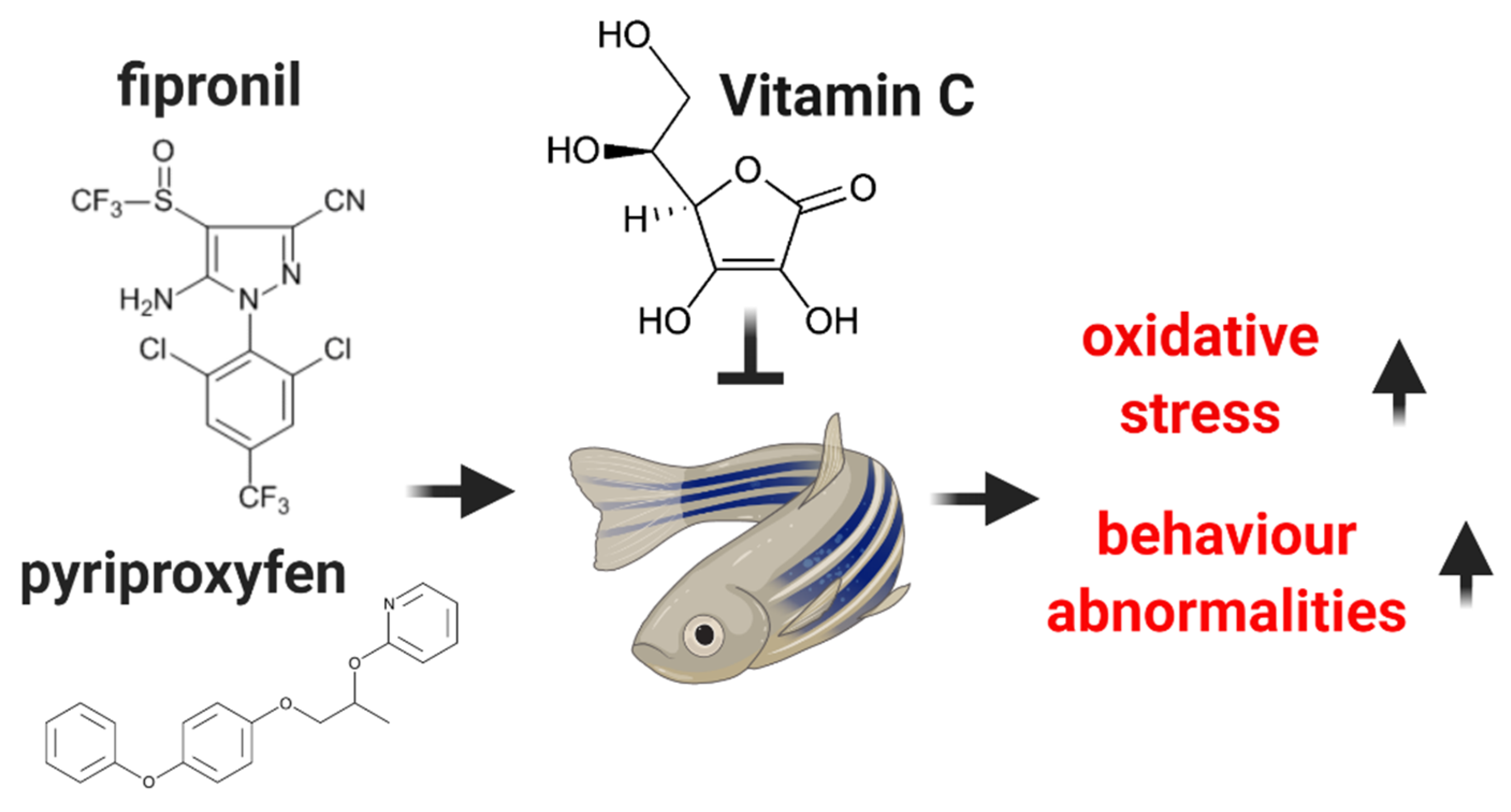
Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Note
2.2. Chemicals
2.3. Animal Housing
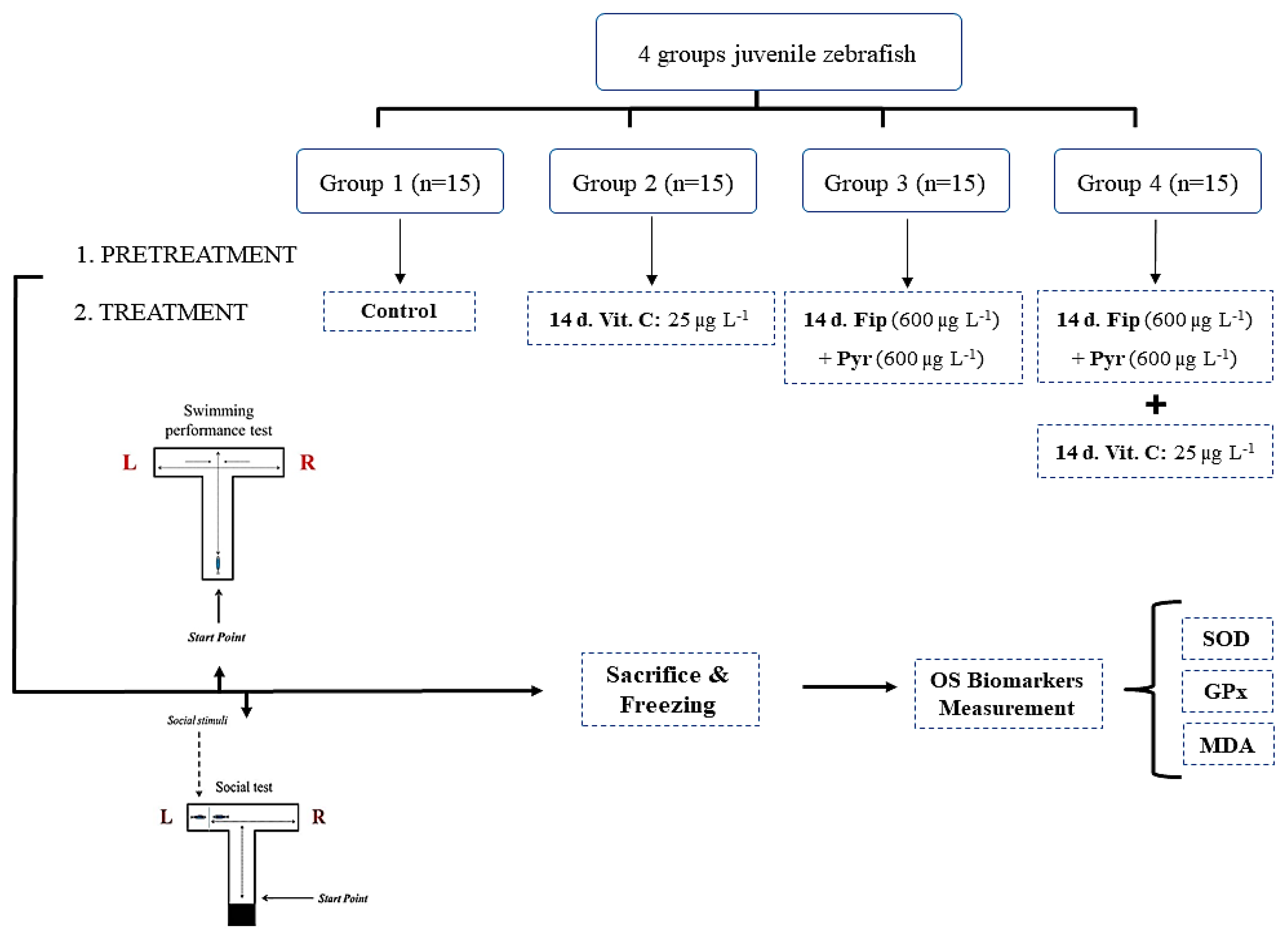
2.4. Experimental Design and Chronic Exposure
2.5. Behavioral Assessment
2.6. Oxidative Stress Evaluation
2.7. Statistical Analysis
3. Results
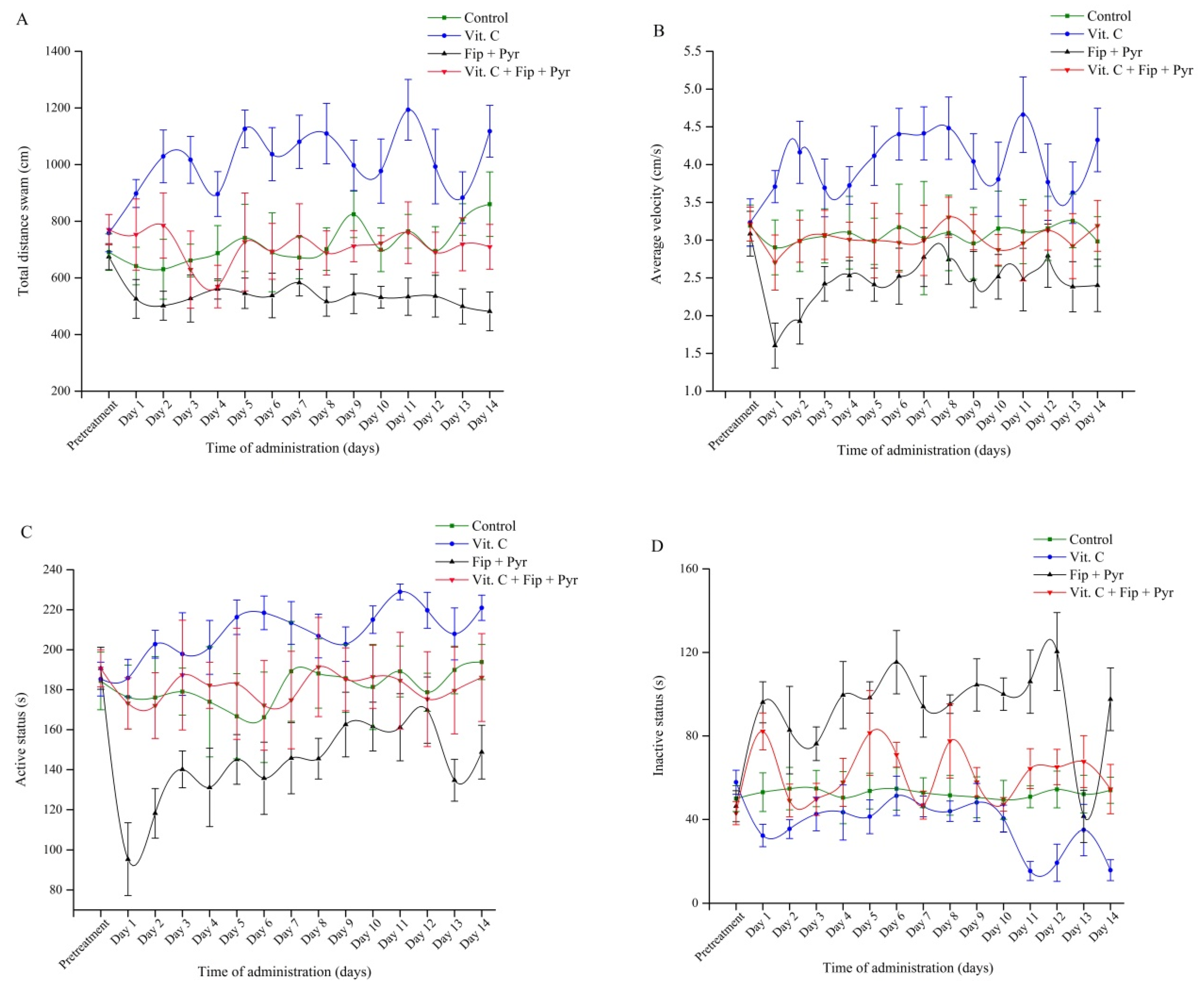
3.1. The Effects of Vitamin C in Insecticide Mixture on Swimming Performances
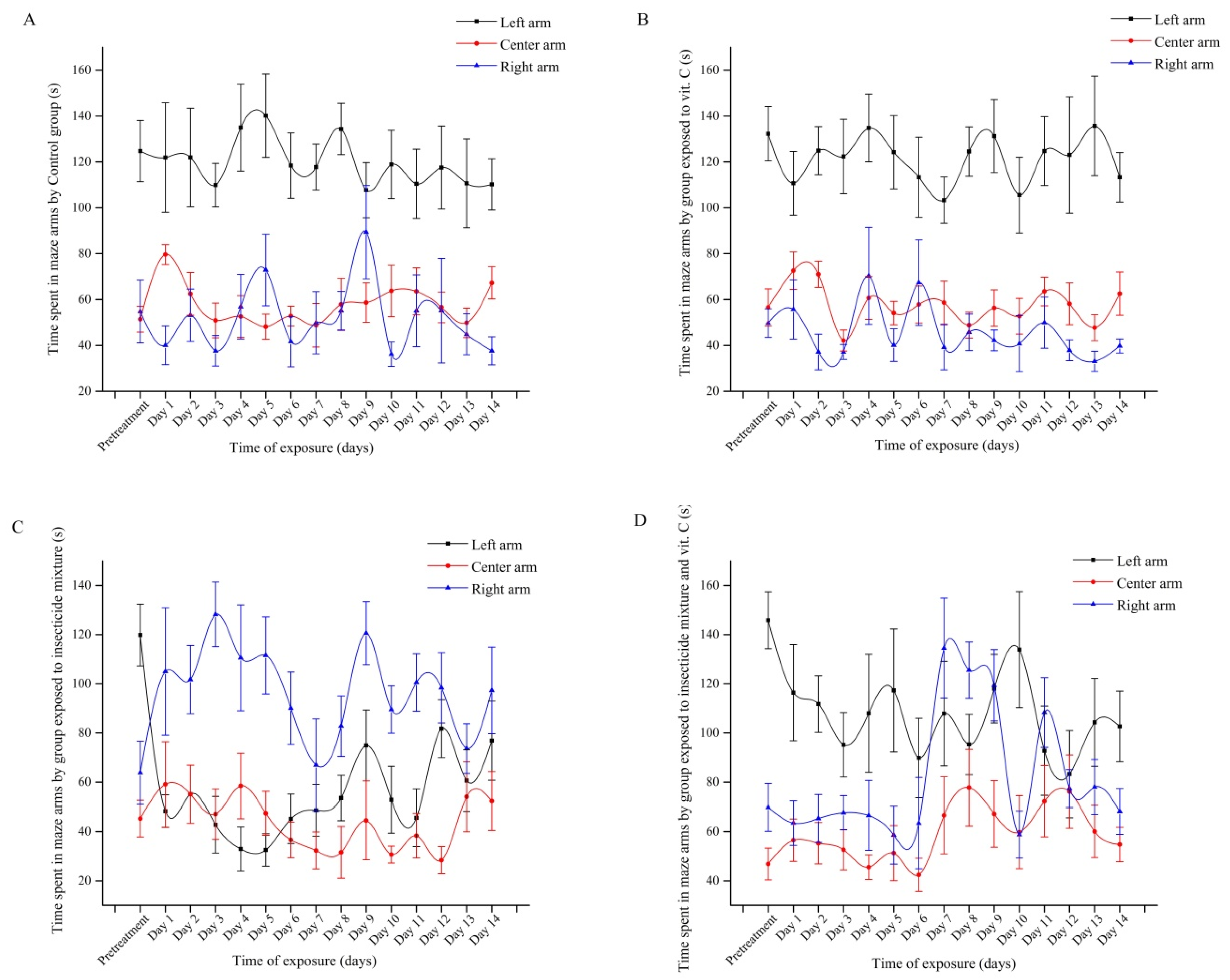
3.2. Effect of Vitamin C on Social Behavior
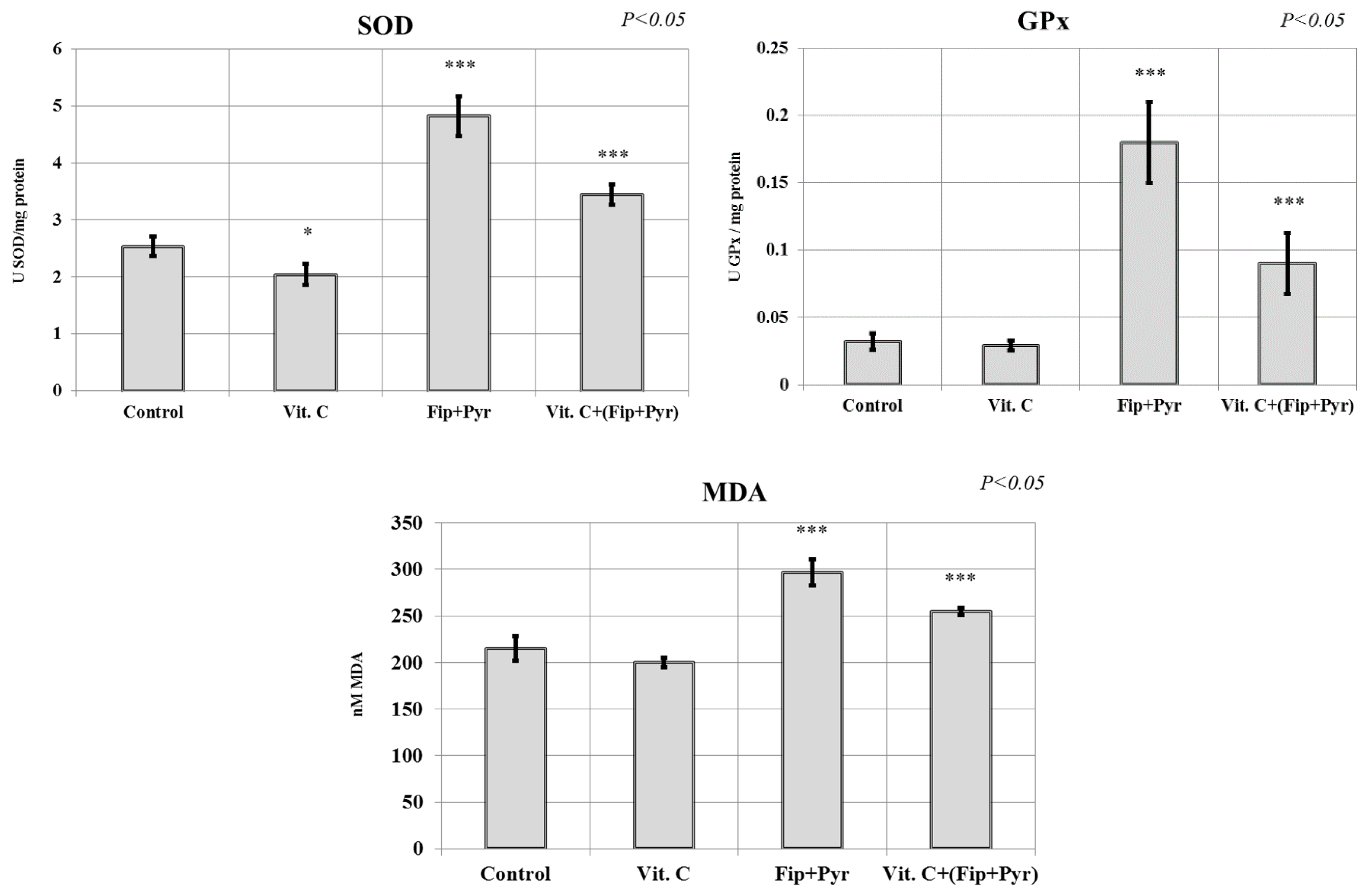
3.3. Oxidative Stress
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chaste, P.; Leboyer, M. Autism Risk Factors: Genes, Environment, and Gene-Environment Interactions. Dialogues Clin. Neurosci. 2012, 14, 281–292. [Google Scholar]
- Modabbernia, A.; Velthorst, E.; Reichenberg, A. Environmental Risk Factors for Autism: An Evidence-Based Review of Systematic Reviews and Meta-Analyses. Mol. Autism 2017, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Hadjkacem, I.; Ayadi, H.; Turki, M.; Yaich, S.; Khemekhem, K.; Walha, A.; Cherif, L.; Moalla, Y.; Ghribi, F. Prenatal, Perinatal and Postnatal Factors Associated with Autism Spectrum Disorder. J. Pediatr. 2016, 92, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.C.; Milatovic, D. Insecticides. In Biomarkers in Toxicology; Academic Press: Cambridge, MA, USA, 2019; 1222p. [Google Scholar]
- Narahashi, T.; Zhao, X.; Ikeda, T.; Salgado, V.L.; Yeh, J.Z. Glutamate-Activated Chloride Channels: Unique Fipronil Targets Present in Insects but Not in Mammals. Pestic. Biochem. Physiol. 2010, 97, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Islam, R.; Lynch, J.W. Mechanism of Action of the Insecticides, Lindane and Fipronil, on Glycine Receptor Chloride Channels. Br. J. Pharmacol. 2012, 165, 2707–2720. [Google Scholar] [CrossRef] [PubMed]
- Robea, M.A.; Nicoara, M.; Plavan, G.; Strugaru, S.A.; Ciobica, A. Fipronil: Mechanisms of Action on Various Organisms and Future Relevance for Animal Models Studies. Surv. Fish. Sci. 2018, 5, 20–31. [Google Scholar] [CrossRef]
- Gibbons, D.; Morrissey, C.; Mineau, P. A Review of the Direct and Indirect Effects of Neonicotinoids and Fipronil on Vertebrate Wildlife. Environ. Sci. Pollut. Res. 2015, 22, 103–118. [Google Scholar] [CrossRef]
- Tingle, C.C.D.; Rother, J.A.; Dewhurst, C.F.; Lauer, S.; King, W.J. Fipronil: Environmental Fate, Ecotoxicology, and Human Health Concerns. Rev. Environ. Contam. Toxicol. 2003, 176, 1–66. [Google Scholar] [CrossRef]
- Ohi, M.; Dalsenter, P.R.; Andrade, A.J.M.; Nascimento, A.J. Reproductive Adverse Effects of Fipronil in Wistar Rats. Toxicol. Lett. 2004, 146, 121–127. [Google Scholar] [CrossRef]
- Stehr, C.M.; Linbo, T.L.; Incardona, J.P.; Scholz, N.L. The Developmental Neurotoxicity of Fipronil: Notochord Degeneration and Locomotor Defects in Zebrafish Embryos and Larvae. Toxicol. Sci. 2006, 92, 270–278. [Google Scholar] [CrossRef]
- Beggel, S.; Werner, I.; Connon, R.E.; Geist, J.P. Sublethal Toxicity of Commercial Insecticide Formulations and Their Active Ingredients to Larval Fathead Minnow (Pimephales Promelas). Sci. Total Environ. 2010, 408, 3169–3175. [Google Scholar] [CrossRef] [PubMed]
- Kitulagodage, M.; Buttemer, W.; Astheimer, L. Adverse Effects of Fipronil on Avian Reproduction and Development: Maternal Transfer of Fipronil to Eggs in Zebra Finch Taeniopygia Guttata and in Ovo Exposure in Chickens Gallus Domesticus. Ecotoxicology 2011, 20, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Qian, Y.; Zhang, X.; Chen, F.; Zhang, Q.; Li, Z.; Zhao, M. A Metabolomic Study of Fipronil for the Anxiety-like Behavior in Zebrafish Larvae at Environmentally Relevant Levels. Environ. Pollut. 2016, 211, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Gong, C.; Zhang, X.; Zhang, Q.; Zhao, M.; Wang, C. Perturbation of Metabonome of Embryo/Larvae Zebrafish after Exposure to Fipronil. Environ. Toxicol. Pharmacol. 2016, 48, 39–45. [Google Scholar] [CrossRef]
- Xu, C.; Niu, L.; Liu, J.; Sun, X.; Zhang, C.; Ye, J.; Liu, W. Maternal Exposure to Fipronil Results in Sulfone Metabolite Enrichment and Transgenerational Toxicity in Zebrafish Offspring: Indication for an Overlooked Risk in Maternal Transfer? Environ. Pollut. 2019, 246, 876–884. [Google Scholar] [CrossRef]
- Kannan, M.; Sellamani, M.; Bojan, N.; Ramesh, M.; Kadirvelu, K. Toxicity Assessment of Pyriproxyfen in Vertebrate Model Zebrafish Embryos (Danio Rerio): A Multi Biomarker Study. Aquat. Toxicol. 2018, 196, 132–145. [Google Scholar] [CrossRef]
- Devillers, J. Fate and Ecotoxicological Effects of Pyriproxyfen in Aquatic Ecosystems. Environ. Sci. Pollut. Res. 2020, 27, 16052–16068. [Google Scholar] [CrossRef]
- Caixeta, E.S.; Silva, C.F.; Santos, V.S.V.; Olegário de Campos Júnior, E.; Pereira, B.B. Ecotoxicological Assessment of Pyriproxyfen under Environmentally Realistic Exposure Conditions of Integrated Vector Management for Aedes Aegypti Control in Brazil. J. Toxicol. Environ. Health A 2016, 79, 799–803. [Google Scholar] [CrossRef]
- Araújo, M.C.; Assis, C.R.D.; Silva, K.C.C.; Souza, K.S.; Azevedo, R.S.; Alves, M.H.M.E.; Silva, L.C.; Silva, V.L.; Adam, M.L.; Carvalho Junior, L.B.; et al. Characterization of Brain Acetylcholinesterase of Bentonic Fish Hoplosternum Littorale: Perspectives of Application in Pesticides and Metal Ions Biomonitoring. Aquat. Toxicol. 2018, 205, 213–226. [Google Scholar] [CrossRef]
- Gusso, D.; Reolon, G.K.; Gonzalez, J.B.; Altenhofen, S.; Kist, L.W.; Bogo, M.R.; Bonan, C.D. Pyriproxyfen Exposure Impairs Cognitive Parameters and Alters Cortisol Levels in Zebrafish. Front. Behav. Neurosci. 2020, 14, 103. [Google Scholar] [CrossRef]
- Lord, C.; Elsabbagh, M.; Baird, G.; Veenstra-Vanderweele, J. Autism Spectrum Disorder. Lancet 2018, 392, 508–520. [Google Scholar] [CrossRef]
- Onaolapo, A.Y.; Onaolapo, O.J. Global Data on Autism Spectrum Disorders Prevalence: A Review of Facts, Fallacies and Limitations. Univers. J. Clin. Med. 2017, 5, 14–23. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Baio, J.; Washington, A.; Patrick, M.; DiRienzo, M.; Christensen, D.L.; Wiggins, L.D.; Pettygrove, S.; Andrews, J.G.; et al. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2016. MMWR Surveill Summ 2020, 69, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Karimi, P.; Kamali, E.; Mousavi, S.M.; Karahmadi, M. Environmental Factors Influencing the Risk of Autism. J. Res. Med. Sci. 2017, 22, 27. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D.; Shamberger, R.J.; Audhya, T. Treatment of Autism Spectrum Children with Thiamine Tetrahydrofurfuryl Disulfide: A Pilot Study. Neuro Endocrinol. Lett. 2002, 23, 303–308. [Google Scholar] [PubMed]
- Lofthouse, N.; Hendren, R.; Hurt, E.; Arnold, L.E.; Butter, E. A Review of Complementary and Alternative Treatments for Autism Spectrum Disorders. Autism Res. Treat. 2012, 2012, 870391. [Google Scholar] [CrossRef]
- Mahmood, L.; Al Saadi, R.; Matthews, L. Dietary and Antioxidant Therapy for Autistic Children: Does It Really Work? Arch. Med. Heal. Sci. 2018, 6, 73–80. [Google Scholar] [CrossRef]
- Marotta, R.; Risolei, M.C.; Messina, G.; Parisi, L.; Carotenuto, M.; Vetri, L.; Roccella, M. The Neurochemistry of Autism. Brain Sci. 2020, 10, 163. [Google Scholar] [CrossRef]
- DeFilippis, M.; Wagner, K.D. Treatment of Autism Spectrum Disorder in Children and Adolescents. Psychopharmacol. Bull. 2016, 46, 18–41. [Google Scholar]
- Mazzone, L.; Giovagnoli, G.; Siracusano, M.; Postorino, V.; Curatolo, P. Drug Treatments for Core Symptoms of Autism Spectrum Disorder: Unmet Needs and Future Directions. J. Pediatr. Neurol. 2017, 15, 134–142. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Frye, R.E. Evidence Linking Oxidative Stress, Mitochondrial Dysfunction, and Inflammation in the Brain of Individuals with Autism. Front. Physiol. 2014, 5, 150. [Google Scholar] [CrossRef]
- Bjørklund, G.; Meguid, N.A.; El-Bana, M.A.; Tinkov, A.A.; Saad, K.; Dadar, M.; Hemimi, M.; Skalny, A.V.; Hosnedlová, B.; Kizek, R.; et al. Oxidative Stress in Autism Spectrum Disorder. Mol. Neurobiol. 2020, 57, 2314–2332. [Google Scholar] [CrossRef] [PubMed]
- Manivasagam, T.; Arunadevi, S.; Essa, M.M.; SaravanaBabu, C.; Borah, A.; Thenmozhi, A.J.; Qoronfleh, M.W. Role of Oxidative Stress and Antioxidants in Autism. Adv. Neurobiol. 2020, 24, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Fraguela, M.; Díaz, M.-L.; Vera, H.; Maragoto, C.; Noris, E.; Blanco, L. Oxidative Stress Markers in Children with Autism Spectrum Disorders. Br. J. Med. Med. Res. 2013, 3, 307–317. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Schlueter, A.K.; Johnston, C.S. Vitamin C: Overview and Update. J. Evid. Based. Complement. Altern. Med. 2011, 16, 49–57. [Google Scholar] [CrossRef]
- Pehlivan, F.E. Vitamin C: An Antioxidant Agent; IntechOpen: Rijeka, Croatia, 2017; pp. 23–35. [Google Scholar]
- Yussif, N.M. Vitamin C. In Vitamin C-An Update on Current Uses and Functions; LeBlanc, J.G., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Li, Y.; Schellhorn, H.E. New Developments and Novel Therapeutic Perspectives for Vitamin C. J. Nutr. 2007, 137, 2171–2184. [Google Scholar] [CrossRef]
- Maxfield, L.; Crane, J.S. Vitamin C Deficiency (Scurvy). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Khan, N.; Furlong-Dillard, J.M.; Buchman, R.F. Scurvy in an Autistic Child: Early Disease on MRI and Bone Scintigraphy Can Mimic an Infiltrative Process. BJR Case Rep. 2015, 1, 20150148. [Google Scholar] [CrossRef]
- Ma, N.S.; Thompson, C.; Weston, S. Brief Report: Scurvy as a Manifestation of Food Selectivity in Children with Autism. J. Autism Dev. Disord. 2016, 46, 1464–1470. [Google Scholar] [CrossRef]
- Saavedra, M.J.; Aziz, J.; Cacchiarelli San Román, N. Scurvy Due to Restrictive Diet in a Child with Autism Spectrum Disorder: Case Report. Arch. Argent. Pediatr. 2018, 116, e684. [Google Scholar] [CrossRef]
- Kinlin, L.M.; Morris, S.; Blanchard, A.; Silver, S. Limp and Difficulty Weight Bearing in a Child with Autism Spectrum Disorder: Scurvy as a Mimicker of Osteomyelitis. Pediatrics 2019, 144, 833. [Google Scholar] [CrossRef]
- Lund, R.M.; Becker, M.L.; Shapiro, S.; Allison, T.; Harris, J.G. Scurvy Presenting with Limp and Weakness: A Case Report. BMC Pediatr. 2019, 19, 228. [Google Scholar] [CrossRef] [PubMed]
- Cermak, S.A.; Curtin, C.; Bandini, L.G. Food Selectivity and Sensory Sensitivity in Children with Autism Spectrum Disorders. J. Am. Diet. Assoc. 2010, 110, 238–246. [Google Scholar] [CrossRef]
- Sharp, W.G.; Postorino, V.; McCracken, C.E.; Berry, R.C.; Criado, K.K.; Burrell, T.L.; Scahill, L. Dietary Intake, Nutrient Status, and Growth Parameters in Children with Autism Spectrum Disorder and Severe Food Selectivity: An Electronic Medical Record Review. J. Acad. Nutr. Diet. 2018, 118, 1943–1950. [Google Scholar] [CrossRef]
- Tomova, A.; Soltys, K.; Kemenyova, P.; Karhanek, M.; Babinska, K. The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota. Int. J. Mol. Sci. 2020, 21, 2797. [Google Scholar] [CrossRef]
- Wang, J.L. Vitamin C in Human Health and Disease; Springer Nature B.V.: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Teame, T.; Zhang, Z.; Ran, C.; Zhang, H.; Yang, Y.; Ding, Q.; Xie, M.; Gao, C.; Ye, Y.; Duan, M.; et al. The Use of Zebrafish (Danio Rerio) as Biomedical Models. Anim. Front. 2019, 9, 68–77. [Google Scholar] [CrossRef]
- Jijie, R.; Solcan, G.; Nicoara, M.; Micu, D.; Strungaru, S.-A. Antagonistic Effects in Zebrafish (Danio Rerio) Behavior and Oxidative Stress Induced by Toxic Metals and Deltamethrin Acute Exposure. Sci. Total Environ. 2020, 698, 134299. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.J.; Jennings, M. Guidance on the Housing and Care of Zebrafish Danio Rerio; Research Animals Department (RSPCA): Horsham, UK, 2011. [Google Scholar]
- Strungaru, S.-A.; Robea, M.A.; Plavan, G.; Todirascu-Ciornea, E.; Ciobica, A.; Nicoara, M. Acute Exposure to Methylmercury Chloride Induces Fast Changes in Swimming Performance, Cognitive Processes and Oxidative Stress of Zebrafish (Danio Rerio) as Reference Model for Fish Community. J. Trace Elem. Med. Biol. 2018, 47, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Strungaru, S.-A.; Plavan, G.; Ciobica, A.; Nicoara, M.; Robea, M.A.; Solcan, C.; Todirascu-Ciornea, E.; Petrovici, A. Acute Exposure to Gold Induces Fast Changes in Social Behavior and Oxidative Stress of Zebrafish (Danio Rerio). J. Trace Elem. Med. Biol. 2018, 50, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Strungaru, S.-A.; Plavan, G.; Ciobica, A.; Nicoara, M.; Robea, M.A.; Solcan, C.; Petrovici, A. Toxicity and Chronic Effects of Deltamethrin Exposure on Zebrafish (Danio Rerio) as a Reference Model for Freshwater Fish Community. Ecotoxicol. Environ. Saf. 2019, 171, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Pena, I.A.; Roussel, Y.; Daniel, K.; Mongeon, K.; Johnstone, D.; Weinschutz Mendes, H.; Bosma, M.; Saxena, V.; Lepage, N.; Chakraborty, P.; et al. Pyridoxine-Dependent Epilepsy in Zebrafish Caused by Aldh7a1 Deficiency. Genetics 2017, 207, 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Liu, Z.; Liu, F.; Ye, Y.; Peng, T.; Fu, Z. Embryonic Exposure to Cadmium (II) and Chromium (VI) Induce Behavioral Alterations, Oxidative Stress and Immunotoxicity in Zebrafish (Danio Rerio). Neurotoxicol. Teratol. 2015, 48, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Peng, L.; Gao, X.; Ji, H.; Ma, J.; Li, Y.; Jiang, S. Effects of Maduramicin on Adult Zebrafish (Danio Rerio): Acute Toxicity, Tissue Damage and Oxidative Stress. Ecotoxicol. Environ. Saf. 2019, 168, 249–259. [Google Scholar] [CrossRef]
- Beasley, V.R. Direct and Indirect Effects of Environmental Contaminants on Amphibians; Elsevier BV: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Wang, X.; Martínez, M.A.; Wu, Q.; Ares, I.; Martínez-Larrañaga, M.R.; Anadón, A.; Yuan, Z. Fipronil Insecticide Toxicology: Oxidative Stress and Metabolism. Crit. Rev. Toxicol. 2016, 46, 876–899. [Google Scholar] [CrossRef] [PubMed]
- Ghazanfar, M.; Shahid, S.; Qureshi, I.Z. Vitamin C Attenuates Biochemical and Genotoxic Damage in Common Carp (Cyprinus Carpio) upon Joint Exposure to Combined Toxic Doses of Fipronil and Buprofezin Insecticides. Aquat. Toxicol. 2018, 196, 43–52. [Google Scholar] [CrossRef]
- Little, E.E.; Archeski, R.D.; Flerov, B.A.; Kozlovskaya, V.I. Behavioral Indicators of Sublethal Toxicity in Rainbow Trout. Arch. Environ. Contam. Toxicol. 1990, 19, 380–385. [Google Scholar] [CrossRef]
- Shelton, J.F.; Hertz-Picciotto, I.; Pessah, I.N. Tipping the Balance of Autism Risk: Potential Mechanisms Linking Pesticides and Autism. Environ. Health Perspect. 2012, 120, 944–951. [Google Scholar] [CrossRef]
- Maharajan, K.; Muthulakshmi, S.; Karthik, C.; Nataraj, B.; Nambirajan, K.; Hemalatha, D.; Jiji, S.; Kadirvelu, K.; Liu, K.-C.; Ramesh, M. Pyriproxyfen Induced Impairment of Reproductive Endocrine Homeostasis and Gonadal Histopathology in Zebrafish (Danio Rerio) by Altered Expression of Hypothalamus-Pituitary-Gonadal (HPG) Axis Genes. Sci. Total Environ. 2020, 735, 139496. [Google Scholar] [CrossRef]
- Truong, L.; Gonnerman, G.; Simonich, M.T.; Tanguay, R.L. Assessment of the Developmental and Neurotoxicity of the Mosquito Control Larvicide, Pyriproxyfen, Using Embryonic Zebrafish. Environ. Pollut. 2016, 218, 1089–1093. [Google Scholar] [CrossRef]
- Clark, K.J.; Boczek, N.J.; Ekker, S.C. Stressing Zebrafish for Behavioral Genetics. Rev. Neurosci. 2011, 22, 49–62. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Panahi, Y.; Sahraei, H.; Johnston, T.P.; Sahebkar, A. The Impact of Stress on Body Function: A Review. EXCLI J. 2017, 16, 1057–1072. [Google Scholar] [CrossRef] [PubMed]
- El-Murr, A.e.I.; Abd El Hakim, Y.; Neamat-Allah, A.N.F.; Baeshen, M.; Ali, H.A. Immune-Protective, Antioxidant and Relative Genes Expression Impacts of β-Glucan against Fipronil Toxicity in Nile Tilapia, Oreochromis Niloticus. Fish. Shellfish Immunol. 2019, 94, 427–433. [Google Scholar] [CrossRef]
- Bjorklund, G.; Waly, M.I.; Al-Farsi, Y.; Saad, K.; Dadar, M.; Rahman, M.M.; Elhoufey, A.; Chirumbolo, S.; Jozwik-Pruska, J.; Kaluzna-Czaplinska, J. The Role of Vitamins in Autism Spectrum Disorder: What Do We Know? J. Mol. Neurosci. 2019, 67, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Dolske, M.C.; Spollen, J.; McKay, S.; Lancashire, E.; Tolbert, L. A Preliminary Trial of Ascorbic Acid as Supplemental Therapy for Autism. Prog. Neuro Psychopharmacol. Biol. Psychiatry 1993, 17, 765–774. [Google Scholar] [CrossRef]
- Adams, J.B.; Audhya, T.; McDonough-Means, S.; Rubin, R.A.; Quig, D.; Geis, E.; Gehn, E.; Loresto, M.; Mitchell, J.; Atwood, S.; et al. Effect of a Vitamin/Mineral Supplement on Children and Adults with Autism. BMC Pediatr. 2011, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.B.; Audhya, T.; Geis, E.; Gehn, E.; Fimbres, V.; Pollard, E.L.; Mitchell, J.; Ingram, J.; Hellmers, R.; Laake, D.; et al. Comprehensive Nutritional and Dietary Intervention for Autism Spectrum Disorder-A Randomized, Controlled 12-Month Trial. Nutrients 2018, 10, 369. [Google Scholar] [CrossRef]
- Kuder, R.S.; Philip, G.H. Antioxidant Enzymatic Activities and Lipid Peroxidation in Liver and Ovary of Zebrafish (Danio Rerio) Exposed to Deltamethrin. Chem. Ecol. 2017, 33, 739–749. [Google Scholar] [CrossRef]
- Al-Harbi, M.S.; El-Rahman, F.A.A.A.; El-Shenawy, N.S.; Al-Mutrafi, W.M. The Beneficial Effects of Ascorbic Acid during Chlorpyrifos-Induced Oxidative Stress and Histopathological Changes in Oreochromis Spilurus. Toxicol. Environ. Health Sci. 2014, 6, 203–216. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Gebhardt, M.; Stewart, A.M.; Cachat, J.M.; Brimmer, M.; Chawla, J.S.; Craddock, C.; Kyzar, E.J.; Roth, A.; Landsman, S.; et al. Towards a Comprehensive Catalog of Zebrafish Behavior 1.0 and Beyond. Zebrafish 2013, 10, 70–86. [Google Scholar] [CrossRef]
- Meshalkina, D.A.; Kizlyk, M.N.; Kysil, E.V.; Collier, A.D.; Echevarria, D.J.; Abreu, M.S.; Barcellos, L.J.G.; Song, C.; Warnick, J.E.; Kyzar, E.J.; et al. Zebrafish Models of Autism Spectrum Disorder. Exp. Neurol. 2018, 299, 207–216. [Google Scholar] [CrossRef]
- Zaluski, R.; Kadri, S.M.; Alonso, D.P.; Martins Ribolla, P.E.; de Oliveira Orsi, R. Fipronil Promotes Motor and Behavioral Changes in Honey Bees (Apis Mellifera) and Affects the Development of Colonies Exposed to Sublethal Doses. Environ. Toxicol. Chem. 2015, 34, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Godinho, A.F.; de Oliveira Souza, A.C.; Carvalho, C.C.; Horta, D.F.; De Fraia, D.; Anselmo, F.; Chaguri, J.L.; Faria, C.A. Memory Impairment Due to Fipronil Pesticide Exposure Occurs at the GABAA Receptor Level, in Rats. Physiol. Behav. 2016, 165, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Purkayastha, P.; Malapati, A.; Yogeeswari, P.; Sriram, D. A Review on GABA/Glutamate Pathway for Therapeutic Intervention of ASD and ADHD. Curr. Med. Chem. 2015, 22, 1850–1859. [Google Scholar] [CrossRef] [PubMed]
- Behuet, S.; Cremer, J.N.; Cremer, M.; Palomero-Gallagher, N.; Zilles, K.; Amunts, K. Developmental Changes of Glutamate and GABA Receptor Densities in Wistar Rats. Front. Neuroanat. 2019, 13, 100. [Google Scholar] [CrossRef]
- Pizzarelli, R.; Cherubini, E. Alterations of GABAergic Signaling in Autism Spectrum Disorders. Neural Plast. 2011, 2011, 297153. [Google Scholar] [CrossRef]
- Roth, F.C.; Draguhn, A. GABA Metabolism and Transport: Effects on Synaptic Efficacy. Neural Plast. 2012, 2012, 805830. [Google Scholar] [CrossRef]
- Samardzic, J.; Jadzic, D.; Hencic, B.; Jancic, J.; Strac, D.S. GABA/Glutamate Balance: A Key for Normal Brain Functioning. In GABA and Glutamate—New Developments in Neurotransmission Research; IntechOpen: Rijeka, Croatia, 2018. [Google Scholar]
- Horder, J.; Petrinovic, M.M.; Mendez, M.A.; Bruns, A.; Takumi, T.; Spooren, W.; Barker, G.J.; Kunnecke, B.; Murphy, D.G. Glutamate and GABA in Autism Spectrum Disorder-a Translational Magnetic Resonance Spectroscopy Study in Man and Rodent Models. Transl. Psychiatry 2018, 8, 106. [Google Scholar] [CrossRef]
- Khalifa, D.; Shahin, O.; Salem, D.; Raafat, O. Serum Glutamate Was Elevated in Children Aged 3–10 Years with Autism Spectrum Disorders When They Were Compared with Controls. Acta Paediatr. 2019, 108, 295–299. [Google Scholar] [CrossRef]
- Sapey-Triomphe, L.-A.; Lamberton, F.; Sonié, S.; Mattout, J.; Schmitz, C. Tactile Hypersensitivity and GABA Concentration in the Sensorimotor Cortex of Adults with Autism. Autism Res. 2019, 12, 562–575. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robea, M.A.; Jijie, R.; Nicoara, M.; Plavan, G.; Ciobica, A.S.; Solcan, C.; Audira, G.; Hsiao, C.-D.; Strungaru, S.-A. Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile. Antioxidants 2020, 9, 944. https://doi.org/10.3390/antiox9100944
Robea MA, Jijie R, Nicoara M, Plavan G, Ciobica AS, Solcan C, Audira G, Hsiao C-D, Strungaru S-A. Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile. Antioxidants. 2020; 9(10):944. https://doi.org/10.3390/antiox9100944
Chicago/Turabian StyleRobea, Madalina Andreea, Roxana Jijie, Mircea Nicoara, Gabriel Plavan, Alin Stelian Ciobica, Carmen Solcan, Gilbert Audira, Chung-Der Hsiao, and Stefan-Adrian Strungaru. 2020. "Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile" Antioxidants 9, no. 10: 944. https://doi.org/10.3390/antiox9100944
APA StyleRobea, M. A., Jijie, R., Nicoara, M., Plavan, G., Ciobica, A. S., Solcan, C., Audira, G., Hsiao, C.-D., & Strungaru, S.-A. (2020). Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile. Antioxidants, 9(10), 944. https://doi.org/10.3390/antiox9100944








