Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies
Abstract
1. Introduction
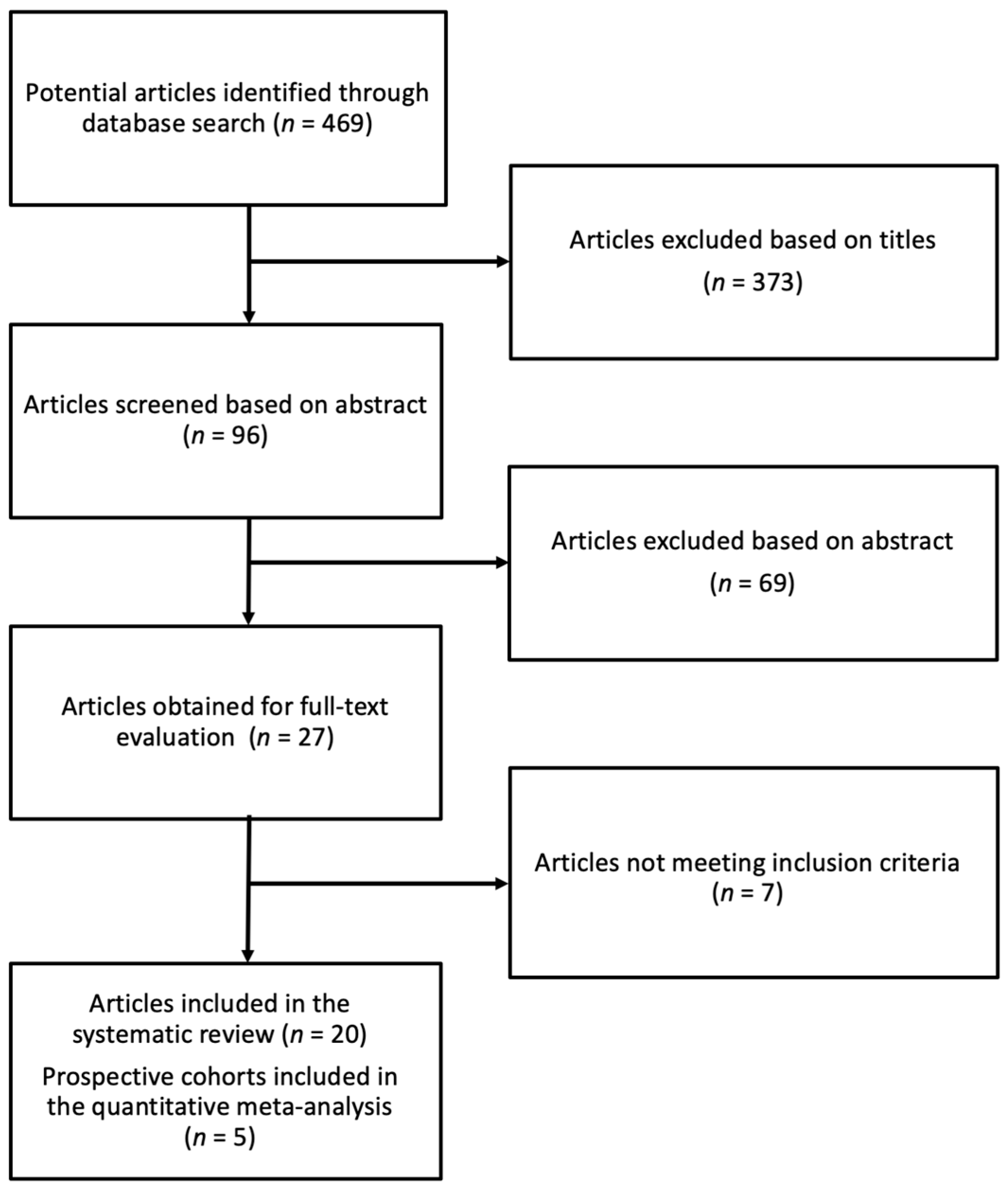
2. Materials and Methods
2.1. Study Selection
2.2. Data Extraction and Study Quality
2.3. Statistical Analysis
3. Results
3.1. Study Characteristics
3.2. Total Polyphenols, Flavonoids, Phenolic Acids, and Blood Pressure/Hypertension
3.3. Phytoestrogens and Blood Pressure/Hypertension
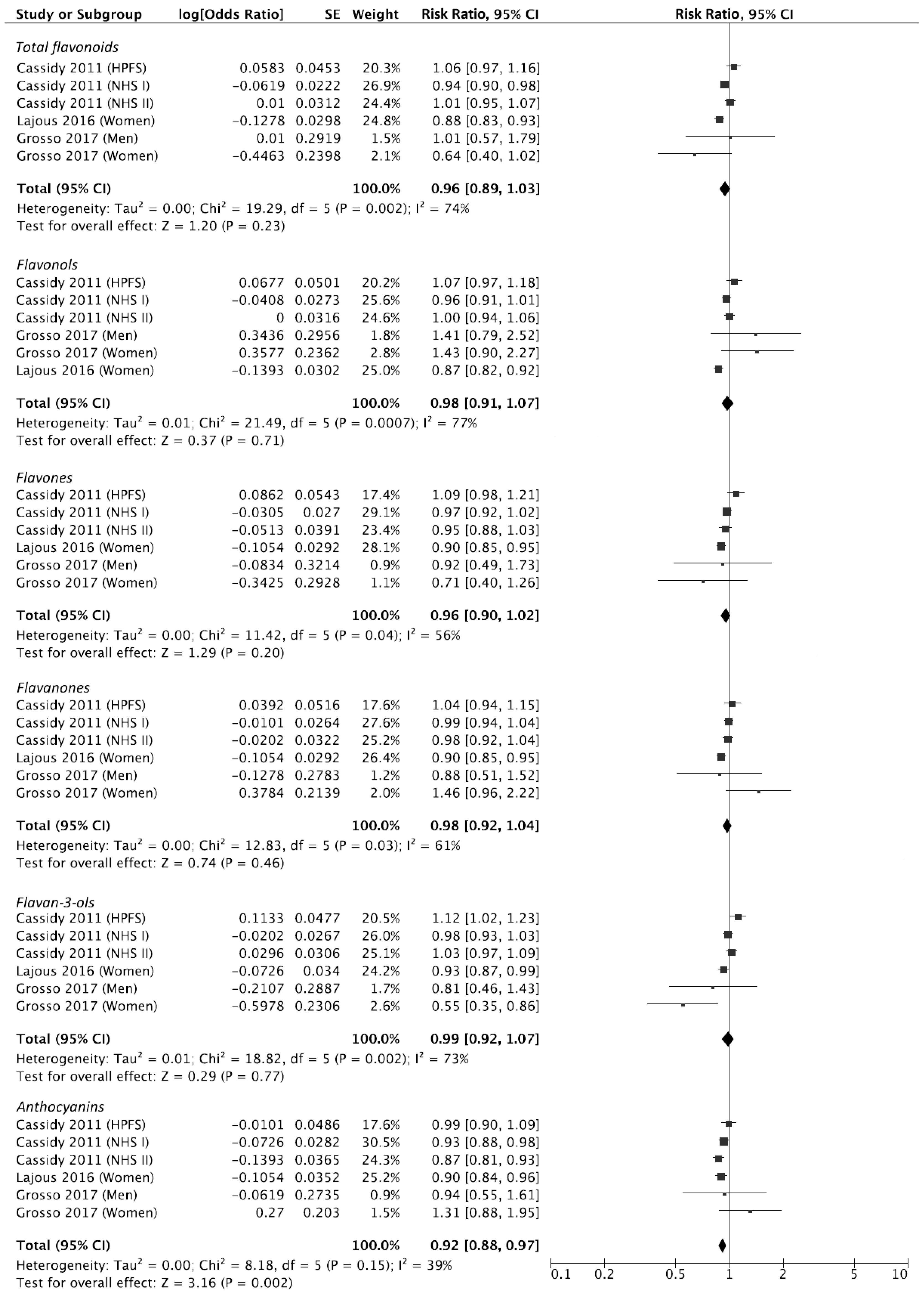
3.4. Quantitative Meta-Analysis for Dietary Flavonoid Intake and Risk of Hypertension
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Del Rio, D.; Costa, L.G.; Lean, M.E.; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis. 2010, 20, 1–6. [Google Scholar] [CrossRef]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; Del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Galvano, F.; Giovannucci, E.L. Dietary flavonoid and lignan intake and mortality in prospective cohort studies: Systematic review and dose-response meta-analysis. Am. J. Epidemiol. 2017, 12, 1304–1316. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Topor-Madry, R.; Szafraniec, K.; Pajak, A. Estimated dietary intake and major food sources of polyphenols in the polish arm of the hapiee study. Nutrition 2014, 30, 1398–1403. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Knaze, V.; Lujan-Barroso, L.; Romieu, I.; Scalbert, A.; Slimani, N.; Hjartaker, A.; Engeset, D.; Skeie, G.; Overvad, K.; et al. Differences in dietary intakes, food sources and determinants of total flavonoids between mediterranean and non-mediterranean countries participating in the european prospective investigation into cancer and nutrition (epic) study. Br. J. Nutr. 2013, 109, 1498–1507. [Google Scholar] [CrossRef]
- Godos, J.; Marventano, S.; Mistretta, A.; Galvano, F.; Grosso, G. Dietary sources of polyphenols in the mediterranean healthy eating, aging and lifestyle (meal) study cohort. Int. J. Food Sci. Nutr. 2017, 68, 750–756. [Google Scholar] [CrossRef]
- Perez-Jimenez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the phenol-explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Medina-Remon, A.; Perez-Jimenez, J.; Martinez-Gonzalez, M.A.; Covas, M.I.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Lapetra, J.; Aros, F.; et al. Dietary intake and major food sources of polyphenols in a spanish population at high cardiovascular risk: The predimed study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 953–959. [Google Scholar] [CrossRef]
- Vitale, M.; Masulli, M.; Rivellese, A.A.; Bonora, E.; Cappellini, F.; Nicolucci, A.; Squatrito, S.; Antenucci, D.; Barrea, A.; Bianchi, C.; et al. Dietary intake and major food sources of polyphenols in people with type 2 diabetes: The tosca.It study. Eur. J. Nutr. 2016. [Google Scholar] [CrossRef]
- Angelino, D.; Godos, J.; Ghelfi, F.; Tieri, M.; Titta, L.; Lafranconi, A.; Marventano, S.; Alonzo, E.; Gambera, A.; Sciacca, S.; et al. Fruit and vegetable consumption and health outcomes: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2019, 1–16. [Google Scholar] [CrossRef]
- Grosso, G.; Bella, F.; Godos, J.; Sciacca, S.; Del Rio, D.; Ray, S.; Galvano, F.; Giovannucci, E.L. Possible role of diet in cancer: Systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nutr. Rev. 2017, 75, 405–419. [Google Scholar] [CrossRef]
- Onvani, S.; Haghighatdoost, F.; Surkan, P.J.; Larijani, B.; Azadbakht, L. Adherence to the healthy eating index and alternative healthy eating index dietary patterns and mortality from all causes, cardiovascular disease and cancer: A meta-analysis of observational studies. J. Hum. Nutr. Diet. 2017, 30, 216–226. [Google Scholar] [CrossRef]
- Godos, J.; Rapisarda, G.; Marventano, S.; Galvano, F.; Mistretta, A.; Grosso, G. Association between polyphenol intake and adherence to the mediterranean diet in sicily, southern italy. NFS J. 2017, 8, 1–7. [Google Scholar] [CrossRef]
- Mocciaro, G.; Ziauddeen, N.; Godos, J.; Marranzano, M.; Chan, M.Y.; Ray, S. Does a mediterranean-type dietary pattern exert a cardio-protective effect outside the mediterranean region? A review of current evidence. Int. J. Food Sci. Nutr. 2017, 5, 524–535. [Google Scholar] [CrossRef]
- Platania, A.; Zappala, G.; Mirabella, M.U.; Gullo, C.; Mellini, G.; Beneventano, G.; Maugeri, G.; Marranzano, M. Association between mediterranean diet adherence and dyslipidaemia in a cohort of adults living in the mediterranean area. Int. J. Food Sci. Nutr. 2017, 5, 608–618. [Google Scholar] [CrossRef]
- Beunza, J.J.; Toledo, E.; Hu, F.B.; Bes-Rastrollo, M.; Serrano-Martinez, M.; Sanchez-Villegas, A.; Martinez, J.A.; Martinez-Gonzalez, M.A. Adherence to the mediterranean diet, long-term weight change, and incident overweight or obesity: The seguimiento universidad de navarra (sun) cohort. Am. J. Clin. Nutr. 2010, 92, 1484–1493. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Garcia-Arellano, A.; Toledo, E.; Salas-Salvado, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schroder, H.; Aros, F.; Gomez-Gracia, E.; et al. A 14-item mediterranean diet assessment tool and obesity indexes among high-risk subjects: The predimed trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef]
- Mendez, M.A.; Popkin, B.M.; Jakszyn, P.; Berenguer, A.; Tormo, M.J.; Sanchez, M.J.; Quiros, J.R.; Pera, G.; Navarro, C.; Martinez, C.; et al. Adherence to a mediterranean diet is associated with reduced 3-year incidence of obesity. J. Nutr. 2006, 136, 2934–2938. [Google Scholar] [CrossRef]
- Mistretta, A.; Marventano, S.; Antoci, M.; Cagnetti, A.; Giogianni, G.; Nolfo, F.; Rametta, S.; Pecora, G.; Marranzano, M. Mediterranean diet adherence and body composition among southern italian adolescents. Obes. Res. Clin. Pract. 2017, 11, 215–226. [Google Scholar] [CrossRef]
- Zappala, G.; Buscemi, S.; Mule, S.; La Verde, M.; D’Urso, M.; Corleo, D.; Marranzano, M. High adherence to mediterranean diet, but not individual foods or nutrients, is associated with lower likelihood of being obese in a mediterranean cohort. Eat. Weight Disord. 2017, 23, 605–614. [Google Scholar] [CrossRef]
- La Verde, M.; Mule, S.; Zappala, G.; Privitera, G.; Maugeri, G.; Pecora, F.; Marranzano, M. Higher adherence to the mediterranean diet is inversely associated with having hypertension: Is low salt intake a mediating factor? Int. J. Food Sci. Nutr. 2018, 69, 235–244. [Google Scholar] [CrossRef]
- Nunez-Cordoba, J.M.; Valencia-Serrano, F.; Toledo, E.; Alonso, A.; Martinez-Gonzalez, M.A. The mediterranean diet and incidence of hypertension: The Seguimiento Universidad de Navarra (SUN) study. Am. J. Epidemiol. 2009, 169, 339–346. [Google Scholar] [CrossRef]
- Bassino, E.; Antoniotti, S.; Gasparri, F.; Munaron, L. Effects of flavonoid derivatives on human microvascular endothelial cells. Nat. Pro.d Res. 2016, 24. [Google Scholar] [CrossRef]
- Fisher, N.D.; Hurwitz, S.; Hollenberg, N.K. Habitual flavonoid intake and endothelial function in healthy humans. J. Am. Coll. Nutr. 2012, 31, 275–279. [Google Scholar] [CrossRef]
- Hemanth Kumar, B.; Dinesh Kumar, B.; Diwan, P.V. Hesperidin, a citrus flavonoid, protects against L-methionine-induced hyperhomocysteinemia by abrogation of oxidative stress, endothelial dysfunction and neurotoxicity in wistar rats. Pharm. Biol. 2017, 55, 146–155. [Google Scholar] [CrossRef]
- Qian, Y.; Babu, P.V.A.; Symons, J.D.; Jalili, T. Metabolites of flavonoid compounds preserve indices of endothelial cell nitric oxide bioavailability under glucotoxic conditions. Nutr. Diabetes 2017, 7, e286. [Google Scholar] [CrossRef]
- Medina-Remon, A.; Zamora-Ros, R.; Rotches-Ribalta, M.; Andres-Lacueva, C.; Martinez-Gonzalez, M.A.; Covas, M.I.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; et al. Total polyphenol excretion and blood pressure in subjects at high cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 323–331. [Google Scholar] [CrossRef]
- Medina-Remon, A.; Tresserra-Rimbau, A.; Pons, A.; Tur, J.A.; Martorell, M.; Ros, E.; Buil-Cosiales, P.; Sacanella, E.; Covas, M.I.; Corella, D.; et al. Effects of total dietary polyphenols on plasma nitric oxide and blood pressure in a high cardiovascular risk cohort. The predimed randomized trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 60–67. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 1999. [Google Scholar]
- Cassidy, A.; O’Reilly, E.J.; Kay, C.; Sampson, L.; Franz, M.; Forman, J.P.; Curhan, G.; Rimm, E.B. Habitual intake of flavonoid subclasses and incident hypertension in adults. Am. J. Clin. Nutr. 2011, 93, 338–347. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Micek, A.; Kozela, M.; Stefler, D.; Bobak, M.; Pajak, A. Dietary polyphenol intake and risk of hypertension in the polish arm of the hapiee study. Eur. J. Nutr. 2017, 4, 1535–1544. [Google Scholar] [CrossRef]
- Lajous, M.; Rossignol, E.; Fagherazzi, G.; Perquier, F.; Scalbert, A.; Clavel-Chapelon, F.; Boutron-Ruault, M.C. Flavonoid intake and incident hypertension in women. Am. J. Clin. Nutr. 2016, 103, 1091–1098. [Google Scholar] [CrossRef]
- Jennings, A.; Welch, A.A.; Fairweather-Tait, S.J.; Kay, C.; Minihane, A.M.; Chowienczyk, P.; Jiang, B.; Cecelja, M.; Spector, T.; Macgregor, A.; et al. Higher anthocyanin intake is associated with lower arterial stiffness and central blood pressure in women. Am. J. Clin. Nutr. 2012, 96, 781–788. [Google Scholar] [CrossRef]
- Wisnuwardani, R.W.; De Henauw, S.; Forsner, M.; Gottrand, F.; Huybrechts, I.; Knaze, V.; Kersting, M.; Donne, C.L.; Manios, Y.; Marcos, A.; et al. Polyphenol intake and metabolic syndrome risk in european adolescents: The HELENA study. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Micek, A.; Stefler, D.; Bobak, M.; Pajak, A. Dietary polyphenols are inversely associated with metabolic syndrome in Polish adults of the HAPIEE study. Eur. J. Nutr. 2016, 4, 1409–1420. [Google Scholar] [CrossRef]
- Sohrab, G.; Hosseinpour-Niazi, S.; Hejazi, J.; Yuzbashian, E.; Mirmiran, P.; Azizi, F. Dietary polyphenols and metabolic syndrome among iranian adults. Int. J. Food Sci. Nutr. 2013, 64, 661–667. [Google Scholar] [CrossRef]
- Miranda, A.M.; Steluti, J.; Fisberg, R.M.; Marchioni, D.M. Association between polyphenol intake and hypertension in adults and older adults: A population-based study in Brazil. PLoS ONE 2016, 11, e0165791. [Google Scholar] [CrossRef]
- Miranda, A.M.; Steluti, J.; Fisberg, R.M.; Marchioni, D.M. Association between coffee consumption and its polyphenols with cardiovascular risk factors: A population-based study. Nutrients 2017, 9, 276. [Google Scholar] [CrossRef]
- Godos, J.; Sinatra, D.; Blanco, I.; Mule, S.; La Verde, M.; Marranzano, M. Association between dietary phenolic acids and hypertension in a mediterranean cohort. Nutrients 2017, 9. [Google Scholar]
- Yang, Y.J.; Kim, Y.J.; Yang, Y.K.; Kim, J.Y.; Kwon, O. Dietary flavan-3-ols intake and metabolic syndrome risk in Korean adults. Nutr. Res. Pract. 2012, 6, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.I.; Steffen, L.M.; Swett, K.; Smith, C.; Burke, L.; Zhou, X.; Shikany, J.M.; Rodriguez, C.J. Dietary total isoflavone intake is associated with lower systolic blood pressure: The Coronary Artery Risk Development in Young Adults (CARDIA) study. J. Clin. Hypertens. 2016, 18, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Bergante, S.; Satriano, A.; Pluchinotta, F.R.; Marranzano, M. Dietary phytoestrogen intake is inversely associated with hypertension in a cohort of adults living in the mediterranean area. Molecules 2018, 23, 368. [Google Scholar] [CrossRef] [PubMed]
- Creus-Cuadros, A.; Tresserra-Rimbau, A.; Quifer-Rada, P.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Fito, M.; Estruch, R.; Gomez-Gracia, E.; Lapetra, J.; et al. Associations between both lignan and yogurt consumption and cardiovascular risk parameters in an elderly population: Observations from a cross-sectional approach in the PREDIMED study. J. Acad. Nutr. Diet. 2017, 117, 609–622 e601. [Google Scholar] [CrossRef]
- Nozue, M.; Shimazu, T.; Sasazuki, S.; Charvat, H.; Mori, N.; Mutoh, M.; Sawada, N.; Iwasaki, M.; Yamaji, T.; Inoue, M.; et al. Fermented soy product intake is inversely associated with the development of high blood pressure: The Japan public health center-based prospective study. J. Nutr. 2017, 147, 1749–1756. [Google Scholar] [CrossRef]
- Woo, H.W.; Kim, M.K.; Lee, Y.H.; Shin, D.H.; Shin, M.H.; Choi, B.Y. Habitual consumption of soy protein and isoflavones and risk of metabolic syndrome in adults ≥ 40 years old: A prospective analysis of the korean multi-rural communities cohort study (MRCOHORT). Eur. J. Nutr. 2018. [Google Scholar] [CrossRef]
- De Kleijn, M.J.; van der Schouw, Y.T.; Wilson, P.W.; Grobbee, D.E.; Jacques, P.F. Dietary intake of phytoestrogens is associated with a favorable metabolic cardiovascular risk profile in postmenopausal U.S.Women: The Framingham study. J. Nutr. 2002, 132, 276–282. [Google Scholar] [CrossRef]
- Kreijkamp-Kaspers, S.; Kok, L.; Bots, M.L.; Grobbee, D.E.; van der Schouw, Y.T. Dietary phytoestrogens and vascular function in postmenopausal women: A cross-sectional study. J. Hypertens. 2004, 22, 1381–1388. [Google Scholar] [CrossRef]
- Witkowska, A.M.; Waskiewicz, A.; Zujko, M.E.; Szczesniewska, D.; Stepaniak, U.; Pajak, A.; Drygas, W. Are total and individual dietary lignans related to cardiovascular disease and its risk factors in postmenopausal women? A nationwide study. Nutrients 2018, 10, 865. [Google Scholar] [CrossRef]
- Jayalath, V.H.; de Souza, R.J.; Sievenpiper, J.L.; Ha, V.; Chiavaroli, L.; Mirrahimi, A.; Di Buono, M.; Bernstein, A.M.; Leiter, L.A.; Kris-Etherton, P.M.; et al. Effect of dietary pulses on blood pressure: A systematic review and meta-analysis of controlled feeding trials. Am. J. hypertens. 2014, 27, 56–64. [Google Scholar] [CrossRef]
- Noad, R.L.; Rooney, C.; McCall, D.; Young, I.S.; McCance, D.; McKinley, M.C.; Woodside, J.V.; McKeown, P.P. Beneficial effect of a polyphenol-rich diet on cardiovascular risk: A randomised control trial. Heart 2016, 102, 1371–1379. [Google Scholar] [CrossRef]
- Sahebkar, A.; Ferri, C.; Giorgini, P.; Bo, S.; Nachtigal, P.; Grassi, D. Effects of pomegranate juice on blood pressure: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2017, 115, 149–161. [Google Scholar] [CrossRef]
- Huang, H.; Chen, G.; Liao, D.; Zhu, Y.; Xue, X. Effects of berries consumption on cardiovascular risk factors: A meta-analysis with trial sequential analysis of randomized controlled trials. Sci. Rep. 2016, 6, 23625. [Google Scholar] [CrossRef]
- Kent, K.; Charlton, K.E.; Jenner, A.; Roodenrys, S. Acute reduction in blood pressure following consumption of anthocyanin-rich cherry juice may be dose-interval dependant: A pilot cross-over study. Int. J. Food. Sci. Nutr. 2016, 67, 47–52. [Google Scholar] [CrossRef]
- Zhu, Y.; Sun, J.; Lu, W.; Wang, X.; Wang, X.; Han, Z.; Qiu, C. Effects of blueberry supplementation on blood pressure: A systematic review and meta-analysis of randomized clinical trials. J. Hum. Hypertens. 2017, 31, 165–171. [Google Scholar] [CrossRef]
- Kou, T.; Wang, Q.; Cai, J.; Song, J.; Du, B.; Zhao, K.; Ma, Y.; Geng, B.; Zhang, Y.; Han, X.; et al. Effect of soybean protein on blood pressure in postmenopausal women: A meta-analysis of randomized controlled trials. Food Funct. 2017, 8, 2663–2671. [Google Scholar] [CrossRef]
- Khosravi-Boroujeni, H.; Nikbakht, E.; Natanelov, E.; Khalesi, S. Can sesame consumption improve blood pressure? A systematic review and meta-analysis of controlled trials. J. Sci. Food Agric. 2017, 97, 3087–3094. [Google Scholar] [CrossRef]
- Ursoniu, S.; Sahebkar, A.; Andrica, F.; Serban, C.; Banach, M.; Lipid and Blood Pressure Meta-analysis Collaboration. Effects of flaxseed supplements on blood pressure: A systematic review and meta-analysis of controlled clinical trial. Clin. Nutr. 2016, 35, 615–625. [Google Scholar] [CrossRef]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Bes-Rastrollo, M.; Galvano, F.; Martinez-Gonzalez, M.A. Long-term coffee consumption is associated with decreased incidence of new-onset hypertension: A dose-response meta-analysis. Nutrients 2017, 9, 890. [Google Scholar] [CrossRef]
- Liu, G.; Mi, X.N.; Zheng, X.X.; Xu, Y.L.; Lu, J.; Huang, X.H. Effects of tea intake on blood pressure: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2014, 112, 1043–1054. [Google Scholar] [CrossRef]
- Serban, C.; Sahebkar, A.; Ursoniu, S.; Andrica, F.; Banach, M. Effect of sour tea (Hibiscus sabdariffa L.) on arterial hypertension: A systematic review and meta-analysis of randomized controlled trials. J. Hypertens. 2015, 33, 1119–1127. [Google Scholar] [CrossRef]
- Liu, J.; Tang, W.; Sang, L.; Dai, X.; Wei, D.; Luo, Y.; Zhang, J. Milk, yogurt, and lactose intake and ovarian cancer risk: A meta-analysis. Nutr. Cancer 2015, 67, 68–72. [Google Scholar] [CrossRef]
- Yarmolinsky, J.; Gon, G.; Edwards, P. Effect of tea on blood pressure for secondary prevention of cardiovascular disease: A systematic review and meta-analysis of randomized controlled trials. Nutr. Rev. 2015, 73, 236–246. [Google Scholar] [CrossRef]
- Zhu, Y.; Bo, Y.; Wang, X.; Lu, W.; Wang, X.; Han, Z.; Qiu, C. The effect of anthocyanins on blood pressure: A prisma-compliant meta-analysis of randomized clinical trials. Medicine 2016, 95, e3380. [Google Scholar] [CrossRef]
- Taku, K.; Lin, N.; Cai, D.; Hu, J.; Zhao, X.; Zhang, Y.; Wang, P.; Melby, M.K.; Hooper, L.; Kurzer, M.S.; et al. Effects of soy isoflavone extract supplements on blood pressure in adult humans: Systematic review and meta-analysis of randomized placebo-controlled trials. J. Hypertens. 2010, 28, 1971–1982. [Google Scholar] [CrossRef]
- Liu, X.X.; Li, S.H.; Chen, J.Z.; Sun, K.; Wang, X.J.; Wang, X.G.; Hui, R.T. Effect of soy isoflavones on blood pressure: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 463–470. [Google Scholar] [CrossRef]
- Khalesi, S.; Irwin, C.; Schubert, M. Flaxseed consumption may reduce blood pressure: A systematic review and meta-analysis of controlled trials. J. Nutr. 2015, 145, 758–765. [Google Scholar] [CrossRef]
- Kay, C.D.; Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cassidy, A. Relative impact of flavonoid composition, dose and structure on vascular function: A systematic review of randomised controlled trials of flavonoid-rich food products. Mol. Nutr. Food Res. 2012, 56, 1605–1616. [Google Scholar] [CrossRef]
- Mena, P.; Dominguez-Perles, R.; Girones-Vilaplana, A.; Baenas, N.; Garcia-Viguera, C.; Villano, D. Flavan-3-ols, anthocyanins, and inflammation. IUBMB Life 2014, 66, 745–758. [Google Scholar] [CrossRef]
- Ahmad, A.; Singhal, U.; Hossain, M.M.; Islam, N.; Rizvi, I. The role of the endogenous antioxidant enzymes and malondialdehyde in essential hypertension. J. Clin. Diagn. Res. 2013, 7, 987–990. [Google Scholar] [CrossRef]
- Brunelli, E.; La Russa, D.; Pellegrino, D. Impaired oxidative status is strongly associated with cardiovascular risk factors. Oxid. Med. Cell. Longev. 2017, 2017, 6480145. [Google Scholar] [CrossRef]
- La Russa, D.; Brunelli, E.; Pellegrino, D. Oxidative imbalance and kidney damage in spontaneously hypertensive rats: Activation of extrinsic apoptotic pathways. Clin. Sci. (Lond.) 2017, 131, 1419–1428. [Google Scholar] [CrossRef]
- Ward, N.C.; Hodgson, J.M.; Puddey, I.B.; Mori, T.A.; Beilin, L.J.; Croft, K.D. Oxidative stress in human hypertension: Association with antihypertensive treatment, gender, nutrition, and lifestyle. Free Radic. Biol. Med. 2004, 36, 226–232. [Google Scholar] [CrossRef]
- Hugel, H.M.; Jackson, N.; May, B.; Zhang, A.L.; Xue, C.C. Polyphenol protection and treatment of hypertension. Phytomedicine 2016, 23, 220–231. [Google Scholar] [CrossRef]
- Clark, J.L.; Zahradka, P.; Taylor, C.G. Efficacy of flavonoids in the management of high blood pressure. Nutr. Rev. 2015, 73, 799–822. [Google Scholar] [CrossRef]
- Ramdath, D.D.; Padhi, E.M.; Sarfaraz, S.; Renwick, S.; Duncan, A.M. Beyond the cholesterol-lowering effect of soy protein: A review of the effects of dietary soy and its constituents on risk factors for cardiovascular disease. Nutrients 2017, 9, 324. [Google Scholar] [CrossRef]
- Rani, N.; Bharti, S.; Bhatia, J.; Nag, T.C.; Ray, R.; Arya, D.S. Chrysin, a ppar-gamma agonist improves myocardial injury in diabetic rats through inhibiting age-rage mediated oxidative stress and inflammation. Chem. Biol. Interact. 2016, 250, 59–67. [Google Scholar] [CrossRef]
- Yuan, X.; Niu, H.T.; Wang, P.L.; Lu, J.; Zhao, H.; Liu, S.H.; Zheng, Q.S.; Li, C.G. Cardioprotective effect of licochalcone d against myocardial ischemia/reperfusion injury in langendorff-perfused rat hearts. PLoS ONE 2015, 10, e0128375. [Google Scholar] [CrossRef]
- De Pascual-Teresa, S.; Moreno, D.A.; Garcia-Viguera, C. Flavanols and anthocyanins in cardiovascular health: A review of current evidence. Int. J. Mol. Sci. 2010, 11, 1679–1703. [Google Scholar] [CrossRef]
- Serraino, I.; Dugo, L.; Dugo, P.; Mondello, L.; Mazzon, E.; Dugo, G.; Caputi, A.P.; Cuzzocrea, S. Protective effects of cyanidin-3-O-glucoside from blackberry extract against peroxynitrite-induced endothelial dysfunction and vascular failure. Life Sci. 2003, 73, 1097–1114. [Google Scholar] [CrossRef]
- Pergola, C.; Rossi, A.; Dugo, P.; Cuzzocrea, S.; Sautebin, L. Inhibition of nitric oxide biosynthesis by anthocyanin fraction of blackberry extract. Nitric Oxide 2006, 15, 30–39. [Google Scholar] [CrossRef]
- Xun, Y.H.; Guo, J.C.; Lou, G.Q.; Jiang, Y.M.; Zhuang, Z.J.; Zhu, M.F.; Luo, Y.; Ma, X.J.; Liu, J.; Bian, D.X.; et al. Non-alcoholic fatty liver disease (NAFLD) fibrosis score predicts 6.6-year overall mortality of chinese patients with nafld. Clin. Exp. Pharmacol. Physiol. 2014, 41, 643–649. [Google Scholar]
- Xu, J.W.; Ikeda, K.; Yamori, Y. Upregulation of endothelial nitric oxide synthase by cyanidin-3-glucoside, a typical anthocyanin pigment. Hypertension 2004, 44, 217–222. [Google Scholar] [CrossRef]
- Martin, S.; Giannone, G.; Andriantsitohaina, R.; Martinez, M.C. Delphinidin, an active compound of red wine, inhibits endothelial cell apoptosis via nitric oxide pathway and regulation of calcium homeostasis. Br. J. Pharmacol. 2003, 139, 1095–1102. [Google Scholar] [CrossRef]
- Kahle, K.; Kraus, M.; Scheppach, W.; Ackermann, M.; Ridder, F.; Richling, E. Studies on apple and blueberry fruit constituents: Do the polyphenols reach the colon after ingestion? Mol. Nutr. Food Res. 2006, 50, 418–423. [Google Scholar] [CrossRef]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Galvano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutr. 2007, 137, 2043–2048. [Google Scholar] [CrossRef]
- Riboli, E.; Slimani, N.; Kaaks, R. Identifiability of food components for cancer chemoprevention. IARC Sci. Publ. 1996, 23–31. [Google Scholar]
| Author, Year | Cohort Name, Country | Study Design | Follow-Up (Years of Study) | Total Population (Analyzed Sample) | Cases (Hypertension/High Blood Pressure) | Sex | Age | Dietary Assessment | Polyphenol Estimation | Polyphenol Classes | Hypertension Assessment | Adjustments |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitale, 2006 [11] | TOSCA.IT, Italy | Cross-sectional | NA | 2573 | NR | MF | 50–75 y (range) | 248-item FFQ | USDA and Phenol-Explorer database | Flavonoids, phenolic acids | Blood pressure measured during clinical visit | Gender, age, BMI, waist and hip circumferences, smoke, alcohol intake, dietary lipids, SFA, MUFA, PUFA, added sugars, and fiber. |
| Cassidy, 2011 [32] | NHS I, USA | Prospective | 14 y (1990–2004) | 46,672 | 17,616 | F | 30–55 y (range) | FFQ | USDA and EuroFIR database, published literature | Flavonoids (and subclasses) | Self-reported | Age and smoking, BMI, PA, alcohol consumption, family history of hypertension, aspirin use, multivitamin use, and intakes of sodium, magnesium, potassium, fiber, whole grain, folate, and caffeine. |
| Cassidy, 2011 [32] | NHS II, USA | Prospective | 14 y (1991–2005) | 87,242 | 11,402 | F | 25–42 y (range) | FFQ | USDA and EuroFIR database, published literature | Flavonoids (and subclasses) | Self-reported | Age and smoking, BMI, PA, alcohol consumption, family history of hypertension, aspirin use, multivitamin use, and intakes of sodium, magnesium, potassium, fiber, whole grain, folate, and caffeine. |
| Cassidy, 2011 [32] | HPFS, USA | Prospective | 14 y (1990–2004) | 23,043 | 5,629 | M | 40–75 y (range) | FFQ | USDA and EuroFIR database, published literature | Flavonoids (and subclasses) | Self-reported | Age and smoking, BMI, PA, alcohol consumption, family history of hypertension, aspirin use, multivitamin use, and intakes of sodium, magnesium, potassium, fiber, whole grain, folate, and caffeine. |
| Grosso, 2017 [33] | HAPIEE, Poland | Prospective | 4 y (2002–2005) | 2725 | 1735 | MF | 45–69 y (range) | 148-item FFQ | Phenol-Explorer database | Total polyphenols, flavonoids (and subclasses), phenolic acids (and subclasses), stilbenes, lignans, others | Blood pressure measured during clinical visit | Age, total energy intake, BMI, PA, educational status, smoking status, alcohol consumption sodium, potassium and fiber intake, menopausal status (women only). |
| Lajous, 2016 [34] | E3N, France | Prospective | 14 y (1993–2008) | 40,574 | 9350 | F | 45–58 y (range) | 208-item FFQ | Phenol-Explorer database | Flavonoids (and subclasses) | Self-reported | Education, family history of hypertension, diabetes, hypercholesterolemia, BMI, smoking, PA metabolic equivalents per week, hormone therapy, alcohol, processed meat, caffeine, magnesium, potassium, and omega-3. |
| Jennings, 2012 [35] | TwinsUK, UK | Cross-sectional | NA | 1898 | NR | F | 18–75 y (range) | 131-item FFQ | USDA and Phenol-Explorer database | Flavonoids (and subclasses) | Blood pressure measured during clinical visit | Age, current smoking, physical activity, BMI, use of hormone replacement therapy, use of blood pressure or statin medication, use of vitamin supplements, use of oral contraceptives, menopausal status, family history of heart disease or hypertension, and intakes of energy, alcohol, SFAs, MUFAs, PUFAs, fiber, and sodium. |
| Wisnuwardani, 2019 [36] | HELENA, multicenter Europe | Cross-sectional | NA | 657 | NR | MF | 12.5–17.5 y (range) | 24 h dietary recalls | Phenol-Explorer database | Total polyphenols | Blood pressure measured during clinical visit | Age, sex, European region, education of mother, education of father, puberty status, BMI, monosaccharides and disaccharides, polysaccharides, fibre, monounsaturated fatty acids, saturated fatty acids, cholesterol, protein, vitamin C, and energy intake. |
| Grosso, 2017 [37] | HAPIEE, Poland | Cross-sectional | NA | 8821 | 5.341 | MF | 45–69 y (range) | 148-item FFQ | Phenol-Explorer database | Total polyphenols, flavonoids, phenolic acids, stilbenes, lignans, others | Blood pressure measured during clinical visit | Age, gender, education, physical activity, smoking status, alcohol drinking, BMI, and total energy intake. |
| Sohrab, 2013 [38] | TLGS, Iran | Cross-sectional | NA | 2618 | NR | MF | 19–84 y (range) | 168-item FFQ | USDA and Phenol-Explorer database | Total polyphenols, flavonoids, phenolic acids, stilbenes, lignans | Blood pressure measured during clinical visit | Age, gender, physical activity, smoking status and educational levels, study center, and total energy intake and BMI. |
| Miranda, 2016 [39] | ISA-Capital Study, Brazil | Cross-sectional | NA | 550 | 267 | MF | ≥12 y | 24 h dietary recalls | Phenol-Explorer database | Total polyphenols, flavonoids, phenolic acids, stilbenes, lignans, others (and subclasses) | Blood pressure measured during home visit | Age, sex, race, educational level, BMI, smoking, physical activity, intake of sodium, fiber, saturated fat, alcohol, total energy intake, misreporting, and other polyphenol components. |
| Miranda, 2017 [40] | ISA-Capital Study, Brazil | Cross-sectional | NA | 557 | NR | MF | 45.1 y (mean) | 24 h dietary recalls | Phenol-Explorer database | Coffee polyphenols (phenolic acids and others) | Blood pressure measured during home visit | Age, sex, race, BMI, smoking, alcohol, physical activity, household per capita income, intake of caffeine, added sugars, total energy intake, and saturated fat, other polyphenol intake. |
| Godos, 2017 [41] | MEAL, Italy | Cross-sectional | NA | 1936 | 976 | MF | ≥18 y | 110-item FFQ | Phenol-Explorer database | Phenolic acids (and subclasses and individual polyphenols) | Blood pressure measured during clinical visit | Age, energy intake, BMI, smoking status, alcohol consumption, physical activity level, educational level, occupational level, menopausal status, fiber, sodium, potassium, magnesium, and calcium intake, adherence to the Mediterranean diet. |
| Yang, 2012 [42] | KNHANES, Korea | Cross-sectional | NA | 4745 | NR | MF | 20–69 y (range) | 24 h dietary recalls | USDA database, Japan Functional Food Factor Database, Rural Development Administration Food Functional Composition Table | Flavanols | Blood pressure measured during clinical visit | Age, BMI, education, current drinking, vitamin/mineral supplement use, functional food use, intakes of total energy, fat, fiber, and carbohydrate. |
| Richardson, 2016 [43] | CARDIA, USA | Cross-sectional | NA | 3142 | 843 | MF | 18–30 y (range) | 100-item FFQ | USDA database | Isoflavones | Blood pressure measured during clinical visit | Age, sex, BMI, smoking, alcohol intake, physical activity, total energy, and sodium intake. |
| Godos, 2018 [44] | MEAL, Italy | Cross-sectional | NA | 1936 | 976 | MF | ≥18 y | 110-item FFQ | Phenol-Explorer database | Total phytoestrogens, isoflavones (and individual polyphenols), lignans (and individual polyphenols) | Blood pressure measured during clinical visit | Age, energy intake, smoking status, alcohol consumption, physical activity level, educational level, occupational level, sodium and potassium intake. |
| Creus-Cuadros, 2017 [45] | PREDIMED, Spain | Cross-sectional | NA | 7169 | NR | MF | ~67 y (mean) | 137-item FFQ | Phenol-Explorer database | Lignans | Blood pressure measured during clinical visit | Recruitment center, sex, age, smoking, soft drinks, carbohydrates, saturated fatty acids, monounsaturated fatty acids, polyunsaturated fatty acids, n3 fatty acids, and dairies. |
| Nozue, 2017 [46] | JPHC II, Japan | Prospective | 5 y (1993–NR) | 4165 | 1335 | MF | 40–69 y (range) | 52-item FFQ | Published literature | Isoflavones | Blood pressure measured during clinical visit | Blood pressure at baseline, age, BMI, alcohol consumption, smoking status, physical activity, energy, energy-adjusted salt, saturated fats, green and yellow vegetables, pickled vegetables, fruits and fish, public health status, and menopausal status. |
| Woo, 2018 [47] | MRCohort, South Korea | Prospective | 3.6 y (median, 2005–2013) | 5509 | NR | MF | ≥40 y | 106-item FFQ | USDA database | Isoflavones | Blood pressure measured during clinical visit | Age, calcium intake, fiber intake, egg consumption in both men and women, soda drink consumption for isoflavones analysis in men and current smoking, higher education, regular exercise, and total energy intake for soy protein/isoflavones analyses in women. |
| de Kleijn, 2002 [48] | Framingham Offspring Study, USA | Cross-sectional | NA | 939 | NR | F | 59 ± 7.5 y (mean) | 130-item FFQ | Published literature | Isoflavones, lignans | Blood pressure measured during clinical visit | Age, BMI, use of hormone replacement therapy, smoking, dietary fiber intake, and potassium intake. |
| Kreijkamp-Kaspers, 2004 [49] | Prospect-EPIC, The Netherlands | Cross-sectional | NA | 301 | 113 | F | 60–75 y (range) | FFQ | Published literature | Isoflavones, lignans | Blood pressure measured during clinical visit | Age, cholesterol lowering medication, use of antihypertensives, BMI, waist/hip ratio, smoking, physical activity, total energy intake, alcohol intake, saturated fat, mono-unsaturated fat, polyunsaturated fat, time postmenopausal, fiber intake, fruit intake, vegetable intake, and vitamin C intake. |
| Witkowska, 2018 [50] | WOBASZ and WOBASZ II, Poland | Cross-sectional | NA | 2599 | 1635 | F | ≥20 y | 24 h dietary recalls | Published literature | Lignans (and individual polyphenols) | Blood pressure measured during clinical visit | Age, smoking, BMI, alcohol intake, education, leisure-time physical activity, glucose level, cholesterol level, menopause hormone therapy, energy intake. |
| Subgroup/Additional Analysis | No. of Cohorts | RR (95% CI) | I2 | Pheterogeneity |
|---|---|---|---|---|
| Total | 5 | 0.96 (0.89, 1.03) | 74% | 0.002 |
| Sex | ||||
| Male | 2 | 1.06 (0.97, 1.16) | 0% | 0.87 |
| Female | 4 | 0.93 (0.87, 1.00) | 76% | 0.005 |
| Geographical area | ||||
| Europe | 2 | 0.88 (0.83, 0.93) | 0% | 0.37 |
| North America | 3 | 0.99 (0.93, 1.07) | 73% | 0.02 |
| Number of cases | ||||
| >10,000 | 2 | 0.97 (0.91, 1.04) | 72% | 0.06 |
| <10,000 | 3 | 0.93 (0.79, 1.10) | 79% | 0.003 |
| Follow-up | ||||
| >10y | 4 | 0.97 (0.90, 1.04) | 82% | 0.0009 |
| <10y | 1 | 0.78 (0.50, 1.21) | 31% | 0.23 |
| Adjusted for smoking | ||||
| No | 0 | NA | NA | NA |
| Yes | 5 | 0.96 (0.89, 1.03) | 74% | 0.002 |
| Adjusted for potassium/sodium | ||||
| No | 0 | NA | NA | NA |
| Yes | 5 | 0.96 (0.89, 1.03) | 74% | 0.002 |
| Adjusted for physical activity | ||||
| No | 0 | NA | NA | NA |
| Yes | 5 | 0.96 (0.89, 1.03) | 74% | 0.002 |
| Adjusted for BMI | ||||
| No | 0 | NA | NA | NA |
| Yes | 5 | 0.96 (0.89, 1.03) | 74% | 0.002 |
| Adjusted for education | ||||
| No | 3 | 0.99 (0.93, 1.07) | 73% | 0.02 |
| Yes | 2 | 0.88 (0.83, 0.93) | 0% | 0.37 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godos, J.; Vitale, M.; Micek, A.; Ray, S.; Martini, D.; Del Rio, D.; Riccardi, G.; Galvano, F.; Grosso, G. Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies. Antioxidants 2019, 8, 152. https://doi.org/10.3390/antiox8060152
Godos J, Vitale M, Micek A, Ray S, Martini D, Del Rio D, Riccardi G, Galvano F, Grosso G. Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies. Antioxidants. 2019; 8(6):152. https://doi.org/10.3390/antiox8060152
Chicago/Turabian StyleGodos, Justyna, Marilena Vitale, Agnieszka Micek, Sumantra Ray, Daniela Martini, Daniele Del Rio, Gabriele Riccardi, Fabio Galvano, and Giuseppe Grosso. 2019. "Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies" Antioxidants 8, no. 6: 152. https://doi.org/10.3390/antiox8060152
APA StyleGodos, J., Vitale, M., Micek, A., Ray, S., Martini, D., Del Rio, D., Riccardi, G., Galvano, F., & Grosso, G. (2019). Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies. Antioxidants, 8(6), 152. https://doi.org/10.3390/antiox8060152








