Untargeted Metabolomic Profiling, Multivariate Analysis and Biological Evaluation of the True Mangrove (Rhizophora mucronata Lam.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Plant Material
2.2. Extraction
2.3. Profile of Bioactive Compounds
2.4. Determination of Antioxidant and Enzyme Inhibitory Effects
2.5. Antimicrobial Properties
2.6. Statistical Analyses
3. Results and Discussion
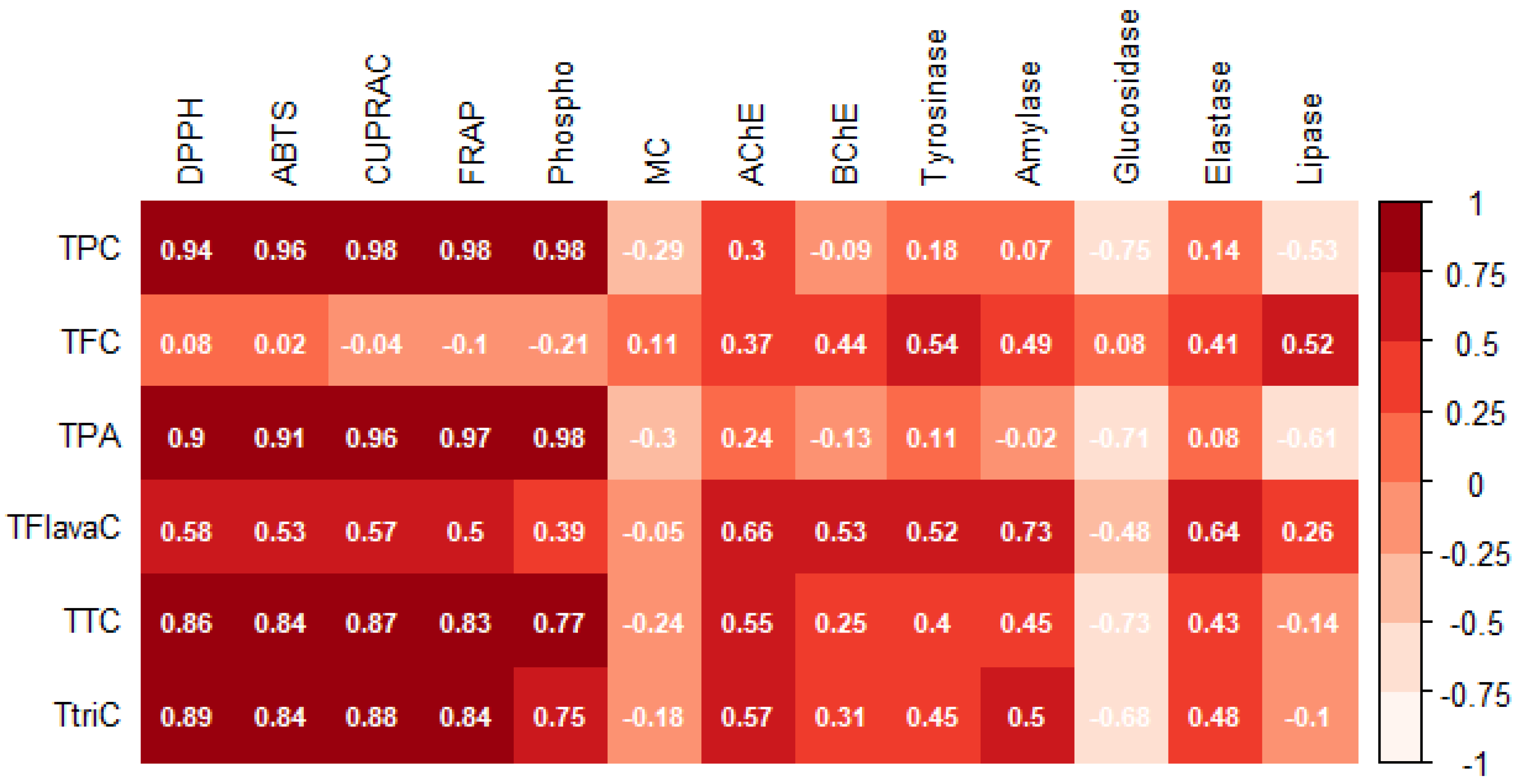
3.1. Spectrophotometric Analysis of Phenolic Compounds
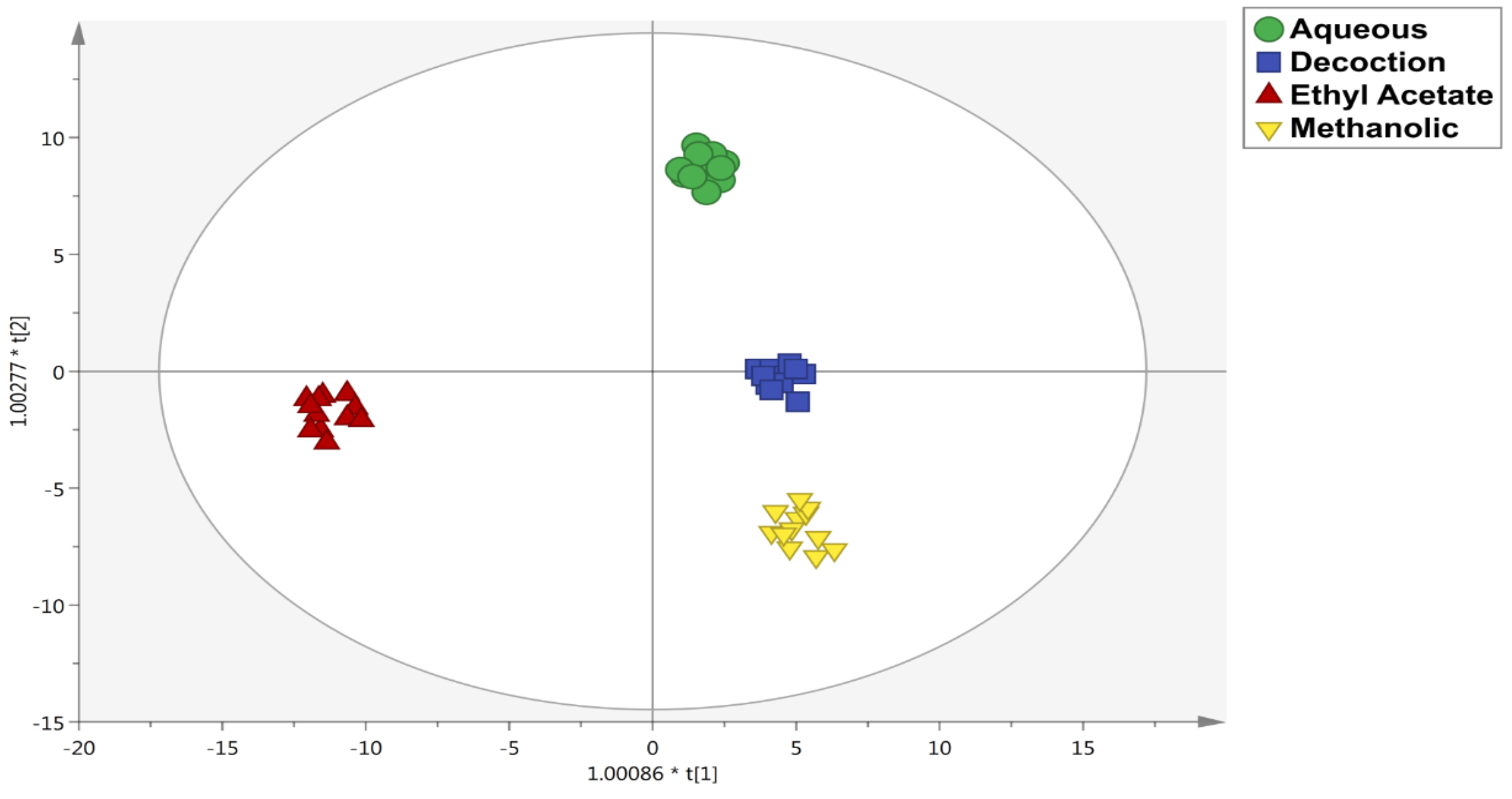
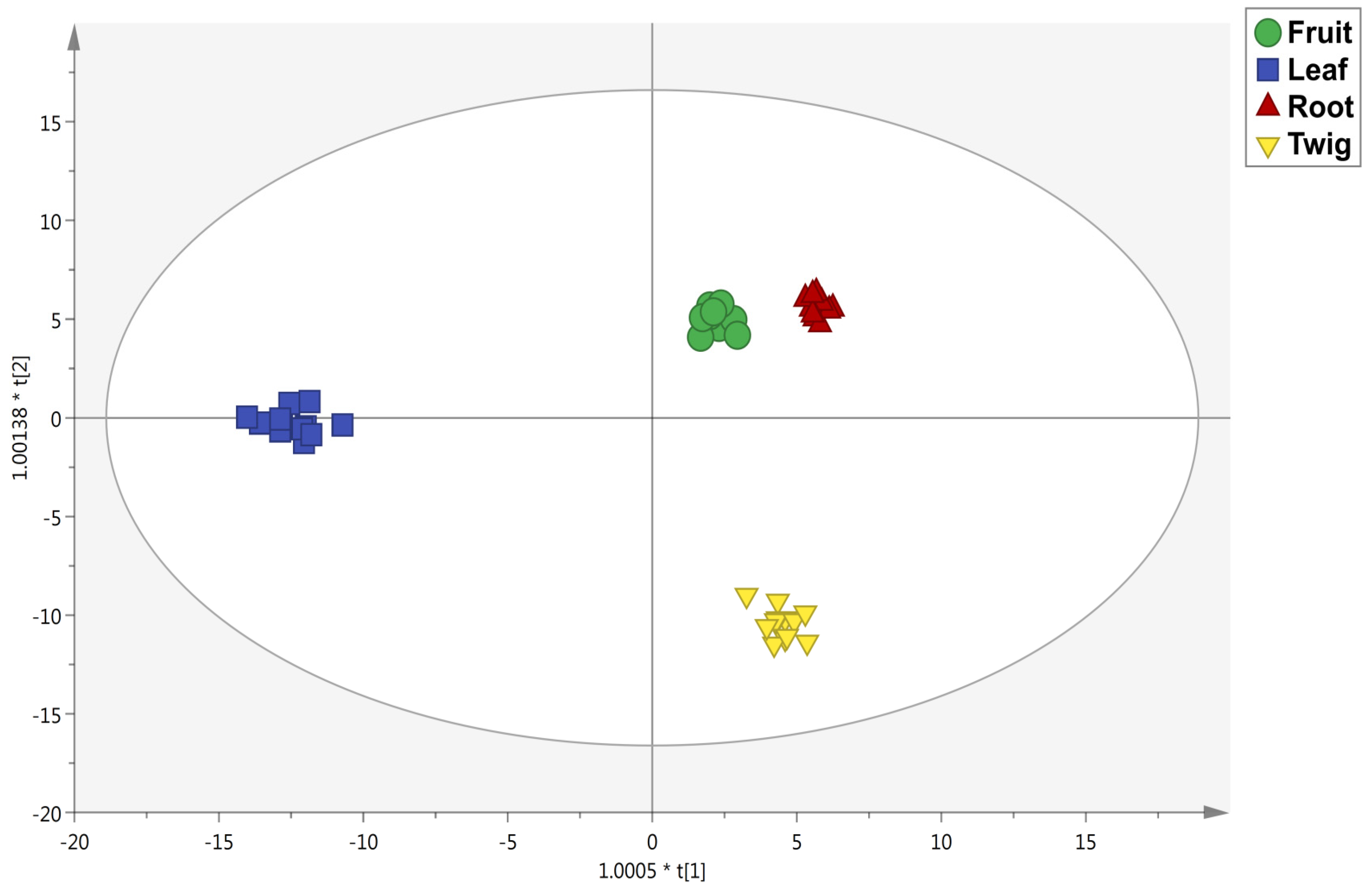
3.2. Untargeted Metabolomic Profiling and Multivariate Statistics
3.3. In Vitro Antioxidant Assays
3.4. Enzymatic Inhibitory Assays
3.5. Antimicrobial Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Network, B.O. Mangroves: Super Forests We Must Protect. Available online: https://blueocean.net/mangroves-super-forests-must-protect/ (accessed on 21 March 2019).
- Hardoko, E.S.; Puspitasari, Y.E.; Amalia, R. Study of ripe Rhizophora mucronata fruit flour as functional food for antidiabetic. Int. Food Res. J. 2015, 22, 953–959. [Google Scholar]
- Gurib-Fakim, A.; Brendler, T. Medicinal and Aromatic Plants of the Indian Ocean Islands, 1st ed.; Medpharm: Guildford, UK, 2004. [Google Scholar]
- Liebezeit, G.; Rau, M.T. New Guinean mangroves—Traditional usage and chemistry of natural products. Senckenberg. Marit. 2006, 36, 1–10. [Google Scholar] [CrossRef]
- Miles, D.H.; Kokpol, U.; Chittawong, V.; Tip-Pyang, S.; Tunsuwan, K.; Nguyen, C. Mangrove forests—The importance of conservation as a bioresource for ecosystem diversity and utilization as a source of chemical constituents with potential medicinal and agricultural value. 1999 IUPAC 1999, 70, 1–9. [Google Scholar]
- Premanathan, M.; Arakaki, R.; Izumi, H.; Kathiresan, K.; Nakano, M.; Yamamoto, N.; Nakashima, H. Antiviral properties of a mangrove plant, Rhizophora apiculata Blume, against human immunodeficiency virus. Antivir. Res. 1999, 44, 113–122. [Google Scholar] [CrossRef]
- Chakraborty, K.; Raola, V.K. Two rare antioxidant and anti-inflammatory oleanenes from loop root Asiatic mangrove Rhizophora mucronata. Phytochemistry 2017, 135, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Kusuma, S.; Kumar, P.A.; Boopalan, K. Potent antimicrobial activity of Rhizophora mucronata. J. Ecobiotechnol. 2012, 3, 40–41. [Google Scholar]
- Adhikari, A.; Ray, M.; Das, A.K.; Sur, T.K. Antidiabetic and antioxidant activity of Rhizophora mucronata leaves (Indian sundarban mangrove): An in vitro and in vivo study. Ayu 2016, 37, 76–81. [Google Scholar] [CrossRef]
- Sur, T.K.; Hazra, A.K.; Bhattacharyya, D.; Hazra, A. Antiradical and antidiabetic properties of standardized extract of Sunderban mangrove Rhizophora mucronata. Pharmacogn. Mag. 2015, 11, 389–394. [Google Scholar] [CrossRef]
- Aljaghthmi, O.; Heba, H.; Zeid, I.A. Bioactive Compounds Extracted from Mangrove Plants (Avicennia marina and Rhizophora mucronata): An Overview. Pathophysiology 2018. [Google Scholar] [CrossRef]
- Banerjee, D.; Chakrabarti, S.; Hazra, A.K.; Banerjee, S.; Ray, J.; Mukherjee, B. Antioxidant activity and total phenolics of some mangroves in Sundarbans. Afr. J. Biotechnol. 2008, 7, 805–810. [Google Scholar]
- Ramanathan, T.A.H. In Studies on Analgesic Activity of a Mangrove Species—Rhizophora mucronata Poir. In Proceedings of the Annual International Conference on Advances in Biotechnology (BIOTECH 2011), Bozen, Italy, 12–13 January 2011. [Google Scholar]
- Sadeer, N.B.; Mahomoodally, M.F.; Zengin, G.; Jeewon, R.; Nazurally, N.; Rengasamy Kannan, R.R.; Albuquerque, R.D.D.G.; Pandian, S.K. Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review. Mar. Drugs 2019, 17, 231. [Google Scholar]
- Uysal, S.; Aktumsek, A. A phytochemical study on Potentilla anatolica: An endemic Turkish plant. Ind. Crops Prod. 2015, 76, 1001–1007. [Google Scholar] [CrossRef]
- Vladimir-Knežević, S.; Blažeković, B.; Bival Štefan, M.; Alegro, A.; Kőszegi, T.; Petrik, J. Antioxidant activities and polyphenolic contents of three selected Micromeria species from Croatia. Molecules 2011, 16, 1454–1470. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Aktumsek, A. Investigation of antioxidant potentials of solvent extracts from different anatomical parts of Asphodeline anatolica E. Tuzlaci: An endemic plant to Turkey. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 481–488. [Google Scholar] [CrossRef]
- Rocchetti, G.; Giuberti, G.; Gallo, A.; Bernardi, J.; Marocco, A.; Lucini, L. Effect of dietary polyphenols on the in vitro starch digestibility of pigmented maize varieties under cooking conditions. Food Res. Int. 2018, 108, 183–191. [Google Scholar] [CrossRef]
- Rocchetti, G.; Lucini, L.; Rodriguez, J.M.L.; Barba, F.J.; Giuberti, G. Gluten-free flours from cereals, pseudocereals and legumes: Phenolic fingerprints and in vitro antioxidant properties. Food Chem. 2019, 271, 157–164. [Google Scholar] [CrossRef]
- Chlapanidas, T.; Faragò, S.; Lucconi, G.; Perteghella, S.; Galuzzi, M.; Mantelli, M.; Avanzini, M.A.; Tosca, M.C.; Marazzi, M.; Vigo, D.; et al. Sericins exhibit ROS-scavenging, anti-tyrosinase, anti-elastase, and in vitro immunomodulatory activities. Int. J. Biol. Macromol. 2013, 58, 47–56. [Google Scholar] [CrossRef]
- Grochowski, D.M.; Uysal, S.; Aktumsek, A.; Granica, S.; Zengin, G.; Ceylan, R.; Locatelli, M.; Tomczyk, M. In vitro enzyme inhibitory properties, antioxidant activities, and phytochemical profile of Potentilla thuringiaca. Phytochem. Lett. 2017, 20, 365–372. [Google Scholar] [CrossRef]
- Zengin, G. A study on in vitro enzyme inhibitory properties of Asphodeline anatolica: New sources of natural inhibitors for public health problems. Ind. Crops Prod. 2016, 83, 39–43. [Google Scholar] [CrossRef]
- Koc, Z.E.; Uysal, A. Investigation of novel monopodal and dipodal oxy-Schiff base triazine from cyanuric chloride: Structural and antimicrobial studies. J. Macromol. Sci. Part A 2016, 53, 111–115. [Google Scholar] [CrossRef]
- Zengin, G.; Uysal, A.; Gunes, E.; Aktumsek, A. Survey of phytochemical composition and biological effects of three extracts from a wild plant (Cotoneaster nummularia Fisch. et Mey.): A potential source for functional food ingredients and drug formulations. PLoS ONE 2014, 9, e113527. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.R. Polyphenols in Plants: Isolation, Purification and Extract Preparation, 2nd ed.; Academic Press: Cambridge, UK, 2019. [Google Scholar]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Delgoda, R.; Murray, J.E. Chapter 7—Evolutionary Perspectives on the Role of Plant Secondary Metabolites. In Pharmacognosy; Badal, S., Delgoda, R., Eds.; Academic Press: Boston, MA, USA, 2017. [Google Scholar]
- Azwanida, N.N. A Review on the Extraction Methods Use in Medicinal Plants, Principle, Strength and Limitation. Med. Aromat. Plants 2015, 4. [Google Scholar] [CrossRef]
- Gololo, S.S. Potential Adverse Effects of Alteration of Phytochemical Accumulation in Fruits and Vegetables. In Phytochemicals—Source of Antioxidants and Role in Disease Prevention; IntechOpen: London, UK, 2018. [Google Scholar]
- Iriti, M.; Faoro, F. Chemical Diversity and Defence Metabolism: How Plants Cope with Pathogens and Ozone Pollution. Int. J. Mol. Sci. 2009, 10, 3371–3399. [Google Scholar] [CrossRef]
- Suganthy, N.; Pandima Devi, K. In vitro antioxidant and anti-cholinesterase activities of Rhizophora mucronata. Pharm. Biol. 2016, 54, 118–129. [Google Scholar] [CrossRef]
- Dutta, S.; Ray, S. Comparative assessment of total phenolic content and in vitro antioxidant activities of bark and leaf methanolic extracts of Manilkara hexandra (Roxb.) Dubard. J. King Saud Univ. Sci. 2018. [Google Scholar] [CrossRef]
- Carocho, M.; Ferreira, I.C.F.R. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef]
- Kim, D.O.; Lee, K.W.; Lee, H.J.; Lee, C.Y. Vitamin C Equivalent Antioxidant Capacity (VCEAC) of Phenolic Phytochemicals. J. Agric. Food Chem. 2002, 50, 3713–3717. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, Y. Reducing power: The measure of antioxidant activities of reductant compounds? Redox Rep. 2004, 9, 213–217. [Google Scholar] [CrossRef]
- Tanaka, M.; Kuei, C.W.; Nagashima, Y.; Taguchi, T. Application of Antioxidative Maillard Reaction Products from Histidine and Glucose to Sardine Products. Nippon Suisan Gakkaishi 1988, 54, 1409–1414. [Google Scholar] [CrossRef]
- Segura Campos, M.R.; Ruiz Ruiz, J.; Chel-Guerrero, L.; Betancur Ancona, D. Coccoloba uvifera (L.) (Polygonaceae) Fruit: Phytochemical Screening and Potential Antioxidant Activity. J. Chem. 2015, 2015, 534954. [Google Scholar] [CrossRef]
- Abdel-Hameed, E.S.S.; Bazaid, S.A.; Salman, M.S. Characterization of the Phytochemical Constituents of Taif Rose and Its Antioxidant and Anticancer Activities. BioMed Res. Int. 2013, 2013, 345465. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C. The fight against non-communicable disease in emerging economies. Nature 2018, 562, S65. [Google Scholar] [CrossRef]
- Tucci, S.A.; Boyland, E.J.; Halford, J.C. The role of lipid and carbohydrate digestive enzyme inhibitors in the management of obesity: A review of current and emerging therapeutic agents. Diabetes Metab. Syndr. Obes. 2010, 3, 125–143. [Google Scholar] [CrossRef]
- Purnell, J. Definitions, Classification, and Epidemiology of Obesity. In Clinical Obesity in Adults and Children, 3rd ed.; Kopelman, P.G., Caterson, I.D., Dietz, W.H., Eds.; Blackwell Publishing Limited: Hoboken, NJ, USA, 2018. [Google Scholar]
- WHO. Obesity. Available online: https://www.who.int/topics/obesity/en/ (accessed on 2 April 2019).
- Pearson, H. In the eye of the beholder. Nature 2003, 424, 990–991. [Google Scholar] [CrossRef]
- Stern, J.S.; Kazaks, A. Obesity: A Reference Handbook, 2nd ed.; ABC-CLIO, LLC: Santa Barbara, CA, USA, 2015. [Google Scholar]
- Ramirez, G.; Zavala, M.; Perez, J.; Zamilpa, A. In Vitro Screening of Medicinal Plants Used in Mexico as Antidiabetics with Glucosidase and Lipase Inhibitory Activities. Evid.-Based Complement. Altern. Med. 2012, 2012, 701261. [Google Scholar] [CrossRef]
- Bustanji, Y.; Mohammad, M.; Hudaib, M.; Tawaha, K.; Almasri, I.; Alkhatib, H.; Issa, A.; Alali, F. Screening of some medicinal plants for their pancreatic lipase inhibitory potential. Jordan J. Pharm. Sci. 2011, 108, 1–16. [Google Scholar]
- Nature. Neurodegenerative Diseases. Available online: https://www.nature.com/subjects/neurodegenerative-diseases (accessed on 2 April 2019).
- Bilska, A.; Kobus-Cisowska, J.; Kmiecik, D.; Danyluk, B.; Kowalski, R.; Szymanowska, D.; Gramza-Michałowska, A.; Szczepaniak, O. Cholinesterase inhibitory activity, antioxidative potential and microbial stability of innovative liver pâté fortified with rosemary extract (Rosmarinus officinalis). Electron. J. Biotechnol. 2019, 40, 22–29. [Google Scholar] [CrossRef]
- Williams, P.; Sorribas, A.; Howes, M.J.R. Natural products as a source of Alzheimer’s drug leads. Nat. Prod. Rep. 2011, 28, 48–77. [Google Scholar] [CrossRef]
- Dorni, C.A.I.; Amalraj, A.; Gopi, S.; Varma, K.; Anjana, S.N. Novel cosmeceuticals from plants—An industry guided review. J. Appl. Res. Med. Aromat. Plants 2017, 7, 1–26. [Google Scholar] [CrossRef]
- Brozyna, A.; Zbytek, B.; Granese, J.; Carlson, J.A.; Ross, J.; Slominski, A. Mechanism of UV-related carcinogenesis and its contribution to nevi/melanoma. Expert Rev. Dermatol. 2007, 2, 451–469. [Google Scholar] [CrossRef]
- Mapunya, M.B.; Nikolova, R.V.; Lall, N. Melanogenesis and Antityrosinase Activity of Selected South African Plants. Evid.-Based Complement. Altern. Med. 2012, 2012, 374017. [Google Scholar] [CrossRef]
- Butt, A.R.S.; Abbasi, M.A.; Aziz Ur, R.; Siddiqui, S.Z.; Hassan, M.; Raza, H.; Shah, S.A.A.; Seo, S.Y. Synthesis and structure-activity relationship of elastase inhibiting novel ethylated thiazole-triazole acetamide hybrids: Mechanistic insights through kinetics and computational contemplations. Bioorgan. Chem. 2019, 86, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Tomasz, A. Multiple-Antibiotic-Resistant Pathogenic Bacteria—A Report on the Rockefeller University Workshop. N. Engl. J. Med. 1994, 330, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Klevens, R.M.; Morrison, M.A.; Nadle, J.; Petit, S.; Gershman, K.; Ray, S.; Harrison, L.H.; Lynfield, R.; Dumyati, G.; Townes, J.M. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. Jama 2007, 298, 1763–1771. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Staphylococcus aureus with reduced susceptibility to vancomycin—United States, 1997. MMWR. Morb. Mortal. Wkly. Rep. 1997, 46, 765. [Google Scholar]
- Hiramatsu, K.; Hanaki, H.; Ino, T.; Yabuta, K.; Oguri, T.; Tenover, F. Methicillin-resistant Staphylococcus aureus clinical strain with reduced vancomycin susceptibility. J. Antimicrob. Chemother. 1997, 40, 135–136. [Google Scholar] [CrossRef]

| Samples | Yield (%) | Total Phenolic Content (mg GAE/g) | Total Flavonoid Content (mg RE/g) | Total Phenolic Acid Content (mg CE/g) | Total Flavanol Content (mg CAE/g) | Total Condensed Tannin Content (mg CAE/g) | Total Triterpenoid Content (mg OAE/g) |
|---|---|---|---|---|---|---|---|
| RLM | 26.88 | 207.05 ± 0.88 c | 31.85 ± 0.31 b | 14.59 ± 0.89 d | 98.77 ± 0.92 b | 173.69 ± 8.36 a | 64.40 ± 4.10 b |
| RRM | 20.90 | 107.98 ± 1.49 h | 2.59 ± 0.09 j,k | 7.89 ± 0.21 g | 52.31 ± 0.56 c | 93.99 ± 0.77 b | 26.72 ± 2.49 e |
| RTM | 14.38 | 220.50 ± 3.33 a | 12.46 ± 0.19 g | 20.73 ± 0.70 a | 107.69 ± 1.16 a | 171.43 ± 2.67 a | 74.86 ± 4.23 a |
| RFM | 6.94 | 79.55 ± 0.73 j | 2.69 ± 0.11 j | 5.55 ± 0.09 i | 21.25 ± 0.28 e | 43.75 ± 2.68 f | 13.78 ± 0.47 g |
| RLD | 21.62 | 173.89 ± 1.43 f | 19.26 ± 0.15 d | 17.63 ± 0.30 c | 12.24 ± 0.08 g,h | 73.52 ± 1.60 d | 31.26 ± 2.01 c,d |
| RRD | 37.14 | 104.54 ± 2.68 h | 2.17 ± 0.07 j,k,l | 9.40 ± 0.37 f | 3.86 ± 0.03 i | 38.78 ± 1.56 f | 14.22 ± 0.41 g |
| RTD | 15.08 | 188.55 ± 0.89 d | 4.08 ± 0.16 i | 19.14 ± 0.93 b | 12.75 ± 0.13 g | 80.69 ± 7.83 c | 34.14 ± 1.00 c,d |
| RFD | 20.44 | 57.29 ± 0.32 k | 1.79 ± 0.05 l | 3.80 ± 0.07 j | 2.07 ± 0.02 j | 19.33 ± 0.39 h,i | 4.81 ± 0.52 i |
| RLA | 29.78 | 178.15 ± 0.83 e | 6.00 ± 0.23 h | 14.27 ± 0.98 d | 12.80 ± 0.04 g | 95.56 ± 4.67 b | 30.15 ± 2.32 d,e |
| RRA | 11.54 | 124.02 ± 1.09 g | 1.65 ± 0.44 l | 11.43 ± 0.43 e | 4.73 ± 0.08 i | 57.44 ± 2.06 e | 19.00 ± 1.62 f |
| RTA | 8.82 | 214.94 ± 0.96 b | 4.09 ± 0.22 i | 21.00 ± 1.18 a | 14.93 ± 0.22 f | 93.64 ± 2.29 b | 35.12 ± 3.09 c |
| RFA | 3.24 | 96.57 ± 0.44 i | 1.87 ± 0.24 k,l | 7.12 ± 0.19 g,h | 2.69 ± 0.06 j | 25.69 ± 0.56 g,h | 9.33 ± 0.97 h |
| RLE | 8.06 | 41.83 ± 1.27 m | 41.67 ± 0.38 a | 1.79 ± 0.05 k | 11.45 ± 0.15 h | 16.94 ± 0.58 i | 10.05 ± 0.49 g,h |
| RRE | 4.54 | 51.14 ± 0.69 l | 14.78 ± 0.30 e | 3.02 ± 0.36 j | 4.16 ± 0.05 i | 30.40 ± 1.72 g | 11.97 ± 0.45 g,h |
| RTE | 1.32 | 97.13 ± 4.16 i | 13.39 ± 0.18 f | 6.23 ± 0.24 h,i | 49.73 ± 1.19 d | 57.03 ± 0.95 e | 22.33 ± 1.52 f |
| RFE | 0.76 | 31.06 ± 0.05 n | 24.36 ± 1.13 c | 1.64 ± 0.10 k | 2.65 ± 0.01 j | 7.95 ± 0.74 j | 10.60 ± 1.66 g,h |
| Samples | Cyanidin Eq. | Luteolin Eq. | Catechin Eq. | Sesamin Eq. | Tyrosol Eq. | Ferulic Acid Eq. |
|---|---|---|---|---|---|---|
| RLM | 11.88 ± 0.80 a | 4.03 ± 0.57 | 48.12 ± 0.7 a | 2.49 ± 0.23 a,b | 4.73 ± 0.05 b | 16.26 ± 0.29 a |
| RRM | 9.64 ± 0.19 a,b | 4.79 ± 1.13 | 12.69 ± 0.7 c | 1.48 ± 0.07 b | 5.84 ± 1.38 b | 14.93 ± 1.25 a,b |
| RTM | 6.07 ± 1.78 b | 4.37 ± 0.81 | 20.18 ± 4.2 b | 3.58 ± 0.17 a | 5.48 ± 0.87 b | 12.54 ± 1.66 b |
| RFM | 4.53 ± 0.16 b | 4.06 ± 2.21 | 8.10 ± 0.2 d | 1.19 ± 0.16 b | 6.04 ± 0.13 a,b | 6.69 ± 0.48 c |
| RLE | 9.70 ± 0.98 a | 4.85 ± 0.51 a | 15.04 ± 3.8 a | 2.35 ± 0.14 a,b | 9.44 ± 0.29 b | 14.18 ± 1.08 b |
| RRE | 6.47 ± 0.32 a,b | 1.81 ± 0.20 b | 8.88 ± 0.4 c | 1.29 ± 0.14 b | 9.21 ± 1.20 b | 4.31 ± 0.59 c |
| RTE | 10.60 ± 0.85 a | 2.27 ± 0.11 b | 11.92 ± 3.3 b | 3.08 ± 0.05 a | 18.50 ± 0.71 a | 16.36 ± 3.24 a,b |
| RFE | 4.26 ± 0.85 b | 1.56 ± 0.20 b | 6.84 ± 0.7 c | 1.71 ± 0.55 b | 14.77 ± 5.55 a | 7.29 ± 4.62 b,c |
| RLD | 9.10 ± 0.75 a | 7.72 ± 0.29 a | 38.56 ± 0.8 a | 1.90 ± 0.22 b | 3.36 ± 0.82 b | 5.56 ± 1.18 c |
| RRD | 6.37 ± 0.50 b | 1.73 ± 0.12 c | 9.41 ± 1.2 c | 1.53 ± 0.05 b | 5.16 ± 0.94 a | 19.24 ± 1.07 a |
| RTD | 7.82 ± 1.17 a,b | 5.37 ± 0.82 b | 15.79 ± 3.2 b | 2.89 ± 0.15 a,b | 4.32 ± 0.09 a,b | 12.88 ± 0.69 b |
| RFD | 5.10 ± 0.65 b | 2.06 ± 0.06 c | 10.10 ± 0.4 c | 2.47 ± 0.73 a,b | 5.86 ± 0.64 a | 10.82 ± 1.12 b |
| RLA | 7.37 ± 0.42 b | 4.06 ± 0.51 a,b | 25.72 ± 0.2 a | 1.93 ± 0.09 a,b | 6.33 ± 0.21 a,b | 5.75 ± 0.911 b |
| RRA | 7.83 ± 0.21 b | 2.23 ± 0.17 c | 16.77 ± 0.2 a,b | 1.40 ± 0.03 a,b | 6.35 ± 1.43 a,b | 8.39 ± 0.27 a |
| RTA | 12.50 ± 0.26 a | 3.16 ± 0.48 b | 12.56 ± 4.1 b | 0.75 ± 0.06 b | 5.86 ± 0.91 a,b | 5.42 ± 1.11 b |
| RFA | 4.01 ± 0.60 c | 1.64 ± 0.21 c | 6.64 ± 0.2 c | 2.56 ± 0.05 a | 4.42 ± 0.12 b | 6.91 ± 0.18 a,b |
| Samples | DPPH (mg TE/g) | ABTS (mg TE/g) | Phosphomolybdenum (mmol TE/g) | Metal Chelating (mg EDTAE/g) | CUPRAC (mg TE/g) | FRAP (mg TE/g) |
|---|---|---|---|---|---|---|
| RLM | 688.96 ± 24.01 b,* | 602.05 ± 9.43 a | 4.13 ± 0.29 d | 10.44 ± 0.45 e | 1050.40 ± 9.11 c | 552.01 ± 20.31 d |
| RRM | 99.52 ± 0.10 g | 141.53 ± 0.10 f | 2.61 ± 0.13 f | 5.57 ± 1.01 f | 418.54 ± 4.39 f | 225.22 ± 1.44 g |
| RTM | 807.07 ± 6.83 a | 514.23 ± 7.95 b | 4.62 ± 0.17 b,c | 15.63 ± 0.56 c,d | 1336.88 ± 15.70 a | 710.18 ± 21.04 a |
| RFM | 97.66 ± 0.21 g | 140.82 ± 0.08 f | 1.81 ± 0.09 g | 26.36 ± 0.51 a | 318.48 ± 5.10 i | 182.34 ± 7.89 i |
| RLD | 469.04 ± 7.42 f | 383.52 ± 9.81 e | 4.32 ± 0.15 d | 11.34 ± 0.84 e | 918.08 ± 8.36 e | 511.93 ± 12.84 e |
| RRD | 96.39 ± 0.38 g | 140.13 ± 0.11 f | 2.65 ± 0.04 f | 3.99 ± 0.79 f | 388.44 ± 0.23 g | 222.87 ± 1.13 g |
| RTD | 523.48 ± 6.13 e | 459.17 ± 22.80 c | 4.69 ± 0.03 b | 6.33 ± 0.09 f | 1001.52 ± 9.53 d | 601.21 ± 3.65 c |
| RFD | 73.25 ± 0.77 h | 107.60 ± 4.86 g | 1.43 ± 0.02 h | 15.27 ± 0.61 d | 194.02 ± 2.05 j | 130.08 ± 1.08 j |
| RLA | 543.33 ± 6.63 d | 408.02 ± 19.70 d | 4.39 ± 0.16 c,d | 11.08 ± 1.98 e | 918.73 ± 8.00 e | 511.86 ± 3.10 e |
| RRA | 94.73 ± 0.14 g | 139.66 ± 0.06 f | 3.25 ± 0.04e | 4.95 ± 0.10f | 427.54 ± 2.31 f | 249.57 ± 0.65 f |
| RTA | 656.32 ± 20.03 c | 602.91 ± 7.03 a | 5.06 ± 0.13a | 17.92 ± 0.96bc | 1082.00 ± 47.96 b | 636.79 ± 3.13 b |
| RFA | 92.55 ± 0.27 g | 139.85 ± 0.04 f | 2.51 ± 0.02f | 18.82 ± 2.48b | 314.59 ± 8.99 i | 200.75 ± 4.69 h |
| RLE | 55.41 ± 0.89 i | 64.44 ± 0.60 h | 1.14 ± 0.03i | 14.92 ± 0.84d | 152.48 ± 3.68 k,l | 71.60 ± 3.01 l |
| RRE | 56.93 ± 0.09 h,i | 70.63 ± 1.42 h | 1.78 ± 0.11g | 18.16 ± 1.65b | 175.70 ± 0.34 j,k | 99.09 ± 2.43 k |
| RTE | 94.99 ± 0.25 g | 140.64 ± 0.19 f | 2.63 ± 0.14f | 15.35 ± 0.21d | 357.95 ± 1.83 h | 197.26 ± 2.62 h,i |
| RFE | 41.89 ± 1.76 i | 36.57 ± 1.53 i | 1.05 ± 0.13i | 17.33 ± 1.98b,c,d | 142.21 ± 6.37 l | 62.17 ± 0.45 l |
| Samples | AChE (mg GALAE/g) | BChE (mg GALAE/g) | Tyrosinase (mg KAE/g) | Amylase (mmol ACAE/g) | Glucosidase (mg ACAE/g) | Lipase (mg OE/g) | Elastase (mg CAE/g) |
|---|---|---|---|---|---|---|---|
| RLM | 4.60 ± 0.02 a | 3.76 ± 0.13 b,c | 144.02 ± 0.74 a | 0.96 ± 0.03 a | na | 57.78 ± 5.00 e | 4.58 ± 0.04 a |
| RRM | 4.61 ± 0.05 a | 4.65 ± 0.11 a | 138.76 ± 1.73 b | 0.75 ± 0.01 b | na | 77.92 ± 7.36 d | 4.50 ± 0.16 a |
| RTM | 4.78 ± 0.03 a | 4.59 ± 0.01 a | 145.31 ± 1.49 a | 0.95 ± 0.01 a | na | 40.53 ± 5.06 c,d | 4.68 ± 0.08 a |
| RFM | 4.46 ± 0.18 a | 3.44 ± 0.05 b,c,d | 136.69 ± 1.22 b,c | 0.70 ± 0.02 c,d | na | 78.77 ± 9.41 c,d | 4.32 ± 0.11 a,b |
| RLD | 3.15 ± 0.10 d,e | 1.20 ± 0.19 f,g | 138.92 ± 0.15 b | 0.24 ± 0.01 g | na | na | 3.47 ± 0.38 c,d |
| RRD | 1.98 ± 0.31 f | 1.48 ± 0.50 e,f | 68.24 ± 0.91 i | 0.11 ± 0.01 h | 30.45 ± 1.51 a,b | 3.56 ± 0.88 g | 3.19 ± 0.21 d,e |
| RTD | 3.54 ± 0.01 c | 2.02 ± 0.66 e | 106.76 ± 2.00 f | 0.46 ± 0.08 f | na | na | 2.89 ± 0.37 e,f |
| RFD | 0.64 ± 0.13 h | 0.51 ± 0.05 g,h | 18.70 ± 0.72 k | 0.12 ± 0.01 h | 30.60 ± 0.85 a,b | 2.86 ± 0.28 g | 2.08 ± 0.49 h |
| RLA | 2.23 ± 0.23 f | 0.32 ± 0.07 h | 119.35 ± 0.93 e | 0.10 ± 0.01 h | na | na | 2.29 ± 0.18 g,h |
| RRA | 1.24 ± 0.15 g | Na | 44.93 ± 1.78 j | 0.14 ± 0.01 h | 31.16 ± 0.28 a | na | 2.55 ± 0.38 f,g,h |
| RTA | 3.54 ± 0.09 c | 2.91 ± 0.34 d | 100.79 ± 4.48 g | 0.43 ± 0.01 f | na | na | 3.78 ± 0.26 b,c |
| RFA | 0.25 ± 0.06 i | na | 72.14 ± 1.06 h | 0.12 ± 0.01 h | 31.16 ± 0.04 a | 7.44 ± 0.22 g | 2.67 ± 0.08 e,f,g |
| RLE | 3.46 ± 0.24 c,d | 3.34 ± 0.16 c,d | 132.58 ± 1.04 d | 0.61 ± 0.01 e | 29.64 ± 0.59 b,c | 83.10 ± 1.96 b,c,d | 3.65 ± 0.54 c,d |
| RRE | 3.64 ± 0.38 c | 4.15 ± 0.87 a,b | 131.37 ± 0.54 d | 0.72 ± 0.01 b,c | 27.61 ± 0.82 d | 88.32 ± 2.18 b | 3.47 ± 0.07 c,d |
| RTE | 4.04 ± 0.09 b | 3.62 ± 0.12 b,c,d | 138.94 ± 0.69 b | 0.73 ± 0.01 b,c | 30.20 ± 0.01 a,b | 86.14 ± 2.24 b,c | 3.49 ± 0.08 c,d |
| RFE | 2.83 ± 0.26 e | 4.68 ± 0.36 a | 134.26 ± 0.70 c,d | 0.66 ± 0.01 d | 28.77 ± 0.13 c | 101.02 ± 1.31 a | 4.25 ± 0.25 a |
| Strains | MIC Values of R. mucronata Extracts (mg/mL) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RLM | RRM | RTM | RFM | RLE | RRE | RTE | RFE | RLA | RRA | RTA | RFA | RLD | RRD | RTD | RFD | Gentamicin (µg/mL) | |
| Escherichia coli ATCC 25922 | - | - | 1.56 | 1.56 | - | - | - | - | - | - | 6.25 | - | - | - | - | 6.25 | 0.312 |
| Pseudomonas aeruginosa ATCC 27853 | - | - | - | - | - | - | - | - | 6.25 | - | 3.13 | - | 6.25 | - | 6.25 | - | 0.039 |
| Klebsiella pneumoniae ATCC 70603 | - | - | 1.56 | - | - | - | - | - | 3.13 | - | 3.13 | - | - | - | 6.25 | - | 1.25 |
| Staphylococcus aureus ATCC 43300 (MRSA) | 0.19 | 0.39 | 0.39 | 3.12 | 3.12 | 1.56 | 3.12 | 3.12 | 0.39 | 1.56 | 0.39 | 1.56 | 0.39 | 0.78 | 0.39 | 6.25 | 0.078 |
| Salmonella enteritidis ATCC 13076 | 1.56 | - | 1.56 | - | - | - | - | - | - | - | 6.25 | - | - | - | 6.25 | - | 0.078 |
| Sarcina lutea ATCC 9341 | 1.56 | - | 1.56 | - | - | - | - | - | - | - | 6.25 | - | 6.25 | - | - | - | 0.039 |
| Proteus mirabilis ATCC 25933 | 0.39 | - | 0.39 | - | - | - | - | - | 1.56 | 6.25 | 0.78 | - | 1.56 | 6.25 | 1.56 | - | 0.312 |
| Bacillus cereus ATCC 11778 | - | - | - | - | - | - | - | - | - | - | 3.13 | - | 6.25 | - | - | - | <0.039 |
| Candida albicans ATCC 26555 | 0.39 | - | 0.19 | 0.78 | - | - | - | - | 0.19 | 0.39 | 0.78 | 0.39 | 0.39 | 0.78 | 0.78 | 6.25 | 0.312 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadeer, N.B.; Rocchetti, G.; Senizza, B.; Montesano, D.; Zengin, G.; Uysal, A.; Jeewon, R.; Lucini, L.; Mahomoodally, M.F. Untargeted Metabolomic Profiling, Multivariate Analysis and Biological Evaluation of the True Mangrove (Rhizophora mucronata Lam.). Antioxidants 2019, 8, 489. https://doi.org/10.3390/antiox8100489
Sadeer NB, Rocchetti G, Senizza B, Montesano D, Zengin G, Uysal A, Jeewon R, Lucini L, Mahomoodally MF. Untargeted Metabolomic Profiling, Multivariate Analysis and Biological Evaluation of the True Mangrove (Rhizophora mucronata Lam.). Antioxidants. 2019; 8(10):489. https://doi.org/10.3390/antiox8100489
Chicago/Turabian StyleSadeer, Nabeelah Bibi, Gabriele Rocchetti, Biancamaria Senizza, Domenico Montesano, Gokhan Zengin, Ahmet Uysal, Rajesh Jeewon, Luigi Lucini, and Mohamad Fawzi Mahomoodally. 2019. "Untargeted Metabolomic Profiling, Multivariate Analysis and Biological Evaluation of the True Mangrove (Rhizophora mucronata Lam.)" Antioxidants 8, no. 10: 489. https://doi.org/10.3390/antiox8100489
APA StyleSadeer, N. B., Rocchetti, G., Senizza, B., Montesano, D., Zengin, G., Uysal, A., Jeewon, R., Lucini, L., & Mahomoodally, M. F. (2019). Untargeted Metabolomic Profiling, Multivariate Analysis and Biological Evaluation of the True Mangrove (Rhizophora mucronata Lam.). Antioxidants, 8(10), 489. https://doi.org/10.3390/antiox8100489









