Effects of Rutin on Wound Healing in Hyperglycemic Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Animal Feeding and Induction of Hyperglycemia
2.3. Animal Wound Healing
2.4. Blood Samples
2.5. Perfusion and Tissue Preparation
2.6. Histopathology and Staining
2.7. Immunohistochemical Staining
2.8. Immunofluorescence Staining
2.9. Preparation of Protein (Protein Lysate) and Western Blot Analysis
2.10. Statistical Analysis
3. Results
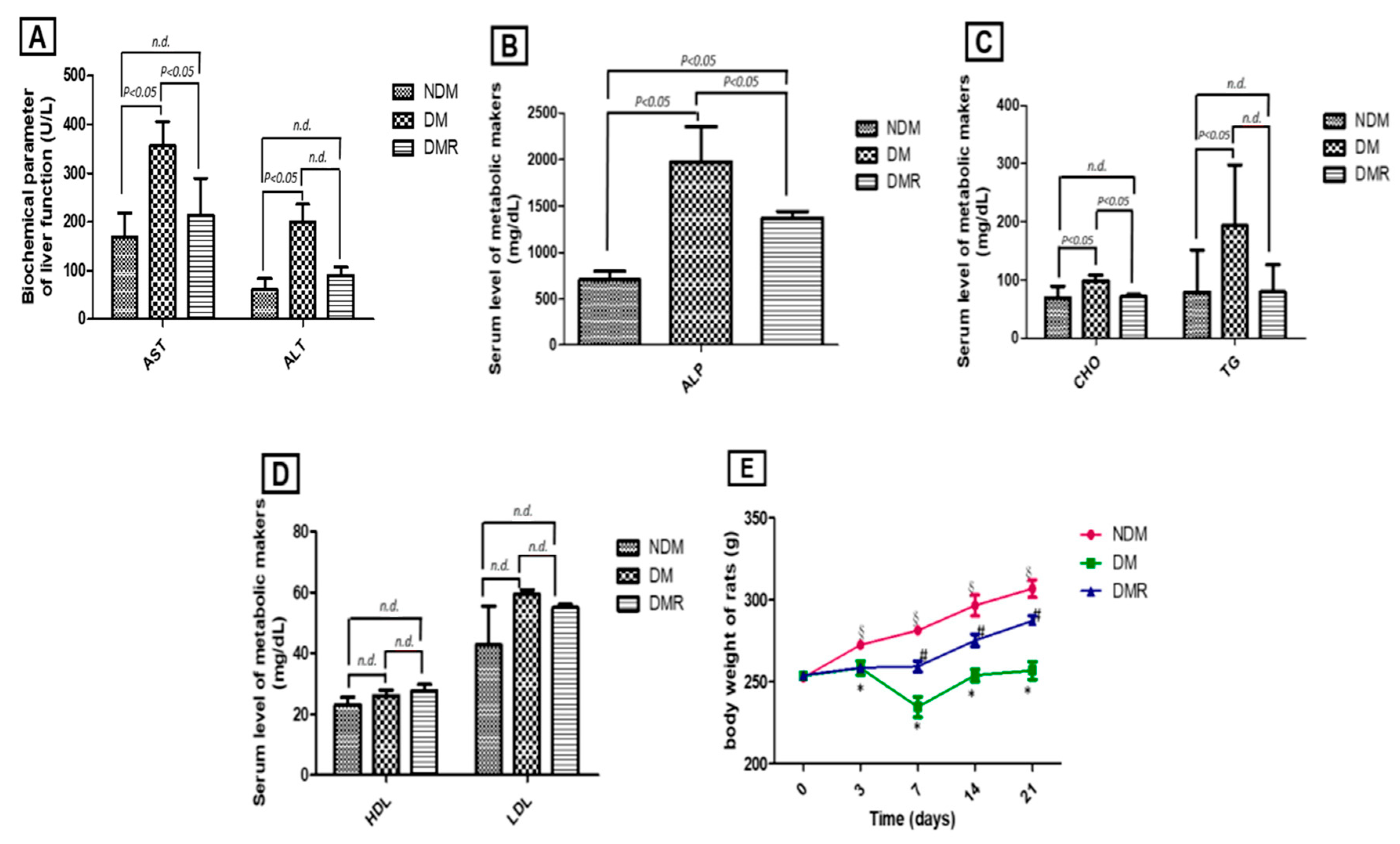
3.1. Rutin Improves Liver Function, Blood Lipid Profile, and Body Weight
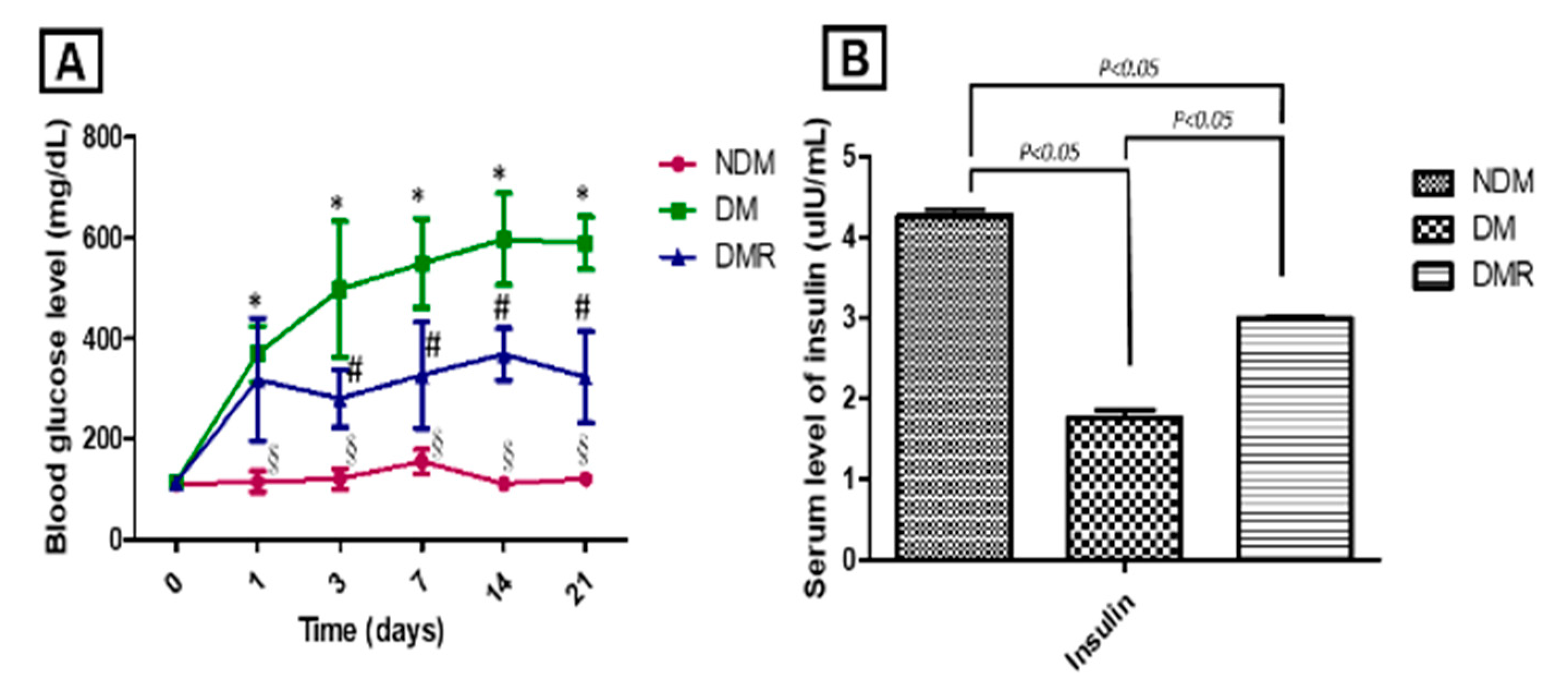
3.2. Rutin Improves Blood Sugar and Maintains Pancreatic Function in STZ-Induced Hyperglycemic Rats
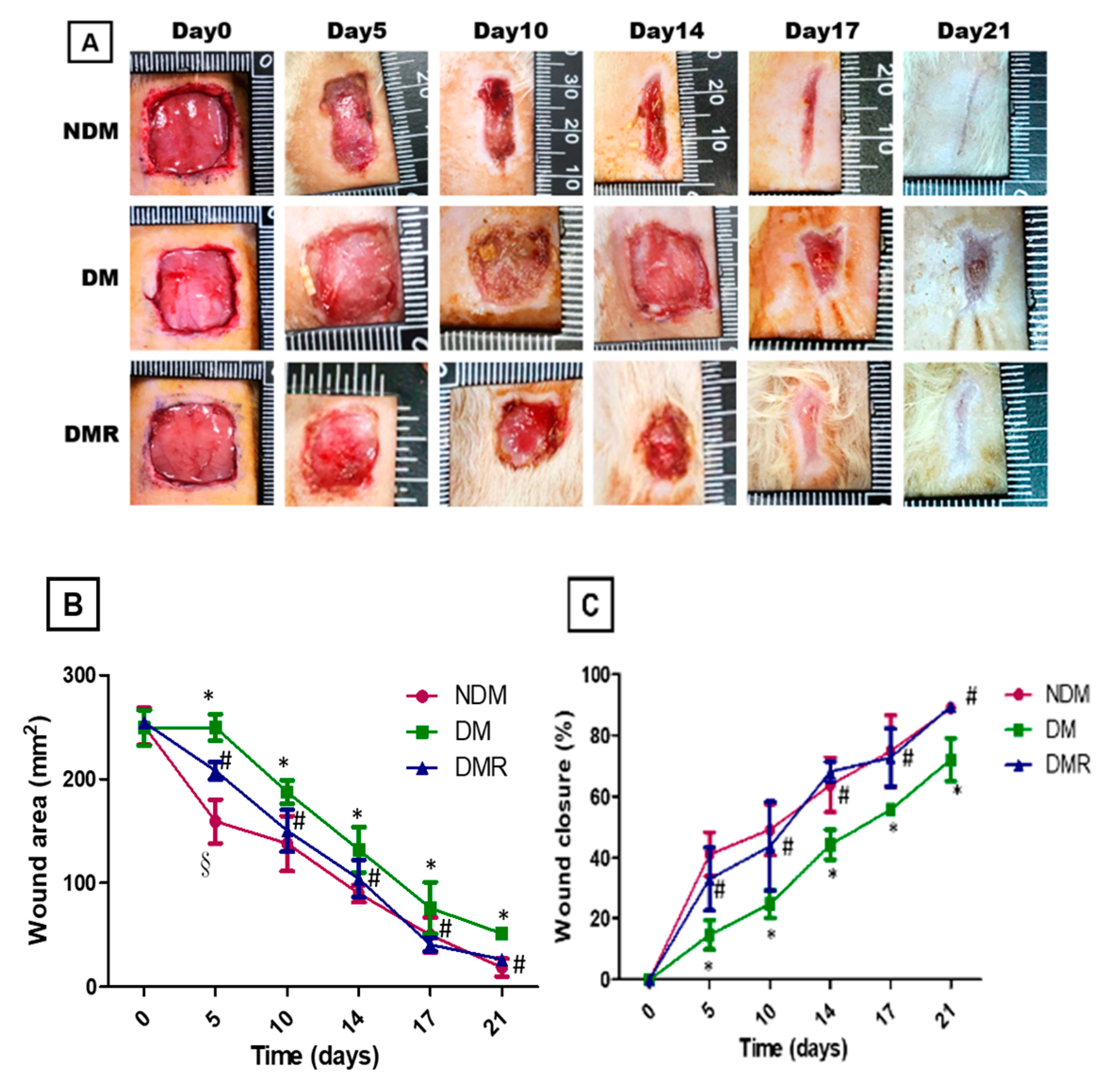
3.3. Rutin Improves Wound Healing in STZ-Induced Hyperglycemic Rats
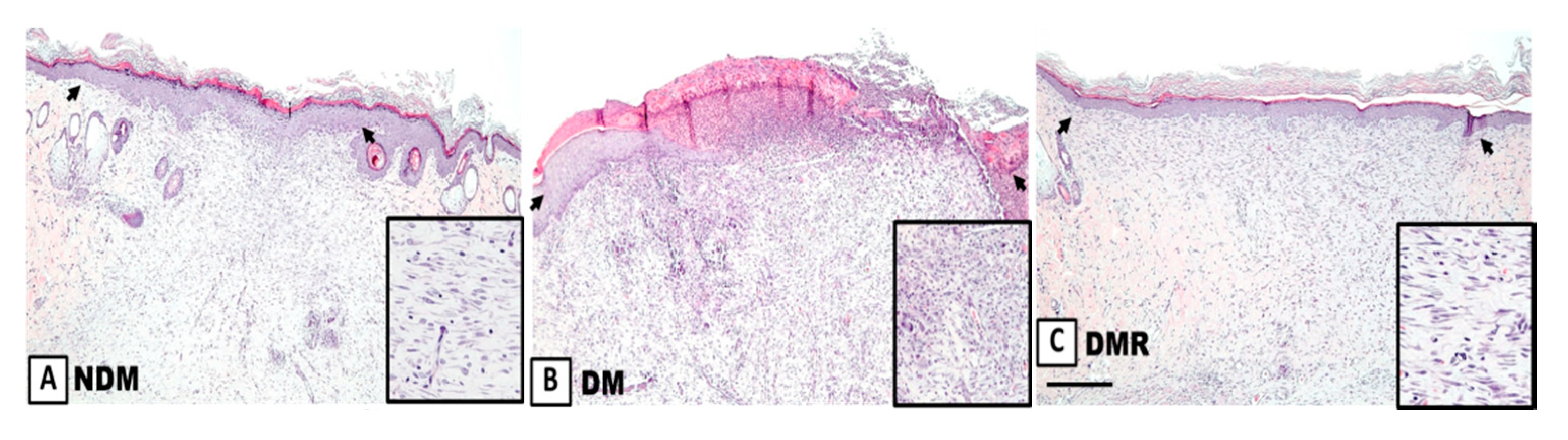
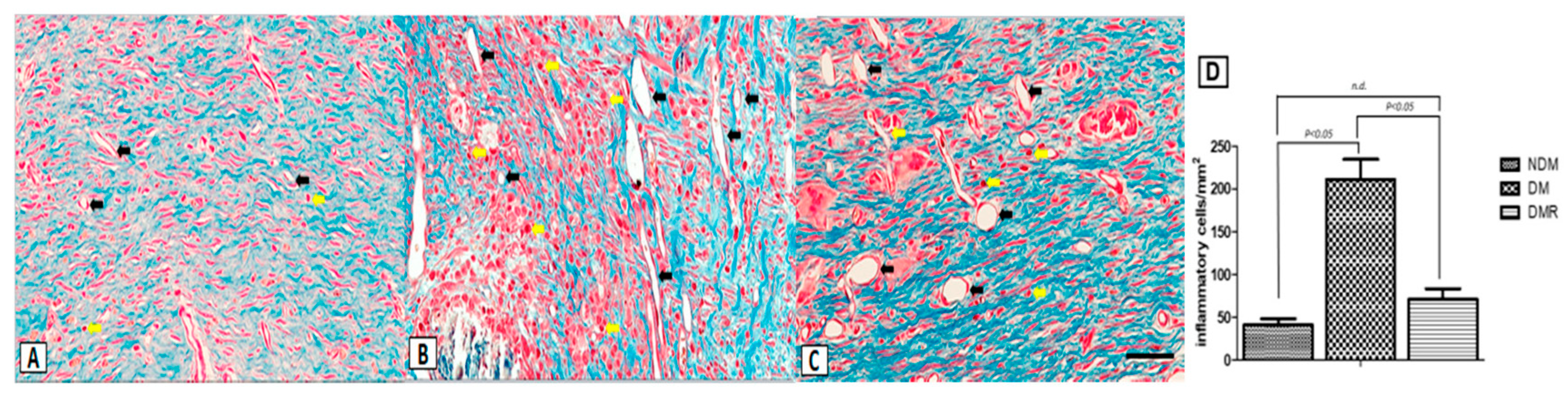
3.4. Rutin Improves Proliferation of Collagen Fibers and Reduces Expression of Inflammatory Cells in the Wounds of STZ-Induced Hyperglycemic Rats
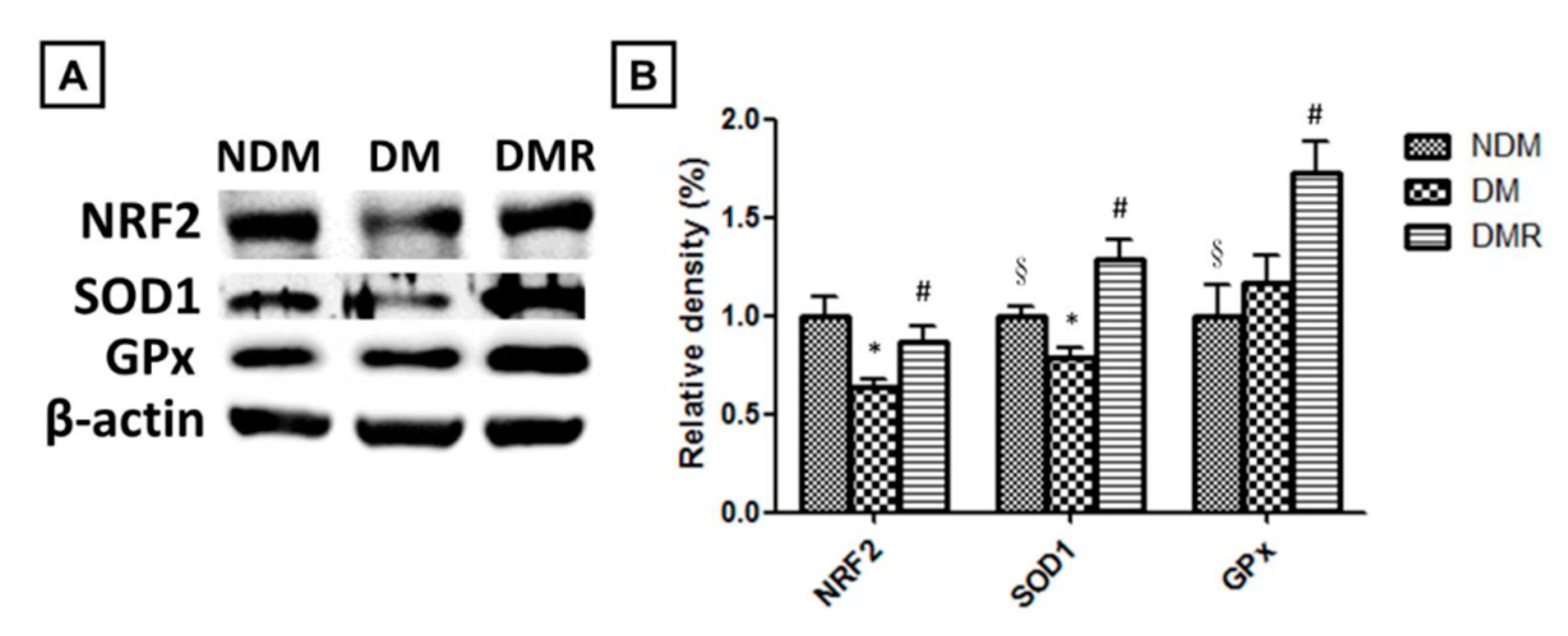
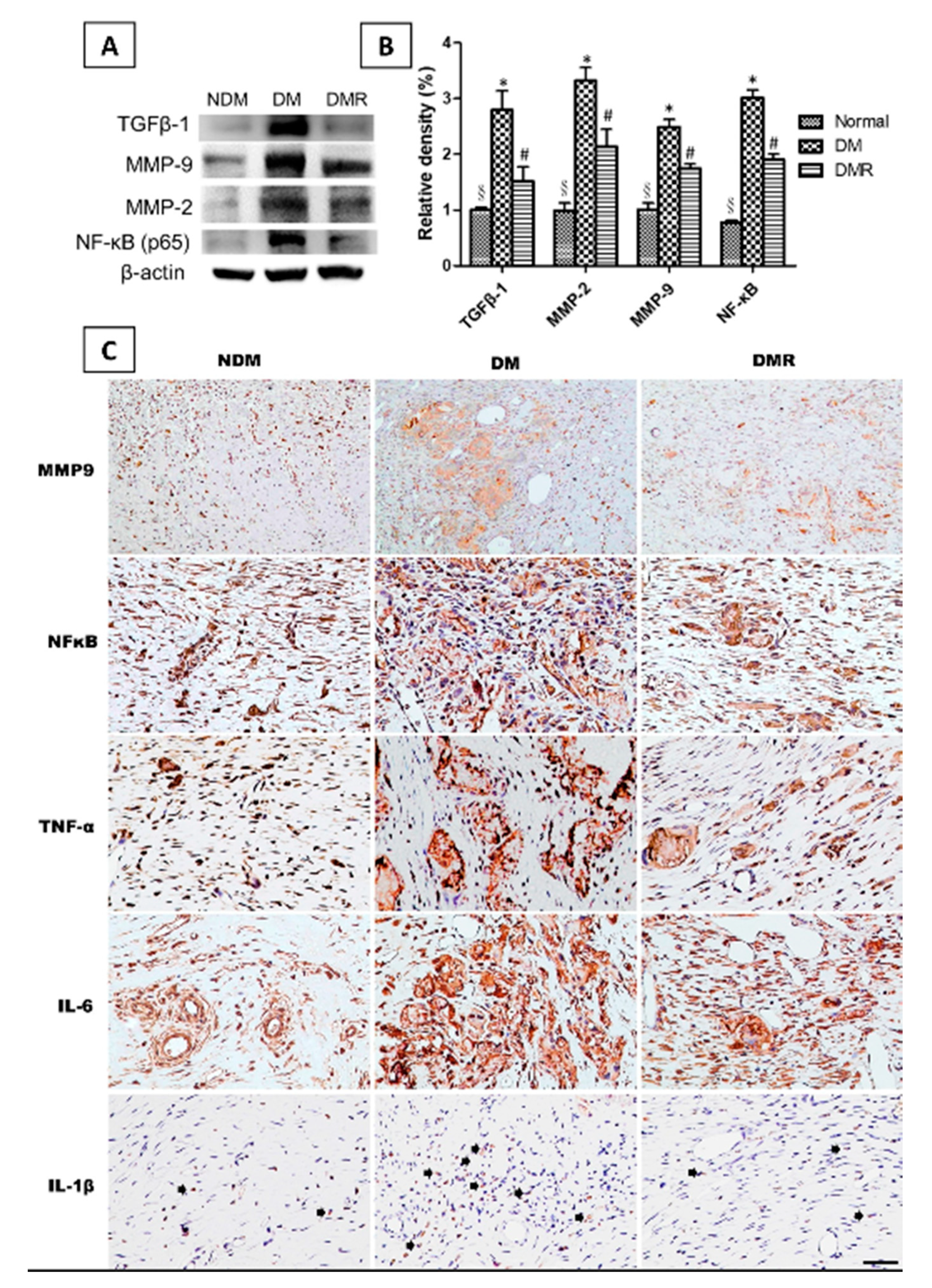
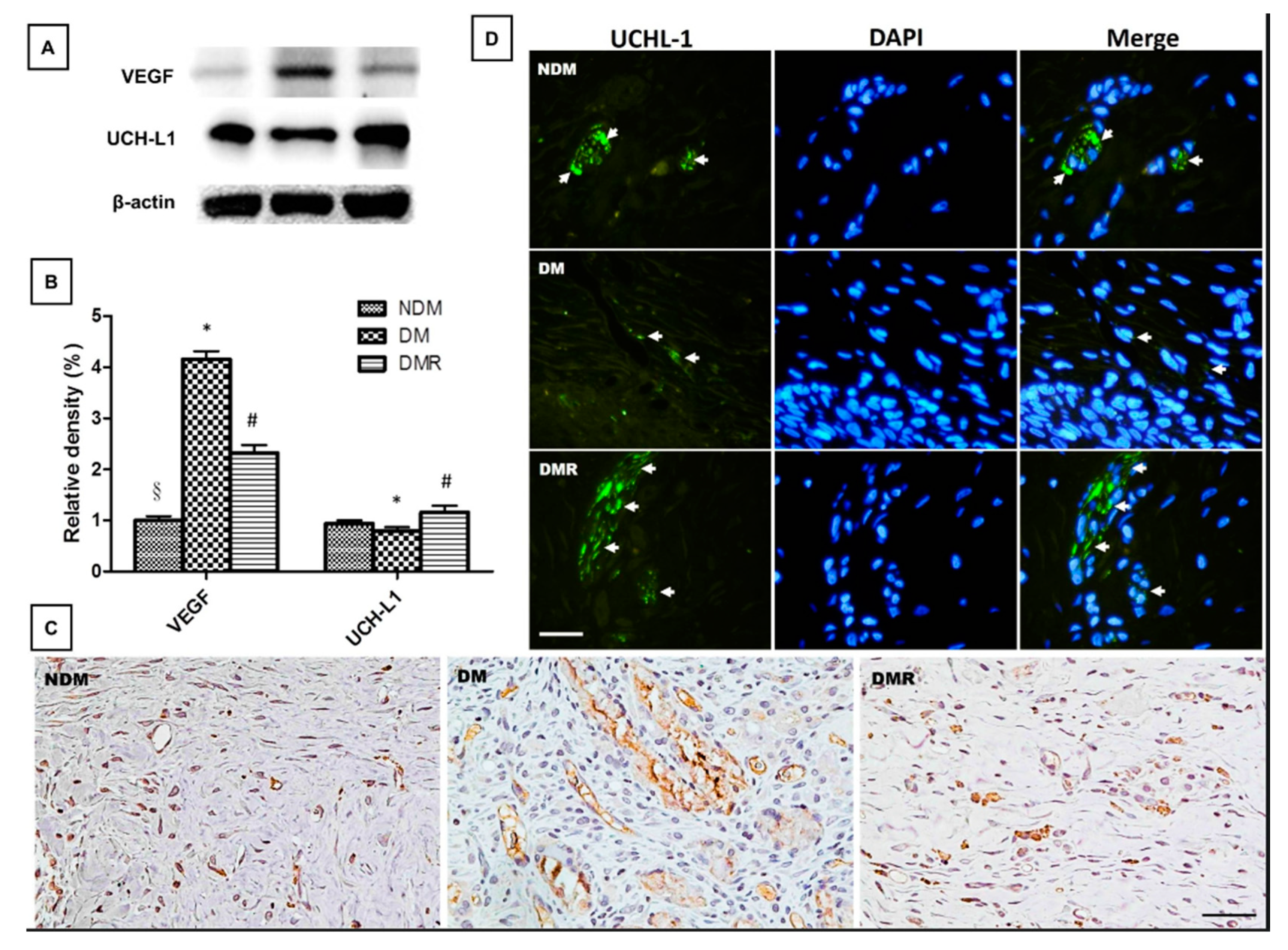
3.5. Rutin Effectively Promotes NRF2, Targets Downstream Antioxidant Enzyme Activities, Suppresses Inflammation-Related Factors, and Facilitates Nerve Growth during Wound Healing
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Federation, Identiy. IDF Diabetes Atlas. 2019. Available online: https://diabetesatlas.org/en/sections/worldwide-toll-of-diabetes.html (accessed on 23 June 2020).
- Ekmektzoglou, K.A.; Zografos, G.C. A concomitant review of the effects of diabetes mellitus and hypothyroidism in wound healing. World J. Gastroenterol. 2006, 12, 2721–2729. [Google Scholar] [CrossRef] [PubMed]
- Elbatreek, M.H.; Pachado, M.P.; Cuadrado, A.; Jandeleit-Dahm, K.; Schmidt, H. Reactive Oxygen Comes of Age: Mechanism-Based Therapy of Diabetic End-Organ Damage. Trends Endocrinol. Metab. 2019, 30, 312–327. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Chang, M. Restructuring of the extracellular matrix in diabetic wounds and healing: A perspective. Pharm. Res. 2016, 107, 243–248. [Google Scholar] [CrossRef]
- Sun, B.K.; Siprashvili, Z.; Khavari, P.A. Advances in skin grafting and treatment of cutaneous wounds. Science 2014, 346, 941–945. [Google Scholar] [CrossRef]
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. The Role of Chemokines in Wound Healing. Int. J. Mol. Sci. 2018, 19, 3217. [Google Scholar] [CrossRef]
- Jaiswal, S.S.; Gambhir, R.P.; Agrawal, A.; Harish, S. Efficacy of topical recombinant human platelet derived growth factor on wound healing in patients with chronic diabetic lower limb ulcers. Indian J. Surg. 2010, 72, 27–31. [Google Scholar] [CrossRef]
- Park, J.W.; Hwang, S.R.; Yoon, I.S. Advanced Growth Factor Delivery Systems in Wound Management and Skin Regeneration. Molecules 2017, 22, 1259. [Google Scholar] [CrossRef]
- Krzyszczyk, P.; Schloss, R.; Palmer, A.; Berthiaume, F. The Role of Macrophages in Acute and Chronic Wound Healing and Interventions to Promote Pro-wound Healing Phenotypes. Front. Physiol. 2018, 9, 419. [Google Scholar] [CrossRef]
- Matsubara, Y.; Kumamoto, H.; Iizuka, Y.; Murakami, T.; Okamoto, K.; Miyake, H.; Yokoi, K. Structure and Hypotensive Effect of Flavonoid Glycosides in Citrus unshiu Peelings. Agric. Biol. Chem. 1985, 49, 909–914. [Google Scholar]
- Kreft, S.; Knapp, M.; Kreft, I. Extraction of Rutin from Buckwheat (Fagopyrum esculentum Moench) Seeds and Determination by Capillary Electrophoresis. J. Agric. Food Chem. 1999, 47, 4649–4652. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.Z.; Feng, M.L.; Hao, X.L.; Zhong, Q.M.; Tong, L.G.; Wang, Z.H. Rutin, quercetin, and free amino acid analysis in buckwheat (Fagopyrum) seeds from different locations. Genet. Mol. Res. 2015, 14, 19040–19048. [Google Scholar] [CrossRef] [PubMed]
- Nafees, S.; Rashid, S.; Ali, N.; Hasan, S.K.; Sultana, S. Rutin ameliorates cyclophosphamide induced oxidative stress and inflammation in Wistar rats: Role of NFkappaB/MAPK pathway. Chem. Biol. Interact. 2015, 231, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Ganeshpurkar, A.; Saluja, A.K. The Pharmacological Potential of Rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Aruna, R.; Geetha, A.; Suguna, P.; Suganya, V. Rutin rich Emblica officinalis Geart. fruit extract ameliorates inflammation in the pancreas of rats subjected to alcohol and cerulein administration. J. Complement. Integr. Med. 2014, 11, 9–18. [Google Scholar] [CrossRef]
- Song, K.; Na, J.Y.; Kim, S.; Kwon, J. Rutin upregulates neurotrophic factors resulting in attenuation of ethanol-induced oxidative stress in HT22 hippocampal neuronal cells. J. Sci. Food Agric. 2015, 95, 2117–2123. [Google Scholar] [CrossRef]
- Shenbagam, M.; Nalini, N. Dose response effect of rutin a dietary antioxidant on alcohol-induced prooxidant and antioxidant imbalance—A histopathologic study. Fundam. Clin. Pharmacol. 2011, 25, 493–502. [Google Scholar] [CrossRef]
- Ziaee, A.; Zamansoltani, F.; Nassiri-Asl, M.; Abbasi, E. Effects of rutin on lipid profile in hypercholesterolaemic rats. Basic Clin. Pharm. Toxicol. 2009, 104, 253–258. [Google Scholar] [CrossRef]
- Niture, N.T.; Ansari, A.A.; Naik, S.R. Anti-hyperglycemic activity of rutin in streptozotocin-induced diabetic rats: An effect mediated through cytokines, antioxidants and lipid biomarkers. Indian J. Exp. Biol. 2014, 52, 720–727. [Google Scholar]
- Qinna, N.A.; Badwan, A.A. Impact of streptozotocin on altering normal glucose homeostasis during insulin testing in diabetic rats compared to normoglycemic rats. Drug Des. Dev. 2015, 9, 2515–2525. [Google Scholar] [CrossRef]
- Cheng, K.-Y.; Lin, Z.-H.; Cheng, Y.-P.; Chiu, H.-Y.; Yeh, N.-L.; Wu, T.-K.; Wu, J.-S. Wound Healing in Streptozotocin-Induced Diabetic Rats Using Atmospheric-Pressure Argon Plasma Jet. Sci. Rep. 2018, 8, 12214. [Google Scholar] [CrossRef] [PubMed]
- Ugwu, E.; Adeleye, O.; Gezawa, I.; Okpe, I.; Enamino, M.; Ezeani, I. Predictors of lower extremity amputation in patients with diabetic foot ulcer: Findings from MEDFUN, a multi-center observational study. J. Foot Ankle Res. 2019, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Hunt, N.A.; Ndip, A.; Lavery, D.C.; Van Houtum, W.; Boulton, A.J. Impact of chronic kidney disease on survival after amputation in individuals with diabetes. Diabetes Care 2010, 33, 2365–2369. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.M.D.; Pereira Leal, A.E.B.; Silva, J.C.; Almeida, J.; de Oliveira, H.P. Influence of Flavonoids on Mechanism of Modulation of Insulin Secretion. Pharm. Mag. 2017, 13, 639–646. [Google Scholar]
- Stanley Mainzen Prince, P.; Kamalakkannan, N. Rutin improves glucose homeostasis in streptozotocin diabetic tissues by altering glycolytic and gluconeogenic enzymes. J. Biochem. Mol. Toxicol. 2006, 20, 96–102. [Google Scholar] [CrossRef]
- Su, W.; Cao, R.; He, Y.C.; Guan, Y.F.; Ruan, X.Z. Crosstalk of Hyperglycemia and Dyslipidemia in Diabetic Kidney Disease. Kidney Dis. (Basel Switz.) 2017, 3, 171–180. [Google Scholar] [CrossRef]
- Femlak, M.; Gluba-Brzózka, A.; Ciałkowska-Rysz, A.; Rysz, J. The role and function of HDL in patients with diabetes mellitus and the related cardiovascular risk. Lipids Health Dis. 2017, 16, 207. [Google Scholar] [CrossRef]
- Goldberg, I.J. Diabetic Dyslipidemia: Causes and Consequences. J. Clin. Endocrinol. Metab. 2001, 86, 965–971. [Google Scholar] [CrossRef]
- Cano Sanchez, M.; Lancel, S.; Boulanger, E.; Neviere, R. Targeting Oxidative Stress and Mitochondrial Dysfunction in the Treatment of Impaired Wound Healing: A Systematic Review. Antioxidants (Basel) 2018, 7, 98. [Google Scholar] [CrossRef]
- Francisqueti-Ferron, F.V.; Ferron, A.J.T.; Garcia, J.L.; Silva, C.; Costa, M.R.; Gregolin, C.S.; Moreto, F.; Ferreira, A.L.A.; Minatel, I.O.; Correa, C.R. Basic Concepts on the Role of Nuclear Factor Erythroid-Derived 2-Like 2 (Nrf2) in Age-Related Diseases. Int. J. Mol. Sci. 2019, 20, 3208. [Google Scholar] [CrossRef]
- Xian, D.; Xiong, X.; Xu, J.; Xian, L.; Lei, Q.; Song, J.; Zhong, J. Nrf2 Overexpression for the Protective Effect of Skin-Derived Precursors against UV-Induced Damage: Evidence from a Three-Dimensional Skin Model. Oxid. Med. Cell. Longev. 2019, 2019, 7021428. [Google Scholar] [CrossRef] [PubMed]
- Beserra, F.P.; Vieira, A.J.; Gushiken, L.F.S.; de Souza, E.O.; Hussni, M.F.; Hussni, C.A.; Nobrega, R.H.; Martinez, E.R.M.; Jackson, C.J.; de Azevedo Maia, G.L.; et al. Lupeol, a Dietary Triterpene, Enhances Wound Healing in Streptozotocin-Induced Hyperglycemic Rats with Modulatory Effects on Inflammation, Oxidative Stress, and Angiogenesis. Oxid. Med. Cell. Longev. 2019, 2019, 3182627. [Google Scholar] [CrossRef] [PubMed]
- Wardyn, J.D.; Ponsford, A.H.; Sanderson, C.M. Dissecting molecular cross-talk between Nrf2 and NF-κB response pathways. Biochem. Soc. Trans. 2015, 43, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.-M.; Lin, H.-C.; Chang, W.-C. Carnosic acid prevents the migration of human aortic smooth muscle cells by inhibiting the activation and expression of matrix metalloproteinase-9. Br. J. Nutr. 2008, 100, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.M.; Lin, H.C. Curcumin prevents human aortic smooth muscle cells migration by inhibiting of MMP-9 expression. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Mobashery, S.; Chang, M. Roles of Matrix Metalloproteinases in Cutaneous Wound Healing. In Wound Healing-New Insights into Ancient Challenges; IntechOpen: London, UK, 2016. [Google Scholar]
- Jude, E.B.; Blakytny, R.; Bulmer, J.; Boulton, A.J.; Ferguson, M.W. Transforming growth factor-beta 1, 2, 3 and receptor type I and II in diabetic foot ulcers. Diabet. Med. A J. Br. Diabet. Assoc. 2002, 19, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J.; Han, G.; Owens, P.; Siddiqui, Y.; Li, A.G. Role of TGFβ-Mediated Inflammation in Cutaneous Wound Healing. J. Investig. Dermatol. Symp. Proc. 2006, 11, 112–117. [Google Scholar] [CrossRef]
- Chawla, A.; Chawla, R.; Jaggi, S. Microvasular and macrovascular complications in diabetes mellitus: Distinct or continuum? Indian J. Endocrinol. Metab. 2016, 20, 546–551. [Google Scholar] [CrossRef]
- Eming, S.A.; Krieg, T.; Davidson, J.M. Inflammation in wound repair: Molecular and cellular mechanisms. J. Investig. Dermatol. 2007, 127, 514–525. [Google Scholar] [CrossRef]
- Deryugina, E.I.; Quigley, J.P. Tumor angiogenesis: MMP-mediated induction of intravasation- and metastasis-sustaining neovasculature. Matrix Biol. 2015, 44–46, 94–112. [Google Scholar] [CrossRef]
- Ansel, J.C. Neuromediators and inflammation. Exp. Dermatol. 2001, 10, 352–353. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.J.; Doran, J.F.; Jackson, P.; Dhillon, A.P.; Rode, J. PGP 9.5—A new marker for vertebrate neurons and neuroendocrine cells. Brain Res. 1983, 278, 224–228. [Google Scholar] [CrossRef]
- Day, I.N.M.; Thompson, R.J. UCHL1 (PGP 9.5): Neuronal biomarker and ubiquitin system protein. Prog. Neurobiol. 2010, 90, 327–362. [Google Scholar] [CrossRef] [PubMed]
- Campbell, L.K.; Thomas, J.R.; Lamps, L.W.; Smoller, B.R.; Folpe, A.L. Protein Gene Product 9.5 (PGP 9.5) Is Not a Specific Marker of Neural and Nerve Sheath Tumors: An Immunohistochemical Study of 95 Mesenchymal Neoplasms. Mod. Pathol. 2003, 16, 963–969. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.-Y.; Huang, C.-N.; Liao, C.-K.; Chang, H.-M.; Kuan, Y.-H.; Tseng, T.-J.; Yen, K.-J.; Yang, K.-L.; Lin, H.-C. Effects of Rutin on Wound Healing in Hyperglycemic Rats. Antioxidants 2020, 9, 1122. https://doi.org/10.3390/antiox9111122
Chen L-Y, Huang C-N, Liao C-K, Chang H-M, Kuan Y-H, Tseng T-J, Yen K-J, Yang K-L, Lin H-C. Effects of Rutin on Wound Healing in Hyperglycemic Rats. Antioxidants. 2020; 9(11):1122. https://doi.org/10.3390/antiox9111122
Chicago/Turabian StyleChen, Li-You, Chien-Ning Huang, Chih-Kai Liao, Hung-Ming Chang, Yu-Hsiang Kuan, To-Jung Tseng, Kai-Jung Yen, Kai-Lin Yang, and Hsing-Chun Lin. 2020. "Effects of Rutin on Wound Healing in Hyperglycemic Rats" Antioxidants 9, no. 11: 1122. https://doi.org/10.3390/antiox9111122
APA StyleChen, L.-Y., Huang, C.-N., Liao, C.-K., Chang, H.-M., Kuan, Y.-H., Tseng, T.-J., Yen, K.-J., Yang, K.-L., & Lin, H.-C. (2020). Effects of Rutin on Wound Healing in Hyperglycemic Rats. Antioxidants, 9(11), 1122. https://doi.org/10.3390/antiox9111122






