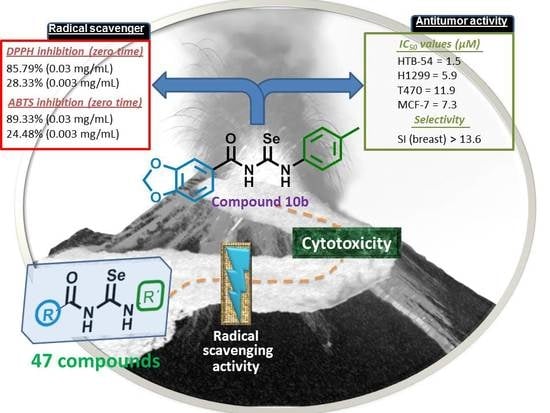
Novel N,N′-Disubstituted Acylselenoureas as Potential Antioxidant and Cytotoxic Agents
Abstract
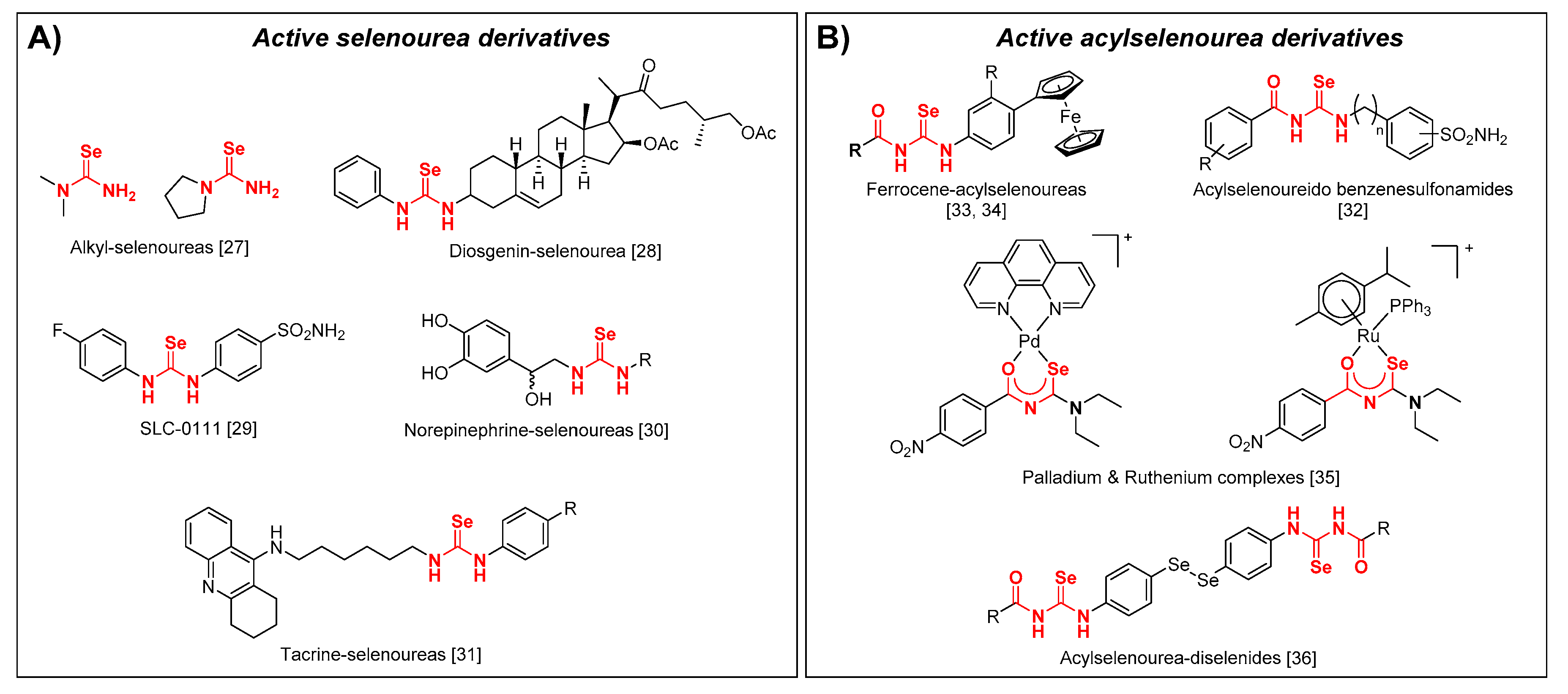
1. Introduction
2. Materials and Methods
2.1. Chemistry
2.1.1. General Information
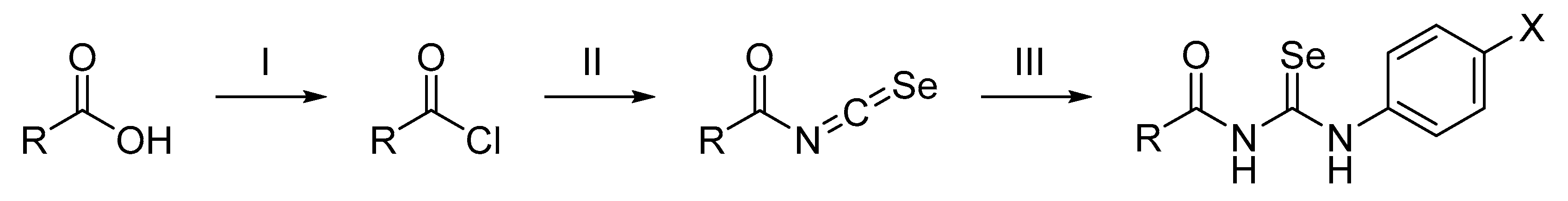
2.1.2. General Procedure for the Preparation of Acylselenourea Derivatives
2.2. Radical Scavenging Activity
2.2.1. DPPH Radical Scavenging Assay
2.2.2. ABTS Radical Scavenging Assay
2.3. Biological Evaluation
2.3.1. Cell Culture Conditions
2.3.2. Cell Viability Assay
2.3.3. Protective Effects against H2O2-Induced Oxidative Stress in HT-29 Cells
3. Results and Discussion
3.1. Chemistry
3.2. Antioxidant Activity
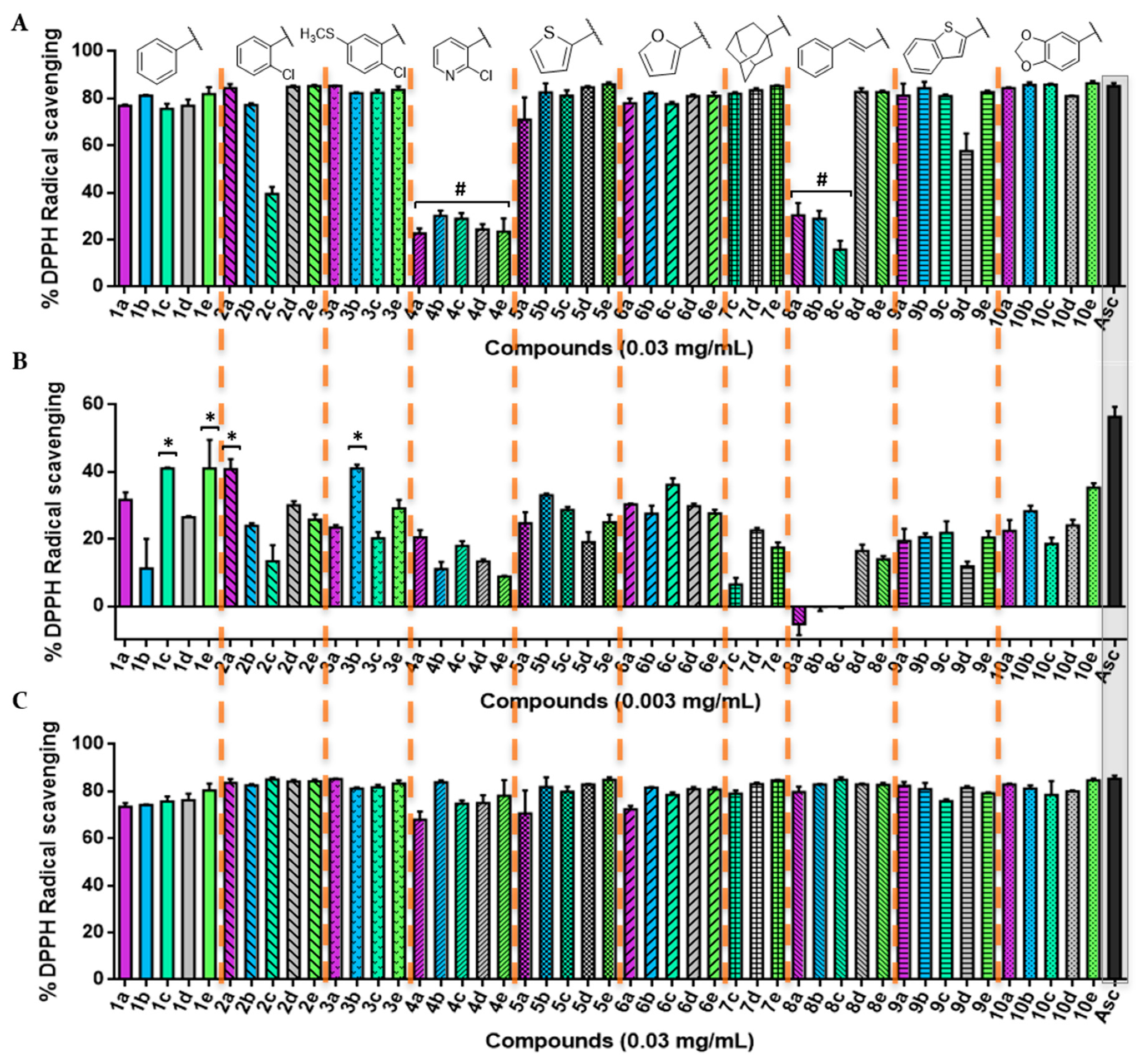
3.2.1. DPPH Radical Scavenging Assay
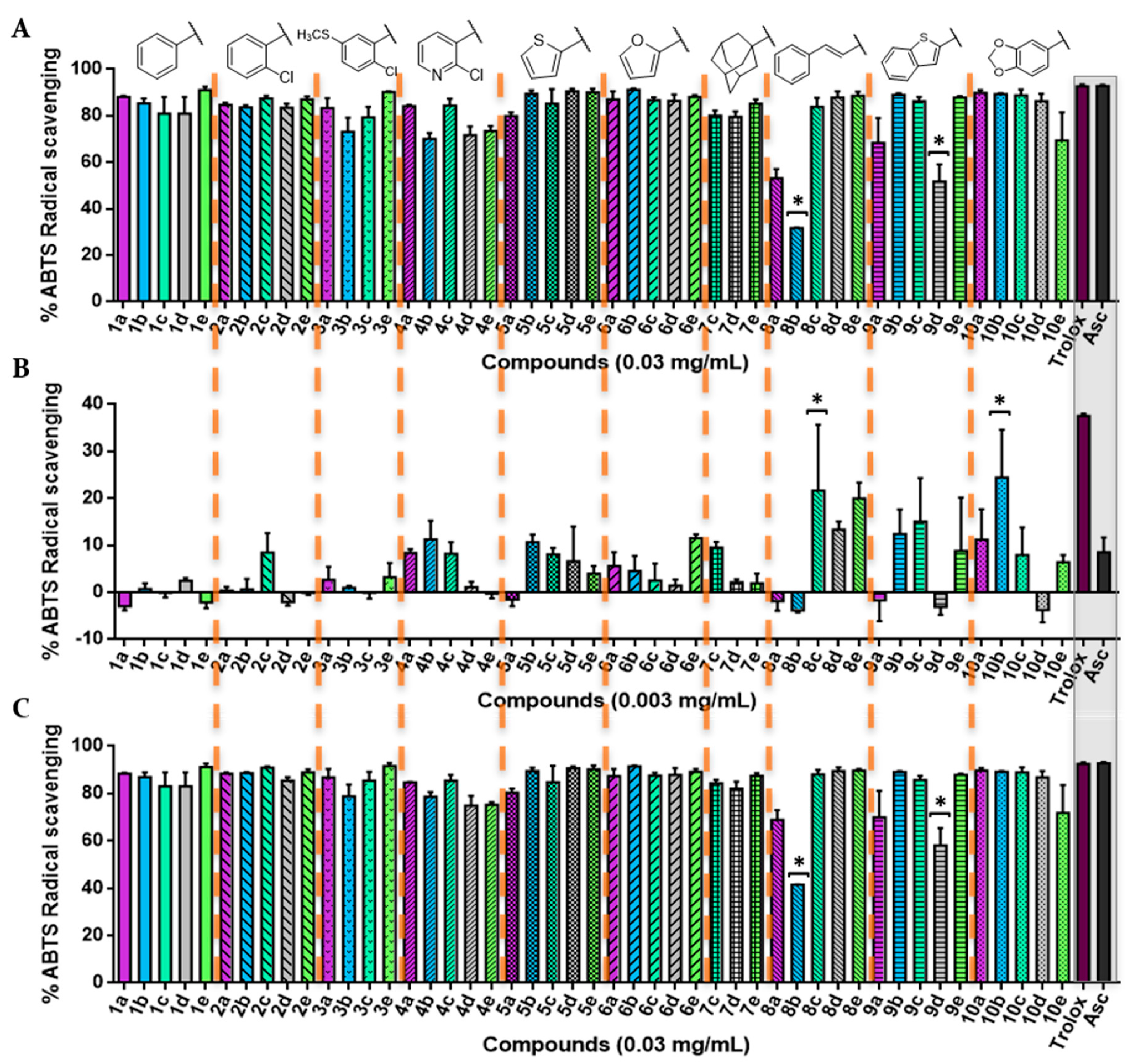
3.2.2. ABTS Radical Scavenging Assay
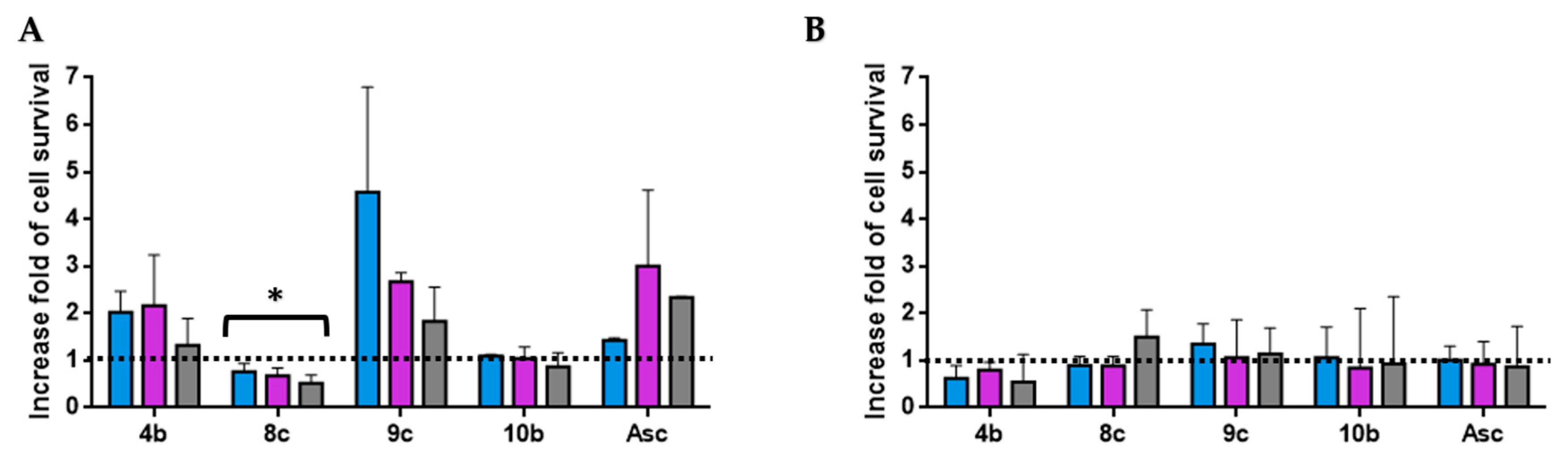
3.2.3. Protective Effects against H2O2-Induced Cell Damage in HT-29 Cells
3.3. Biological Evaluation
Cytotoxic Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kensler, T.W.; Trush, M.A. Role of oxygen radicals in tumor promotion. Environ. Mutagen. 1984, 6, 593–616. [Google Scholar] [CrossRef]
- Lewis, A.M.; Ough, M.; Du, J.; Tsao, M.S.; Oberley, L.W.; Cullen, J.J. Targeting NAD(P)H:Quinone Oxidoreductase (NQO1) in pancreatic cancer. Mol. Carcinog. 2017, 56, 1825–1834. [Google Scholar] [CrossRef]
- Kahroba, H.; Shirmohamadi, M.; Hejazi, M.S.; Samadi, N. The Role of Nrf2 signaling in cancer stem cells: From stemness and self-renewal to tumorigenesis and chemoresistance. Life Sci. 2019, 239, 116986. [Google Scholar] [CrossRef]
- Ismail, T.; Kim, Y.; Lee, H.; Lee, D.S.; Lee, H.S. Interplay between mitochondrial peroxiredoxins and ROS in cancer development and progression. Int. J. Mol. Sci. 2019, 20, 4407. [Google Scholar] [CrossRef]
- Assi, M. The differential role of reactive oxygen species in early and late stages of cancer. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 313, R646–R653. [Google Scholar] [CrossRef]
- Moldogazieva, N.T.; Lutsenko, S.V.; Terentiev, A.A. Reactive oxygen and nitrogen species-induced protein modifications: Implication in carcinogenesis and anticancer therapy. Cancer Res. 2018, 78, 6040–6047. [Google Scholar] [CrossRef]
- Mao, X.; Xiao, X.; Chen, D.; Yu, B.; He, J. Tea and Its components prevent cancer: A review of the redox-related mechanism. Int. J. Mol. Sci. 2019, 20, 5249. [Google Scholar] [CrossRef]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell. Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef]
- Short, S.P.; Williams, C.S. Selenoproteins in tumorigenesis and cancer progression. Adv. Cancer Res. 2017, 136, 49–83. [Google Scholar]
- Mohammadi, F.; Soltani, A.; Ghahremanloo, A.; Javid, H.; Hashemy, S.I. The thioredoxin system and cancer therapy: A review. Cancer Chemother. Pharmacol. 2019, 84, 925–935. [Google Scholar] [CrossRef]
- Russo, M.W.; Murray, S.C.; Wurzelmann, J.I.; Woosley, J.T.; Sandler, R.S. Plasma selenium levels and the risk of colorectal adenomas. Nutr. Cancer 1997, 28, 125–129. [Google Scholar] [CrossRef]
- Brinkman, M.; Reulen, R.C.; Kellen, E.; Buntinx, F.; Zeegers, M.P. Are men with low selenium levels at increased risk of prostate cancer? Eur. J. Cancer 2006, 42, 2463–2471. [Google Scholar] [CrossRef]
- Vinceti, M.; Filippini, T.; Cilloni, S.; Crespi, C.M. The epidemiology of selenium and human cancer. Adv. Cancer Res. 2017, 136, 1–48. [Google Scholar]
- Collery, P. Strategies for the development of selenium-based anticancer drugs. J. Trace Elem. Med. Biol. 2018, 50, 498–507. [Google Scholar] [CrossRef]
- Tan, H.W.; Mo, H.Y.; Lau, A.T.Y.; Xu, Y.M. Selenium species: Current status and potentials in cancer prevention and therapy. Int. J. Mol. Sci. 2018, 20, 75. [Google Scholar] [CrossRef]
- Clark, L.C.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Chalker, D.K.; Chow, J.; Davis, L.S.; Glover, R.A.; Graham, G.F.; Gross, E.G.; et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA 1996, 276, 1957–1963. [Google Scholar] [CrossRef]
- Clark, L.C.; Dalkin, B.; Krongrad, A.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Witherington, R.; Herlong, J.H.; Janosko, E.; Carpenter, D.; et al. Decreased incidence of prostate cancer with selenium supplementation: Results of a double-blind cancer prevention trial. Br. J. Urol. 1998, 81, 730–734. [Google Scholar] [CrossRef]
- Duffield-Lillico, A.J.; Reid, M.E.; Turnbull, B.W.; Combs, G.F., Jr.; Slate, E.H.; Fischbach, L.A.; Marshall, J.R.; Clark, L.C. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: A summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiol. Biomark. Prev. 2002, 11, 630–639. [Google Scholar]
- Lippman, S.M.; Klein, E.A.; Goodman, P.J.; Lucia, M.S.; Thompson, I.M.; Ford, L.G.; Parnes, H.L.; Minasian, L.M.; Gaziano, J.M.; Hartline, J.A.; et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2009, 301, 39–51. [Google Scholar] [CrossRef]
- Vinceti, M.; Vicentini, M.; Wise, L.A.; Sacchettini, C.; Malagoli, C.; Ballotari, P.; Filippini, T.; Malavolti, M.; Rossi, P.G. Cancer incidence following long-term consumption of drinking water with high inorganic selenium content. Sci. Total Environ. 2018, 635, 390–396. [Google Scholar] [CrossRef]
- Rayman, M.P.; Winther, K.H.; Pastor-Barriuso, R.; Cold, F.; Thvilum, M.; Stranges, S.; Guallar, E.; Cold, S. Effect of long-term selenium supplementation on mortality: Results from a multiple-dose, randomised controlled trial. Free Radic. Biol. Med. 2018, 127, 46–54. [Google Scholar] [CrossRef]
- Bartolini, D.; Sancineto, L.; Fabro de Bem, A.; Tew, K.D.; Santi, C.; Radi, R.; Toquato, P.; Galli, F. Selenocompounds in cancer therapy: An overview. Adv. Cancer Res. 2017, 136, 259–302. [Google Scholar] [PubMed]
- Ali, W.; Álvarez-Pérez, M.; Marć, M.A.; Salardón-Jiménez, N.; Handzlik, J.; Domínguez-Álvarez, E.J.C.P.R. The anticancer and chemopreventive activity of selenocyanate-containing compounds. Curr. Pharmacol. Rep. 2018, 4, 468–481. [Google Scholar] [CrossRef]
- Alvarez-Perez, M.; Ali, W.; Marc, M.A.; Handzlik, J.; Dominguez-Alvarez, E. Selenides and diselenides: A review of their anticancer and chemopreventive activity. Molecules 2018, 23, 628. [Google Scholar] [CrossRef]
- Ruberte, A.C.; Sanmartin, C.; Aydillo, C.; Sharma, A.K.; Plano, D. Development and therapeutic potential of selenazo compounds. J. Med. Chem. 2019. [Google Scholar] [CrossRef]
- Plano, D.; Sanmartin, C.; Moreno, E.; Prior, C.; Calvo, A.; Palop, J.A. Novel potent organoselenium compounds as cytotoxic agents in prostate cancer cells. Bioorg. Med. Chem. Lett. 2007, 17, 6853–6859. [Google Scholar] [CrossRef]
- Plano, D.; Baquedano, Y.; Ibanez, E.; Jimenez, I.; Palop, J.A.; Spallholz, J.E.; Sanmartin, C. Antioxidant-prooxidant properties of a new organoselenium compound library. Molecules 2010, 15, 7292–7312. [Google Scholar] [CrossRef]
- Ibanez, E.; Plano, D.; Font, M.; Calvo, A.; Prior, C.; Palop, J.A.; Sanmartin, C. Synthesis and antiproliferative activity of novel symmetrical alkylthio- and alkylseleno-imidocarbamates. Eur. J. Med. Chem. 2011, 46, 265–274. [Google Scholar] [CrossRef]
- Plano, D.; Ibanez, E.; Calvo, A.; Palop, J.A.; Sanmartin, C. Novel library of selenocompounds as kinase modulators. Molecules 2011, 16, 6349–6364. [Google Scholar] [CrossRef]
- Font, M.; Zuazo, A.; Anso, E.; Plano, D.; Sanmartin, C.; Palop, J.A.; Martinez-Irujo, J.J. Novel structural insights for imidoselenocarbamates with antitumoral activity related to their ability to generate methylselenol. Bioorg. Med. Chem. 2012, 20, 5110–5116. [Google Scholar] [CrossRef]
- Ibanez, E.; Stoedter, M.; Hofmann, P.J.; Plano, D.; Calvo, A.; Nguewa, P.A.; Palop, J.A.; Sanmartin, C.; Schomburg, L. Structure- and cell-specific effects of imidoselenocarbamates on selenoprotein expression and activity in liver cells in culture. Metallomics 2012, 4, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Lamberto, I.; Plano, D.; Moreno, E.; Font, M.; Palop, J.A.; Sanmartin, C.; Encio, I. Bisacylimidoselenocarbamates cause G2/M arrest associated with the modulation of CDK1 and Chk2 in human breast cancer MCF-7 cells. Curr. Med. Chem. 2013, 20, 1609–1619. [Google Scholar] [CrossRef] [PubMed]
- Alcolea, V.; Plano, D.; Encio, I.; Palop, J.A.; Sharma, A.K.; Sanmartin, C. Chalcogen containing heterocyclic scaffolds: New hybrids with antitumoral activity. Eur. J. Med. Chem. 2016, 123, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Alcolea, V.; Karelia, D.N.; Pandey, M.K.; Plano, D.; Singh, P.; Palop, J.A.; Amin, S.; Sanmartin, C.; Sharma, A.K. Identification of a novel quinoxaline-isoselenourea targeting the STAT3 pathway as a potential melanoma therapeutic. Int. J. Mol. Sci. 2019, 20, 521. [Google Scholar] [CrossRef]
- Plano, D.; Ibanez, E.; Palop, J.A.; Sanmartin, C. Synthesis, characterization, crystal structure and cytotoxicity of 2,4-bis(selenomethyl) quinazoline. Struct. Chem. 2011, 22, 1233–1240. [Google Scholar] [CrossRef]
- Moreno, E.; Plano, D.; Lamberto, I.; Font, M.; Encio, I.; Palop, J.A.; Sanmartin, C. Sulfur and selenium derivatives of quinazoline and pyrido[2,3-d]pyrimidine: Synthesis and study of their potential cytotoxic activity in vitro. Eur. J. Med. Chem. 2012, 47, 283–298. [Google Scholar] [CrossRef]
- Moreno, E.; Doughty-Shenton, D.; Plano, D.; Font, M.; Encio, I.; Palop, J.A.; Sanmartin, C. A dihydroselenoquinazoline inhibits S6 ribosomal protein signalling, induces apoptosis and inhibits autophagy in MCF-7 cells. Eur. J. Pharm. Sci. 2014, 63, 87–95. [Google Scholar] [CrossRef]
- Tsukagoshi, H.; Koketsu, M.; Kato, M.; Kurabayashi, M.; Nishina, A.; Kimura, H. Superoxide radical-scavenging effects from polymorphonuclear leukocytes and toxicity in human cell lines of newly synthesized organic selenium compounds. FEBS J. 2007, 274, 6046–6054. [Google Scholar] [CrossRef]
- Romero-Hernandez, L.L.; Merino-Montiel, P.; Montiel-Smith, S.; Meza-Reyes, S.; Vega-Baez, J.L.; Abasolo, I.; Schwartz, S., Jr.; Lopez, O.; Fernandez-Bolanos, J.G. Diosgenin-based thio(seleno)ureas and triazolyl glycoconjugates as hybrid drugs. Antioxidant and antiproliferative profile. Eur. J. Med. Chem. 2015, 99, 67–81. [Google Scholar] [CrossRef]
- Angeli, A.; Tanini, D.; Peat, T.S.; Di Cesare Mannelli, L.; Bartolucci, G.; Capperucci, A.; Ghelardini, C.; Supuran, C.T.; Carta, F. Discovery of new selenoureido analogues of 4-(4-fluorophenylureido)benzenesulfonamide as carbonic anhydrase inhibitors. ACS Med. Chem. Lett. 2017, 8, 963–968. [Google Scholar] [CrossRef]
- Marset, A.; Begines, P.; Lopez, O.; Maya, I.; Garcia-Aranda, N.; Schwartz, S., Jr.; Abasolo, I.; Fernandez-Bolanos, J.G. Design of chalcogen-containing norepinephrines: Efficient GPx mimics and strong cytotoxic agents against HeLa cells. Future Med. Chem. 2016, 8, 2185–2195. [Google Scholar] [CrossRef] [PubMed]
- Roldan-Pena, J.M.; Alejandre-Ramos, D.; Lopez, O.; Maya, I.; Lagunes, I.; Padron, J.M.; Pena-Altamira, L.E.; Bartolini, M.; Monti, B.; Bolognesi, M.L.; et al. New tacrine dimers with antioxidant linkers as dual drugs: Anti-Alzheimer’s and antiproliferative agents. Eur. J. Med. Chem. 2017, 138, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Angeli, A.; Carta, F.; Bartolucci, G.; Supuran, C.T. Synthesis of novel acyl selenoureido benzensulfonamides as carbonic anhydrase I, II, VII and IX inhibitors. Bioorg. Med. Chem. 2017, 25, 3567–3573. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.A.; Badshah, A.; Pezzuto, J.M.; Ahmed, N.; Kondratyuk, T.P.; Park, E.J. Ferrocene incorporated selenoureas as anticancer agents. J. Photochem. Photobiol. B Biol. 2015, 148, 197–208. [Google Scholar] [CrossRef]
- Hussain, R.A.; Badshah, A.; Ahmed, N.; Pezzuto, J.M.; Kondratyuk, T.P.; Park, E.-J.; Hussain, I. Synthesis, characterization and biological applications of selenoureas having ferrocene and substituted benzoyl functionalities. Polyhedron 2019, 170, 12–24. [Google Scholar] [CrossRef]
- Molter, A.; Kathrein, S.; Kircher, B.; Mohr, F. Anti-tumour active gold(i), palladium(ii) and ruthenium(ii) complexes with thio- and selenoureato ligands: A comparative study. Dalton Trans. 2018, 47, 5055–5064. [Google Scholar] [CrossRef]
- Garnica, P.; Encio, I.; Plano, D.; Palop, J.A.; Sanmartin, C. Combined acylselenourea-diselenide structures: New potent and selective antitumoral agents as autophagy activators. ACS Med. Chem. Lett. 2018, 9, 306–311. [Google Scholar] [CrossRef]
- Viegas-Junior, C.; Danuello, A.; Da Silva Bolzani, V.; Barreiro, E.J.; Fraga, C.A. Molecular hybridization: A useful tool in the design of new drug prototypes. Curr. Med. Chem. 2007, 14, 1829–1852. [Google Scholar] [CrossRef]
- Cesarini, S.; Spallarossa, A.; Ranise, A.; Schenone, S.; Rosano, C.; La Colla, P.; Sanna, G.; Busonera, B.; Loddo, R. N-acylated and N,N′-diacylated imidazolidine-2-thione derivatives and N,N′-diacylated tetrahydropyrimidine-2(1H)-thione analogues: Synthesis and antiproliferative activity. Eur. J. Med. Chem. 2009, 44, 1106–1118. [Google Scholar] [CrossRef]
- Marquise, N.; Chevallier, F.; Nassar, E.; Frédérich, M.; Ledoux, A.; Halauko, Y.S.; Ivashkevich, O.A.; Matulis, V.E.; Roisnel, T.; Dorcet, V.; et al. Substituted azafluorenones: Access from dihalogeno diaryl ketones by palladium-catalyzed auto-tandem processes and evaluation of their antibacterial, antifungal, antimalarial and antiproliferative activities. Tetrahedron 2016, 72, 825–836. [Google Scholar] [CrossRef]
- Worawalai, W.; Doungwichitrkul, T.; Rangubpit, W.; Taweechat, P.; Sompornpisut, P.; Phuwapraisirisan, P. Furofuran lignans as a new series of antidiabetic agents exerting alpha-glucosidase inhibition and radical scarvenging: Semisynthesis, kinetic study and molecular modeling. Bioorg. Chem. 2019, 87, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Spasova, M.; Kortenska-Kancheva, V.; Totseva, I.; Ivanova, G.; Georgiev, L.; Milkova, T. Synthesis of cinnamoyl and hydroxycinnamoyl amino acid conjugates and evaluation of their antioxidant activity. J. Pept. Sci. 2006, 12, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Kristian, P.; Koscik, D.; Gonda, J. Heterocycles with a pyrido 3,2-E-1,3-selenazine and pyrido 3,4-E -1,3-selenazine ring-systems. Collect. Czech. Chem. Commun. 1983, 48, 3567–3574. [Google Scholar] [CrossRef]
- Hua, G.; Cordes, D.B.; Du, J.; Slawin, A.M.Z.; Woollins, J.D. Diverse derivatives of selenoureas: A synthetic and single crystal structural study. Molecules 2018, 23, 2143. [Google Scholar] [CrossRef]
- Hu, H.Z.; Wang, S.Y.; Shi, D.Y.; Zhong, B.L.; Huang, X.; Shi, C.W.; Shao, Z.W. Lycorine exerts antitumor activity against osteosarcoma cells in vitro and in vivo xenograft model through the JAK2/STAT3 pathway. Onco Targets Ther. 2019, 12, 5377–5388. [Google Scholar] [CrossRef]
- Sujarwo, W.; Keim, A.P. Spondias pinnata (L. f.) Kurz. (Anacardiaceae): Profiles and applications to diabetes. In Bioactive Food as Dietary Interventions for Diabetes, 2nd ed.; Watson, R.R., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA, 2019; Chapter 27; pp. 395–405. [Google Scholar] [CrossRef]
- Zafar, M.S.; Quarta, A.; Marradi, M.; Ragusa, A. Recent developments in the reduction of oxidative stress through antioxidant polymeric formulations. Pharmaceutics 2019, 11, 505. [Google Scholar] [CrossRef]
- Svinyarov, I.; Bogdanov, M.G. One-pot synthesis and radical scavenging activity of novel polyhydroxylated 3-arylcoumarins. Eur. J. Med. Chem. 2014, 78, 198–206. [Google Scholar] [CrossRef]
- Romano, B.; Plano, D.; Encio, I.; Palop, J.A.; Sanmartin, C. In vitro radical scavenging and cytotoxic activities of novel hybrid selenocarbamates. Bioorg. Med. Chem. 2015, 23, 1716–1727. [Google Scholar] [CrossRef]
- Teng, Y.; Li, X.; Yang, K.; Li, X.; Zhang, Z.; Wang, L.; Deng, Z.; Song, B.; Yan, Z.; Zhang, Y.; et al. Synthesis and antioxidant evaluation of desmethylxanthohumol analogs and their dimers. Eur. J. Med. Chem. 2017, 125, 335–345. [Google Scholar] [CrossRef]
- Yao, K.; Zhang, L.; Ye, P.P.; Tang, X.J.; Zhang, Y.D. Protective effect of magnolol against hydrogen peroxide-induced oxidative stress in human lens epithelial cells. Am. J. Chin. Med. 2009, 37, 785–796. [Google Scholar] [CrossRef]
- Fernandes, A.P.; Gandin, V. Selenium compounds as therapeutic agents in cancer. Biochim. Biophys. Acta 2015, 1850, 1642–1660. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Lipinski, B. Pathophysiological significance of protein hydrophobic interactions: An emerging hypothesis. Med. Hypotheses 2018, 110, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Lipinski, B.; Blazejak, S. Application of sodium selenite in the prevention and treatment of cancers. Cells 2017, 6, 39. [Google Scholar] [CrossRef] [PubMed]
- Tavadyan, L.A.; Sahakyan, A.D.; Harutyunyan, L.A.; Tonikyan, A.G.; Manukyan, Z.O. Antiradical activity of dimethyl selenoxide and sodium selenite. Russ. Chem. Bull. 2013, 62, 1586–1589. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Leite, A.C.L.; Da Silva, K.P.; De Souza, I.A.; De Araujo, J.M.; Brondani, D.J. Synthesis, antitumour and antimicrobial activities of new peptidyl derivatives containing the 1,3-benzodioxole system. Eur. J. Med. Chem. 2004, 39, 1059–1065. [Google Scholar] [CrossRef]
- Tu, S.H.; Wu, C.H.; Chen, L.C.; Huang, C.S.; Chang, H.W.; Chang, C.H.; Lien, H.M.; Ho, Y.S. In vivo antitumor effects of 4,7-dimethoxy-5-methyl-1,3-benzodioxole isolated from the fruiting body of antrodia camphorata through activation of the p53-mediated p27/Kip1 signaling pathway. J. Agric. Food Chem. 2012, 60, 3612–3618. [Google Scholar] [CrossRef]
- Wei, P.L.; Tu, S.H.; Lien, H.M.; Chen, L.C.; Chen, C.S.; Wu, C.H.; Huang, C.S.; Chang, H.W.; Chang, C.H.; Tseng, H.; et al. The in vivo antitumor effects on human COLO 205 cancer cells of the 4,7-dimethoxy-5-(2-propen-1-yl)-1,3-benzodioxole (apiole) derivative of 5-substituted 4,7-dimethoxy-5-methyl-l,3-benzodioxole (SY-1) isolated from the fruiting body of Antrodia camphorata. J. Cancer Res. Ther. 2012, 8, 532–536. [Google Scholar]

| Compound | IC50 (µM) 1 | |||||
|---|---|---|---|---|---|---|
| Cell Lines | ||||||
| HTB-54 | H1299 | T47D | MCF-7 | 184B5 | SI 2 | |
| 5d | 14.6 ± 6.3 | 7.5 ± 0.2 | 6.1 ± 0.4 | 4.2 ± 0.2 | >100 | >23.7 |
| 6a | 26.1 ± 2.3 | 30.2 ± 7.2 | 7.8 ± 1.0 | 3.5 ± 0.6 | 52.2 ± 3.8 | 14.7 |
| 9e | >100 | 8.7 ± 1.4 | 7.1 ± 0.6 | 9.8 ± 0.9 | >100 | >10.2 |
| 10a | >100 | 11.2 ± 0.8 | 8.0 ± 1.0 | 4.4 ± 1.0 | >100 | >22.9 |
| 10b | 1.5 ± 1.8 | 5.9 ± 1.4 | 11.9 ± 1.0 | 7.3 ± 0.3 | >100 | >13.6 |
| 10c | 5.2 ± 4.0 | 7.3 ± 0.2 | 12.9 ± 1.5 | 8.5 ± 0.6 | 53.8 ± 10.5 | 6.3 |
| 10d | 23.4 ± 3.2 | 5.7 ± 0.1 | 11.8 ± 1.9 | 7.9 ± 0.7 | >100 | >12.7 |
| 10e | 2.9 ± 3.2 | 48.6 ± 0.9 | 17.4 ± 3.1 | 11.7 ± 1.5 | 73.1 ± 5.2 | 6.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruberte, A.C.; Ramos-Inza, S.; Aydillo, C.; Talavera, I.; Encío, I.; Plano, D.; Sanmartín, C. Novel N,N′-Disubstituted Acylselenoureas as Potential Antioxidant and Cytotoxic Agents. Antioxidants 2020, 9, 55. https://doi.org/10.3390/antiox9010055
Ruberte AC, Ramos-Inza S, Aydillo C, Talavera I, Encío I, Plano D, Sanmartín C. Novel N,N′-Disubstituted Acylselenoureas as Potential Antioxidant and Cytotoxic Agents. Antioxidants. 2020; 9(1):55. https://doi.org/10.3390/antiox9010055
Chicago/Turabian StyleRuberte, Ana Carolina, Sandra Ramos-Inza, Carlos Aydillo, Irene Talavera, Ignacio Encío, Daniel Plano, and Carmen Sanmartín. 2020. "Novel N,N′-Disubstituted Acylselenoureas as Potential Antioxidant and Cytotoxic Agents" Antioxidants 9, no. 1: 55. https://doi.org/10.3390/antiox9010055
APA StyleRuberte, A. C., Ramos-Inza, S., Aydillo, C., Talavera, I., Encío, I., Plano, D., & Sanmartín, C. (2020). Novel N,N′-Disubstituted Acylselenoureas as Potential Antioxidant and Cytotoxic Agents. Antioxidants, 9(1), 55. https://doi.org/10.3390/antiox9010055