Enhancement of Minor Ginsenosides Contents and Antioxidant Capacity of American and Canadian Ginsengs (Panax quinquefolius) by Puffing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemicals
2.3. Puffing Process
2.4. Color Measurement
2.5. Extraction Yield
- W1 = Weight of empty aluminum dish (g)
- W2 = Weight of aluminum dish and solid (g)
- A = Weight of dried ginseng (g)
- E = Total volume of extract (mL)
- E′ = Used volume of extract (mL).
2.6. Crude Saponin Content
- W1 = Weight of the dried sample and flask (mg)
- W2 = Weight of the flask (mg)
- W3 = Weight of total dried ginseng (g)
- A = Weight of total concentration (g)
- B = Weight of used concentration (g).
2.7. Ginsenoside Profile
2.8. Antioxidant Activity
2.8.1. DPPH Radical Scavenging Activity
- Areference = absorbance of the blank
- Asample = absorbance of the sample
- Areference = mixture of 0.1 mL of 80% MeOH and 2.9 mL of DPPH radical solution.
2.8.2. ABTS Radical Scavenging Activity
2.9. Total Phenolic Content
2.10. Total Flavonoid Content
2.11. Acidic Polysaccharides
2.12. Maillard Reaction Products
2.13. Statistical Analysis
3. Results and Discussion
3.1. Morphology
3.2. Extraction Yield and Crude Saponin Content
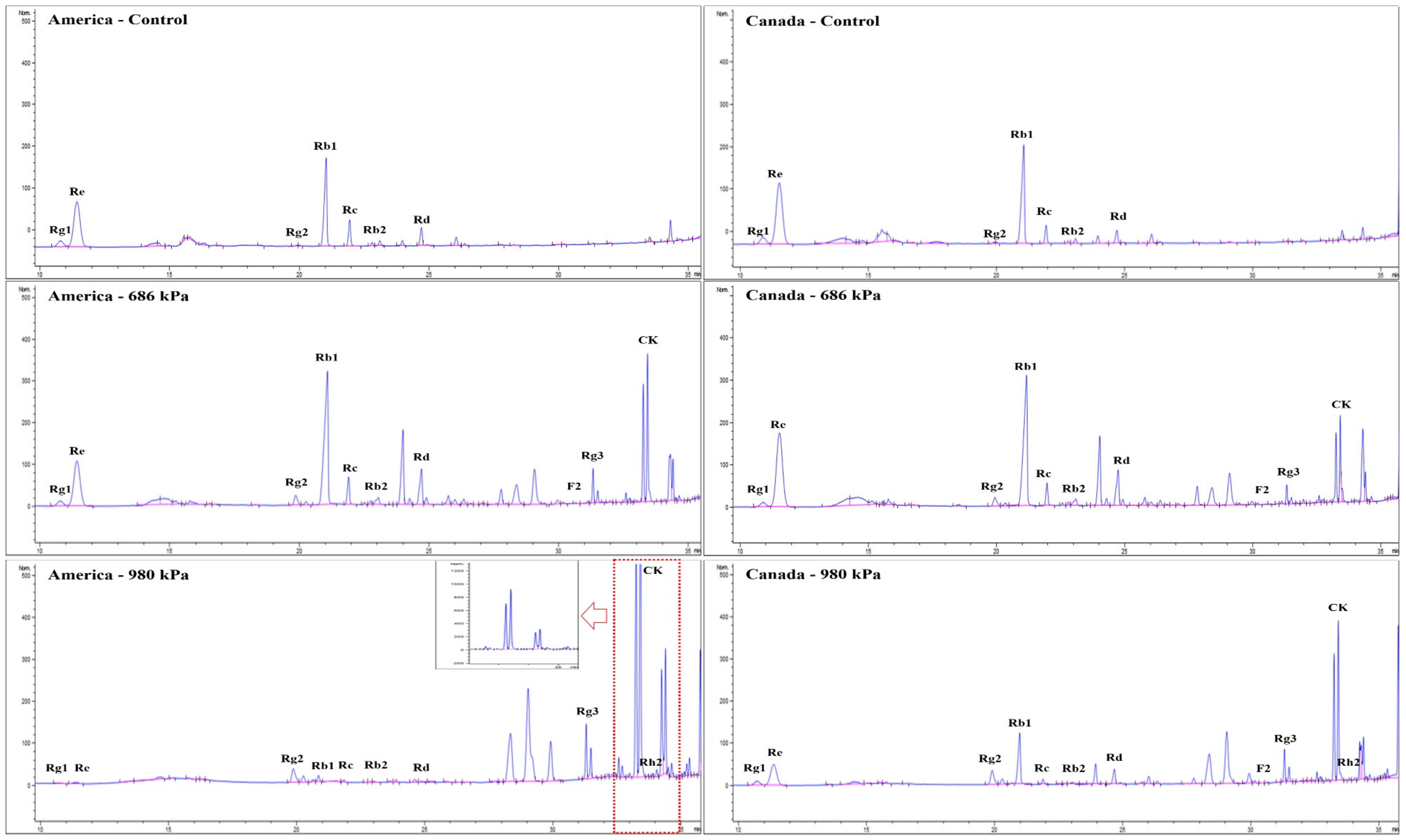
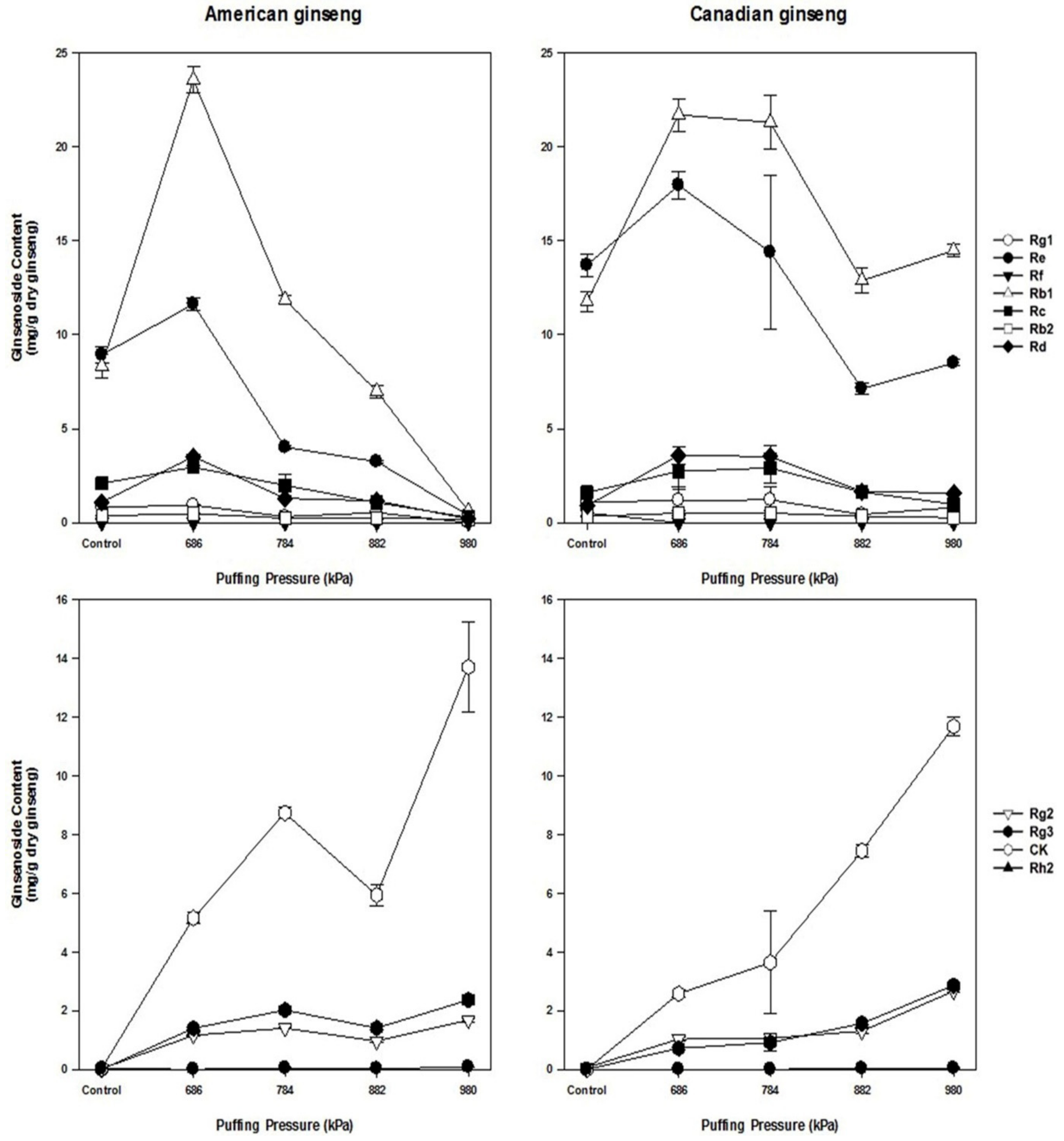
3.3. Changes in Ginsenosides
3.4. Antioxidant Activity, TPC, and TFC
3.5. Acidic Polysaccharides and MRPs
3.6. Relationships between Antioxidant Activities, TPC, TFC, MRPs, and Acidic Polysaccharides
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ang-Lee, M.K.; Moss, J.; Yuan, C.-S. Herbal medicines and perioperative care. Jama 2001, 286, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Attele, A.S.; Wu, J.A.; Yuan, C.-S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Wu, J.; Zhong, J.-J. Production of ginseng and its bioactive components in plant cell culture: Current technological and applied aspects. J. Biotechnol. 1999, 68, 89–99. [Google Scholar] [CrossRef]
- Lee, S.-J.; Ko, W.-G.; Kim, J.-H.; Sung, J.-H.; Lee, S.-J.; Moon, C.-K.; Lee, B.-H. Induction of apoptosis by a novel intestinal metabolite of ginseng saponin via cytochrome c-mediated activation of caspase-3 protease. Biochem. Pharmacol. 2000, 60, 677–685. [Google Scholar] [CrossRef]
- Park, J. Recent studies on the chemical constituents of Korean ginseng (Panax ginseng CA Meyer). Korean J. Ginseng Sci. 1996, 20, 389–415. [Google Scholar]
- Park, C.-K.; Jeon, B.-S.; Yang, J.-W. The chemical components of Korean ginseng. Food Ind. Nutr. 2003, 8, 10–23. [Google Scholar]
- Yang, X.-D.; Yang, Y.-Y.; Ouyang, D.-S.; Yang, G.-P. A review of biotransformation and pharmacology of ginsenoside compound K. Fitoterapia 2015, 100, 208–220. [Google Scholar] [CrossRef]
- Huang, B.-M.; Chen, T.-B.; Xiao, S.-Y.; Zha, Q.-L.; Luo, P.; Wang, Y.-P.; Cui, X.-M.; Liu, L.; Zhou, H. A new approach for authentication of four ginseng herbs and their related products based on the simultaneous quantification of 19 ginseng saponins by UHPLC-TOF/MS coupled with OPLS-DA. RSC Adv. 2017, 7, 46839–46851. [Google Scholar] [CrossRef]
- Nah, S.-Y. Ginseng ginsenoside pharmacology in the nervous system: Involvement in the regulation of ion channels and receptors. Front. Physiol. 2014, 5, 98. [Google Scholar] [CrossRef]
- Chen, C.-F.; Chiou, W.-F.; Zhang, J.-T. Comparison of the pharmacological effects of Panax ginseng and Panax quinquefolium. Acta Pharmacol. Sin. 2008, 29, 1103. [Google Scholar] [CrossRef]
- Hall, T.; Lu, Z.; Yat, P. Ginseng evaluation program part I: Standardized phase report. J. Am. Bot. Counc. Herb. 2001, 52, 26–45. [Google Scholar]
- Yuan, C.-S.; Wang, C.-Z.; Wicks, S.M.; Qi, L.-W. Chemical and pharmacological studies of saponins with a focus on American ginseng. J. Ginseng Res. 2010, 34, 160. [Google Scholar] [CrossRef] [PubMed]
- Assinewe, V.A.; Baum, B.R.; Gagnon, D.; Arnason, J.T. Phytochemistry of wild populations of Panax quinquefolius L. (North American ginseng). J. Agric. Food Chem. 2003, 51, 4549–4553. [Google Scholar] [CrossRef] [PubMed]
- Helms, S. Cancer prevention and therapeutics: Panax ginseng. Altern. Med. Rev. 2004, 9, 259–274. [Google Scholar] [PubMed]
- Kim, K.T.; Yoo, K.M.; Lee, J.W.; Eom, S.H.; Hwang, I.K.; Lee, C.Y. Protective effect of steamed American ginseng (Panax quinquefolius L.) on V79-4 cells induced by oxidative stress. J. Ethnopharmacol. 2007, 111, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.-H.; Kang, P.-S.; Kim, J.-W.; Kwon, J.; Oh, S.-H. Water extracts of cultured mountain ginseng stimulate immune cells and inhibit cancer cell proliferation. Food Sci. Biotechnol. 2006, 15, 369–373. [Google Scholar]
- Wang, C.-Z.; Li, B.; Wen, X.-D.; Zhang, Z.; Yu, C.; Calway, T.D.; He, T.-C.; Du, W.; Yuan, C.-S. Paraptosis and NF-κB activation are associated with protopanaxadiol-induced cancer chemoprevention. BMC Complement. Altern. Med. 2013, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- An, Y.-E.; Ahn, S.-C.; Yang, D.-C.; Park, S.-J.; Kim, B.-Y.; Baik, M.-Y. Chemical conversion of ginsenosides in puffed red ginseng. LWT Food Sci. Technol. 2011, 44, 370–374. [Google Scholar] [CrossRef]
- Hoseney, R.C. Principles of Cereal Science and Technology; American Association of Cereal Chemists (AACC): Saint Paul, MN, USA, 1994. [Google Scholar]
- Mishra, G.; Joshi, D.; Panda, B.K. Popping and puffing of cereal grains: A review. J. Grain Process. Storage 2014, 1, 34–46. [Google Scholar]
- Kim, S.-T.; Jang, J.-H.; Kwon, J.-H.; Moon, K.-D. Changes in the chemical components of red and white ginseng after puffing. Korean J. Food Preserv. 2009, 16, 355–361. [Google Scholar]
- Yoon, S.-R.; Lee, M.-H.; Park, J.-H.; Lee, I.-S.; Kwon, J.-H.; Lee, G.-D. Changes in physicochemical compounds with heating treatment of ginseng. J. Korean Soc. Food Sci. Nutr. 2005, 34, 1572–1578. [Google Scholar]
- Kim, S.-B.; Do, J.-R.; Lee, Y.-W.; Gu, Y.-S.; Kim, C.-N.; Park, Y.-H. Nitrite-scavenging effects of roasted-barley extracts according to processing conditions. Korean J. Food Sci. Technol. 1990, 22, 748–752. [Google Scholar]
- Ryu, G.; Lee, J. Development of extrusion process on red ginseng from raw ginseng and its products. Final Rep. Ventur. Res. Minist. Health Welf. Seoul 2003, 9–10. [Google Scholar]
- Kim, J.-H.; Ahn, S.-C.; Choi, S.-W.; Hur, N.-Y.; Kim, B.-Y.; Baik, M.-Y. Changes in effective components of ginseng by puffing. Appl. Biol. Chem. 2008, 51, 188–193. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Van den Berg, R.; Haenen, G.R.; van den Berg, H.; Bast, A. Applicability of an improved Trolox equivalent antioxidant capacity (TEAC) assay for evaluation of antioxidant capacity measurements of mixtures. Food Chem. 1999, 66, 511–517. [Google Scholar] [CrossRef]
- Hossain, A.; Moon, H.K.; Kim, J.-K. Antioxidant properties of Korean major persimmon (Diospyros kaki) leaves. Food Sci. Biotechnol. 2018, 27, 177–184. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Nam, T.G.; Kim, D.-O.; Eom, S.H. Effects of light sources on major flavonoids and antioxidant activity in common buckwheat sprouts. Food Sci. Biotechnol. 2018, 27, 169–176. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Do, J.; Lee, H.; Lee, S.; Jang, J.; Lee, S.; Sung, H. Colorimetric determination of acidic polysaccharide from Panax ginseng, its extraction condition and stability. Korean J. Ginseng Sci. 1993, 17, 139–144. [Google Scholar]
- Kim, K.T.; Yoo, K.M. Effect of hot water boiling and autoclaving on physicochemical properties of American ginseng (Panax quinquefolium L.). J. Ginseng Res. 2009, 33, 40–47. [Google Scholar]
- Hong, H.-D.; Kim, Y.-C.; Rho, J.-H.; Kim, K.-T.; Lee, Y.-C. Changes on physicochemical properties of Panax ginseng CA Meyer during repeated steaming process. J. Ginseng Res. 2007, 31, 222–229. [Google Scholar]
- Hong, H.-D.; Kim, Y.-C.; Kim, S.-S.; Sim, G.-S.; Han, C.-K. Effect of puffing on quality characteristics of red ginseng tail root. J. Ginseng Res. 2007, 31, 147–153. [Google Scholar]
- Choi, Y.; Lee, S.; Chun, J.; Lee, H.; Lee, J. Influence of heat treatment on the antioxidant activities and polyphenolic compounds of Shiitake (Lentinus edodes) mushroom. Food Chem. 2006, 99, 381–387. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef]
- Yang, S.-J.; Woo, K.-S.; Yoo, J.-S.; Kang, T.-S.; Noh, Y.-H.; Lee, J.-S.; Jeong, H.-S. Change of Korean ginseng components with high temperature and pressure treatment. Korean J. Food Sci. Technol. 2006, 38, 521–525. [Google Scholar]
- Ko, S.; Choi, K.; Kim, H. Comparison of proximate composition, mineral nutrient, amino acid and free sugar contents of several Panax species. Korean J. Ginseng Sci. 1996, 20, 36–41. [Google Scholar]

| Sample (kPa) | L | a | b | Extraction Yield (%) | Crude Saponin Content (mg/g Dried Ginseng) |
|---|---|---|---|---|---|
| A-Control | 76.86 ± 1.55 a,* | 8.34 ± 0.15 d | 16.99 ± 0.53 d | 35.03 ± 8.49 d | 72.44 ± 5.19 e |
| A-686 | 54.66 ± 10.14 b | 12.91 ± 1.15 ab | 27.64 ± 2.21 ab | 46.86 ± 12.07 ab | 141.20 ± 26.16 a |
| A-784 | 47.56 ± 14.73 bc | 12.20 ± 1.00 bc | 25.97 ± 3.90 abc | 44.84 ± 5.74 b | 126.56 ± 12.26 abc |
| A-882 | 44.87 ± 10.28 bc | 11.73 ± 2.29 bc | 24.55 ± 5.16 bc | 40.44 ± 4.16 c | 107.12 ± 17.39 d |
| A-980 | 39.44 ± 9.58 c | 10.78 ± 2.51 c | 21.52 ± 6.24 c | 46.19 ± 3.20 ab | 122.10 ± 14.78 bcd |
| C-Control | 78.67 ± 1.92 a | 8.41 ± 0.28 d | 16.77 ± 1.04 d | 38.57 ± 3.92 cd | 104.33 ± 22.08 d |
| C-686 | 54.26 ± 4.08 b | 14.27 ± 0.50 a | 30.56 ± 1.02 a | 46.95 ± 4.43 ab | 129.44 ± 17.68 ab |
| C-784 | 53.53 ± 7.51 b | 13.28 ± 1.05 ab | 28.69 ± 2.36 ab | 47.32 ± 3.54 ab | 115.86 ± 13.42 bcd |
| C-882 | 48.04 ± 3.86 b | 13.41 ± 1.30 ab | 27.85 ± 2.55 ab | 49.44 ± 5.26 a | 109.61 ± 9.29 cd |
| C-980 | 39.82 ± 2.42 c | 11.94 ± 1.24 bc | 23.86 ± 2.68 bc | 49.20 ± 5.04 a | 126.33 ± 10.66 abc |
| Sample (kPa) | DPPH (mg VCE/ g Dried Ginseng) | ABTS (mg VCE/ g Dried Ginseng) | TPC (mg GAE/ g Dried Ginseng) | TFC (mg CE/ g Dried Ginseng) | AP (mg GA/ g Dried Ginseng) | MRPs |
|---|---|---|---|---|---|---|
| A-Control | 0.29 ± 0.11 b* | 0.97 ± 0.15 d | 1.11 ± 0.06 f | 0.44 ± 0.05 e | 1.78 ± 0.27 c | 0.186 ± 0.014 c |
| A-686 | 1.88 ± 0.26 a | 10.06 ± 4.88 c | 9.24 ± 3.60 e | 2.39 ± 1.17 de | 2.60 ± 0.61 bc | 0.295 ± 0.085 bc |
| A-784 | 1.97 ± 0.16 a | 14.58 ± 5.23 bc | 14.46 ± 5.05 cde | 3.84 ± 1.66 bcd | 2.87 ± 0.37 b | 0.422 ± 0.129 b |
| A-882 | 1.89 ± 0.28 a | 15.37 ± 3.99 bc | 16.05 ± 4.80 cd | 4.82 ± 2.15 abc | 2.81 ± 0.10 bc | 0.442 ± 0.033 b |
| A-980 | 1.67 ± 0.33 a | 39.71 ± 1.02 a | 24.23 ± 5.89 a | 5.73 ± 2.11 ab | 5.09 ± 1.21 a | 0.694 ± 0.209 a |
| C-Control | 0.41 ± 0.20 b | 1.24 ± 0.19 d | 1.65 ± 0.22 f | 0.56 ± 0.06 e | 1.79 ± 0.42 c | 0.215 ± 0.043 c |
| C-686 | 2.04 ± 0.15 a | 13.04 ± 3.70 bc | 12.51 ± 4.69 de | 3.54 ± 1.04 cd | 3.11 ± 0.39 b | 0.317 ±0.075 bc |
| C-784 | 1.94 ± 0.18 a | 17.58 ± 7.58 b | 12.87 ± 2.39 cde | 4.20 ± 1.41 abcd | 3.02 ± 0.15 b | 0.400 ± 0.140 b |
| C-882 | 1.74 ± 0.22 a | 17.28 ± 0.34 b | 18.48 ± 3.02 bc | 4.91 ± 1.73 abc | 3.25 ± 0.45 b | 0.616 ± 0.146 a |
| C-980 | 1.85 ± 0.23 a | 39.64 ± 3.13 a | 21.33 ± 6.21 ab | 6.19 ± 1.34 a | 4.69 ± 0.69 a | 0.645 ± 0.204 a |
| American Ginseng | MRPs | DPPH | ABTS | TPC | TFC | AP |
|---|---|---|---|---|---|---|
| MRPs | ||||||
| DPPH | 0.548 | |||||
| ABTS | 0.982 ** | 0.471 | ||||
| TPC | 0.981 ** | 0.693 | 0.942 * | |||
| TFC | 0.938 * | 0.737 | 0.868 | 0.981 ** | ||
| AP | 0.965 ** | 0.444 | 0.997 *** | 0.918 * | 0.831 | |
| Canadian Ginseng | ||||||
| MRPs | ||||||
| DPPH | 0.548 | |||||
| ABTS | 0.835 | 0.613 | ||||
| TPC | 0.937 * | 0.793 | 0.88 ** | |||
| TFC | 0.910 * | 0.820 | 0.911 * | 0.989 ** | ||
| AP | 0.842 | 0.705 | 0.982 ** | 0.926 * | 0.941 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.-S.; Jeon, S.-J.; Youn, S.J.; Lee, H.; Park, Y.-J.; Kim, D.-O.; Kim, B.-Y.; Kim, W.; Baik, M.-Y. Enhancement of Minor Ginsenosides Contents and Antioxidant Capacity of American and Canadian Ginsengs (Panax quinquefolius) by Puffing. Antioxidants 2019, 8, 527. https://doi.org/10.3390/antiox8110527
Kim M-S, Jeon S-J, Youn SJ, Lee H, Park Y-J, Kim D-O, Kim B-Y, Kim W, Baik M-Y. Enhancement of Minor Ginsenosides Contents and Antioxidant Capacity of American and Canadian Ginsengs (Panax quinquefolius) by Puffing. Antioxidants. 2019; 8(11):527. https://doi.org/10.3390/antiox8110527
Chicago/Turabian StyleKim, Min-Soo, Sung-Joon Jeon, So Jung Youn, Hyungjae Lee, Young-Joon Park, Dae-Ok Kim, Byung-Yong Kim, Wooki Kim, and Moo-Yeol Baik. 2019. "Enhancement of Minor Ginsenosides Contents and Antioxidant Capacity of American and Canadian Ginsengs (Panax quinquefolius) by Puffing" Antioxidants 8, no. 11: 527. https://doi.org/10.3390/antiox8110527
APA StyleKim, M.-S., Jeon, S.-J., Youn, S. J., Lee, H., Park, Y.-J., Kim, D.-O., Kim, B.-Y., Kim, W., & Baik, M.-Y. (2019). Enhancement of Minor Ginsenosides Contents and Antioxidant Capacity of American and Canadian Ginsengs (Panax quinquefolius) by Puffing. Antioxidants, 8(11), 527. https://doi.org/10.3390/antiox8110527





