Evaluation of the AGE/sRAGE Axis in Patients with Multiple Myeloma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Control Subjects
2.2. AGEs Analysis
2.3. sRAGE Analysis
2.4. Statistical Analysis
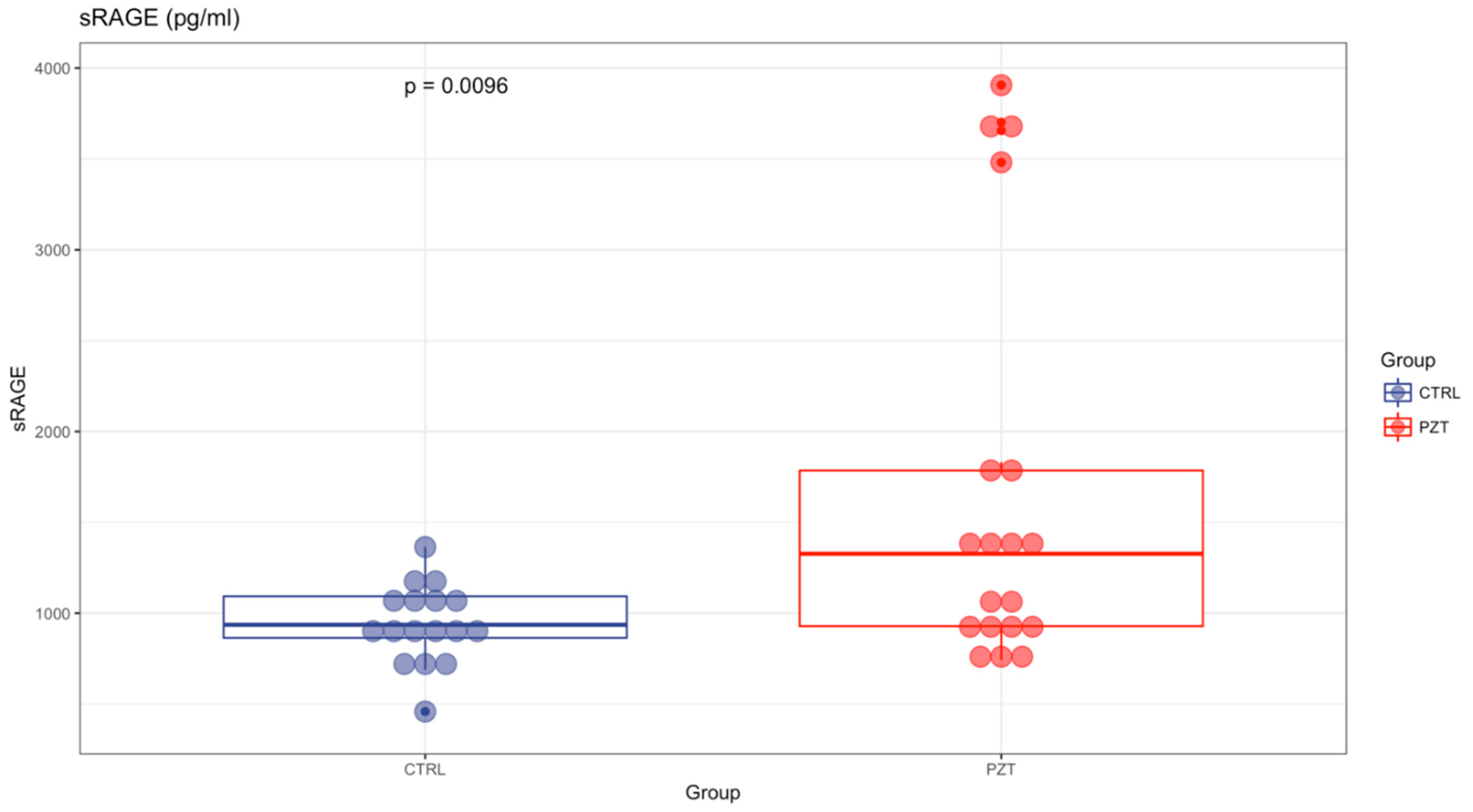
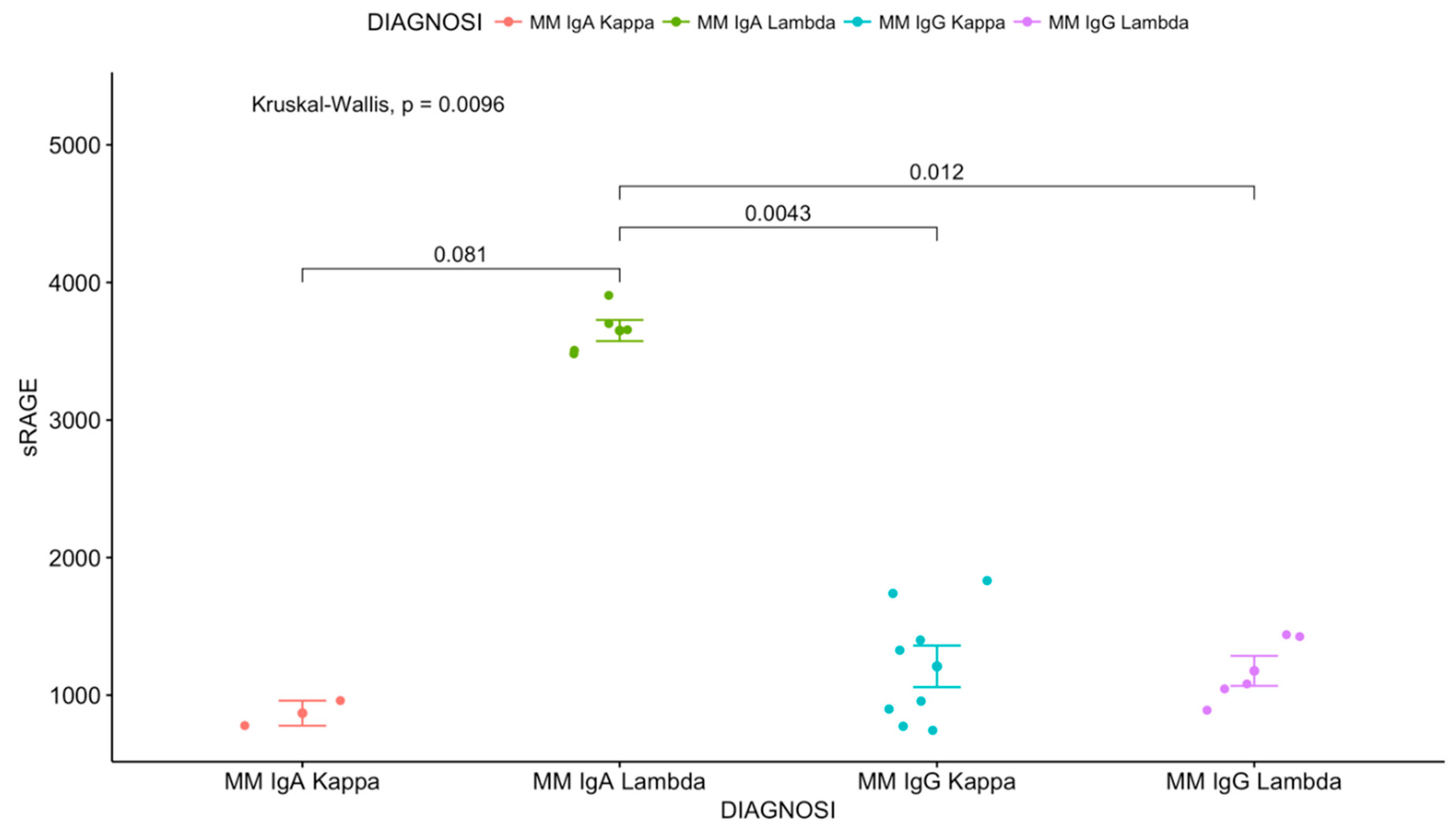
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gaens, K.H.; Stehouwer, C.D.; Schalkwijk, C.G. Advanced glycation endproducts and its receptor for advanced glycation endproducts in obesity. Curr. Opin. Lipidol. 2013, 24, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Khan, M.S.; Akhter, F.; Khan, M.S.; Khan, A.; Ashraf, J.M.; Pandey, R.P.; Shahab, U. Glycoxidation of biological macromolecules: A critical approach to halt the menace of glycation. Glycobiology 2014, 24, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Akhter, F.; Khan, M.S.; Alatar, A.A.; Faisal, M.; Ahmad, S. Antigenic role of the adaptive immune response to d-ribose glycated LDL in diabetes, atherosclerosis and diabetes atherosclerotic patients. Life Sci. 2016, 151, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, L.; Rylott, E.L.; Bruce, N.C.; Edwards, R.; Grogan, G. Structural evidence for Arabidopsis glutathione transferase AtGSTF2 functioning as a transporter of small organic ligands. FEBS Openbio 2017, 7, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Tesarova, P.; Kalousova, M.; Trnkova, B.; Soukupova, J.; Argalasova, S.; Mestek, O.; Petruzelka, L.; Zima, T. Carbonyl and oxidative stress in patients with breast cancer—Is there a relation to the stage of the disease? Neoplasma 2007, 54, 219–224. [Google Scholar] [PubMed]
- Yan, S.F.; Ramasamy, R.; Schmidt, A.M. Mechanisms of disease: Advanced glycation end-products and their receptor in inflammation and diabetes complications. Nat. Clin. Pract. Cardiovasc. Med. 2008, 4, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Sims, G.P.; Rowe, D.C.; Rietdijk, S.T.; Herbst, R.; Coyle, A.J. HMGB1 and RAGE in inflammation and cancer. Ann. Rev. Immunol. 2010, 28, 367–388. [Google Scholar] [CrossRef] [PubMed]
- Prasad, C.; Imrhan, V.; Marotta, F.; Juma, S.; Vijayagopal, P. Lifestyle and Advanced Glycation End Products (AGEs) Burden: Its Relevance to Healthy Aging. Aging Dis. 2014, 5, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, Y.; Matsui, T.; Takeuchi, M.; Yamagishi, S. Metformin inhibits advanced glycation end products (AGEs)-induced growth and VEGF expression in MCF-7 breast cancer cells by suppressing AGEs receptor expression via AMP-activated protein kinase. Horm. Metab. Res. 2013, 45, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Jing, R.; Wang, H.; Jiang, S.; Zhang, Z. Up-regulation of the Receptor for Advanced Glycation End Product (RAGE) in Esophageal Cancer and Down-regulation in Lung Cancer and Their Relationship to Clinicopathological Features. Lab. Med. 2008, 39, 661–667. [Google Scholar] [CrossRef]
- Demling, N.; Ehrhardt, C.; Kasper, M.; Laue, M.; Knels, L.; Rieber, E.P. Promotion of cell adherence and spreading: A novel function of RAGE, the highly selective differentiation marker of human alveolar epithelial type I cells. Cell Tissue Res. 2006, 323, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Raucci, A.; Cugusi, S.; Antonelli, A.; Barabino, S.M.; Monti, L.; Bierhaus, A.; Reiss, K.; Saftig, P.; Bianchi, M.E. A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J. 2008, 22, 3716–3727. [Google Scholar] [CrossRef] [PubMed]
- Galichet, A.; Weibel, M.; Heizmann, C.W. Calcium-regulated intramembrane proteolysis of the RAGE receptor. Biochem. Biophys. Res. Commun. 2008, 370, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Bukulin, M.; Kojro, E.; Roth, A.; Metz, V.V.; Fahrenholz, F.; Nawroth, P.P.; Bierhaus, A.; Postina, R. Receptor for advanced glycation end products is subjected to protein ectodomain shedding by metalloproteinases. J. Biol. Chem. 2008, 283, 35507–35516. [Google Scholar] [CrossRef] [PubMed]
- Tesarova, P.; Kalousova, M.; Jachymova, M.; Mestek, O.; Petruzelka, L.; Zima, T. Receptor for advanced glycation end products (RAGE)—Soluble form (sRAGE) and gene polymorphisms in patients with breast cancer. Cancer Investig. 2007, 25, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Gangemi, S.; Allegra, A.; Aguennouz, M.; Alonci, A.; Speciale, A.; Cannavo, A.; Cristani, M.; Russo, S.; Spatari, G.; Alibrandi, A.; et al. Relationship between advanced oxidation protein products, advanced glycation end products, and S-nitrosylated proteins with biological risk and MDR-1 polymorphisms in patients affected by B-chronic lymphocytic leukemia. Cancer Investig. 2012, 30, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Imbesi, S.; Musolino, C.; Allegra, A.; Saija, A.; Morabito, F.; Calapai, G.; Gangemi, S. Oxidative stress in oncohematologic diseases: An update. Expert Rev. Hematol. 2013, 6, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Gangemi, S.; Allegra, A.; Alonci, A.; Cristani, M.; Russo, S.; Speciale, A.; Penna, G.; Spatari, G.; Cannavo, A.; Bellomo, G.; et al. Increase of novel biomarkers for oxidative stress in patients with plasma cell disorders and in multiple myeloma patients with bone lesions. Inflamm. Res. 2012, 61, 1063–1067. [Google Scholar] [CrossRef] [PubMed]
- Musolino, C.; Alonci, A.; Allegra, A.; Saija, A.; Penna, G.; Cannavo, A.; Cristani, M.; Saitta, S.; Gangemi, S. Increase in serum protein carbonyl groups is associated with more advanced stage of disease in multiple myeloma patients. Biomarkers 2011, 16, 718–719. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.; Moreb, J.; Baitinger, C.; Singer, C.; Caudle, R.M. Advanced glycation endproducts (AGEs) in saliva of patients with multiple myeloma—A pilot study. Leuk. Lymphoma 2017, 58, 2934–2938. [Google Scholar] [CrossRef] [PubMed]
- Zilin, S.; Naifeng, L.; Bicheng, L.; Jiping, W. The determination of AGE-peptides by flow injection assay, a practical marker of diabetic nephropathy. Clin. Chim. Acta 2001, 313, 69–75. [Google Scholar] [CrossRef]
- Villa, M.; Parravano, M.; Micheli, A.; Gaddini, L.; Matteucci, A.; Mallozzi, C.; Facchiano, F.; Malchiodi-Albedi, F.; Pricci, F. A quick, simple method for detecting circulating fluorescent advanced glycation end-products: Correlation with in vitro and in vivo non-enzymatic glycation. Metab. Clin. Exp. 2017, 71, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Herwig, N.; Belter, B.; Pietzsch, J. Extracellular S100A4 affects endothelial cell integrity and stimulates transmigration of A375 melanoma cells. Biochem. Biophys. Res. Commun. 2016, 477, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Weinstein, S.J.; Albanes, D.; Taylor, P.R.; Graubard, B.I.; Virtamo, J.; Stolzenberg-Solomon, R.Z. Evidence that serum levels of the soluble receptor for advanced glycation end products are inversely associated with pancreatic cancer risk: A prospective study. Cancer Res. 2011, 71, 3582–3589. [Google Scholar] [CrossRef] [PubMed]
- Khorramdelazad, H.; Bagheri, V.; Hassanshahi, G.; Karami, H.; Moogooei, M.; Zeinali, M.; Abedinzadeh, M. S100A12 and RAGE expression in human bladder transitional cell carcinoma: A role for the ligand/RAGE axis in tumor progression? Asian Pac. J. Cancer Prev. 2015, 16, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Piperi, C.; Adamopoulos, C.; Papavassiliou, A.G. Potential of glycative stress targeting for cancer prevention. Cancer Lett. 2017, 390, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, E.; Roy, B. Altered Mitochondrial Signalling and Metabolism in Cancer. Front. Oncol. 2017, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-W.; Karin, M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Investig. 2007, 117, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-L.; Tai, C.-J.; Huang, C.-W.; Chang, F.-R.; Wang, J.-Y. Efficacy of Low-Molecular-Weight Fucoidan as a Supplemental Therapy in Metastatic Colorectal Cancer Patients: A Double-Blind Randomized Controlled Trial. Mar. Drugs 2017, 15, 122. [Google Scholar] [CrossRef] [PubMed]
- Mailankody, S.; Kazandjian, D.; Korde, N.; Roschewski, M.; Manasanch, E.; Bhutani, M.; Tageja, N.; Kwok, M.; Zhang, Y.; Zingone, A.; et al. Baseline mutational patterns and sustained MRD negativity in patients with high-risk smoldering myeloma. Blood Adv. 2017, 1, 1911–1918. [Google Scholar] [CrossRef] [PubMed]
- Rojas, A.; Figueroa, H.; Morales, E. Fueling inflammation at tumor microenvironment: The role of multiligand/RAGE axis. Carcinogenesis 2010, 31, 334–341. [Google Scholar] [CrossRef]
- Terzic, J.; Grivennikov, S.; Karin, E.; Karin, M. Inflammation and colon cancer. Gastroenterology 2010, 138, 2101–2114. [Google Scholar] [CrossRef]
- Rojas, A.; González, I.; Morales, E.; Pérez-Castro, R.; Romero, J.; Figueroa, H. Diabetes and cancer: Looking at the multiligand/RAGE axis. World J. Diabetes 2011, 2, 108–113. [Google Scholar] [CrossRef]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Bazhin, A.V.; Philippov, P.P.; Karakhanova, S. Reactive Oxygen Species in Cancer Biology and Anticancer Therapy. Oxid. Med. Cell. Longev. 2016, 2016, 2. [Google Scholar] [CrossRef]
- Gangemi, S.; Minciullo, P.; Magliacane, D.; Saitta, S.; Loffredo, S.; Saija, A.; Cristani, M.; Marone, G.; Triggiani, M. Oxidative stress markers are increased in patients with mastocytosis. Allergy 2015, 70, 436–442. [Google Scholar] [CrossRef]
- Smedsrød, B.; Melkko, J.; Araki, N.; Hiroyuki, S.; Horiuchi, S. Advanced glycation end products are eliminated by scavenger-receptor-mediated endocytosis in hepatic sinusoidal Kupffer and endothelial cells. Biochem. J. 1997, 322, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.N.; Qin, A.; Xu, J.; Wang, X. In situ accumulation of advanced glycation endproducts (AGEs) in bone matrix and its correlation with osteoclastic bone resorption. Bone 2011, 49, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Franke, S.; Ruster, C.; Pester, J.; Hofmann, G.; Oelzner, P.; Wolf, G. Advanced glycation end products affect growth and function of osteoblasts. Clin. Exp. Rheumatol. 2011, 29, 650–660. [Google Scholar] [PubMed]
- Zhou, Z.; Xiong, W.C. RAGE and its ligands in bone metabolism. Front. Biosci. 2011, 3, 768–776. [Google Scholar]
- Yamagishi, S.; Nakamura, K.; Matsui, T. Regulation of advanced glycation end product (AGE)-receptor (RAGE) system by PPAR-gamma agonists and its implication in cardiovascular disease. Pharmacol. Res. 2009, 60, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Engblom, C.; Pfirschke, C.; Zilionis, R.; Da Silva Martins, J.; Bos, S.A.; Courties, G.; Rickelt, S.; Severe, N.; Baryawno, N.; Faget, J.; et al. Osteoblasts remotely supply lung tumors with cancer-promoting SiglecF(high) neutrophils. Science 2017, 358. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.J.; Niu, H.X.; Hou, F.F.; Zhang, L.; Fu, N.; Nagai, R.; Lu, X.; Chen, B.H.; Shan, Y.X.; Tian, J.W. Advanced oxidation protein products activate vascular endothelial cells via a RAGE-mediated signaling pathway. Antioxid. Redox Signal. 2008, 10, 1699–1712. [Google Scholar] [CrossRef] [PubMed]
- Jing, R.; Cui, M.; Wang, J.; Wang, H. Receptor for advanced glycation end products (RAGE) soluble form (sRAGE): A new biomarker for lung cancer. Neoplasma 2010, 57, 55. [Google Scholar] [CrossRef] [PubMed]
- Matzner, Y.; Benbassat, J.; Polliack, A. Prognostic factors in multiple myeloma: A retrospective study using conventional statistical methods and a computer program. Acta Haematol. 1978, 60, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Geng, C.; Chen, W. Clinical characteristics of a group of patients with multiple myeloma who had two different λ light chains by immunofixation electrophoresis: A retrospective study from a single center. Exp. Ther. Med. 2015, 9, 1895–1900. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Duan, Z.; Tinker, L.; Sangi-Haghpeykar, H.; Strickler, H.; Ho, G.Y.; Gunter, M.J.; Rohan, T.; Logsdon, C.; White, D.L. A prospective study of soluble receptor for advanced glycation end-products and colorectal cancer risk in postmenopausal women. Cancer Epidemiol. 2016, 42, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Galliera, E.; Marazzi, M.; Vianello, E.; Drago, L.; Luzzati, A.; Bendinelli, P.; Maroni, P.; Tacchini, L.; Desiderio, M.A.; Corsi, M.R. Circulating sRAGE in the diagnosis of osteolytic bone metastasis. J. Biol. Regul. Homeost. Agents 2016, 30, 1203–1208. [Google Scholar] [PubMed]
- Geroldi, D.; Falcone, C.; Emanuele, E. Soluble receptor for advanced glycation end products: From disease marker to potential therapeutic target. Curr. Med. Chem. 2006, 13, 1971–1978. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Adachi, H.; Nakamura, K.; Matsui, T.; Jinnouchi, Y.; Takenaka, K.; Takeuchi, M.; Enomoto, M.; Furuki, K.; Hino, A.; et al. Positive association between serum levels of advanced glycation end products and the soluble form of receptor for advanced glycation end products in nondiabetic subjects. Metab: Clin. Exp. 2006, 55, 1227–1231. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Greten, F.R. NF-kappaB: Linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. 2005, 5, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Imaizumi, T. Serum levels of soluble form of receptor for advanced glycation end products (sRAGE) may reflect tissue RAGE expression in diabetes. Arterioscler. Thromb. Vasc. Biol. 2007, 27, e32, author reply e33-34. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Yamagishi, S.; Adachi, H.; Matsui, T.; Kurita-Nakamura, Y.; Takeuchi, M.; Inoue, H.; Imaizumi, T. Circulating advanced glycation end products (AGEs) and soluble form of receptor for AGEs (sRAGE) are independent determinants of serum monocyte chemoattractant protein-1 (MCP-1) levels in patients with type 2 diabetes. Diabetes Metab. Res. Rev. 2008, 24, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Tesarova, P.; Kalousova, M.; Zima, T.; Tesar, V. HMGB1, S100 proteins and other RAGE ligands in cancer—Markers, mediators and putative therapeutic targets. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2016, 160, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bongarzone, S.; Savickas, V.; Luzi, F.; Gee, A.D. Targeting the Receptor for Advanced Glycation Endproducts (RAGE): A Medicinal Chemistry Perspective. J. Med. Chem. 2017, 60, 7213–7232. [Google Scholar] [CrossRef] [PubMed]


| Values and Parameters | sRAGE | AGE | age | MC | BM PCs | CREATIN. | Beta2Microg. | Calcemia | Hb |
|---|---|---|---|---|---|---|---|---|---|
| sRAGE | 1 | ||||||||
| AGE | 0.37 | 1 | |||||||
| age (yrs) | 0.15 | 0.0062 | 1 | ||||||
| MC | 0.23 | −0.15 | 0.0061 | 1 | |||||
| BM PCs_ | 0.50 * | 0.19 | −0.29 | 0.48 * | 1 | ||||
| CREATININE | −0.081 | 0.49 * | 0.17 | 0.15 | −0.04 | 1 | |||
| Beta2Microg. | 0.33 | −0.022 | 0.46 * | 0.26 | −0.29 | 0.29 | 1 | ||
| Calcemia | 0.61 *** | 0.40 * | 0.12 | −0.19 | 0.23 | −0.21 | 0.096 | 1 | |
| Hb | −0.37 | 0.17 | −0.53 * | −0.058 *** | −0.31 | −0.052 | −0.49 * | −0.18 | 1 |
| Model/Variables Using Variables Most Highly Correlated with sRAGE | Adjusted R-square |
|---|---|
| BM PCs + Calc | 0.447 |
| BM PCs + Beta2Microglob. + Calc | 0.613 |
| BM PCs + Beta2 Microglob. + Calc: PCs | 0.746 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allegra, A.; Musolino, C.; Pace, E.; Innao, V.; Di Salvo, E.; Ferraro, M.; Casciaro, M.; Spatari, G.; Tartarisco, G.; Allegra, A.G.; et al. Evaluation of the AGE/sRAGE Axis in Patients with Multiple Myeloma. Antioxidants 2019, 8, 55. https://doi.org/10.3390/antiox8030055
Allegra A, Musolino C, Pace E, Innao V, Di Salvo E, Ferraro M, Casciaro M, Spatari G, Tartarisco G, Allegra AG, et al. Evaluation of the AGE/sRAGE Axis in Patients with Multiple Myeloma. Antioxidants. 2019; 8(3):55. https://doi.org/10.3390/antiox8030055
Chicago/Turabian StyleAllegra, Alessandro, Caterina Musolino, Elisabetta Pace, Vanessa Innao, Eleonora Di Salvo, Maria Ferraro, Marco Casciaro, Giovanna Spatari, Gennaro Tartarisco, Andrea Gaetano Allegra, and et al. 2019. "Evaluation of the AGE/sRAGE Axis in Patients with Multiple Myeloma" Antioxidants 8, no. 3: 55. https://doi.org/10.3390/antiox8030055
APA StyleAllegra, A., Musolino, C., Pace, E., Innao, V., Di Salvo, E., Ferraro, M., Casciaro, M., Spatari, G., Tartarisco, G., Allegra, A. G., & Gangemi, S. (2019). Evaluation of the AGE/sRAGE Axis in Patients with Multiple Myeloma. Antioxidants, 8(3), 55. https://doi.org/10.3390/antiox8030055







