1. Introduction
Environmental factors like polluted water, air, soil, UV radiations, intake of pesticides by food consumption, all might result in production of reactive oxygen species (ROS) [
1,
2,
3,
4]. Imbalance between ROS production and their scavenging by antioxidant apparatus cause oxidative stress which is the base of damages to cell structure, proteins, lipids, and DNA [
5,
6,
7]. A number of transcription factors are activated via ROS, which results in expressing proteins regulating inflammation, survival of tumor cells, cellular transformation, proliferation and invasion, metastasis and angiogenesis [
8]. The disturbance of the homeostasis of cellular machinery caused by ROS leads to various pathological disorders, among which cancer is doubtless the most serious and worrying. Cancer has become a public health concern in developed as well as developing countries on a massive level. In India, it is a major cause of adult deaths, with more than 70% of lethal cancers occurring in people in the productive ages of 30–69 years [
9]. The World Cancer Report 2014, launched by The International Agency for Research on Cancer (IARC), tells that the worldwide burden of cancer has reached to approximately 14 million new cases annually, and this will further rise to around 22 million per year in next two decades. Further, cancer deaths have been expected to rise from 8.2 million (approximately) to 13 million every year [
10].
Among other classes of health promoting compounds, polyphenols from fruits, vegetables and medicinal plants represent a peerless class of antioxidants which can counteract ROS-triggered damages. The cancer preventive activities of various phytochemicals lie in their ability to affect cellular defenses like detoxification, scavenging ROS, as well as induction of antitumor and anti-inflammatory responses. Thus, antioxidants from medicinal plants present themselves as the safer option to chemically formulated anticancer drugs [
11,
12,
13]. A huge diversity of polyphenol structures are produced by plants, and along with contributing to the color, flavor, odor, bitterness and astringency of food, these possess several biological activities like anti-allergic, anti-microbial, anti-viral, antioxidant, anti-inflammatory, anti-mutagenic, and anti-proliferative [
14].
Prevention is better than cure, and cancer, though being a major cause of deaths worldwide, is a disease which can largely be prevented by modulating dietary supplements [
15]. Among the reported cancer cases, 5–10% are linked to genetics and the other 90–95% cancers originate from various environmental and lifestyle reasons and are preventable [
16]. The practice of using medicinal plant parts has been demonstrated since ancient times to treat a wide array of illnesses [
17] and it is the need of the hour to return to plant based diet and to re-evaluate the effectiveness of herbal treatments against several dreaded diseases. Also, synthetic drugs and the pharmaceutical products, being used in human and veterinary medicine have certain side effects and are an emerging class of environmental contaminants [
18]. Even though polyphenols are prevalent in the medicinal plant parts, not all people worldwide can ensure an adequate intake due to different reasons. The extraction, identification and separation of the antioxidant polyphenolic compounds from various plant parts is undertaken to help manufacturing dietary antioxidants with effective influence to be used as supplements to the normal diet [
19].
The genus
Bauhinia belongs to family Fabaceae and it includes an approximate of 350 shrubs, lianas and small tree species which are recognized easily with their bilobed leaves as well as palmate venation. Due to its bilobed leaves, the genus was named
Bauhinia by Linnaeus to honor Swiss Botanists, Johann and Gaspard, who were identical twins [
20]. The genus is broadly distributed in countries in Asia, South America, and Africa. Plant parts are often used in traditional medicine for infections, inflammations and diabetes.
Bauhinia species like
B. rufescens,
B. reticulata, are used traditionally for treatment of ulcers, roundworm infections, conjunctivitis, dysentery, blood-poisoning, lung and skin diseases [
21,
22].
Bauhinia variegata Linn., a medium sized, deciduous tree, is also known as Kanchanara in Sanskrit; Kovidara in Hindi and Raktakanchan in Marathi. Almost all the parts find uses inside the traditional medicine against various ailments like leprosy, piles, asthma, ulcer, liver complaints and snake bite. It also finds its uses in the treatment of skin diseases, wound healing, obesity, stomatitis, dyspepsia, flatulence and is used as tonic, astringent and laxative as well [
23]. An alcoholic extract of
B. variegata stem bark has shown significant hepatoprotective activity against liver damage caused by carbon tetrachloride [
24]. The ethanolic extract of
B. variegata stem has shown antitumor activity against Dalton’s Ascytic lymphoma and Ehrlich ascites carcinoma in swiss albino mice [
25,
26]. Isolation of quercetin, rutin, apigenin and apigenin 7-O-glucoside has been reported from the different plant parts of
B. variegata [
27]. Literature survey suggests that although
B. variegata plant parts have many medicinal uses, however, few studies have been undertaken to validate the therapeutic claim in a scientific manner. To introduce
B. variegata to the list of potential sources of chemo-preventive phytochemicals, the present work was undertaken for the isolation and characterization of bioactive compounds from bark extracts and to estimate their antioxidant and cytotoxic potential.
4. Discussion
Natural plant products have traditionally been an imperative source of lead molecules in drug discovery. Despite this fact, the interest of the pharmaceutical industries in natural products has declined in the past. There has been observed a switching away from natural products to combinatorial chemistry for drug development [
36]. The reason behind this change seems to be the fact that chemical agents were far easier to synthesize, to get patented and furthermore these ensured greater profitability for industries. However, synthetic drug side effects became a major health concern leading to withdrawal of certain drugs from the market.
Earlier, the isolation and characterization of bioactive compounds from different plant parts was a long, gradual process, contingent upon the convolution of the study. Nowadays, however, pace of bioassay-guided fractionation has escalated by use of instrumentation like higher magnetic field-strength NMR, high-performance liquid chromatography (HPLC), liquid chromatography mass spectroscopy (LC-MS), etc. [
37]. Drug discovery from natural sources is gaining pace in correspondence with rise in identification of new lead compounds.
The chemical composition of the plants belonging to genus
Bauhinia shows a high proportion of flavonoids, terpenes, steroids and alkaloids. Isolation of Lupeol, 5, 7-dimethoxyflavonone-4–O–α– L–rhamnopyranosyl-β-D-glucopyranoside, β-sitosterol, kaempferol-3- glucoside from the stem of the have been reported [
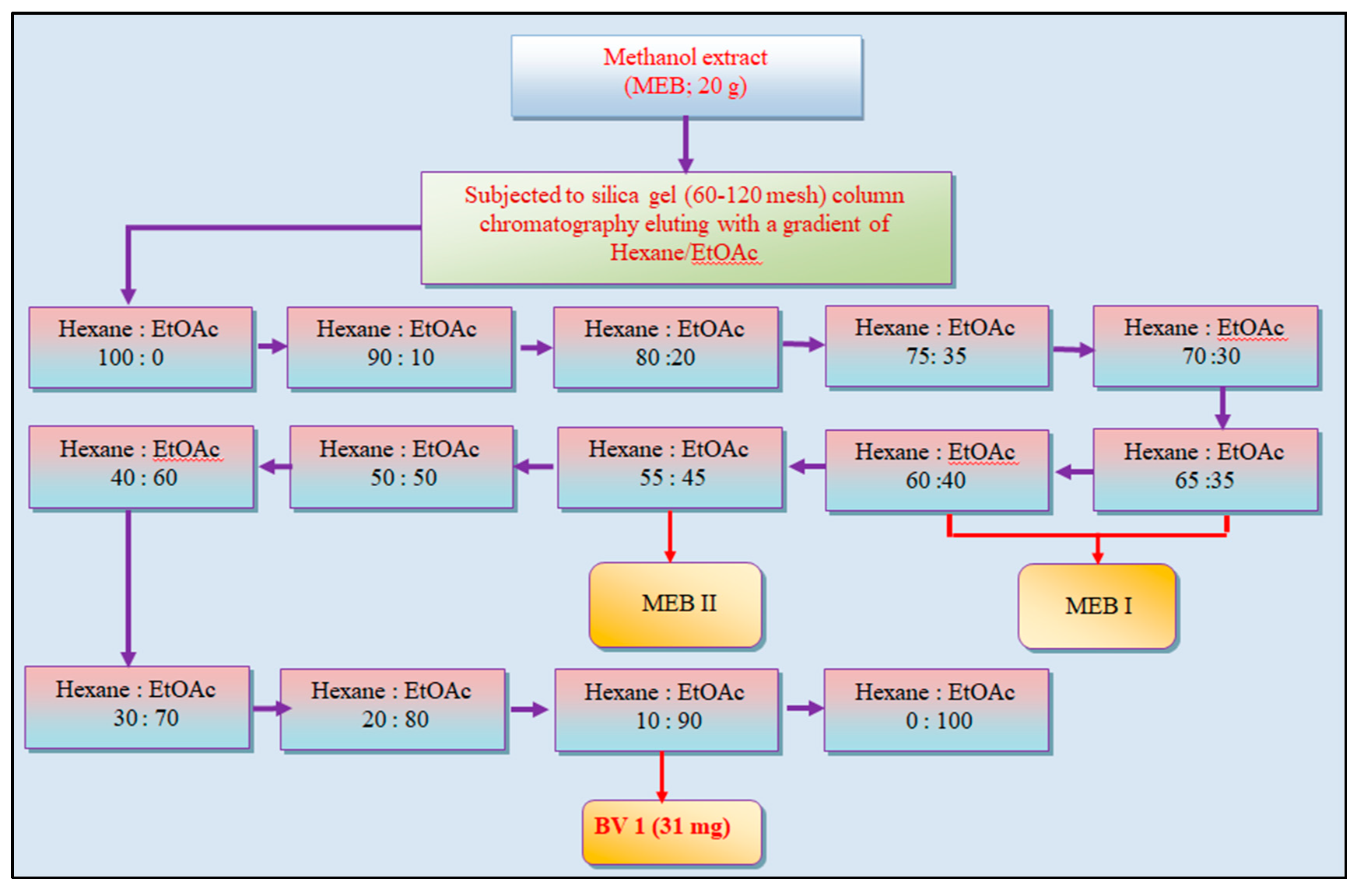
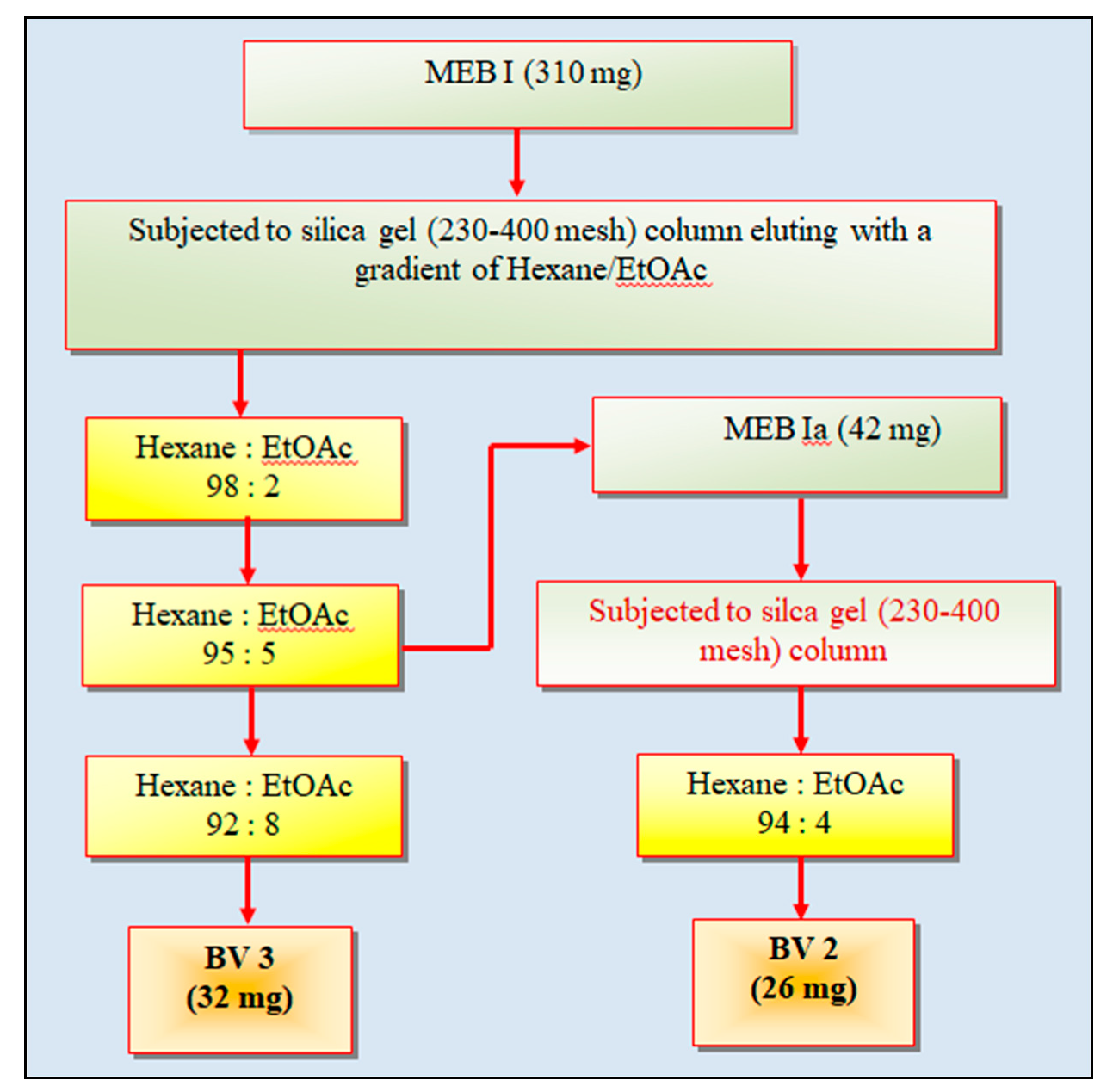
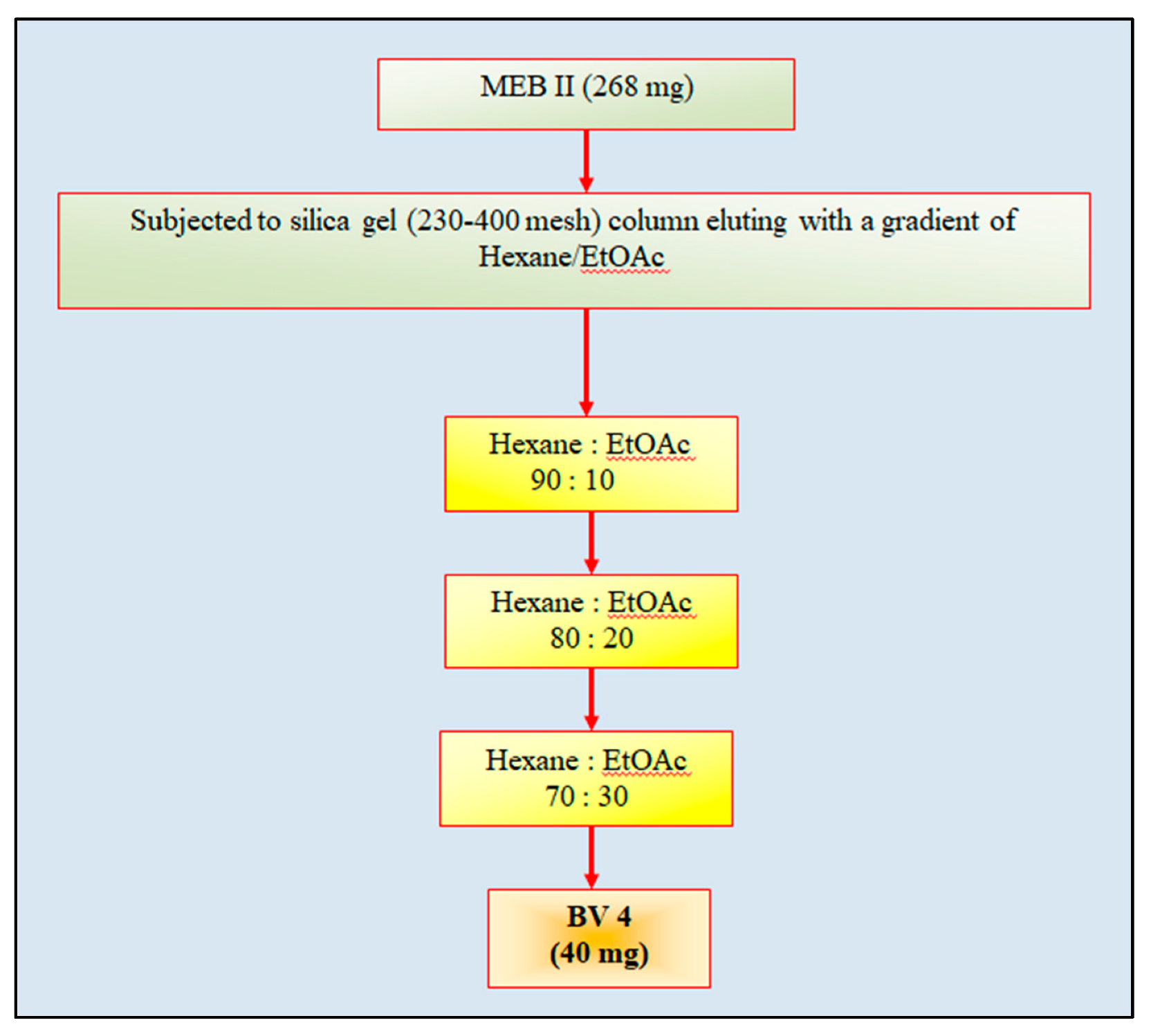
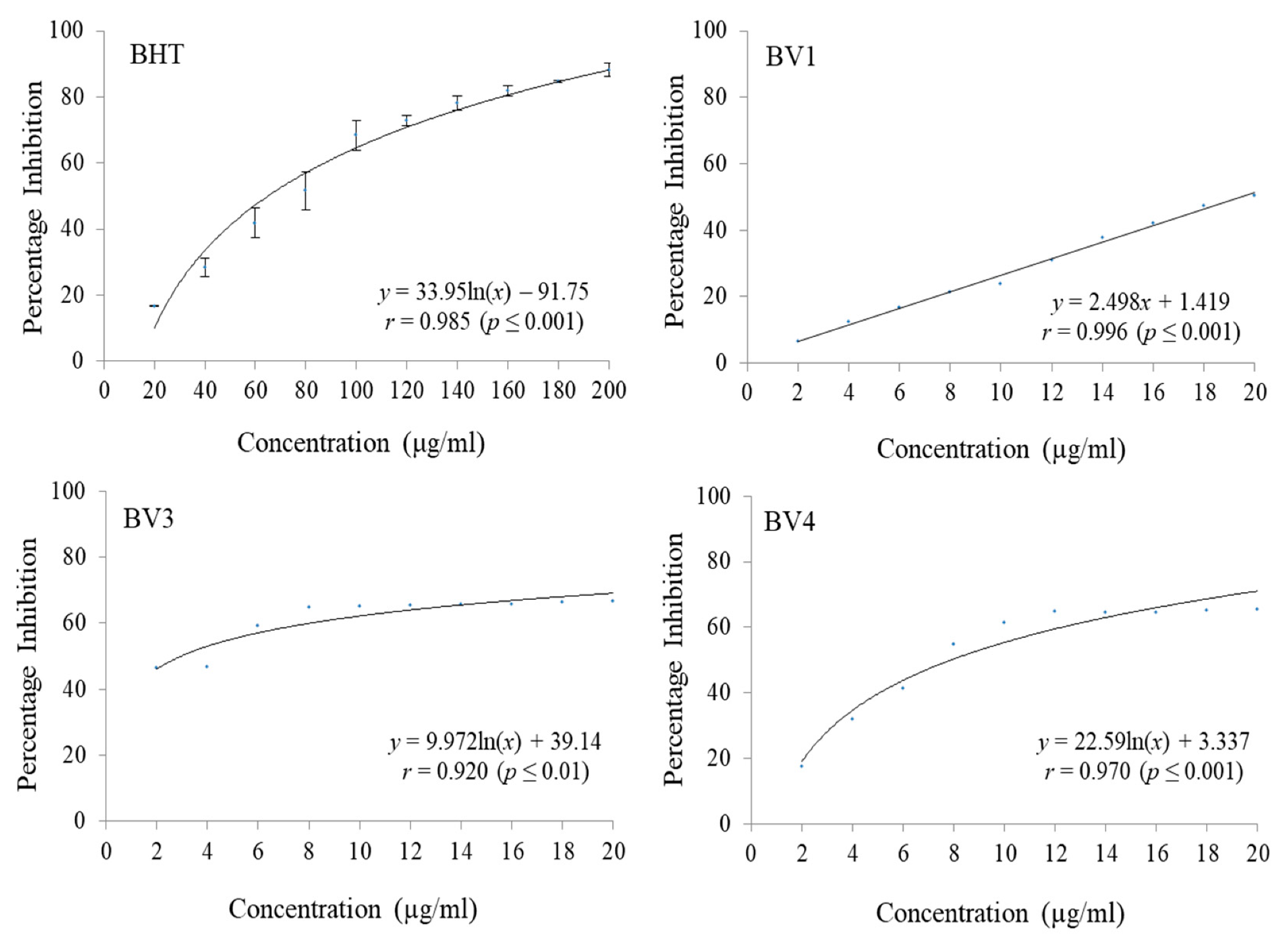
38]. In the present study, pure fractions isolated from MEB through silica gel column chromatography, i.e., BV1, BV2, BV3 and BV4, were characterized as kaempferol, stigmasterol, protocatechuic acid-methyl ester (PCA-ME) and protocatechuic acid (PCA) respectively, and were subjected to analysis of their antioxidant potential and cytotoxic activity against different cell lines.
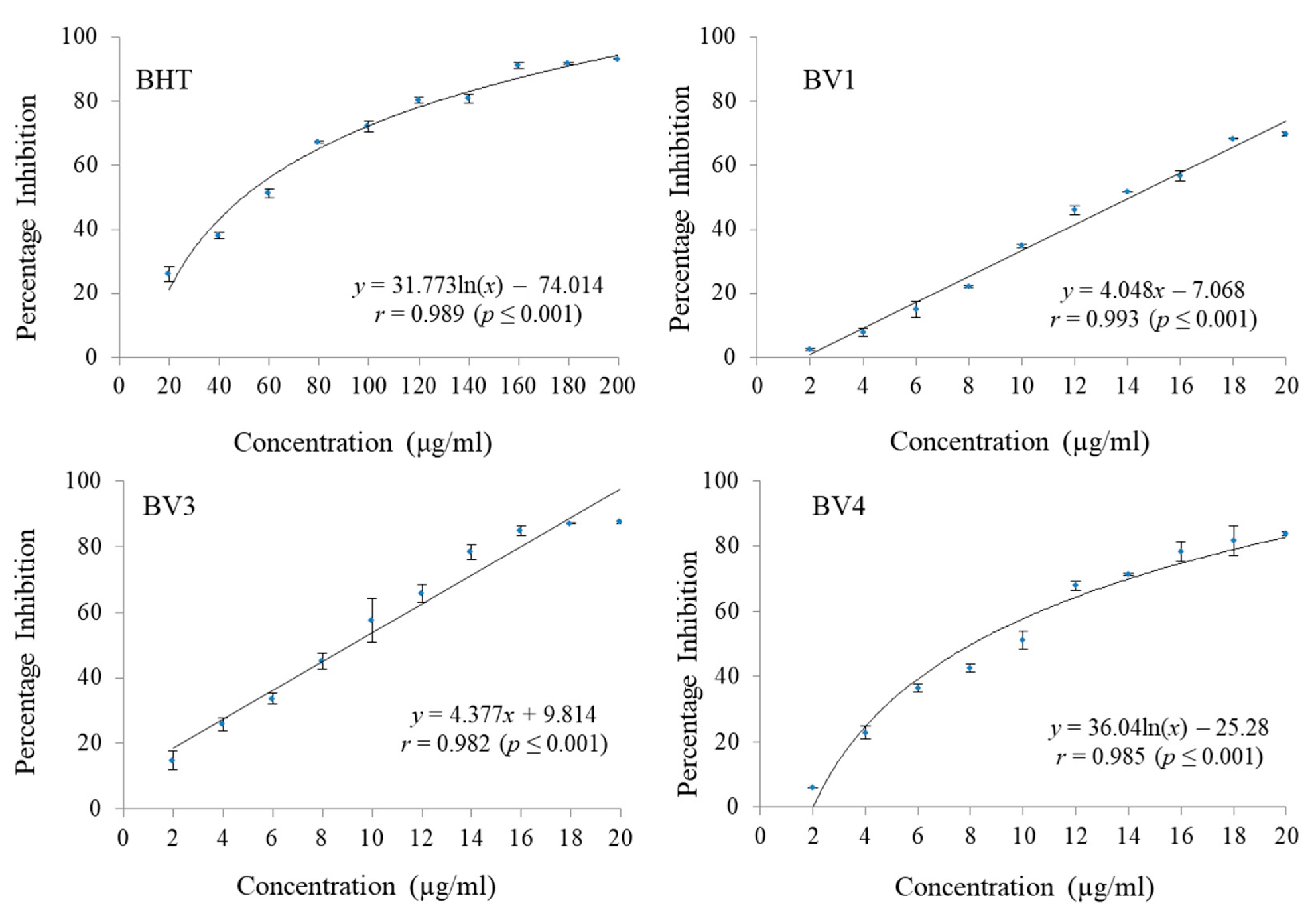
Kaempferol has been reported to have very significant antioxidant activity [
39]. It is a flavonoid frequently found in plant-based foods as well as in plant parts utilized in folk medicine. It has a double bond at C2-C3 and an oxo group at C4, and hydroxyl groups at C3, C5 and C4’, which provide the structural bases for antioxidant activity [
40,
41,
42]. In the present study, it significantly inhibited ABTS and DPPH free radicals, which is in conjugation with a number of studies reporting that kaempferol, and its glycosides, and various plants parts having kaempferol possess antioxidative potential [
43]. Stigmasterol, known as the Wulzen anti-stiffness factor, belongs to the group of plant sterols having similarity to animal cholesterol. The manufacture of vitamin D3 and semi-synthetic progesterone, which is a valuable human hormone, involves stigmasterol as a precursor [
44]. In the present investigation, when analyzed for its activity against ABTS and DPPH free radicals, it did not show any considerable antioxidative activity. Similar results were obtained by Shanthakumar et al. [
45] where stigmasterol was found to possess a very high IC
50 of 220µg/mL against DPPH radicals.
Protocatechuic acid has wide natural distribution and shares structural similarity with caffeic acid, vanillic acid, gallic acid, syringic acid which are well acknowledged antioxidants [
46]. In the present investigation, both PCA and PCA-ME showed considerable antioxidant and cytotoxic activities. The PCA-ME had a higher potential to scavenge free radicals as compared to PCA. In case of ABTS assay, PCA-ME possessed an IC
50 of 8.76 ± 1.23 µg/mL as compared to PCA having an IC
50 of 9.39 ± 0.45 µg/mL. Similar was the case with DPPH assay where the IC
50 values were 4.46 ± 0.21 µg/mL and 7.28 ± 1.64 µg/mL for PCA-ME and PCA respectively.
Literature reports also suggest lower radical scavenging activity of PCA compared to PCA-ME in alcoholic solvents. In an experiment carried out by Saito and Kwabata [
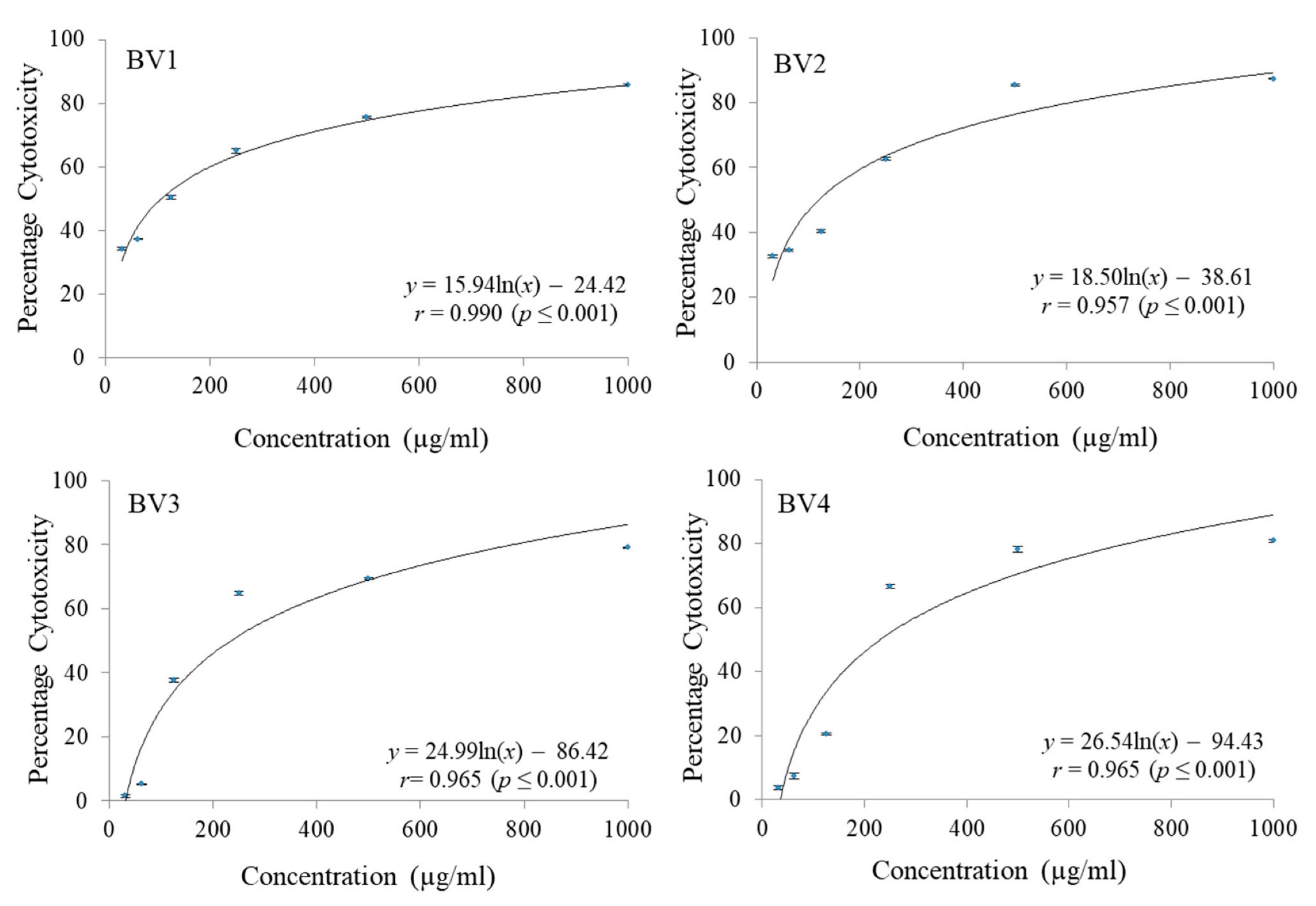
47], it was found that PCAME scavenged 5 equivalents of DPPH radical in contrast to 2 equivalents of radical scavenging by PCA in methanol in 30 min. Further, kaempferol, stigmasterol, PCA-ME and PCA were subjected to analysis of their cytotoxic prospective against C6, MCF-7 and HCT-15 cell lines. A chemically induced rat brain tumor was used to clone C6 rat glioma cell line, and is categorized as an undifferentiated astrocytic cell type [
48]. It is extensively used in in vitro models to study glial cell properties. The MCF-7 has been taken from a patient’s pleural effusion suffering with metastatic breast cancer [
49]. In the present investigation, kaempferol exhibited dose dependent increase in the cytotoxic capacity against different cell lines under study. Kaempferol showed maximum cytotoxicity against C-6 cell line, followed by MCF-7 and HCT-15 cell line. Earlier reports by Diantini et al. [
50] stated that kaempferol-3
-O-rhamnoside hinders the proliferation of MCF-7 cells dose-dependent mode and also promotes apoptosis. Among A549 and MCF-7 cells, induction of MAPK (mitogen-activated protein kinase) has been considered to be an essential factor in kaempferol initiated apoptosis. Also, the MAPK activation mediated by kaempferol may avoid impairment to DNA leading to cell transformation. Kaempferol was further found to increase expression of haeme oxygenase (HO)-1 gene, resulting in a surge in the antioxidant capacity of cells [
51]. In HeLa cells, kaempferol blocked the glucose cellular intake [
52]. A cytostatic activity of 24.8–64.7 μM by kaempferol was observed in PC3, HeLa and K562 human cancer cells [
53]. Luo et al. [
54] reported kaempferol to initiate activation of p53 thus inducing apoptosis in ovarian cancer cells. It has also inhibited quinine reductase 2 (IC
50 33.6 μM) for NF-κB activity [
55]. Similarly, Kim and Choi [
56] reported kaempferol to inhibit growth of MCF-7 cells via regulating cell cycle and apoptosis-related genes. Combining kaempferol with chemotherapeutic drugs has produced better therapeutic effects than the drugs alone, and this combination also reduces the toxicity of chemotherapeutic drugs [
57]. Imran et al. [
58] reported that kaempferol-rich food decreases the risk of development of skin, liver, and colon cancers via apoptosis, cell cycle arrest, downregulating epithelial-mesenchymal transition (EMT) related markers as well as the phosphoinositide 3-kinase/protein kinase B signaling pathways. They suggested that cancer fighting properties of kaempferol are extraordinary as it specifically targets cancerous cells without damaging healthy cells.
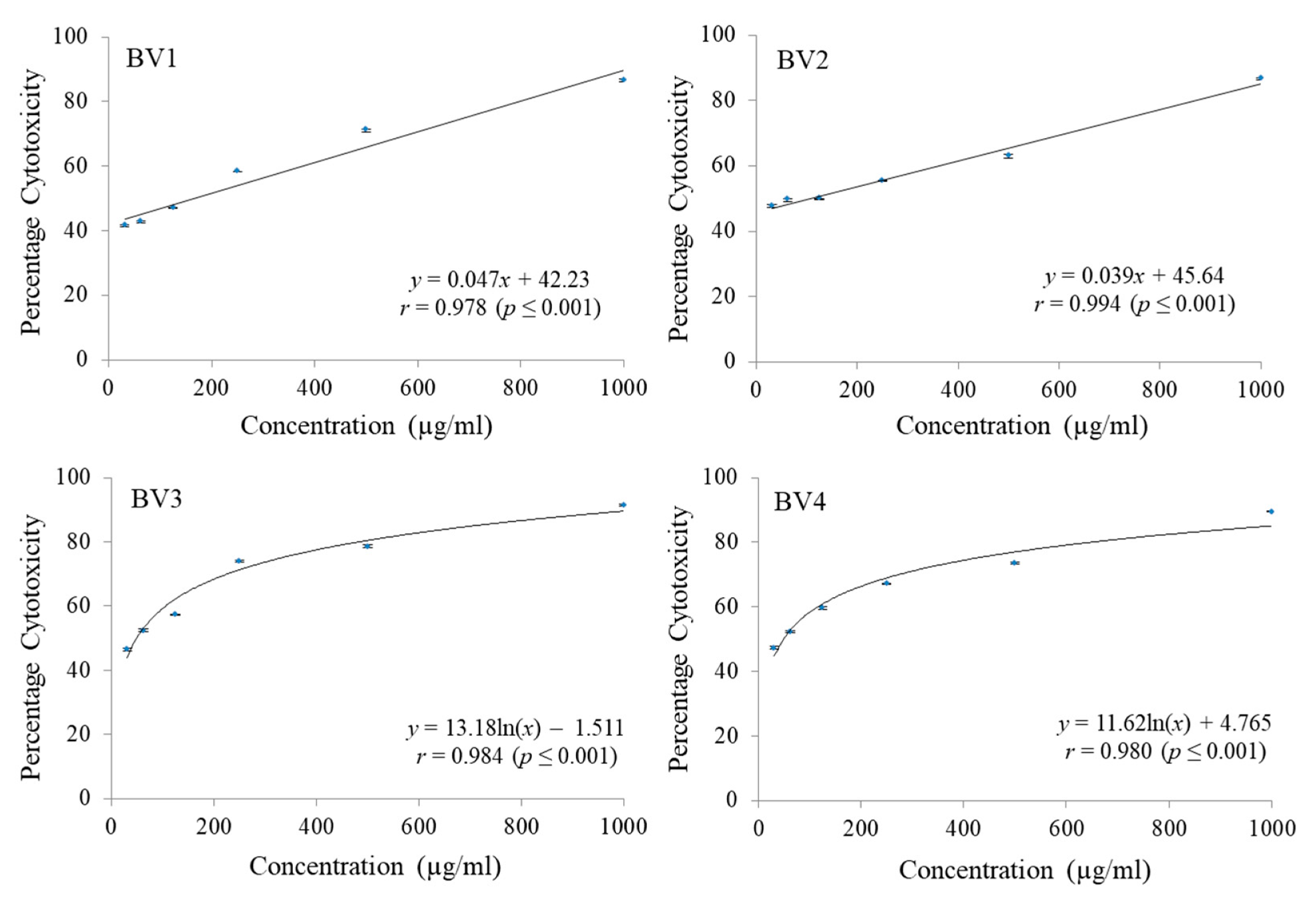
In case of stigmasterol, when evaluated for cytotoxic potential, it was observed that it showed moderate activity against the cell lines under investigation showing GI
50 values of 173.97 ± 0.81 µg/mL, 132.01 ± 0.55 µg/mL, 151.20 ± 0.74 µg/mL for C-6, MCF-7 and HCT-15 cell lines respectively. Malikova et al. [
59] established that stigmasterol showed very weak to undetectable activity against the T-lymphoblastic leukaemia CEM, MCF-7 (breast carcinoma), A-549 (lung carcinoma), K562 (chronic myeloid leukaemia), HeLa (cervical carcinoma), G361 (malignant melanoma) and HOS (osteosarcoma) cell lines and had a moderate activity against multiple myeloma RPMI 8226 cell line. However, in a study conducted by Ali et al. [
60], a reduction in size of tumor and number of papillomas was observed with an oral administration of stigmasterol to Swiss albino mice in which had skin tumors induced through topical application of 7,12-dimethylbenz[a]anthracene (DMBA).
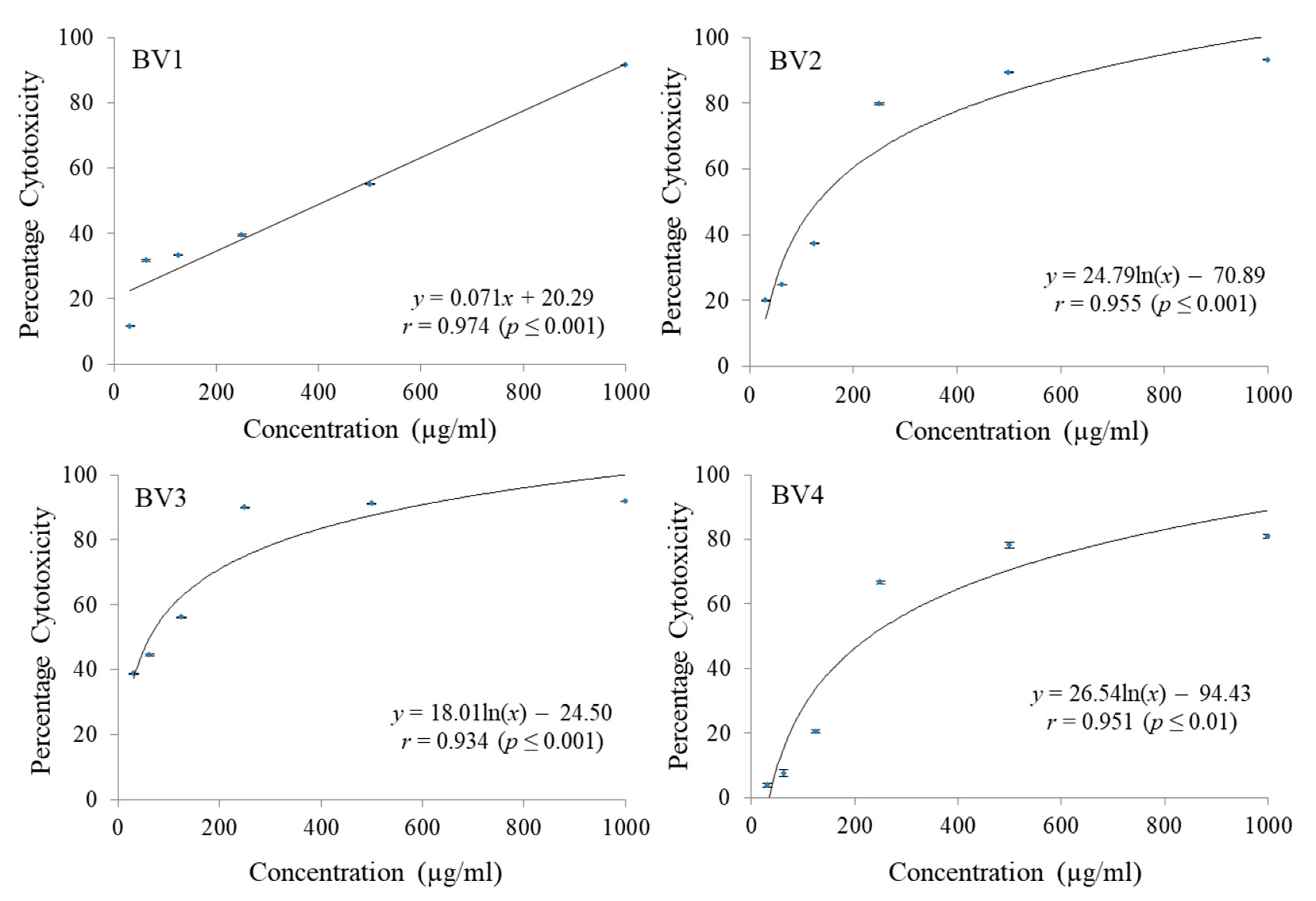
PCA have shown considerable cytotoxic potential against MCF-7 cell line (GI
50 49.14 ± 0.23 µg/mL), while PCA-ME have shown significant cytotoxic activity against MCF-7 (GI
50 54.18 ± 0.16 µg/mL) as well as against HCT-15 cell line (GI
50 92.17 ± 1.17 µg/mL). The mechanism of cytotoxic action of PCA has been proposed to be associated with antioxidant activity. It can influence metabolism phases 1 and 2 of various carcinogens thus blocking specific binding sites of the carcinogens with the DNA. This leads to the prevention of adduct formation that could have resulted into mutations and neoplastic transformations [
61]. Various researchers have reported PCA to have significant antioxidant, antibacterial, anti-inflammatory, analgesic and antiseptic properties [
62,
63]. In a study conducted by Bullo et al. [
64], PCA and chlorogenic acid (CA) were loaded into an anticancer nanocarrier graphene oxide-polyethylene glycol (GOPEG) and tested in phosphate buffer saline to assess the release properties and also tested against HEPG2 (liver cancer), HT-29 cells (human colon cancer) and normal 3T3 cells (fibroblast cells). It was found that PCA and CA were released in a sustained manner, having strong activity against cancerous cells and biocompatibility with normal cells.
The phytochemical screening of
B. variegata has shown the occurrence of flavonoids, terpenoids, reducing sugars, steroids, saponins, tannins, and cardiac glycosides [
65]. In earlier studies,
B. variegata has depicted chemopreventive activity against liver tumors induced by
N-nitrosodiethylamine and human cancer cell lines. It was found that orally administrating ethanol extract of
B. variegata suppressed liver tumor. A decrease in serum glutamate pyruvate transaminase, lipid peroxidase, total bilirubin, glutathione peroxidase and glutathione
S-transferase levels was observed. Further, an increase in superoxide dismutase, catalase and total proteins was also observed [
66]. A phytochemical investigation of the polar fractions of methanol extract of
B. variegata bark revealed that the fractions were rich in phenolic and flavonoid content and a statistically significant correlation was observed between phenolic/flavonoid content and the antioxidant activity of the fractions [
67]. The essential oil of
B. variegata flowers has been reported to contain a substantial amount of nerolidol, α-bisabolol and β-bisabolene which have significant medicinal properties [
68].