UHPLC–Q/Orbitrap/MS/MS Fingerprinting, Free Radical Scavenging, and Antimicrobial Activity of Tessaria absinthiodes (Hook. & Arn.) DC. (Asteraceae) Lyophilized Decoction from Argentina and Chile
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material
2.3. Lyophilized Decoction Preparation of T. absinthiodes (TLD)
2.4. UHPLC–DAD–MS Instrument
2.5. LC Parameters and MS Parameters
2.6. Determination of Total Phenolic (TP) and Flavonoid (F) Content
2.7. Antioxidant Activity
2.7.1. DPPH Scavenging Activity
2.7.2. Ferric-Reducing Antioxidant Power Assay (FRAP)
2.7.3. Trolox Equivalent Antioxidant Activity (TEAC) Assay
2.7.4. Lipid Peroxidation in Human Erythrocytes
2.8. Antibacterial Activity
2.9. Statistical Analysis
3. Results and discussion
3.1. UHPLC–OT Analysis of Tessaria Absinthiodes Decoctions from San Juan and Mendoza Provinces (Argentina) and Chile
3.1.1. Phenolic Acids
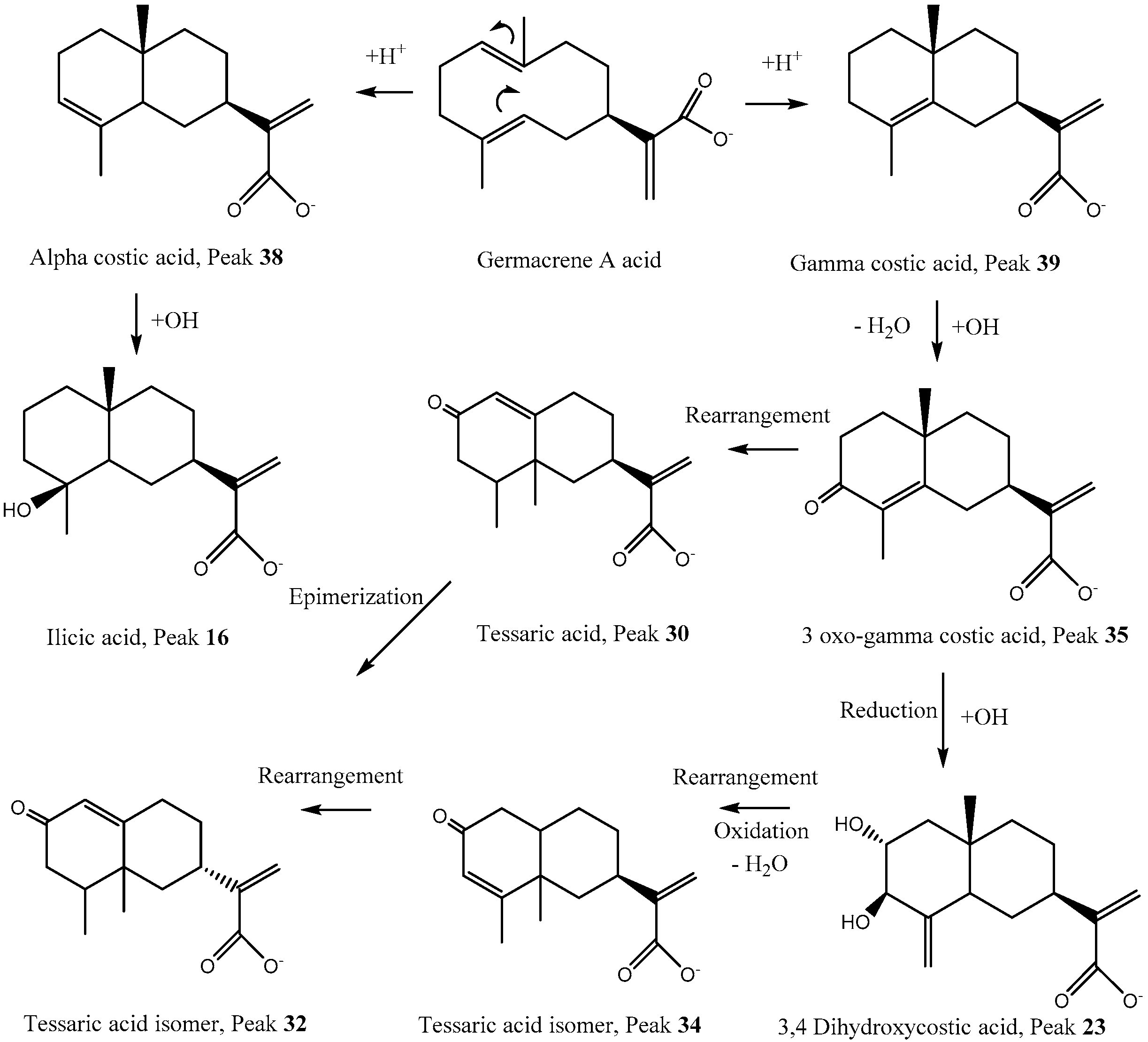
3.1.2. Sesquiterpenes
3.1.3. Oxylipins or Fatty Acids
3.1.4. Other Compounds
3.2. Total Phenolic and Flavonoid Contents and Antioxidant and Antibacterial Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). WHO Traditional Medicine Strategy: 2014–2023; WHO Press: Geneva, Switzerland, 2013; Available online: https://www.who.int/medicines/publications/traditional/trm_strategy14_23/en/ (accessed on 25 September 2019).
- Kayser, O. Ethnobotany and Medicinal Plant Biotechnology: From Tradition to Modern Aspects of Drug Development. Planta Med. 2018, 84, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Richieri, M.; Ladio, A.H.; Beeskow, A.M. Traditional knowledge and self-sufficiency: Rural herbal medicine in Central Plain of Chubut (Argentine). Boletín Latinoam. y del Caribe de Plantas Med. y Aromáticas 2013, 12, 44–58. [Google Scholar]
- Feresin, G.E.; Tapia, A.; Gimenez, A.; Ravelo, A.G.; Zacchino, S.; Sortino, M.; Schmeda-Hirschmann, G. Constituents of the Argentinian medicinal plant Baccharis grisebachii and their antimicrobial activity. J. Ethnopharmacol. 2003, 89, 73–80. [Google Scholar] [CrossRef]
- Lima, B.; Sánchez, M.; Agüero, M.B.; Tapia, A.; Palermo, J.A.; Feresin, G.E. Antibacterial activity of extracts and compounds isolated from the Andean medicinal plant Azorella cryptantha collected in San Juan province, Argentina. Ind. Crop. Prod. 2015, 64, 152–157. [Google Scholar] [CrossRef]
- Sanz, M.B.K.; Donadel, O.J.; Rossomando, P.C.; Tonn, C.E.; Guerreiro, E. Sesquiterpenes from Tessaria absinthioides. Phytochemistry 1997, 44, 897–900. [Google Scholar] [CrossRef]
- Barboza, G.E.; Cantero, J.J.; Núñez, C.; Pacciaroni, A.; Ariza Espinar, L. Medicinal plants: A general review, a phytochemical, and ethnopharmacological screening of the native argentine flora. Kurtziana 2009, 34, 365. [Google Scholar]
- Bailac, P.; Duschatzky, C.; Carrascull, A.; Ponzi, M. Composition of the Essential Oils of Tessaria absinthioides (Hook et Arn.) D. Candole. J. Essent. Oil Res. 2013, 10, 89–91. [Google Scholar] [CrossRef]
- Campos-Navarro, R.; Scarpa, G.F. The cultural-bound disease “empacho” in Argentina. A comprehensive botanico-historical and ethnopharmacological review. J. Ethnopharmacol. 2013, 148, 349–360. [Google Scholar] [CrossRef]
- Madaleno, I.M.; Delatorre-Herrera, J. Popular medicine of Iquique, Tarapacá. Idesia 2013, 31, 67–78. [Google Scholar] [CrossRef]
- García, M.; Sosa, M.E.; Donadel, O.J.; Giordano, O.S.; Tonn, C.E. Effects of some sesquiterpenes on the stored-product insect Tenebrio molitor (Coleoptera: Tenebrionidae). Rev. Soc. Entomol. Argent. 2003, 62, 17–26. [Google Scholar]
- García, M.; Sosa, M.E.; Donadel, O.J.; Giordano, O.S.; Tonn, C.E. Allelochemical effects of eudesmane and eremophilane sesquiterpenes on Tribolium castaneum larvae. J. Chemical. Ecol. 2003, 29, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Donadel, O.J.; Guerreiro, E.; María, A.O.; Wendel, G.; Enrizc, R.D.; Giordano, O.S.; Tonn, C.E. Gastric cytoprotective activity of ilicic aldehyde: Structure–activity relationships. Bioorg. Med. Chem. Lett. 2005, 15, 3547–3550. [Google Scholar] [CrossRef] [PubMed]
- Visintini, J.M.F.; Redko, F.; Muschietti, L.V.; Campos, R.H.; Martino, V.S.; Cavallaro, L.V. In vitro antiviral activity of plant extracts from Asteraceae medicinal plants. Virol. J. 2013, 27, 245. [Google Scholar] [CrossRef] [PubMed]
- Torres Carro, R.; Isla, M.I.; Ríos, J.L.; Giner, R.M.; Alberto, M.R. Anti-inflammatory properties of hydroalcoholic extracts of Argentine Puna plants. Food Res. Int. 2017, 67, 230–237. [Google Scholar] [CrossRef]
- Persia, F.A.; Rinaldini, E.; Carrión, A.; Hapon, M.B.; Gamarra-Luques, C. Evaluation of cytotoxic and antitumoral properties of Tessaria absinthioides (Hook & Arn) DC, “pájaro bobo”, aqueous extract. Medicina 2017, 77, 283–290. [Google Scholar]
- Cornejo, A.; Salgado, F.; Caballero, J.; Vargas, R.; Simirgiotis, M.; Areche, C. Secondary metabolites in Ramalina terebrata detected by UHPLC/ESI/MS/MS and identification of parietin as tau protein inhibitor. Int. J. Mol. Sci. 2016, 17, 1303. [Google Scholar] [CrossRef]
- Quispe, C.; Bórquez, J.; Villalobos, M.; Simirgiotis, M. Chemical Composition and Antioxidant Activity of Aloe vera from the Pica Oasis (Tarapacá, Chile) by UHPLC-Q/Orbitrap/MS/MS. J. Chem. 2018, 2018, 6123850. [Google Scholar] [CrossRef]
- Simirgiotis, M.J.; Quispe, C.; Mocan, A.; Villatoro, J.M.; Areche, C.; Bórquez, J.; Sepúlveda, B.; Echiburu-Chau, C. UHPLC high resolution orbitrap metabolomic fingerprinting of the unique species Ophryosporus triangularis meyen from the atacama desert, Northern Chile. Rev. Bras. de Farmacogn. 2017, 27, 179–187. [Google Scholar] [CrossRef]
- Luna, L.; Simirgiotis, M.J.; Lima, B.; Bórquez, J.; Feresin, G.E.; Tapia, A. UHPLC-MS metabolome fingerprinting: The isolation of main compounds and antioxidant activity of the andean species Tetraglochin ameghinoi (Speg.) Speg. Molecules 2018, 23, 793. [Google Scholar] [CrossRef]
- Gómez, J.; Simirgiotis, M.J.; Lima, B.; Paredes, J.D.; Villegas Gabutti, C.M.; Gamarra-Luques, C.; Bórquez, J.; Luna, L.; Wendel, G.H.; Maria, A.O.; et al. Antioxidant, Gastroprotective, Cytotoxic Activities and UHPLC PDA-Q Orbitrap Mass Spectrometry Identification of Metabolites in Baccharis grisebachii Decoction. Molecules 2019, 24, 1085. [Google Scholar] [CrossRef]
- Simirgiotis, M.J.; Quispe, C.; Areche, C.; Sepulveda, B. Phenolic Compounds in Chilean Mistletoe (Quintral, Tristerix tetrandus) Analyzed by UHPLC-Q/Orbitrap/MS/MS and Its Antioxidant Properties. Molecules 2016, 21, 245. [Google Scholar] [CrossRef] [PubMed]
- Simirgiotis, M.J.; Quispe, C.; Bórquez, J.; Schmeda-Hirschmann, G.; Avendaño, M.; Sepúlveda, B.; Winterhalter, P. Fast high resolution Orbitrap MS fingerprinting of the resin of Heliotropium taltalense Phil. from the Atacama Desert. Ind. Crop. Prod. 2016, 85, 159–166. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef]
- Tolonen, A.; Joutsamo, T.; Mattlla, S.; Kamarainen, T.; Jalonen, J. Identification of isomeric dicaffeoylquinic acids from Eleutherococcus senticosus using HPLC-ESI/TOF/MS and 1H-NMR methods. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 2002, 13, 316–328. [Google Scholar] [CrossRef]
- Lee, S.A.; Jung, E.B.; Lee, S.H.; Kim, Y.J.; Bang, H.; Seo, S.J.; Choi, Y.W.; Kim, M.H.; Lee, M.W.; Lee, C.S. 3, 4,5-tricaffeoylquinic Acid inhibits the lipopolysaccharide-stimulated production of inflammatory mediators in keratinocytes. Pharmacology 2012, 90, 183–192. [Google Scholar] [CrossRef]
- Bursal, E.; Aras, A.; Kilic, O. Evaluation of antioxidant capacity of endemic plant Marrubium astracanicum subsp. macrodon: Identification of its phenolic contents by using HPLC-MS/MS. Nat. Prod. Res. 2019, 33, 1975–1979. [Google Scholar] [CrossRef]
- Kurina Sanz, M.B.; Donadel, O.J. Biotransformation of eudesmanes by Tessaria absinthiodes cell suspension cultures. Plant Celltissue Organ Cult. 2003, 73, 123–129. [Google Scholar] [CrossRef]
- Bringmann, G.; Herzberg, D.; Adam, G.; Balkenhohl, F.; Paust, J. A Short and Productive Synthesis of (R)-α-lipoic Acid. Z. fur Nat. Sect. B J. Chem. Sci. 1999, 54, 655–661. [Google Scholar] [CrossRef]
- Tsevegsuren, N.; Edrada, R.; Lin, W.; Ebel, R.; Torre, C.; Ortlepp, S.; Wray, V.; Proksch, P. Biologically active natural products from Mongolian medicinal plants Scorzonera divaricata and Scorzonera pseudodivaricata. J. Nat. Prod. 2007, 70, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach, 3rd ed.; John Wiley & Sons, Ltd.: New York, NY, USA, 2009; ISBN 0471496413. [Google Scholar]
- Jiménez-Sánchez, C.; Lozano-Sánchez, J.; Rodríguez-Pérez, C.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Comprehensive, untargeted, and qualitative RP-HPLC-ESI-QTOF/MS2 metabolite profiling of green asparagus (Asparagus officinalis). J. Food Compos. Anal. 2016, 46, 78–87. [Google Scholar] [CrossRef]
- Simirgiotis, M.J.; Ramirez, J.E.; Hirschmann, G.S.; Kennelly, E.J. Bioactive coumarins and HPLC-PDA-ESI-ToF-MS metabolic profiling of edible queule fruits (Gomortega keule), an endangered endemic Chilean species. Food Res. Int. 2013, 54, 532–543. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Kiokias, S.; Varzakas, T.; Oreopoulou, V. In vitro activity of vitamins, flavanoids, and natural phenolic antioxidants against the oxidative deterioration of oil-based systems. Crit. Rev. Food Sci. Nutr. 2008, 48, 78–93. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beveragesand spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Pokorný, J.; Yanishlieva, N.; Gordon, M. Antioxidants in Food: Practical Applications; Elsevier: Washington, DC, USA, 2001; Volume 1, pp. 86–145. [Google Scholar]
- Abdel-Salam, O.M.E.; Sleem, A.A.; Shaffie, N.M. Protection against carbon tetrachloride-induced liver damage by citric acid. Cell Biol. Res. Ther. 2015, 4, 1. [Google Scholar] [CrossRef]
- Abdel-Salam, O.M.E.; Youness, E.R.; Mohammed, N.A.; Yassen, N.N.; Khadrawy, Y.A.; El-Toukhy, S.E.; Sleem, A.A. Novel neuroprotective and hepatoprotective effects of citric acid in acute malathion intoxication. Asian Pac. J. Trop. Med. 2016, 9, 1181–1194. [Google Scholar] [CrossRef]
- Tungsanga, K.; Sriboonlue, P.; Futrakul, P.; Yachantha, C.; Tosukhowong, P. Renal tubular cell damage and oxidative stress in renal stone patients and the effect of potassium citrate treatment. Urol. Res. 2005, 33, 65–69. [Google Scholar] [CrossRef]
- Puntel, R.L.; Roos, D.H.; Grotto, D.; Garcia, S.C.; Nogueira, C.W.; Rocha, J.B. Antioxidant properties of Krebs cycle intermediates against malonate pro-oxidant activity in vitro: A comparative study using the colorimetric method and HPLC analysis to determine malondialdehyde in rat brain homogenates. Life Sci. 2007, 81, 51–62. [Google Scholar] [CrossRef]
- Abdel-Salam, O.M.E.; Youness, E.R.; Mohammed, N.A.; Morsy, S.M.Y.; Omara, E.A.; Sleem, A.A. Citric acid effects on brain and liver oxidative stress in lipopolysaccharide-treated mice. J. Med. Food 2014, 17, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Prince, P.S.M.; Rajakumar, S.; Dhanasekar, K. Protective effects of vanillic acid on electrocardiogram, lipid peroxidation, antioxidants, proinflammatory markers and histopathology in isoproterenol induced. Eur. J. Pharmacol. 2011, 668, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Vinothiya, K.; Ashokkumar, N. Modulatory effect of vanillic acid on antioxidant status in high fat diet-induced changes in diabetic hypertensive rats. Biomed. Pharmacother. 2017, 87, 640–652. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.; He, C.; Li, X.X.; Zhou, L.Y.; Liu, R.J.; Zhang, S.; Li, G.Q.; Chen, Z.C.; Zhang, P.F. Ginnalin A from Kujin tea (Acer tataricum subsp. ginnala) exhibits a colorectal cancer chemoprevention effect via activation of the Nrf2/HO-1 signaling pathway. Food Funct. 2018, 9, 2809–2819. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, I.; Bassiouni, Y.A.; Badr, A.M.; Ali, R.A. Telmisartan and/or Chlorogenic Acid Attenuates Fructose-Induced Non-Alcoholic Fatty Liver Disease in Rats: Implications of Cross-Talk between Angiotensin, the sphingosine kinase/sphingoine-1-phosphate pathway, and TLR4 receptors. Biochem. Pharmacol. 2019, 164, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Shen, D.; Tang, X.; Li, X.; Wo, D.; Yan, H.; Song, R.; Feng, J.; Li, P.; Zhang, J. Chlorogenic acid prevents isoproterenol-induced hypertrophy in neonatal rat myocytes. Toxicol. Lett. 2014, 226, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Ruifeng, G.; Yunhe, F.; Zhengkai, W.; Yimeng, L.; Minjun, Y.; Xiaojing, S.; Zhengtao, Y.; Naisheng, Z. Chlorogenic acid attenuates lipopolysaccharide-induced mice mastitis by suppressing TLR4-mediated NF-κB signaling pathway. Eur. J. Pharmacol. 2014, 729, 54–58. [Google Scholar]
- Shi, H.; Dong, L.; Jiang, J.; Zhao, J.; Zhao, G.; Dang, X.; Lu, X.; Jia, M. Chlorogenic acid reduces liver inflammation and fibrosis through inhibition of toll-like receptor 4signaling pathway. Toxicology 2013, 303, 107–114. [Google Scholar] [CrossRef]
- Rezende, C.O., Jr.; Rigotto, C.; Caneschia, W.; Rezende, C.A.M.; Hyarica, M.L.; Couria, M.R.C.; Simões, C.M.O.; de Almeida, M.V. Anti-HSV-1 and antioxidant activities of dicaffeoyl and digalloyl esters of quinic acid. Biomed. Prev. Nutr. 2014, 4, 35–38. [Google Scholar] [CrossRef]
- Kim, J.Y.; Cho, J.Y.; Ma, Y.K.; Park, K.Y.; Lee, S.H.; Ham, K.S.; Lee, H.J.; Park, K.H.; Moon, J.H. Dicaffeoylquinic acid derivatives and flavonoid glucosides from glasswort (Salicornia herbacea L.) and their antioxidative activity. Food Chem. 2011, 125, 55–62. [Google Scholar] [CrossRef]
- Kim, S.S.; Park, R.Y.; Jeon, H.J.; Kwon, Y.S.; Chun, W. Neuroprotective Effects of 3,5-Dicaffeoylquinic Acid on Hydrogen Peroxide-induced Cell Death in SH-SY5Y Cells. Phytother. Res. 2005, 19, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Hernández, V.; Mañez, S.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Anti-inflammatory profile of dehydrocostic acid, a novel sesquiterpenes acid with a pharmacophoric conjugated diene. Eur. J. Pharm. Sci. 2005, 26, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Middleton, E.; Kanndasamy, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implication for inflammation, heart disease and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- García, L.; Rojo, D.; García, L.V.; Hernández, M. Plantas con propiedades antiinflamatorias. Centro de Investigaciones Biomédicas “Victoria de Girón”. Rev. Cubana. Invest. Biomed. 2002, 21, 214–216. [Google Scholar]
- Özden, P.; Avci, E.; Vural, H. Survey of the Apoptotic Effect of Ginnalin A on Hep3b Human Hepatocellular Carcinoma Cell Line. Multidiscip. Digit. Publ. Inst. Proc. 2017, 1, 1064. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; M100-S22; Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2012. [Google Scholar]
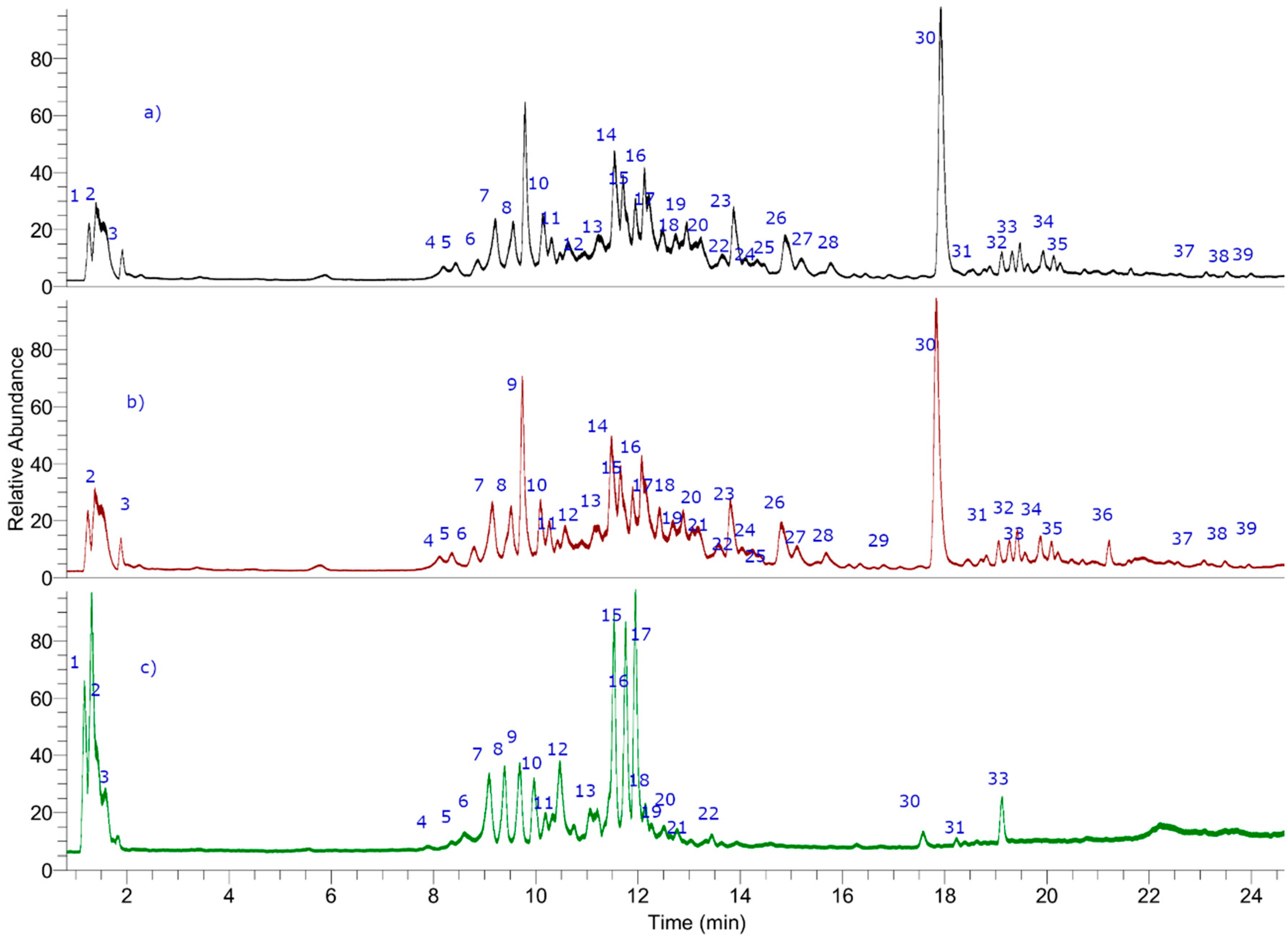
| Peak | Retention Time (min) | UV Max | Tentative Identification | Elemental Composition [M−H] | Measured Mass (m/z) | Theoretical Mass (m/z) | Ac Curacy (δppm) | MSn Ions (δppm) |
|---|---|---|---|---|---|---|---|---|
| 1 | 1.21 | - | Quinic acid *,a,b,c | C7H11O6− | 191.05579 | 191.05501 | 4.03 | 144.00844 |
| 2 | 1.31 | - | Manoheptulose a,b,c | C7H13O7− | 209.06633 | 209.06558 | 3.37 | 153.01857 |
| 3 | 1.82 | - | Citric acid a,b,c | C6H7O7− | 191.01863 | 191.01939 | 3.76 | 144.00844 |
| 4 | 7.26 | - | Unknown a,b,c | C16H11O15N3− | 365.01859 | 365.01847 | 0.23 | - |
| 5 | 8.47 | 330 | Unknown a,b,c | C4H7O12− | 246.99167 | 246.99320 | −6.2 | 152.01080 |
| 6 | 8.73 | 239–320 | CQA (chlorogenic acid) * a,b,c | C16H17O9− | 353.08671 | 353.08786 | 3.86 | 275.0235, 191.05481 (quinic acid), 707.18115 (2M−H) |
| 7 | 9.55 | 223 | 3-Hydroxysuberic acid a,b,c | C8H13O5− | 189.07645 | 189.07575 | 3.69 | - |
| 8 | 10.03 | 330 | Vanillic acid * a,b,c | C9H7O4− | 179.03465 | 179.03389 | 4.28 | 135.04436 |
| 9 | 10.07 | 283 | 2, 5, 9-trihydroxy-3-O-arabinosyl-tessaric acid a,b,c | C20H31O10− | 431.19241 | 431.19117 | 2.87 | 311.11367, 135.04433 |
| 10 | 10.36 | 330 | Unknown a,b,c | C10H9O15N3− | 411.00293 | 411.00282 | 0.32 | - |
| 11 | 10.43 | 288–346 | Hymenoxynin a,b,c | C21H34O9− | 429.21313 | 429.21191 | 2.85 | 267.21184 (M−hexose moiety) |
| 12 | 11.12 | 330 | Unknown a,b,c | C15H7O11N− | 377.00092 | 377.00136 | −1.18 | |
| 13 | 11.48 | 330 | Unknown a,b | C10H9O15N3− | 411.00296 | 411.00282 | 0.34 | |
| 14 | 11.62 | 239–320 | 1′,5′-di-CQA(cynarin) a,b,c | C25H23O12− | 515.11945 | 515.11840 | 2.02 | 191.05551 (quinic acid), 179.03429 |
| 15 | 11.82 | 239–320 | 3′,5′-di-CQA a,b,c | C25H23O12− | 515.11951 | 515.11840 | 2.14 | 191.05562 (quinic acid), 179.03429 |
| 16 | 11.90 | 255–354 | Ilicic acid a,b,c | C15H23O3− | 251.16516 | 251.16417 | 4.5 | 233.15470 (M–H2O), 207.17544 (M−CO2), 171.95076 |
| 17 | 12.00 | 239–320 | 4′,5′-di-CQA a,b,c | C25H23O12− | 515.11840 | 515.11945 | 2.14 | 191.05551 (quinic acid), 179.03429 |
| 18 | 12.21 | 255–365 | Eudesmane 4(15), 11(13)-dien-12, 5βolide * a,b,c | C15H19O3− | 247.13380 | 247.13287 | 3.77 | 205.15968 (M−CO2), 149.09645 |
| 19 | 12.57 | 289–329 | 3′,4′,5′-tri-CQA a,b,c | C34H39O15− | 677.15033 | 677.15119 | 14.30 | 515.11963 (Di-CQA), 191.05561 (quinic acid) |
| 20 | 12.81 | 278 | Unknown a,b,c | C30H33O15N10− | 773.13715 | 773.21214 | −0.08 | |
| 21 | 12.96 | 278 | Unknown a,b,c | C26H8ON10− | 476.08734 | 476.08771 | −0.76 | |
| 22 | 13.21 | 278 | Unknown a,b,c | C12HO16N14− | 953.17645 | 953.17632 | −0.98 | 476.08722 |
| 23 | 13.46 | 278 | 3,4-Dihydroxy-costic acid a,b | C15H21O4− | 265.14459 | 265.14344 | 4.32 | 247.13395 (M−H2O) |
| 24 | 14.04 | 278 | Unknown a,b | C28H23O12N9− | 677.15039 | 677.14741 | 4.40 | |
| 25 | 14.70 | 323 | Ginnalin A a,b | C20H19O13− | 467.08200 | 467.08202 | −0.03 | 249.08006 |
| 26 | 16.20 | 278 | 3,5-Dihydroxy-costic acid a,b | C15H21O4− | 265.14456 | 265.14344 | 4.24 | 247.13392 (M−H2O) |
| 27 | 15.55 | 265–329 | Tetra-CQA a,b | C43H35O18− | 839.17999 | 839.18289 | 3.43 | 191.05552 (quinic acid), 179.03423 |
| 28 | 15.94 | 235 | Scorzonerin (C30H36O11−) a,b | C30H36O11− | 571.21872 | 571.21739 | 1.63 | 467.08185, 327.21765 |
| 29 | 16.48 | 222 | Trihydroxy-octadecadienoic acid a | C18H31O5− | 327.21790 | 327.21660 | 3.95 | 283.22787(M−CO2) |
| 30 | 17.69 | 283 | Tessaric acid *,a,b,c | C15H19O3− | 247.13379 | 247.13287 | 3.71 | 205.15979 (M−CO2), 149.09644 |
| 31 | 18.34 | 222 | Trihydroxy-octadecaenoic acid a,b | C18H33O5− | 329.23225 | 329.23364 | 4.22 | 285.24352(M−CO2) |
| 32 | 19.09 | 283 | Tessaric acid isomer a,b | C15H19O3− | 247.13383 | 247.13287 | 3.90 | 205.15972 (M−CO2), 163.11223 |
| 33 | 19.25 | 218 | 5-Acetyl-3-hydroxy-4-dihydro-costic acid a,b,c | C17H25O5− | 309.17090 | 309.16965 | 4.03 | 291.16019 (M−H2O), 267.16018(M-acetyl moiety) 152.08374 |
| 34 | 19.47 | 283 | Tessaric acid isomer a,b | C15H19O3− | 247.13383 | 247.13287 | 3.90 | 205.15979 (M−CO2), 162.01357 |
| 35 | 19.96 | 283 | 3-oxo-gamma costic acid a,b | C15H19O3− | 247.13383 | 247.13287 | 3.90 | 231.13903, 233.11812, 219.13902, 215.00955, 149.09644 |
| 36 | 21.53 | 218 | Eupatorine a,b | C13H27O8− | 343.08258 | 343.08123 | −4.11 | 329.06663 (M−CH3), 315.0533 (M−2CH3), 313.03531 |
| 37 | 22.53 | 218 | Unknown a,b | C13H27O8− | 311.16876 | 311.17004 | −4.11 | |
| 38 | 23.41 | 335 | Gamma costic acid a,b | C15H21O2− | 233.15453 | 233.15361 | 3.94 | 215.00955, (M–H2O) 205.15973(M−CO) |
| 39 | 23.97 | 335 | Alpha Costic acid a,b | C15H21O2− | 233.15451 | 233.15361 | 3.87 | 215.00953, (M−H2O) 205.15965 (M−CO) |
| Assay | TLDSJ | TLDM | TLDCH |
|---|---|---|---|
| Content of phenols | |||
| Total phenolics (mg GAE/g TLD) | 94.84 ± 12.76 | 185.53 ± 43.9 | 64.36 ± 5.53 |
| Flavonoids (mg QE/g TLD) | 9.10 ± 0.90 | 18.18 ± 5.3 | 19.16 ± 0.66 |
| Antioxidant | |||
| DPPH (EC50 in µg TLD/mL) | 42.39 ± 8.21 | 41,6 ± 0.75 | 43.11 ± 4.04 |
| FRAP (mM TE/g TLD) | 0.89 ± 0.07 | 1.93 ± 0.3 | 0.98 ± 0.11 |
| TEAC (mg TE/g TLD) | 0.97 ± 0.04 | 1.29 ± 0.02 | 0.89 ± 0.085 |
| Percentage LP (at 250 µg TLD/mL) | 88.79 ± 11.53 | 86.77 ± 4.17 | 97.64 ± 1.9 a |
| Antibacterial Assay | TLD (MICs in µg/mL) | |||
|---|---|---|---|---|
| TLDCH | TLDM | TLDSJ | Cefotaxime | |
| Staphylococcus aureus methicillin-sensitive ATCC 29213 | >2500 | >2500 | >2500 | 0.5 |
| Staphylococcus aureus, methicillin-resistant ATCC 43300 | >2500 | 2500 | >2500 | 0.5 |
| Staphylococcus aureus methicillin-resistant-MQ-1 | >2500 | 2000 | >2500 | 0.5 |
| Staphylococcus aureus methicillin-resistant-MQ-2 | 2500 | 2000 | >2500 | 0.75 |
| Streptococcus pyogenes-1 | >2500 | >2500 | >2500 | 0.25 |
| Escherichia coli ATCC 25922 | >2500 | >2500 | >2500 | 1.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez, J.; Simirgiotis, M.J.; Lima, B.; Gamarra-Luques, C.; Bórquez, J.; Caballero, D.; Feresin, G.E.; Tapia, A. UHPLC–Q/Orbitrap/MS/MS Fingerprinting, Free Radical Scavenging, and Antimicrobial Activity of Tessaria absinthiodes (Hook. & Arn.) DC. (Asteraceae) Lyophilized Decoction from Argentina and Chile. Antioxidants 2019, 8, 593. https://doi.org/10.3390/antiox8120593
Gómez J, Simirgiotis MJ, Lima B, Gamarra-Luques C, Bórquez J, Caballero D, Feresin GE, Tapia A. UHPLC–Q/Orbitrap/MS/MS Fingerprinting, Free Radical Scavenging, and Antimicrobial Activity of Tessaria absinthiodes (Hook. & Arn.) DC. (Asteraceae) Lyophilized Decoction from Argentina and Chile. Antioxidants. 2019; 8(12):593. https://doi.org/10.3390/antiox8120593
Chicago/Turabian StyleGómez, Jessica, Mario J. Simirgiotis, Beatriz Lima, Carlos Gamarra-Luques, Jorge Bórquez, Duilio Caballero, Gabriela Egly Feresin, and Alejandro Tapia. 2019. "UHPLC–Q/Orbitrap/MS/MS Fingerprinting, Free Radical Scavenging, and Antimicrobial Activity of Tessaria absinthiodes (Hook. & Arn.) DC. (Asteraceae) Lyophilized Decoction from Argentina and Chile" Antioxidants 8, no. 12: 593. https://doi.org/10.3390/antiox8120593
APA StyleGómez, J., Simirgiotis, M. J., Lima, B., Gamarra-Luques, C., Bórquez, J., Caballero, D., Feresin, G. E., & Tapia, A. (2019). UHPLC–Q/Orbitrap/MS/MS Fingerprinting, Free Radical Scavenging, and Antimicrobial Activity of Tessaria absinthiodes (Hook. & Arn.) DC. (Asteraceae) Lyophilized Decoction from Argentina and Chile. Antioxidants, 8(12), 593. https://doi.org/10.3390/antiox8120593








