1. Introduction
Potato (
Solanum tuberosum L.) is the sole major tuber crop and the fourth most important food crop worldwide [
1,
2,
3]. The total world potato production was estimated at 388,191,000 t in 2017 and almost a third of all potatoes were harvested in China and India [
4]. The nutritional quality of potato is important to human health whose staple food is potato [
5]. The potato contains minerals, proteins and antioxidant compounds such as carotenoids, phenolics, flavonoids, vitamins C and E, among others [
5,
6,
7]. These phytochemicals in potato have attracted more and more attention from the public because of their effects on promotion of physical well-being [
2,
8].
Wide diversity in the phytochemicals and nutritional compositions in potato has been reported [
9]. For example, the free phenolic components in potato have been widely investigated. Ramamurthy et al. reported that the chlorogenic, neochlorogenic, cryptochlorogenic,
p-coumaric and ferulic acids were detected in potato [
10]. Im et al. reported that the caffeic acid, chlorogenic acid, cinnamic acid, ferulic acid, gallic acid, and protocatechuic acid were richly present in potato peel [
11]. Xu et al. [
12], Albishi et al. [
13] and Kim et al. [
14] reported that chlorogenic, neochlorogenic, cryptochlorogenic, caffeic,
p-coumaric, ferulic, vanillic acids, quercetin glucoside and tryptophan were detected in potato flesh. Although the contents of the different phenolic components were varied in different reports, chlorogenic acid was the most abundant [
11,
12,
13,
14]. The variation in phenolic acids may be due to different genotypes, and different extraction and analytical methods.
Phenolic acids and flavonoids are the most common phenolic compounds, which are generally present in both free and bound forms [
15]. Chu et al. reported the free and bound phenolic content of 10 kinds of vegetables, and found that free phenolic acids accounted for 60.1%, 62.4%, 67.1% and 73.8% of the total phenolic acids in potato, carrot, cabbage and cucumber [
16]. However, most of the studies only investigated the free phenolics based on the soluble extraction and neglected the bound phenolics, so the total phenolic content (TPC) of potato and antioxidant activities were underestimated in those works, especially for the colored potato. Anthocyanins are one kind of flavonoid, which are present in the colored potato and exhibit a range of biological activities [
17,
18,
19]. Nemś et al. indicated that pelargonidin derivatives were present in greatest quantity in the red-fleshed potatoes, and petunidin and malvidin were the most abundant anthocyanins in the purple potatoes [
20].
Although many assays have been conducted to determine the phenolic acids, anthocyanins and antioxidant capacity in potato, the individual phenolic acid composition and antioxidant capacity in the bound form were rarely reported. Furthermore, the relationships between the content of individual phenolic acid, TPC and antioxidant capacity have not been clearly understood.
The objectives of this study were: (1) to investigate the composition and levels of phenolic acids, and the antioxidant capacity of both free and bound fractions of potato accessions; (2) to analyze the relationships between the content of individual phenolic acid and TPC and antioxidant activities; and (3) to assess the composition of anthocyanin and total anthocyanin contents from red and purple flesh potatoes. The results from this study may further contribute to our understanding of the genetic diversity in the phenolics and processing techniques to benefit the desired nutritional qualities of potato crops.
2. Materials and Methods
2.1. Sample Preparation
The 14 potato cultivars were planted and harvested in the Zhejiang University farm in Hangzhou, China, in 2016 (
Table 1) [
1]. After harvesting, representative samples were obtained by selecting around 15 tubers with similar size for each cultivar, which were washed and peeled manually. Then they were cut to pieces and quickly stored at −80 °C. The potato pieces were put in a freeze dryer for 2 days at −50 °C. The dried potato samples were ground into powder and stored at 4 °C until analyzed.
2.2. Chemicals
Phenolic acid standards (caffeic acid, chlorogenic acid, p-coumaric acid, cryptochlorogenic acid, ferulic acid and neochlorogenic acid), Folin–Ciocalteu reagent, sodium hydroxide, 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), 1,1-diphenyl-2-picrylhydrazyl radical 2,2-diphenyl-1-(2,4,6-trinitrophenyl) hydrazyl (DPPH), 2,2-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid diammonium salt (ABTS), and gallic acid were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). Methanol (HPLC grade) was bought from the Fisher Scientific Co. (Ottawa, ON, Canada). Acetic acid (HPLC grade) was bought from Macklin (Shanghai, China). Ethyl acetate, hexanes, hydrochloric acid, potassium chloride, potassium persulfate, sodium acetate, sodium carbonate, and sodium sulfate were purchased from the Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China).
2.3. Extraction of Free/Soluble Phenolics and Bound Phenolics
The extraction method of free/soluble phenolics was performed according to the procedure described by Pang et al. [
21]. Briefly, one gram potato powder (dry basis) was first defatted with 10 mL of hexanes. Then potato powder was extracted two times using 20 mL of 80% methanol at room temperature. Each time, the extraction mixture was shaken for 30 min. Then, the mixture was subsequently centrifuged at 8000×
g for five min. After accomplishing centrifugation, the supernatants were collected and the pH adjusted to 1.5–2.0. The supernatant was then concentrated by a rotary evaporator (IKA RV10 digital V, Staufen, Germany) at 37 °C. The concentrated fraction was extracted with 60 mL ethyl acetate three times. The pooled extracts were evaporated with the same rotary evaporator at 35 °C, then 5 mL of 50% methanol was used to dissolve the dried extracts, which were stored at –20 °C and used as crude free fraction for phenolic compounds analysis.
The solid residue after extracting soluble phenolics was used to extract bound phenolics. The residue was first digested with 20 mL of NaOH (4 M) for 2 h at room temperature. The pH was adjusted to 1.5–2.0 using concentrated HCl, the mixture was then extracted three times using 60 mL of ethyl acetate. The pooled ethyl acetate extractions were evaporated and dissolved in methanol according to the same procedures stated above for free extraction [
21].
2.4. Total Phenolic Content (TPC)
The TPC was measured using the colorimetric method with Folin–Ciocalteu reagent [
21,
22]. The TPC was expressed as milligrams of gallic acid equivalent (mg GAE) per 100 g of potato powder.
2.5. Determination of DPPH• Radical and ABT•+ Radical Cation Scavenging Activities
The DPPH assay was accomplished with the procedures described by Yamaguchi et al. with minor modification [
21,
23]. Briefly, 200 μL of the diluted free or bound extracts were added to 3 mL DPPH• radical solution (100 μM), which was prepared in methanol. The reaction was kept in the dark at room temperature for 30 min, and a spectrophotometer was used to measure the absorbance at 517 nm. The DPPH scavenging activity was expressed with inhibition (percent) of DPPH• absorbance [
21].
The assay of ABTS•
+ radical cation scavenging activity was conducted according to the procedure of Re et al. with slight modification [
21,
22,
24]. First, 7 mM ABTS and 2.45 mM potassium per sulfate was mixed at room temperature in dark for 20 h to generate ABTS•
+ radical cation. Then, methanol was used to dilute the ABTS•
+ mixture to an absorbance around 0.700 at 734 nm. Last, 3.9 mL of ABTS+ solution was added to 0.1 mL of the extracts which were first diluted appropriately. The reaction mixture was kept at room temperature for 6 min, then the absorbance was recorded at 734 nm by a spectrophotometer.
Trolox (0.5 mM) was served as a reference antioxidant. The results of DPPH radical scavenging activities and ABTS•+ radical cation scavenging activities were expressed as µM of Trolox equivalents (TE) per 100 g of potato powder using a standard curve of Trolox.
2.6. Total Anthocyanin Content (TAC)
TAC assay was carried out using a pH differential method [
25,
26,
27]. The anthocyanin was extracted with 0.5 g potato powder mixed with 15 mL methanol to 1 M HCl (85:15,
v/v) three times under dark condition, and each time held on a shaker for 20 min. Then supernatants were collected using a centrifuge at 6000×
g for 15 min at room temperature. The TAC was expressed as cyanidin-3-glucoside equivalent.
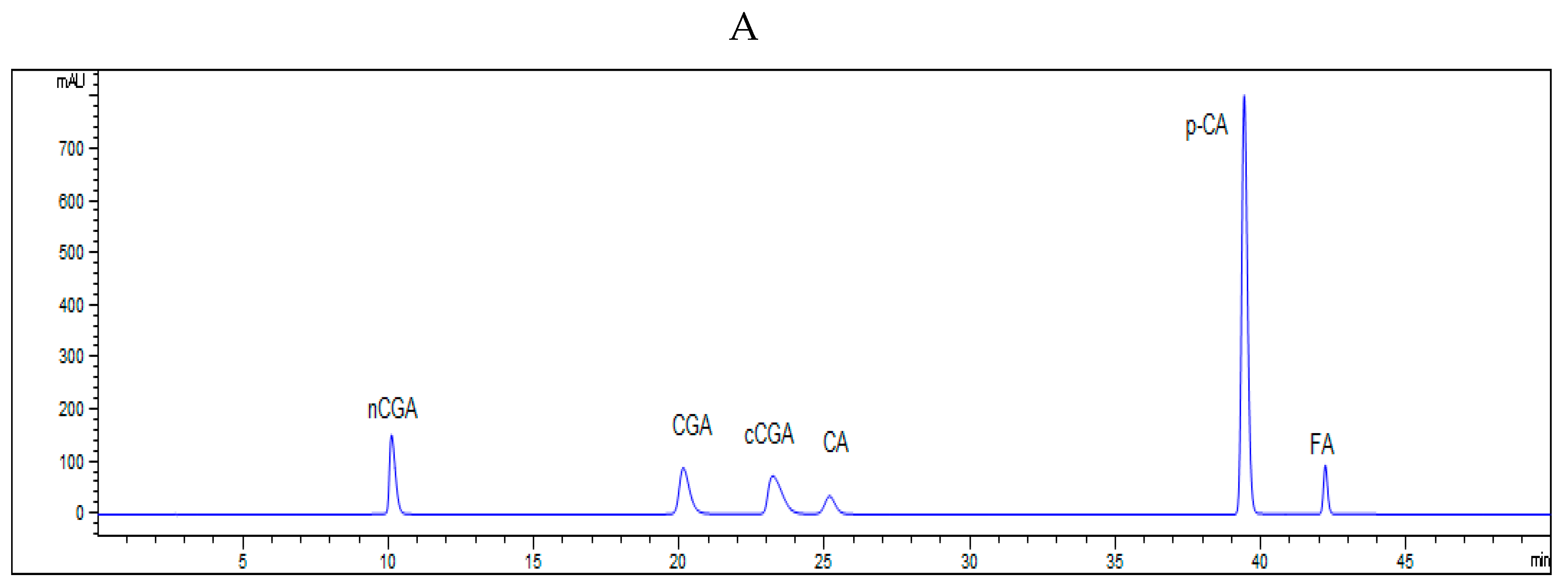
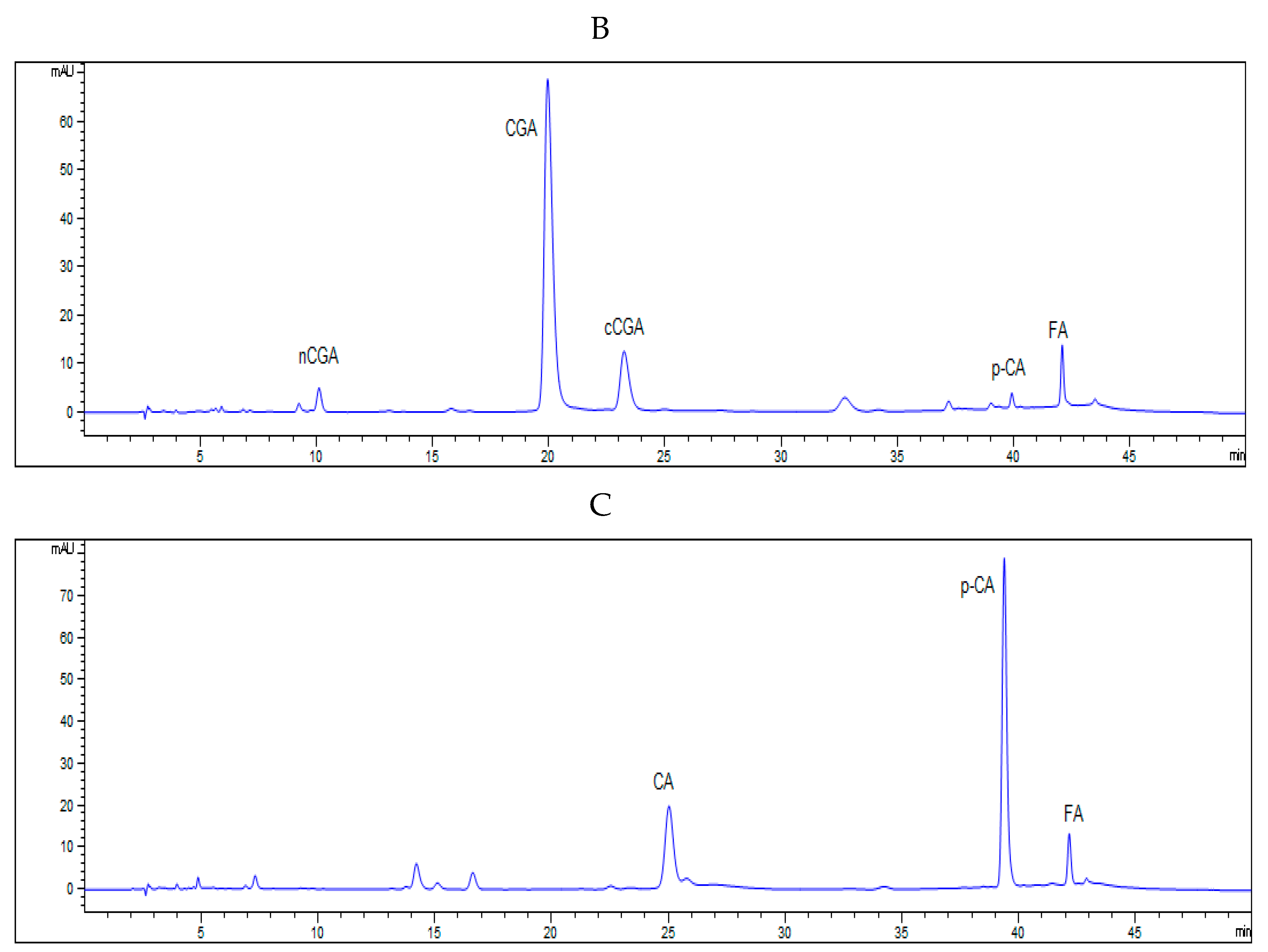
2.7. HPLC Analysis of Phenolic Acids
An HPLC system (Waters Associate, Milford, MA, USA) was used for separation of phenolic acids [
21]. The 5 µL extraction was injected by an autosampler. The mobile phase consisted of 0.1% acetic acid in water (A) and 0.1% acetic acid in methanol (B), and the flow rate was 1 mL/min. A 45 min linear gradient was set as follows: 0–1 min, 10–15% B, 1–30 min, 15–18% B, 30–40 min, 18–40% B, 40–45 min, 40–10% B. The phenolic acids were detected at a wavelength of 280 nm. The content of individual phenolic acid was quantified using external calibration curve.
2.8. LC-MS/MS Analysis of Anthocyanin
The phenolics were analyzed by a UPLC-TOF-MS/MS system, which consisted of an ultrahigh pressure chromatograph (UPLC) (Shimadzu Co., Ltd., Kyoto, Japan) and an ESI 5600 triple quadrupole time of flight (TOF) mass spectrometer (Sciex Co., Ltd., Framingham, MA, USA). A Zorbax SB-C18 column (3.5 μm, 2.1 × 100 mm) (Agilent Technologies, Inc., Palo Alto, CA, USA) was used for separation of the anthocyanins. The same injection volume and mobile phase as in separation of phenolic acids were used, but the flow rate was 0.5 mL/min. A linear gradient with the following proportions of solvent B was used: 0–1 min, 10–15% B; 1–10 min, 15–20% B; 10–15 min, 20–95% B; 15–18 min, 95–10% B. MS parameters were set as follows: capillary voltage, 2000 V; sample cone voltage, 30 V; source temperature, 150 °C; desolvation temperature, 250 °C; desolvation gas (nitrogen gas) flow rate, 900 L/h; data acquisition range, m/z 100–1000 Da; ionization mode, positive. The MS/MS spectra were acquired by using collision energy of 35 V.
2.9. Statistical Analysis
The results were presented as mean ± standard deviation (SD), in which all the measurements were accomplished at least in duplicate. Duncan’s multiple range test of ANOVA and correlation analysis were conducted using the SPSS 20.0 software (SPSS, Inc., Chicago IL, USA).
4. Conclusions
The TPC, phenolic acids content and antioxidant activities in the free and bound fractions of potatoes with different flesh color were systematically assayed. The total free TPC was significantly higher than the bound TPC in all cultivars. Colored potatoes had higher TPC than yellow and white flesh potatoes, which also had higher antioxidant capacity than yellow and white potatoes. The free and bound TPC were strongly positively correlated with antioxidant activity of free and bound fractions. Chlorogenic acid, neochlorogenic acid, cryptochlorogenic acid, caffeic acid, p-coumaric acid and ferulic acid were detected in the free fraction. Chlorogenic acid was the most abundant phenolic acid and accounted for 35.21–81.78% of the total free phenolic acid contents. The caffeic acid, p-coumaric acid and ferulic acid were detected in the bound fraction of the colored potatoes. In the free fraction, the content of each individual phenolic acid was positively correlated with antioxidant activity. In the bound fraction, caffeic acid and p-coumaric acid contents were positively correlated with antioxidant activity. The TAC of purple flesh potato was higher than red flesh potato. The TAC was not positively correlated with free TPC. This study promotes the further understanding of the correlations among the TPC, antioxidant activity and phenolic acid content in the free and bound fraction in potatoes with different flesh color and provides the theoretical basis for breeders to screen higher functional varieties based on the correlations reported in this study.