Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Preparation of the Extracts
2.2. Determination of Extracts’ Polyphenolic Content Using HPLC and Ultra Performance Liquid Chromatography–Tandem Mass Spectrometer (UPLC-MS-MS) Analysis
2.3. Analytical Method Validation
2.4. Assessment of the Total Polyphenolic Content of the Extracts
2.5. Total Flavonoid Content of the Extracts
2.6. Free Radical Scavenging Activity
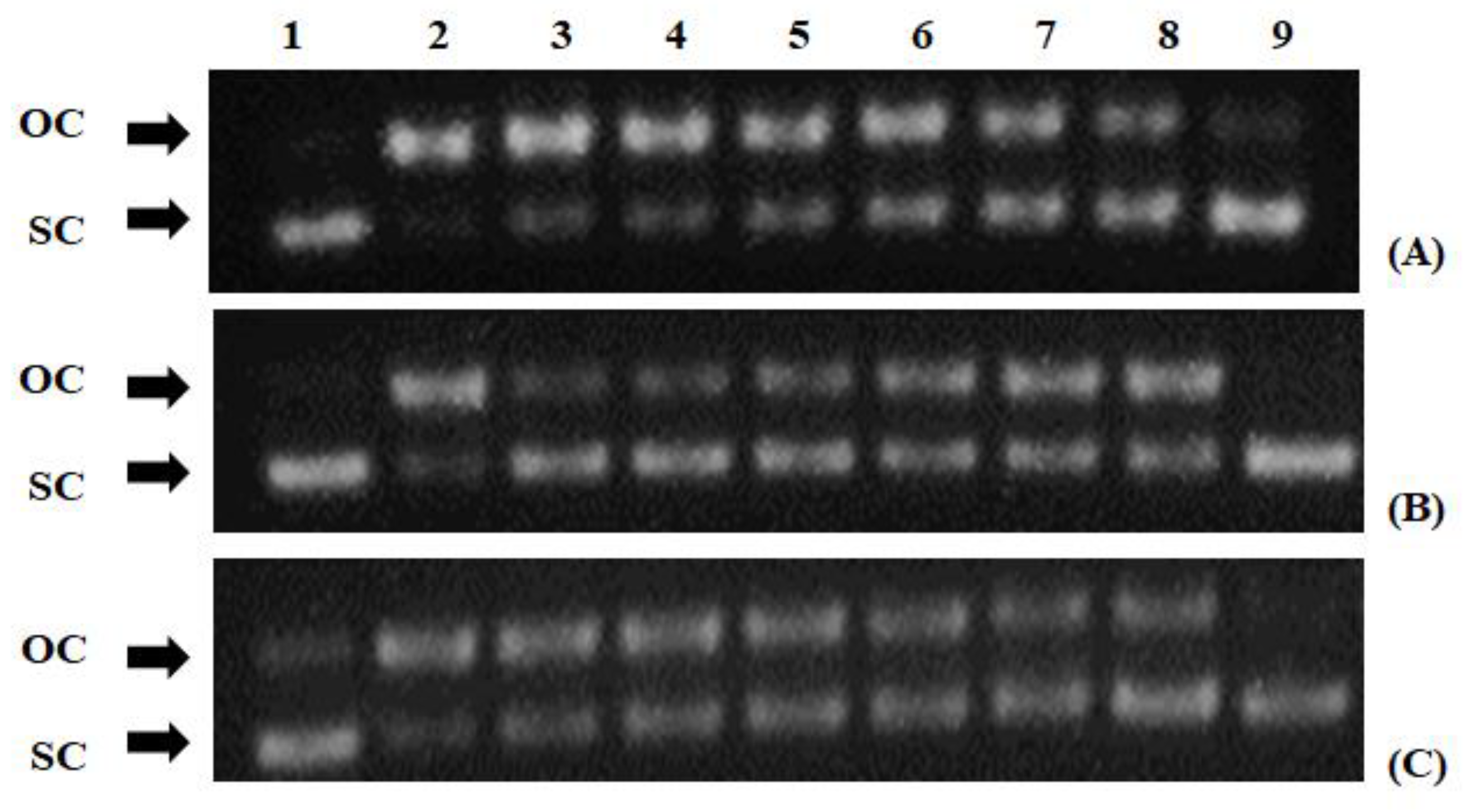
2.7. Peroxyl Radical-Induced DNA Plasmid Strand Cleavage
2.8. Cell Culture Conditions
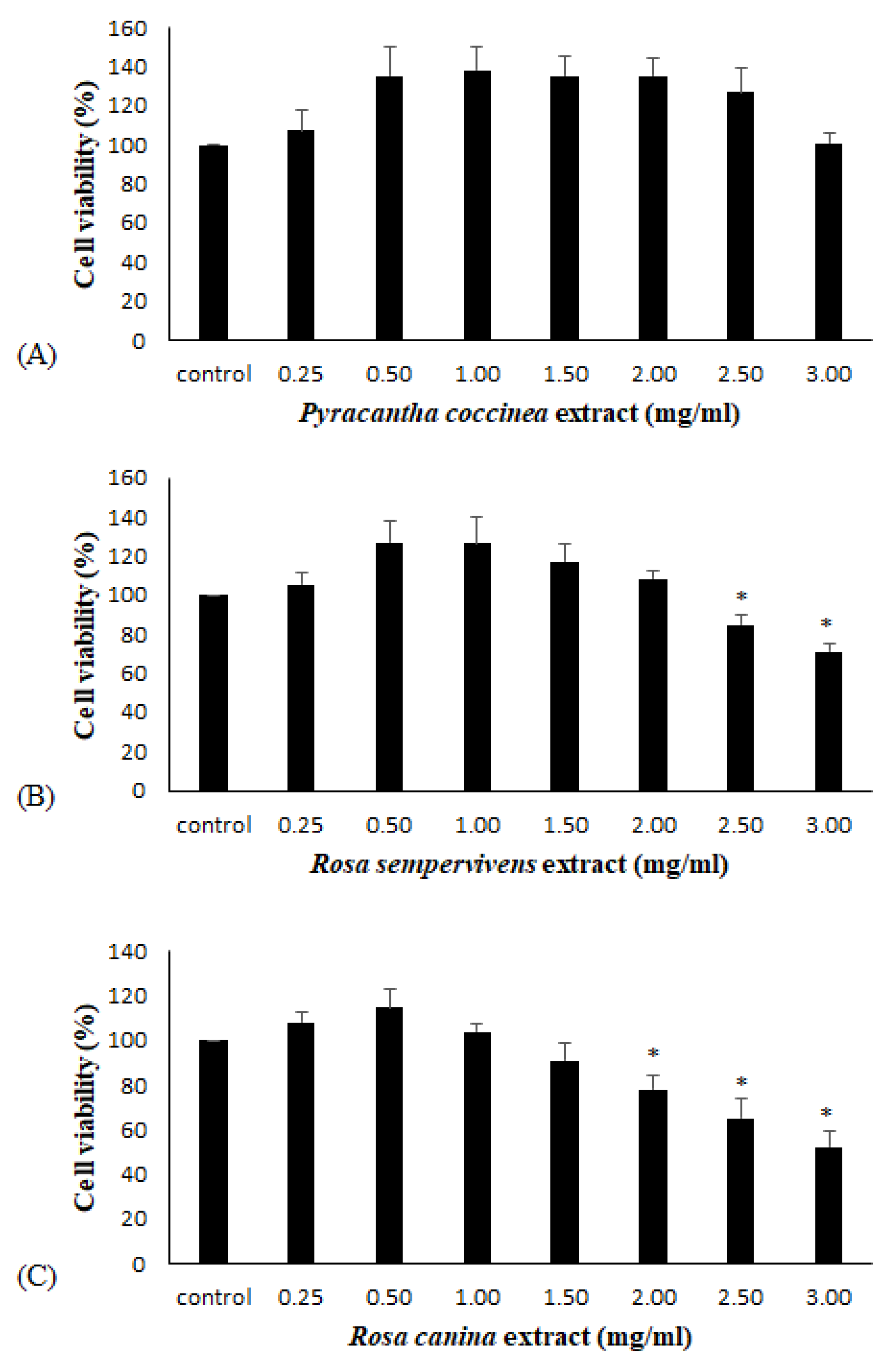
2.9. XTT Cell Viability Assay
2.10. Treatment of EA.hy926 Cells with the Extracts
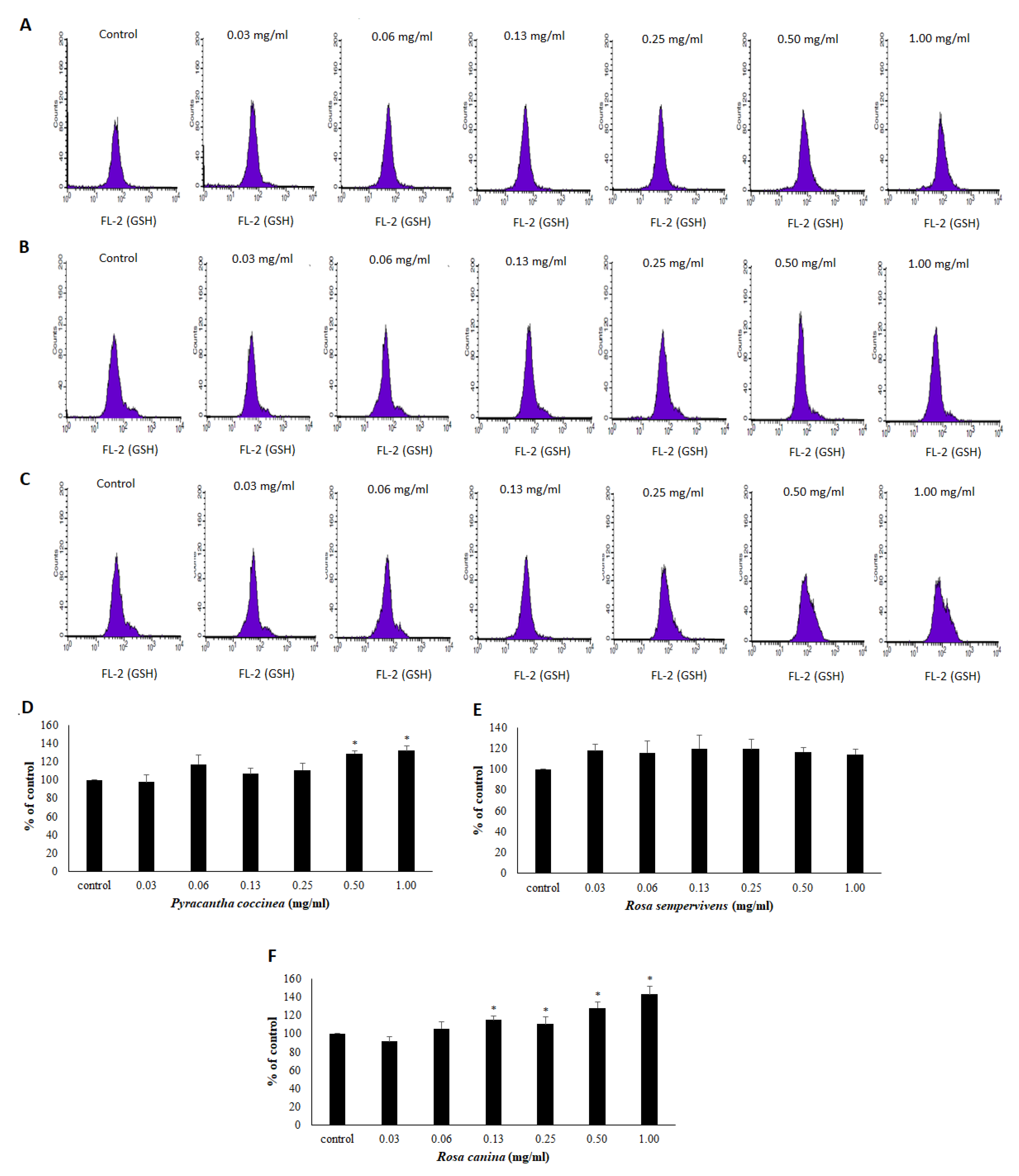
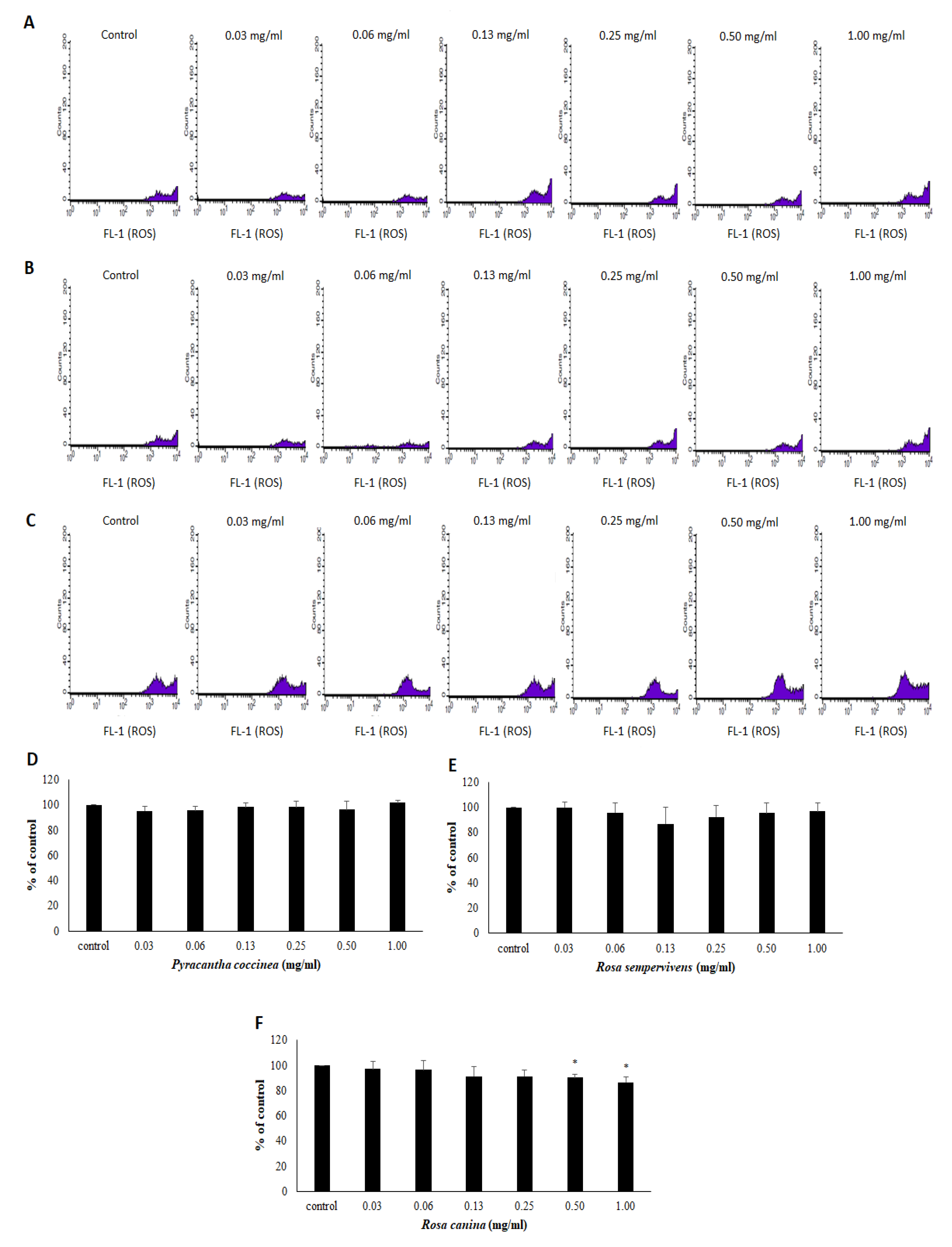
2.11. Assessment of GSH and ROS Levels by Flow Cytometry Analysis in Endothelial Cells
2.12. Statistical Analysis
3. Results
3.1. Extract Composition in Bioactive Compounds
3.2. Free Radical Scavenging Activity of the Extracts
3.3. Effects of Extracts on the Antioxidant Status of Endothelial Cells
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Dupre-Crochet, S.; Erard, M.; Nüβe, O. ROS production in phagocytes: Why, when, and where? J. Leukoc. Biol. 2013, 94, 657–670. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and other reactive species in disease. In Encyclopedia of Life Sciences; Wiley, John & Sons: London, UK, 2001; pp. 1–7. [Google Scholar]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Oxid. Med. Cell Longev. 2016, 2016, e4350965. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L.; Zeller, M.; Cottin, Y.; Vergely, C. Diabetes, oxidative stress and therapeutic strategies. Biochim. Biophys. Acta 2014, 1840, 2709–2729. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, B. Pathophysiology of oxidative stress in diabetes mellitus. J. Diabetes Complic. 2001, 15, 203–210. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and antioxidants: Updating a personal view. Nutr. Rev. 2012, 70, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Deanfield, J.E.; Halcox, J.P.; Rabelink, T.J. Endothelial function and dysfunction: Testing and clinical relevance. Circulation 2007, 115, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Victor, V.M.; Rocha, M.; Solá, E.; Bañuls, C.; Garcia-Malpartida, K.; Hernández-Mijares, A. Oxidative stress, endothelial dysfunction and atherosclerosis. Curr. Pharm. Des. 2009, 15, 2988–3002. [Google Scholar] [CrossRef]
- Closa, D.; Folch-Puy, E. Oxygen free radicals and the systemic inflammatory response. IUBMB Life 2004, 56, 185–191. [Google Scholar] [CrossRef]
- Muller, M.M.; Griesmacher, A. Markers of endothelial dysfunction. Clin. Chem. Lab. Med. 2000, 38, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Yoon, S.; Jung, K.J.; Kim, C.H.; Son, T.G.; Kim, M.S.; Kim, Y.J.; Lee, J.; Yu, B.P.; Chung, H.Y. Upregulation of aortic adhesion molecules during aging. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 232–244. [Google Scholar] [CrossRef]
- Karpinska, A.; Gromadzka, G. Oxidative stress and natural antioxidant mechanisms: The role in neurodegeneration. From molecular mechanisms to therapeutic strategies. Postepy Hig. Med. Dosw. 2013, 67, 43–53. [Google Scholar] [CrossRef]
- Landete, J.M. Dietary intake of natural antioxidants: Vitamins and polyphenols. Crit. Rev. Food Sci. Nutr. 2013, 53, 706–721. [Google Scholar] [CrossRef] [PubMed]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M. Updated knowledge about polyphenols: Functions, bioavailability, metabolism, and health. Crit. Rev. Food Sci. Nutr. 2012, 52, 936–948. [Google Scholar] [CrossRef]
- Jiménez, S.; Jiménez-Moreno, N.; Luquin, A.; Laguna, M.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Chemical composition of rosehips from different Rosa species: An alternative source of antioxidants for food industry. Food Addict. Contam. Part A 2017, 34, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Roman, I.; Stanila, A.; Stanila, S. Bioactive compounds and antioxidant activity of Rosa canina L. Biotypes from spontaneous flora of Transylvania. Chem. Cent. J. 2013, 7, 73. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.; Barros, L.; Calhelha, R.C.; Carvalho, A.M.; Queiroz, M.J.R.; Ferreira, I.C. Bioactivity of different enriched phenolic extracts of wild fruits from northeastern portugal: A comparative study. Plant Foods Hum. Nutr. 2014, 69, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Nađpal, J.D.; Lesjak, M.M.; Šibul, F.S.; Anačkov, G.T.; Četojević-Simin, D.D.; Mimica-Dukić, N.M.; Beara, I.N. Comparative study of biological activities and phytochemical composition of two rose hips and their preserves: Rosa canina L. and Rosa arvensis Huds. Food Chem. 2016, 192, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Serteser, A.; Kargioğlu, M.; Gök, V.; Bağci, Y.; Ozcan, M.M.; Arslan, D. Determination of antioxidant effects of some plant species wild growing in Turkey. Int. J. Food Sci. Nutr. 2008, 59, 643–651. [Google Scholar] [CrossRef]
- Tumbas, V.T.; Canadanovic-Brunet, J.M.; Cetojevic-Simin, D.D.; Cetkovic, G.S.; Ethilas, S.M.; Gille, L. Effect of rosehip (Rosa canina L.) phytochemicals on stable free radicals and human cancer cells. J. Sci. Food Agric. 2012, 92, 1273–1281. [Google Scholar] [CrossRef]
- Andersson, S.C.; Olsson, M.E.; Gustavsson, K.E.; Johansson, E.; Rumpunen, K. Tocopherols in rose hips (Rosa spp.) during ripening. J. Sci. Food Agric. 2012, 92, 2116–2121. [Google Scholar] [CrossRef] [PubMed]
- Keser, S. Antiradical activities and phytochemical compounds of firethorn (Pyracantha coccinea) fruit extracts. Nat. Prod. Res. 2014, 28, 1789–1794. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Jia, Z.; Tang, M.C.; Wu, J.M. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Cano, A.; Hernández-Ruíz, J.; García-Cánovas, F.; Acosta, M.; Arnao, M.B. An end-point method for estimation of the total antioxidant activity in plant material. Phytochem. Anal. 1998, 9, 196–202. [Google Scholar] [CrossRef]
- Chang, S.T.; Wu, J.H.; Wang, S.Y.; Kang, P.L.; Yang, N.S.; Shyur, L.F. Antioxidant activity of extracts from Acacia confusa bark and heartwood. J. Agric. Food Chem. 2001, 49, 3420–3424. [Google Scholar] [CrossRef] [PubMed]
- Priftis, A.; Panagiotou, E.M.; Lakis, K.; Plika, C.; Halabalaki, M.; Ntasi, G.; Veskoukis, A.S.; Stagos, D.; Skaltsounis, L.A.; Kouretas, D. Roasted and green coffee extracts show antioxidant and cytotoxic activity in myoblast and endothelial cell lines in a cell specific manner. Food Chem. Toxicol. 2018, 114, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Kerasioti, E.; Stagos, D.; Priftis, A.; Aivazidis, S.; Tsatsakis, A.M.; Hayes, A.W.; Kouretas, D. Antioxidant effects of whey protein on muscle C2C12 cells. Food Chem. 2014, 155, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Goutzourelas, N.; Stagos, D.; Demertzis, N.; Mavridou, P.; Karterolioti, H.; Georgadakis, S.; Kerasioti, E.; Aligiannis, N.; Skaltsounis, L.; Statiri, A.; et al. Effects of polyphenolic grape extract on the oxidative status of muscle and endothelial cells. Hum. Exp. Toxicol. 2014, 33, 1099–1112. [Google Scholar] [CrossRef]
- Kerasioti, E.; Stagos, D.; Georgatzi, V.; Bregou, E.; Priftis, A.; Kafantaris, I.; Kouretas, D. Antioxidant Effects of Sheep Whey Protein on Endothelial Cells. Oxid. Med. Cell Longev. 2016, 2016, e6585737. [Google Scholar] [CrossRef]
- Ayati, Z.; Amiri, M.S.; Ramezani, M.; Delshad, E.; Sahebkar, A.; Emami, S.A. Phytochemistry, traditional uses and pharmacological profile of rose hip: A review. Curr. Pharm. Des. 2018, 24, 4101–4124. [Google Scholar] [CrossRef]
- Nađpal, J.D.; Lesjak, M.M.; Mrkonjić, Z.O.; Majkić, T.M.; Četojević-Simin, D.D.; Mimica-Dukić, N.M.; Beara, I.N. Phytochemical composition and in vitro functional properties of three wild rose hips and their traditional preserves. Food Chem. 2018, 241, 290–300. [Google Scholar] [CrossRef]
- Gao, X.; Bjo, L.; Ttrajkovski, V.; Uggla, M. Evaluation of antioxidant activities of rosehip ethanol extracts in different test systems. J. Sci. Food Agric. 2000, 80, 2021–2027. [Google Scholar] [CrossRef]
- Koczka, N.; Stefanovits-Banyai, E.; Ombodi, A. Total Polyphenol Content and Antioxidant Capacity of Rosehips of Some Rosa Species. Medicines 2018, 5, 84. [Google Scholar] [CrossRef]
- Bhave, A.; Schulzova, V.; Chmelarova, H.; Mrnka, L.; Hajslova, J. Assessment of rosehips based on the content of their biologically active compounds. J. Food Drug Anal. 2017, 25, 681–690. [Google Scholar] [CrossRef]
- Elmastas, M.; Demir, A.; Genç, N.; Dölek, Ü.; Günes, M. Changes in flavonoid and phenolic acid contents in some Rosa species during ripening. Food Chem. 2017, 25, 154–159. [Google Scholar] [CrossRef]
- Cunja, V.; Mikulic-Petkovsek, M.; Zupan, A.; Stampar, F.; Schmitzer, V. Frost decreases content of sugars, ascorbic acid and some quercetin glycosides but stimulates selected carotenes in Rosa canina hips. J. Plant Physiol. 2015, 178, 55–63. [Google Scholar] [CrossRef]
- Wenzig, E.M.; Widowitz, U.; Kunert, O.; Chrubasik, S.; Bucar, F.; Knauder, E.; Bauer, R. Phytochemical composition and in vitro pharmacological activity of two rose hip (Rosa canina L.) preparations. Phytomedicine 2008, 15, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Fusi, J.; Bianchi, S.; Daniele, S.; Pellegrini, S.; Martini, C.; Galetta, F.; Giovannini, L.; Franzoni, F. An in vitro comparative study of the antioxidant activity and SIRT1 modulation of natural compounds. Biomed. Pharmacother. 2018, 101, 805–819. [Google Scholar] [CrossRef]
- Yan, X.T.; Li, W.; Sun, Y.N.; Yang, S.Y.; Lee, S.H.; Chen, J.B.; Jang, H.D.; Kim, Y.H. Identification and biological evaluation of flavonoids from the fruits of Prunus mume. Bioorg. Med. Chem. Lett. 2014, 24, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.N.; Jang, H.D. Flavonol content in the water extract of the mulberry (Morus alba L.) leaf and their antioxidant capacities. J. Food Sci. 2011, 76, C869–C873. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; León-Carmona, J.R.; Alvarez-Idaboy, J.R. Influence of the environment on the protective effects of guaiacol derivatives against oxidative stress: Mechanisms, kinetics, and relative antioxidant activity. J. Phys. Chem. B 2012, 116, 7129–7137. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Kang, H.J.; Kim, S.Z.; Kwon, T.O.; Jeong, S.I.; Jang, S.I. Antioxidant effect of astragalin isolated from the leaves of Morus alba L. against free radical-induced oxidative hemolysis of human red blood cells. Arch. Pharm. Res. 2013, 36, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Vasantha Rupasinghe, H.P.; Yasmin, A. Inhibition of oxidation of aqueous emulsions of omega-3 fatty acids and fish oil by phloretin and phloridzin. Molecules 2010, 15, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Marino, T.; Galano, A.; Russo, N. Radical scavenging ability of gallic acid toward OH and OOH radicals. Reaction mechanism and rate constants from the density functional theory. J. Phys. Chem. B 2014, 118, 10380–10389. [Google Scholar] [CrossRef] [PubMed]
- Aquilano, K.; Baldelli, S.; Ciriolo, M.R. Glutathione: New roles in redox signaling for an old antioxidant. Front. Pharmacol. 2014, 5, 196. [Google Scholar] [CrossRef]
- Kirkeskov, B.; Christensen, R.; Bügel, S.; Bliddal, H.; Danneskiold-Samsøe, B.; Christensen, L.P.; Andersen, J.R. The effects of rose hip (Rosa canina) on plasma antioxidative activity and C-reactive protein in patients with rheumatoid arthritis and normal controls: A prospective cohort study. Phytomedicine 2011, 18, 953–958. [Google Scholar] [CrossRef]
- Li, H.B.; Yi, X.; Gao, J.M.; Ying, X.X.; Guan, H.Q.; Li, J.C. The mechanism of hyperoside protection of ECV-304 cells against tert-butyl hydroperoxide-induced injury. Pharmacology 2008, 82, 105–113. [Google Scholar] [CrossRef]
- Gong, G.; Qin, Y.; Huang, W.; Zhou, S.; Yang, X.; Li, D. Rutin inhibits hydrogen peroxide-induced apoptosis through regulating reactive oxygen species mediated mitochondrial dysfunction pathway in human umbilical vein endothelial cells. Eur. J. Pharmacol. 2010, 628, 27–35. [Google Scholar] [CrossRef]
- Elliott, S.J.; Koliwad, S.K. Redox control of ion channel activity in vascular endothelial cells by glutathione. Microcirculation 1997, 4, 341–347. [Google Scholar] [CrossRef]
- Espinosa-Díez, C.; Miguel, V.; Vallejo, S.; Sánchez, F.J.; Sandoval, E.; Blanco, E.; Cannata, P.; Peiró, C.; Sánchez-Ferrer, C.F.; Lamas, S. Role of glutathione biosynthesis in endothelial dysfunction and fibrosis. Redox Biol. 2018, 14, 88–99. [Google Scholar] [CrossRef]
- Jiménez, S.; Gascón, S.; Luquin, A.; Laguna, M.; Ancin-Azpilicueta, C.; Rodríguez-Yoldi, M.J. Rosa canina Extracts Have Antiproliferative and Antioxidant Effects on Caco-2 Human Colon Cancer. PLoS ONE 2016, 11, e0159136. [Google Scholar]
- Zhang, T.; Mu, Y.; Yang, M.; Al Maruf, A.; Li, P.; Li, C.; Dai, S.; Lu, J.; Dong, Q. (+)-Catechin prevents methylglyoxal-induced mitochondrial dysfunction and apoptosis in EA.hy926 cells. Arch. Physiol. Biochem. 2017, 123, 121–127. [Google Scholar] [CrossRef]
- Ruijters, E.J.; Weseler, A.R.; Kicken, C.; Haenen, G.R.; Bast, A. The flavanol (−)-epicatechin and its metabolites protect against oxidative stress in primary endothelial cells via a direct antioxidant effect. Eur. J. Pharmacol. 2013, 715, 147–153. [Google Scholar] [CrossRef]
- Li, Q.; Zhao, W.; Zeng, X.; Hao, Z. Ursolic Acid Attenuates Atherosclerosis in ApoE(−/−) Mice: Role of LOX-1 Mediated by ROS/NF-κB Pathway. Molecules 2018, 23, 1101. [Google Scholar] [CrossRef]
- Cavalera, M.; Axling, U.; Rippe, C.; Swärd, K.; Holm, C. Dietary rose hip exerts antiatherosclerotic effects and increases nitric oxide-mediated dilation in ApoE-null mice. J. Nutr. Biochem. 2017, 44, 52–59. [Google Scholar] [CrossRef]
| Compound | R. caninaa | R. semprevirensa | P. coccineaa |
|---|---|---|---|
| Quinic acid | 1102.59 ± 38.91 | 389.95 ± 10.29 | ND |
| (+)-Catechin | 134.75 ± 1.02 | 25.48 ± 0.68 | 7.93 ± 0.31 |
| Gallic acid | 2.21 ± 0.09 | 0.44 ± 0.03 | ND |
| Protocatechuic acid | 2.09 ± 0.06 | ND | ND |
| Syringic acid | ND | ND | 6.23 ± 0.17 |
| Caffeic acid | ND | ND | 1.49 ± 0.08 |
| (−)-Epicatechin | 120.99 ± 1.18 | 34.01 ± 0.51 | 10.23 ± 1.10 |
| Hyperoside | 308.11 ± 7.10 | 8.31 ± 0.19 | 170.72 ± 3.49 |
| Rutin | 25.64 ± 0.48 | 2.62 ± 0.14 | 25.82 ± 0.98 |
| Chlorogenic acid | ND | ND | 4.82 ± 0.06 |
| Taxifolin | ND | ND | 0.09 ± 0.02 |
| p-Coumaric acid | 2.44 ± 0.07 | ND | ND |
| Vanillin | ND | ND | 7.89 ± 0.06 |
| Astragalin | 172.48 ± 7.48 | 9.16 ± 0.10 | 9.13 ± 0.05 |
| Phloridzin | 3.41 ± 0.10 | ND | ND |
| Eriodictyol | ND | 0.05 ± 0.01 | 0.06 ± 0.01 |
| Quercitrin | ND | 0.44 ± 0.07 | ND |
| Quercetin | 0.67 ± 0.05 | 0.19 ± 0.02 | 0.06 ± 0.01 |
| Genistein | ND | 0.03 ± 0.01 | ND |
| Kaempferol | 0.46 ± 0.02 | ND | 0.05 ± 0.01 |
| Isorhamnetin | ND | ND | ND |
| Isosakuranetin | ND | ND | 0.03 ± 0.01 |
| Betulinic acid | 0.47 ± 0.03 | ND | ND |
| Ursolic acid | 138.23 ± 4.44 | ND | ND |
| TPC b | 290.00 ± 2.10 d | 267.67 ± 1.78 e | 226.93 ± 1.11 f |
| TFC c | 118.56 ± 1.69 g | 65.78 ± 0.93 h | 99.16 ± 1.22 i |
| Plant Extracts | IC50 (μg/mL) | ||
|---|---|---|---|
| DPPH a | ABTS a | ROO b | |
| Rosa sempervivens (fruit) | 130 ± 7.8 a,* | 85 ± 10.0 d,* | 570 ± 51.3 g,* |
| Rosa canina (fruit) | 100 ± 7.0 b,* | 60 ± 6.6 e,* | 530 ± 68.9 g,* |
| Pyracantha coccinea (fruit) | 500 ± 40.0 c,* | 140 ± 4.2 f,* | 950 ± 47.5 h,* |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kerasioti, E.; Apostolou, A.; Kafantaris, I.; Chronis, K.; Kokka, E.; Dimitriadou, C.; Tzanetou, E.N.; Priftis, A.; Koulocheri, S.D.; Haroutounian, S.A.; et al. Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells. Antioxidants 2019, 8, 92. https://doi.org/10.3390/antiox8040092
Kerasioti E, Apostolou A, Kafantaris I, Chronis K, Kokka E, Dimitriadou C, Tzanetou EN, Priftis A, Koulocheri SD, Haroutounian SA, et al. Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells. Antioxidants. 2019; 8(4):92. https://doi.org/10.3390/antiox8040092
Chicago/Turabian StyleKerasioti, Efthalia, Anna Apostolou, Ioannis Kafantaris, Konstantinos Chronis, Eleana Kokka, Christina Dimitriadou, Evangelia N. Tzanetou, Alexandros Priftis, Sofia D. Koulocheri, Serkos A. Haroutounian, and et al. 2019. "Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells" Antioxidants 8, no. 4: 92. https://doi.org/10.3390/antiox8040092
APA StyleKerasioti, E., Apostolou, A., Kafantaris, I., Chronis, K., Kokka, E., Dimitriadou, C., Tzanetou, E. N., Priftis, A., Koulocheri, S. D., Haroutounian, S. A., Kouretas, D., & Stagos, D. (2019). Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells. Antioxidants, 8(4), 92. https://doi.org/10.3390/antiox8040092








