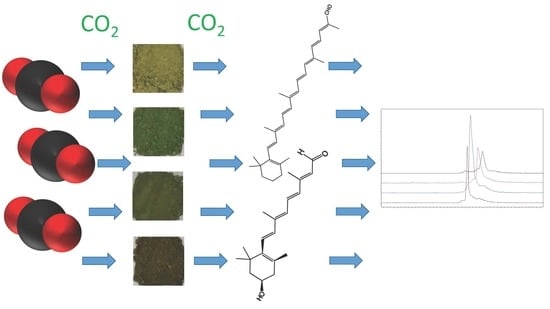
First Apocarotenoids Profiling of Four Microalgae Strains
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Strain Selection and Biomass Production
2.3. Sample Preparation
2.4. SFE-SFC-MS Instrumentation
2.5. SFE-SFC-MS Analytical Conditions
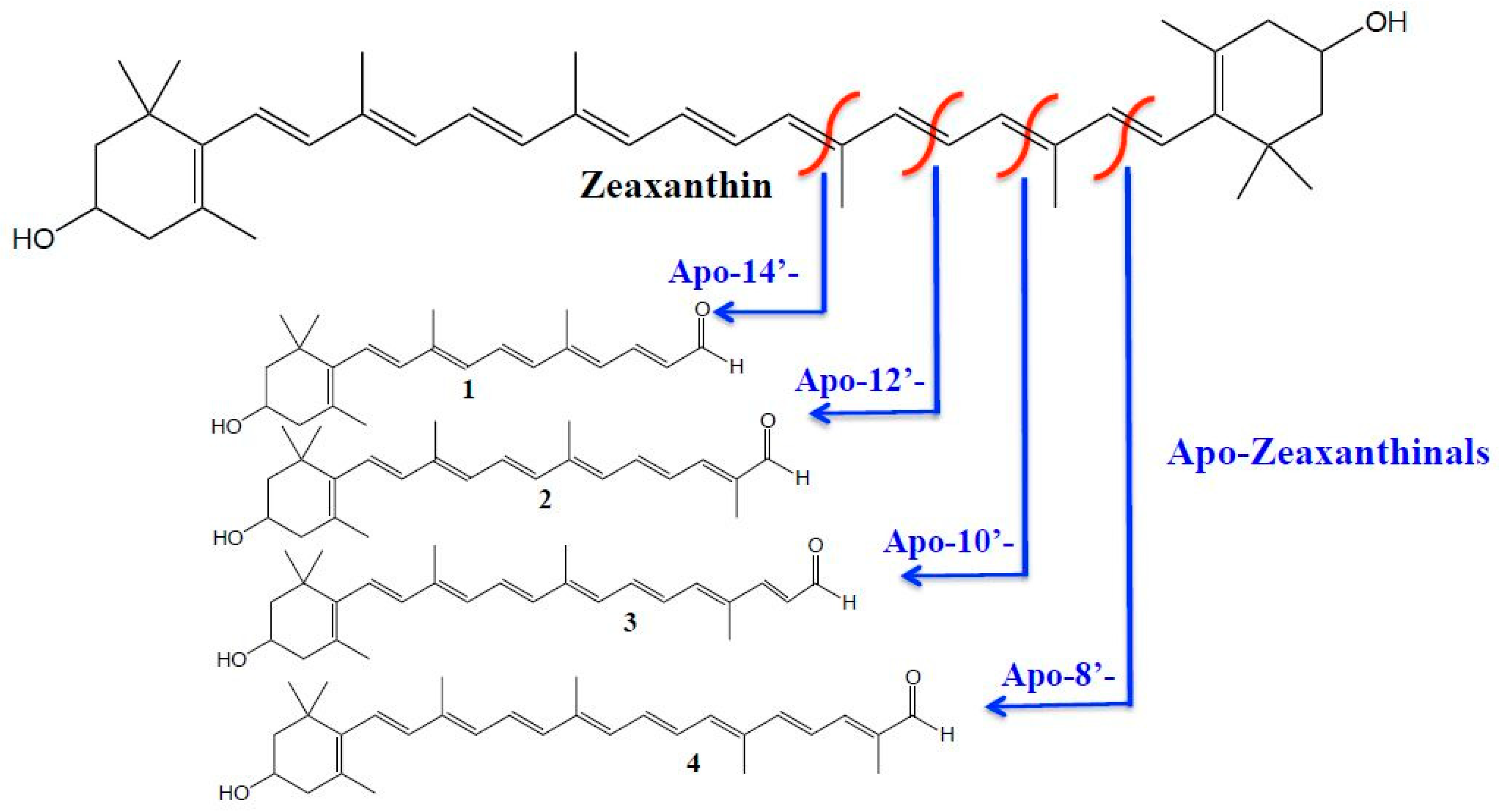
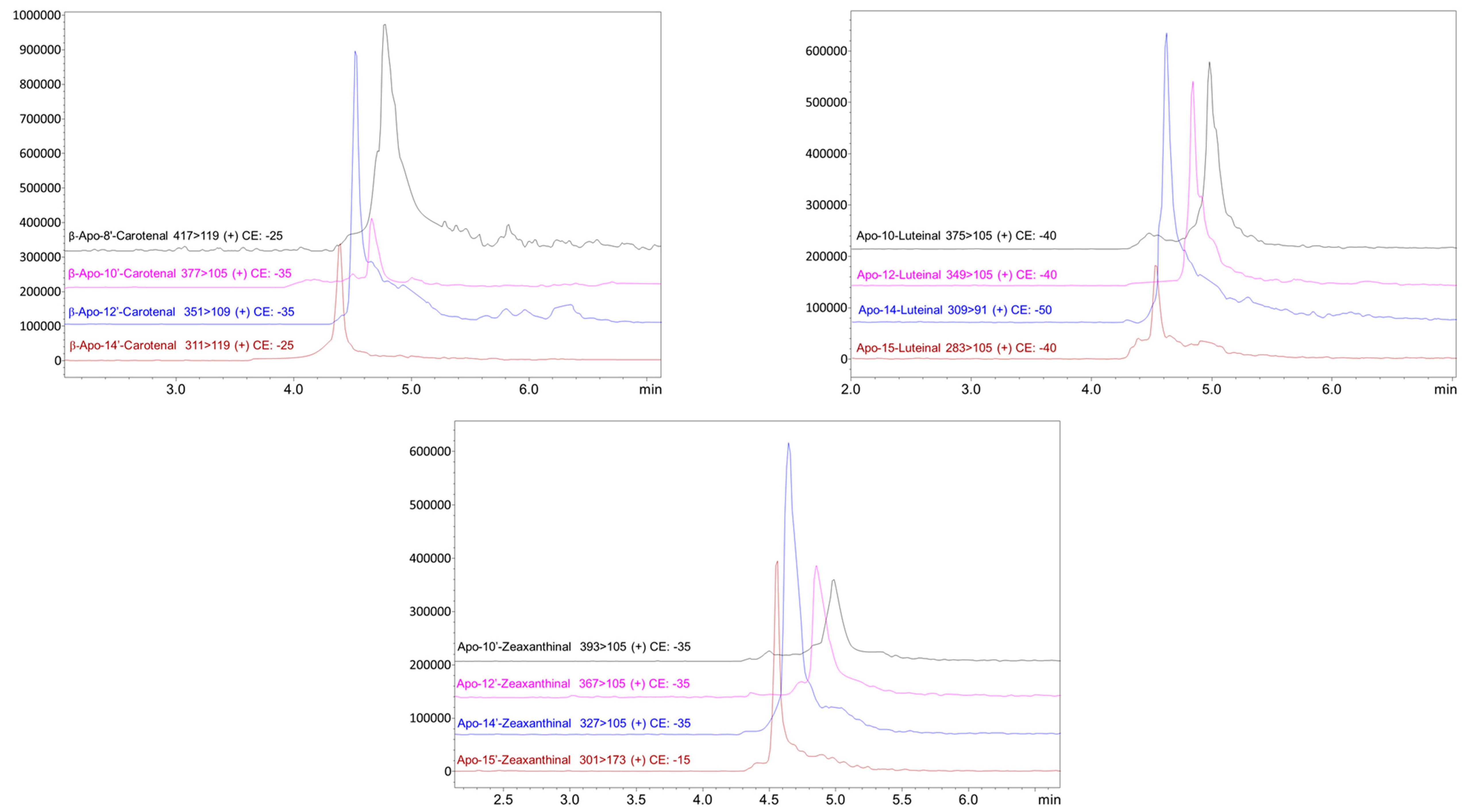
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gong, M.; Bassi, A. Carotenoids from microalgae: A review of recent developments. Biotechnol. Adv. 2016, 34, 1396–1412. [Google Scholar] [CrossRef] [PubMed]
- Matos, J.; Cardoso, C.; Bandarra, N.M.; Afonso, C. Microalgae as healthy ingredients for functional food: A review. Food Funct. 2017, 8, 2672–2685. [Google Scholar] [CrossRef] [PubMed]
- Sathasivam, R.; Ki, J.-S. A review of the biological activities of microalgal carotenoids and their potential use in healthcare and cosmetic industries. Mar. Drugs 2018, 16, 26. [Google Scholar] [CrossRef] [PubMed]
- Mc Gee, D.; Archer, L.; Paskuliakova, A.; Mc Coy, G.R.; Fleming, G.T.A.; Gillespie, E.; Touzet, N. Rapid chemotaxonomic profiling for the identification of high-value carotenoids in microalgae. J. Appl. Phycol. 2018, 30, 385–399. [Google Scholar] [CrossRef]
- Maroneze, M.M.; Jacob-Lopez, E.; Queiroz Zepka, L.; Roca, M.; Perez-Galvez, A. Esterified carotenoids as new food components in cyanobacteria. Food Chem. 2019, 287, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Minhas, A.K.; Hodgson, P.; Barrow, C.; Adholeya, A. A review on the assessment of stress conditions for simultaneous production of microalgal lipids and carotenoids. Front. Microbiol. 2016, 7, 546. [Google Scholar] [CrossRef]
- Liu, J.; Sun, Z.; Gerken, H. Recent Advances in Microalgae Biotechnology; OMICS Group eBooks: Foster City, CA, USA, 2014. [Google Scholar]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids Volume 5: Nutrition and Health; Birkhauser: Basel, Switzerland, 2009. [Google Scholar]
- Young, A.J.; Lowe, G.L. Carotenoids-Antioxidant Properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef]
- Ceron-Garcia, M.C.; Gonzalez-Lopez, C.V.; Camacho-Rodriguez, J.; Lopez-Rosales, L.; Garcia-Camacho, F.; Molina-Grima, E. Maximizing carotenoid extraction from microalgae used as food additives and determined by liquid chromatography (HPLC). Food Chem. 2018, 257, 316–324. [Google Scholar] [CrossRef]
- Abrahamsson, V.; Rodriguez-Meizoso, I.; Turner, C. Determination of carotenoids in microalgae using supercritical fluid extraction and chromatography. J. Chromatogr. A 2012, 1250, 63–68. [Google Scholar] [CrossRef]
- Pour Hosseini, S.R.; Tavakoli, O.; Sarrafzadeh, M.H. Experimental optimization of SC-CO2 extraction of carotenoids from Dunaliella salina. J. Supercrit. Fluids 2017, 121, 89–95. [Google Scholar] [CrossRef]
- Hou, X.; Rivers, J.; Leon, P.; McQuinn, R.P.; Pogson, B.J. Synthesis and function of apocarotenoid signals in plants. Trends Plant Sci. 2016, 21, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, D.; Donato, P.; Dugo, P.; Mondello, L. Recent analytical techniques advances in the carotenoids and their derivatives determination in various matrixes. J. Agric. Food Chem. 2018, 66, 3302–3307. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, A.; Harrison, E.H. Carotenoid metabolism in mammals, including man: Formation, occurrence, and function of apocarotenoids. J. Lipid Res. 2013, 54, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.H.; Quadro, L. Apocarotenoids: Emerging roles in mammals. Annu. Rev. Nutr. 2018, 38, 153–172. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, M.; Giuffrida, D.; Dugo, P.; Mondello, L. Direct online extraction and determination by supercritical fluid extraction with chromatography and mass spectrometry of targeted carotenoids from Habanero peppers (Capsicum chinense Jacq.). J. Sep. Sci. 2017, 40, 3905–3913. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, D.; Zoccali, M.; Arigò, A.; Cacciola, F.; Osorio-Roa, C.; Dugo, P.; Mondello, L. Comparison of different analytical techniques for the analysis of carotenoids in tamarillo (Solanum betaceum Cav.). Arch. Biochem. Biophys. 2018, 646, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, E.B.; Rodriguez-Amaya, D.B. Formation of apocarotenals and epoxycarotenoids from β-carotene by chemical reactions and by autoxidation in model system and processed foods. Food Chem. 2007, 101, 563–572. [Google Scholar] [CrossRef]
- Giuffrida, D.; Zoccali, M.; Giofrè, S.V.; Dugo, P.; Mondello, L. Apocarotenoids determination in Capsicum chinense Jacq. cv Habanero, by supercritical fluid chromatography-mass spectrometry. Food Chem. 2017, 231, 316–323. [Google Scholar] [CrossRef]
- Zoccali, M.; Giuffrida, D.; Salafia, F.; Giofrè, S.V.; Mondello, L. Carotenoids and apocarotenoids determination in intact human blood samples by online supercritical fluid extraction-supercritical fluid chromatography-tandem mass spectrometry. Anal. Chim. Acta 2018, 1032, 40–47. [Google Scholar] [CrossRef]
- Guillard, R.R.L.; Hargraves, P.E. Stichochrysis immobilis is a diatom, not a chrysophyte. Phycologia 1993, 32, 234–236. [Google Scholar] [CrossRef]
- Harris, E.H. The Chlamydomonas Sourcebook. A comprehensive Guide to Biology and Laboratory Use, 2nd ed.; Academic Press Inc.: San Diego, CA, USA, 1989. [Google Scholar]
- Singh, S.; Kate, B.N.; Banerjee, U.C. Bioactive compounds from cyanobacteria and microalgae: An overview. Crit. Rev. Biotechnol. 2005, 25, 73–95. [Google Scholar] [CrossRef] [PubMed]
- Galasso, C.; Corinaldesi, C.; Sansone, C. Carotenoids from marine organisms: Biological functions and industrial applications. Antioxidants 2017, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Fanning, K.; Netzel, M.; Turner, W.; Li, Y.; Schenk, P.M. Profiling of carotenoids and antioxidant capacity of microalgae from subtropical coastal and brackish waters. Food Chem. 2014, 165, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Skjånes, K.; Rebours, C.; Lindblad, P. Potential for green microalgae to produce hydrogen, pharmaceuticals and other high value products in a combined process. Crit. Rev. Biotechnol. 2013, 33, 172–215. [Google Scholar] [CrossRef] [PubMed]
- Forján, E.; Garbayo, I.; Casal, C.; Vílchez, C. Enhancement of carotenoid production in Nannochloropsis by phosphate and sulphur limitation. Commun. Curr. Res. Educ. Top. Trends Appl. Microbiol. 2007, 1, 356–364. [Google Scholar]
- Safafar, H.; van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef] [PubMed]
- Di Lena, G.; Casini, I.; Lucarini, M.; Lombardi-Boccia, G. Carotenoid profiling of five microalgae species from large-scale production. Food Res. Int. 2019, 120, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Montero, L.; Sedghi, M.; Garcia, Y.; Almeida, C.; Safi, C.; Engelen-Smit, N.; Cifuentes, A. Pressurized liquid extraction of pigments from Chlamydomonas sp. and chemical characterization by HPLC-MS/MS. J. Anal. Test. 2018, 2, 149–157. [Google Scholar] [CrossRef]
- Cordero, B.F.; Obraztsova, L.; Couso, I.; Leon, R.; Vargas, M.A.; Rodriguez, H. Enhancement of lutein production in Chlorella sorokiniana (Chlorophyta) by improvement of culture conditions and random mutagenesis. Mar. Drugs 2011, 9, 1607–1624. [Google Scholar] [CrossRef] [PubMed]
- Lubian, L.M. Nannochloropsis (Eustigmatophyceae) as source of commercially valuable pigments. J. Appl. Phycol. 2000, 12, 249–255. [Google Scholar] [CrossRef]
- D’amico, S.; Collins, T.; Marx, J.C.; Feller, G.; Gerday, C. Psychrophilic microorganism: Challenges for life. EMBO Rep. 2006, 7, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Sommella, E.; Conte, G.M.; Salviati, E.; Pepe, G.; Bertamino, A.; Ostacolo, C.; Sansone, F.; del Prete, F.; Aquino, R.P.; Campiglia, P. Fast profiling of natural pigments in different Spirulina (Arthrospira platensis) dietary supplements by DI-FT-ICR and evaluation of their antioxidant potential by pre-column DPPH-UHPLC assay. Molecules 2018, 23, 1152. [Google Scholar] [CrossRef] [PubMed]
- Hornero-Mendez, D. Occurrence of carotenoid esters in food. In Carotenoid Esters in Food: Physical, Chemical and Biological Properties; Mercadante, A.Z., Ed.; Royal Society of Chemistry: Cambridge, UK, 2019; Chapter 7; pp. 182–284. [Google Scholar]

| Apocarotenoids | SIM (−) | MRM Transition (CE) | ||
|---|---|---|---|---|
| m/z | Quantifier | Qualifier | Q/q % | |
| β-Apo-8’-Carotenal | 416 | + 417>119 (−25) | + 417>105 (−35) | 73 |
| β-Apo-10’-Carotenal | 376 | + 377>105 (−35) | + 377>119 (−30) | 79 |
| β-Apo-12’-Carotenal | 350 | + 351>105 (−35) | + 351>119 (−25) | 74 |
| β-Apo-14’-Carotenal | 310 | + 311>105 (−25) | + 311>119 (−25) | 77 |
| Apo-8’-Zeaxanthinal | 432 | + 433>119 (−30) | + 433>105 (−35) | 95 |
| Apo-10’-Zeaxanthinal | 392 | + 393>105 (−35) | + 393>119 (−25) | 92 |
| Apo-12’-Zeaxanthinal | 366 | + 367>105 (−35) | + 367>119 (−30) | 80 |
| Apo-14’-Zeaxanthinal | 326 | + 327>105 (−35) | + 327>119 (−30) | 61 |
| Apo-15-Zeaxanthinal | 300 | + 301>173 (−15) | + 301>105 (−30) | 57 |
| Apo-8-Luteinal | 432 | + 415>119 (−40) | + 415>91 (−50) | 95 |
| Apo-10-Luteinal | 392 | + 375>105 (−40) | + 375>91 (−50) | 91 |
| Apo-12-Luteinal | 366 | + 349>105 (−40) | + 349>91 (−50) | 90 |
| Apo-14-Luteinal | 326 | + 309>91 (−50) | + 309>105 (−40) | 55 |
| Apo-15-Luteinal | 300 | + 283>105 (−40) | + 283>91 (−50) | 95 |
| Apo-8’-violaxanthin | 448 | n.d. | n.d. | |
| Apo-12’-violaxanthal | 382 | n.d. | n.d. | |
| Apo-14’-violaxanthal | 342 | n.d. | n.d. | |
| Apo-15’-violaxanthal | 316 | n.d. | n.d. | |
| Apo-8’-Fucoxanthinal | 464 | n.d. | n.d. | |
| Apo-10’-Fucoxanthinal | 424 | n.d. | n.d. | |
| Apo-14’-Fucoxanthinal | 358 | n.d. | n.d. | |
| Apo-15’-Fucoxanthinal | 332 | n.d. | n.d. | |
| Apocarotenoids-Esters | SIM (−) | MRM transition (CE) | ||
| Apo-10’-Zeaxanthinal-C4:0 | 462 | + 463>105 (−40) | + 463>119 (−35) | 71 |
| Apo-10’-Zeaxanthinal-C10:0 | 546 | + 547>105 (−35) | + 547>119 (−30) | 87 |
| Apo-10’-Zeaxanthinal-C12:0 | 574 | + 575>105 (−35) | + 575>119 (−30) | 75 |
| Apo-10’-Zeaxanthinal-C14:0 | 602 | + 603>105 (−40) | + 603>119 (−30) | 77 |
| Apo-8’-Zeaxanthinal-C8:0 | 558 | + 559>105 (−40) | + 559>119 (−40) | 70 |
| Apo-8’-Zeaxanthinal-C10:0 | 586 | + 587>119 (−40) | + 587>105 (−40) | 81 |
| Apo-8’-Zeaxanthinal-C12:0 | 614 | + 615>105 (−40) | + 615>119 (−40) | 79 |
| Compound | Chlorella sorokiana NIVA-CHL 176 | Nanochloropsis gaditana CCMP 526 | Tetraselmis chui SAG 8-6 | Chlamydomonas sp. CCMP 2294 |
|---|---|---|---|---|
| Apo-8’-Zeaxanthinal | - | × | - | × |
| Apo-10’-Zeaxanthinal | × | - | - | × |
| Apo-12’-Zeaxanthinal | × | × | × | × |
| Apo-14’-Zeaxanthinal | × | - | × | × |
| Apo-15’-Zeaxanthinal | - | × | × | × |
| β-Apo-8’-Carotenal | × | - | × | - |
| β-Apo-10’-Carotenal | × | × | - | × |
| β-Apo-12’-Carotenal | × | × | × | × |
| β-Apo-14’-Carotenal | × | - | × | × |
| Apo-10’-Zeaxanthinal -C4:0 | × | × | × | × |
| Apo-10’-Zeaxanthinal -C10:0 | × | × | - | × |
| Apo-10’-Zeaxanthinal -C12:0 | × | - | - | × |
| Apo-10’-Zeaxanthinal -C14:0 | × | - | × | × |
| Apo-8’-Zeaxanthinal-C8:0 | × | × | × | × |
| Apo-8’-Zeaxanthinal-C10:0 | × | × | - | × |
| Apo-8’-Zeaxanthinal-C12:0 | × | × | - | × |
| Apo-8-Luteinal | - | × | × | - |
| Apo-10-Luteinal | × | × | × | × |
| Apo-12-Luteinal | × | × | × | × |
| Apo-14-Luteinal | × | - | × | × |
| Apo-15-Luteina | × | - | - | × |
| Apo-8’-Violaxanthin | × | - | × | × |
| Apo-12’-Violaxanthal | × | × | × | × |
| Apo-14’-Violaxanthal | × | - | × | × |
| Apo-15’-Violaxanthal | - | × | × | × |
| Apo-8’-Fucoxanthinal | - | × | × | × |
| Apo-10’-Fucoxanthinal | - | × | - | - |
| Apo-14’-Fucoxanthinal | × | - | - | - |
| Apo-15’-Fucoxanthinal | × | - | × | × |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zoccali, M.; Giuffrida, D.; Salafia, F.; Socaciu, C.; Skjånes, K.; Dugo, P.; Mondello, L. First Apocarotenoids Profiling of Four Microalgae Strains. Antioxidants 2019, 8, 209. https://doi.org/10.3390/antiox8070209
Zoccali M, Giuffrida D, Salafia F, Socaciu C, Skjånes K, Dugo P, Mondello L. First Apocarotenoids Profiling of Four Microalgae Strains. Antioxidants. 2019; 8(7):209. https://doi.org/10.3390/antiox8070209
Chicago/Turabian StyleZoccali, Mariosimone, Daniele Giuffrida, Fabio Salafia, Carmen Socaciu, Kari Skjånes, Paola Dugo, and Luigi Mondello. 2019. "First Apocarotenoids Profiling of Four Microalgae Strains" Antioxidants 8, no. 7: 209. https://doi.org/10.3390/antiox8070209
APA StyleZoccali, M., Giuffrida, D., Salafia, F., Socaciu, C., Skjånes, K., Dugo, P., & Mondello, L. (2019). First Apocarotenoids Profiling of Four Microalgae Strains. Antioxidants, 8(7), 209. https://doi.org/10.3390/antiox8070209