1. Introduction
Atherosclerosis is considered a chronic inflammatory process and involves a complex pathophysiological effect, including endothelial dysfunction, low-density lipoprotein (LDL) oxidation, foam cell formation, and vascular smooth muscle cell (VSMC) proliferation and migration at different stages of this disease [
1,
2]. Elevated plasma LDL concentration contributes to the initiation of atherosclerosis [
3]. Oxidized LDL triggers endothelial cells to release chemokines in contribution to recruitment of monocytes, resulting in the transformation of the lipid-laden macrophages into foam cells [
3]. In the lesion progression, these activated macrophages still secrete proinflammatory cytokines, especially tumor necrosis factor-alpha (TNF-α), which enhances VSMC migration and proliferation [
1,
3]. Subsequently, VSMC transforms and proliferates into foam cells, and thus the accumulation of foam cells leading to fatty streaks results in the formation of atherosclerotic plaques [
2]. Thus, inhibition of abnormal VSMC migration and proliferation is an attractive strategy for clinical therapy of atherosclerosis and restenosis after percutaneous coronary interventions.
VSMC is normally quiescent, but upon vascular injury, it transforms into a more synthetic phenotype with progressively increasing capacity for activation, proliferation, and migration [
1,
4]. In the atherosclerotic process, VSMC migrates from the media to the intima, forms the neointima progressively with abundant levels of extracellular matrix (ECM) proteins, and then eventually leads to plaque formation [
2]. Identification of key proteins involved in the process, such as matrix metalloproteinases (MMPs), is vital for understanding atherosclerosis and devising new therapies. MMPs are a subfamily of the metzincin superfamily of endogenous proteinases that break down components of ECM. Among them, the gelatinases (MMP-2 and MMP-9) degrade efficiently native collagen types IV and laminin, and promote a VSMC migratory phenotype [
5]. Moreover, the gene expression of MMPs is majorly regulated by the transcriptional factors, such as activator protein-1 (AP-1) or nuclear factor-kappaB (NF-κB) through the serine/threonine protein kinase PKB (also known as Akt) or extracellular signal-regulated kinase (ERK) pathways, or by the MMP protein activators or inhibitors. One review study concluded that oxidative stress could enhance MMP activity and expression [
6], and recent studies further indicate that MMP-mediated ECM remodeling is modulated by reactive oxygen species (ROS) [
7]. Hence, MMPs and their regulatory signaling have been considered as promising targets for anti-atherosclerotic agents [
8].
In arterial media, VSMC is at low proliferative indices (<0.05%) and remains in the G0/G1 phase of the cell cycle [
4]. However, VSMC re-enters into the cell cycle from the quiescent state to proliferate under the stimulation of several cytokines in pathological processes, which plays an important role in the development of atherosclerosis [
1]. VSMC begins to divide in response to cytokines, exits the G1 phase, and then enters the S phase. During the G1/S transition, cyclin D1/cyclin-dependent kinase (cdk) 4 and cyclin E/cdk2 complexes are required. The complexes participate in the hyperphosphorylation of retinoblastoma (Rb) tumor suppressor, leading phosphorylated Rb (p-Rb) to release E2F transcription factor, allowing the cells to progress into S phase [
9]. The kinase activities of these cyclin/cdk complexes are regulated by cdk inhibitors (cki), including p16, p21, and p27. The gatekeeper of the mammalian cell cycle, p53, plays a key role in controlling G0/G1 arrest through its downstream factor, such as p21 [
10].
Previous studies have reported that
Hibiscus leaf, an edible part of
H. sabdariffa Linne (
Malvaceae) [
11], possesses hypoglycemic [
12], hypolipidemic [
13,
14], and antioxidant [
13,
15] effects, as demonstrated by various experimental models (
Table S1). For the standardization of each extract, our studies also indicated that (–)-epicatechin gallate (ECG; 16.5 ± 5.6%) was identified to be present in the highest level in
Hibiscus leaf polyphenols (HLPs), followed by ellagic acid (EA; 10.31 ± 3.43%) and catechin (Cat; 7.4 ± 2.6%), and traces of only quercetin (Que; 0.8 ± 0.4%) and ferulic acid (FA; 0.7 ± 0.3%) were detected (
Table S2) [
14]. In this regard, the aqueous and methanol extracts of
H. sabdariffa leaves showed anti-atherogenic effects in hyperlipidemia animals induced by cholesterol [
11,
12], and inhibited foam cell formation, as well as protected endothelial cells from injury in vitro [
11,
16]. Our recent studies also revealed that
Hibiscus leaf aqueous extract, due to its high content in polyphenols, has apoptotic and anti-migratory effects on prostate cancer cells [
17,
18]. However, little information is available on the isolation and characterization of a polyphenolic extract of
H. sabdariffa leaves. In the present study, HLP was partially characterized by biochemical and spectroscopic assays, and evaluated for the ability to inhibit TNF-α-stimulated VSMC dysfunction.
Many studies have indicated that plant-derived polyphenols have various pharmacological and biological effects, such as antioxidant, anti-inflammatory, anti-hyperlipidemia, anti-diabetes, anti-atherogenic, and anti-tumor abilities [
19]. Furthermore, although the protective effects of HLPs on endothelial cells and macrophages have been demonstrated previously, the in vivo function and the molecular target of HLPs on VSMC remain to be elucidated in cardiovascular microenvironment. Using a model of VSMC exposed to TNF-α and the well-established atherosclerotic rabbit experiment, to our knowledge, this is the first report revealing the TNF-α-antagonist potential of HLPs in vitro and in vivo.
2. Materials and Methods
2.1. Preparation of HLP and Detection of Polyphenolic Compounds
One hundred grams of
H. sabdariffa L. (Malvaceae) dried leaves, obtained from Taitung City, Taitung Country, Taiwan, were extracted three times with methanol (300 mL) at 50 °C for 3 h, and the samples were filtered after each extraction. The methanol was evaporated under reduced pressure, and the residue was dissolved in 500 mL of distilled water at 50 °C and extracted with 200 mL of hexane to remove pigments. The aqueous phase was extracted three times with 180 mL of ethyl acetate, and the solvent was removed from the extract with a vacuum rotary evaporator. The residue was re-dissolved in 250 mL of distilled water and was lyophilized to obtain about 2.5 g of HLP. The polyphenolic components of HLP were further analyzed as follows. All reagents and pure compounds were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). Total phenolic acid content was determined by the Folin-Ciocalteau method [
20] using gallic acid (GA) as a standard. To start, 0.1 mg of HLP was first dissolved in a tube with 1 mL of distilled water, and 0.5 mL of Folin-Ciocalteu reagent (2 N) was added and mixed thoroughly. After 3 min, 3 mL of Na
2CO
3 solution (2%) was added, and the mixture was allowed to stand for 15 min. The absorbance of the mixture at 750 nm was measured on a spectrophotometer (Beckman Coulter DU 730, Brea, CA, USA). The concentration of total flavonoid was assayed according to the Jia method [
21]. A standard curve using rutin (Rut) was also prepared. Next, 0.5 mL of HLP (1 mg/mL) was diluted with 1.25 mL of distilled water. Afterwards, 75 μL of NaNO
2 solution (5%) was added to the mixture. After an interval of 6 min, 150 μL of AlCl
3·6H
2O solution (10%) was added, and the mixture was allowed to stand for another 5 min. Then, 0.5 mL of NaOH (1 M) and 2.5 mL of distilled water were added. The solution was mixed, and the absorbance was immediately measured against the prepared control at 510 nm. The polyphenolic components of HLP were confirmed by high performance liquid chromatography (HPLC) system using a Hewlett-Packard Vectra 436/33 N system with a diode array detector (all from Waters Corp., Milford, MA, USA). The HLP was filtered through a 0.45 μm filter disc, and then 20 μL of HLP was injected onto a 5 μm RP-18 column (4.6 × 150 mm i.d.; Phenomenex, Inc., Torrance, CA, USA). The mobile phase contained two solvents, including solvent A (formic acid/water = 10:90) and solvent B (formic acid/acetonitrile/water = 10:30:60), run by a linear gradient method at room temperature as follows: From 10% solvent B to 40% solvent B (flow rate = 1.0 mL/min) over 25 min. The chromatography was monitored at 240 and 345 nm, and an ultraviolet (UV) spectrum (Beckman Coulter Inc., Brea, CA, USA) was collected to confirm peak purity. The HPLC analysis of 10 kinds of standard polyphenols showed the retention times (RT) as follows: GA (4.58 min), protocatechuic acid (PCA, 7.50 min), Cat (9.39 min), ECG (11.21 min), EA (13.29 min), Rut (14.01 min), ρ-coumaric acid (CA, 14.44 min), FA (15.28 min), Que (21.57 min), and naringenin (Nar, 24.48 min), respectively. Consistent with our previous study [
16], the yield of HLP was approximately 25.0%, and polyphenols were indeed present in HLP (
Table S2).
2.2. Cell Culture
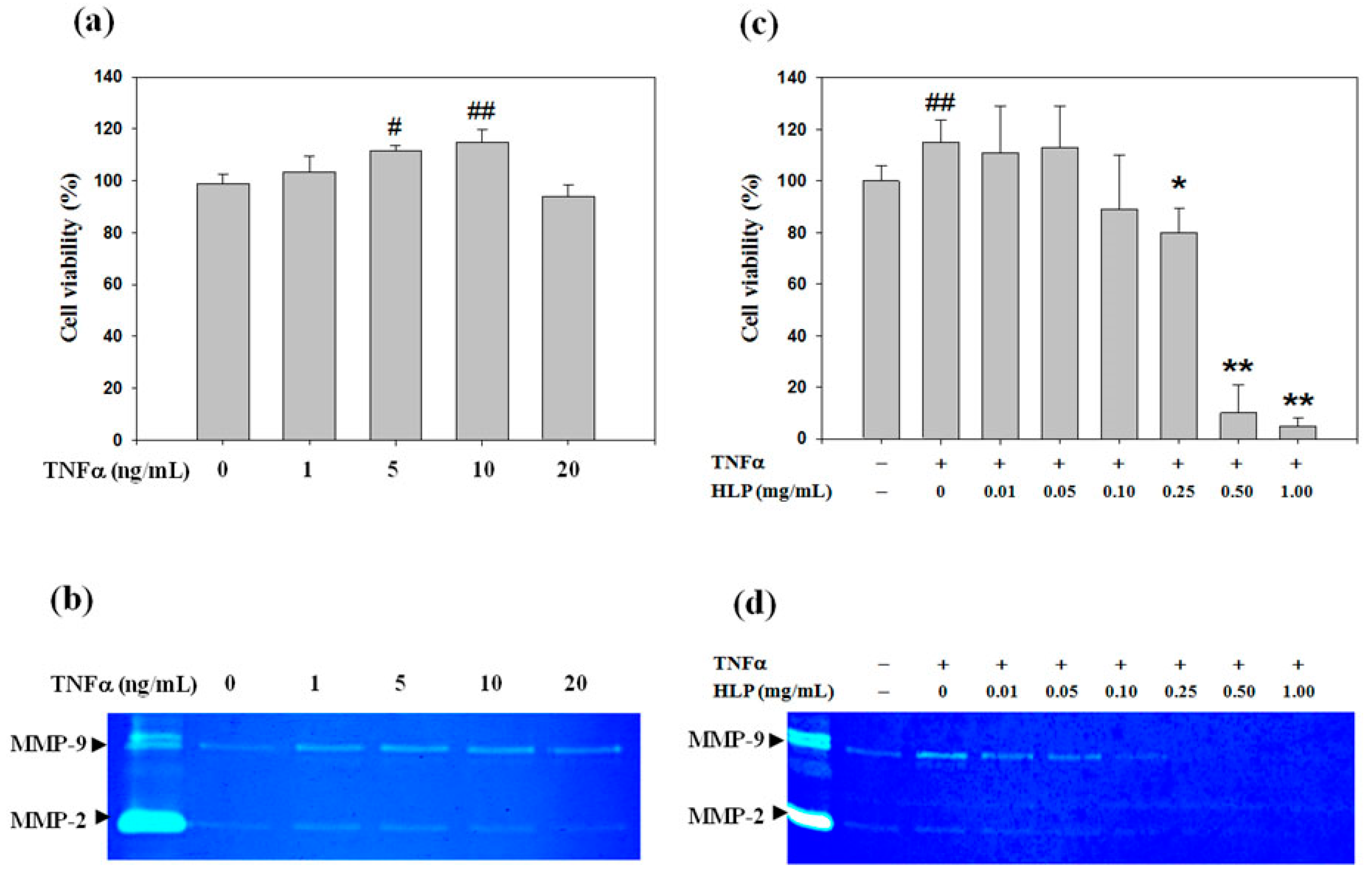
A rat VSMC cell line A7r5 was purchased from the Bioresource Collection and Research Center. A7r5 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 1% penicillin-streptomycin mixed antibiotics, 1% glutamine, and 1.5 g/L sodium bicarbonate (all regents from Hyclone, Logan, Utah, USA). All cell cultures were maintained at 37 °C under 95% moisturized air with 5% CO2. Before cell treatments, A7r5 cells were seeded onto each 60 mm Petri dish (Corning Inc, Corning, NY, USA) at a density of 105 for 24 h. For induction of VSMC dysfunction, A7r5 cells at 70% confluence were serum-starved for 24 h and treated with TNF-α (10 ng/mL; Sigma-Aldrich, St Louis, MO, USA) for 24 h.
2.3. 3-(4,5-Dimethylthiazol-Zyl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay
A7r5 cells were seeded onto each 24-well plate (Corning Inc, Corning, NY, USA) at a density of 10
5 cells/mL, and treated with various concentrations of TNF-α (0–20 ng/mL) alone or TNF-α (10 ng/mL) in combination with HLP (0–1.0 mg/mL) for 24 or 48 h. Thereafter, the medium was changed, MTT (0.1 mg/mL, Sigma, St. Louis, MO, USA) was added for next 4-h incubation. The viable cell number was directly proportional to the formazan production, which was solubilized in isopropanol and detected at 563 nm with a spectrophotometer. The MTT assay was used to evaluate the effect of TNF-α alone or TNF-α and/or HLP on cell viability, and to determine the non-cytotoxic doses of HLP, as described by Chen et al. [
16].
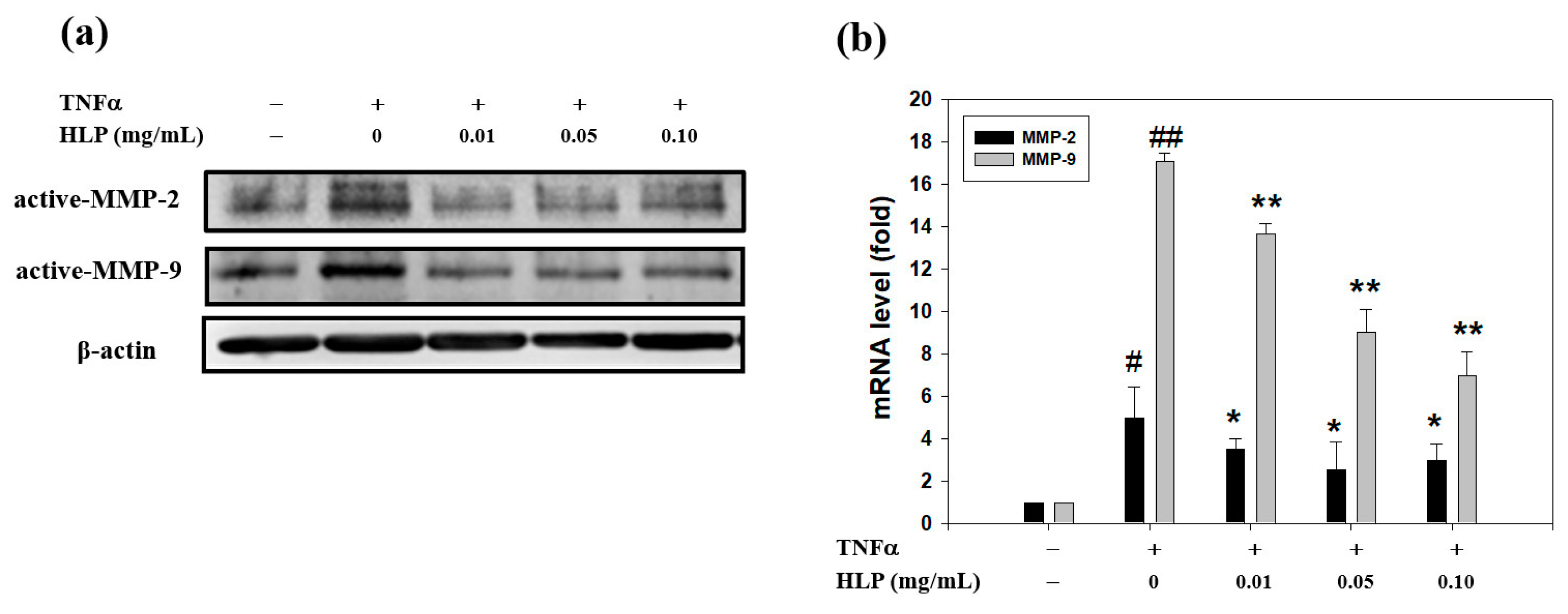
2.4. Gelatin Zymography Protease Assay
The activities of MMP-2 and MMP-9 in the serum-free conditioned medium were evaluated by gelatin zymography according to a previously described method by Huang et al. [
22]. In short, samples were prepared with standard sodium dodecyl sulfate (SDS)-gel loading buffer containing 0.01% SDS (Sigma-Aldrich, St Louis, MO, USA). The prepared samples (25 μg total protein) were not boiled before loading, but subjected to electrophoresis on 8% SDS polyacrylamide gels (1.0-mm-thick, acrylamide/bis-acrylamide = 30/1.2) containing 0.1% gelatin (Sigma-Aldrich, St Louis, MO, USA). After electrophoresis, the gel was washed twice with 100 mL distilled water containing 2% Triton X-100 (Sigma-Aldrich, St Louis, MO, USA) on a shaker for 30 min at room temperature to remove SDS, and incubated in 100 mL reaction buffer (0.02% NaN
3, 10 mM CaCl
2 and 40 mM Tris-HCl (pH 8.0)) at 37 °C for 12 h. The gel was further stained with Coomassie brilliant blue R-250 dye (Abcam plc, Cambridge, UK) followed by destaining with methanol/acetic acid/water (50:75:875,
v/v/v).
2.5. Real-Time Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Total RNAs were extracted using a TRIzol reagent (Invitrogen, Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions, as described by Chiu et al. [
18]. In general, the mRNA levels were analyzed by quantitative real-time RT-PCR using a Bio-Rad iCycler system (Bio-Rad, Hercules, CA, USA), and normalized to the housekeeping gene, β-actin. The sequences of primers (MDBio Inc., Taipei, Taiwan) used in the experiments are listed in
Table S3.
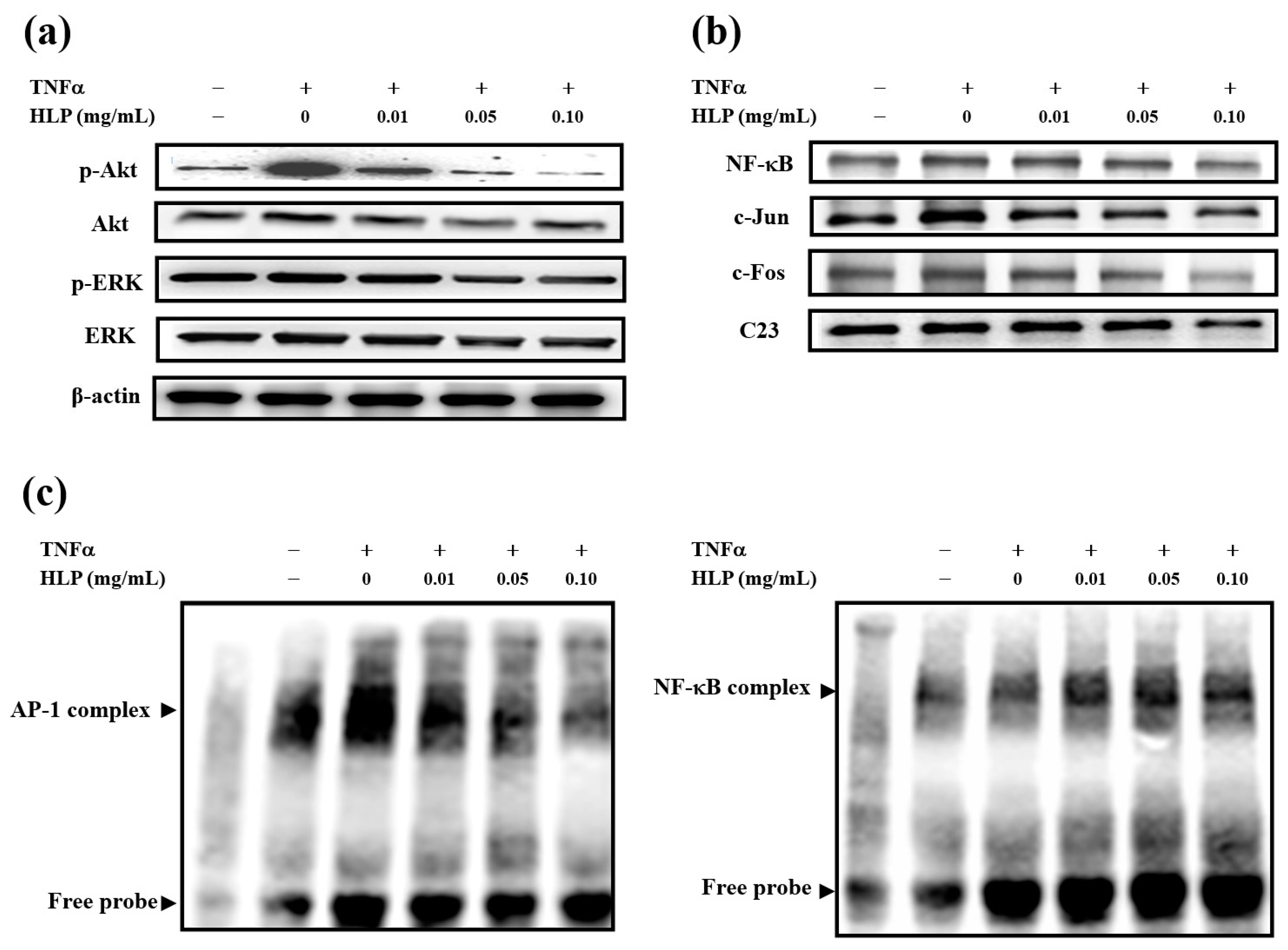
2.6. Protein Isolation and Western Blotting
The preparation of cytosolic and nuclear fractions of the cells was performed using the Nuclear and Cytoplasmic Extraction Reagent Kit (Thermo Scientific, Rockford, IL, USA), described by Chiu et al. [
18]. In brief, the harvested cells were washed with phosphate-buffered saline (PBS) and incubated on ice in Reagent A for 2 min. Reagent B was added, and the mixture was further incubated on ice for 5 min. Reagent C was added, and the contents were mixed by inverting the tube several times, followed by centrifugation (700×
g) at 4 °C for 10 min. The supernatant (cytosol) was collected and centrifuged (12,000×
g) at 4 °C for 15 min. Then, the nuclear pellet was washed twice with wash buffer (10 mM Tris-HCl (pH 7.5), 0.4% Nonidet P-40, and 10 mM KCl) to remove non-lysed cells. A protease inhibitor cocktail (Bio-Rad Labs., Hercules, CA, USA) was added to all solutions before use. Western blot analysis was carried out according to a previously described method by Chen et al. [
16]. Whole cell lysate was prepared using sample buffer containing 2% SDS, 10% glycerol, 5% β-mercaptoethanol, and 50 mM Tris-HCl (pH 6.8), and then extracted using sonication. Equal amounts of proteins were separated by 8–15% SDS–polyacrylamide gels and transferred to nitrocellulose membranes (Bio-Rad Labs., Hercules, CA, USA). In order to block non-specific binding, the nitrocellulose membranes were incubated with 5% nonfat dry milk for 1–2 h at 4 °C, and then overnight with polyclonal first antibodies against MMP-2, MMP-9, p-Akt, Akt, p-ERK, ERK, c-Jun, c-Fos, NF-κB, p-p53, p53, p21, p27, p16, PCNA (proliferating cell nuclear antigen), E2F, and p-Rb were from Santa Cruz Biotech (CA, USA). In the subsequent day, the blots were incubated with the appropriate horseradish peroxidase-conjugated secondary antibodies (goat anti-rabbit IgG or goat anti-mouse IgG), from Sigma-Aldrich (St Louis, MO, USA), for 1 h, and detection was performed using an enhanced chemiluminescence (ECL) reagent (Amersham, Arlington Heights, IL, USA). The cytosolic and nuclear protein were respectively determined by Western blotting using anti-β-actin and anti-C23 antibodies, purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA),as loading controls. Protein level was quantified by densitometry using FUJIFILM-Multi Gauge V2.2 software (Fujifilm, Kyoto, Japan).
2.7. AP-1 and NF-κB Binding Assay
DNA-binding activities of AP-1 and NF-κB in nuclear extracts were assayed by electrophoretic mobility shift assay (EMSA) with biotin-labeled double-stranded AP-1 or NF-κB oligonucleotides (MDBio Inc., Taipei, Taiwan), as described by Chiu et al. [
18]. EMSA was carried out by using the Lightshift kit from Pierce (Rockford, IL, USA). Binding reactions containing 10 μg of nuclear extracts, 1 μg poly (dI·dC), 12.5 μg poly-
l-lysine, 2 pmol of oligonucleotide probe, and 2 μL of 10× binding buffer were incubated for 20 min at room temperature. Protein-DNA complexes were separated by electrophoresis on a 6% non-denaturing acrylamide gel, transferred to positively charged nylon membranes (Millipore, Bedford, MA, USA), and then UV cross-linked. Gel shifts were visualized with a streptavidin-horseradish peroxidase followed using chemiluminescent detection.
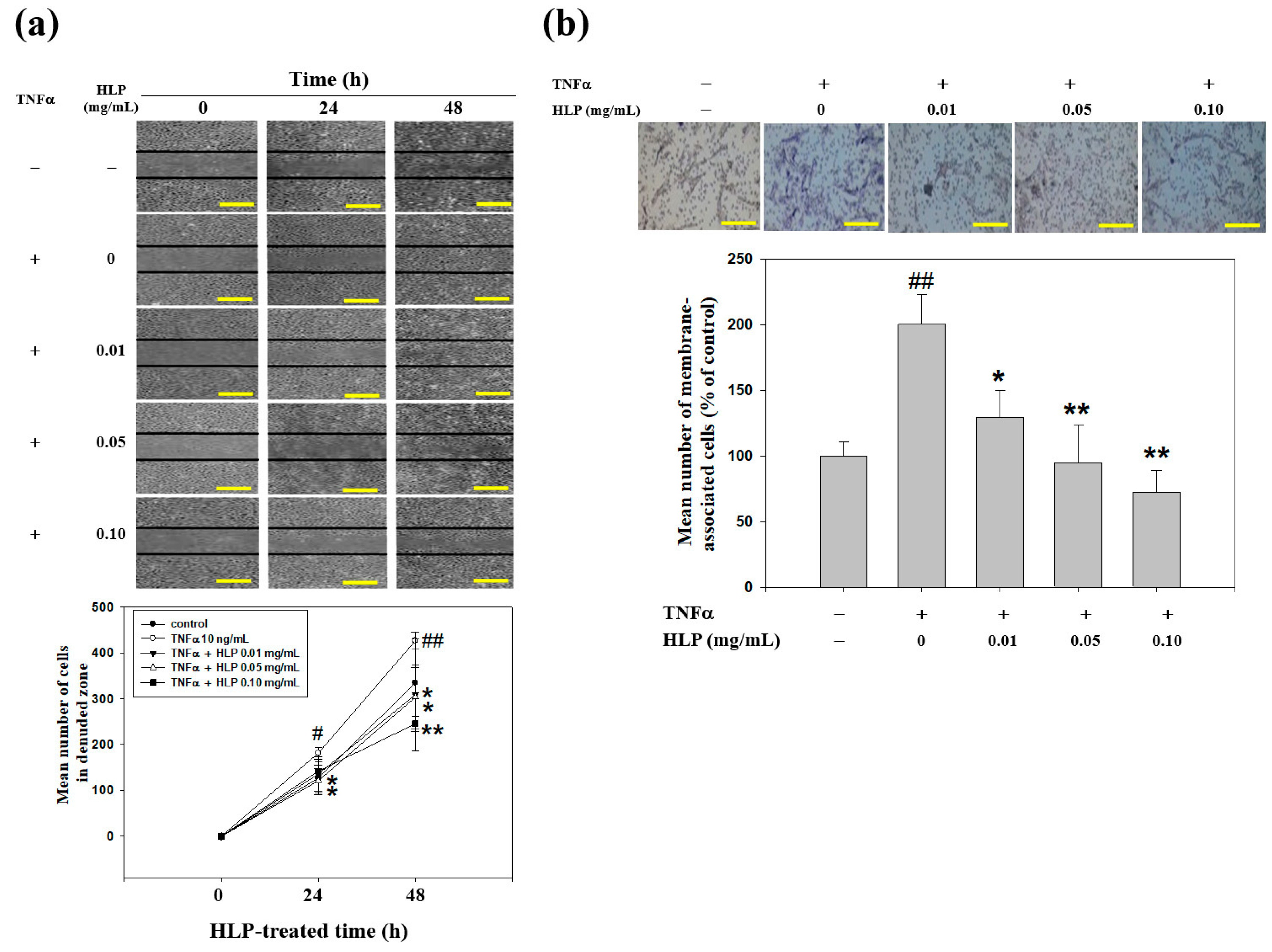
2.8. Wound-Healing Migration Assay
To study the possibility that HLP alter migration of VSMC-treated TNF-α, the cell medium was replaced with serum-containing medium following the treatments of TNF-α (10 ng/mL) in the absence or presence various concentrations (0, 0.01, 0.05, and 0.10 mg/mL) of HLP, and the monolayers were wounded using scraping with a 20-μL pipette tip. At the indicated times (0, 24, and 48 h) after scraping, the above-treated cells were washed twice in PBS (pH 7.4). The cells were photographed using a phase-contrast microscope (Olympus, Tokyo, Japan) [
23].
2.9. Boyden Chamber Invasion Assay
To test the effect of HLP on the in vitro invasiveness of VSMC-treated TNF-α, a modified Boyden chamber (Neuro Probe, Cabin John, MD, USA) invasion assay coating with a layer of Matrigel (25 mg/50 mL; Sigma-Aldrich, St Louis, MO, USA) was used [
23], and was applied to polycarbonate membrane filters with an 8.0 μm pore size (Nucleopore, Pleasanton, CA, USA). Afterwards, the membrane was fixed with methanol, and then stained with 10% Giemsa (Sigma-Aldrich, St Louis, MO, USA). The image of cells invaded through the membrane was capture and counted under the light microscope.
2.10. Cell Growth Curve Analysis
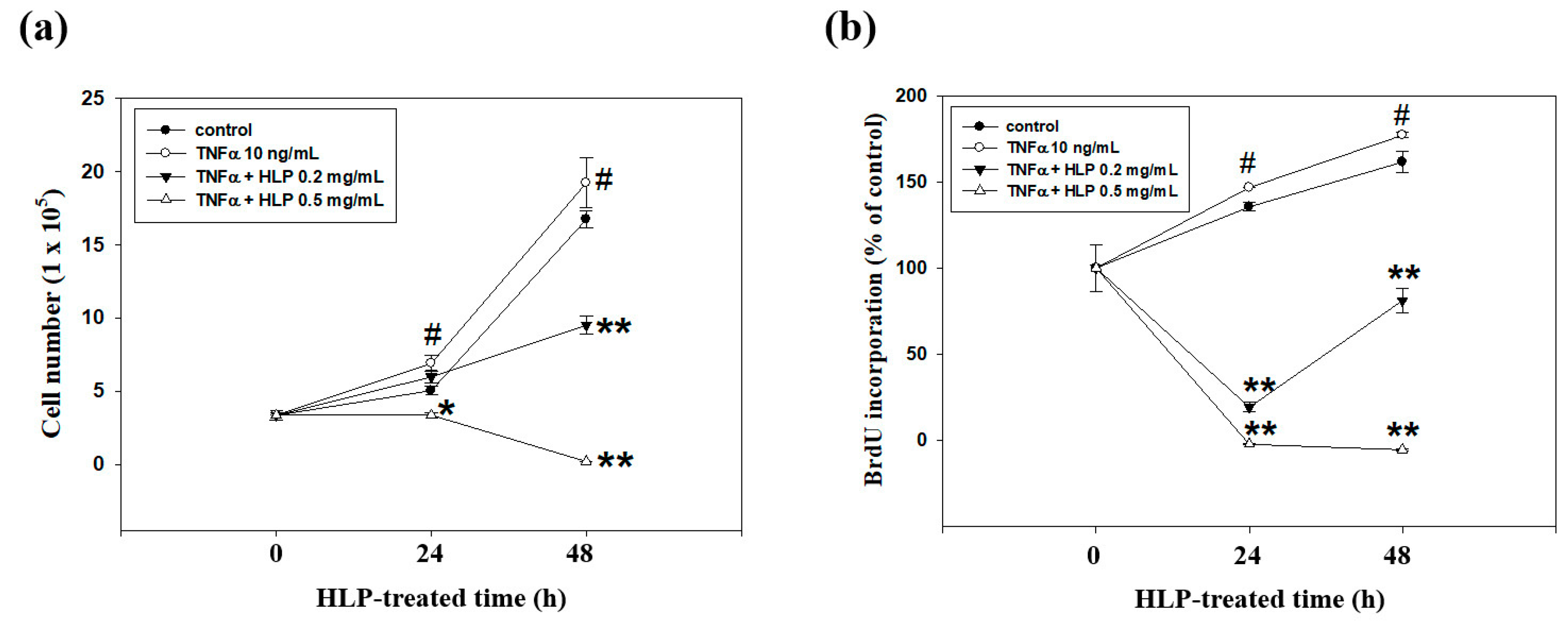
VSMC was seeded into a 6-well culture plate at a density of 7 × 104 cells/mL, and then incubated with TNF-α (10 ng/mL) in the absence or presence various concentrations (0, 0.2, and 0.5 mg/mL) of HLP for 24 h. The cell numbers were further counted using the Corning Cell Counter with a reusable glass counting chamber (Corning Inc, Corning, NY, USA) each day for 2 days. On the basis of the mean number of cells in these wells, the growth curves were formed.
2.11. Bromodeoxyuridine (BrdU) Cell Proliferation Assay
To identify the cells in S phase of cell cycle, the BrdU cell proliferation assay (Oncogene, Cambridge, MA, USA) was carried out according to the manufacturer’s manual. In brief, A7r5 cells were seeded into a 96-well plate (4 × 103 cells/well) and grown in DMEM medium supplemented with 5% FBS overnight. The cells were rinsed once with serum-free medium, and then treated with TNF-α (10 ng/mL) in the presence or absence of various concentrations (0.2 and 0.5 mg/mL) of HLP in serum-free medium for 24 h. In most of the experiments, pulse labeling of synthesized DNA was used. For this, the BrdU label was added 1 h before the experimental end. The cells were fixed, denatured, and probed with anti-BrdU antibody. Absorbance was determined at dual wavelengths of 450 and 540 nm in a microplate reader system (Bio-Rad Labs., Hercules, CA, USA). Proliferative value (BrdU incorporation) was expressed as a percentage of absorbance of the treated cells to the absorbance of the non-treated control cells. The BrdU incorporation of the control group was set to 100%.
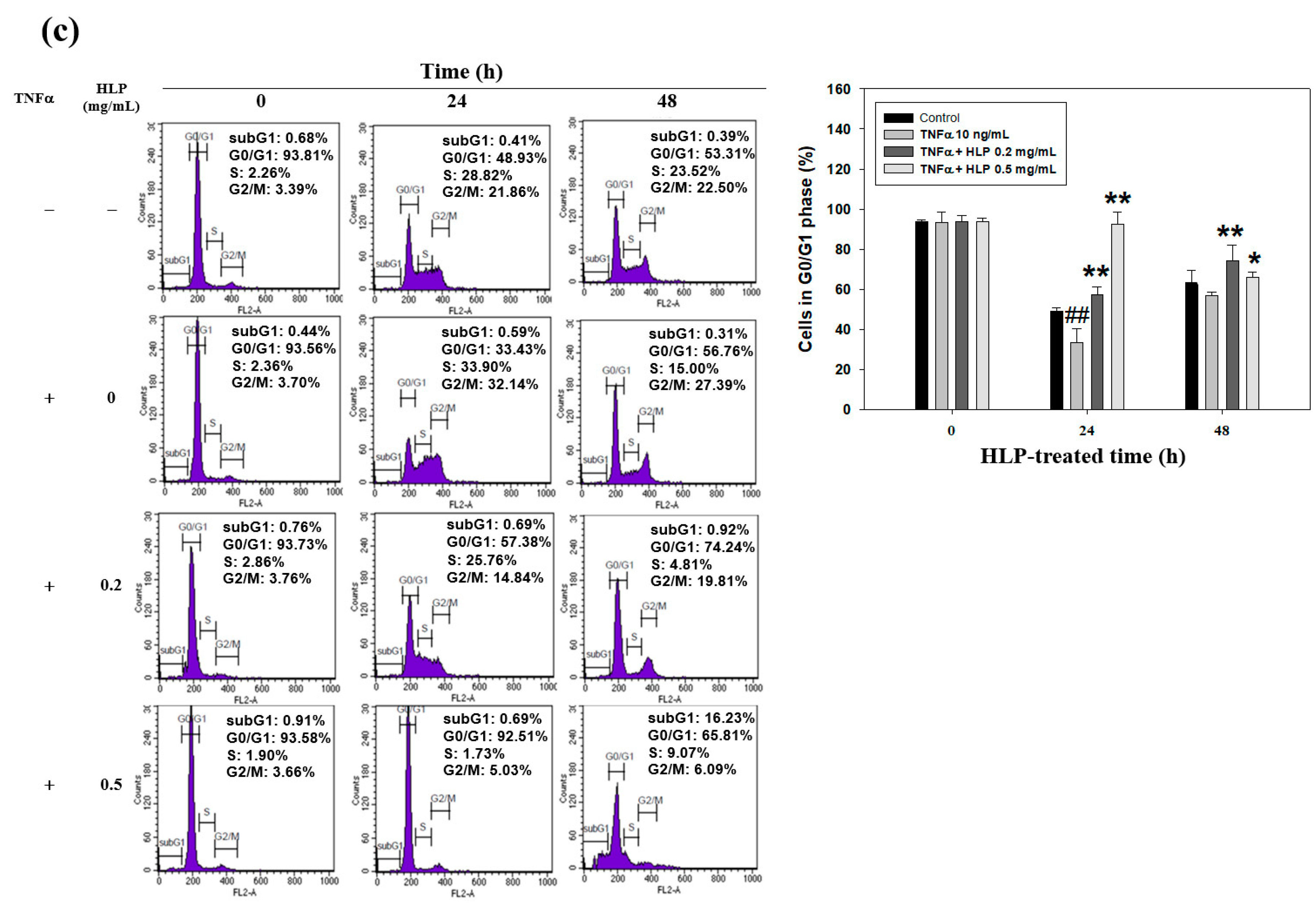
2.12. Cell Cycle Analysis by DNA Content
The quantification of cell cycle distribution was examined using a FACScan laser flow cytometer (Becton Dickinson, San Jose, CA, USA). The VSMC was treated with TNF-α (10 ng/mL) in the absence or presence various concentrations (0.2 and 0.5 mg/mL) of HLP for 24 h; collected, rinsed with PBS twice; fixed in 70% ethanol at –20 °C overnight; and then stained with propidium iodide (PI) solution (20 μg/mL of PI, 20 μg/mL of RNase A, and 0.1% Triton X-100; all chemicals from Sigma-Aldrich, St Louis, MO, USA) for 20 min in the dark at room temperature. Each phase of cell cycle was presented as the cell number versus the DNA content as indicated by the intensity of fluorescence, and gated into subG1, G0/G1, S, and G2/M phases with CELLQuest Version 3.3 software (Becton Dickinson, San Jose, CA, USA).
2.13. Immunoprecipitation
For detection of protein-protein interaction, immunoprecipitation was carried out. In short, 500 μg of protein from cell lysates was precleared with protein A–agarose beads (Pierce Biotechnology, Rockford, IL, USA), followed by immunoprecipitation using polyclonal antibodies against cdk2 or E2F, purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA). Immune complexes were harvested with protein A, and immunoprecipitated proteins were then assayed by Western blotting, as above. Immunodetection was performed using polyclonal anti-cyclin E or anti-Rb antibodies (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA).
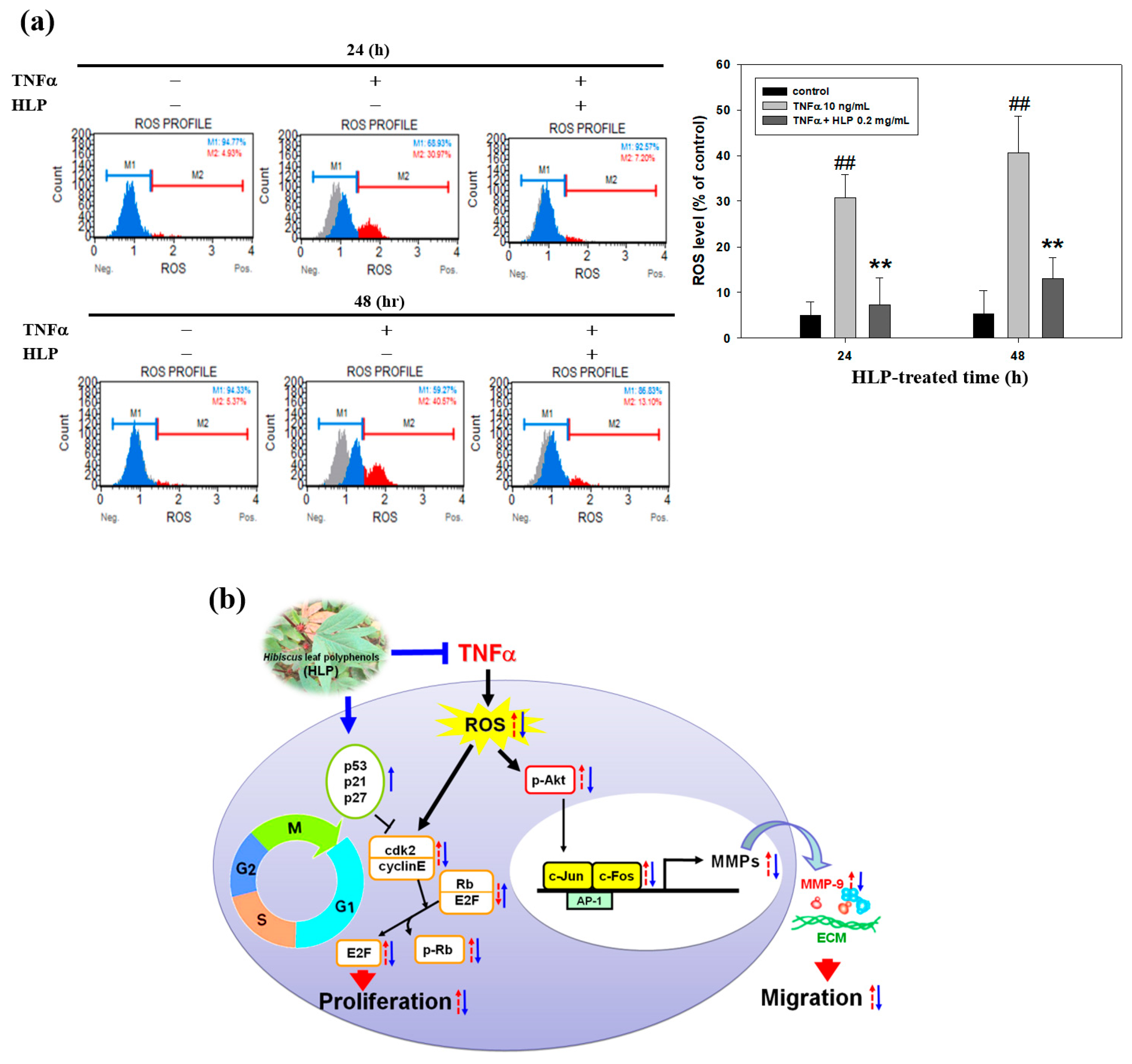
2.14. Intracellular ROS Assay
The fluorescent probe, dichlorofluorescin diacetate (DCFH-DA), purchased from Enzo Life Sciences Inc. (Farmingdale, NY, USA), was used to determine the effect of HLP on intracellular ROS generation by TNF-α stimulation. In brief, the confluent A7r5 cells in the 6-well plates at 105 cells/well were treated with TNF-α (10 ng/mL) in the absence or presence various concentrations (0.2 and 0.5 mg/mL) of HLP for 24 h. After removing the treated cells from the wells, the cells were incubated with 2 µM of DCFH-DA at 37 °C for 30 min. The fluorescence intensity of intracellular ROS production was evaluated at an excitation and emission wavelength of 485 and 530 nm, respectively, using Muse™ Cell Analyzer (EMD Millipore Corporation, Merck Life Sciences, KGaA, Darmstadt, Germany). Values were expressed relative to the fluorescence signal of the control.
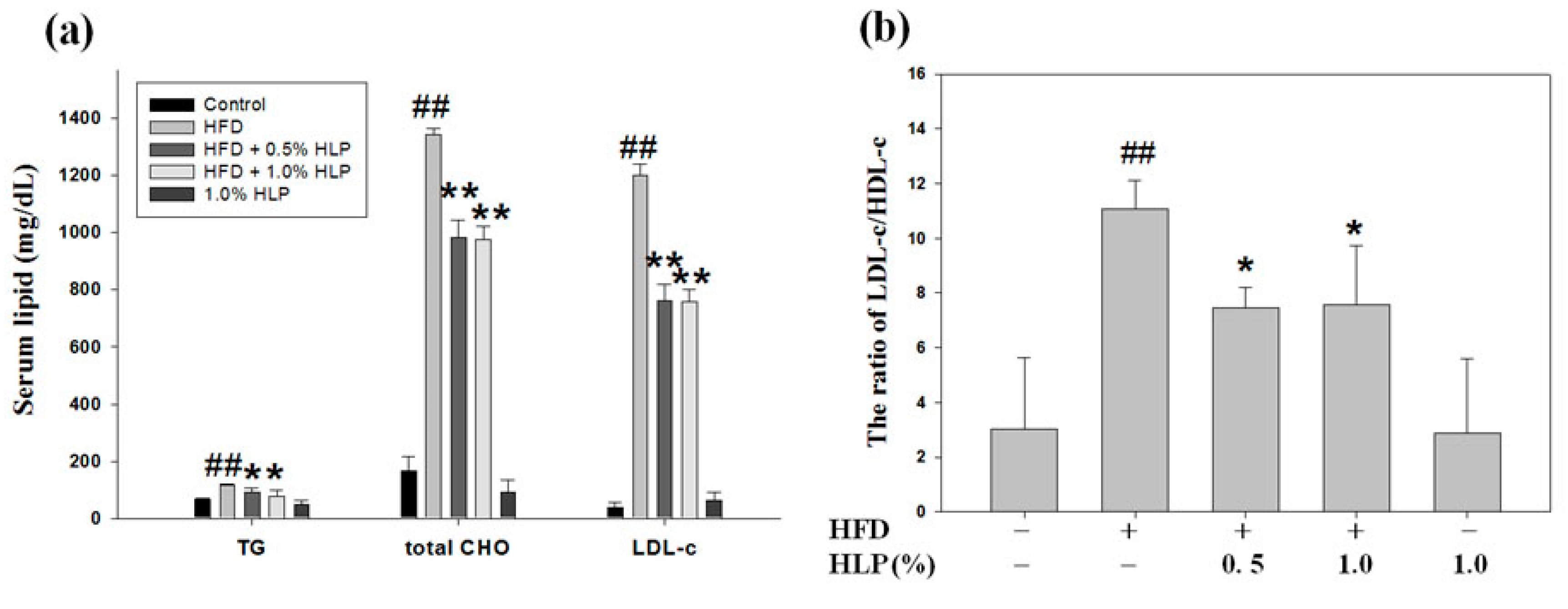
2.15. Evaluation of Atherosclerotic Lesions In Vivo
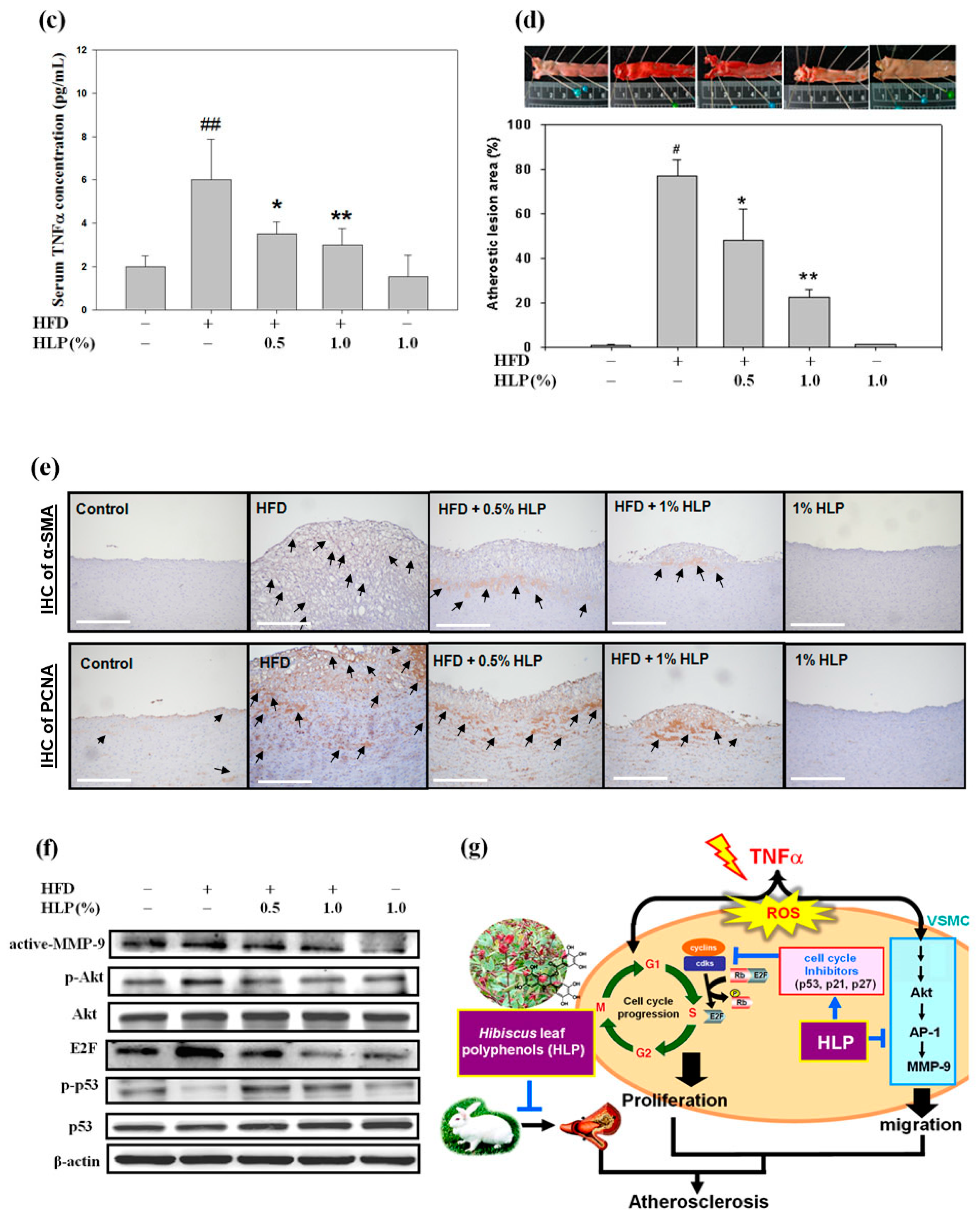
New Zealand white male rabbits weighing between 1800 and 2200 g were randomly divided into five experimental groups as follows: Group I, normal control group (Purina Lab Diet 5031); group II, high-fat diet (HFD); group III, HFD with 0.5% HLP group (HFD + 0.5% HLP); group IV, HFD with 1% HLP group (HFD + 1% HLP); and group V, normal diet with 1% HLP group (cytotoxicity group of HLP). The rabbits in groups II, III, and IV were fed on a HFD containing 95.7% standard Purina Chow (Purina Mills Inc., Louis, MI, USA), 1.3% cholesterol, and 3% lard oil (Sigma-Aldrich, St Louis, MO, USA) for 25 weeks to induce the atherosclerotic process., In groups III, IV, and V, the rabbits were treated with oral feeding 0.5% or 1% HLP at the same time. The dose regimen for these groups was based on a previous study published by Chiu et al. [
18]. For the care and use of laboratory animals, the use of all rabbits was reviewed and approved by Chung Shan Medical University animal care committee according to the guidelines of the Institutional Animal Care and Use Committee (IACUC approval number: 893). After 25 weeks of supplementation, aortic arches from each rabbit were collected and then stained with hematoxylin and eosin (H & E) for the pathological analysis. Serum was also collected and stored at –80 °C until measurements of serum biochemical parameters and TNF-α using a cytoscreen immunoassay kit (BioSource International, Camarillo, CA, USA). For immunohistochemistry (IHC), commercial monoclonal anti-alpha smooth muscle actin (α-SMA, a marker of VSMC migration), obtained from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA), and anti- PCNA (a marker of VSMC proliferation) were used for target detection in the paraffin-embedded tissues.
2.16. Statistical Analysis
In vitro data are reported as means ± standard deviation (SD) of three independent experiments. The in vivo effect of each treatment was analyzed from 6 rabbits (n = 6) in each group. Statistical significances of difference throughout this study were evaluated by one-way analysis of variance (ANOVA). p < 0.05 was considered statistically significant.
4. Discussion
H. sabdariffa Linne (
Malvaceae), an attractive plant believed to be native to African countries, is cultivated in both Southern and Eastern Taiwan [
26]. The calyces of the plant are typically used in foods and beverages, such as jam, jellies, and teas [
15,
26]. Previous studies have shown that various extracts of calyces of
H. sabdariffa L., including
H. sabdariffa aqueous extracts (HSEs),
H. sabdariffa anthocyanins (HAs), and its polyphenol-rich extracts (HPEs), have been reported to exhibit a wide variety of activities against hypertension, inflammation, liver disorders, diabetes, cancer, atherosclerosis, and other metabolic syndromes [
26]. While the focus has been on the calyx, the leaves of this plant are also consumed as a leafy vegetable in many countries [
15]. The
Hibiscus leaf has been also reported to exert many biologic effects, including antioxidant [
13,
15], anti-hyperlipidemic [
13,
14], anti-cancer [
17,
18], anti-atherosclerotic [
11,
16], and anti-inflammatory [
15] activities, as shown in
Table S1. Our recent studies have indicated that HLP, a methanol extract of
Hibiscus leaf, is rich in polyphenols [
16], including ECG and other polyphenols (Cat, EA, Que, and FA;
Table S2). In the literature, ECG-enriched HLP exhibited to inhibit ox-LDL uptake and lipid-laden foam cell formation, promoted cholesterol efflux [
16], and reduced ox-LDL-mediated endothelial cell injury and apoptosis [
11], so HLP was expected to have potential as an anti-atherogenic agent. ECG, one of the major tea catechins, plays an important protective role in the cardiovascular system, and has been reported to possess anti-atherogenic properties in in vitro and in vivo studies [
27]. It has been shown that the anti-atherosclerotic effect of Cat is associated with their antioxidant, anti-hypertensive, hypolipidaemic, and anti-mutagenic effects [
28]. These Cat have been indicated to suppress the LDL oxidation and the foam cell formation in in vivo atherosclerotic lesions [
27], and MMP-2 activities in cell culture supernatant of pulmonary VSMC [
29]. Previous studies have indicated that EA, a polyphenolic compound present in berries, scavenged free radicals and improved lipid peroxidation [
30]. According to the past and present works, the findings cooperatively show the anti-atherosclerotic activities of HLP may be contributed by their biological properties of these polyphenolic components.
In the comparison of components between both extracts, polyphenolic extracts from flowers (HPE) and leaves (HLP) of
H. sabdariffa L., the total flavonoid content of HPE and HLP was estimated to near to 20% and 75%, respectively, via Jia method [
16,
22]. The results show the polyphenolic extract of
Hibiscus leaves exhibited an about 3.8-fold content of flavonoids compared to that of its flowers. In addition to the above, HLP seems to possess stronger protective properties from VSMC dysfunction than HPE [
22], including the inhibitory effects on MMP expression and cell migration (
Figure 2 and
Figure 4). Huang et al. demonstrated that HPE at the doses of 0.01 and 0.10 mg/mL reduced high glucose-stimulated cell migration about 30% and 80%, respectively, by measuring the wound-healing assay [
22]. In this study, above the dose of HLP at 0.01 mg/mL could completely reverse the TNF-α-increased proportion of cell migration (
Figure 4a). These data suggest that HLP could exert the anti-migratory effect at lower doses than HPE.
Atherosclerosis is a multistep and chronic inflammatory process that involves interactions between various soluble mediators, endothelial cells, monocytes, and VSMCs. Monocyte-derived growth factors and cytokines further affect the vascular wall by stimulating VSMC migration and proliferation [
31]. The inflammatory cytokine TNF-α has been shown to play a vital role in the disruption of the vascular circulation, and its increased expression induces the production of ROS, resulting in endothelial cell injury and VSMC dysfunction [
32]. The blockade of TNF-α has been demonstrated to improve cardiovascular morbidity and mortality in chronic inflammatory disease [
33]. The model of TNF-α-stimulated VSMCs has been applied to mimic the VSMC dysfunction during atherosclerotic development [
31]. Therefore, the atheroprotective effects of HLP were investigated in a model of VSMCs exposed to TNF-α in vitro. The extract, at a concentration in a range of 0.01–0.10 mg/mL, possessed inhibitory effects on VSMC migration, as evidenced by the results of the decreased activities and expressions of MMPs, the levels of key migratory proteins, and the wound-healing and invasive abilities of VSMCs (
Figure 1d and
Figure 2,
Figure 3 and
Figure 4). The effect of the higher concentrations (>0.10 mg/mL) showed that it repressed cell growth and DNA synthesis, as well as enhanced cell apoptosis (
Figure 5). Similarly, Won et al. reported that the mechanisms of catechins action against cardiovascular diseases include the inhibition of VSMC proliferation [
34]. Further studies have also indicated that many polyphenols prevented atherosclerosis through inhibiting proliferation and/or inducing apoptosis of VSMC [
35]. The findings of this study reveal, for the first time, the protective effects of HLP on this model, examined in each test, provide bifunctional results of HLP. Therefore, it is convincing that HLP could potentially be used in the treatment of atherosclerosis.
As mentioned above, the migration of VSMCs from medial to intima contributes to the formation of atherosclerosis [
2]. MMPs, a family of proteinases, promote ECM degradation, which, in turn, facilitates cell migration. Activated Akt induced AP-1, which is required for the production of MMPs [
36]. However, upon TNF-α stimulation, the mechanism(s) mediating MMP activation and the MMP-regulated downstream signals have yet to be clarified. TNF-α promoted VSMC chemotaxis via Akt and MAPK activation, as reported by Chan et al. [
31]. The Akt antagonist PTEN was also shown to be involved in VSMC migration [
37]. Consistent with previous reports, this study confirmed that it is therefore possible that HLP inhibits TNF-α-stimulated MMP-9 activation by downregulating Akt/AP-1 pathway, and subsequently prevents VSMC migration. Furthermore, previous studies have indicated that oxidative stress modulated by ROS promotes VSMC dysfunction developing atherosclerosis and also induces MMP-mediated ECM remodeling and cell cycle progression [
6,
7,
38]. In this study, TNF-α increased the production of intracellular ROS, especially of the H
2O
2 level, and this might influence the overall signaling pathways of VSMC migration/proliferation. In this regard, HLP attenuated ROS generation against TNF-α stimulation (
Figure 7), and it might contribute to MMP inhibition and cell cycle regulation by HLP. Further investigations are needed to clarify this issue. Consistent with previous reports and our past results, as shown in
Table S4, this study confirmed that HLP possesses strong antioxidant ability in inhibiting the VSMC dysfunction and atherosclerotic development.
Next, to study the mechanism(s) of HLP-inhibited VSMC proliferation, the regulation of cell cycle arrest was examined. VSMCs begin to divide in response to mitogens and enter the S phase upon vascular injury [
9]. Cdks, working in conjunction with their activating subunits (cyclins), provide the driving force for cell cycle transitions [
34]. The kinase activities of these cyclin/cdk complexes are regulated by cdi, including Ink4 proteins (p16, p18, and p19) and Cip/Kip proteins (p21, p27, and p57), and their upstream factor, the gatekeeper of mammalian cell cycle, p53 [
10,
39]. As expected, HLP treatments induced a G0/G1 phase growth arrest by inducing the cki-mediated cell cycle regulation (
Figure 6). In addition, PCNA expression is increased in unstable atherosclerotic plaque [
40]; moreover, PCNA is highly expressed in human VSMCs [
41]. Consistent with previous reports, our studies showed that TNF-α significantly reduced A7r5 cell growth and increased PCNA expression (
Figure 6b). Further data demonstrate a marked and dose-dependent reduction in neointimal expression of PCNA after fed with HLP in HFD-treated rabbits (
Figure 8e), indicating that HLP ameliorates atherosclerosis by reducing PCNA expression, both in vitro and in vivo.