, D.A. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003–2004 National Health and Nutrition Examination Survey (NHANES). Am. J. Clin. Nutr. 2009, 90, 1252–1263. [Google Scholar] [CrossRef]
- Rowe, S.; Carr, A.C. Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern? Nutrients 2020, 12, 2008. [Google Scholar] [CrossRef]
- Villalpando, S.; Montalvo-Velarde, I.; Zambrano, N.; Garcia-Guerra, A.; Ramirez-Silva, C.I.; Shamah-Levy, T.; Rivera, J.A. Vitamins A, and C and folate status in Mexican children under 12 years and women 12–49 years: A probabilistic national survey. Salud Publica Mex 2003, 45, S508–S519. [Google Scholar] [CrossRef] [PubMed]
- Garcia, O.P.; Ronquillo, D.; Mdel, C.C.; Camacho, M.; Long, K.Z.; Rosado, J.L. Zinc, vitamin A, and vitamin C status are associated with leptin concentrations and obesity in Mexican women: Results from a cross-sectional study. Nutr. Metab. 2012, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L.; Corey, P.N.; El-Sohemy, A. Vitamin C deficiency in a population of young Canadian adults. Am. J. Epidemiol. 2009, 170, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, R.D.; Vashist, P.; Gupta, S.K.; Young, I.S.; Maraini, G.; Camparini, M.; Jayanthi, R.; John, N.; Fitzpatrick, K.E.; Chakravarthy, U.; et al. Inverse association of vitamin C with cataract in older people in India. Ophthalmology 2011, 118, 1958–1965.e2. [Google Scholar] [CrossRef]
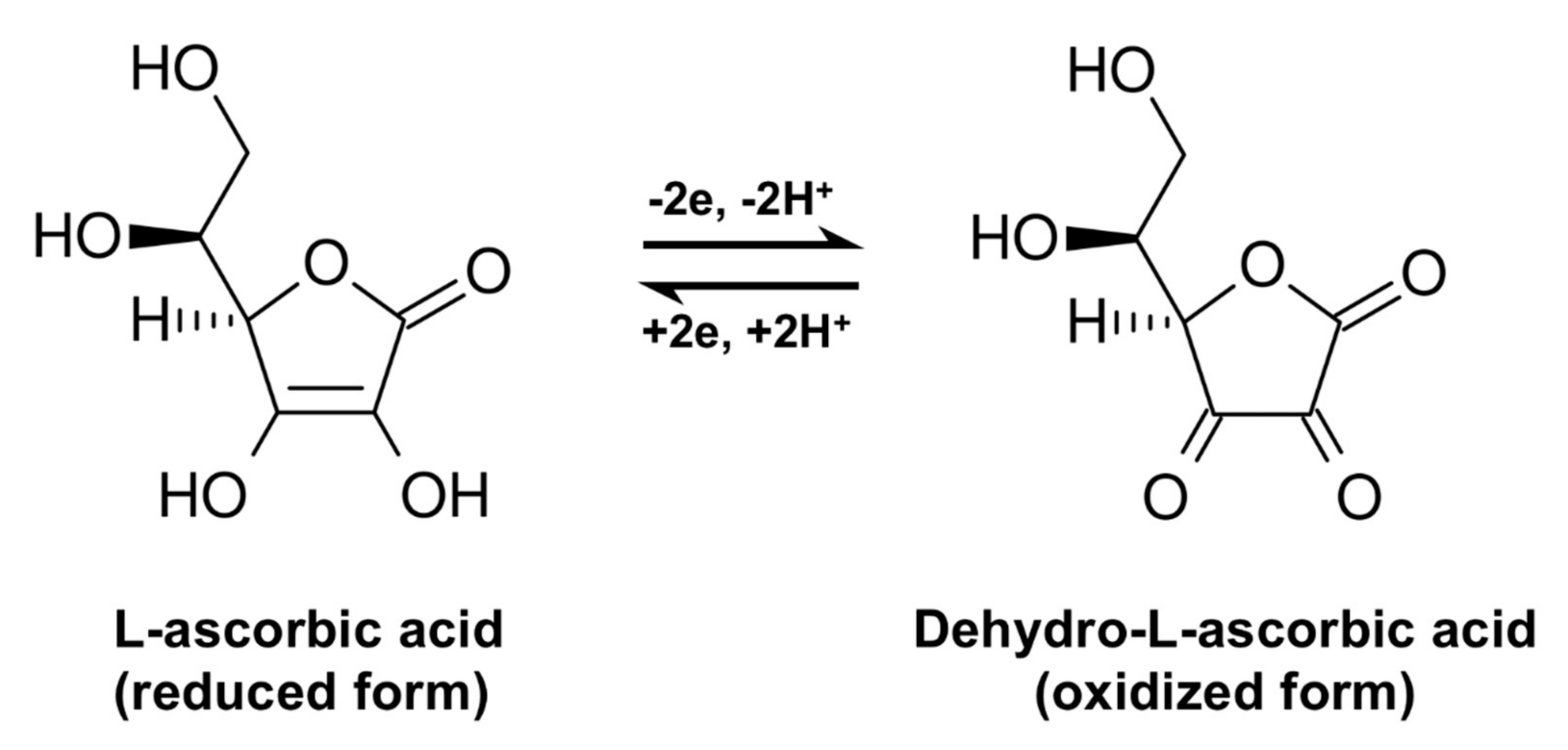
- Nishikawa, Y.; Kurata, T. Interconversion between dehydro-L-ascorbic acid and L-ascorbic acid. Biosci. Biotechnol. Biochem. 2000, 64, 476–483. [Google Scholar] [CrossRef]
- Dewhirst, R.A.; Fry, S.C. The oxidation of dehydroascorbic acid and 2,3-diketogulonate by distinct reactive oxygen species. Biochem. J. 2018, 475, 3451–3470. [Google Scholar] [CrossRef]
- Sauberlich, H.E. Pharmacology of vitamin C. Annu. Rev. Nutr. 1994, 14, 371–391. [Google Scholar] [CrossRef]
- Arrigoni, O.; De Tullio, M.C. Ascorbic acid: Much more than just an antioxidant. Biochim. Biophys. Acta 2002, 1569, 1–9. [Google Scholar] [CrossRef]
- Pauling, L. Ascorbic acid. Lancet 1979, 1, 615. [Google Scholar] [CrossRef]
- Przybylo, M.; Langner, M. On the physiological and cellular homeostasis of ascorbate. Cell Mol. Biol. Lett. 2020, 25, 32. [Google Scholar] [CrossRef]
- Rose, R.C. Solubility properties of reduced and oxidized ascorbate as determinants of membrane permeation. Biochim. Biophys. Acta 1987, 924, 254–256. [Google Scholar] [CrossRef]
- Zhitkovich, A. Nuclear and Cytoplasmic Functions of Vitamin C. Chem. Res. Toxicol. 2020, 33, 2515–2526. [Google Scholar] [CrossRef] [PubMed]
- Tsukaguchi, H.; Tokui, T.; Mackenzie, B.; Berger, U.V.; Chen, X.Z.; Wang, Y.; Brubaker, R.F.; Hediger, M.A. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature 1999, 399, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Sotiriou, S.; Gispert, S.; Cheng, J.; Wang, Y.; Chen, A.; Hoogstraten-Miller, S.; Miller, G.F.; Kwon, O.; Levine, M.; Guttentag, S.H.; et al. Ascorbic-acid transporter Slc23a1 is essential for vitamin C transport into the brain and for perinatal survival. Nat. Med. 2002, 8, 514–517. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, V.S.; Srinivasan, P.; Wildman, A.J.; Marchant, J.S.; Said, H.M. Molecular mechanism(s) involved in differential expression of vitamin C transporters along the intestinal tract. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, G340–G347. [Google Scholar] [CrossRef] [PubMed]
- Macias, R.I.; Hierro, C.; de Juan, S.C.; Jimenez, F.; Martin, F.G.-S.; Marin, J.J. Hepatic expression of sodium-dependent vitamin C transporters: Ontogeny, subtissular distribution and effect of chronic liver diseases. Br. J. Nutr. 2011, 106, 1814–1825. [Google Scholar] [CrossRef]
- Larsson, N.; Rankin, G.D.; Bicer, E.M.; Roos-Engstrand, E.; Pourazar, J.; Blomberg, A.; Mudway, I.S.; Behndig, A.F. Identification of vitamin C transporters in the human airways: A cross-sectional in vivo study. BMJ Open 2015, 5, e006979. [Google Scholar] [CrossRef]
- Wang, Y.; Mackenzie, B.; Tsukaguchi, H.; Weremowicz, S.; Morton, C.C.; Hediger, M.A. Human vitamin C (L-ascorbic acid) transporter SVCT1. Biochem. Biophys. Res. Commun. 2000, 267, 488–494. [Google Scholar] [CrossRef]
- Santulli, G. Adrenal Glands: From Pathophysiology to Clinical Evidence; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2015. [Google Scholar]
- Portugal, C.C.; Socodato, R.; Canedo, T.; Silva, C.M.; Martins, T.; Coreixas, V.S.; Loiola, E.C.; Gess, B.; Rohr, D.; Santiago, A.R.; et al. Caveolin-1-mediated internalization of the vitamin C transporter SVCT2 in microglia triggers an inflammatory phenotype. Sci. Signal. 2017, 10. [Google Scholar] [CrossRef]
- Ludke, A.R.; Sharma, A.K.; Akolkar, G.; Bajpai, G.; Singal, P.K. Downregulation of vitamin C transporter SVCT-2 in doxorubicin-induced cardiomyocyte injury. Am. J. Physiol. Cell Physiol. 2012, 303, C645–C653. [Google Scholar] [CrossRef]
- Portugal, C.C.; da Encarnacao, T.G.; Socodato, R.; Moreira, S.R.; Brudzewsky, D.; Ambrosio, A.F.; Paes-de-Carvalho, R. Nitric oxide modulates sodium vitamin C transporter 2 (SVCT-2) protein expression via protein kinase G (PKG) and nuclear factor-kappaB (NF-kappaB). J. Biol. Chem. 2012, 287, 3860–3872. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Chae, J.S.; Shin, H.; Shin, Y.; Song, H.; Kim, Y.; Yoo, B.C.; Roh, K.; Cho, S.; Kil, E.J.; et al. Hormetic dose response to L-ascorbic acid as an anti-cancer drug in colorectal cancer cell lines according to SVCT-2 expression. Sci. Rep. 2018, 8, 11372. [Google Scholar] [CrossRef] [PubMed]
- Malo, C.; Wilson, J.X. Glucose modulates vitamin C transport in adult human small intestinal brush border membrane vesicles. J. Nutr. 2000, 130, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Nualart, F.; Mack, L.; Garcia, A.; Cisternas, P.; Bongarzone, E.R.; Heitzer, M.; Jara, N.; Martinez, F.; Ferrada, L.; Espinoza, F.; et al. Vitamin C Transporters, Recycling and the Bystander Effect in the Nervous System: SVCT2 versus Gluts. J. Stem. Cell Res. Ther. 2014, 4, 209. [Google Scholar] [CrossRef]
- Vera, J.C.; Rivas, C.I.; Fischbarg, J.; Golde, D.W. Mammalian facilitative hexose transporters mediate the transport of dehydroascorbic acid. Nature 1993, 364, 79–82. [Google Scholar] [CrossRef]
- Corpe, C.P.; Eck, P.; Wang, J.; Al-Hasani, H.; Levine, M. Intestinal dehydroascorbic acid (DHA) transport mediated by the facilitative sugar transporters, GLUT2 and GLUT8. J. Biol. Chem. 2013, 288, 9092–9101. [Google Scholar] [CrossRef]
- Dong, W.; Tian, C.; Jiao, Y.; Blackwell, S.; Lou, G.; Postlethwaite, A.; Gu, W.; Sun, D. Multiple genome analyses reveal key genes in Vitamin C and Vitamin D synthesis and transport pathways are shared. Sci. Rep. 2019, 9, 16811. [Google Scholar] [CrossRef]
- Lukawski, M.; Dalek, P.; Witkiewicz, W.; Przybylo, M.; Langner, M. Experimental evidence and physiological significance of the ascorbate passive diffusion through the lipid bilayer. Chem. Phys. Lipids 2020, 232, 104950. [Google Scholar] [CrossRef]
- Pozzer, D.; Invernizzi, R.W.; Blaauw, B.; Cantoni, O.; Zito, E. Ascorbic Acid Route to the Endoplasmic Reticulum: Function and Role in Disease. Antioxid. Redox Signal. 2020. [Google Scholar] [CrossRef]
- Nygaard, G. On a Novel, Simplified Model Framework Describing Ascorbic Acid Concentration Dynamics. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2019, 2019, 2880–2886. [Google Scholar]
- Bozonet, S.M.; Carr, A.C. The Role of Physiological Vitamin C Concentrations on Key Functions of Neutrophils Isolated from Healthy Individuals. Nutrients 2019, 11, 1363. [Google Scholar] [CrossRef] [PubMed]
- Corpe, C.P.; Tu, H.; Eck, P.; Wang, J.; Faulhaber-Walter, R.; Schnermann, J.; Margolis, S.; Padayatty, S.; Sun, H.; Wang, Y.; et al. Vitamin C transporter Slc23a1 links renal reabsorption, vitamin C tissue accumulation, and perinatal survival in mice. J. Clin. Investig. 2010, 120, 1069–1083. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Padayatty, S.J.; Espey, M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011, 2, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, S.M.; Katz, A.; Wesley, R.A.; Levine, M. Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, B.; Illing, A.C.; Hediger, M.A. Transport model of the human Na+-coupled L-ascorbic acid (vitamin C) transporter SVCT1. Am. J. Physiol. Cell Physiol. 2008, 294, C451–C459. [Google Scholar] [CrossRef] [PubMed]
- Wohlrab, C.; Phillips, E.; Dachs, G.U. Vitamin C Transporters in Cancer: Current Understanding and Gaps in Knowledge. Front. Oncol. 2017, 7, 74. [Google Scholar] [CrossRef]
- Roa, F.J.; Pena, E.; Inostroza, E.; Sotomayor, K.; Gonzalez, M.; Gutierrez-Castro, F.A.; Maurin, M.; Sweet, K.; Labrousse, C.; Gatica, M.; et al. Data on SVCT2 transporter expression and localization in cancer cell lines and tissues. Data Br. 2019, 25, 103972. [Google Scholar] [CrossRef]
- Lykkesfeldt, J. On the effect of vitamin C intake on human health: How to (mis)interprete the clinical evidence. Redox Biol. 2020, 34, 101532. [Google Scholar] [CrossRef]
- Wilson, J.X. Regulation of vitamin C transport. Annu. Rev. Nutr. 2005, 25, 105–125. [Google Scholar] [CrossRef]
- Stephenson, C.M.; Levin, R.D.; Spector, T.; Lis, C.G. Phase I clinical trial to evaluate the safety, tolerability, and pharmacokinetics of high-dose intravenous ascorbic acid in patients with advanced cancer. Cancer Chemother. Pharmacol. 2013, 72, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Bangham, A.D.; Horne, R.W. Negative Staining of Phospholipids and Their Structural Modification by Surface-Active Agents as Observed in the Electron Microscope. J. Mol. Biol. 1964, 8, 660–668. [Google Scholar] [CrossRef]
- Yao, X.; Fan, X.; Yan, N. Cryo-EM analysis of a membrane protein embedded in the liposome. Proc. Natl. Acad. Sci. USA 2020, 117, 18497–18503. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D.W. From “banghasomes“ to liposomes: A memoir of Alec Bangham, 1921–2010. FASEB J. 2010, 24, 1308–1310. [Google Scholar] [CrossRef] [PubMed]
- Mozafari, M.R. Liposomes: An overview of manufacturing techniques. Cell. Mol. Biol. Lett. 2005, 10, 711–719. [Google Scholar] [PubMed]
- Madni, A.; Sarfraz, M.; Rehman, M.; Ahmad, M.; Akhtar, N.; Ahmad, S.; Tahir, N.; Ijaz, S.; Al-Kassas, R.; Lobenberg, R. Liposomal drug delivery: A versatile platform for challenging clinical applications. J. Pharm. Pharm. Sci. 2014, 17, 401–426. [Google Scholar] [CrossRef]
- Karn, P.R.; Cho, W.; Hwang, S.J. Liposomal drug products and recent advances in the synthesis of supercritical fluid-mediated liposomes. Nanomedicine 2013, 8, 1529–1548. [Google Scholar] [CrossRef]
- Nayerhoda, R.; Hill, A.; Pfeifer, B.A. Liposomal Dual Delivery of Both Polysaccharide and Protein Antigens. Methods Mol. Biol. 2021, 2183, 477–487. [Google Scholar]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015, 10, 975–999. [Google Scholar] [CrossRef]
- Kauffman, K.J.; Dorkin, J.R.; Yang, J.H.; Heartlein, M.W.; DeRosa, F.; Mir, F.F.; Fenton, O.S.; Anderson, D.G. Optimization of Lipid Nanoparticle Formulations for mRNA Delivery in Vivo with Fractional Factorial and Definitive Screening Designs. Nano Lett. 2015, 15, 7300–7306. [Google Scholar] [CrossRef]
- Kamaly, N.; Miller, A.D. Paramagnetic liposome nanoparticles for cellular and tumour imaging. Int. J. Mol. Sci. 2010, 11, 1759–1776. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Shin, Y.; Lee, W.; Whang, K.; Kim, D.; Lee, L.P.; Choi, J.W.; Kang, T. General and programmable synthesis of hybrid liposome/metal nanoparticles. Sci. Adv. 2016, 2, e1601838. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Liu, C.; Liu, W.; Yu, H.; Zheng, H.; Zhou, W.; Hu, Y. Preparation and characterization of nanoliposomes entrapping medium-chain fatty acids and vitamin C by lyophilization. Int. J. Mol. Sci. 2013, 14, 19763–19773. [Google Scholar] [CrossRef] [PubMed]
- Moribe, K.; Limwikrant, W.; Higashi, K.; Yamamoto, K. Drug nanoparticle formulation using ascorbic Acid derivatives. J. Drug. Deliv. 2011, 2011, 138929. [Google Scholar] [CrossRef] [PubMed]
- Preiss, M.R.; Bothun, G.D. Stimuli-responsive liposome-nanoparticle assemblies. Expert Opin. Drug Deliv. 2011, 8, 1025–1040. [Google Scholar] [CrossRef]
- Ross, C.; Taylor, M.; Fullwood, N.; Allsop, D. Liposome delivery systems for the treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 8507–8522. [Google Scholar] [CrossRef]
- Ickenstein, L.M.; Garidel, P. Lipid-based nanoparticle formulations for small molecules and RNA drugs. Expert Opin. Drug Deliv. 2019, 16, 1205–1226. [Google Scholar] [CrossRef]
- Seleci, M.; Seleci, D.A.; Scheper, T.; Stahl, F. Theranostic Liposome-Nanoparticle Hybrids for Drug Delivery and Bioimaging. Int. J. Mol. Sci. 2017, 18, 1415. [Google Scholar] [CrossRef]
- Zylberberg, C.; Gaskill, K.; Pasley, S.; Matosevic, S. Engineering liposomal nanoparticles for targeted gene therapy. Gene Ther. 2017, 24, 441–452. [Google Scholar] [CrossRef]
- Michel, R.; Gradzielski, M. Experimental aspects of colloidal interactions in mixed systems of liposome and inorganic nanoparticle and their applications. Int. J. Mol. Sci. 2012, 13, 11610–11642. [Google Scholar] [CrossRef]
- Zamani, P.; Momtazi-Borojeni, A.A.; Nik, M.E.; Oskuee, R.K.; Sahebkar, A. Nanoliposomes as the adjuvant delivery systems in cancer immunotherapy. J. Cell. Physiol. 2018, 233, 5189–5199. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Compton, R.G. Investigation of single-drug-encapsulating liposomes using the nano-impact method. Angew. Chem. Int. Ed. Engl. 2014, 53, 13928–13930. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Seike, Y.; Saeki, A.; Kozawa, T.; Takeuchi, F.; Tsubaki, M. A pulse radiolysis study of the dynamics of ascorbic acid free radicals within a liposomal environment. ChemPhysChem 2014, 15, 2994–2997. [Google Scholar] [CrossRef] [PubMed]
- Vanaja, K.; Wahl, M.A.; Bukarica, L.; Heinle, H. Liposomes as carriers of the lipid soluble antioxidant resveratrol: Evaluation of amelioration of oxidative stress by additional antioxidant vitamin. Life Sci. 2013, 93, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Song, W.; Shen, L.; Qiu, N.; Hu, M.; Liu, Y.; Liu, Q.; Huang, L. Vasodilator Hydralazine Promotes Nanoparticle Penetration in Advanced Desmoplastic Tumors. ACS Nano 2019, 13, 1751–1763. [Google Scholar] [CrossRef] [PubMed]
- Attia, M.; Essa, E.A.; Zaki, R.M.; Elkordy, A.A. An Overview of the Antioxidant Effects of Ascorbic Acid and Alpha Lipoic Acid (in Liposomal Forms) as Adjuvant in Cancer Treatment. Antioxidants 2020, 9, 359. [Google Scholar] [CrossRef] [PubMed]
- Felice, B.; Prabhakaran, M.P.; Rodriguez, A.P.; Ramakrishna, S. Drug delivery vehicles on a nano-engineering perspective. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 41, 178–195. [Google Scholar] [CrossRef]
- Tavano, L.; Muzzalupo, R.; Picci, N.; de Cindio, B. Co-encapsulation of antioxidants into niosomal carriers: Gastrointestinal release studies for nutraceutical applications. Colloids Surf. B Biointerfaces 2014, 114, 82–88. [Google Scholar] [CrossRef]
- Castro, R.A.; Silva-Barcellos, N.M.; Licio, C.S.; Souza, J.B.; Souza-Testasicca, M.C.; Ferreira, F.M.; Batista, M.A.; Silveira-Lemos, D.; Moura, S.L.; Frezard, F.; et al. Association of liposome-encapsulated trivalent antimonial with ascorbic acid: An effective and safe strategy in the treatment of experimental visceral leishmaniasis. PLoS ONE 2014, 9, e104055. [Google Scholar] [CrossRef]
- Prantl, L.; Eigenberger, A.; Gehmert, S.; Haerteis, S.; Aung, T.; Rachel, R.; Jung, E.M.; Felthaus, O. Enhanced Resorption of Liposomal Packed Vitamin C Monitored by Ultrasound. J. Clin. Med. 2020, 9, 1616. [Google Scholar] [CrossRef]
- Liu, N.; Park, H.J. Factors effect on the loading efficiency of Vitamin C loaded chitosan-coated nanoliposomes. Colloids Surf. B Biointerfaces 2010, 76, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, P.; Zou, Y.X.; Luo, Z.G.; Tamer, T.M. Co-encapsulation of Vitamin C and beta-Carotene in liposomes: Storage stability, antioxidant activity, and in vitro gastrointestinal digestion. Food Res. Int. 2020, 136, 109587. [Google Scholar] [CrossRef] [PubMed]
- Khalili, A.; Alipour, S.; Fathalipour, M.; Purkhosrow, A.; Mashghoolozekr, E.; Bayat, G.; Nekooeian, A.A. Liposomal and Non-Liposomal Formulations of Vitamin C: Comparison of the Antihypertensive and Vascular Modifying Activity in Renovascular Hypertensive Rats. Iran. J. Med. Sci. 2020, 45, 41–49. [Google Scholar]
- Carita, A.C.; Fonseca-Santos, B.; Shultz, J.D.; Michniak-Kohn, B.; Chorilli, M.; Leonardi, G.R. Vitamin C: One compound, several uses. Advances for delivery, efficiency and stability. Nanomedicine 2020, 24, 102117. [Google Scholar] [CrossRef] [PubMed]
- Lukawski, M.; Dalek, P.; Borowik, T.; Forys, A.; Langner, M.; Witkiewicz, W.; Przybylo, M. New oral liposomal vitamin C formulation: Properties and bioavailability. J. Liposome Res. 2020, 30, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Maione-Silva, L.; de Castro, E.G.; Nascimento, T.L.; Cintra, E.R.; Moreira, L.C.; Cintra, B.A.S.; Valadares, M.C.; Lima, E.M. Ascorbic acid encapsulated into negatively charged liposomes exhibits increased skin permeation, retention and enhances collagen synthesis by fibroblasts. Sci. Rep. 2019, 9, 522. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Fu, Q.; Zhao, Y.; Zhang, L.; Yue, Q.; Hai, L.; Guo, L.; Wu, Y. Ascorbic acid-modified brain-specific liposomes drug delivery system with “lock-in” function. Chem. Phys. Lipids 2019, 224, 104727. [Google Scholar] [CrossRef] [PubMed]
- Parhizkar, E.; Rashedinia, M.; Karimi, M.; Alipour, S. Design and development of vitamin C-encapsulated proliposome with improved in-vitro and ex-vivo antioxidant efficacy. J. Microencapsul. 2018, 35, 301–311. [Google Scholar] [CrossRef]
- Jiao, Z.; Wang, X.; Yin, Y.; Xia, J.; Mei, Y. Preparation and evaluation of a chitosan-coated antioxidant liposome containing vitamin C and folic acid. J. Microencapsul. 2018, 35, 272–280. [Google Scholar] [CrossRef]
- Miura, Y.; Fuchigami, Y.; Hagimori, M.; Sato, H.; Ogawa, K.; Munakata, C.; Wada, M.; Maruyama, K.; Kawakami, S. Evaluation of the targeted delivery of 5-fluorouracil and ascorbic acid into the brain with ultrasound-responsive nanobubbles. J. Drug Target 2018, 26, 684–691. [Google Scholar] [CrossRef]
- Tsai, W.C.; Rizvi, S.S.H. Simultaneous microencapsulation of hydrophilic and lipophilic bioactives in liposomes produced by an ecofriendly supercritical fluid process. Food Res. Int. 2017, 99, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Duconge, J.; Miranda-Massari, J.R.; Gonzalez, M.J.; Jackson, J.A.; Warnock, W.; Riordan, N.H. Pharmacokinetics of vitamin C: Insights into the oral and intravenous administration of ascorbate. P. R. Health Sci. J. 2008, 27, 7–19. [Google Scholar] [PubMed]
- Davis, J.L.; Paris, H.L.; Beals, J.W.; Binns, S.E.; Giordano, G.R.; Scalzo, R.L.; Schweder, M.M.; Blair, E.; Bell, C. Liposomal-encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia-Reperfusion Injury. Nutr. Metab. Insights 2016, 9, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Gopi, S.; Balakrishnan, P. Evaluation and Clinical Comparison Studies on Liposomal and Non-Liposomal Ascorbic Acid (Vitamin C) and their Enhanced Bioavailability. J. Liposome Res. 2020, 1–35. [Google Scholar] [CrossRef]
- Ashor, A.W.; Siervo, M.; Lara, J.; Oggioni, C.; Mathers, J.C. Antioxidant vitamin supplementation reduces arterial stiffness in adults: A systematic review and meta-analysis of randomized controlled trials. J. Nutr. 2014, 144, 1594–1602. [Google Scholar] [CrossRef]
- Moran, J.P.; Cohen, L.; Greene, J.M.; Xu, G.; Feldman, E.B.; Hames, C.G.; Feldman, D.S. Plasma ascorbic acid concentrations relate inversely to blood pressure in human subjects. Am. J. Clin. Nutr. 1993, 57, 213–217. [Google Scholar] [CrossRef]
- Myint, P.K.; Luben, R.N.; Wareham, N.J.; Khaw, K.T. Association between plasma vitamin C concentrations and blood pressure in the European prospective investigation into cancer-Norfolk population-based study. Hypertension 2011, 58, 372–379. [Google Scholar] [CrossRef]
- Pfister, R.; Sharp, S.J.; Luben, R.; Wareham, N.J.; Khaw, K.T. Plasma vitamin C predicts incident heart failure in men and women in European Prospective Investigation into Cancer and Nutrition-Norfolk prospective study. Am. Heart J. 2011, 162, 246–253. [Google Scholar] [CrossRef]
- Heller, R.; Munscher-Paulig, F.; Grabner, R.; Till, U. L-Ascorbic acid potentiates nitric oxide synthesis in endothelial cells. J. Biol. Chem. 1999, 274, 8254–8260. [Google Scholar] [CrossRef]
- Huang, A.; Vita, J.A.; Venema, R.C.; Keaney, J.F., Jr. Ascorbic acid enhances endothelial nitric-oxide synthase activity by increasing intracellular tetrahydrobiopterin. J. Biol. Chem. 2000, 275, 17399–17406. [Google Scholar] [CrossRef]
- Baker, T.A.; Milstien, S.; Katusic, Z.S. Effect of vitamin C on the availability of tetrahydrobiopterin in human endothelial cells. J. Cardiovasc. Pharmacol. 2001, 37, 333–338. [Google Scholar] [CrossRef] [PubMed]
- D’Uscio, L.V.; Milstien, S.; Richardson, D.; Smith, L.; Katusic, Z.S. Long-term vitamin C treatment increases vascular tetrahydrobiopterin levels and nitric oxide synthase activity. Circ. Res. 2003, 92, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Tie, G.; Messina, L.M. Tetrahydrobiopterin, L-arginine and vitamin C actsynergistically to decrease oxidative stress, increase nitricoxide and improve blood flow after induction of hindlimb ischemia in the rat. Mol. Med. 2012, 18, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Ulker, S.; McKeown, P.P.; Bayraktutan, U. Vitamins reverse endothelial dysfunction through regulation of eNOS and NAD(P)H oxidase activities. Hypertension 2003, 41, 534–539. [Google Scholar] [CrossRef]
- Ladurner, A.; Schmitt, C.A.; Schachner, D.; Atanasov, A.G.; Werner, E.R.; Dirsch, V.M.; Heiss, E.H. Ascorbate stimulates endothelial nitric oxide synthase enzyme activity by rapid modulation of its phosphorylation status. Free Radic. Biol. Med. 2012, 52, 2082–2090. [Google Scholar] [CrossRef]
- Yuan, Q.; Yang, J.; Santulli, G.; Reiken, S.R.; Wronska, A.; Kim, M.M.; Osborne, B.W.; Lacampagne, A.; Yin, Y.; Marks, A.R. Maintenance of normal blood pressure is dependent on IP3R1-mediated regulation of eNOS. Proc. Natl. Acad. Sci. USA 2016, 113, 8532–8537. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Aspect. Med. 2009, 30, 1–12. [Google Scholar] [CrossRef]
- Lushchak, V.I. Glutathione homeostasis and functions: Potential targets for medical interventions. J Amino Acids 2012, 2012, 736837. [Google Scholar] [CrossRef]
- Martensson, J.; Han, J.; Griffith, O.W.; Meister, A. Glutathione ester delays the onset of scurvy in ascorbate-deficient guinea pigs. Proc. Natl. Acad. Sci. USA 1993, 90, 317–321. [Google Scholar] [CrossRef]
- Guaiquil, V.H.; Vera, J.C.; Golde, D.W. Mechanism of vitamin C inhibition of cell death induced by oxidative stress in glutathione-depleted HL-60 cells. J. Biol. Chem. 2001, 276, 40955–40961. [Google Scholar] [CrossRef]
- Shang, F.; Lu, M.; Dudek, E.; Reddan, J.; Taylor, A. Vitamin C and vitamin E restore the resistance of GSH-depleted lens cells to H2O2. Free Radic. Biol. Med. 2003, 34, 521–530. [Google Scholar] [CrossRef]
- Waly, M.I.; Al-Attabi, Z.; Guizani, N. Low Nourishment of Vitamin C Induces Glutathione Depletion and Oxidative Stress in Healthy Young Adults. Prev. Nutr. Food Sci. 2015, 20, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.A.; Aon, M.A.; Frasier, C.R.; Sloan, R.C.; Maloney, A.H.; Anderson, E.J.; O’Rourke, B. Cardiac arrhythmias induced by glutathione oxidation can be inhibited by preventing mitochondrial depolarization. J. Mol. Cell. Cardiol. 2010, 48, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Rimm, E.B.; Stampfer, M.J.; Ascherio, A.; Giovannucci, E.; Colditz, G.A.; Willett, W.C. Vitamin E consumption and the risk of coronary heart disease in men. N. Engl. J. Med. 1993, 328, 1450–1456. [Google Scholar] [CrossRef]
- Osganian, S.K.; Stampfer, M.J.; Rimm, E.; Spiegelman, D.; Hu, F.B.; Manson, J.E.; Willett, W.C. Vitamin C and risk of coronary heart disease in women. J. Am. Coll. Cardiol. 2003, 42, 246–252. [Google Scholar] [CrossRef]
- Knekt, P.; Ritz, J.; Pereira, M.A.; O’Reilly, E.J.; Augustsson, K.; Fraser, G.E.; Goldbourt, U.; Heitmann, B.L.; Hallmans, G.; Liu, S.; et al. Antioxidant vitamins and coronary heart disease risk: A pooled analysis of 9 cohorts. Am. J. Clin. Nutr. 2004, 80, 1508–1520. [Google Scholar] [CrossRef]
- Lee, D.H.; Folsom, A.R.; Harnack, L.; Halliwell, B.; Jacobs, D.R., Jr. Does supplemental vitamin C increase cardiovascular disease risk in women with diabetes? Am. J. Clin. Nutr. 2004, 80, 1194–1200. [Google Scholar] [CrossRef]
- Khaw, K.T.; Bingham, S.; Welch, A.; Luben, R.; Wareham, N.; Oakes, S.; Day, N. Relation between plasma ascorbic acid and mortality in men and women in EPIC-Norfolk prospective study: A prospective population study. European Prospective Investigation into Cancer and Nutrition. Lancet 2001, 357, 657–663. [Google Scholar] [CrossRef]
- Myint, P.K.; Luben, R.N.; Welch, A.A.; Bingham, S.A.; Wareham, N.J.; Khaw, K.T. Plasma vitamin C concentrations predict risk of incident stroke over 10 y in 20 649 participants of the European Prospective Investigation into Cancer Norfolk prospective population study. Am. J. Clin. Nutr. 2008, 87, 64–69. [Google Scholar] [CrossRef]
- Ascherio, A.; Rimm, E.B.; Hernan, M.A.; Giovannucci, E.; Kawachi, I.; Stampfer, M.J.; Willett, W.C. Relation of consumption of vitamin E, vitamin C, and carotenoids to risk for stroke among men in the United States. Ann. Intern. Med. 1999, 130, 963–970. [Google Scholar] [CrossRef]
- Salonen, R.M.; Nyyssonen, K.; Kaikkonen, J.; Porkkala-Sarataho, E.; Voutilainen, S.; Rissanen, T.H.; Tuomainen, T.P.; Valkonen, V.P.; Ristonmaa, U.; Lakka, H.M.; et al. Antioxidant Supplementation in Atherosclerosis Prevention, S., Six-year effect of combined vitamin C and E supplementation on atherosclerotic progression: The Antioxidant Supplementation in Atherosclerosis Prevention (ASAP) Study. Circulation 2003, 107, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.R.; Albert, C.M.; Gaziano, J.M.; Zaharris, E.; MacFadyen, J.; Danielson, E.; Buring, J.E.; Manson, J.E. A randomized factorial trial of vitamins C and E and beta carotene in the secondary prevention of cardiovascular events in women: Results from the Women’s Antioxidant Cardiovascular Study. Arch. Intern. Med. 2007, 167, 1610–1618. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.A.; Hudes, E.S.; Tice, J.A. Relation of serum ascorbic acid to mortality among US adults. J. Am. Coll. Nutr. 2001, 20, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.A.; Hudes, E.S.; Browner, W.S. Serum ascorbic acid and cardiovascular disease prevalence in U.S. adults. Epidemiology 1998, 9, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Roussel, A.M.; Favier, A.; Briancon, S. The SU.VI.MAX Study: A randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch. Intern. Med. 2004, 164, 2335–2342. [Google Scholar] [CrossRef]
- Sesso, H.D.; Buring, J.E.; Christen, W.G.; Kurth, T.; Belanger, C.; MacFadyen, J.; Bubes, V.; Manson, J.E.; Glynn, R.J.; Gaziano, J.M. Vitamins E and C in the prevention of cardiovascular disease in men: The Physicians’ Health Study II randomized controlled trial. JAMA 2008, 300, 2123–2133. [Google Scholar] [CrossRef] [PubMed]
- Heart Protection Study Collaborative, G. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: A randomised placebo-controlled trial. Lancet 2002, 360, 23–33. [Google Scholar]
- Buijsse, B.; Jacobs, D.R., Jr.; Steffen, L.M.; Kromhout, D.; Gross, M.D. Plasma Ascorbic Acid, A Priori Diet Quality Score, and Incident Hypertension: A Prospective Cohort Study. PLoS ONE 2015, 10, e0144920. [Google Scholar] [CrossRef]
- Yochum, L.A.; Folsom, A.R.; Kushi, L.H. Intake of antioxidant vitamins and risk of death from stroke in postmenopausal women. Am. J. Clin. Nutr. 2000, 72, 476–483. [Google Scholar] [CrossRef]
- Del Rio, D.; Agnoli, C.; Pellegrini, N.; Krogh, V.; Brighenti, F.; Mazzeo, T.; Masala, G.; Bendinelli, B.; Berrino, F.; Sieri, S.; et al. Total antioxidant capacity of the diet is associated with lower risk of ischemic stroke in a large Italian cohort. J. Nutr. 2011, 141, 118–123. [Google Scholar] [CrossRef]
- Ye, Z.; Song, H. Antioxidant vitamins intake and the risk of coronary heart disease: Meta-analysis of cohort studies. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Kubota, Y.; Iso, H.; Date, C.; Kikuchi, S.; Watanabe, Y.; Wada, Y.; Inaba, Y.; Tamakoshi, A.; Group, J.S. Dietary intakes of antioxidant vitamins and mortality from cardiovascular disease: The Japan Collaborative Cohort Study (JACC) study. Stroke 2011, 42, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Li, J.; Yuan, Z. Effect of antioxidant vitamin supplementation on cardiovascular outcomes: A meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e56803. [Google Scholar] [CrossRef] [PubMed]
- Juraschek, S.P.; Guallar, E.; Appel, L.J.; Miller, E.R., 3rd. Effects of vitamin C supplementation on blood pressure: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 95, 1079–1088. [Google Scholar] [CrossRef]
- Chen, G.C.; Lu, D.B.; Pang, Z.; Liu, Q.F. Vitamin C intake, circulating vitamin C and risk of stroke: A meta-analysis of prospective studies. J. Am. Heart Assoc. 2013, 2, e000329. [Google Scholar] [CrossRef]
- Ashor, A.W.; Lara, J.; Mathers, J.C.; Siervo, M. Effect of vitamin C on endothelial function in health and disease: A systematic review and meta-analysis of randomised controlled trials. Atherosclerosis 2014, 235, 9–20. [Google Scholar] [CrossRef]
- Al-Khudairy, L.; Flowers, N.; Wheelhouse, R.; Ghannam, O.; Hartley, L.; Stranges, S.; Rees, K. Vitamin C supplementation for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2017, 3, CD011114. [Google Scholar] [CrossRef]
- Ran, L.; Zhao, W.; Tan, X.; Wang, H.; Mizuno, K.; Takagi, K.; Zhao, Y.; Bu, H. Association between Serum Vitamin C and the Blood Pressure: A Systematic Review and Meta-Analysis of Observational Studies. Cardiovasc. Ther. 2020, 2020, 4940673. [Google Scholar] [CrossRef]
- Guan, Y.; Dai, P.; Wang, H. Effects of vitamin C supplementation on essential hypertension: A systematic review and meta-analysis. Medicine 2020, 99, e19274. [Google Scholar] [CrossRef]
- Jayedi, A.; Rashidy-Pour, A.; Parohan, M.; Zargar, M.S.; Shab-Bidar, S. Dietary and circulating vitamin C, vitamin E, beta-carotene and risk of total cardiovascular mortality: A systematic review and dose-response meta-analysis of prospective observational studies. Public Health Nutr. 2019, 22, 1872–1887. [Google Scholar] [CrossRef]
- Bird, J.K.; Murphy, R.A.; Ciappio, E.D.; McBurney, M.I. Risk of Deficiency in Multiple Concurrent Micronutrients in Children and Adults in the United States. Nutrients 2017, 9, 655. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Cipolletta, E.; Sorriento, D.; Del Giudice, C.; Anastasio, A.; Monaco, S.; Maione, A.S.; Condorelli, G.; Puca, A.; Trimarco, B.; et al. CaMK4 Gene Deletion Induces Hypertension. J. Am. Heart Assoc. 2012, 1, e001081. [Google Scholar] [CrossRef] [PubMed]
- Iaccarino, G.; Ciccarelli, M.; Sorriento, D.; Cipolletta, E.; Cerullo, V.; Iovino, G.L.; Paudice, A.; Elia, A.; Santulli, G.; Campanile, A.; et al. AKT participates in endothelial dysfunction in hypertension. Circulation 2004, 109, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. The pathogenesis of atherosclerosis—An update. N. Engl. J. Med. 1986, 314, 488–500. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef]
- Sardu, C.; Gambardella, J.; Morelli, M.B.; Wang, X.; Marfella, R.; Santulli, G. Hypertension, Thrombosis, Kidney Failure, and Diabetes: Is COVID-19 an Endothelial Disease? A Comprehensive Evaluation of Clinical and Basic Evidence. J. Clin. Med. 2020, 9, 1417. [Google Scholar] [CrossRef]
- Celermajer, D.S.; Adams, M.R.; Clarkson, P.; Robinson, J.; McCredie, R.; Donald, A.; Deanfield, J.E. Passive smoking and impaired endothelium-dependent arterial dilatation in healthy young adults. N. Engl. J. Med. 1996, 334, 150–154. [Google Scholar] [CrossRef]
- Marti, C.N.; Gheorghiade, M.; Kalogeropoulos, A.P.; Georgiopoulou, V.V.; Quyyumi, A.A.; Butler, J. Endothelial dysfunction, arterial stiffness, and heart failure. J. Am. Coll. Cardiol. 2012, 60, 1455–1469. [Google Scholar] [CrossRef]
- Matarese, A.; Gambardella, J.; Sardu, C.; Santulli, G. miR-98 Regulates TMPRSS2 Expression in Human Endothelial Cells: Key Implications for COVID-19. Biomedicines 2020, 8, 462. [Google Scholar] [CrossRef]
- Aquila, G.; Morelli, M.B.; Vieceli Dalla Sega, F.; Fortini, F.; Nigro, P.; Caliceti, C.; Ferracin, M.; Negrini, M.; Pannuti, A.; Bonora, M.; et al. Heart rate reduction with ivabradine in the early phase of atherosclerosis is protective in the endothelium of ApoE-deficient mice. J. Physiol. Pharmacol. 2018, 69, 35–52. [Google Scholar]
- Gambardella, J.; Sorriento, D.; Bova, M.; Rusciano, M.; Loffredo, S.; Wang, X.; Petraroli, A.; Carucci, L.; Mormile, I.; Oliveti, M.; et al. Role of Endothelial G Protein-Coupled Receptor Kinase 2 in Angioedema. Hypertension 2020, 76, 1625–1636. [Google Scholar] [CrossRef] [PubMed]
- Sorriento, D.; Santulli, G.; Del Giudice, C.; Anastasio, A.; Trimarco, B.; Iaccarino, G. Endothelial cells are able to synthesize and release catecholamines both in vitro and in vivo. Hypertension 2012, 60, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, J.; Morelli, M.B.; Wang, X.J.; Santulli, G. Pathophysiological mechanisms underlying the beneficial effects of physical activity in hypertension. J. Clin. Hypertens. 2020, 22, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Munro, J.M.; Cotran, R.S. The pathogenesis of atherosclerosis: Atherogenesis and inflammation. Lab. Investig. 1988, 58, 249–261. [Google Scholar] [PubMed]
- Forman, J.P.; Choi, H.; Curhan, G.C. Fructose and vitamin C intake do not influence risk for developing hypertension. J. Am. Soc. Nephrol. 2009, 20, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Block, G.; Jensen, C.D.; Norkus, E.P.; Hudes, M.; Crawford, P.B. Vitamin C in plasma is inversely related to blood pressure and change in blood pressure during the previous year in young Black and White women. Nutr. J. 2008, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Bruno, R.M.; Daghini, E.; Ghiadoni, L.; Sudano, I.; Rugani, I.; Varanini, M.; Passino, C.; Emdin, M.; Taddei, S. Effect of acute administration of vitamin C on muscle sympathetic activity, cardiac sympathovagal balance, and baroreflex sensitivity in hypertensive patients. Am. J. Clin. Nutr. 2012, 96, 302–308. [Google Scholar] [CrossRef]
- Heitzer, T.; Just, H.; Munzel, T. Antioxidant vitamin C improves endothelial dysfunction in chronic smokers. Circulation 1996, 94, 6–9. [Google Scholar] [CrossRef]
- Fuller, C.J.; Grundy, S.M.; Norkus, E.P.; Jialal, I. Effect of ascorbate supplementation on low density lipoprotein oxidation in smokers. Atherosclerosis 1996, 119, 139–150. [Google Scholar] [CrossRef]
- Van Hoydonck, P.G.; Schouten, E.G.; Manuel, Y.K.B.; van Campenhout, A.; Hoppenbrouwers, K.P.; Temme, E.H. Does vitamin C supplementation influence the levels of circulating oxidized LDL, sICAM-1, sVCAM-1 and vWF-antigen in healthy male smokers? Eur. J. Clin. Nutr. 2004, 58, 1587–1593. [Google Scholar] [CrossRef]
- Salonen, J.T.; Nyyssonen, K.; Salonen, R.; Lakka, H.M.; Kaikkonen, J.; Porkkala-Sarataho, E.; Voutilainen, S.; Lakka, T.A.; Rissanen, T.; Leskinen, L.; et al. Antioxidant Supplementation in Atherosclerosis Prevention (ASAP) study: A randomized trial of the effect of vitamins E and C on 3-year progression of carotid atherosclerosis. J. Intern. Med. 2000, 248, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Valls, N.; Gormaz, J.G.; Aguayo, R.; Gonzalez, J.; Brito, R.; Hasson, D.; Libuy, M.; Ramos, C.; Carrasco, R.; Prieto, J.C.; et al. Amelioration of persistent left ventricular function impairment through increased plasma ascorbate levels following myocardial infarction. Redox Rep. 2016, 21, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ranadive, S.M.; Joyner, M.J.; Walker, B.G.; Taylor, J.L.; Casey, D.P. Effect of vitamin C on hyperoxia-induced vasoconstriction in exercising skeletal muscle. J. Appl. Physiol. 2014, 117, 1207–1211. [Google Scholar] [CrossRef] [PubMed]
- Barak, O.F.; Caljkusic, K.; Hoiland, R.L.; Ainslie, P.N.; Thom, S.R.; Yang, M.; Jovanov, P.; Dujic, Z. Differential influence of vitamin C on the peripheral and cerebral circulation after diving and exposure to hyperoxia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R759–R767. [Google Scholar] [CrossRef]
- Johnson, B.D.; Mather, K.J.; Newcomer, S.C.; Mickleborough, T.D.; Wallace, J.P. Vitamin C prevents the acute decline of flow-mediated dilation after altered shear rate patterns. Appl. Physiol. Nutr. Metab. 2013, 38, 268–274. [Google Scholar] [CrossRef]
- Thosar, S.S.; Bielko, S.L.; Wiggins, C.C.; Klaunig, J.E.; Mather, K.J.; Wallace, J.P. Antioxidant vitamin C prevents decline in endothelial function during sitting. Med. Sci. Monit. 2015, 21, 1015–1021. [Google Scholar]
- De Marchi, S.; Prior, M.; Rigoni, A.; Zecchetto, S.; Rulfo, F.; Arosio, E. Ascorbic acid prevents vascular dysfunction induced by oral glucose load in healthy subjects. Eur. J. Intern. Med. 2012, 23, 54–57. [Google Scholar] [CrossRef]
- Batista, G.M.S.; Rocha, H.N.M.; Storch, A.S.; Garcia, V.P.; Teixeira, G.F.; Mentzinger, J.; Gomes, E.A.C.; Velasco, L.L.; Nobrega, A.C.L.; Rocha, N.G. Ascorbic acid inhibits vascular remodeling induced by mental stress in overweight/obese men. Life Sci. 2020, 250, 117554. [Google Scholar] [CrossRef]
- Plotnick, M.D.; D’Urzo, K.A.; Gurd, B.J.; Pyke, K.E. The influence of vitamin C on the interaction between acute mental stress and endothelial function. Eur. J. Appl. Physiol. 2017, 117, 1657–1668. [Google Scholar] [CrossRef]
- Richards, J.C.; Crecelius, A.R.; Larson, D.G.; Dinenno, F.A. Acute ascorbic acid ingestion increases skeletal muscle blood flow and oxygen consumption via local vasodilation during graded handgrip exercise in older adults. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H360–H368. [Google Scholar] [CrossRef]
- Meredith, M.E.; Qu, Z.C.; May, J.M. Ascorbate reverses high glucose- and RAGE-induced leak of the endothelial permeability barrier. Biochem. Biophys. Res. Commun. 2014, 445, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.H.; Rhea, E.M.; Qu, Z.C.; Hecker, M.R.; May, J.M. Intracellular ascorbate tightens the endothelial permeability barrier through Epac1 and the tubulin cytoskeleton. Am. J. Physiol. Cell Physiol. 2016, 311, C652–C662. [Google Scholar] [CrossRef] [PubMed]
- Ulker, E.; Parker, W.H.; Raj, A.; Qu, Z.C.; May, J.M. Ascorbic acid prevents VEGF-induced increases in endothelial barrier permeability. Mol. Cell. Biochem. 2016, 412, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Chelazzi, C.; Villa, G.; Mancinelli, P.; De Gaudio, A.R.; Adembri, C. Glycocalyx and sepsis-induced alterations in vascular permeability. Crit. Care 2015, 19, 26. [Google Scholar] [CrossRef]
- Fernandez-Sarmiento, J.; Salazar-Pelaez, L.M.; Carcillo, J.A. The Endothelial Glycocalyx: A Fundamental Determinant of Vascular Permeability in Sepsis. Pediatr. Crit. Care Med. 2020, 21, e291–e300. [Google Scholar] [CrossRef]
- Kang, Q.; Chen, Y.; Zhang, X.; Yu, G.; Wan, X.; Wang, J.; Bo, L.; Zhu, K. Heat shock protein A12B protects against sepsis-induced impairment in vascular endothelial permeability. J. Surg. Res. 2016, 202, 87–94. [Google Scholar] [CrossRef]
- Li, Z.; Yin, M.; Zhang, H.; Ni, W.; Pierce, R.W.; Zhou, H.J.; Min, W. BMX Represses Thrombin-PAR1-Mediated Endothelial Permeability and Vascular Leakage During Early Sepsis. Circ. Res. 2020, 126, 471–485. [Google Scholar] [CrossRef]
- Mironova, G.D.; Belosludtseva, N.V.; Ananyan, M.A. Prospects for the use of regulators of oxidative stress in the comprehensive treatment of the novel Coronavirus Disease 2019 (COVID-19) and its complications. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 8585–8591. [Google Scholar]
- Schonrich, G.; Raftery, M.J.; Samstag, Y. Devilishly radical NETwork in COVID-19: Oxidative stress, neutrophil extracellular traps (NETs), and T cell suppression. Adv. Biol. Regul. 2020, 77, 100741. [Google Scholar] [CrossRef]
- Suhail, S.; Zajac, J.; Fossum, C.; Lowater, H.; McCracken, C.; Severson, N.; Laatsch, B.; Narkiewicz-Jodko, A.; Johnson, B.; Liebau, J.; et al. Role of Oxidative Stress on SARS-CoV (SARS) and SARS-CoV-2 (COVID-19) Infection: A Review. Protein J. 2020, 1–13. [Google Scholar] [CrossRef]
- Montel-Hagen, A.; Kinet, S.; Manel, N.; Mongellaz, C.; Prohaska, R.; Battini, J.L.; Delaunay, J.; Sitbon, M.; Taylor, N. Erythrocyte Glut1 triggers dehydroascorbic acid uptake in mammals unable to synthesize vitamin C. Cell 2008, 132, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Russo, T.A.; Kwon, O.; Chanock, S.; Rumsey, S.C.; Levine, M. Ascorbate recycling in human neutrophils: Induction by bacteria. Proc. Natl. Acad. Sci. USA 1997, 94, 13816–13819. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.R.; Kapoor, A.; Rath, M.; Thomas, S.A. What is the role of supplementation with ascorbic acid, zinc, vitamin D, or N-acetylcysteine for prevention or treatment of COVID-19? Clevel. Clin. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rowe, S. The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19. Nutrients 2020, 12, 3286. [Google Scholar] [CrossRef]
- Gillani, S.W.; Sulaiman, S.A.S.; Abdul, M.I.M.; Baig, M.R. Combined effect of metformin with ascorbic acid versus acetyl salicylic acid on diabetes-related cardiovascular complication; a 12-month single blind multicenter randomized control trial. Cardiovasc. Diabetol. 2017, 16, 103. [Google Scholar] [CrossRef]
- Ceriello, A.; Novials, A.; Ortega, E.; Canivell, S.; La Sala, L.; Pujadas, G.; Bucciarelli, L.; Rondinelli, M.; Genovese, S. Vitamin C further improves the protective effect of glucagon-like peptide-1 on acute hypoglycemia-induced oxidative stress, inflammation, and endothelial dysfunction in type 1 diabetes. Diabetes Care 2013, 36, 4104–4108. [Google Scholar] [CrossRef]
- Bae, M.; Kim, H. Mini-Review on the Roles of Vitamin C, Vitamin D, and Selenium in the Immune System against COVID-19. Molecules 2020, 25, 5346. [Google Scholar] [CrossRef]
- Bronte, V.; Zanovello, P. Regulation of immune responses by L-arginine metabolism. Nat. Rev. Immunol. 2005, 5, 641–654. [Google Scholar] [CrossRef]
- Wijnands, K.A.; Castermans, T.M.; Hommen, M.P.; Meesters, D.M.; Poeze, M. Arginine and citrulline and the immune response in sepsis. Nutrients 2015, 7, 1426–1463. [Google Scholar] [CrossRef]
- Kuck, J.L.; Bastarache, J.A.; Shaver, C.M.; Fessel, J.P.; Dikalov, S.I.; May, J.M.; Ware, L.B. Ascorbic acid attenuates endothelial permeability triggered by cell-free hemoglobin. Biochem. Biophys. Res. Commun. 2018, 495, 433–437. [Google Scholar] [CrossRef]
- Lundblad, C.; Bentzer, P. Effects of L-arginine on cerebral blood flow, microvascular permeability, number of perfused capillaries, and brain water content in the traumatized mouse brain. Microvasc. Res. 2007, 74, 1–8. [Google Scholar] [CrossRef] [PubMed]
- May, J.M.; Qu, Z.C. Nitric oxide mediates tightening of the endothelial barrier by ascorbic acid. Biochem. Biophys. Res. Commun. 2011, 404, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, T.; Date, C.; Kokubo, Y.; Yoshiike, N.; Matsumura, Y.; Tanaka, H. Serum vitamin C concentration was inversely associated with subsequent 20-year incidence of stroke in a Japanese rural community. The Shibata study. Stroke 2000, 31, 2287–2294. [Google Scholar] [CrossRef]
- Michels, A.J.; Frei, B. Myths, artifacts, and fatal flaws: Identifying limitations and opportunities in vitamin C research. Nutrients 2013, 5, 5161–5192. [Google Scholar] [CrossRef]
- Henriquez-Sanchez, P.; Sanchez-Villegas, A.; Doreste-Alonso, J.; Ortiz-Andrellucchi, A.; Pfrimer, K.; Serra-Majem, L. Dietary assessment methods for micronutrient intake: A systematic review on vitamins. Br. J. Nutr. 2009, 102, S10–S37. [Google Scholar] [CrossRef]
- Cahill, L.E.; El-Sohemy, A. Vitamin C transporter gene polymorphisms, dietary vitamin C and serum ascorbic acid. J. Nutrigenet. Nutrigenom. 2009, 2, 292–301. [Google Scholar] [CrossRef]
- Horska, A.; Mislanova, C.; Bonassi, S.; Ceppi, M.; Volkovova, K.; Dusinska, M. Vitamin C levels in blood are influenced by polymorphisms in glutathione S-transferases. Eur. J. Nutr. 2011, 50, 437–446. [Google Scholar] [CrossRef]
- Cahill, L.E.; El-Sohemy, A. Haptoglobin genotype modifies the association between dietary vitamin C and serum ascorbic acid deficiency. Am. J. Clin. Nutr. 2010, 92, 1494–1500. [Google Scholar] [CrossRef]
| Clinical Study | Study Population | Vitamin C Concentration Range | Fasting Blood Sample (Assay Used) | Adjustments | Outcome | Ref. |
|---|---|---|---|---|---|---|
| EPIC-Norfolk | 19,496 healthy men and women from Norfolk (US) | 25.9–79.2 μmol/L | Non-fasting venous blood (fluorometric assay) | Age, systolic BP, cholesterol, smoking, diabetes, and supplement use | A mean follow-up of 4 years showed that 20 μmol/L rising in plasma Vitamin C concentration was associated with ~20% reduced risk of all-cause mortality | [121,139] |
| EPIC-Norfolk | 20,299 healthy men and women from Norfolk (US) | 41–66 μmol/L | Non-fasting venous blood (fluorometric assay) | Age, sex, smoking, BMI, physical activity, systolic BP, diabetes, cholesterol, MI, social class, alcohol, and supplement used | A mean follow-up of 12.8 years showed that every 20 μmol/L increase in plasma Vitamin C was associated with a 9% relative reduction in the risk of HF. | [140] |
| HPFS—Health Professionals Follow-up Study | 39,910 healthy US male, health professionals | 95–116 mg/dL | Not reported | Age, season, smoking, energy intake, alcohol, hypertension, history of MI, profession, BMI, and physical activity | After a mean follow-up of 8 years, Vitamin C intake was not associated with a lower risk of CHD and stroke | [141] |
| NHS I—Nurses’ Health Study 1 | 85,118 healthy US female nurses | 70–704 mg/dL | Not reported | Age, energy intake, supplements use, alcohol, smoking status, and diabetes | Data from 16 years of follow-up indicate that Vitamin C supplement is associated with a significantly lower risk of CHD | [138] |
| ASAP—Antioxidant Supplementation in Atherosclerosis Prevention | 520 men and post-menopausal women, with hyper-cholesterolemia | Not specified | Not specified (HPLC assay) | Sex, baseline HDL cholesterol | A 6–year follow-up indicated that Vitamin C supplementation reduced the slope of the mean carotid artery intima-media thickness progression | [142] |
| Women’s Antioxidant Cardiovascular Study | 8171 female health professionals with CVD, or 3 or more CVD risk factors | Not specified | Not reported | Compliance-adjusted analyses | After a follow-up of 9.4 years, no overall effects of Vitamin C (500 mg/day) on all major cardiovascular events and stroke | [143] |
| NHANES II | 8417 US men and women | 17–79.5 μmol/L | Non-fasting venous blood (colorimetric assay) | Age, sex, race, education, physical activity, dietary energy and fat intake, cholesterol, BMI, alcohol, smoking, diabetes, hypertension, CHD, stroke, cancer, use of aspirin, Vitamin E intake | Decreased risk of all-cause mortality | [144] |
| NHANES III | 6624 US men and women | 17–85.2 μmol/L | Non-fasting venous blood (colorimetric assay) | Age, sex, race, education, physical activity, smoking, alcohol, cholesterol, BMI, diabetes, hypertension, use of aspirin, Vitamin E intake | Top Vitamin C group had a 27% lower CVD risk compared to bottom group | [145] |
| SU.VI.MAX study | 13,017 French adults | Not specified | Fasting venous blood (continuous flow segmented by air bubbles) | Bonferroni adjustment for multiple mean comparisons | After a follow-up of 7.5 years, no effect of Vitamin C supplementation (120 mg/d) on CVD; lower all-cause mortality in men | [146] |
| PHS II—Physicians Health Study II | 14,641 healthy US male physicians | Not specified | Not reported | Age, PHS cohort, randomized β–carotene, Vitamin E or Vitamin C, and multivitamin assignments | A mean follow-up of ~8 years showed that Vitamin C supplementation (500 mg/day) had no significant effect on the incidence of CVD, stroke, or mortality for cardiovascular events | [147] |
| Heart Protection Study | 20,536 British men and women with CHD, occlusive arterial disease, or diabetes | Not specified | Non-fasting venous blood | Not specified | After a follow-up of 5 years, Vitamin C supplementation (250 mg/d) had no significant effects on CVD | [148] |
| CARDIA | 2884 healthy US men and women | 24–70 μmol/L | Fasting venous blood (HPLC assay) | Age, sex, race, center, education, smoking, alcohol, physical activity, a priori diet quality score and food consumption, BMI, waist circumference, history of diabetes, systolic BP | Higher plasma Vitamin C, due to better diet quality, is inversely related to high BP. | [149] |
| Iowa Women’s Health Study | 1923 post-menopausal diabetic women from U.S. (55 to 69 years) | 82–≥679 mg/day | Not reported | Age, total energy intake, history of hypertension, BMI, waist-hip ratio, smoking status and amount smoked, alcohol consumption, physical activity score, hormone replacement therapy, main type and duration of diabetes, medications | After a follow-up of 11 years, an association between increased mortality for CVD or CHD and Vitamin C ≥ 300 mg/day was reported. Vitamin C intake was unrelated to CVD and stroke mortality in nondiabetic women. | [150] |
| Italian section of the EPIC Study | 41,620 Italians previously diagnosed with stroke or myocardial infarction (44 to 61 years) | 83–201 mg/day | Not reported | Age, sex, hypertension, smoking, alcohol, education, energy intake, waist circumference, obesity, and physical activity | After 7.9 years of follow-up, Vitamin C was associated with a lower risk of ischemic stroke | [151] |
| Meta-Analysis Topic | Number of Studies | Total Participants (n) | Trial Duration | Vitamin C Supplement Dosage | Outcome | Ref. |
|---|---|---|---|---|---|---|
| Coronary Heart Disease | 14 cohorts | 374,488 | mean duration: 10 y. | Not reported | Dietary Vitamin C had an inverse association with CHD risk, but supplement intake of Vitamin C had no significant association with CHD risk. | [154] |
| Hypertension | 29 trials | 1407 | mean duration: 8 wks. | 60 to 4000 mg/day (median 500 mg/day) | Daily supplementation of Vitamin C in hypertensive participants reduced systolic BP by 3.84 mm Hg and DBP by 1.48 mm Hg. | [159] |
| Coronary Heart Disease | 15 trials | 188,209 | from 1.4 y. to 12 y. | 120 to 1000 mg/day | Vitamin C in association with Vitamin E and beta-carotene had no effect on CHD or other major cardiovascular events. Vitamin C in association with Vitamin E and beta-carotene had no effect on stroke risk compared to placebo. | [156] |
| Stroke | 16 cohorts | 217,454 participants for studies on Vitamin C intake 29,648 participants for studies on circulating Vitamin C | from 6.1 y. to 30 y. | 45 to 1167 mg/day | 19% lower risk of stroke (pooled analysis of 2 studies) comparing the highest to the lowest dietary Vitamin C intakes. 38% lower risk of stroke (pooled analysis of 6 studies) with the highest versus lowest circulating Vitamin C concentrations. | [160] |
| Endothelial function | 44 trials | 1324 | from 1 d. to 8 wks. | 500 to 2000 mg/day | A significant positive effect of Vitamin C supplementation on endothelial function measured by flow-mediated dilation (19 trials), evaluating pulse-wave (5 trials) and forearm blood flow (20 trials). | [161] |
| Arterial stiffness | 22 trials | 1909 | from 1 d. to 7 y. (mean duration: 56 d.) | 120 to 4000 mg/day (mean 2000 mg/day) | Association of Vitamin C with Vitamin E and β–carotene improved arterial stiffness irrespective of the age group | [161] |
| Cardiovascular events | 8 trials | 15,445 | mean duration: 8 y. | – | Inconsistent or low-quality (downgraded) evidence for the correlation between Vitamin C supplementation and major cardiovascular events or cardiovascular mortality | [157] |
| Hypertension | 11 cross-sectional studies and 7 case-control studies | 22,200 | – | – | Serum Vitamin C level was significantly lower in hypertensive compared to normotensive subjects. Inverse correlation between serum Vitamin C concentration and SBP or DBP. | [162] |
| Hypertension | 8 trials | 614 | from 4 wks. to 24 wks. | 300 to 1000 mg/day | Significantly reduced SBP and DBP in the subgroups of age ≥35 and ≥60 years with Vitamin C supplementation ≥500 mg/day for 6 weeks or more. | [163] |
| Cardiovascular mortality | 15 trials | 320,548 | from 4 to 22y. | 50 to 450 mg/day | Higher Vitamin C intake is associated with a lower risk of cardiovascular mortality | [164] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morelli, M.B.; Gambardella, J.; Castellanos, V.; Trimarco, V.; Santulli, G. Vitamin C and Cardiovascular Disease: An Update. Antioxidants 2020, 9, 1227. https://doi.org/10.3390/antiox9121227
Morelli MB, Gambardella J, Castellanos V, Trimarco V, Santulli G. Vitamin C and Cardiovascular Disease: An Update. Antioxidants. 2020; 9(12):1227. https://doi.org/10.3390/antiox9121227
Chicago/Turabian StyleMorelli, Marco B., Jessica Gambardella, Vanessa Castellanos, Valentina Trimarco, and Gaetano Santulli. 2020. "Vitamin C and Cardiovascular Disease: An Update" Antioxidants 9, no. 12: 1227. https://doi.org/10.3390/antiox9121227
APA StyleMorelli, M. B., Gambardella, J., Castellanos, V., Trimarco, V., & Santulli, G. (2020). Vitamin C and Cardiovascular Disease: An Update. Antioxidants, 9(12), 1227. https://doi.org/10.3390/antiox9121227