Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin
Abstract
1. Introduction
2. Factors Affecting Zeaxanthin Bioavailability
2.1. Thermal Processing
2.2. Esterification
2.3. Dietary Polysaccharides
2.4. Dietary Lipids
2.5. Food Matrix
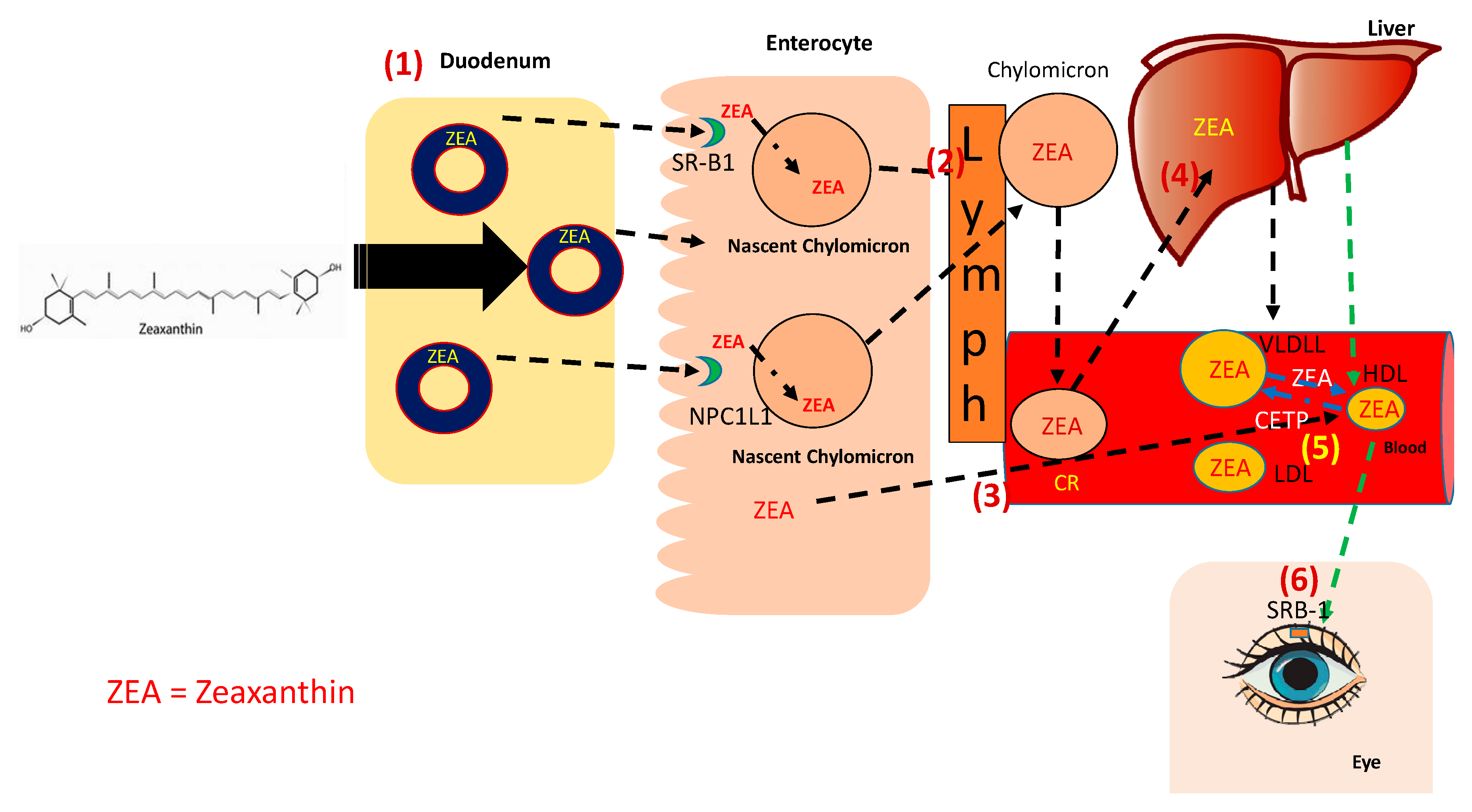
3. Absorption and Metabolism of Zeaxanthin
3.1. Digestion in the Intestinal Lumen
3.2. Absorption by Enterocytes
3.3. Efflux from Enterocytes
3.4. Transport in Blood
3.5. Tissue Distribution
3.6. Uptake of Zeaxanthin by the Eyes
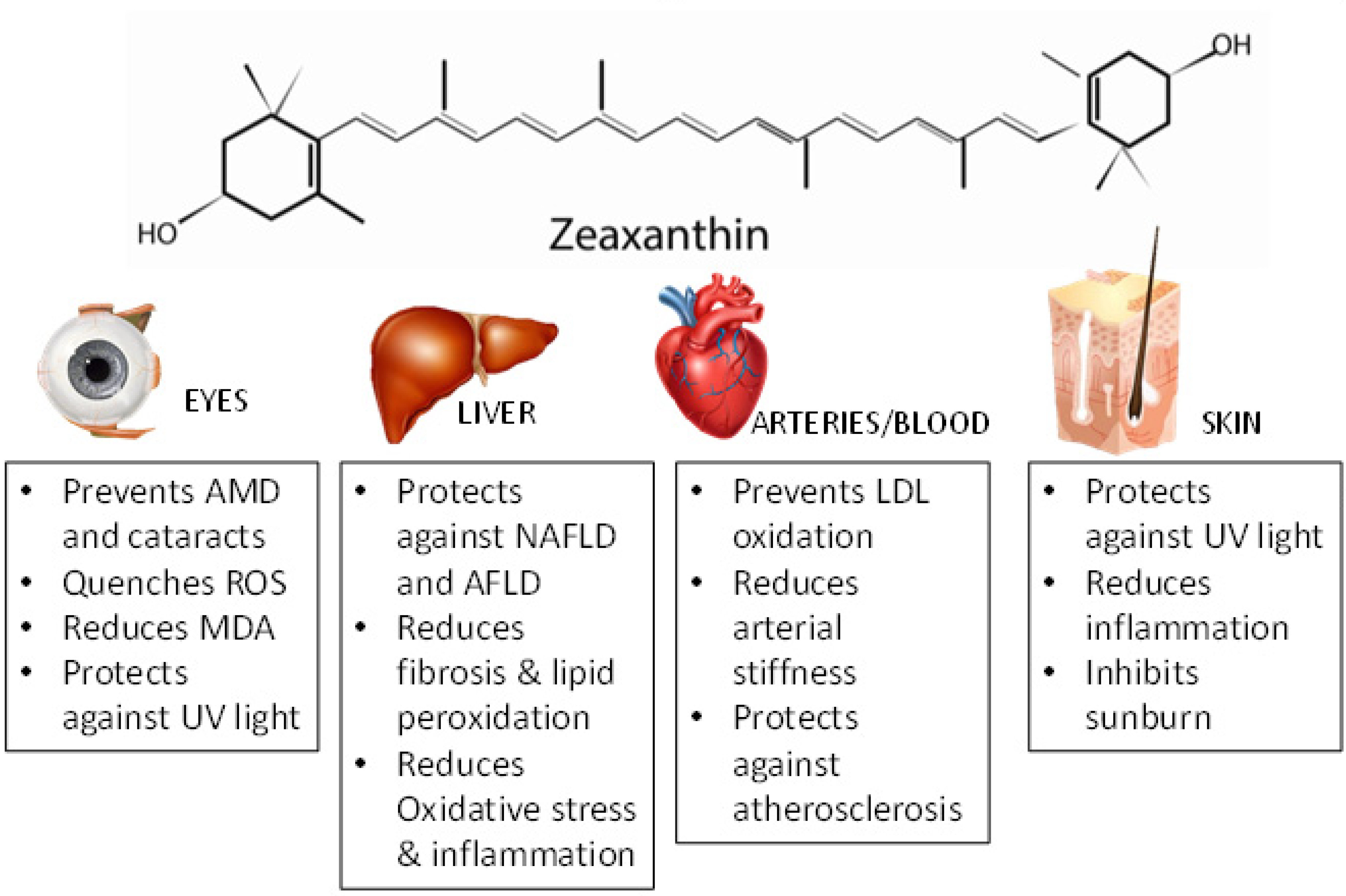
4. Effects of Zeaxanthin in Human Health
4.1. Antioxidant Properties of Zeaxanthin
4.2. Lutein and Eye Health
4.3. Zeaxanthin and Liver Diseases
4.4. Zeaxanthin and Atherosclerosis
4.5. Zeaxanthin and Skin Health
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sajilata, M.G.; Singhal, R.S.; Kamat, M.Y. The Carotenoid Pigment Zeaxanthin—A Review. Compr. Rev. Food Sci. Food Saf. 2008, 7, 29–49. [Google Scholar] [CrossRef]
- Sparrow, J.R.; Kim, S.R. The carotenoids of macular pigment and bisretinoid lipofuscin precursors in photoreceptor outer segments. In Carotenoids: Physical, Chemical and Biological Functions and Properties; CRC Press: Boca Raton, FL, USA, 2009; pp. 355–363. [Google Scholar]
- Nwachukwu, I.D.; Udenigwe, C.C.; Aluko, R.E. Lutein and zeaxanthin: Production technology, bioavailability, mechanisms of action, visual function, and health claim status. Trends Food Sci. Technol. 2016, 49, 74–84. [Google Scholar] [CrossRef]
- Delgado-Pelayo, R.; Hornero-Méndez, D. Identification and Quantitative Analysis of Carotenoids and Their Esters from Sarsaparilla (Smilax aspera L.) Berries. J. Agric. Food Chem. 2012, 60, 8225–8232. [Google Scholar] [CrossRef] [PubMed]
- Billsten, H.H.; Bhosale, P.; Yemelyanov, A.; Bernstein, P.S.; Polivka, T. Photophysical Properties of Xanthophylls in Carotenoproteins from Human Retina. Photochem. Photobiol. 2003, 78, 138–145. [Google Scholar] [CrossRef]
- Mozaffarieh, M.; Sacu, S.; Wedrich, A. The role of the carotenoids, lutein and zeaxanthin, in protecting against age-related macular degeneration: A review based on controversial evidence. Nutr. J. 2003, 2, 20. [Google Scholar] [CrossRef] [PubMed]
- Maiani, G.; Castón, M.J.P.; Catasta, G.; Toti, E.; Cambrodon, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Behsnilian, D.; Schlemmer, U.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef] [PubMed]
- Nimalaratne, C.; Lopes-Lutz, D.; Schieber, A.; Wu, J. Effect of domestic cooking methods on egg yolk xanthophylls. J. Agric. Food Chem. 2012, 60, 12547–12552. [Google Scholar] [CrossRef] [PubMed]
- Torregrosa, F.; Cortés, C.; Esteve, M.J.; Frigola, A. Effect of High-Intensity Pulsed Electric Fields Processing and Conventional Heat Treatment on Orange−Carrot Juice Carotenoids. J. Agric. Food Chem. 2005, 53, 9519–9525. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.; Chung, W.Y.; Wang, J.; Richelle, M.; Bucheli, P. Enhanced bioavailability of zeaxanthin in a milk-based formulation of wolfberry (Gou Qi Zi; Fructus barbarum L.). Br. J. Nutr. 2006, 96, 154–160. [Google Scholar] [CrossRef]
- Khoo, H.E.; Prasad, K.N.; Kong, K.W.; Jiang, Y.; Ismail, A. Carotenoids and their isomers: Color pigments in fruits and vegetables. Molecules 2011, 16, 1710–1738. [Google Scholar] [CrossRef]
- Schieber, A.; Carle, R. Occurrence of carotenoid cis-isomers in food: Technological, analytical, and nutritional implications. Trends Food Sci. Technol. 2005, 16, 416–422. [Google Scholar] [CrossRef]
- Pérez-Gálvez, A.; Martin, H.D.; Sies, H.; Stahl, E. Incorporation of carotenoids from paprika oleoresin into human chylomicrons. Br. J. Nutr. 2003, 89, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Breithaupt, D.E.; Weller, P.; Wolters, M.; Hahn, A. Comparison of plasma responses in human subjects after the ingestion of 3R,3R’-zeaxanthin dipalmitate from wolfberry (Lycium barbarum) and non-esterified 3R,3R’-zeaxanthin using chiral high-performance liquid chromatography. Br. J. Nutr. 2004, 91, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Hempel, J.; Fischer, A.; Fischer, M.; Hogel, J.; Bosy-Westphal, A.; Carle, R.; Schweiggeert, R.M. Effect of aggregation form on bioavailability of zeaxanthin in humans: A randomized cross-over study. Br. J. Nutr. 2017, 118, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.; Beck, M.; Elliott, J.; Etherve, S.; Roberts, R.; Schalch, W. Effects of formulation on the bioavailability of lutein and zeaxanthin: A randomized, double-blind, cross-over, comparative, single-dose study in healthy subjects. Eur. J. Nutr. 2013, 52, 1381–1391. [Google Scholar] [CrossRef] [PubMed]
- Mackie, A.R.; Macierzanka, A.; Aarak, K.; Rigby, N.M.; Parker, R.; Channell, G.A.; Harding, S.E.; Bajka, B.H. Sodium alginate decreases the permeability of intestinal mucus. Food Hydrocoll. 2013, 52, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Furr, H.C.; Clark, R.M. Intestinal absorption and tissue distribution of carotenoids. J. Nutr. Biochem. 1997, 8, 364–377. [Google Scholar] [CrossRef]
- Roodenburg, A.J.; Leenen, R.; van het Hof, K.H.; Weststrate, J.A.; Tijburg, L.B. Amount of fat in the diet affects bioavailability of lutein esters but not of α-carotene, β-carotene, and vitamin E in humans. Am. J. Clin. Nutr. 2000, 71, 1187–1193. [Google Scholar] [CrossRef] [PubMed]
- Handelman, G.J.; Nightingale, Z.D.; Lichtenstein, A.H.; Schaefer, E.J.; Blumberg, J.B. Lutein and zeaxanthin concentrations in plasma after dietary supplementation with egg yolk. Am. J. Clin. Nutr. 1999, 70, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Gleize, B.; Tourniaire, F.; Depezay, L.; Bott, R.; Nowicki, M.; Albino, L.; Lairon, D.; Kesse-Guyot, E.; Galan, P.; Hercberg, S.; et al. Effect of type of TAG fatty acids on lutein and zeaxanthin bioavailability. Br. J. Nutr. 2013, 110, 1–10. [Google Scholar] [CrossRef]
- Ballesteros, M.N.; Cabrera, R.M.; Saucedo, M.S.; Fernandez, M.L. Dietary cholesterol does not increase biomarkers for chronic disease in a pediatric population at risk from Northern Mexico. Am. J. Clin. Nutr. 2004, 80, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.M.; Herron, K.L.; Waters, D.; Fernandez, M.L. Hypo- and hyper-response to egg cholesterol predicts lutein and beta-carotene plasma concentrations in men and women. J. Nutr. 2006, 136, 601–607. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Norris, G.H.; Millar, C.; Blesso, C.; Fernandez, M.L. Intake of up to 3 eggs per day is associated with changes in HDL function and increased plasma antioxidants in healthy young adults. J. Nutr. 2017, 147, 323–339. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.M.; Waters, D.; Clark, R.M.; Contois, J.H.; Fernandez, M.L. Plasma LDL and HDL characteristics and carotenoid content are positively influenced by egg consumption in an elderly population. Nutr. Met. 2006, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Mutungi, G.; Waters, D.; Ratliff, J.; Puglisi, M.J.; Clark, R.M.; Volek, J.S.; Fernandez, M.L. Eggs distinctly modulate plasma carotenoid and lipoprotein subclasses in adult men following a carbohydrate restricted diet. J. Nutr. Biochem. 2010, 21, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C.N.; Andersen, C.J.; Bolling, B.; Fernandez, M.L. Egg intake improves carotenoid status by increasing HDL cholesterol in adults with metabolic syndrome. Food Funct. 2013, 4, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Tyssandier, V.; Reboul, E.; Dumas, J.F.; Bouteloup-Demage, C.; Armand, C.; Marcand, J.; Sallas, M.; Borel, P. Processing of vegetable-borne carotenoids in the human stomach and duodenum. Am. J. Physiol. Gastrointest. Physiol. 2003, 284, G913–G923. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Grolier, P.; Armand, M.; Partier, A.; Lafont, H.; Lairon, D.; Azais-Braesco, V. Carotenoids in biological emulsions: Solubility, surface-to-core distribution, and release from lipid droplets. J. Lipid Res. 1996, 37, 250–261. [Google Scholar]
- Weller, P.; Breithaupt, D.E. Identification and Quantification of Zeaxanthin Esters in Plants Using Liquid Chromatography−Mass Spectrometry. J. Agric. Food Chem. 2003, 51, 7044–7049. [Google Scholar] [CrossRef]
- Wingerath, T.; Sies, H.; Stahl, W. Xanthophyll esters in human skin. Arch. Biochem. Biophys. 1998, 355, 271–274. [Google Scholar] [CrossRef]
- Chitchumroonchokchai, C.; Failla, M.L. Hydrolysis of Zeaxanthin Esters by Carboxyl Ester Lipase during Digestion Facilitates Micellarization and Uptake of the Xanthophyll by Caco-2 Human Intestinal Cells. J. Nutr. 2006, 136, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Deming, D.M.; Erdman, J.W. Mammalian carotenoid absorption and metabolism. Pure Appl. Chem. 1999, 71, 2213–2223. [Google Scholar] [CrossRef]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Abou, L.; Mikail, C.; Ghiringhelli, O.; Andre, M.; Portugal, H.; Jourdeheuil-Rahmani, D.; Amiot, M.J.; Lairot, D.; Borel, P. Lutein transport by Caco-2 TC-7 cells occurs partly by a facilitated process involving the scavenger receptor class B type I (SR-BI). Biochem. J. 2005, 387, 455. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Suzuki, R.; Kobayashi, M.; Itagaki, S.; Hirano, T.; Noda, T.; Mizuno, S.; Sugawara, M.; Iseki, K. Involvement of cholesterol membrane transporter Niemann-Pick C1-like 1 in the intestinal absorption of lutein. J. Pharm. Pharm. 2012, 15, 256–264. [Google Scholar] [CrossRef]
- Borel, P. Genetic variations involved in interindividual variability in carotenoid status. Mol. Nutr. Food Res. 2012, 56, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Niesor, E.J.; Chaput, E.; Mary, J.L.; Staempfli, A.; Topp, A.; Stauffer, A.; Wang, H.; Durrwell, A. Effect of compounds affecting ABCA1 expression and CETP activity on the HDL pathway involved in intestinal absorption of lutein and zeaxanthin. Lipids 2014, 49, 1233–1243. [Google Scholar] [CrossRef]
- Li, B.; Vachali, P.; Frederick, J.M.; Bernstein, P.S. Identification of StARD3 as a lutein-binding protein in the macula of the primate retina. Biochemistry 2011, 50, 2541–2549. [Google Scholar] [CrossRef]
- Bhosale, P.; Li, B.; Sharifzadeh, M.; Gellermann, W.; Frederick, J.M.; Tsuchida, K.; Bernstein, P.S. Purification and partial characterization of a lutein-binding protein from human retina. Biochemistry 2009, 48, 4798–4807. [Google Scholar] [CrossRef]
- Desmarchelier, C.; Borel, P. Overview of carotenoid bioavailability determinants: From dietary factors to host genetic variations. Trends Food Sci. Technol. 2017, 69, 270–280. [Google Scholar] [CrossRef]
- Tyssandier, V.; Choubert, G.; Grolier, P.; Borel, P. Carotenoids, mostly the xanthophylls, exchange between plasma lipoproteins. Int. J. Vitam. Nutr. Res. 2002, 72, 300–308. [Google Scholar] [CrossRef]
- Kiokias, S.; Proestos, C.; Varzakas, T. A review of the structure, biosynthesis, absorption of carotenoids-analysis and properties of their common natural extracts. Curr. Res. Nutr. Food Sci. J. 2016, 4, 25–37. [Google Scholar] [CrossRef]
- Jeon, S.; Li, Q.; Rubakhin, S.S.; Sweedleer, J.V.; Smith, J.W.; Neuringer, M.; Kuchan, M.; Erdman, J.W., Jr. 13C-lutein is differentially distributed in tissues of an adult female rhesus macaque following a single oral administration: A pilot study. Nutr. Res. 2019, 61, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Shyam, R.; Gorusupudi, A.; Nelson, K.; Hoevath, M.P.; Bernstein, P.S. RPE65 has an additional function as the lutein to meso-zeaxanthin isomerase in the vertebrate eye. Proc. Natl. Acad. Sci. USA 2017, 114, 10882–10887. [Google Scholar] [CrossRef] [PubMed]
- Connor, W.E.; Duell, P.B.; Kean, R.; Wang, Y. The prime role of HDL to transport lutein into the retina: Evidence from HDL-deficient WHAM chicks having a mutant ABCA1 transporter. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4226–4231. [Google Scholar] [CrossRef] [PubMed]
- During, A.; Doraiswamy, S.; Harrison, E.H. Xanthophylls are preferentially taken up compared with β-carotene by retinal cells via a SRBI-dependent mechanism. J. Lipid Res. 2008, 49, 1715–1724. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Stahl, W. Vitamins E and C, beta-carotene, and other carotenoids as antioxidants. Am. J. Clin. Nutr. 1995, 62, 1315S–1321S. [Google Scholar] [CrossRef] [PubMed]
- Ribaya-Mercado, J.D.; Blumberg, J.B. Lutein and zeaxanthin and their potential roles in disease prevention. J. Am. Coll. Nutr. 2004, 23, 567S–587S. [Google Scholar] [CrossRef] [PubMed]
- Ojima, F.; Sakamoto, H.; Ishiguro, Y.; Terao, J. Consumption of carotenoids in photosensitized oxidation of human plasma and plasma low-density lipoprotein. Free Rad. Biol. Med. 1993, 15, 377–384. [Google Scholar] [CrossRef]
- Dhalla, N.S.; Temsah, R.M.; Netticadan, T. Role of oxidative stress in cardiovascular diseases. J. Hypertens. 2000, 18, 655–673. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A. Oxidative stress in cataracts. Pathophysiology 2006, 13, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Tuzcu, M.; Orhan, C.; Muz, O.E.; Sahin, N.; Juturu, V.; Sahin, V. Lutein and zeaxanthin isomers modulates lipid metabolism and the inflammatory state of retina in obesity-induced high-fat diet rodent model. BMC Ophthalmol. 2017, 17, 129. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Zou, X.; Gao, J.; Zheng, Y.; Wang, X.; Chen, C.; Cao, K.; Xu, J.; Li, Y.; Lu, W.; Liu, J.; et al. Zeaxanthin induces Nrf2-mediated phase II enzymes in protection of cell death. Cell Death Dis. 2014, 5, e1218. [Google Scholar] [CrossRef] [PubMed]
- Bhosale, P.; Bernstein, P.S. Synergistic effects of zeaxanthin and its binding protein in the prevention of lipid membrane oxidation. Biochim. Et Biophys. Acta (BBA) Mol. Basis Dis. 2005, 1740, 116–121. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, S.H.; Min, D.B. Effects, quenching mechanisms, and kinetics of carotenoids in chlorophyll-sensitized photooxidation of soybean oil. J. Agric. Food Chem. 1990, 38, 1630–1634. [Google Scholar] [CrossRef]
- Kim, S.R.; Nakanishi, K.; Itagaki, Y.; Sparrow, J.R. Photooxidation of A2-PE, a photoreceptor outer segment fluorophore, and protection by lutein and zeaxanthin. Exp. Eye Res. 2006, 82, 828–839. [Google Scholar] [CrossRef]
- Cantrell, A.; McGarvey, D.J.; Truscott, T.G.; Rancan, F.; Bohm, F. Singlet oxygen quenching by dietary carotenoids in a model membrane environment. Arch. Biochem. Biophys. 2003, 412, 47–54. [Google Scholar] [CrossRef]
- Rapp, L.M.; Maple, S.S.; Choi, J.H. Lutein and Zeaxanthin Concentrations in Rod Outer Segment Membranes from Perifoveal and Peripheral Human Retina. Investig. Ophtalmol. Vis. Sci. 2000, 41, 1200–1209. [Google Scholar]
- Khachik, F.; Bernstein, P.S.; Garland, D.L. Identification of lutein and zeaxanthin oxidation products in human and monkey retinas. Investig. Ophtalmol. Vis. Sci. 1997, 38, 1802–1811. [Google Scholar]
- National Eye Institute. Age-Related Macular Degeneration (AMD). Available online: https://nei.nih.gov/eyedata/amd (accessed on 15 June 2019).
- Kovach, J.L.; Schwartz, S.G.; Flynn, H.W., Jr.; Scott, I.U. Anti-VEGF Treatment Strategies for Wet AMD. J. Ophthalmol. 2012, 2012, 786870. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Mayne, S.T.; Gomez, C.M.; Tibor, S.E.; Twaroska, E.E. Macular pigment in donor eyes with and without AMD: A case-control study. Investig. Ophtalmol. Vis. Sci. 2001, 42, 235–240. [Google Scholar]
- Nolan, J.; Kenny, R.; O’Regan, C.; Cronin, H.; Loughman, J.; Connolly, E.E.; Kearney, P.; Loane, E.; Beatty, S. Macular Pigment Optical Density in an Ageing Irish Population: The Irish Longitudinal Study on Ageing. Ohptalmic. Res. 2010, 44, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Lutein and zeaxanthin status and risk of age-related macular degeneration. Investig. Ophtalmol. Vis. Sci. 2003, 44, 2461–2465. [Google Scholar] [CrossRef] [PubMed]
- Akuffo, K.O.; Nolan, J.M.; Howard, A.N.; Moran, R.; Stack, J.; Klein, B.E.; Meuer, S.M.; Sabour-Pickett, S.; Thurnham, D.I.; Beatty, S. Sustained supplementation and monitored response with differing carotenoid formulations in early age-related macular degeneration. Eye 2015, 29, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Garber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary Carotenoids, Vitamins A, C, and E, and Advanced Age-Related Macular Degeneration. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef]
- Young, R.W. Pathophysiology of age-related macular degeneration. Surv. Ophtalmol. 1987, 31, 291–306. [Google Scholar] [CrossRef]
- Chang, J.Y.; Bora, P.S.; Bora, N.S. Prevention of Oxidative Stress-Induced Retinal Pigment Epithelial Cell Death by the PPAR Agonists, 15-Deoxy-Delta 12, 14-Prostaglandin. PPAR Res. 2008, 2008, 720163. [Google Scholar] [CrossRef]
- Keller, J.N.; Dimayuga, E.; Chen, Q.; Thorpe, J.; Gee, J.; Ding, Q. Autophagy, proteasomes, lipofuscin, and oxidative stress in the aging brain. Int. J. Biochem. Cell Biol. 2004, 36, 2376–2391. [Google Scholar] [CrossRef]
- Nociari, M.M.; Kiss, S.; Rodriguez-Boulan, E. Lipofuscin Accumulation into and Clearance from Retinal Pigment Epithelium Lysosomes: Physiopathology and Emerging Therapeutics. In Lysosomes-Associated Diseases and Methods to Study Their Function; IntechOpen: London, UK, 2017. [Google Scholar]
- Sundelin, S.P.; Nilsson, S.E.G. Lipofuscin-formation in retinal pigment epithelial cells is reduced by antioxidants. Free Radic. Biol. Med. 2001, 31, 217–225. [Google Scholar] [CrossRef]
- Bhosale, P.; Serban, B.; Bernstein, P.S. Retinal carotenoids can attenuate formation of A2E in the retinal pigment epithelium. Arch. Biochem. Biophys. 2009, 483, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R.; West, S.; Munoz, B.; Rosenthal, F.S.; Bressler, S.B.; Bressler, N.M. The long-term effects of visible light on the eye. Arch. Ophtalmol. 1992, 110, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Ann. Rev. Nutr. 2003, 23, 171–201. [Google Scholar] [CrossRef] [PubMed]
- Junghans, A.; Sies, H.; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Bioiphys. 2001, 391, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Barker, F.M., II; Snodderly, D.M.; Johnson, E.J.; Schalchk, W.; Koepcke, W.; Gerss, J.; Neuringer, M. Nutritional Manipulation of Primate Retinas, V: Effects of Lutein, Zeaxanthin, and n–3 Fatty Acids on Retinal Sensitivity to Blue-Light–Induced Damage. Investig. Ophtalmol. Vis. Sci. 2011, 52, 3934–3942. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.-H. Nonalcoholic fatty liver disease: Molecular mechanisms for the hepatic steatosis. Clin. Mol. Hepatol. 2013, 19, 210–215. [Google Scholar] [CrossRef]
- Byrne, C.D.; Targher, G. NAFLD: A multisystem disease. J. Hepatol. 2015, 62, S47–S64. [Google Scholar] [CrossRef] [PubMed]
- Sayiner, M.; Koenig, A.; Henry, L.; Younossi, Z.M. Epidemiology of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis in the United States and the Rest of the World. Clin. Liver Dis. 2016, 20, 205–214. [Google Scholar] [CrossRef]
- Li, S.; Tan, H.-Y.; Wang, N.; Zhang, Z.-J.; Lao, L.; Wong, C.-W.; Feng, Y. The Role of Oxidative Stress and Antioxidants in Liver Diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef]
- Angulo, P. Nonalcoholic Fatty Liver Disease. N. Engl. J. Med. 2014, 346, 1221–1231. [Google Scholar] [CrossRef]
- Nassir, F.; Rector, R.S.; Hammoud, G.M.; Ibdah, J.A. Pathogenesis and Prevention of Hepatic Steatosis. Gastroenterol. Hepatol. (N. Y.) 2015, 11, 167–175. [Google Scholar]
- Murillo, A.G.; Fernandez, M.L. Potential of Dietary Non-Provitamin A Carotenoids in the Prevention and Treatment of Diabetic Microvascular Complications. Adv. Nutr. 2016, 7, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Lawler, T.; Mares, J. Dietary Carotenoids and Non-Alcoholic Fatty Liver Disease among US Adults, NHANES 2003–2014. Nutrients 2019, 11, 1101. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.M.; Hall, J.D.; Patel, J.; Lee, J.R.; Marcus, D.M.; Sridhar, S.; Sridhar, S.; Romero, M.J.; Labazi, M.; Caldwell, R.W.; et al. Protective effects of the carotenoid zeaxanthin in experimental nonalcoholic steatohepatitis. Dig. Dis. Sci. 2009, 54, 1460–1464. [Google Scholar] [CrossRef] [PubMed]
- Toshikuni, N.; Tsutsumi, M.; Arisawa, T. Clinical differences between alcoholic liver disease and nonalcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 8393–8406. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Wang, J.; Xing, F.; Han, T.; Jiao, R.; Liong, E.C.; Fung, M.L.; So, K.F.; Tipoe, G.L. Zeaxanthin dipalmitate therapeutically improves hepatic functions in an alcoholic fatty liver disease model through modulating MAPK pathway. PLoS ONE 2014, 9, e95214. [Google Scholar] [CrossRef]
- Gao, H.; Lv, Y.; Liu, Y.; Li, J.; Wang, X.; Zhou, Z.; Ouyang, S.; Guo, Y.; Zhang, J.; Hao, X.; et al. Wolfberry-derived zeaxanthin dipalmitate attenuates ethanol-induced hepatic damage. Mol. Nutr. Food Res. 2019, 63, 1–47. [Google Scholar] [CrossRef]
- Torres, N.; Guevara-Cruz, M.; Velazquez-Villegas, L.A.; Tovar, A.R. Nutrition and Atherosclerosis. Arch. Med. Res. 2015, 46, 408–426. [Google Scholar] [CrossRef]
- Barquera, S.; Pedroza-Tobías, A.; Medina, C.; Hernández-Barrera, L.; Bibbins-Domingo, K.; Lozano, R.; Moran, A.E. Global Overview of the Epidemiology of Atherosclerotic Cardiovascular Disease. Arch. Med. Res. 2015, 46, 328–338. [Google Scholar] [CrossRef]
- Aziz, M.; Yadav, K. Pathogenesis of Atherosclerosis A Review. Med. Clin. Rev. 2016, 2, 1–6. [Google Scholar]
- Carmena, R. Atherogenic Lipoprotein Particles in Atherosclerosis. Circulation 2004, 109, III-2–III-7. [Google Scholar] [CrossRef]
- Violi, F.; Micheletta, F.; Luliano, L. Antioxidants and atherosclerosis: An overview. Eur. Hear. J. Suppl. 2002, 4, B17–B21. [Google Scholar] [CrossRef][Green Version]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and Atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative Stress in Atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; Riccioni, G. Carotenoids: Potential allies of cardiovascular health? Food Nutr. Res. 2015, 59, 26762. [Google Scholar]
- Wang, W.; Connor, S.L.; Johnson, E.J.; Klein, M.L.; Hughes, S.; Connor, W.E. Effect of dietary lutein and zeaxanthin on plasma carotenoids and their transport in lipoproteins in age-related macular degeneration. Am. J. Clin. Nutr. 2007, 85, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Ouimet, M.; Barrett, T.J.; Fisher, E.A. Review Basic Mechanisms and Their Roles in Vascular Health and Disease. Circ. Res. 2019, 124, 1505–1518. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chung, S.; Mccullough, M.L.; Song, W.O.; Fernandez, M.L.; Koo, S.I.; Chun, O.K. Dietary Carotenoids Are Associated with Cardiovascular Disease Risk Biomarkers Mediated by Serum Carotenoid. J. Nutr. 2014, 144, 1067–1074. [Google Scholar] [CrossRef]
- Carpenter, K.L.H.; Van Der Veen, C.; Hird, R.; Dennis, I.F.; Ding, T.; Mitchinson, M.J. The carotenoids beta-carotene, canthaxanthin and zeaxanthin inhibit macrophage-mediated LDL oxidation. FEBS Lett. 1997, 401, 262–266. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Taguchi, C.; Saita, E.; Suzuki-sugihara, N.; Nishiyama, H.; Wang, W.; Masuda, Y.; Kondo, K. Additional consumption of one egg per day increases serum lutein plus zeaxanthin concentration and lowers oxidized low-density lipoprotein in moderately hypercholesterolemic males. Food Res. Int. 2017, 99, 944–949. [Google Scholar] [CrossRef]
- Dwyer, J.H.; Navab, M.; Dwyer, K.M.; Hassan, K.; Sun, P.; Shircore, A.; Hama-Levy, S.; Hough, G.; Wang, X.; Drake, T.; et al. Oxygenated Carotenoid Lutein and Progression of Early Atherosclerosis: The Los Angeles Atherosclerosis Study. Circulation 2001, 103, 2922–29277. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.H.; Paul-labrador, M.J.; Fan, J.; Shircore, A.M.; Merz, C.N.B.; Dwyer, K.M. Progression of Carotid Intima-Media Thickness and Plasma Antioxidants: The Los Angeles Atherosclerosis Study. Atheroscler. Thromb. Vasc. Biol. 2004, 24, 313–319. [Google Scholar] [CrossRef]
- Zou, Z.; Xu, X.; Huang, Y.; Xiao, X.; Ma, L.; Sun, T.; Dong, P.; Wang, X.; Lin, X. High serum level of lutein may be protective against early atherosclerosis: The Beijing atherosclerosis study. Atherosclerosis 2011, 219, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Kijlstra, A.; Tian, Y.; Kelly, E.; Berendschot, T.T.J.M. Lutein: More than just a filter for blue light. Prog. Retin. Eye Res. 2012, 31, 303–315. [Google Scholar] [CrossRef]
- Marie, M.; Bigot, K.; Angebault, C.; Barrau, C.; Gondouin, P.; Pagan, D.; Fouquet, S.; Villette, T.; Lenaers, G.; Picaud, S.; et al. Light action spectrum on oxidative stress and mitochondrial damage in A2E-loaded retinal pigment epithelium cells. Cell Death Dis. 2018, 9, 287. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.; Green, J.; Lewis, B. Lutein and zeaxanthin in eye and skin health. Clin. Dermatol. 2009, 27, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.L. Lutein, Zeaxanthin, and Skin Health. Am. J. Lifestyle Med. 2013, 7, 182–185. [Google Scholar] [CrossRef]
- Astner, S.; An, W.; Goukassian, D.; Pathak, M.A. Dietary Lutein/Zeaxanthin Decreases Ultraviolet B-Induced Epidermal Hyperproliferation and Acute In£ammation in Hairless Mice. J. Investig. Dermatol. 2003, 121, 399–405. [Google Scholar]
- Palombo, P.; Fabrizi, G.; Ruocco, V.; Ruocco, E.; Fluhr, J.; Roberts, R.; Morganti, P. Beneficial Long-Term Effects of Combined Oral/Topical Antioxidant Treatment with the Carotenoids Lutein and Zeaxanthin on Human Skin: A Double-Blind, Placebo-Controlled Study. Ski. Pharmacol. Physiol. 2007, 20, 199–210. [Google Scholar] [CrossRef]
- Johnson, E.J. A possible role for lutein and zeaxanthin in cognitive function in the elderly. Am. J. Clin. Nutr. 2012, 96, 1161S–1165S. [Google Scholar] [CrossRef] [PubMed]
| Factor | Bioavailability | Population Studied | Reference |
|---|---|---|---|
| Thermal Processing | ↑ lower temperatures | In vitro | [7] |
| ↑ lower temperatures | In vitro | [8] | |
| ↑ higher temperatures | In vitro | [9] | |
| ↑ higher temperatures | In vitro | [10] | |
| Esterification | ↑ esterified vs. Free | 12 healthy volunteers | [14] |
| Esterification | ↑ esterified vs. Free | 16 healthy volunteers | [15] |
| Lipids | ↑ SFA vs. PUFA | 11 hypercholesterolemic men | [20] |
| ↑ SFA compared to PUFA or MUFA | Rats | [21] | |
| Carbohydrates | ↑ SMZ vs. AMZ | 48 healthy volunteers | [16] |
| Eggs | ↑ eggs vs. egg substitute | 224 volunteers including children, healthy young and old, overweight/obese and metabolic syndrome participants | [22,23,24,25,26,27] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murillo, A.G.; Hu, S.; Fernandez, M.L. Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin. Antioxidants 2019, 8, 390. https://doi.org/10.3390/antiox8090390
Murillo AG, Hu S, Fernandez ML. Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin. Antioxidants. 2019; 8(9):390. https://doi.org/10.3390/antiox8090390
Chicago/Turabian StyleMurillo, Ana Gabriela, Siqi Hu, and Maria Luz Fernandez. 2019. "Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin" Antioxidants 8, no. 9: 390. https://doi.org/10.3390/antiox8090390
APA StyleMurillo, A. G., Hu, S., & Fernandez, M. L. (2019). Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin. Antioxidants, 8(9), 390. https://doi.org/10.3390/antiox8090390






