The Influence of In Vitro Gastrointestinal Digestion on the Chemical Composition and Antioxidant and Enzyme Inhibitory Capacities of Carob Liqueurs Obtained with Different Elaboration Techniques
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
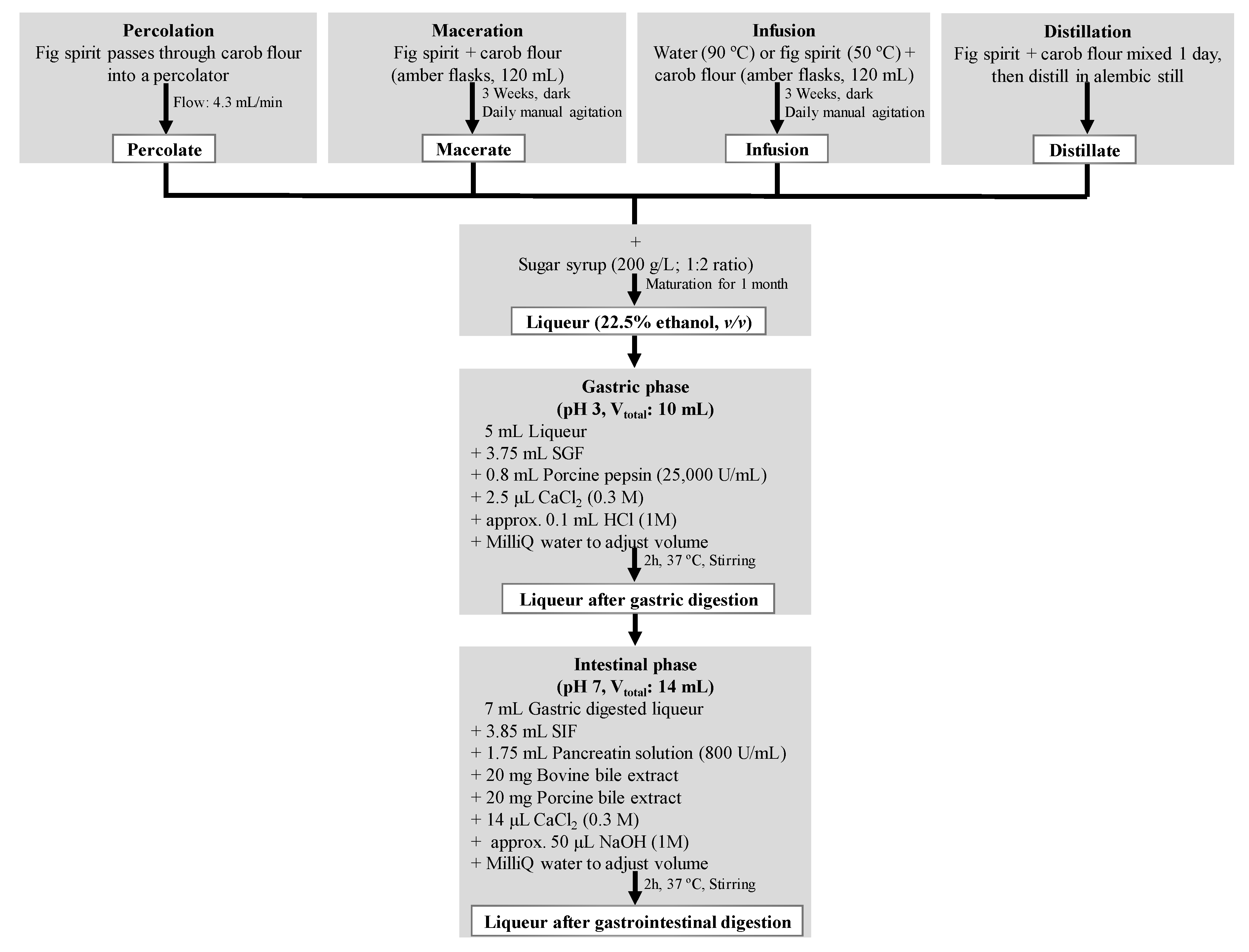
2.2. Production and Digestion Processes of Carob Liqueurs
2.3. Identification and Quantification of the Phenolic and Furanic Compounds by HPLC-PDA Analysis
2.4. Total Phenolic (TPC) and Flavonoid (TFC) Contents, and Antioxidant Capacities by Trolox Equivalent Antioxidant Capacity (TEAC) and Oxygen Radical Absorption Capacity (ORAC) Assays
2.4.1. TPC by Folin-Ciocalteu Method
2.4.2. TFC by Aluminium Chloride Method
2.4.3. TEAC Method (ABTS Free Radical Scavenging Assay)
2.4.4. ORAC Assay
2.5. Enzyme Inhibitory Capacities
2.5.1. Acetylcholinesterase (AChE) Activity Method
2.5.2. Tyrosinase Inhibition
2.5.3. α-Amylase Assay
2.5.4. α-Glucosidase Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Stability of the Phenolic and Furanic Compounds of Carob Liqueurs during Simulated In Vitro Gastrointestinal Digestion
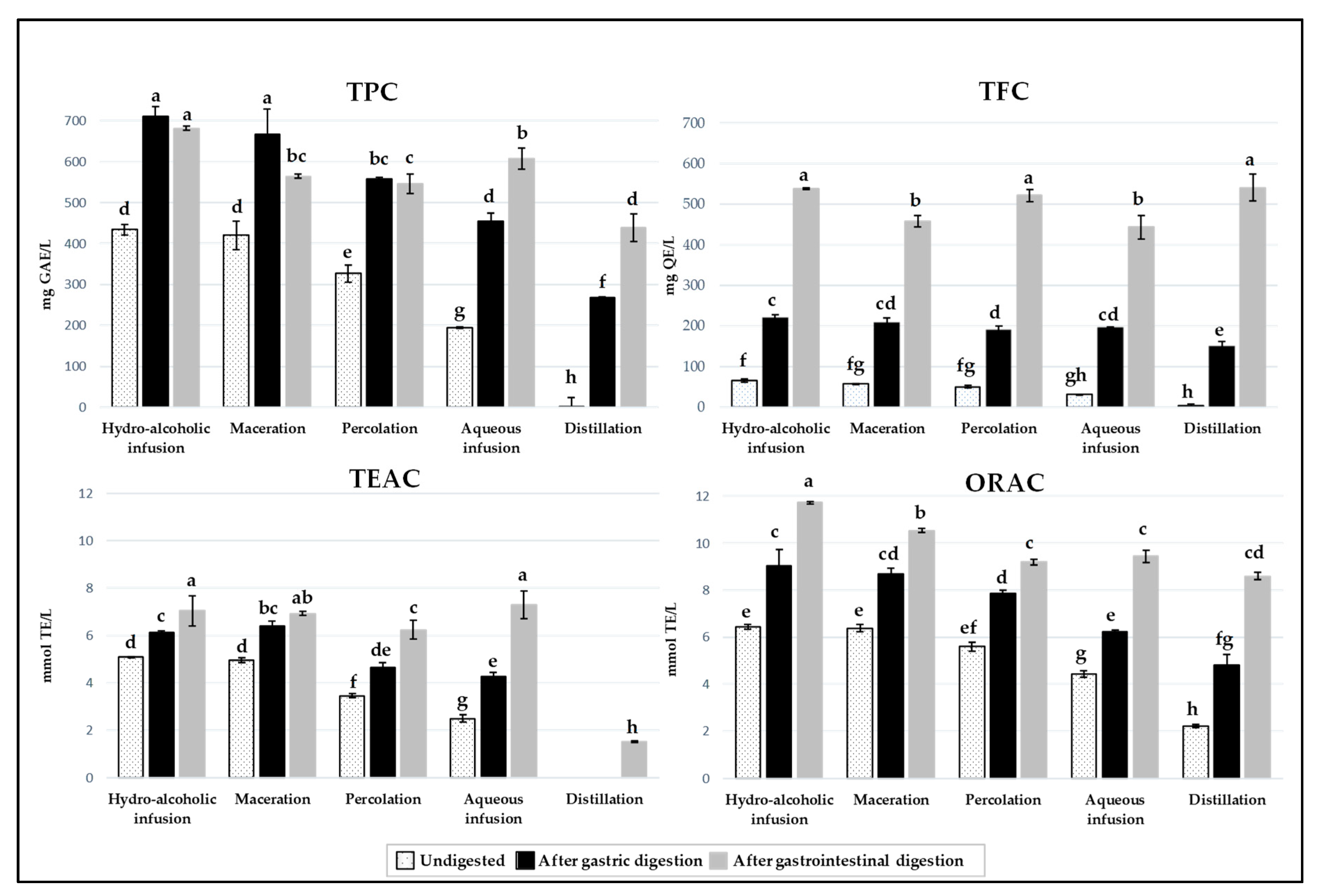
3.2. Evaluation of the Total Phenolic and Flavonoid Contents and Antioxidant Capacity of Carob Liqueurs
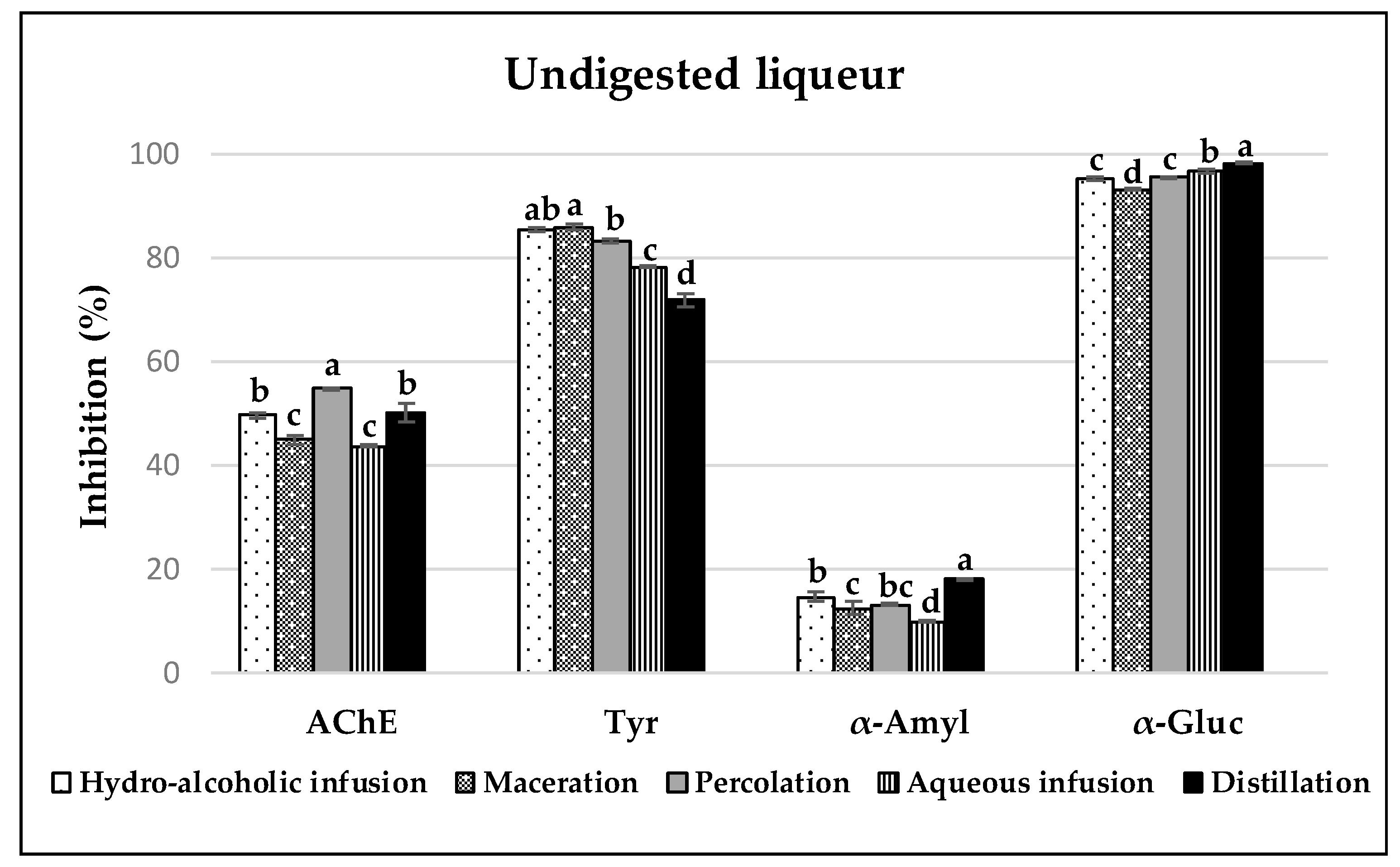
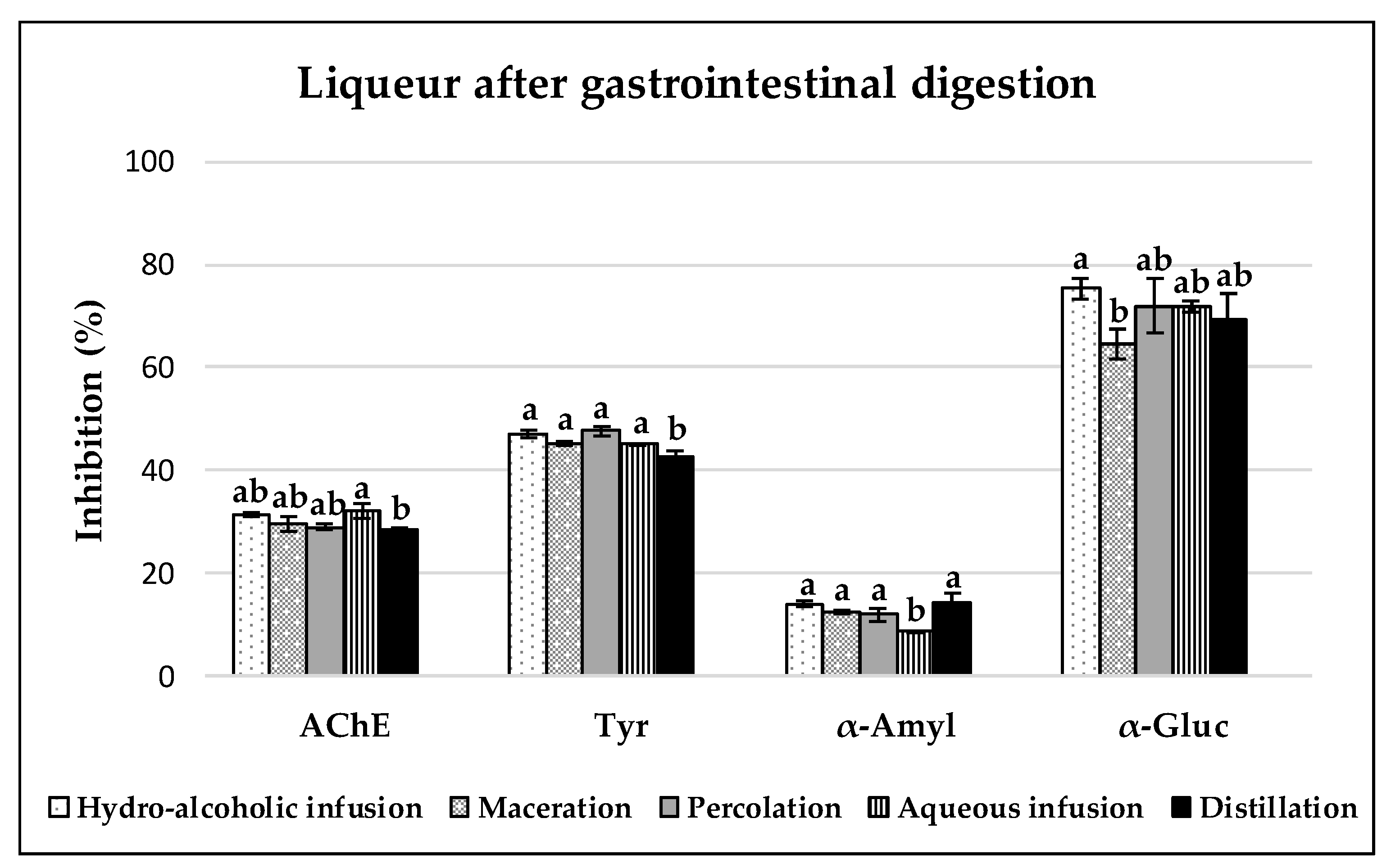
3.3. Enzyme Inhibitory Capacities of Carob Liqueurs
3.3.1. Acetylcholinesterase (AchE) Inhibition
3.3.2. Tyrosinase Inhibition
3.3.3. α-Amylase and α-Glucosidase Inhibition
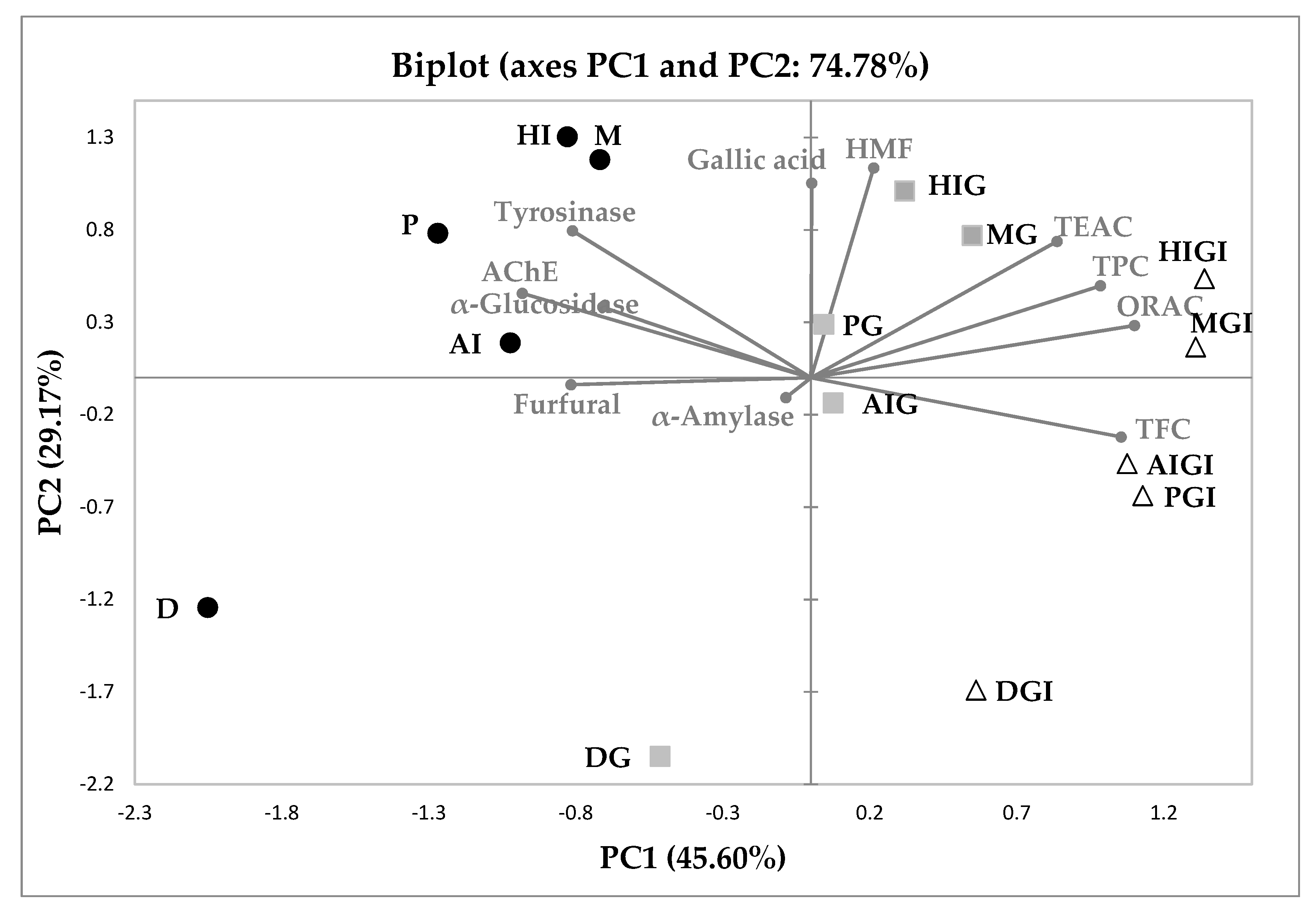
3.4. Principal Component Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Custódio, L.; Escapa, A.L.; Fernandes, E.; Fajardo, A.; Aligué, R.; Alberício, F.; Neng, N.; Florêncio Nogueira, J.M.; Romano, A. Phytochemical profile, antioxidant and cytotoxic activities of the carob tree (Ceratonia siliqua L.) germ flour extracts. Plant Foods Hum. Nutr. 2011, 66, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Youssef, M.K.E.; El-Manfaloty, M.M.; Ali, H.M. Assessment of proximate chemical composition, nutritional status, fatty acid composition and phenolic compounds of carob (Ceratonia siliqua L.). Food Public Health 2013, 3, 304–308. [Google Scholar] [CrossRef]
- Čepo, D.V.; Mornar, A.; Nigović, B.; Kremer, D.; Radanović, D.; Dragojević, I.V. Optimization of roasting conditions as an useful approach for increasing antioxidant activity of carob powder. LWT-Food Sci. Technol. 2014, 58, 578–586. [Google Scholar] [CrossRef]
- Ydjedd, S.; Bouriche, S.; López-Nicolás, R.; Sánchez-Moya, T.; Frontela-Saseta, C.; Ros-Berruezo, G.; Rezgui, F.; Louaileche, H.; Kati, D.E. Effect of in vitro gastrointestinal digestion on encapsulated and nonencapsulated phenolic compounds of carob (Ceratonia siliqua L.) pulp extracts and their antioxidant capacity. J. Agric. Food Chem. 2017, 65, 827–835. [Google Scholar] [CrossRef]
- Goulas, V.; Hadjisolomou, A. Dynamic changes in targeted phenolic compounds and antioxidant potency of carob fruit (Ceratonia siliqua L.) products during in vitro digestion. LWT-Food Sci. Technol. 2019, 101, 269–275. [Google Scholar] [CrossRef]
- Custódio, L.; Fernandes, E.; Escapa, A.L.; Fajardo, A.; Aligué, R.; Alberício, F.; Neng, N.R.; Nogueira, J.M.F.; Romano, A. Antioxidant and cytotoxic activities of carob tree fruit pulps are strongly influenced by gender and cultivar. J. Agric. Food Chem. 2011, 59, 7005–7012. [Google Scholar] [CrossRef]
- Custódio, L.; Patarra, J.; Alberício, F.; Neng, N.R.; Nogueira, J.M.F.; Romano, A. In vitro antioxidant and inhibitory activity of water decoctions of carob tree (Ceratonia siliqua L.) on cholinesterases, α-amylase and α-glucosidase. Nat. Prod. Res. 2015, 29, 2155–2159. [Google Scholar] [CrossRef]
- Corsi, L.; Avallone, R.; Cosenza, F.; Farina, F.; Baraldi, C.; Baraldi, M. Antiproliferative effects of Ceratonia siliqua L. on mouse hepatocellular carcinoma cell line. Fitoterapia 2002, 73, 674–684. [Google Scholar] [CrossRef]
- Rodríguez-Solana, R.; Salgado, J.M.; Pérez-Santín, E.; Romano, A. Effect of carob variety and roasting on the antioxidant capacity, and the phenolic and furanic contents of carob liquors. J. Sci. Food Agric. 2019, 99, 2697–2707. [Google Scholar] [CrossRef]
- Rodríguez-Solana, R.; Vázquez-Araújo, L.; Salgado, J.M.; Domínguez, J.M.; Cortés-Diéguez, S. Optimization of the process of aromatic and medicinal plant maceration in grape marc distillates to obtain herbal liqueurs and spirits. J. Sci. Food Agric. 2016, 96, 4760–4771. [Google Scholar] [CrossRef]
- Śliwińska, M.; Wiśniewska, P.; Dymerski, T.; Wardencki, W.; Namieśnik, J. The flavour of fruit spirits and fruit liqueurs: A review. Flavour Fragr. J. 2015, 30, 197–207. [Google Scholar] [CrossRef]
- Naviglio, D.; Scarano, P.; Ciaravolo, M.; Gallo, M. Rapid Solid-Liquid Dynamic Extraction (RSLDE): A powerful and greener alternative to the latest solid-liquid extraction techniques. Foods 2019, 8, 245. [Google Scholar] [CrossRef] [PubMed]
- Coldea, T.E.; Mudura, E. Valorisation of aromatic plants in beverage industry: A review. HOP Med. Plants 2015, 23, 25–33. [Google Scholar]
- Gulling, R.; Vargas, P. Cordials from Your Kitchen: Easy, Elegant Liqueurs You Can Make & Give; Storey Publishing: North Adams, MA, USA, 1997; p. 10. [Google Scholar]
- Li, W.; Beta, T. Evaluation of antioxidant capacity and aroma quality of anthograin liqueur. Food Chem. 2011, 127, 968–975. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects–A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Christou, C.; Poulli, E.; Yiannopoulos, S.; Agapiou, A. GC–MS analysis of D-pinitol in carob: Syrup and fruit (flesh and seed). J. Chromatogr. B 2019, 1116, 60–64. [Google Scholar] [CrossRef]
- Nasar-Abbas, S.M.; e-Huma, Z.; Vu, T.H.; Khan, M.K.; Esbenshade, H.; Jayasena, V. Carob Kibble: A Bioactive-Rich Food Ingredient. Compr. Rev. Food Sci. Saf. 2016, 15, 63–72. [Google Scholar] [CrossRef]
- Attri, S.; Singh, N.; Singh, T.R.; Goel, G. Effect of in vitro gastric and pancreatic digestion on antioxidant potential of fruit juices. Food Biosci. 2017, 17, 1–6. [Google Scholar] [CrossRef]
- Ryan, L.; Prescott, S.L. Stability of the antioxidant capacity of twenty-five commercially available fruit juices subjected to an in vitro digestion. Int. J. Food Sci. Technol. 2010, 45, 1191–1197. [Google Scholar] [CrossRef]
- Gutiérrez-grijalva, E.P.; Ambriz-pére, D.L.; Leyva-lópez, N.; Castillo-lópez, R.I.; Heredia, J.B. Review: Dietary phenolic compounds, health benefits and bioaccessibility. Arch. Latinoam. Nutr. 2016, 66, 87–100. [Google Scholar]
- Ortega, N.; Macià, A.; Romero, M.P.; Reguant, J.; Motilva, M.J. Matrix composition effect on the digestibility of carob flour phenols by an in-vitro digestion model. Food Chem. 2011, 124, 65–71. [Google Scholar] [CrossRef]
- Mehta, M.; Adem, A.; Sabbagh, M. New acetylcholinesterase inhibitors for Alzheimer’s disease. Int. J. Alzh. Dis. 2012. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Treis, A.; Patenge, N.; Fiesel, F.C.; Springer, W.; Kahle, P.J. Parkin protects against tyrosinase-mediated dopamine neurotoxicity by suppressing stress-activated protein kinase pathways. J. Neurochem. 2008, 105, 1700–1715. [Google Scholar] [CrossRef] [PubMed]
- Balogun, F.O.; Ashafa, A.O.T. Aqueous root extracts of Dicoma anomala (Sond.) extenuates postprandial hyperglycaemia in vitro and its modulation on the activities of carbohydrate-metabolizing enzymes in streptozotocin-induced diabetic Wistar rats. S. Afr. J. Bot. 2017, 112, 102–111. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Correig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food–an international consensus. Food funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef]
- Jara-Palacios, M.J.; Gonçalves, S.; Hernanz, D.; Heredia, F.J.; Romano, A. Effects of in vitro gastrointestinal digestion on phenolic compounds and antioxidant activity of different white winemaking byproducts extracts. Food Res. Int. 2018, 109, 433–439. [Google Scholar] [CrossRef]
- Otles, S.; Yalcin, B. Phenolic compounds analysis of root, stalk, and leaves of nettle. Sci. World J. 2012, 2012, 564367. [Google Scholar] [CrossRef]
- Woisky, R.G.; Salatino, A. Analysis of propolis: Some parameters and procedures for chemical quality control. J. Apic. Res. 1998, 37, 99–105. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Gillespie, K.; Chae, J.; Ainsworth, E. Rapid measurement of total antioxidant capacity in plants. Nat. Protoc. 2007, 2, 867–870. [Google Scholar] [CrossRef]
- Ellman, G.L.; Lourtney, D.K.; Andres, V.; Gmelin, G. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Ingkaninan, K.; Yuyaem, T.; Thongnoi, W. Anti-cholinesterase activity in Stephania venosa Spreng. Naresuan Univ. J. 2001, 9, 35–40. [Google Scholar]
- Masuda, T.; Yamashita, D.; Takeda, Y.; Yonemori, S. Screening for tyrosinase inhibitors among extracts of seashore plants and identification of potent inhibitors from Garcinia subelliptica. Biosci. Biotechnol. Biochem. 2005, 69, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Houghton, P.J.; Soumyanath, A. A-Amylase inhibitory activity of some Malaysian plants used to treat diabetes; with particular reference to Phyllanthus amarus. J. Ethnopharmacol. 2006, 107, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.I.; Apostolidis, E.; Shetty, K. In vitro studies of eggplant (Solanum melongena) phenolics as inhibitors of key enzymes relevant for type 2 Diabetes and hypertension. Bioresour. Technol. 2008, 99, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Papagiannopoulos, M.; Wollseifen, H.R.; Mellenthin, A.; Haber, B.; Galensa, R. Identification and quantification of polyphenols in Carob Fruits (Ceratonia siliqua L.) and derived products by HPLC-UV-ESI/MS n. J. Agric. Food Chem. 2004, 52, 3784–3791. [Google Scholar] [CrossRef] [PubMed]
- Owen, R.W.; Haubner, R.; Hull, W.E.; Erben, G.; Spiegelhalder, B.; Bartsch, H.; Haber, B. Isolation and structure elucidation of the major individual polyphenols in carob fibre. Food Chem. Toxicol. 2003, 41, 1727–1738. [Google Scholar] [CrossRef]
- Daneshfar, A.; Ghaziaskar, H.S.; Homayoun, N. Solubility of gallic acid in methanol, ethanol, water, and ethyl acetate. J. Chem. Eng. Data 2008, 53, 776–778. [Google Scholar] [CrossRef]
- Kallithraka, S.; Garcia-Viguera, C.; Bridle, P.; Bakker, J. Survey of solvents for the extraction of grape seed phenolics. Phytochem. Anal. 1995, 6, 265–267. [Google Scholar] [CrossRef]
- Lingua, M.S.; Wunderlin, D.A.; Baroni, M.V. Effect of simulated digestion on the phenolic components of red grapes and their corresponding wines. J. Funct. Foods 2018, 44, 86–94. [Google Scholar] [CrossRef]
- Chen, G.L.; Chen, S.G.; Chen, F.; Xie, Y.Q.; Han, M.D.; Luo, C.X.; Zhao, Y.Y.; Gao, Y.Q. Nutraceutical potential and antioxidant benefits of selected fruit seeds subjected to an in vitro digestion. J. Funct. Foods 2016, 20, 317–331. [Google Scholar] [CrossRef]
- Hamzalıoğlu, A.; Gökmen, V. Formation and elimination reactions of 5-hydroxymethylfurfural during in vitro digestion of biscuits. Food Res. Int. 2017, 99, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Solana, R.; Carlier, J.D.; Costa, M.C.; Romano, A. Multi-element characterisation of carob, fig and almond liqueurs by MP-AES. J. Inst. Brew. 2018, 124, 300–309. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kim, D.B.; Lee, J.; Cho, J.H.; Kim, B.; Choi, H.S.; Lee, B.H.; Lee, O.H. Antioxidant activity and anti-adipogenic effects of wild herbs mainly cultivated in Korea. Molecules 2013, 18, 12937–12950. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Torun, H.; Ayaz, S.; Correia, P.J.; Alaiz, M.; Sanz, C.; Grúz, J.; Strnad, M. Determination of chemical composition of anatolian carob pod (Ceratonia siliqua L.): Sugars, amino and organic acids, minerals and phenolic compounds. J. Food Qual. 2007, 30, 1040–1055. [Google Scholar] [CrossRef]
- Gubbuk, H.; Kafkas, E.; Guven, D.; Gunes, E. Physical and phytochemical profile of wild and domesticated carob (Ceratonia siliqua L.) genotypes. Span. J. Agric. Res. 2010, 8, 1129–1136. [Google Scholar] [CrossRef]
- Kowalski, S. Changes of antioxidant activity and formation of 5-hydroxymethylfurfural in honey during thermal and microwave processing. Food Chem. 2013, 141, 1378–1382. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Cengiz, M.; Uren, M.C.; Ceylan, O.; Orenc, T.; Tepe, B. Phenolic composition, enzyme inhibitory, and antioxidant activity of Bituminaria bituminosa. Food Sci. Biotechnol. 2016, 25, 1299–1304. [Google Scholar] [CrossRef]
- Russo, D.; Valentão, P.; Andrade, P.; Fernandez, E.; Milella, L. Evaluation of antioxidant, antidiabetic and anticholinesterase activities of Smallanthus sonchifolius landraces and correlation with their phytochemical profiles. Int. J. Mol. Sci. 2015, 16, 17696–17718. [Google Scholar] [CrossRef] [PubMed]
- Howes, M.J.R.; Perry, N.S.; Houghton, P.J. Plants with traditional uses and activities, relevant to the management of Alzheimer’s disease and other cognitive disorders. Phytother. Res. 2003, 17, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Khadhri, A.; Bouali, I.; Belkhir, S.; Mokded, R.; Smiti, S.; Falé, P.; Araújo, M.E.M.; Serralheiro, M.L.M. In vitro digestion, antioxidant and antiacetylcholinesterase activities of two species of Ruta: Ruta chalepensis and Ruta Montana. Pharm. Biol. 2017, 55, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Yur, S.; Tekin, M.; Göger, F.; Başer, K.H.; Özek, T.; Özek, G. Composition and potential of Tanacetum haussknechtii Bornm. Grierson as antioxidant and inhibitor of acetylcholinesterase, tyrosinase, and α-amylase enzymes. Int. J. Food Prop. 2017, 20 (Suppl. 3), S2359–S2378. [Google Scholar] [CrossRef]
- Abidar, S.; Yildiz, O.; Degirmenci, A.; Amakran, A.; El Maadoudi, M.; Nhiri, M. Glucose-mediated protein glycation: Contribution of methanolic extract of Ceratonia siliqua L. in protection and in vitro potential inhibition of acetylcholinesterase. J. Food Biochem. 2019, 43, e13009. [Google Scholar] [CrossRef]
- Cha, S.H.; KO, S.C.; Kim, D.; Jeon, Y.J. Screening of marine algae for potential tyrosinase inhibitor: Those inhibitors reduced tyrosinase activity and melanin synthesis in zebrafish. J. Dermatol. 2011, 38, 354–363. [Google Scholar] [CrossRef]
- Fachinetti, N.; Rigon, R.B.; Eloy, J.O.; Sato, M.R.; dos Santos, K.C.; Chorilli, M. Comparative study of glyceryl behenate or polyoxyethylene 40 stearate-based lipid carriers for trans-resveratrol delivery: Development, characterization and evaluation of the in vitro tyrosinase inhibition. AAPS PharmSciTech. 2018, 19, 1401–1409. [Google Scholar] [CrossRef]
- Ferri, M.; Rondini, G.; Calabretta, M.M.; Michelini, E.; Vallini, V.; Fava, F.; Roda, A.; Minnucci, G.; Tassoni, A. White grape pomace extracts, obtained by a sequential enzymatic plus ethanol-based extraction, exert antioxidant, anti-tyrosinase and anti-inflammatory activities. New Biotechnol. 2017, 39, 51–58. [Google Scholar] [CrossRef]
- Momtaz, S. Tyrosinase Inhibitors Isolated from Ceratonia siliqua (L.) and Sideroxylon inerme (L.). Master’s Thesis, University of Pretoria, Pretoria, South Africa, 2007. [Google Scholar]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. In vitro antioxidant, anti-diabetic, cholinesterase and tyrosinase inhibitory potential of fresh juice from Citrus hystrix and C. maxima fruits. Food Sci. Hum. Wellness 2014, 3, 16–25. [Google Scholar] [CrossRef]
- Dong, H.Q.; Li, M.; Zhu, F.; Liu, F.L.; Huang, J.B. Inhibitory potential of trilobatin from Lithocarpus polystachyus Rehd against α-glucosidase and α-amylase linked to type 2 diabetes. Food Chem. 2012, 130, 261–266. [Google Scholar] [CrossRef]
- Oboh, G.; Ogunsuyi, O.B.; Ogunbadejo, M.D.; Adefegha, S.A. Influence of gallic acid on α-amylase and α-glucosidase inhibitory properties of acarbose. J. Food Drug Anal. 2016, 24, 627–634. [Google Scholar] [CrossRef] [PubMed]
| Compound | RT (min) | Calibration Range (mg/L) | Regression Equation y = ax + b | r2 | LOD (mg/L) | LOQ (mg/L) |
|---|---|---|---|---|---|---|
| Gallic acid | 4.75 | 5–55 | y = 33.901x − 15.493 | 0.999 | 1.97 | 6.58 |
| HMF | 6.45 | 1–35 | y = 123.37x + 63.257 | 0.999 | 1.26 | 4.20 |
| Furfural | 9.65 | 1–20 | y = 149.39x + 19.349 | 1 | 0.40 | 1.32 |
| Liqueur Elaboration | Digestion Process | Concentration (mg/L) | ||
|---|---|---|---|---|
| Method of Extraction | Liqueur | Gallic Acid | HMF | Furfural |
| Hydro-alcoholic infusion | Undigested | 95.25 ± 1.39 a | 61.95 ± 0.73 a | 17.80 ± 0.07 c,d |
| After gastric digestion | 93.57 ± 1.38 a | 61.30 ± 0.36 a | 17.63 ± 0.22 c,d | |
| After gastrointestinal digestion | 86.86 ± 4.37 a,b,c | 52.49 ± 1.27 b | 14.02 ± 1.65 f | |
| Maceration | Undigested | 94.60 ± 3.27 a | 58.98 ± 0.57 a | 16.96 ± 0.62 d,e |
| After gastric digestion | 92.05 ± 0.30 a,b | 59.20 ± 0.02 a | 17.56 ± 0.04 c,d | |
| After gastrointestinal digestion | 82.75 ± 8.06 b,c,d | 42.57 ± 0.65 d | 11.09 ± 0.05 g | |
| Percolation | Undigested | 76.02 ± 9.17 b | 47.60 ± 6.13 c | 19.25 ± 0.27 a,b,c |
| After gastric digestion | 68.82 ± 7.31 e | 45.46 ± 4.60 c,d | 18.26 ± 1.69 b,c,d | |
| After gastrointestinal digestion | <LOQ | 28.33 ± 2.34 g | 11.01 ± 1.85 g | |
| Undigested | 74.96 ± 4.67 d,e | 33.21 ± 1.33 e,f | 12.77 ± 0.98 f,g | |
| Aqueous infusion | After gastric digestion | 79.70 ± 4.05 c,d | 35.72 ± 0.83 e | 17.12 ± 1.24 c,d,e |
| After gastrointestinal digestion | <LOD | 29.77 ± 0.57 f,g | 12.88 ± 0.85 f,g | |
| Distillation | Undigested | n.d. | <LOD | 20.86 ± 1.84 a |
| After gastric digestion | n.d. | <LOD | 20.25 ± 0.58 a,b | |
| After gastrointestinal digestion | n.d. | <LOD | 14.97 ± 0.95 e,f | |
| Fig spirit | Undigested | n.d. | n.d. | 38.51 ± 0.70 |
| Variables | GA | TPC | TFC | TEAC | ORAC | F | HMF | AchE | Tyr | α-Gluc | α-Amyl |
|---|---|---|---|---|---|---|---|---|---|---|---|
| GA | 1 | 0.370 | −0.296 | 0.478 | 0.202 | 0.031 | 0.891 | 0.243 | 0.603 | 0.127 | 0.114 |
| TPC | 1 | 0.615 | 0.862 | 0.914 | −0.405 | 0.598 | −0.560 | −0.277 | −0.500 | 0.128 | |
| TFC | 1 | 0.473 | 0.838 | −0.699 | −0.135 | −0.782 | −0.829 | −0.443 | −0.233 | ||
| TEAC | 1 | 0.811 | −0.563 | 0.741 | −0.315 | −0.069 | −0.169 | −0.203 | |||
| ORAC | 1 | −0.629 | 0.399 | −0.640 | −0.514 | −0.424 | −0.129 | ||||
| F | 1 | −0.088 | 0.509 | 0.461 | 0.053 | 0.595 | |||||
| HMF | 1 | 0.203 | 0.537 | 0.125 | 0.001 | ||||||
| AchE | 1 | 0.809 | 0.833 | −0.198 | |||||||
| Tyr | 1 | 0.606 | −0.559 | ||||||||
| α-Gluc | 1 | 0.021 | |||||||||
| α-Amyl | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Solana, R.; Coelho, N.; Santos-Rufo, A.; Gonçalves, S.; Pérez-Santín, E.; Romano, A. The Influence of In Vitro Gastrointestinal Digestion on the Chemical Composition and Antioxidant and Enzyme Inhibitory Capacities of Carob Liqueurs Obtained with Different Elaboration Techniques. Antioxidants 2019, 8, 563. https://doi.org/10.3390/antiox8110563
Rodríguez-Solana R, Coelho N, Santos-Rufo A, Gonçalves S, Pérez-Santín E, Romano A. The Influence of In Vitro Gastrointestinal Digestion on the Chemical Composition and Antioxidant and Enzyme Inhibitory Capacities of Carob Liqueurs Obtained with Different Elaboration Techniques. Antioxidants. 2019; 8(11):563. https://doi.org/10.3390/antiox8110563
Chicago/Turabian StyleRodríguez-Solana, Raquel, Natacha Coelho, Antonio Santos-Rufo, Sandra Gonçalves, Efrén Pérez-Santín, and Anabela Romano. 2019. "The Influence of In Vitro Gastrointestinal Digestion on the Chemical Composition and Antioxidant and Enzyme Inhibitory Capacities of Carob Liqueurs Obtained with Different Elaboration Techniques" Antioxidants 8, no. 11: 563. https://doi.org/10.3390/antiox8110563
APA StyleRodríguez-Solana, R., Coelho, N., Santos-Rufo, A., Gonçalves, S., Pérez-Santín, E., & Romano, A. (2019). The Influence of In Vitro Gastrointestinal Digestion on the Chemical Composition and Antioxidant and Enzyme Inhibitory Capacities of Carob Liqueurs Obtained with Different Elaboration Techniques. Antioxidants, 8(11), 563. https://doi.org/10.3390/antiox8110563








