Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats
Abstract
1. Introduction
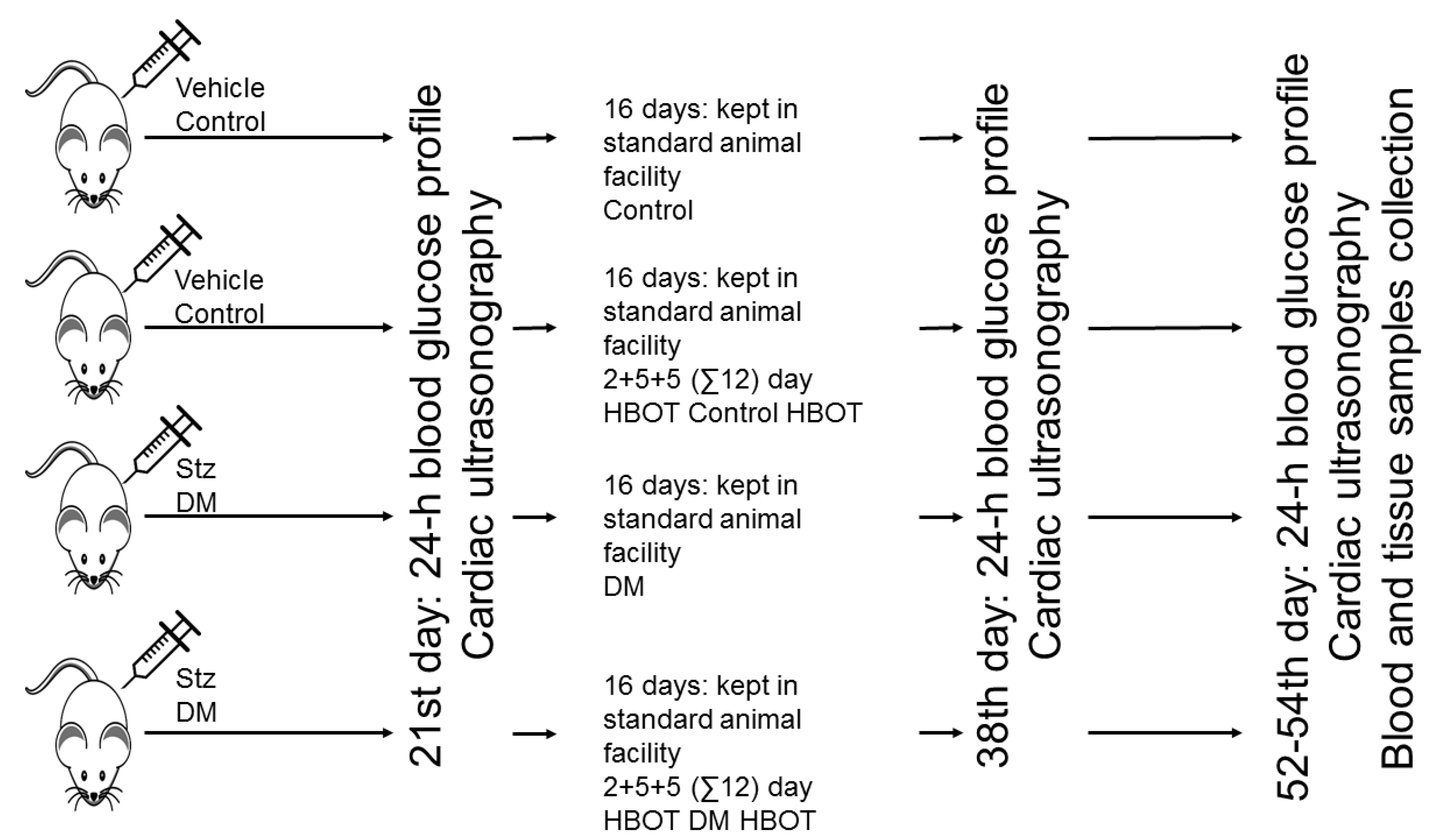
2. Materials and Methods
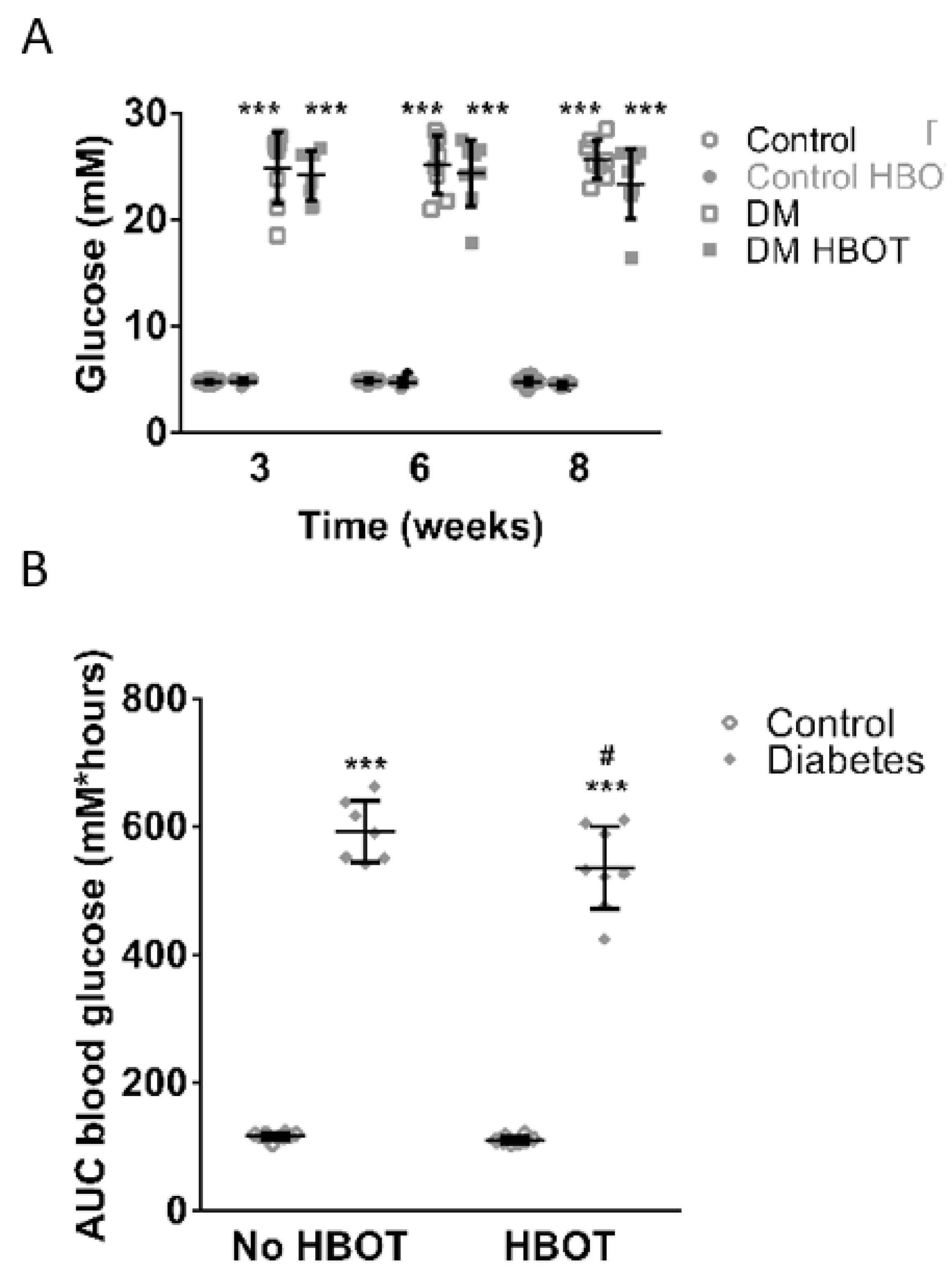
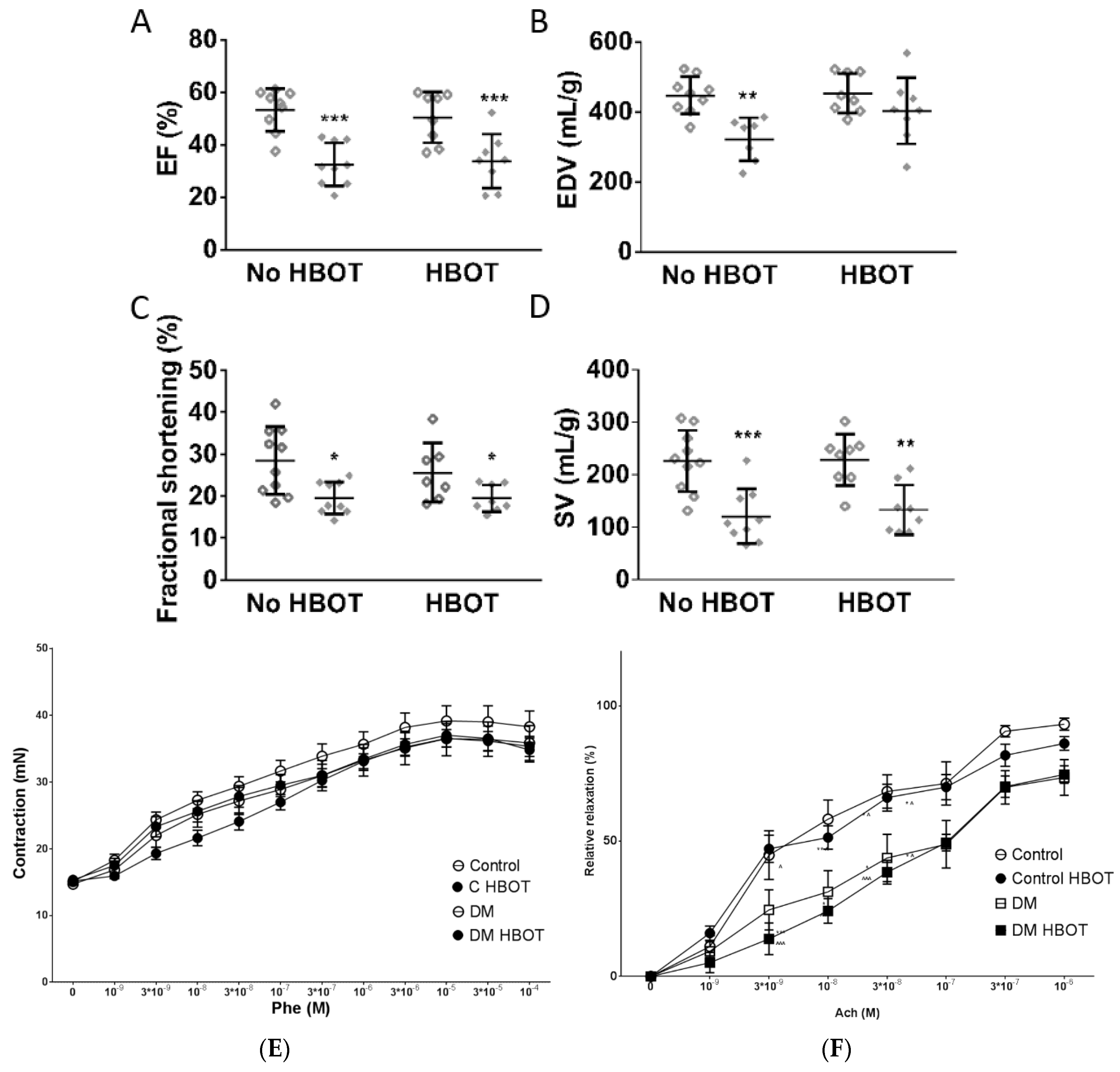
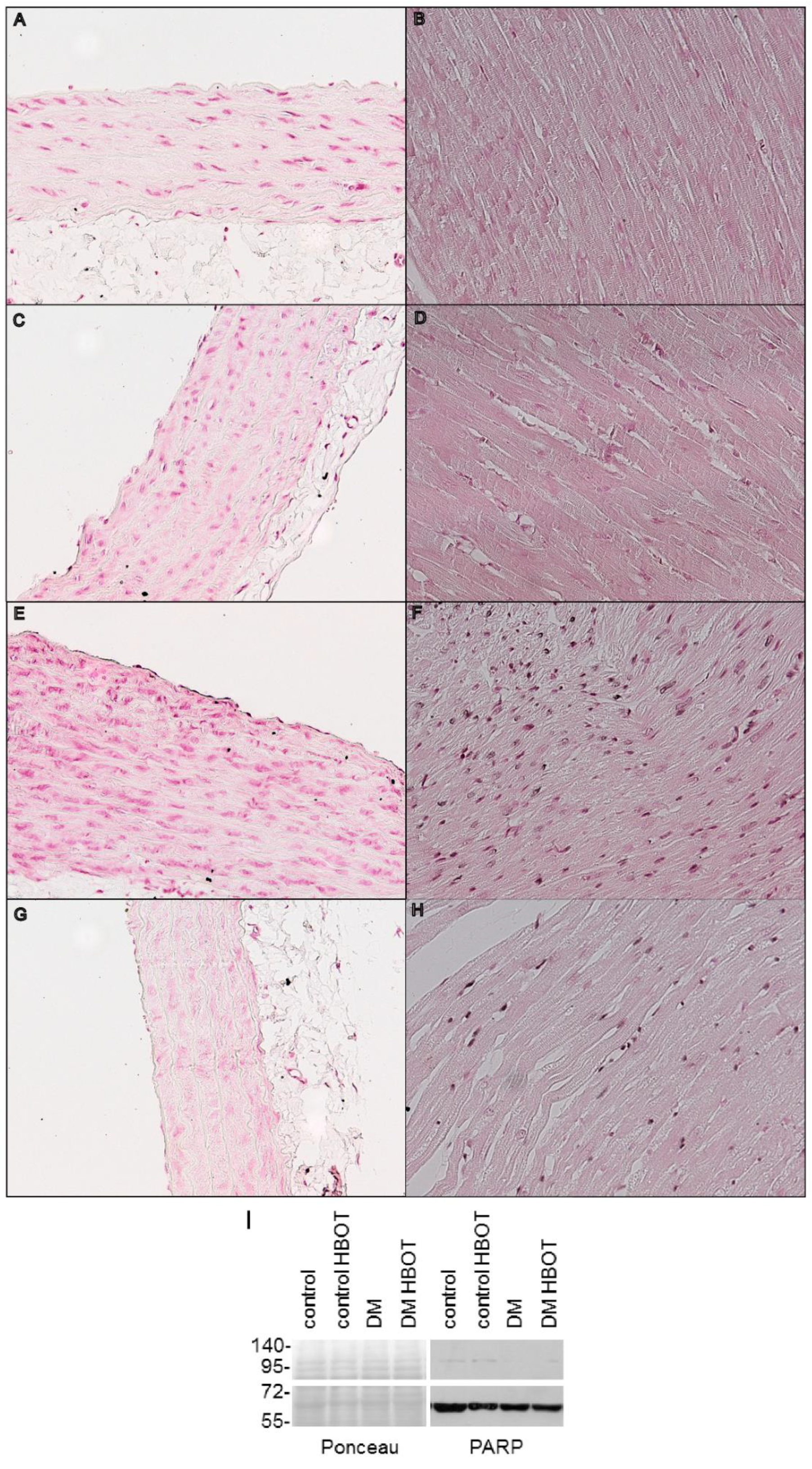
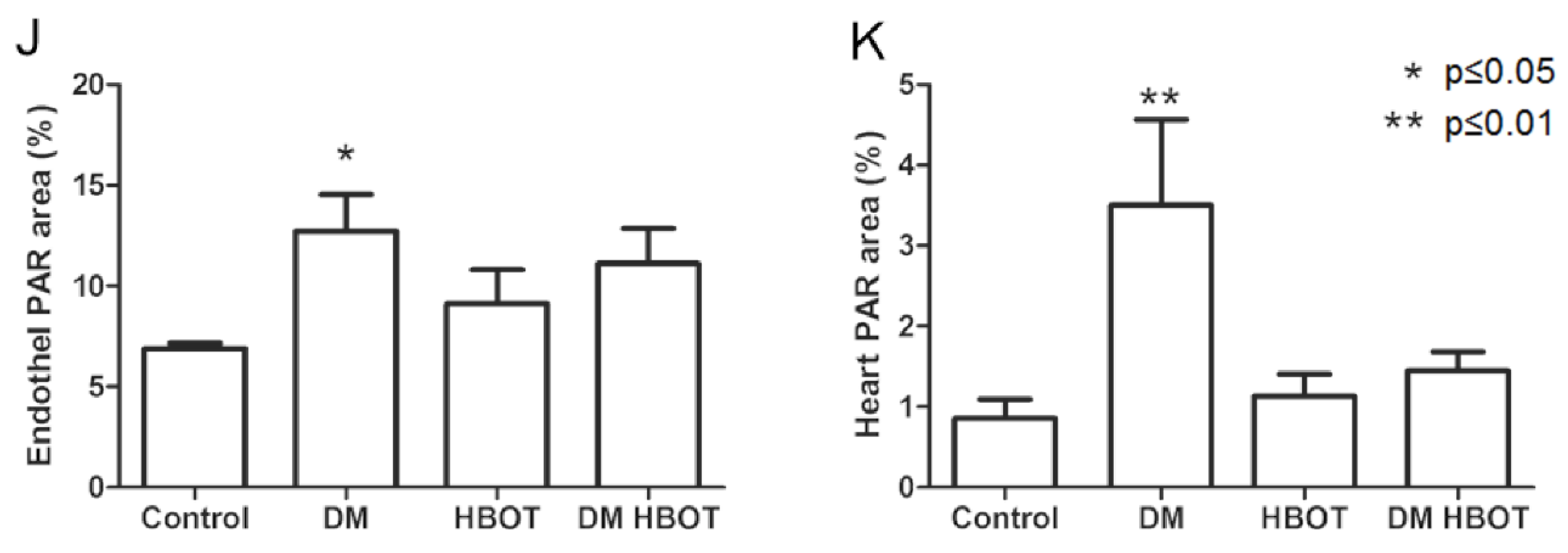
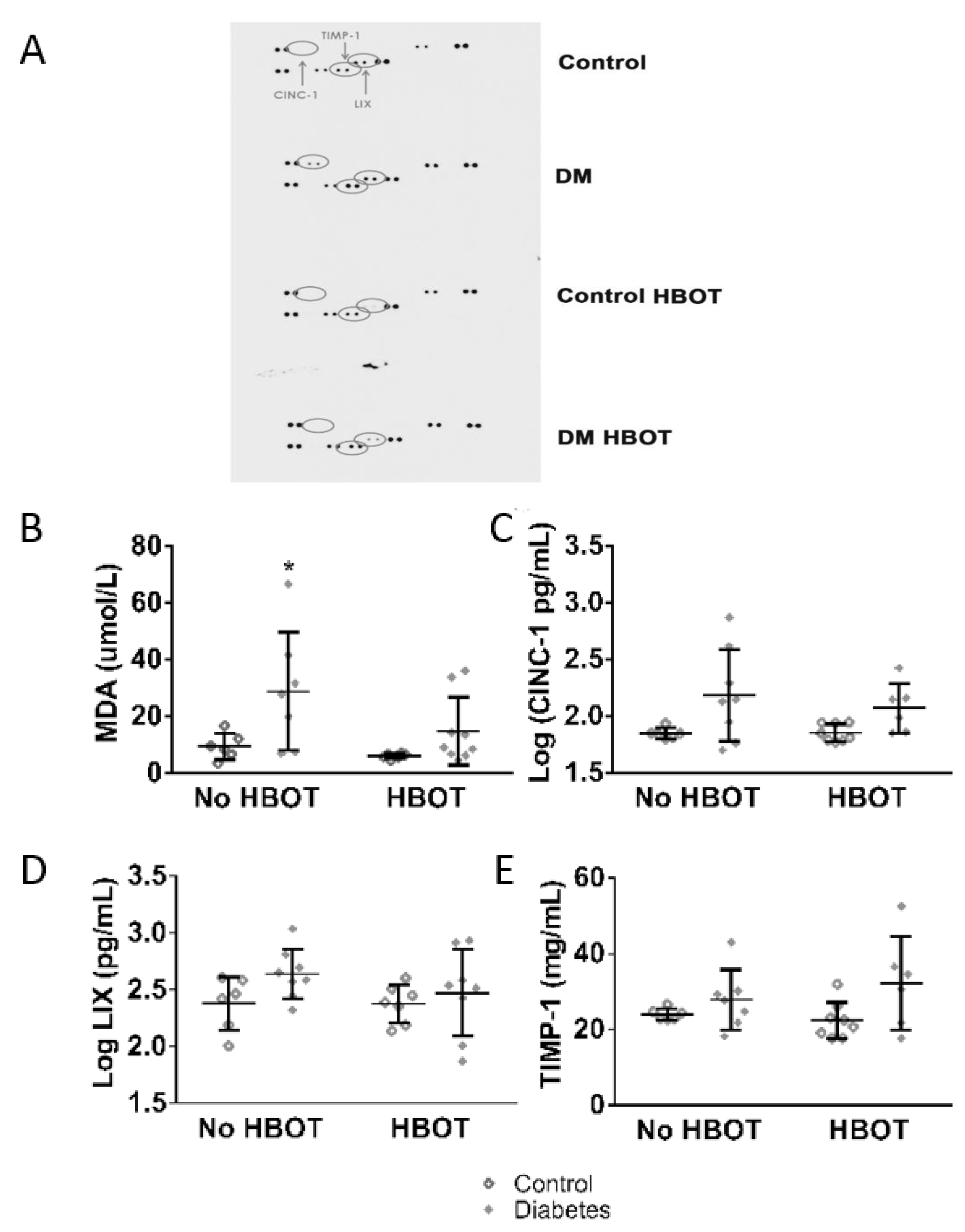
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Ach | acetylcholine |
| CINC-1 | cytokine-induced neutrophil chemoattractant-1 |
| DM | diabetes mellitus; |
| EDV | end diastolic volume |
| EF | ejection fraction |
| FS | fractional shortening |
| HBOT | Hyperbaric oxygen therapy |
| LIX | lipopolysaccharide-inducible |
| CXC | chemokine |
| MDA | malonyl-dialdehyde |
| MMP | matrix metalloproteinase |
| NO | nitric oxide |
| ONOO | peroxynitrite |
| PAR | poly(ADP-ribose) |
| PARP | poly(ADP-ribose) polymerase |
| Phe | phenylephrine |
| ROS | reactive oxygen species |
| SV | stroke volume |
| TBA | thiobarbituric acid |
| TBARS | thiobarbituric acid reactive substances |
| TIMP-1 | tissue inhibitor of metalloproteases-1 |
References
- Kanhai, A.; Losito, J.M. Hyperbaric oxygen therapy for lower-extremity soft-tissue sports injuries. J. Am. Podiatr. Med. Assoc. 2003, 93, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, A.; Duzgun, A.P.; Erdogan, K.; Ozkan, M.B.; Coskun, F. Efficacy of Hyperbaric Oxygen Therapy in Diabetic Foot Ulcers Based on Wagner Classification. J. Foot Ankle Surg. 2018, 57, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Fuller, A.M.; Giardina, C.; Hightower, L.E.; Perdrizet, G.A.; Tierney, C.A. Hyperbaric oxygen preconditioning protects skin from UV-A damage. Cell Stress Chaperones 2013, 18, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Gasier, H.G.; Fothergill, D.M. Oxidative stress, antioxidant defenses and nitric oxide production following hyperoxic exposures. Undersea Hyperb. Med. J. Undersea Hyperb. Med. Soc. Inc. 2013, 40, 125–134. [Google Scholar]
- Ma, L.; Li, P.; Shi, Z.; Hou, T.; Chen, X.; Du, J. A prospective, randomized, controlled study of hyperbaric oxygen therapy: Effects on healing and oxidative stress of ulcer tissue in patients with a diabetic foot ulcer. Ostomy Wound Manag. 2013, 59, 18–24. [Google Scholar]
- Wang, K.; Ahmad, S.; Cai, M.; Rennie, J.; Fujisawa, T.; Crispi, F.; Baily, J.; Miller, M.R.; Cudmore, M.; Hadoke, P.W.; et al. Dysregulation of hydrogen sulfide producing enzyme cystathionine gamma-lyase contributes to maternal hypertension and placental abnormalities in preeclampsia. Circulation 2013, 127, 2514–2522. [Google Scholar] [CrossRef]
- Efrati, S.; Gall, N.; Bergan, J.; Fishlev, G.; Bass, A.; Berman, S.; Hamad-Abu, R.; Feigenzon, M.; Weissgarten, J. Hyperbaric oxygen, oxidative stress, NO bioavailability and ulcer oxygenation in diabetic patients. Undersea Hyperb. Med. J. Undersea Hyperb. Med. Soc. Inc. 2009, 36, 1–12. [Google Scholar]
- Tsuneyama, K.; Chen, Y.C.; Fujimoto, M.; Sasaki, Y.; Suzuki, W.; Shimada, T.; Iizuka, S.; Nagata, M.; Aburada, M.; Chen, S.Y. Advantages and disadvantages of hyperbaric oxygen treatment in mice with obesity hyperlipidemia and steatohepatitis. Sci. World J. 2011, 11, 2124–2135. [Google Scholar] [CrossRef]
- De Smet, G.H.J.; Kroese, L.F.; Menon, A.G.; Jeekel, J.; van Pelt, A.W.J.; Kleinrensink, G.J.; Lange, J.F. Oxygen therapies and their effects on wound healing. Wound Repair. Regen. 2017, 25, 591–608. [Google Scholar] [CrossRef]
- Kendall, A.C.; Whatmore, J.L.; Winyard, P.G.; Smerdon, G.R.; Eggleton, P. Hyperbaric oxygen treatment reduces neutrophil-endothelial adhesion in chronic wound conditions through S-nitrosation. Wound Repair. Regen. 2013, 21, 860–868. [Google Scholar] [CrossRef]
- Zhang, Q.; Gould, L.J. Hyperbaric oxygen reduces matrix metalloproteinases in ischemic wounds through a redox-dependent mechanism. J. Investig. Derm. 2014, 134, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Tepic, S.; Petkovic, A.; Srejovic, I.; Jeremic, N.; Zivkovic, V.; Loncarevic, S.; Bradic, J.; Jakovljevic, V.; Zivkovc, M. Impact of hyperbaric oxygenation on oxidative stress in diabetic patients. Undersea Hyperb. Med. J. Undersea Hyperb. Med. Soc. Inc. 2018, 45, 9–17. [Google Scholar] [CrossRef]
- Szabo, C. PARP as a Drug Target for the Therapy of Diabetic Cardiovascular Dysfunction. Drug News Perspect. 2002, 15, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Szabo, C.; Mabley, J.G.; Moeller, S.M.; Shimanovich, R.; Pacher, P.; Virag, L.; Soriano, F.G.; Van Duzer, J.H.; Williams, W.; Salzman, A.L.; et al. Part I: Pathogenetic role of peroxynitrite in the development of diabetes and diabetic vascular complications: Studies with FP15, a novel potent peroxynitrite decomposition catalyst. Mol. Med. 2002, 8, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Padgaonkar, V.; Giblin, F.J.; Reddan, J.R.; Dziedzic, D.C. Hyperbaric oxygen inhibits the growth of cultured rabbit lens epithelial cells without affecting glutathione level. Exp. Eye Res. 1993, 56, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Inal, V.; Mas, M.R.; Isik, A.T.; Comert, B.; Aydn, S.; Mas, N.; Deveci, M.S.; Tasci, I.; Yamanel, L.; Cinar, E.; et al. A new combination therapy in severe acute pancreatitis—hyperbaric oxygen plus 3-aminobenzamide: An experimental study. Pancreas 2015, 44, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Matsunami, T.; Sato, Y.; Hasegawa, Y.; Ariga, S.; Kashimura, H.; Sato, T.; Yukawa, M. Enhancement of reactive oxygen species and induction of apoptosis in streptozotocin-induced diabetic rats under hyperbaric oxygen exposure. Int. J. Clin. Exp. Pathol. 2011, 4, 255–266. [Google Scholar]
- Matsunami, T.; Sato, Y.; Sato, T.; Yukawa, M. Antioxidant status and lipid peroxidation in diabetic rats under hyperbaric oxygen exposure. Physiol. Res. 2010, 59, 97–104. [Google Scholar]
- Matsunami, T.; Sato, Y.; Morishima, T.; Mano, Y.; Yukawa, M. Enhancement of glucose toxicity by hyperbaric oxygen exposure in diabetic rats. Tohoku J. Exp. Med. 2008, 216, 127–132. [Google Scholar] [CrossRef]
- Matsunami, T.; Sato, Y.; Ariga, S.; Sato, T.; Kashimura, H.; Hasegawa, Y.; Yukawa, M. Regulation of oxidative stress and inflammation by hepatic adiponectin receptor 2 in an animal model of nonalcoholic steatohepatitis. Int. J. Clin. Exp. Pathol. 2010, 3, 472–481. [Google Scholar]
- Lenzen, S. The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia 2008, 51, 216–226. [Google Scholar] [CrossRef]
- Faleo, G.; Fotino, C.; Bocca, N.; Molano, R.D.; Zahr-Akrawi, E.; Molina, J.; Villate, S.; Umland, O.; Skyler, J.S.; Bayer, A.L.; et al. Prevention of autoimmune diabetes and induction of beta-cell proliferation in NOD mice by hyperbaric oxygen therapy. Diabetes 2012, 61, 1769–1778. [Google Scholar] [CrossRef][Green Version]
- Karadurmus, N.; Sahin, M.; Tasci, C.; Naharci, I.; Ozturk, C.; Ilbasmis, S.; Dulkadir, Z.; Sen, A.; Saglam, K. Potential benefits of hyperbaric oxygen therapy on atherosclerosis and glycaemic control in patients with diabetic foot. Endokrynol. Pol. 2010, 61, 275–279. [Google Scholar]
- Varga, Z.V.; Giricz, Z.; Liaudet, L.; Hasko, G.; Ferdinandy, P.; Pacher, P. Interplay of oxidative, nitrosative/nitrative stress, inflammation, cell death and autophagy in diabetic cardiomyopathy. Biochim. Biophys. Acta 2015, 1852, 232–242. [Google Scholar] [CrossRef]
- Ray Chaudhuri, A.; Nussenzweig, A. The multifaceted roles of PARP1 in DNA repair and chromatin remodelling. Nat. Rev. Mol. Cell Biol. 2017, 18, 610–621. [Google Scholar] [CrossRef]
- Ayvaz, S.; Kanter, M.; Aksu, B.; Sahin, S.H.; Uzun, H.; Erboga, M.; Pul, M. The effects of hyperbaric oxygen application against cholestatic oxidative stress and hepatic damage after bile duct ligation in rats. J. Surg. Res. 2013, 183, 146–155. [Google Scholar] [CrossRef]
- Zakaria, E.M.; El-Bassossy, H.M.; El-Maraghy, N.N.; Ahmed, A.F.; Ali, A.A. PARP-1 inhibition alleviates diabetic cardiac complications in experimental animals. Eur. J. Pharmacol. 2016, 791, 444–454. [Google Scholar] [CrossRef]
- Li, F.; Drel, V.R.; Szabo, C.; Stevens, M.J.; Obrosova, I.G. Low-dose poly(ADP-ribose) polymerase inhibitor-containing combination therapies reverse early peripheral diabetic neuropathy. Diabetes 2005, 54, 1514–1522. [Google Scholar] [CrossRef]
- Minchenko, A.G.; Stevens, M.J.; White, L.; Abatan, O.I.; Komjati, K.; Pacher, P.; Szabo, C.; Obrosova, I.G. Diabetes-induced overexpression of endothelin-1 and endothelin receptors in the rat renal cortex is mediated via poly(ADP-ribose) polymerase activation. FASEB J. 2003, 17, 1514–1516. [Google Scholar] [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef]
- Yamakoshi, K.; Yagishita, K.; Tsuchimochi, H.; Inagaki, T.; Shirai, M.; Poole, D.C.; Kano, Y. Microvascular oxygen partial pressure during hyperbaric oxygen in diabetic rat skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R1512–R1520. [Google Scholar] [CrossRef]
- Kahler, W.; Koch, I.; Wohlrab, C.; Kowalski, J.; Witte, J.; Koch, A. Influence of hyperoxia and physical exercise on *OH-radical stress in humans as measured by dihydroxylated benzoates (DHB) in urine. Undersea Hyperb. Med. J. Undersea Hyperb. Med. Soc. Inc. 2013, 40, 231–238. [Google Scholar]
- Soejima, Y.; Hu, Q.; Krafft, P.R.; Fujii, M.; Tang, J.; Zhang, J.H. Hyperbaric oxygen preconditioning attenuates hyperglycemia-enhanced hemorrhagic transformation by inhibiting matrix metalloproteinases in focal cerebral ischemia in rats. Exp. Neurol. 2013, 247, 737–742. [Google Scholar] [CrossRef]
- Williams, L.J.; Nye, B.G.; Wende, A.R. Diabetes-Related Cardiac Dysfunction. Endocrinol. Metab. 2017, 32, 171–179. [Google Scholar] [CrossRef]
- Oyaizu, T.; Enomoto, M.; Yamamoto, N.; Tsuji, K.; Horie, M.; Muneta, T.; Sekiya, I.; Okawa, A.; Yagishita, K. Hyperbaric oxygen reduces inflammation, oxygenates injured muscle, and regenerates skeletal muscle via macrophage and satellite cell activation. Sci. Rep. 2018, 8, 1288. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benkő, R.; Miklós, Z.; Ágoston, V.A.; Ihonvien, K.; Répás, C.; Csépányi-Kömi, R.; Kerék, M.; Béres, N.J.; Horváth, E.M. Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats. Antioxidants 2019, 8, 607. https://doi.org/10.3390/antiox8120607
Benkő R, Miklós Z, Ágoston VA, Ihonvien K, Répás C, Csépányi-Kömi R, Kerék M, Béres NJ, Horváth EM. Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats. Antioxidants. 2019; 8(12):607. https://doi.org/10.3390/antiox8120607
Chicago/Turabian StyleBenkő, Rita, Zsuzsanna Miklós, Viktor Antal Ágoston, Katrine Ihonvien, Csaba Répás, Roland Csépányi-Kömi, Margit Kerék, Nóra Judit Béres, and Eszter Mária Horváth. 2019. "Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats" Antioxidants 8, no. 12: 607. https://doi.org/10.3390/antiox8120607
APA StyleBenkő, R., Miklós, Z., Ágoston, V. A., Ihonvien, K., Répás, C., Csépányi-Kömi, R., Kerék, M., Béres, N. J., & Horváth, E. M. (2019). Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats. Antioxidants, 8(12), 607. https://doi.org/10.3390/antiox8120607






