Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview
Abstract
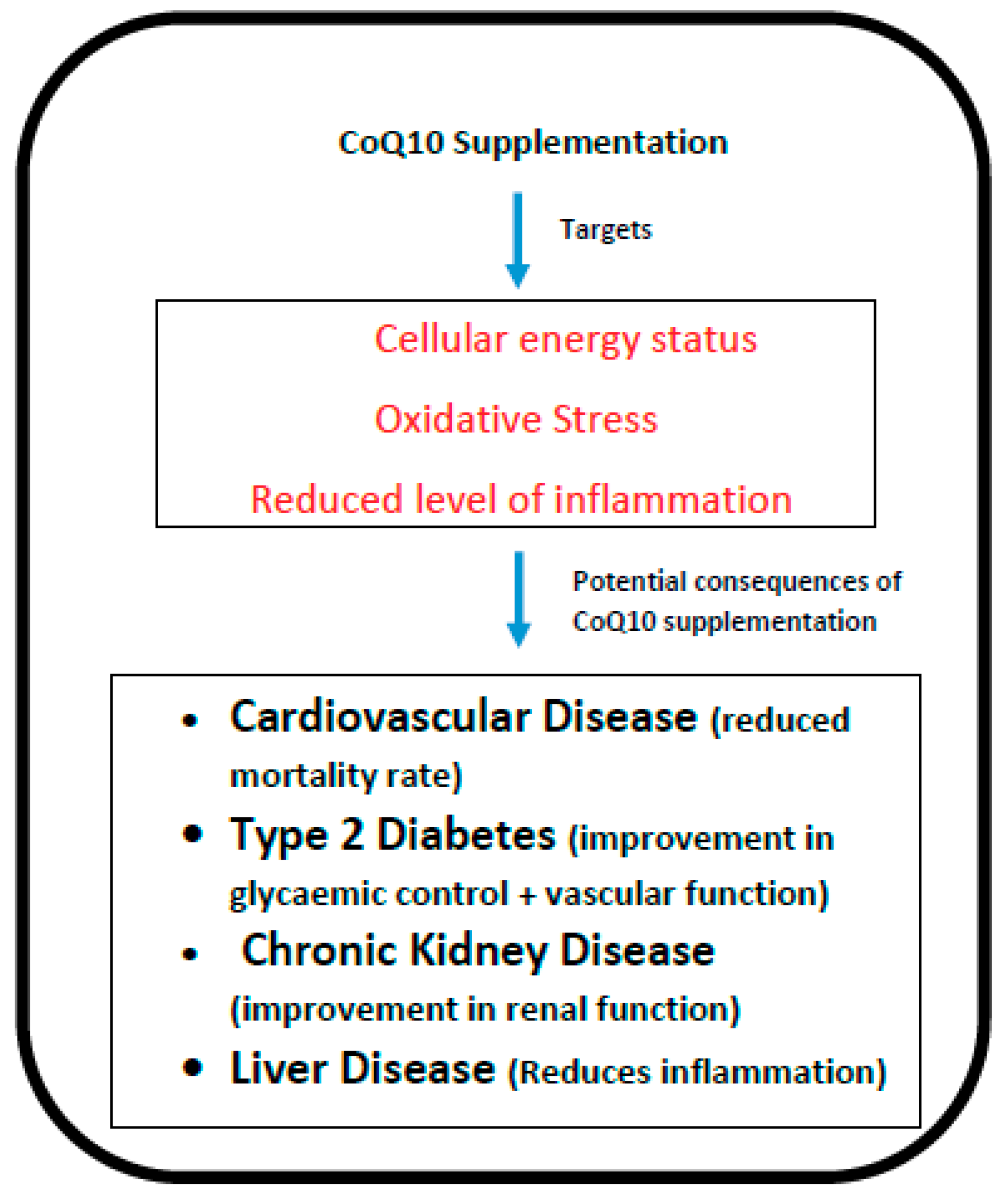
:1. Introduction
2. Cardiovascular Disease
3. Diabetes
4. Chronic Kidney Disease (CKD)
5. Liver Disease
6. CoQ10 Supplementation: Importance of Product Quality and Bioavailability
7. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- DuGoff, E.H.; Canudas-Romo, V.; Buttorff, C.; Leff, B.; Anderson, G.F. Multiple chronic conditions and life expectancy: A life table analysis. Med. Care 2014, 52, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, I.P. CoQ10 as a therapy for mitochondrial disease. Int. J. Biochem. Cell Biol. 2014, 49, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Feng, Y.; Chen, G.C.; Qin, L.Q.; Fu, C.L.; Chen, L.H. Effects of coenzyme Q10 supplementation on inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2017, 119, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Bo, Y.; Lu, Y.; Liu, C.; Zhang, L. Effects of Coenzyme Q10 on Markers of Inflammation: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0170172. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, C.; Lindner, I.; Rimbach, G.; Niklowitz, P.; Menke, T.; Döring, F. Functions of coenzyme Q10 in inflammation and gene expression. Biofactors 2008, 32, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Bysted, A.; Hłlmer, G. The coenzyme Q10 content of the average Danish diet. Int. J. Vitam. Nutr. Res. 1997, 67, 123–129. [Google Scholar] [PubMed]
- Kalén, A.; Appelkvist, E.L.; Dallner, G. Age-related changes in the lipid compositions of rat and human tissues. Lipids 1989, 24, 579–584. [Google Scholar] [CrossRef]
- Hernández-Camacho, J.D.; Bernier, M.; López-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef]
- Mantle, D. CoQ10 to treat and prevent heart disease. Br. J. Cardiac. Nurs. 2015, 10, 382–387. [Google Scholar] [CrossRef]
- Mantle, D. CoQ10 and cardiovascular disease: An overview. Br. J. Cardiol. 2015, 22, 1–5. [Google Scholar]
- Flowers, N.; Hartley, L.; Todkill, D.; Stranges, S.; Rees, K. Co-enzyme Q10 supplementation for primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef]
- Mortensen, S.A.; Rosenfeldt, F.; Kumar, A.; Dolliner, P.; Filipiak, K.J.; Pella, D.; Alehagen, U.; Steurer, G.; Littarru, G.P. The effect of CoQ10 on morbidity and mortality in chronic heart failure: The Q-SYMBIO randomized double blind trial. JACC 2014, 2, 641–649. [Google Scholar] [PubMed]
- Alehagen, U.; Johansson, P.; Björnstedt, M.; Rosén, A.; Dahlström, U. Cardiovascular mortality and N-terminal proBNP reduced after combined selenium and CoQ10 supplementation: A 5 year prospective randomised double blind placebo controlled trial among elderly Swedish citizens. Int. J. Cardiol. 2013, 167, 1860–1866. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Dahlström, Ö.; Dahlström, U.; Alehagen, U. Effect of selenium and CoQ10 on the cardiac biomarker NT-proBNP. Scand. Cardiovadc. J. 2013, 47, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Lindahl, T.L.; Aaseth, J.; Svensson, E.; Johansson, P. Levels of sP-selectin and hs-CRP decrease with dietary intervention with selenium and CoQ10. PLoS ONE 2015, e0137680. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Aaseth, J.; Johansson, P. Less increase of copeptin and MR-proADM due to intervention with selenium and CoQ10 for 4 years—Follow up results of a randomised controlled trial in elderly citizens. Biofactors 2015, 41, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Dahlström, Ö.; Dahlström, U.; Alehagen, U. Improved health related quality of life and more days out of hospital with supplementation with selenium and CoQ10 combined. J. Nutr. Health Aging 2015, 19, 870–877. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Johansson, P. Reduced cardiovascular mortality 10 years after supplementaion with selenium and CoQ10 for 4 years: Follow up results of a randomised controlled trial in elderly citizens. PLoS ONE 2015, 10, e0141641. [Google Scholar] [CrossRef]
- Okuyama, H.; Langsjoen, P.H.; Hamazaki, T.; Ogushi, Y.; Hama, R.; Kobayashi, T.; Uchino, H. Statins stimulate atherosclerosis and heart failure: Pharmacological mechanisms. Expert Rev. Clin. Pharmacol. 2015, 8, 189–199. [Google Scholar] [CrossRef]
- Alehagen, U.; Alexander, J.; Aaseth, J. Supplementation with selenium and CoQ10 reduces cardiovascular mortality in elderly with low selenium status. PLoS ONE 2016, 10, e0157541. [Google Scholar] [CrossRef]
- Mantle, D. CoQ10 supplementation for diabetes and its complications: An overview. Br. J. Diabetes 2017, 17, 1–4. [Google Scholar] [CrossRef]
- El-Ghoroury, E.A.; Raslan, H.M.; Badawy, E.A. Malondialdehyde and CoQ10 in platelets and serum in type II diabetes: Correlation with glycemic control. Blood Coagul. Fibrinolysis 2009, 20, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.G.; Forsen, T.J.; Mortensen, S.A.; Rohde, M. Effect of CoQ10 administration on metabolic control in type II diabetes mellitus. Biofactors 1999, 9, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.M.; Watts, G.F.; Playford, D.A.; Burke, V.; Croft, K.D. CoQ10 improves blood pressure and glycaemic control in subjects with type II diabetes. Eur. J. Clin. Nutr. 2002, 56, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Koladouz-Mohammadi, R.; Hosseinzadeh-Attar, M.; Eshraghian, M. Effect of CoQ10 supplementation on metabolic status of type II diabetic patients. Minerva Gastroenterol. Dietol. 2013, 59, 231–235. [Google Scholar]
- Zahedi, H.; Eghtesadi, S.; Seifirad, S.; Rezaee, N.; Shidfar, F.; Heydari, I.; Golestan, B.; Jazayeri, S. Effects of CoQ10 supplementation on lipid profiles and glycemic control in patients with type II diabetes. J. Diabetes Metabol. Disord. 2014, 13, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh-Attar, M.; Mohammadi, R.; Eshraghian, I.M. Reduction in asymmetric dimethylarginine plasma levels by CoQ10 supplementation in patients with type II diabetes. Minerva Endocrinol. 2015, 40, 259–266. [Google Scholar]
- Zarei, P.; Rezvanfar, M.R.; Ansarihadipour, H.; Delavar, M.; Abdollahi, M.; Khosrowbeygi, A. Effects of coenzyme Q10 supplementation on the serum levels of amylase, adenosine deaminase, catalase, and total antioxidant capacity in women with type 2 diabetes mellitus: A randomized, double-blind placebo-controlled trial. J. Res. Med. Sci. 2018. [Google Scholar] [CrossRef]
- Yen, C.H.; Chu, Y.J.; Lee, B.J.; Lin, Y.C.; Lin, P.T. Effect of liquid ubiquinol supplementation on glucose, lipids and antioxidant capacity in type 2 diabetes patients: A double-blind, randomised, placebo-controlled trial. Br. J. Nutr. 2018, 120, 57–63. [Google Scholar] [CrossRef]
- Akbari-Fakhrabadi, M.; Ghotrom, A.; Khosravi, H. Effect of CoQ10 on oxidative stress, glycemic control and inflammation in diabetic neuropathy: A double blind randomised clinical trial. Int. J. Vitam. Nutr. Res. 2014, 84, 252–260. [Google Scholar] [CrossRef]
- Raygan, F.; Rezavandi, Z.; Dadkhah Tehrani, S.; Farrokhian, A.; Asemi, Z. The effects of CoQ10 administration on glucose homeostasis parameters, lipid profiles, biomarkers of inflammation and oxidative stress in patients with metabolic syndrome. Eur. J. Nutr. 2016, 55, 2357–2364. [Google Scholar] [CrossRef] [PubMed]
- Mehrdadi, P.; Kolahdouz Mohammadi, R.; Alipoor, E.; Eshraghian, M.R.; Esteghamati, A.; Hosseinzadeh-Attar, M.J. Effect of CoQ10 supplementation on circulating levels of adipokine adipolin in overweight and obese patients withy type 2 diabetes. Exp. Clin. Endocrinol. Diabetes 2016, 125, 156–162. [Google Scholar] [PubMed]
- Yoo, J.Y.; Yum, K.S. Effect of Coenzyme Q10 on Insulin Resistance in Korean Patients with Prediabetes: A Pilot Single-Center, Randomized, Double-Blind, Placebo-Controlled Study. Biomed. Res. Int. 2018, 1613247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Yang, K.L.; Zeng, L.T.; Wu, X.H.; Huang, H.Y. Effectiveness of Coenzyme Q10 Supplementation for Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int. J. Endocrinol. 2018, 6484839. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.M.; Asaad, G.F.; Salam, R.M.; El-Abhar, H.S.; Arbid, M.S. Novel CoQ10 antidiabetic mechanisms underlie its positive effect: Modulation of insulin and adiponectin receptors, tyrosine kinase, glucose transporters and visfatin in insulin resistant/diabetic rats. PLoS ONE 2014, 9, e89169. [Google Scholar] [CrossRef] [PubMed]
- Heidari, A.; Hamidi, G.; Soleimani, A.; Aghadavod, E.; Asemi, Z. Effects of Coenzyme Q10 Supplementation on Gene Expressions Related to Insulin, Lipid, and Inflammation Pathways in Patients With Diabetic Nephropathy. Iran. J. Kidney Dis. 2018, 12, 14–21. [Google Scholar] [PubMed]
- Ates, O.; Bileu, H.; Keies, S. Plasma CoQ10 levels in type II diabetics with retinopathy. Int. J. Ophthalmol. 2013, 6, 675–679. [Google Scholar]
- Montano, S.J.; Grünler, J.; Nair, D.; Tekle, M.; Fernandes, A.P.; Hua, X.; Holmgren, A.; Brismar, K.; Ungerstedt, J.S. Glutaredoxin mediated redox effects of CoQ10 treatment in type 1 and type 2 diabetes patients. BBA Clin. 2015, 4, 14–20. [Google Scholar] [CrossRef]
- Hamilton, S.J.; Chew, G.T.; Watts, G.F. CoQ10 improves endothelial dysfunction in statin treated type II diabetic patients. Diabetes Care 2000, 32, 810–812. [Google Scholar] [CrossRef]
- Brault, M.; Ray, J.; Gomez, Y.H.; Mantzoros, C.S.; Daskalopoulou, S.S. Statin treatment and new onset diabetes: A review of proposed mechanisms. Metabolism 2014, 63, 735–745. [Google Scholar] [CrossRef]
- Cederberg, H.; Yaluri, N.; Modi, S. Increased risk of diabetes with statin treatment is associated with impaired insulin sensitivity and insulin secretion: A 6 year follow up study of the METSIM cohort. Diabetologia 2015, 58, 1109–1117. [Google Scholar] [CrossRef]
- Chan, D.C.; Pang, J.; Watts, D.F. Pathogenesis and management of the diabetogenic effect of statins: A role for adiponectin and CoQ10? Curr. Atheroscler. Rep. 2015, 17, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Gansean, S.; Ito, M.K. CoQ10 ameliorates reduction in GLUT4 transporter expression induced by simvastatin in 3T3-L1 adipocytes. Metab. Syndr. Relat. Disord. 2013, 11, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Kuhlman, A.B.; Morville, T.; Dohlmann, T.L.; Hansen, M.; Kelly, B.; Helge, J.W.; Dela, F. Coenzyme Q10 does not improve peripheral insulin sensitivity in statin-treated men and women; the LIFESTAT study. Appl. Physiol. Nutr. Metab. 2018. [Google Scholar] [CrossRef]
- Mantle, D.; Milford, D.; Hargreaves, I.P. Chronic kidney disease and CoQ10 supplementation: An overview. J. Kidney Care 2018, in press. [Google Scholar]
- Triolo, L.; Lippa, S.; De Sole, P.; Mori, R. Serum CoQ10 in uremic patients on chronic hemodialysis. Nephron 1994, 66, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Macunluoglu, B.; Kaya, Y.; Atakan, A.; Ari, E.; Kaspar, C.; Demir, H. Serum CoQ10 levels are associted with coronary flow reserve in hemodialysis patients. Hemodial. Int. 2013, 17, 339–345. [Google Scholar] [CrossRef]
- Yeung, C.K.; Billings, F.T.; Claessens, A.J.; Roshanravan, B.; Linke, L.; Sundell, M.B.; Ahmad, S.; Shao, B.; Shen, D.D.; Ikizler, T.A.; et al. CoQ10 dose escalation study in hemodialysis patients: Safety, tolerability and effect on oxidative stress. BMC Nephrol. 2015, 16, 183–191. [Google Scholar] [CrossRef]
- Singh, R.B.; Kumar, A.; Niaz, M.A.; Singh, R.G.; Gujrati, S.; Singh, V.P. Randomised double blind placebo controlled trial of CoQ10 in chronic renal failure. J. Nutr. Environ. Med. 2000, 10, 281–288. [Google Scholar] [CrossRef]
- Hargreaves, I.P.; Al Sharhrani, M.; Wainright, L.; Heales, S.J. Drug induced mitochondrial toxicity. Drug Saf. 2016, 39, 661–674. [Google Scholar] [CrossRef]
- Zahed, N.S.; Ghassami, M.; Nikbahkt, H. Effects of CoQ10 supplementation on C-reactive protein and homocysteine as inflammatory markers in hemodialysis patients: A randomised controlled trial. J. Nephropathol. 2016, 5, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, M.; Alleva, R.; Solenghi, M.; Littarru, G.P. Distribution of antioxidants among blood components and lipoproteins: Significance of lipids/CoQ10 ratio as a possible marker of increased risk for atherosclerosis. Biofactors 1999, 9, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Lippa, S.; Colacicco, L.; Bondanini, F.; Calla, C.; Gozzo, M.L.; Ciccariello, M. Plasma levels of CoQ10, vitamin E and lipids in uremic patients on conservative therapy and hemodialysis treatment: Some biochemical and clinical implications. Clin. Chim. Acta 2000, 292, 81–91. [Google Scholar] [CrossRef]
- Bakhshayeshkaram, M.; Lankarani, K.B.; Mirhosseini, N.; Tabrizi, R.; Akbari, M.; Dabbaghmanesh, M.H.; Asemi, Z. The effects of coenzyme Q10 supplementation on metabolic profiles of patients with chronic kidney disease: A systematic review and meta-analysis of randomized controlled trials. Curr. Pharm. Des. 2018. [Google Scholar] [CrossRef] [PubMed]
- Macunluoglu, B.; Atakan, A.; Ari, E.; Kaya, Y.; Kaspar, C.; Demir, C. Epicardial fat tissue thickness is correlated with diminished levels of CoQ10, a major antioxidant in hemodialysis patients. Clin. Biochem. 2014, 47, 1231–1234. [Google Scholar] [CrossRef] [PubMed]
- Francque, S.M.; van der Graaff, D.; Kwanten, W.J. NAFLD and cardiovascular risk:pathophysiological mechanisms and implications. J. Hepatol. 2016, 65, 425–443. [Google Scholar] [CrossRef] [PubMed]
- Sîrbu, O.; Floria, M.; Dăscălița, P.; Şorodoc, V.; Şorodoc, L. NAFLD from the cardiologist perspective. Anatol. J. Cardiol. 2016, 16, 534–541. [Google Scholar] [PubMed]
- Milić, S.; Lulić, D.; Štimac, D.; Ružić, A.; Zaputović, L. Cardiac manifestations in alcoholic liver disease. Postgrad. Med. J. 2016, 92, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Preedy, V.R. Free radicals as mediators of alcohol toxicity. Advers. Drug React. Toxicol. Rev. 1999, 18, 235–253. [Google Scholar]
- Spahis, S.; Delvin, E.; Borys, J.M.; Levy, E. Oxidative stress as a critical factor in nonalcoholic fatty liver disease pathogenesis. Antioxid. Redox Signal. 2017, 26, 519–541. [Google Scholar] [CrossRef]
- Fouad, A.A.; Jresat, I. Hepatoprotective effect of CoQ10 in rats with acetaminophen toxicity. Environ. Toxicol. Pharmacol. 2012, 33, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Pokharel, Y.R.; Lim, S.C.; Han, H.K.; Ryu, C.S.; Kim, S.K.; Kwak, M.K.; Kang, K.W. Inhibition of liver fibrosis by CoQ10: Role of Nrf2 activation in inhibiting transforming growth factor beta expression. Toxicol. Appl. Pharmacol. 2009, 240, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.A.; Shoheib, Z.S.; Abdel-Aleem, G.A.; Shareef, M.M. Experimental schistosomal hepatitis: Protective effect of CoQ10. Exp. Parasitol. 2008, 120, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Tarry-Adkins, J.L.; Fernandez-Twinn, D.S.; Hargreaves, I.P.; Neergheen, V.; Aiken, C.E.; Martin-Gronert, M.S.; McConnell, J.M.; Ozanne, S.E. CoQ10 prevents hepatic fibrosis, inflammation and oxidative stress in a rat model of poor maternal nutrition. Am. J. Clin. Nutr. 2016, 103, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Yesilova, Z.; Yaman, H.; Oktenli, C.; Ozcan, A.; Uygun, A.; Cakir, E.; Sanisoglu, S.Y.; Erdil, A.; Ates, Y.; Aslan, M.; et al. Systemic markers of lipid peroxidation and antioxidants in patients with non-alcoholic fatty liver disease. Am. J. Gastroenterol. 2005, 100, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Farhangi, M.A.; Alipour, B.; Jafarvand, E.; Khoshbaten, M. Oral CoQ10 supplementation in patients with NAFLD: Effects on serum vaspin, chemerin, pentraxin, insulin resistance and oxidative stress. Arch. Med. Res. 2014, 45, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Farsi, F.; Mohammadshahi, M.; Alavinejad, P.; Rezazadeh, A.; Zarei, M.; Engali, K.A. Functions of CoQ10 supplementation on liver enzymes, markers of inflammation and adipokines in patients with NAFLD: A double blind randomized placebo controlled clinical trial. J. Am. Coll. Nutr. 2016, 35, 346–353. [Google Scholar] [CrossRef]
- López-Lluch, G.; Del Pozo-Cruz, J.; Sánchez-Cuesta, A.; Cortés-Rodríguez, A.B.; Navas, P. Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutrition 2019, 57, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Madmani, M.E.; Yusuf, A.; Tamr, K.; Madmani, Y.; Essali, A.; Kadro, W. CoQ10 for heart failure. Cochrane Database Syst. Rev. 2014, CD008684. [Google Scholar] [CrossRef]
- Lei, L.; Liu, Y. Efficacy of coenzyme Q10 in patients with cardiac failure: A meta-analysis of clinical trials. BMC Cardiovasc. Disord. 2017, 17, 196. [Google Scholar] [CrossRef]
- Tabrizi, R.; Akbari, M.; Sharifi, N.; Lankarani, K.B.; Moosazadeh, M.; Kolahdooz, F.; Taghizadeh, M.; Asemi, Z. The Effects of Coenzyme Q10 Supplementation on Blood Pressures Among Patients with Metabolic Diseases: A Systematic Review and Meta-analysis of Randomized Controlled Trials. High Blood Press Cardiovasc Prev. 2018, 25, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Jorat, M.V.; Tabrizi, R.; Mirhosseini, N.; Lankarani, K.B.; Akbari, M.; Heydari, S.T.; Mottaghi, R.; Asemi, Z. The effects of coenzyme Q10 supplementation on lipid profiles among patients with coronary artery disease: A systematic review and meta-analysis of randomized controlled trials. Lipids Health Dis. 2018, 17, 230. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, M.; Radenkovic, M. A meta-analysis of randomised and placebo controlled clinical trials suggests that Coq10 at low dose improves glucose and HbA1c levels. Nutr. Res. 2017, 38, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Judy, W. Ubiquinone and ubiquinol. In Proceedings of the Eighth Conference of the International CoQ10 Association, Bologna, Italy, 8–11 October 2015. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantle, D.; Hargreaves, I. Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants 2019, 8, 44. https://doi.org/10.3390/antiox8020044
Mantle D, Hargreaves I. Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants. 2019; 8(2):44. https://doi.org/10.3390/antiox8020044
Chicago/Turabian StyleMantle, David, and Iain Hargreaves. 2019. "Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview" Antioxidants 8, no. 2: 44. https://doi.org/10.3390/antiox8020044
APA StyleMantle, D., & Hargreaves, I. (2019). Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants, 8(2), 44. https://doi.org/10.3390/antiox8020044




