Preparation and Evaluation of Resveratrol-Loaded Composite Nanoparticles Using a Supercritical Fluid Technology for Enhanced Oral and Skin Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
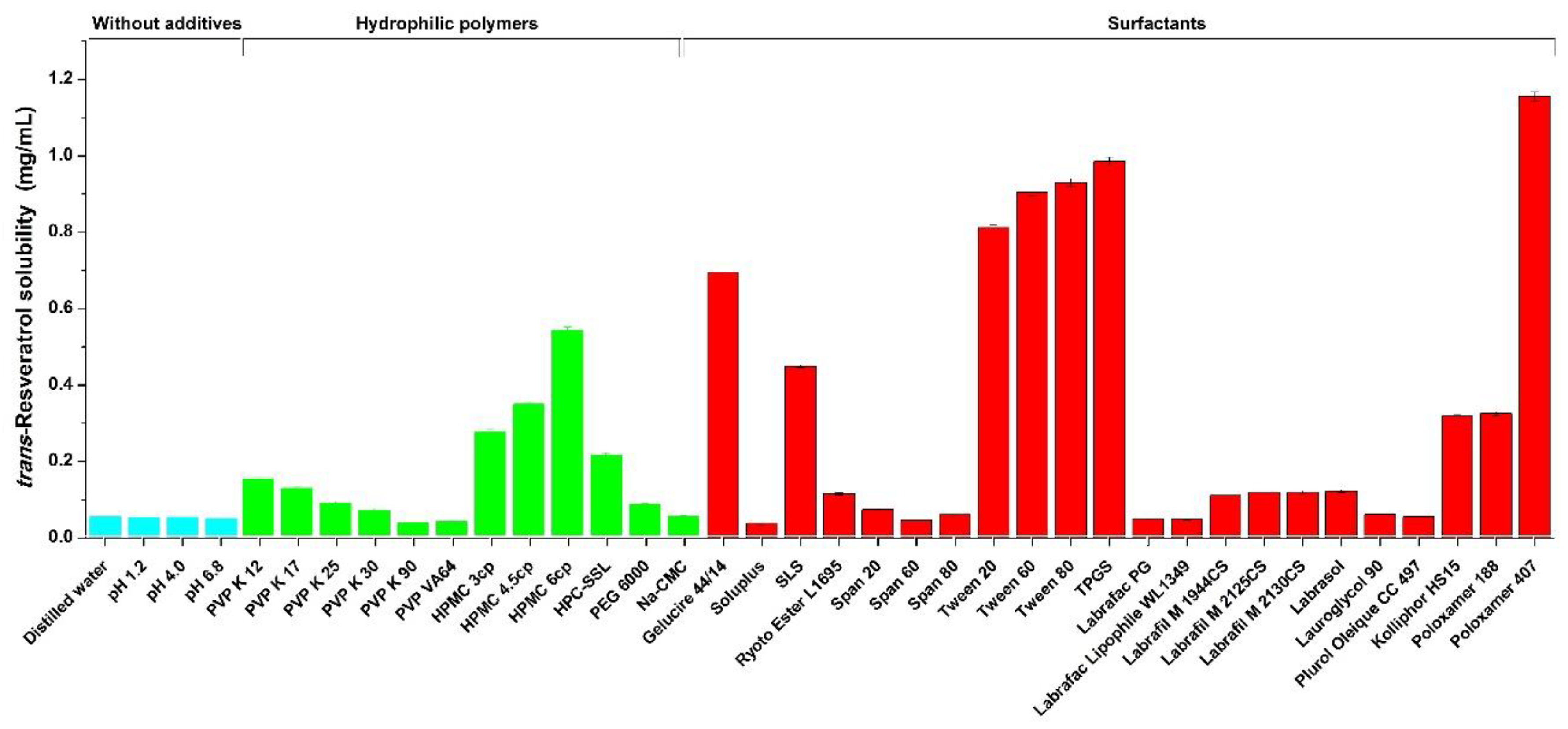
2.2. Solubility Studies of Trans-Resveratrol in Aqueous Solutions Containing Various Additives
2.3. Nanoparticle Preparation Using an SAS Process
2.4. Trans-Resveratrol Content Analysis
2.5. Scanning Electron Microscopy (SEM)
2.6. Particle Size Measurements
2.7. Specific Surface Area Measurements
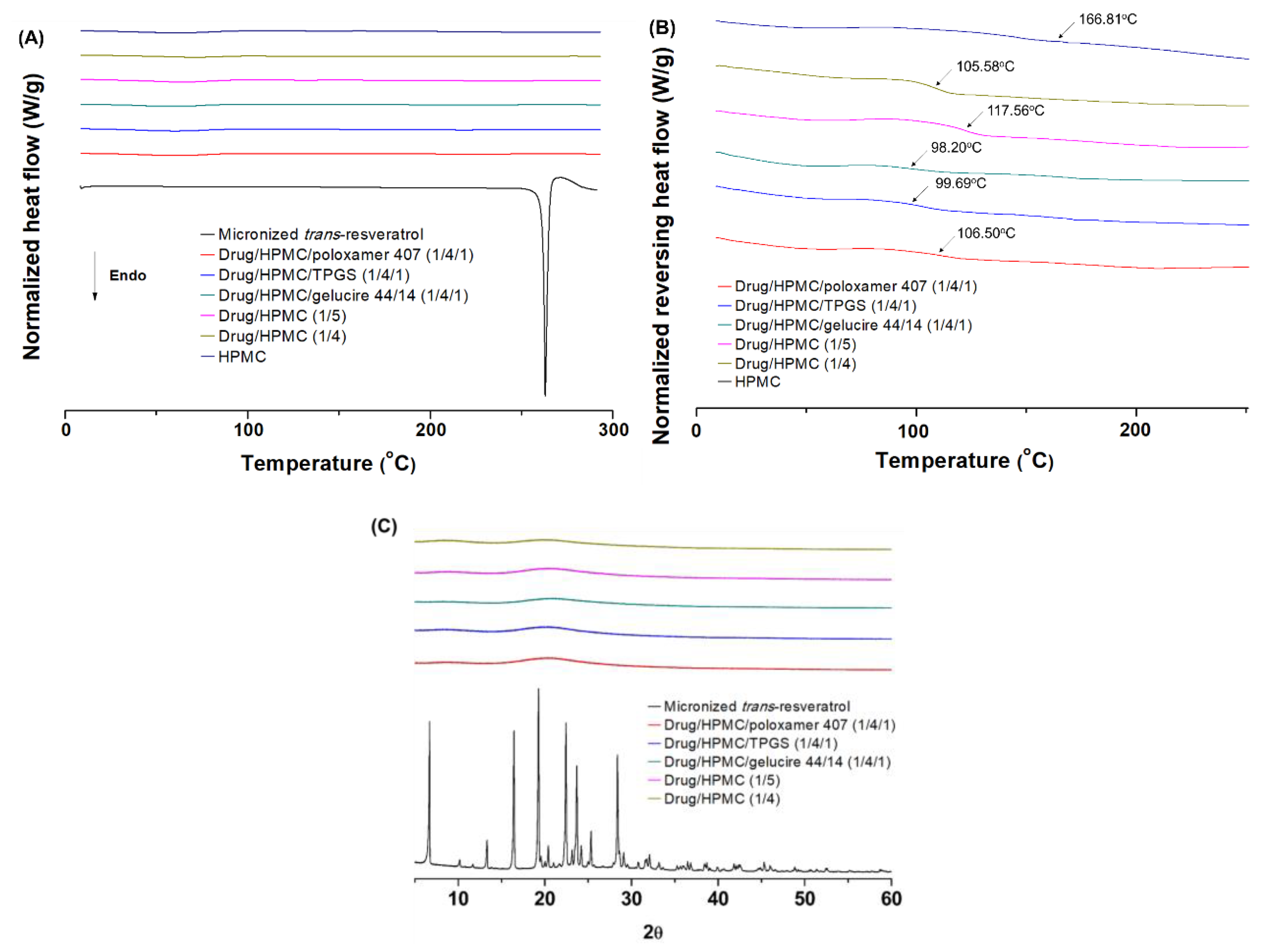
2.8. Differential Scanning Calorimetry (DSC)
2.9. Powder X-ray Diffraction (PXRD)
2.10. Kinetic Solubility Study
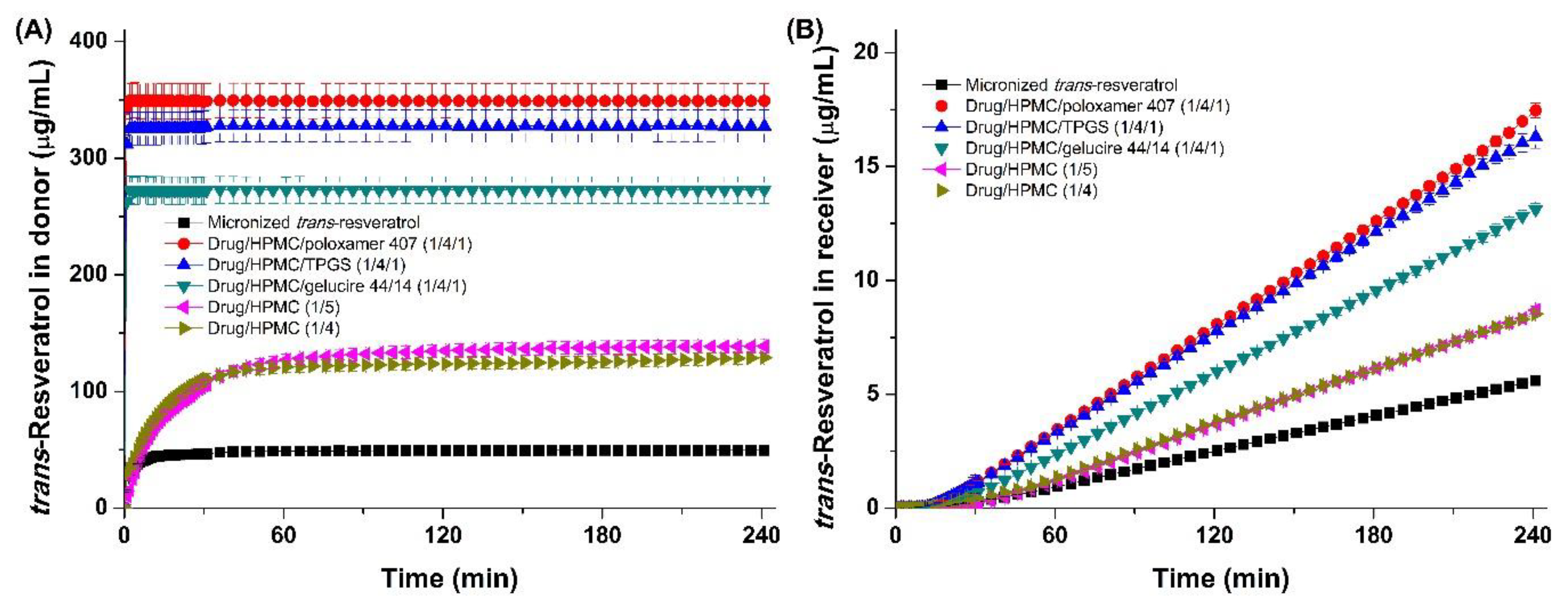
2.11. Flux Measurements via In Vitro Dissolution and Permeation Studies
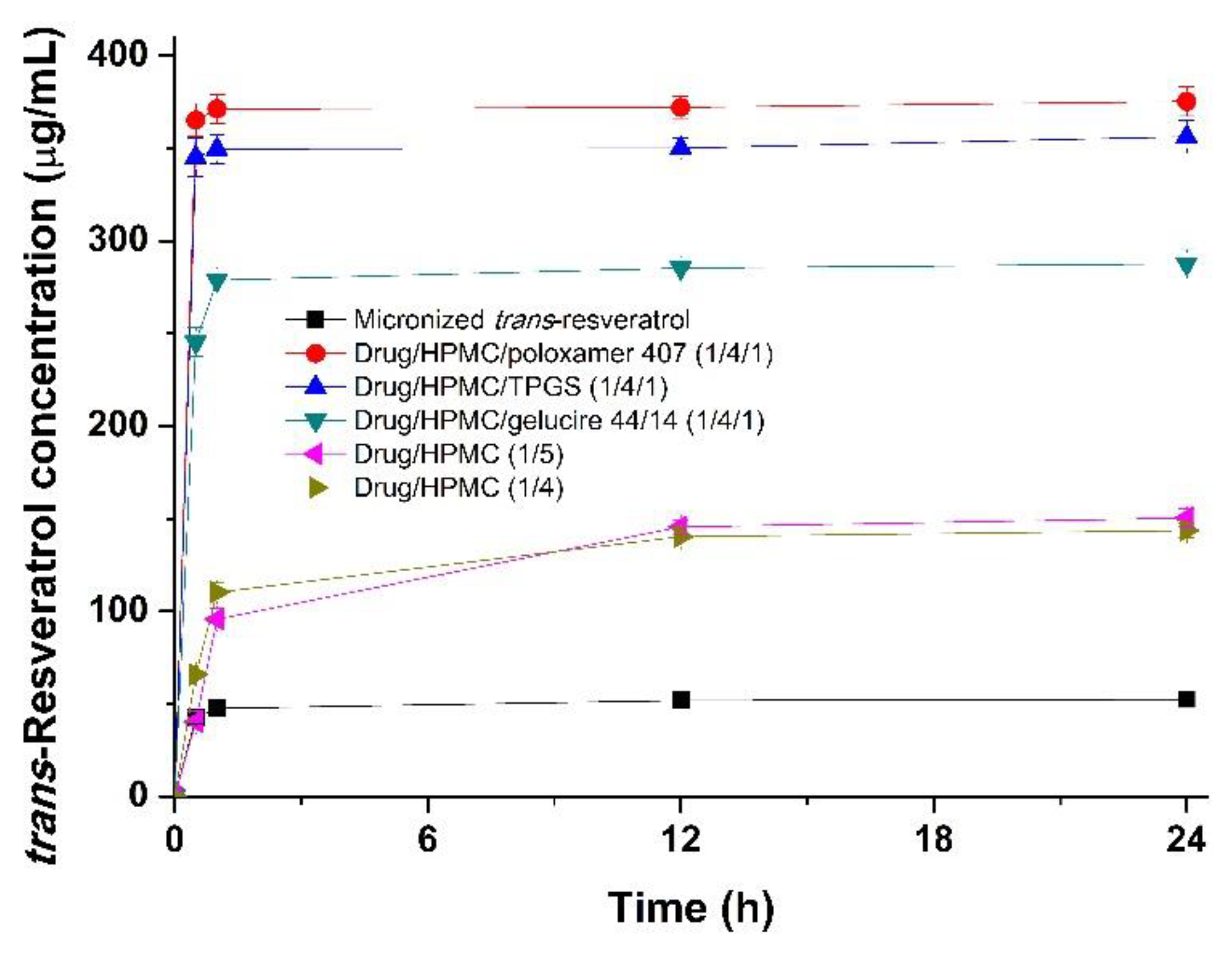
2.12. Pharmacokinetic Study of Oral Delivery in Rats
2.13. Ex Vivo Skin Permeation Study of Skin Delivery
2.14. Data Analysis
3. Results and Discussions
3.1. Preparation and Characterization of Trans-Resveratrol Composite Nanoparticles
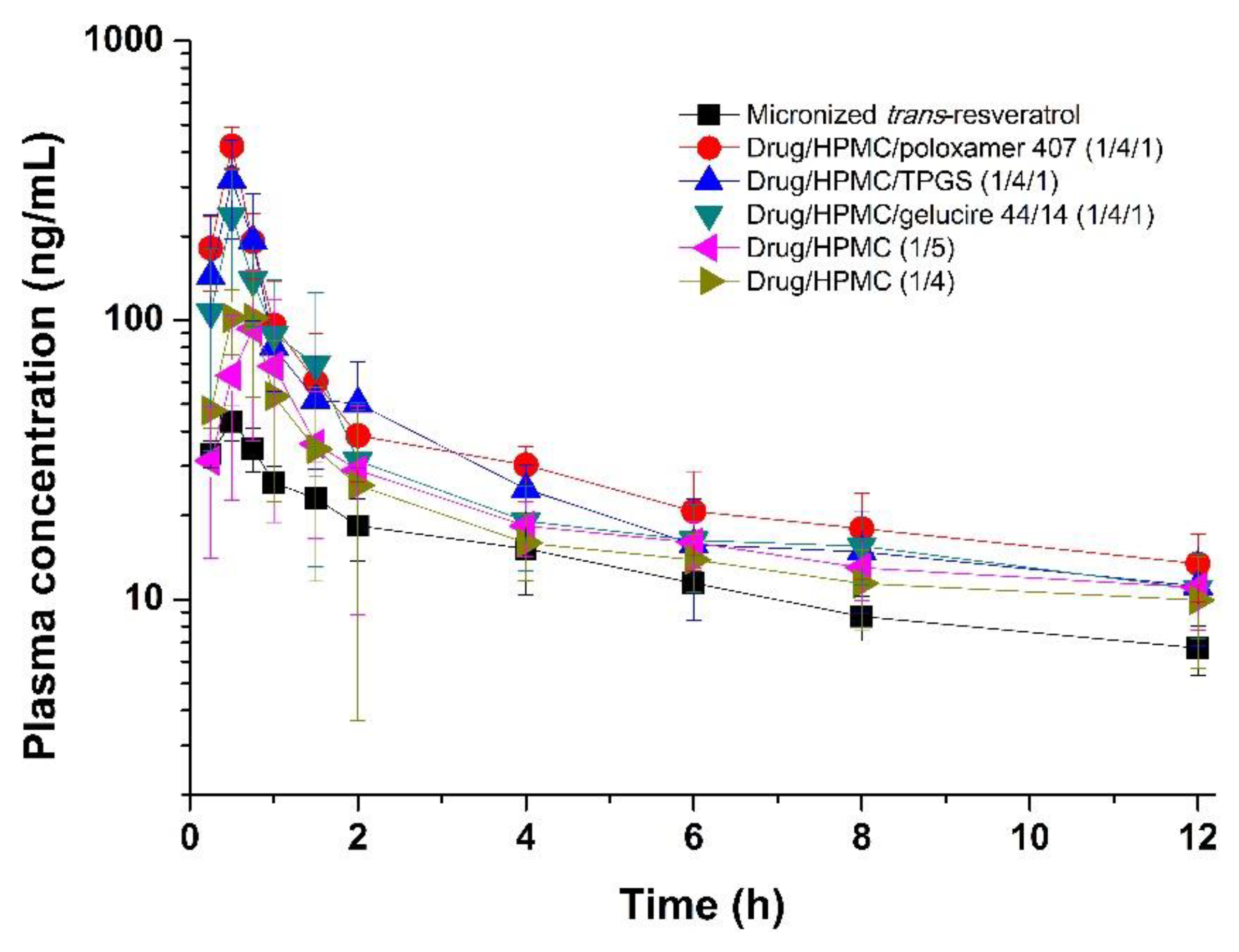
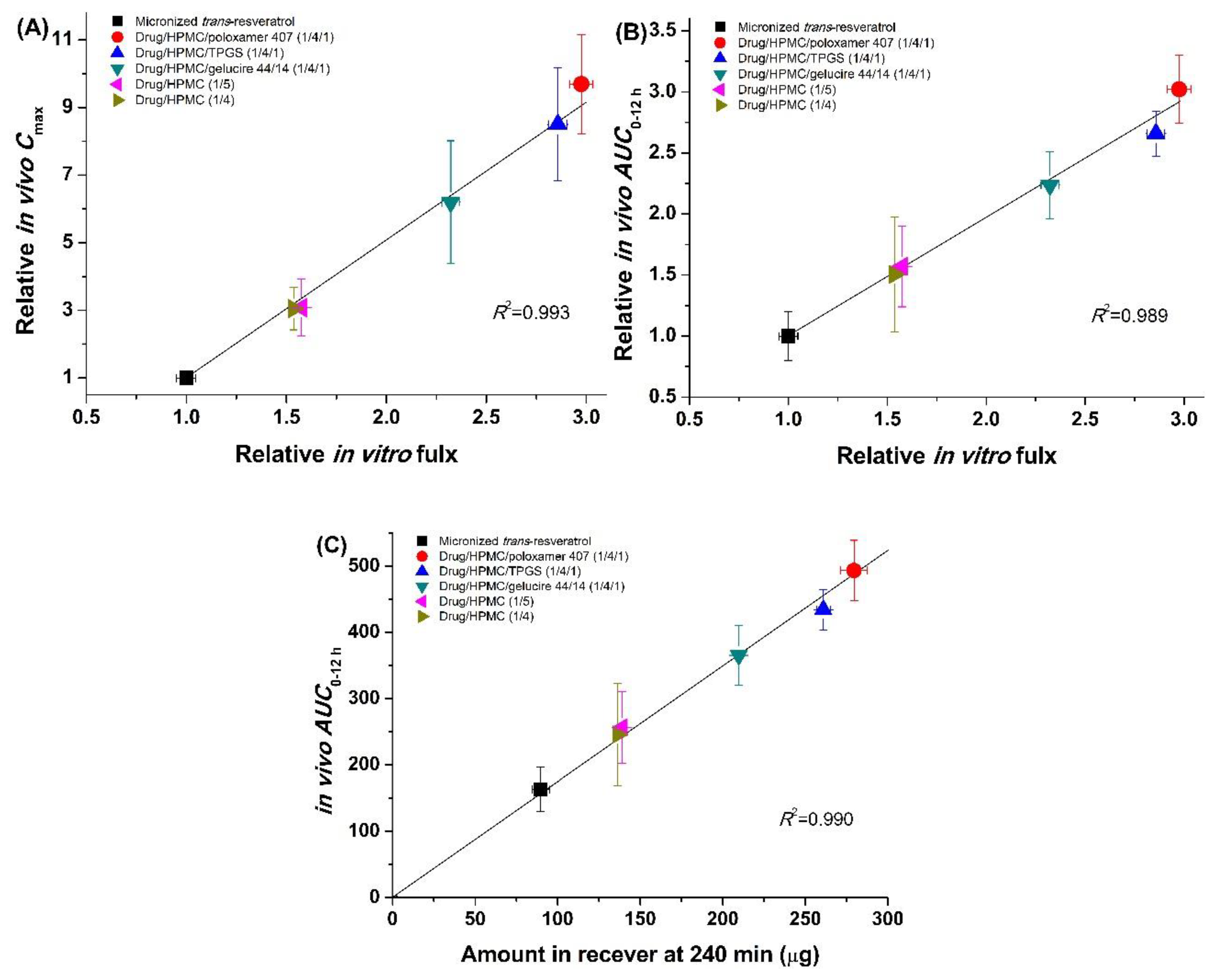
3.2. Use of Trans-Resveratrol Composite Nanoparticles for Oral Delivery
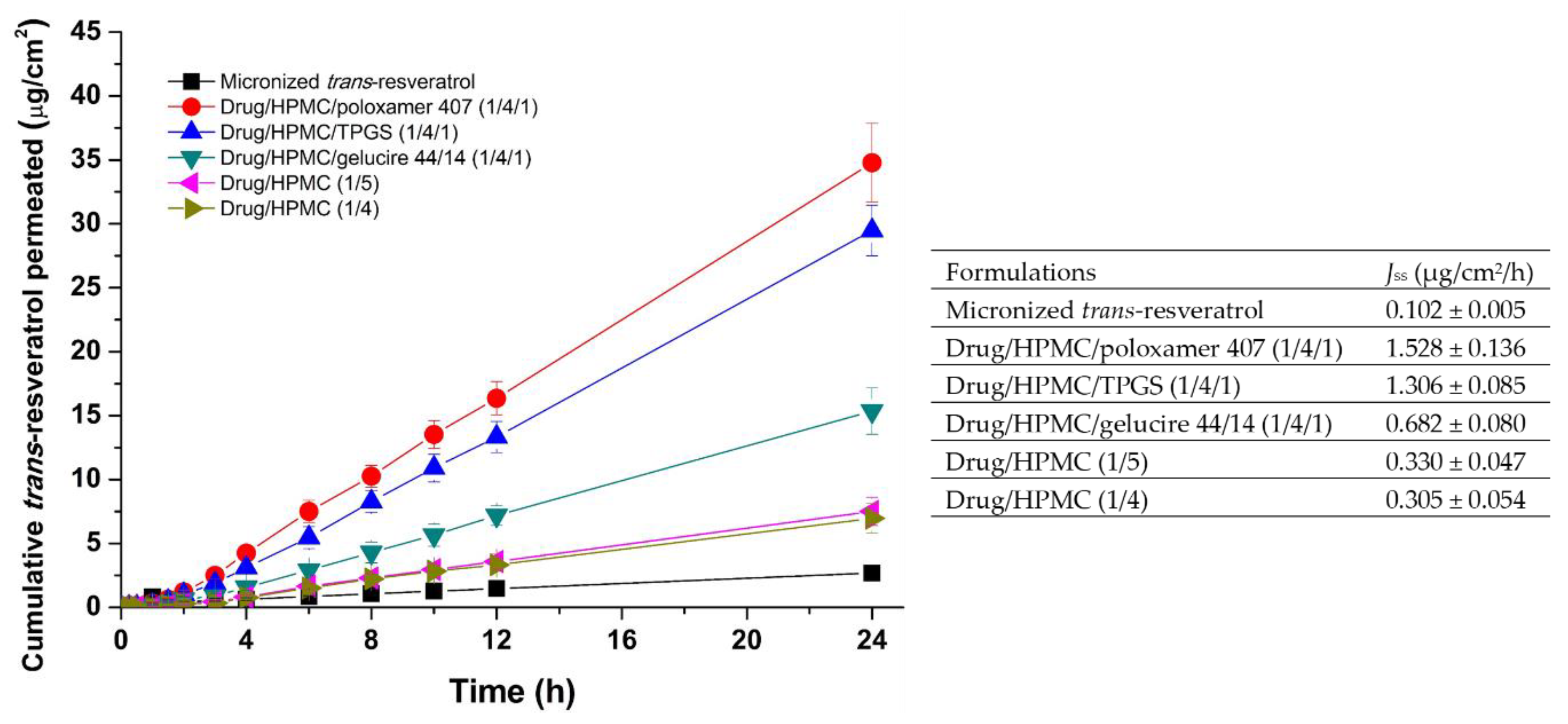
3.3. Utilization of Trans-Resveratrol Composite Nanoparticles for Skin Delivery
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Amri, A.; Chaumeil, J.C.; Sfar, S.; Charrueau, C. Administration of resveratrol: What formulation solutions to bioavailability limitations? J. Control. Release 2012, 158, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shi, M.; Ye, J.-H.; Zheng, X.-Q.; Lu, J.-L.; Liang, Y.-R. Photo-induced chemical reaction of trans-resveratrol. Food Chem. 2015, 171, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef] [PubMed]
- Na, J.-I.; Shin, J.-W.; Choi, H.-R.; Kwon, S.-H.; Park, K.-C. Resveratrol as a Multifunctional Topical Hypopigmenting Agent. Int. J. Mol. Sci. 2019, 20, 956. [Google Scholar] [CrossRef]
- Fonseca, J.; Moradi, F.; Valente, A.J.F.; Stuart, J.A. Oxygen and Glucose Levels in Cell Culture Media Determine Resveratrol’s Effects on Growth, Hydrogen Peroxide Production, and Mitochondrial Dynamics. Antioxidants 2018, 7, 157. [Google Scholar] [CrossRef]
- Chimento, A.; De Amicis, F.; Sirianni, R.; Sinicropi, M.S.; Puoci, F.; Casaburi, I.; Saturnino, C.; Pezzi, V. Progress to Improve Oral Bioavailability and Beneficial Effects of Resveratrol. Int. J. Mol. Sci. 2019, 20, 1381. [Google Scholar] [CrossRef]
- Marier, J.F.; Vachon, P.; Gritsas, A.; Zhang, J.; Moreau, J.P.; Ducharme, M.P. Metabolism and disposition of resveratrol in rats: Extent of absorption, glucuronidation, and enterohepatic recirculation evidenced by a linked-rat model. J. Pharmacol. Exp. Ther. 2002, 302, 369–373. [Google Scholar] [CrossRef]
- Berman, A.Y.; Motechin, R.A.; Wiesenfeld, M.Y.; Holz, M.K. The therapeutic potential of resveratrol: A review of clinical trials. Npj Precis. Oncol. 2017, 1, 35. [Google Scholar] [CrossRef]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef]
- Mamadou, G.; Charrueau, C.; Dairou, J.; Nzouzi, N.L.; Eto, B.; Ponchel, G. Increased intestinal permeation and modulation of presystemic metabolism of resveratrol formulated into self-emulsifying drug delivery systems. Int. J. Pharm. 2017, 521, 150–155. [Google Scholar] [CrossRef]
- Intagliata, S.; Modica, M.N.; Santagati, L.M.; Montenegro, L. Strategies to Improve Resveratrol Systemic and Topical Bioavailability: An Update. Antioxidants 2019, 8, 244. [Google Scholar] [CrossRef] [PubMed]
- Hajjar, B.; Zier, K.-I.; Khalid, N.; Azarmi, S.; Löbenberg, R. Evaluation of a microemulsion-based gel formulation for topical drug delivery of diclofenac sodium. J. Pharm. Investig. 2018, 48, 351–362. [Google Scholar] [CrossRef]
- Jadhav, P.; Bothiraja, C.; Pawar, A. Resveratrol-piperine loaded mixed micelles: Formulation, characterization, bioavailability, safety and in vitro anticancer activity. RSC Adv. 2016, 6, 112795–112805. [Google Scholar] [CrossRef]
- Zu, Y.; Zhang, Y.; Wang, W.; Zhao, X.; Han, X.; Wang, K.; Ge, Y. Preparation and in vitro/in vivo evaluation of resveratrol-loaded carboxymethyl chitosan nanoparticles. Drug Deliv. 2016, 23, 981–991. [Google Scholar] [CrossRef]
- Dhakar, N.K.; Matencio, A.; Caldera, F.; Argenziano, M.; Cavalli, R.; Dianzani, C.; Zanetti, M.; López-Nicolás, J.M.; Trotta, F. Comparative Evaluation of Solubility, Cytotoxicity and Photostability Studies of Resveratrol and Oxyresveratrol Loaded Nanosponges. Pharmaceutics 2019, 11, 545. [Google Scholar] [CrossRef]
- Gartziandia, O.; Lasa, A.; Pedraz, J.L.; Miranda, J.; Portillo, M.P.; Igartua, M.; Hernández, R.M. Preparation and Characterization of Resveratrol Loaded Pectin/Alginate Blend Gastro-Resistant Microparticles. Molecules 2018, 23, 1886. [Google Scholar] [CrossRef]
- Shimojo, A.A.M.; Fernandes, A.R.V.; Ferreira, N.R.E.; Sanchez-Lopez, E.; Santana, M.H.A.; Souto, E.B. Evaluation of the Influence of Process Parameters on the Properties of Resveratrol-Loaded NLC Using 22 Full Factorial Design. Antioxidants 2019, 8, 272. [Google Scholar] [CrossRef]
- Aguiar, G.P.S.; Arcari, B.D.; Chaves, L.M.P.C.; Magro, C.D.; Boschetto, D.L.; Piato, A.L.; Lanza, M.; Oliveira, J.V. Micronization of trans-resveratrol by supercritical fluid: Dissolution, solubility and in vitro antioxidant activity. Ind. Crops Prod. 2018, 112, 1–5. [Google Scholar] [CrossRef]
- Calvo-Castro, L.A.; Schiborr, C.; David, F.; Ehrt, H.; Voggel, J.; Sus, N.; Behnam, D.; Bosy-Westphal, A.; Frank, J. The oral bioavailability of trans-resveratrol from a grapevine-shoot extract in healthy humans is significantly increased by micellar solubilization. Mol. Nutr. Food Res. 2018, 62, 1701057. [Google Scholar] [CrossRef]
- Spogli, R.; Bastianini, M.; Ragonese, F.; Iannitti, R.G.; Monarca, L.; Bastioli, F.; Nakashidze, I.; Brecchia, G.; Menchetti, L.; Codini, M.; et al. Solid dispersion of resveratrol supported on magnesium dihydroxide (resv@mdh) microparticles improves oral bioavailability. Nutrients 2018, 10, 1925. [Google Scholar] [CrossRef]
- He, H.; Zhang, Q.; Wang, J.-R.; Mei, X. Structure, physicochemical properties and pharmacokinetics of resveratrol and piperine cocrystals. CrystEngComm 2017, 19, 6154–6163. [Google Scholar] [CrossRef]
- Singh, D.; Bedi, N.; Tiwary, A.K. Enhancing solubility of poorly aqueous soluble drugs: Critical appraisal of techniques. J. Pharm. Investig. 2018, 48, 509–526. [Google Scholar] [CrossRef]
- Ma, X.; Williams, R.O., III. Polymeric nanomedicines for poorly soluble drugs in oral delivery systems: An update. J. Pharm. Investig. 2018, 48, 61–75. [Google Scholar]
- Abuzar, S.M.; Hyun, S.-M.; Kim, J.-H.; Park, H.J.; Kim, M.-S.; Park, J.-S.; Hwang, S.-J. Enhancing the solubility and bioavailability of poorly water-soluble drugs using supercritical antisolvent (SAS) process. Int. J. Pharm. 2018, 538, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; McClements, D.J. Fabrication of biopolymer nanoparticles by antisolvent precipitation and electrostatic deposition: Zein-alginate core/shell nanoparticles. Food Hydrocoll. 2015, 44, 101–108. [Google Scholar] [CrossRef]
- Ha, E.-S.; Kim, J.-S.; Lee, S.-K.; Sim, W.-Y.; Jeong, J.-S.; Kim, M.S. Solubility and modeling of telmisartan in binary solvent mixtures of dichloromethane and (methanol, ethanol, n-propanol, or n-butanol) and its application to the preparation of nanoparticles using the supercritical antisolvent technique. J. Mol. Liq. 2019, 295, 111710. [Google Scholar] [CrossRef]
- Ha, E.-S.; Kim, J.-S.; Lee, S.-K.; Sim, W.-Y.; Jeong, J.-S.; Kim, M.S. Equilibrium solubility and solute-solvent interactions of carvedilol (Form I) in twelve mono solvents and its application for supercritical antisolvent precipitation. J. Mol. Liq. 2019, 294, 111622. [Google Scholar] [CrossRef]
- Tsinman, K.; Tsinman, O.; Lingamaneni, R.; Zhu, S.; Riebesehl, B.; Grandeury, A.; Juhnke, M.; Van Eerdenbrugh, B. Ranking Itraconazole formulations based on the flux through artificial lipophilic membrane. Pharm. Res. 2018, 35, 161. [Google Scholar] [CrossRef]
- Das, S.; Ng, K.Y. Quantification of trans-resveratrol in rat plasma by a simple and sensitive high performance liquid chromatography method and its application in pre-clinical study. J. Liq. Chromatogr. Relat. Technol. 2011, 34, 1399–1414. [Google Scholar] [CrossRef]
- Alonso, C.; Martı, M.; Barba, C.; Carrer, V.; Rubio, L.; Coderch, L. Skin permeation and antioxidant efficacy of topically applied resveratrol. Arch. Dermatol. Res. 2017, 309, 423–431. [Google Scholar] [CrossRef]
- Zupančič, Š.; Lavrič, Z.; Kristl, J. Stability and solubility of trans-resveratrol are strongly influenced by pH and temperature. Euro. J. Pharm. Biopharm. 2015, 93, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Wegiel, L.A.; Mosquera-Giraldo, L.I.; Mauer, L.J.; Edgar, K.J.; Taylor, L.S. Phase Behavior of Resveratrol Solid Dispersions Upon Addition to Aqueous media. Pharm. Res. 2015, 32, 3324–3337. [Google Scholar] [CrossRef] [PubMed]
- Ha, E.-S.; Choo, G.-H.; Baek, I.-H.; Kim, M.-S. Formulation, Characterization, and in Vivo Evaluation of Celecoxib-PVP Solid Dispersion Nanoparticles Using Supercritical Antisolvent Process. Molecules 2014, 19, 20325–20339. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-S.; Ha, E.-S.; Kim, J.-S.; Baek, I.; Yoo, J.-W.; Jung, Y.; Moon, H.R. Development of megestrol acetate solid dispersion nanoparticles for enhanced oral delivery by using a supercritical antisolvent process. Drug Des. Dev. Ther. 2015, 9, 4269–4277. [Google Scholar] [CrossRef]
- Ha, E.S.; Choo, G.H.; Baek, I.H.; Kim, J.S.; Cho, W.; Jung, Y.S.; Jin, S.E.; Hwang, S.J.; Kim, M.S. Dissolution and bioavailability of lercanidipine-hydroxypropylmethyl cellulose nanoparticles with surfactant. Int. J. Biol. Macromol. 2015, 72, 188–222. [Google Scholar] [CrossRef]
- Ansari, K.A.; Vavia, P.R.; Trotta, F.; Cavalli, R. Cyclodextrin-Based Nanosponges for Delivery of Resveratrol: In Vitro Characterisation, Stability, Cytotoxicity and Permeation Study. AAPS PharmSciTech 2011, 12, 279–286. [Google Scholar] [CrossRef]
- Aguiar, G.P.S.; Boschetto, D.L.; Chaves, L.M.P.C.; Arcari, B.D.; Piato, A.L.; Oliveira, J.V.; Lanza, M. Trans-resveratrol micronization by SEDS technique. Ind. Crops Prod. 2016, 89, 350–355. [Google Scholar] [CrossRef]
- Liu, X.; Feng, X.; Williams, R.O., III; Zhang, F. Characterization of amorphous solid dispersions. J. Pharm. Investig. 2018, 48, 19–41. [Google Scholar] [CrossRef]
- Chang, C.W.; Wong, C.Y.; Wu, Y.T.; Hsu, M.C. Development of a solid dispersion system for improving the oral bioavailability of resveratrol in rats. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 239–249. [Google Scholar] [CrossRef]
- Singh, S.K.; Makadia, V.; Sharma, S.; Rashid, M.; Shahi, S.; Mishra, P.R.; Wahajuddin, M.; Gayen, J.R. Preparation and in-vitro/in-vivo characterization of trans-resveratrol nanocrystals for oral administration. Drug Deliv. Transl. Res. 2017, 7, 395–407. [Google Scholar] [CrossRef]
- Maier-Salamon, A.; Hagenauer, B.; Wirth, M.; Gabor, F.; Szekeres, T.; Jager, W. Increased transport of resveratrol across monolayers of the human intestinal Caco-2 cells is mediated by inhibition and saturation of metabolites. Pharm. Res. 2006, 23, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.N.; Verma, P.R.P. Enhancement of in vitro dissolution and pharmacodynamic potential of olanzapine using solid SNEDDS. J. Pharm. Investig. 2018, 48, 269–278. [Google Scholar] [CrossRef]
- Goo, B.; Sim, W.-Y.; Ha, E.-S.; Kim, M.-S.; Cho, C.-W.; Hwang, S.-J. Preparation of Spray-dried Emulsion of Sirolimus for Enhanced Oral Bioavailability. Bull. Korean Chem. Soc. 2018, 39, 1215–1218. [Google Scholar] [CrossRef]
- Baek, I.H.; Kim, M.S. Improved supersaturation and oral absorption of dutasteride by amorphous solid dispersions. Chem. Pharm. Bull. 2012, 60, 1468–1473. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wu, T.; Qi, Y.; Zhang, Z. Recent advances in the application of vitamin E TPGS for drug delivery. Theranostics 2018, 8, 464–485. [Google Scholar] [CrossRef]
- Herpin, M.J.; Smyth, H.D.C. Super-heated aqueous particle engineering (SHAPE): A novel method for the micronization of poorly water soluble drugs. J. Pharm. Investig. 2018, 48, 135–142. [Google Scholar] [CrossRef]
- Som, I.; Bhatia, K.; Yasir, M. Status of surfactants as penetration enhancers in transdermal drug delivery. J. Pharm. Bioallied Sci. 2012, 4, 2–9. [Google Scholar]
- Rastogi, V.; Yadav, P.; Verma, N.; Verma, A. Preparation and characterization of transdermal mediated microemulsion delivery of T4 bacteriophages against E.coli bacteria: A novel anti-microbial approach. J. Pharm. Investig. 2018, 48, 394–407. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kim, M.-S.; Baek, I.-H. Enhanced Bioavailability of Tadalafil after Intranasal Administration in Beagle Dogs. Pharmaceutics 2018, 10, 187. [Google Scholar] [CrossRef]
- Teichmann, A.; Otberg, N.; Jacobi, U.; Sterry, W.; Lademann, J. Follicular penetration: Development of a method to block the follicles selectively against the penetration of topically applied substances. Skin Pharmacol. Physiol. 2006, 19, 216–223. [Google Scholar] [CrossRef]

| Formulation | Encapsulation Efficiency (%) | Mean Particle Size (nm) | Specific Surface Area (m2/g) |
|---|---|---|---|
| Micronized trans-resveratrol | - | 2631.4 ± 203.1 | 2.98 ± 0.3 |
| Drug/HPMC/poloxamer 407 | 98.9 ± 0.6 | 258.5 ± 19.5 | 40.2 ± 1.6 |
| Drug/HPMC/TPGS | 99.1 ± 0.8 | 293.4 ± 16.9 | 36.4 ± 1.1 |
| Drug/HPMC/gelucire 44/14 | 97.2 ± 1.2 | 291.7 ± 15.2 | 33.1 ± 1.3 |
| Drug/HPMC (1:5) | 98.2 ± 0.9 | 188.2 ± 6.9 | 60.4 ± 2.3 |
| Drug/HPMC (1:4) | 99.9 ± 0.3 | 181.5 ± 8.5 | 63.2 ± 1.9 |
| Formulation | In Vitro Flux (μg/cm2/min) | In Vivo Pharmacokinetic Data | ||
|---|---|---|---|---|
| AUC0→12 h (ng·h/mL) | Cmax (ng/mL) | Tmax (h) | ||
| Micronized trans-resveratrol | 0.266 ± 0.006 | 163.4 ± 33.1 | 43.4 ± 6.3 | 1.0 ± 0.5 |
| Drug/HPMC/poloxamer 407 | 0.792 ± 0.013 a,b,c,d | 493.6 ± 45.4 a,b,c,d | 420.3 ± 63.4 a,b,c | 0.5 ± 0.1 |
| Drug/HPMC/TPGS | 0.761 ± 0.016 a,b,c | 434.3 ± 30.1 a,b,c | 368.7 ± 72.1 a,b | 0.5 ± 0.2 |
| Drug/HPMC/gelucire 44/14 | 0.618 ± 0.012 a,b | 365.4 ± 45.0 a,b | 268.7 ± 78.9 a | 0.7 ± 0.4 |
| Drug/HPMC (1:5) | 0.419 ± 0.012 a | 256.6 ± 53.8 a | 133.5 ± 36.7 | 0.9 ± 0.3 |
| Drug/HPMC (1:4) | 0.409 ± 0.004 a | 245.9 ± 76.9 a | 132.2 ± 27.3 | 0.7 ± 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, E.-S.; Sim, W.-Y.; Lee, S.-K.; Jeong, J.-S.; Kim, J.-S.; Baek, I.-h.; Choi, D.H.; Park, H.; Hwang, S.-J.; Kim, M.-S. Preparation and Evaluation of Resveratrol-Loaded Composite Nanoparticles Using a Supercritical Fluid Technology for Enhanced Oral and Skin Delivery. Antioxidants 2019, 8, 554. https://doi.org/10.3390/antiox8110554
Ha E-S, Sim W-Y, Lee S-K, Jeong J-S, Kim J-S, Baek I-h, Choi DH, Park H, Hwang S-J, Kim M-S. Preparation and Evaluation of Resveratrol-Loaded Composite Nanoparticles Using a Supercritical Fluid Technology for Enhanced Oral and Skin Delivery. Antioxidants. 2019; 8(11):554. https://doi.org/10.3390/antiox8110554
Chicago/Turabian StyleHa, Eun-Sol, Woo-Yong Sim, Seon-Kwang Lee, Ji-Su Jeong, Jeong-Soo Kim, In-hwan Baek, Du Hyung Choi, Heejun Park, Sung-Joo Hwang, and Min-Soo Kim. 2019. "Preparation and Evaluation of Resveratrol-Loaded Composite Nanoparticles Using a Supercritical Fluid Technology for Enhanced Oral and Skin Delivery" Antioxidants 8, no. 11: 554. https://doi.org/10.3390/antiox8110554
APA StyleHa, E.-S., Sim, W.-Y., Lee, S.-K., Jeong, J.-S., Kim, J.-S., Baek, I.-h., Choi, D. H., Park, H., Hwang, S.-J., & Kim, M.-S. (2019). Preparation and Evaluation of Resveratrol-Loaded Composite Nanoparticles Using a Supercritical Fluid Technology for Enhanced Oral and Skin Delivery. Antioxidants, 8(11), 554. https://doi.org/10.3390/antiox8110554









