Beneficial Effects of Alpha-Lipoic Acid on Hypertension, Visceral Obesity, UCP-1 Expression and Oxidative Stress in Zucker Diabetic Fatty Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Protocols
2.3. Measurement of Metabolic Parameters
2.4. Measurement of Superoxide Anion
2.5. Adipocyte Morphometry
2.6. UCP-1 Protein Immuno-Staining
2.7. Statistical Analysis
3. Results
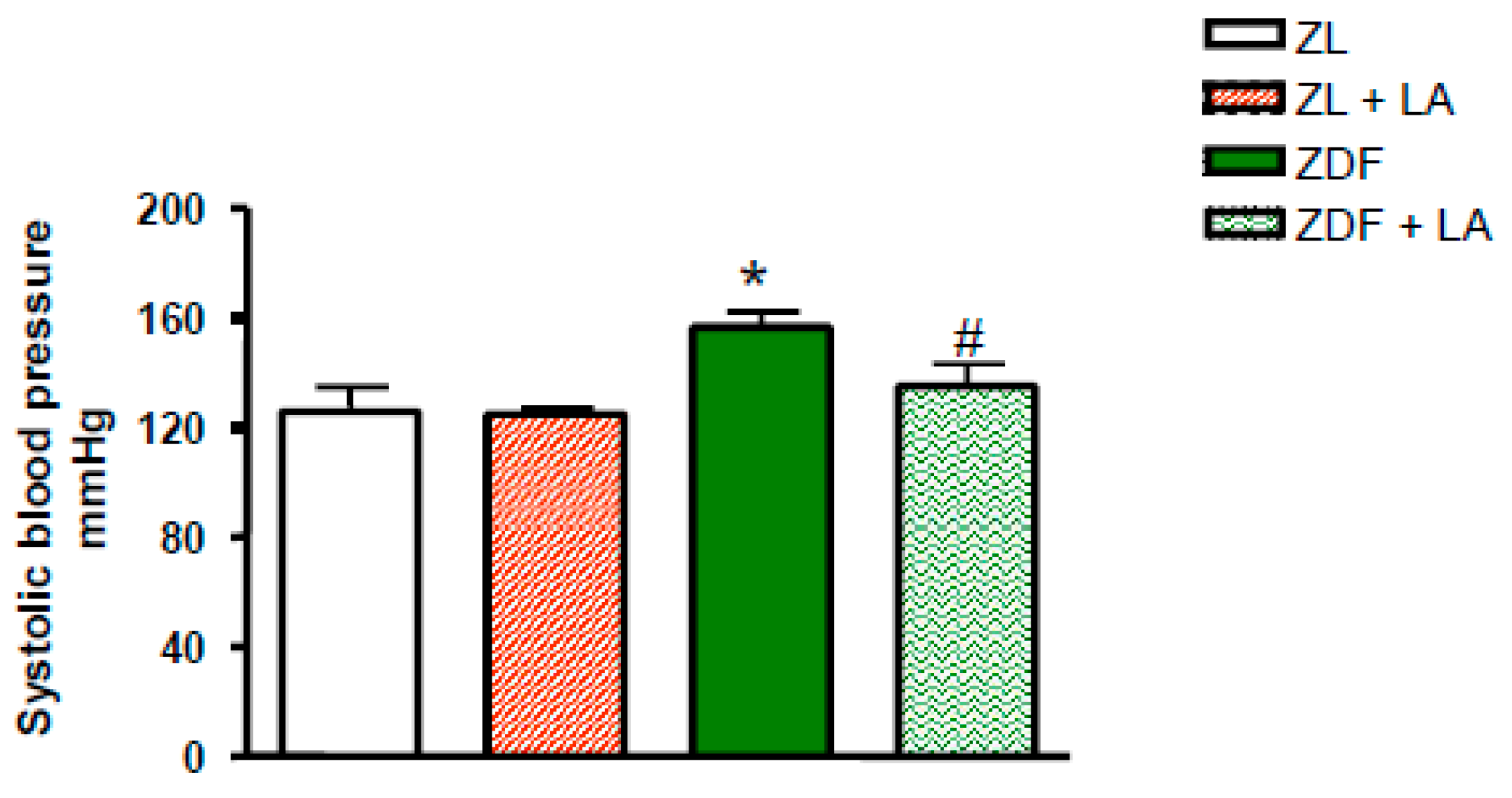
3.1. Blood Pressure
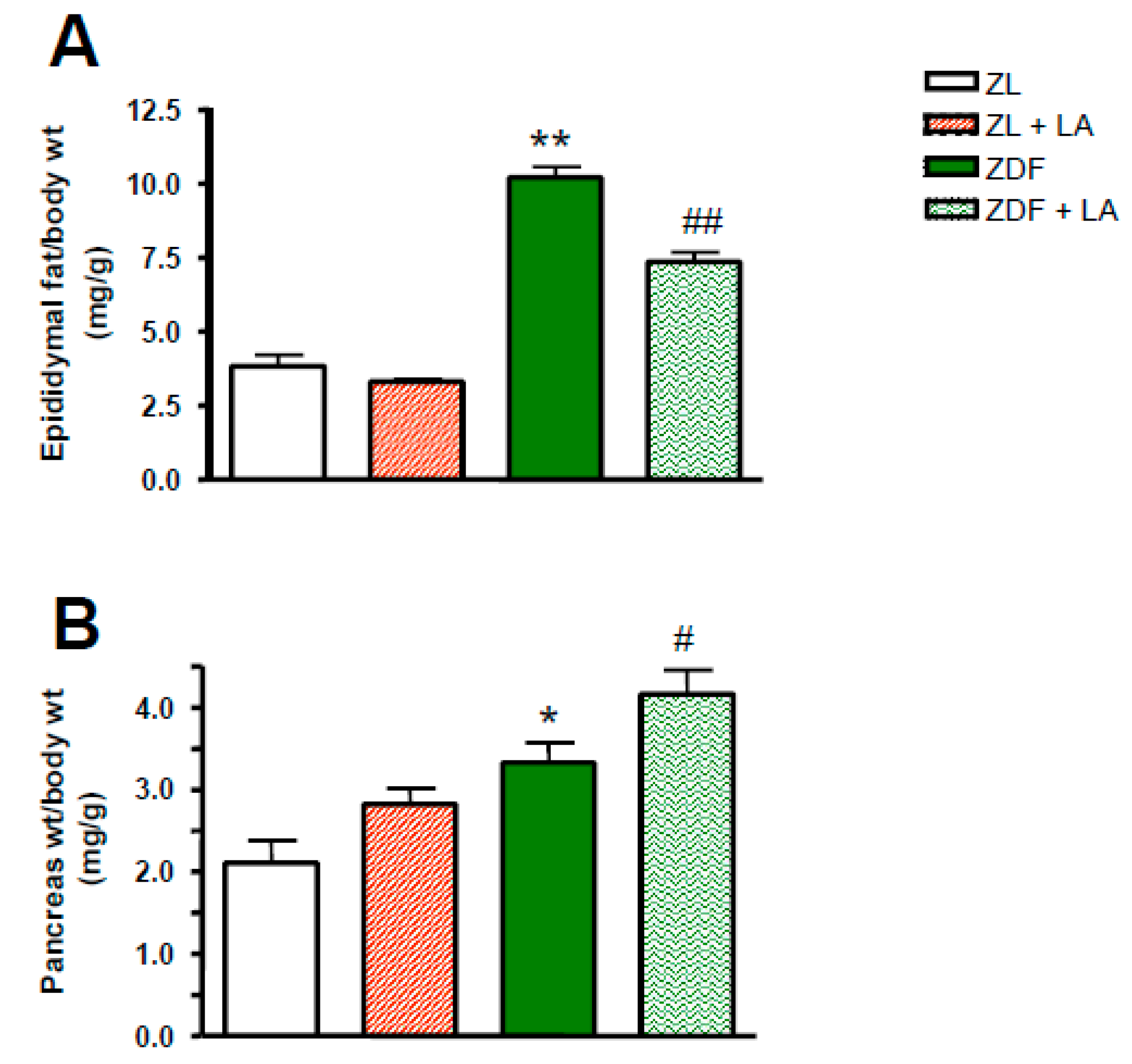
3.2. Body Weight in Relation to the Weight of Epididymis and Pancreas
3.3. Metabolic Parameters
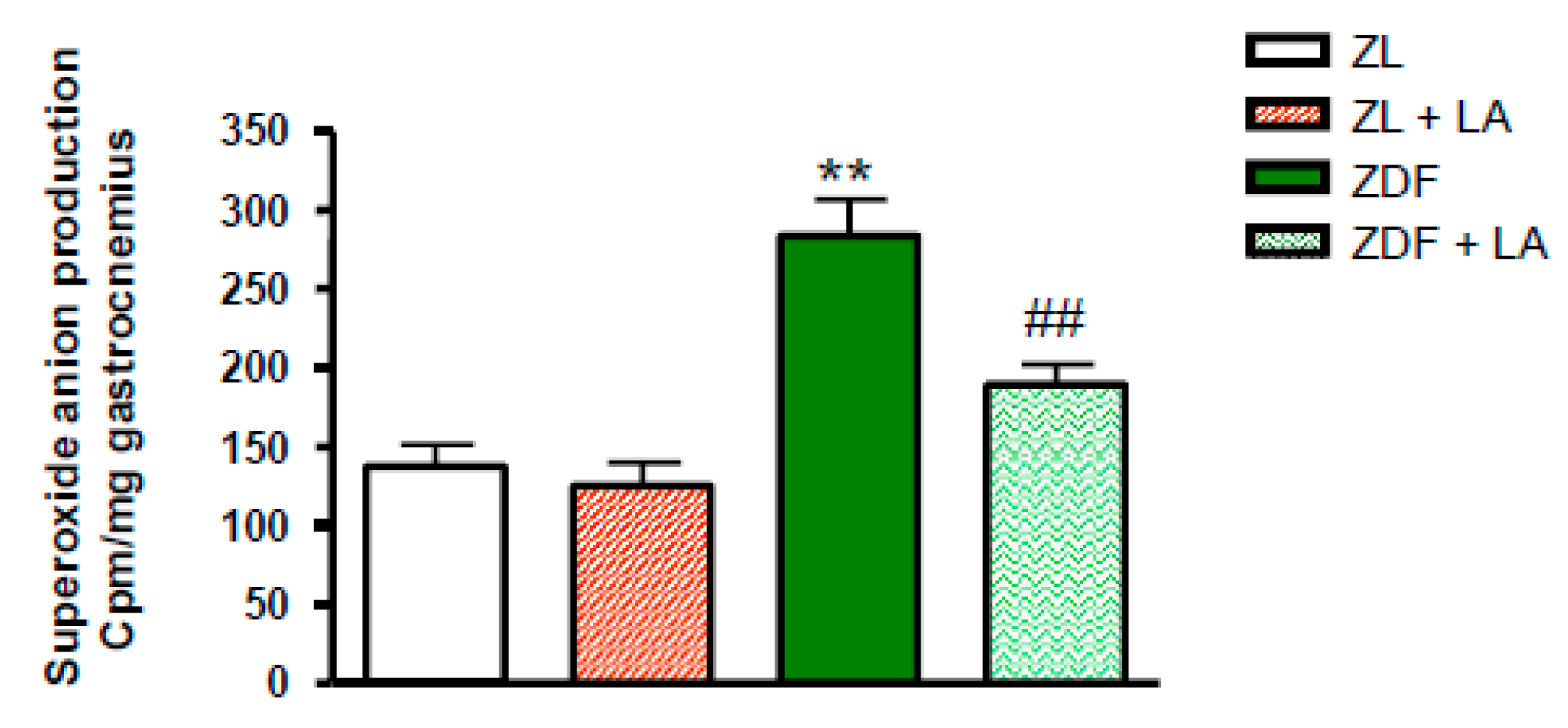
3.4. Oxidative Stress
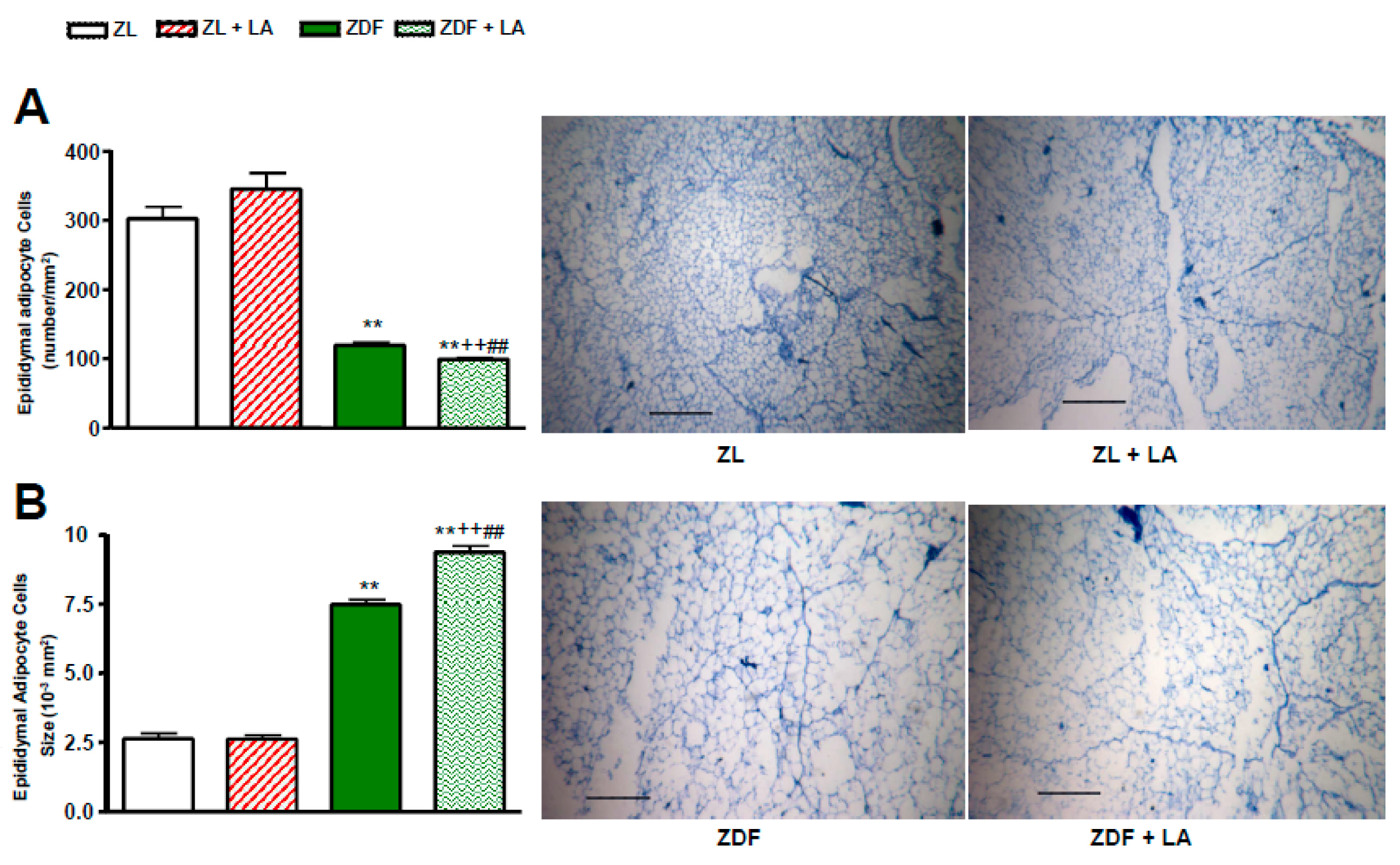
3.5. Number and Size of Epididymal Adipocyte Cells
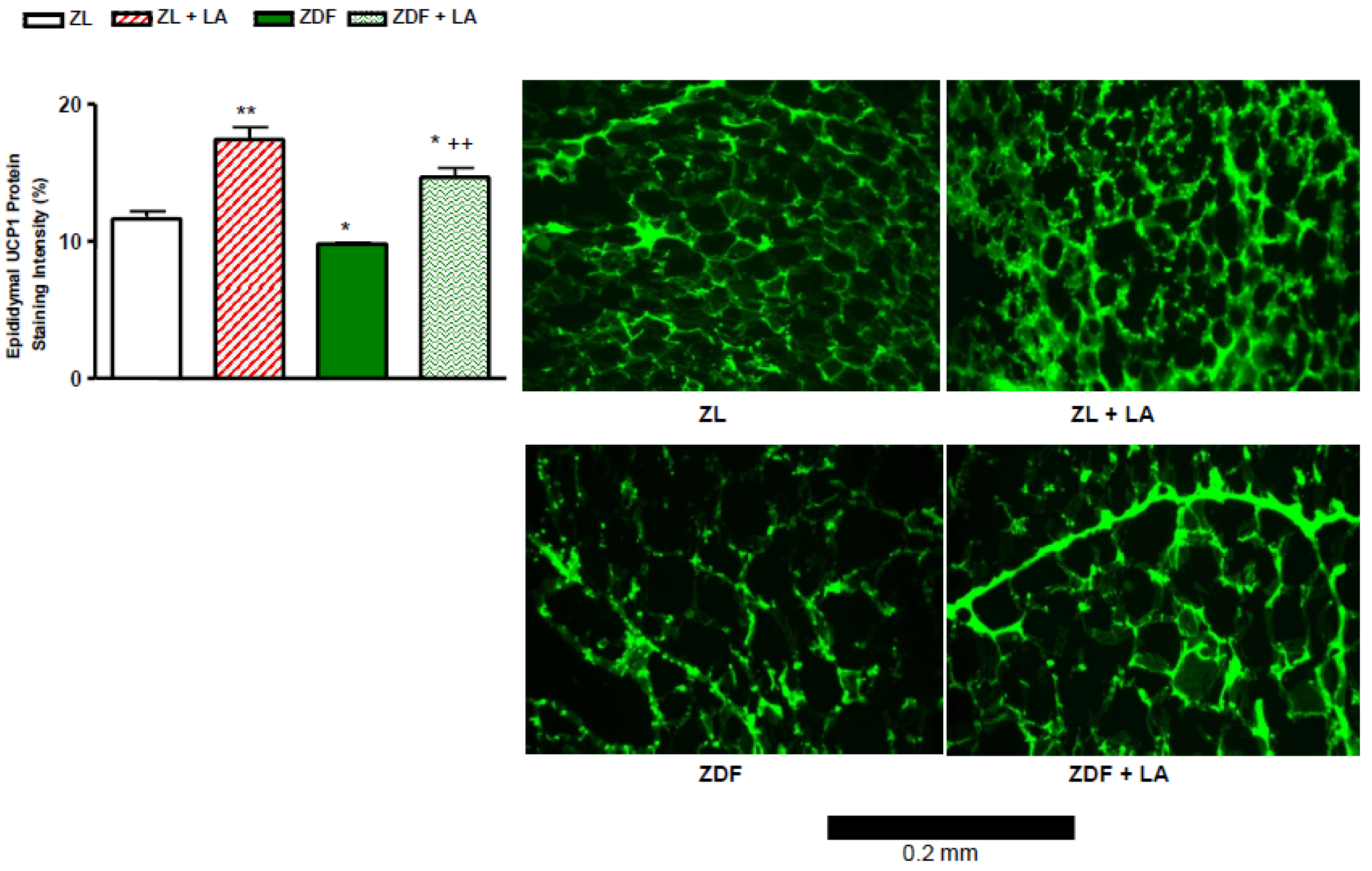
3.6. Epididymal UCP-1 Protein Staining
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data availability
Abbreviations
References
- Baynes, J.W. Role of oxidative stress in development of complications in diabetes. Diabetes 1991, 40, 405–412. [Google Scholar] [CrossRef]
- Jun, T.; Ke-yan, F.; Catalano, M. Increased superoxide anion production in humans: A possible mechanism for the pathogenesis of hypertension. J. Hum. Hypertens. 1996, 10, 305–309. [Google Scholar] [PubMed]
- Ceriello, A.; Motz, E. Isoxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arter. Thromb. Vasc. Biol. 2004, 24, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Urakawa, H.; Katsuki, A.; Sumida, Y.; Gabazza, E.C.; Murashima, S.; Morioka, K.; Maruyama, N.; Kitagawa, N.; Tanaka, T.; Hori, Y.; et al. Oxidative stress is associated with adiposity and insulin resistance in men. J. Clin. Endocrinol. Metab. 2003, 88, 4673–4676. [Google Scholar] [CrossRef] [PubMed]
- El Midaoui, A.; Ismael, M.A.; Lu, H.; Fantus, I.G.; de Champlain, J.; Couture, R. Comparative effects of N-acetyl-L-cysteine and ramipril on arterial hypertension, insulin resistance, and oxidative stress in chronically glucose-fed rats. Can. J. Physiol. Pharm. 2008, 86, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Oltman, C.L.; Davidson, E.P.; Coppey, L.J.; Kleinschmidt, T.L.; Lund, D.D.; Adebara, E.T.; Yorek, M.A. Vascular and neural dysfunction in Zucker diabetic fatty rats: A difficult condition to reverse. Diabetes Obes. Metab. 2008, 10, 64–74. [Google Scholar] [CrossRef]
- Midaoui, A.E.; Talbot, S.; Lahjouji, K.; Dias, J.P.; Fantus, I.G.; Couture, R. Effects of Alpha-Lipoic Acid on Oxidative Stress and Kinin Receptor Expression in Obese Zucker Diabetic Fatty Rats. J. Diabetes Metab. 2015, 6, 1–7. [Google Scholar] [CrossRef]
- Stanford, K.I.; Middelbeek, R.J.; Townsend, K.L.; An, D.; Nygaard, E.B.; Hitchcox, K.M.; Markan, K.R.; Nakano, K.; Hirshman, M.F.; Tseng, Y.H.; et al. Brown adipose tissue regulates glucose homeostasis and insulin sensitivity. J. Clin. Investig. 2013, 123, 215–223. [Google Scholar] [CrossRef]
- Stanford, K.I.; Goodyear, L.J. The therapeutic potential of brown adipose tissue. Hepatobiliary Surg. Nutr. 2013, 2, 286–287. [Google Scholar] [CrossRef]
- Peschechera, A.; Eckel, J. “Browning” of adipose tissue--regulation and therapeutic perspectives. Arch. Physiol. Biochem. 2013, 119, 151–160. [Google Scholar] [CrossRef]
- Petrovic, N.; Walden, T.B.; Shabalina, I.G.; Timmons, J.A.; Cannon, B.; Nedergaard, J. Chronic peroxisome proliferator-activated receptor gamma (PPAR-gamma) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J. Biol. Chem. 2010, 285, 7153–7164. [Google Scholar] [CrossRef] [PubMed]
- Ohno, H.; Shinoda, K.; Spiegelman, B.M.; Kajimura, S. PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell. Metab. 2012, 15, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Vernochet, C.; Peres, S.B.; Davis, K.E.; McDonald, M.E.; Qiang, L.; Wang, H.; Scherer, P.E.; Farmer, S.R. C/EBP-alpha and the corepressors CtBP1 and CtBP2 regulate repression of select visceral white adipose genes during induction of the brown phenotype in white adipocytes by peroxisome proliferator-activated receptor gamma agonists. Mol. Cell. Biol. 2009, 29, 4714–4728. [Google Scholar] [CrossRef]
- Lapa, C.; Arias-Loza, P.; Hayakawa, N.; Wakabayashi, H.; Werner, R.A.; Chen, X.; Shinaji, T.; Herrmann, K.; Pelzer, T.; Higuchi, T. Whitening and Impaired Glucose Utilization of Brown Adipose Tissue in a Rat Model of Type 2 Diabetes Mellitus. Sci. Rep. 2017, 7, 16795. [Google Scholar] [CrossRef] [PubMed]
- Lodge, J.K.; Youn, H.; Handelman, G. Natural sources of lipoic acid: Determination of lipoyllysine released from protease-digested tissues by high performance liquid chromatography incorporating electrochemical detection. J. Appl. Nutr. 1997, 49, 3–11. [Google Scholar]
- Rochette, L.; Ghibu, S.; Richard, C.; Zeller, M.; Cottin, Y.; Vergely, C. Direct and indirect antioxidant properties of α-lipoic acid and therapeutic potential. Mol. Nutr. Food. Res. 2013, 57, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Biewenga, G.P.; Haenen, G.R.; Bast, A. The pharmacology of the antioxidant lipoic acid. Gen. Pharm. 1997, 29, 315–331. [Google Scholar] [CrossRef]
- Podda, M.; Grundmann-Kollmann, M. Low molecular weight antioxidants and their role in skin ageing. Clin. Exp. Derm. 2001, 26, 578–582. [Google Scholar] [CrossRef]
- Midaoui, A.E.; Elimadi, A.; Wu, L.; Haddad, P.S.; de Champlain, J. Lipoic acid prevents hypertension, hyperglycemia, and the increase in heart mitochondrial superoxide production. Am. J. Hypertens. 2003, 16, 173–179. [Google Scholar] [CrossRef]
- Jacob, S.; Streeper, R.S.; Fogt, D.L.; Hokama, J.Y.; Tritschler, H.J.; Dietze, G.J.; Henriksen, E.J. The antioxidant alpha-lipoic acid enhances insulin-stimulated glucose metabolism in insulin-resistant rat skeletal muscle. Diabetes 1996, 45, 1024–1029. [Google Scholar] [CrossRef]
- Song, K.H.; Song, K.H.; Lee, W.J.; Koh, J.M.; Kim, H.S.; Youn, J.Y.; Park, H.S.; Koh, E.H.; Kim, M.S.; Youn, J.H.; et al. Alpha-Lipoicacidpreventsdiabetes mellitus in diabetes-proneobeserats. Biochem. Biophys. Res. Commun. 2005, 326, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Seo, E.Y.; Ha, A.W.; Kim, W.K. α-Lipoic acid reduced weight gain and improved the lipid profile in rats fed with high fat diet. Nutr. Res. Pract. 2012, 6, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, P.; Del Guerra, S.; Marselli, L.; Lupi, R.; Masini, M.; Pollera, M.; Bugliani, M.; Boggi, U.; Vistoli, F.; Mosca, F.; et al. Pancreatic islets from type 2 diabetic patients have functional defects and increased apoptosis that are ameliorated by metformin. J. Clin. Endocrinol. Metab. 2004, 89, 5535–5541. [Google Scholar] [CrossRef] [PubMed]
- El Midaoui, A.; Wu, L.; Wang, R.; de Champlain, J. Modulation of cardiac and aortic peroxisome proliferator-activated receptor-gamma expression by oxidative stress in chronically glucose-fed rats. Am. J. Hypertens. 2006, 19, 407–412. [Google Scholar] [CrossRef][Green Version]
- El Midaoui, A.; Lungu, C.; Wang, H.; Wu, L.; Robillard, C.; Deblois, D.; Couture, R. Impact of α-lipoic acid on liver peroxisome proliferator-activated receptor-α, vascular remodeling, and oxidative stress in insulin-resistant rats. Can. J. Physiol. Pharmacol. 2011, 89, 743–751. [Google Scholar] [CrossRef]
- Sato, N.; Kaneko, M.; Tamura, M.; Kurumatani, H. The prostacyclin analog beraprost sodium ameliorates characteristics of metabolic syndrome in obese Zucker (fatty) rats. Diabetes 2010, 59, 1092–1100. [Google Scholar] [CrossRef]
- El Midaoui, A.; Haddad, Y.; Filali-Zegzouti, Y.; Couture, R. Argan Oil as an Effective Nutri-Therapeutic Agent in Metabolic Syndrome: A Preclinical Study. Int. J. Mol. Sci. 2017, 18, 2492. [Google Scholar] [CrossRef]
- Ohara, Y.; Peterson, T.E.; Harrison, D.G. Hypercholesterolemia increases endothelial superoxide anion production. J. Clin. Investig. 1993, 91, 2546–2551. [Google Scholar] [CrossRef]
- El Akoum, S.; Haddad, Y.; Couture, R. Impact of pioglitazone and bradykinin type 1 receptor antagonist on type 2 diabetes in high-fat diet-fed C57BL/6J mice. Obes. Sci. Pract. 2017, 3, 352–362. [Google Scholar] [CrossRef]
- Hunt, J.V.; Wolff, S.P. Oxidative glycation and free radical production: a causal mechanism of diabetic complications. Free. Radic. Res. Commun. 1991, 12–13, 115–123. [Google Scholar] [CrossRef]
- Gopaul, N.K.; Manraj, M.D.; Hebe, A.; Yan, S.L.K.; Johnston, A.; Carrier, M.J.; Anggård, E.E. Oxidative stress could precede endothelial dysfunction and insulin resistance in Indian Mauritians with impaired glucose metabolism. Diabetologia 2001, 44, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Estrada, D.E.; Ewart, H.S.; Tsakiridis, T.; Volchuk, A.; Ramlal, T.; Tritschler, H.; Klip, A. Stimulation of glucose uptake by the natural coenzyme alpha-lipoic acid/thioctic acid: Participation of elements of the insulin signaling pathway. Diabetes 1996, 45, 1798–1804. [Google Scholar] [CrossRef] [PubMed]
- El Midaoui, A.; de Champlain, J. Prevention of hypertension, insulin resistance, and oxidative stress by alpha-lipoic acid. Hypertension 2002, 39, 303–330. [Google Scholar] [CrossRef] [PubMed]
- Streeper, R.S.; Henriksen, E.J.; Jacob, S.; Hokama, J.Y.; Fogt, D.L.; Tritschler, H.J. Differential effects of lipoic acid stereoisomers on glucose metabolism in insulin-resistant skeletal muscle. Am. J. Physiol. 1997, 273, E185–E191. [Google Scholar] [CrossRef]
- Saengsirisuwan, V.; Perez, F.R.; Sloniger, J.A.; Maier, T.; Henriksen, E.J. Interactions of exercise training and alpha-lipoic acid on insulin signaling in skeletal muscle of obese Zucker rats. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E529–E536. [Google Scholar] [CrossRef]
- El Midaoui, A.; de Champlain, J. Effects of glucose and insulin on the development of oxidative stress and hypertension in animal models of type 1 and type 2 diabetes. J. Hypertens. 2005, 23, 581–588. [Google Scholar] [CrossRef]
- Jacob, S.; Ruus, P.; Hermann, R.; Tritschler, H.J.; Maerker, E.; Renn, W.; Augustin, H.J.; Dietze, G.J.; Rett, K. Oral administration of RAC-alpha-lipoic acid modulates insulin sensitivity in patients with type-2 diabetes mellitus: A placebo-controlled pilot trial. Free. Radic. Biol. Med. 1999, 27, 309–314. [Google Scholar] [CrossRef]
- Jacob, S.; Henriksen, E.J.; Schiemann, A.L.; Simon, I.; Clancy, D.E.; Tritschler, H.J.; Jung, W.I.; Augustin, H.J.; Dietze, G.J. Enhancement of glucose disposal in patients with type 2 diabetes by alpha-lipoic acid. Arzneimittelforschung 1995, 45, 872–874. [Google Scholar]
- Kocak, G.; Aktan, F.; Canbolat, O.; Ozogul, C.; Elbeg, S.; Yildizoglu-Ari, N. Alpha-lipoic acid treatment ameliorates metabolic parameters, blood pressure, vascular reactivity and morphology of vessels already damaged by streptozotocin-diabetes. Diabetes. Nutr. Metab. 2000, 13, 308–318. [Google Scholar]
- Vessières, E.; Guihot, A.L.; Toutain, B.; Maquigneau, M.; Fassot, C.; Loufrani, L.; Henrion, D. COX-2-derived prostanoids and oxidative stress additionally reduce endothelium-mediated relaxation in old type 2 diabetic rats. PLoS ONE 2013, 8, e68217. [Google Scholar] [CrossRef]
- Muellenbach, E.A.; Diehl, C.J.; Teachey, M.K.; Lindborg, K.A.; Archuleta, T.L.; Harrell, N.B.; Andersen, G.; Somoza, V.; Hasselwander, O.; Matuschek, M.; et al. Interactions of the advanced glycation end product inhibitor pyridoxamine and the antioxidant alpha-lipoic acid on insulin resistance in the obese Zucker rat. Metabolism 2008, 57, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Zalba, G.; Beaumont, F.J.; San José, G.; Fortuño, A.; Fortuño, M.A.; Etayo, J.C.; Díez, J. Vascular NADH/NADPH oxidase is involved in enhanced superoxide production in spontaneously hypertensive rats. Hypertension 2000, 35, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Beswick, R.A.; Dorrance, A.M.; Leite, R.; Webb, R.C. NADH/NADPH oxidase and enhanced superoxide production in the mineralocorticoid hypertensive rat. Hypertension 2001, 38, 1107–1111. [Google Scholar] [CrossRef] [PubMed]
- Fukui, T.; Ishizaka, N.; Rajagopalan, S.; Laursen, J.B.; Capers, Q., IV; Taylor, W.R.; Harrison, D.G.; de Leon, H.; Wilcox, J.N.; Griendling, K.K. p22phox mRNA expression and NADPH oxidase activity are increased in aortas from hypertensive rats. Circ. Res. 1997, 80, 45–51. [Google Scholar] [CrossRef]
- Pashaj, A.; Yi, X.; Xia, M.; Canny, S.; Riethoven, J.J.; Moreau, R. Characterization of genome-wide transcriptional changes in liver and adipose tissues of ZDF (fa/fa) rats fed R-α-lipoic acid by next-generation sequencing. Physiol. Genom. 2013, 45, 1136–1143. [Google Scholar] [CrossRef]
- Fernández Vázquez, G.; Reiter, R.J.; Agil, A. Melatonin increases brown adipose tissue mass and function in Zücker diabetic fatty rats: Implications for obesity control. J. Pineal Res. 2018, 64, e12472. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor- alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef]
- Blüher, M. Adipose tissue dysfunction in obesity. Exp. Clin. Endocrinol. Diabetes 2009, 117, 241–250. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Tian, Y.F.; Hsieh, C.H.; Hsieh, Y.J.; Chen, Y.T.; Peng, Y.J.; Hsieh, P.S. α-Lipoic acid prevents mild portal endotoxaemia-induced hepatic inflammation and β cell dysfunction. Eur. J. Clin. Investig. 2012, 42, 637–648. [Google Scholar] [CrossRef]
- Van Bree, B.W.; Lenaers, E.; Nabben, M.; Briedé, J.J.; Jörgensen, J.A.; Schaart, G.; Schrauwen, P.; Hoeks, J.; Hesselink, M.K. A genistein-enriched diet neither improves skeletal muscle oxidative capacity nor prevents the transition towards advanced insulin resistance in ZDF rats. Sci. Rep. 2016, 6, 22854. [Google Scholar] [CrossRef] [PubMed]
| Metabolic Parameters | ZL | ZL + LA | ZDF | ZDF + LA |
|---|---|---|---|---|
| Final body weight(g) | 312 ± 6 | 305 ± 6 | 365 ± 9 ** | 326 ± 5 ## |
| Plasma glucose (mmol/L) | 5.8 ± 0.4 | 5.3 ± 0.3 | 16.8 ± 1.8 ** | 12.9 ± 1.7 ** |
| Plasma insulin (pmol/L) | 471.5 ± 36.5 | 520.6 ± 130.2 | 2098.9 ± 313.5 ** | 2087.0 ± 253.9 ** |
| HOMA | 18.0 ± 1.9 | 17.5 ± 4.3 | 271.9 ± 30.9 ** | 182.4 ± 50.9 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Midaoui, A.; Fantus, I.G.; Ait Boughrous, A.; Couture, R. Beneficial Effects of Alpha-Lipoic Acid on Hypertension, Visceral Obesity, UCP-1 Expression and Oxidative Stress in Zucker Diabetic Fatty Rats. Antioxidants 2019, 8, 648. https://doi.org/10.3390/antiox8120648
El Midaoui A, Fantus IG, Ait Boughrous A, Couture R. Beneficial Effects of Alpha-Lipoic Acid on Hypertension, Visceral Obesity, UCP-1 Expression and Oxidative Stress in Zucker Diabetic Fatty Rats. Antioxidants. 2019; 8(12):648. https://doi.org/10.3390/antiox8120648
Chicago/Turabian StyleEl Midaoui, Adil, I. George Fantus, Ali Ait Boughrous, and Réjean Couture. 2019. "Beneficial Effects of Alpha-Lipoic Acid on Hypertension, Visceral Obesity, UCP-1 Expression and Oxidative Stress in Zucker Diabetic Fatty Rats" Antioxidants 8, no. 12: 648. https://doi.org/10.3390/antiox8120648
APA StyleEl Midaoui, A., Fantus, I. G., Ait Boughrous, A., & Couture, R. (2019). Beneficial Effects of Alpha-Lipoic Acid on Hypertension, Visceral Obesity, UCP-1 Expression and Oxidative Stress in Zucker Diabetic Fatty Rats. Antioxidants, 8(12), 648. https://doi.org/10.3390/antiox8120648






